Abstract
The highly conserved NendoU replicative domain of nidoviruses (arteriviruses, coronaviruses, and roniviruses) belongs to a small protein family whose cellular branch is prototyped by XendoU, a Xenopus laevis endoribonuclease involved in nucleolar RNA processing. Recently, sequence-specific in vitro endoribonuclease activity was demonstrated for the NendoU-containing nonstructural protein (nsp) 15 of several coronaviruses. To investigate the biological role of this novel enzymatic activity, we have characterized a comprehensive set of arterivirus NendoU mutants. Deleting parts of the NendoU domain from nsp11 of equine arteritis virus was lethal. Site-directed mutagenesis of conserved residues exerted pleiotropic effects. In a first-cycle analysis, replacement of two conserved Asp residues in the C-terminal part of NendoU rendered viral RNA synthesis and virus production undetectable. In contrast, mutagenesis of other conserved residues, including two putative catalytic His residues that are absolutely conserved in NendoU and cellular homologs, produced viable mutants displaying reduced plaque sizes (20 to 80% reduction) and reduced yields of infectious progeny of up to 5 log units. A more detailed analysis of these mutants revealed a moderate reduction in RNA synthesis, with subgenomic RNA synthesis consistently being more strongly affected than genome replication. Our data suggest that the arterivirus nsp11 is a multifunctional protein with a key role in viral RNA synthesis and additional functions in the viral life cycle that are as yet poorly defined.
Nidoviruses are enveloped, positive-strand RNA viruses that have been grouped together on the basis of similarities in genome organization, the use of similar strategies for nonstructural and structural protein expression, and the presumed common ancestry of key replicative enzymes (2, 3, 5, 12, 24). The order Nidovirales currently includes the families Arteriviridae, Coronaviridae, and Roniviridae. Notwithstanding their taxonomic unification, important differences between nidovirus subgroups exist at the levels of genome size, virion architecture, biology of virus-host interactions, and pathogenesis.
Nidovirus RNA synthesis in infected cells entails both amplification of the viral genome and the production of an extensive set of subgenomic (sg) mRNAs, presumably each from their specific negative-stranded intermediates (17-19). RNA synthesis is driven by a set of nidovirus enzymes commonly referred to as “replicase,” which include several activities that are rare or lacking in other RNA viruses (23). The replicase is expressed from the incoming genome by translation of the large open reading frames (ORFs) 1a and 1b into polyproteins pp1a and pp1ab. The latter polypeptide is a C-terminally extended version of pp1a and is derived from a −1 ribosomal frameshift from ORF1a into overlapping ORF1b. The core of the nidovirus replicase polyprotein is formed by a conserved array of enzymatic domains consisting of (from N to C terminus) one or more papain-like “accessory proteinases,” a chymotrypsin-like “main proteinase,” the RNA-dependent RNA polymerase (RdRp), a helicase that contains an N-terminal zinc-binding domain, and an endoribonuclease called NendoU (for Nidovirus-specific endoribonuclease; see below) (11, 24, 39). For nidovirus replicase polyproteins of over 6,500 amino acids encoded by Coronaviridae and Roniviridae, which have genomes of over 25 kilobases, several additional enzymatic activities have been predicted (9, 9, 23, 35, 37). The fact that these enzymes have not been found in the proteomes of the Arteriviridae, which have smaller genomes of between 12 and 16 kilobases, was linked to the postulated functional hierarchy of the predicted nidovirus “RNA-processing enzymes,” with the nidovirus-wide conserved domain (NendoU) playing a key role in the initiation of a cascade of reactions (23).
The replicase polyproteins of equine arteritis virus (EAV), the arterivirus prototype, are autocatalytically cleaved into nonstructural proteins (nsps) 1 to 12 by three proteinases residing in nsp1, nsp2, and nsp4 (for a review, see reference 40). The EAV structural proteins are encoded by seven ORFs in the 3′ proximal quarter of the genome and are expressed from a nested set of sg mRNAs. These not only are 3′ coterminal with the genome but also carry a common 211-nucleotide (nt), 5′ leader sequence which is identical to the 5′ proximal region of the genome (6). The sg mRNAs are produced via a unique mechanism that presumably involves discontinuous extension of minus-strand RNA synthesis to produce sg-length minus-strand templates for the synthesis of sg mRNAs (17-19, 22). The discontinuous step in minus-strand RNA synthesis, during which sequences that are noncontiguous in the antigenome are fused to give the sg-length minus-strand RNAs, is guided by a base-pairing interaction between complementary transcription-regulating sequences (TRSs) present at the 3′ end of the nascent minus strand (anti-body TRS) and in the leader region of the genomic template (leader TRS) (17, 27, 32).
Four conserved domains, residing in proteins currently known as nsp9 to nsp11, were originally identified when the EAV ORF1b-encoded polypeptide was compared to its coronavirus and torovirus counterparts (5) (Fig. 1A): the RdRp in nsp9, a putative multinuclear zinc finger and an RNA helicase domain that are combined in nsp10 (20, 21), and a nidovirus-wide conserved domain in the C-terminal part of nsp11 (between His-2961 and Phe-3039, using the pp1ab amino acid numbering). This latter domain has no known homologs in other groups of RNA viruses, and thus it can be regarded as a genetic marker of the Nidovirales. A distant relationship between this nidovirus-specific domain and a family of cellular proteins prototyped by a Xenopus laevis endoribonuclease (XendoU) (15), was recently identified (23). XendoU is a Mn2+-dependent RNase that cleaves at U stretches and releases 2′,3′-cyclic phosphodiester products upon the processing of intron-encoded small nucleolar RNAs (15). Its nidovirus homolog was dubbed “NendoU” (13), and using severe acute respiratory syndrome-coronavirus (SARS-CoV) and three other coronaviruses, two groups independently verified the in vitro endoribonuclease activity of the coronavirus nsp15 replicase subunit, which contains the NendoU domain (1, 13). Also, coronavirus NendoU was shown to be Mn2+ dependent, produce molecules with 2′,3′-cyclic phosphate ends, and cleave upstream and downstream of uridylates in GU or GUU sequences. Ivanov and colleagues (13) reported a preference of NendoU for double-stranded over single-stranded RNA molecules, but a slight preference for single-stranded RNA was claimed by Bhardwaj and colleagues (1). Three residues (His-162, His-178, and Lys-224 in XendoU) that are absolutely conserved in proteins of the XendoU/NendoU family were implicated in catalysis (10, 23). Their replacement was not tolerated in coronavirus NendoU (13) and XendoU (10). By using an infectious cDNA clone of human coronavirus 229E (HCoV-229E), a single substitution of a conserved NendoU Asp (Asp-6408) (Fig. 1B), which also abolished endoribonuclease function in the in vitro assay, was found to completely block viral RNA synthesis and virus production (13). This amino acid and a second conserved Asp residue (Asp-6435 in HCoV-229E) (Fig. 1B) reside in the C-terminal part of the NendoU domain, which is well conserved in all nidoviruses but cannot be confidently aligned with the corresponding region in the cellular homologs (see below).
FIG. 1.
Nidovirus replicase comparison and sequence alignment of the EAV NendoU domain. (A) The replicase gene organization of the arteriviruses EAV and porcine reproductive and respiratory syndrome virus (PRRSV) and coronaviruses HCoV-229E and SARS-CoV are depicted in the form of the polyprotein pp1ab. The border between ORF1a- and ORF1b-encoded residues is indicated as RFS (ribosomal frameshift). Gray and black arrows represent the sites that are cleaved by accessory papain-like proteinases (AP) and the main (“3C-like”) proteinases (MP), respectively. The proteolytic cleavage products are numbered, and within the cleavage products, the location of domains that have been identified as structurally or functionally related are indicated in black boxes. These include the four nidovirus-wide conserved domains encoded by ORF1b (RdRp, zinc-binding domain [Z], helicase [HEL], and NendoU [N]), putative transmembrane domains (TM), coronavirus-specific conserved domains (Ac, X, Y, ExoN, and MT), and one SARS-CoV specific insertion (SUD). For details, see references 11, 23, 24, 39 and references therein. Note that the arterivirus and coronavirus polyproteins are drawn to different scales. (B) Sequence alignment of the most conserved regions of the NendoU domain of several nidoviruses with cellular proteins from the XendoU family. Subdomains A (present in all proteins) and B (nidovirus-specific) are indicated. Key conserved residues are shown in bold. In the EAV sequence (bottom line), arrows indicate the amino acids targeted in this study by site-directed mutagenesis; amino acid numbers refer to their position in EAV pp1ab. Abbreviations and accession numbers are as follows: Npun 0562, hypothetical protein of Nostoc punctiforme, ZP_00106190; Poliv smB, pancreatic protein of Paralichthys olivaceus, BAA88246; Celeg Pp11, placental protein 11-like precursor of Caenorhabditis elegans, NP_492590; Xlaev endoU, endoU protein of Xenopus laevis, CAD45344; SARS-CoV, NC_004718; HCoV-229E, human coronavirus 229E, NC_002645; TGEV, transmissible gastroenteritis virus, NC_002306; MHV-A59, mouse hepatitis virus A59, NC_001846; BCoV-LUN, bovine coronavirus LUN, AF391542; IBV-B, avian infectious bronchitis virus Beaudette, NC_001451; EToV, equine torovirus, X52374; LDV-C, lactate dehydrogenase-elevating virus type C, NC_002534; PRRSV-LV, porcine reproductive and respiratory syndrome virus, strain Lelystad, M96262; PRRSV-VR, porcine reproductive and respiratory syndrome virus, strain ATCC VR2332, AAD12125; SHFV, simian hemorrhagic fever virus, NC_003092; and EAVB equine arteritis virus strain Bucyrus, NC_002532.
To obtain more information on the role of NendoU in the life cycle of nidoviruses in general and of arteriviruses in particular, we performed an extensive site-directed mutagenesis study. Using our EAV reverse genetics system, the domain was partially or completely deleted and a variety of point mutations were introduced at the positions of key conserved residues, including those presumably involved in catalysis. Mutants were tested for genome and sg RNA synthesis and, when viable, production of progeny virus. Whereas deletions and certain point mutations blocked all detectable viral RNA synthesis, other substitutions resulted in attenuated but viable virus mutants. A more detailed analysis of the latter mutants suggested a specific link between the NendoU domain and viral sg mRNA synthesis.
MATERIALS AND METHODS
Mutagenesis of nsp11 in the EAV full-length cDNA clone.
Mutations in the EAV nsp11 NendoU domain were introduced in an appropriate shuttle vector by standard site-directed PCR mutagenesis as described by Landt et al. (14). Restriction fragments containing the desired mutations were transferred to pEAV211, a wild-type (wt) EAV full-length cDNA (26), after which all PCR-derived sequences were verified to exclude the introduction of unwanted additional nucleotide changes. The mutations engineered in the nsp11-coding region are listed in Table 1. pEAV211 was used as the wt control in all experiments. The NendoU deletion mutant was constructed by mutagenesis PCR using a sense primer in which nucleotide 9091 was fused to nucleotide 9338 of the EAV genome, thereby making an in-frame deletion of the sequences encoding pp1ab residues His-2963 to Asp-3038. Similarly, mutants lacking the N-terminal part (His-2963 to Lys-3007) or the C-terminal part (Ser-3011 to Asp-3038) of the NendoU domain were constructed. The PCR products containing the deletions were transferred to pEAV211 by using appropriate restriction sites.
TABLE 1.
Overview of the genotype and first-cycle phenotype of EAV NendoU mutants used in this studya
| Construct | Mutation | wt codon | Mutant codon | Nsp3 IFAb | N IFAb | Infectious progeny (PFU/ml)c | Summary of phenotyped |
|---|---|---|---|---|---|---|---|
| pEAV211 | none (wild type) | + | + | + (∼107) | Wild type | ||
| H2963A | His-2963→Ala | CAU | GCA | + | + | + (∼102) | Small plaque size |
| H2963Q | His-2963→Gln | CAU | CAG | + | + | + (∼103) | Small plaque size |
| H2978A | His-2978→Ala | CAU | GCC | + | + | + (∼103) | Small plaque size |
| H2978Q | His-2978→Gln | CAU | CAA | + | + | + (∼102) | Small plaque size |
| H2963A/H2978A | His-2963→Ala | CAU | GCA | + | + | + (∼102) | Small plaque size |
| His-2978→Ala | CAU | GCC | |||||
| S2982A | Ser-2982→Ala | UCC | GCC | + | + | + (∼107) | Wild type |
| D2992A | Asp-2992→Ala | GAC | GCA | − | − | − | Non viable |
| D2992N | Asp-2992→Asn | GAC | AAC | + | + | + (∼107) | Wild type |
| D2992G | Asp-2992→Gly | GAC | GGC | + | + | + (∼107) | Wild type |
| K3004A | Lys-3004→Ala | AAA | GCA | + | + | + (∼106-107) | Wild type |
| K3004H | Lys-3004→His | AAA | CAU | + | + | + (∼106-107) | Wild type |
| K3004R | Lys-3004→Arg | AAA | AGA | + | + | + (∼107) | Wild type |
| K3007A | Lys-3007→Ala | AAA | GCC | + | + | + (∼102-103) | Small plaque size |
| K3007H | Lys-3007→His | AAA | CAU | + | + | + (∼102-103) | Small plaque size |
| K3007R | Lys-3007→Arg | AAA | CGG | + | + | + (∼103) | Small plaque size |
| S3011A | Ser-3011→Ala | AGC | GCC | + | + | + (∼105) | Intermediate plaque size |
| S3011T | Ser-3011→Thr | AGC | ACC | + | + | + (∼107) | Wild type |
| S3011C | Ser-3011→Cys | AGC | UGC | + | + | + (∼105) | Intermediate plaque size |
| D3014A | Asp-3014→Ala | GAU | GCU | − | − | − | Nonviable |
| D3014E | Asp-3014→Glu | GAU | GAG | − | − | − | Nonviable |
| D3014N | Asp-3014→Asn | GAU | AAC | − | − | − | Nonviable |
| S3030A | Ser-3030→Ala | AGU | GCC | + | + | + (∼106) | Intermediate plaque size |
| S3030T | Ser-3030→Thr | AGU | ACG | + | + | + (∼105) | Intermediate plaque size |
| S3030C | Ser-3030→Cys | AGU | UGC | + | + | + (∼106) | Intermediate plaque size |
| D3038A | Asp-3038→Ala | GAU | GCU | − | − | − | Nonviable |
| D3038E | Asp-3038→Glu | GAU | GAA | − | − | − | Nonviable |
| D3038N | Asp-3038→Asn | GAU | AAU | − | − | − | Nonviable |
Data from two independent transfection experiments are summarized. +, positive; −, negative.
Transfected cells were analyzed after one cycle of replication at 19 h posttransfection. IFA, immunofluorescence assay.
Results indicate the detection of infectious progeny (plaque assays) in medium from transfected cells harvested at 19 h posttransfection; approximate virus titers are shown between brackets.
Nonviable indicates that no replication detected in immunofluorescence assays for nsp3 and N protein (see Fig. 4), and no infectious progeny were detected at 19 h posttransfection; small plaques were approximately 20% of the wt plaque size. The diameter of intermediate-size plaques ranged from 30 to 80% of the wt plaque size.
RNA transfection, EAV infection, immunoprecipitation, and immunofluorescence analysis.
Baby hamster kidney (BHK-21; ATCC CCL10) cells were electroporated with in vitro-derived RNA transcripts of wt and mutant EAV full-length cDNA clones as described previously (29). Infection of the BHK-21 cells with EAV-containing supernatants harvested after transfection, 35S-labeling of protein synthesis, and immunoprecipitation analysis of transfected cells were performed by using the methods of de Vries et al. (7). To ensure the quantitative nature of the immunoprecipitation, lysate and antiserum were titrated and it was confirmed that all of the target protein had been precipitated by performing a control reimmunoprecipitation on the supernatant of the primary immunoprecipitation. Immunofluorescence analyses with EAV-specific antisera against nsp3 (98E3) and N (MAb 3E2) were performed according to the method of van der Meer et al. (28). To monitor and compare transfection efficiencies, cells were labeled with anti-nsp3 serum 98E3 and Hoechst 33258 (1 μg/ml final concentration) to stain nuclei (17). For plaque assays, subconfluent monolayers of BHK-21 cells were infected with 10-fold serial dilutions of wt or mutant EAV. Following a 1-h incubation, a 1% agar overlay was applied and cells were incubated at 39.5°C. Plaques were stained with 0.75% (wt/vol) crystal violet 4 days postinfection. To test nonviable mutants for a possible temperature-sensitive phenotype, plaque assays were also performed at 34°C.
RNA isolation and analysis.
Intracellular RNA was isolated by using the acidic phenol method as described by Van Marle et al. (32). RNA was separated in denaturing agarose-formaldehyde gels and detected by hybridization to a 32P-labeled probe E154 (5′-TTGGTTCCTGGGTGGCTAATAACTACTT-3′), which is complementary to the 3′ end of the EAV genome and recognizes the genome and all sg mRNAs. Phosphorimager screens were exposed to the hybridized gels and scanned with a Personal Molecular Imager FX (Bio-Rad). Band intensities were quantified with Quantity One v4.2.2 software (Bio-Rad).
The presence of the original NendoU mutation(s) in progeny virus was verified in the following way: supernatant harvested at the indicated time point was used to infect fresh BHK-21 cells. At 24 h postinfection, total RNA was isolated and used in reverse transcription (RT)-PCR experiments with E533 (5′-CAATCACAGAAATAGCAAAATCAGC-3′, nt 9799 to 9823) as antisense primer for RT and PCR and E519 (5′-CGAGGAAAAGTTTGCCGCCGC-3′, nt 9082 to 9102) as sense primer for PCR. The PCR products were purified and sequenced directly using standard protocols.
RESULTS
Deletion of the EAV NendoU domain is lethal.
The boundaries of EAV nsp11 are Ser-2838 and Glu-3056 (using pp1ab numbering) (30), with residues His-2961 to Phe-3039 forming the conserved 78-residue core of the NendoU domain. An alignment of the EAV NendoU domain with cellular and nidovirus homologs is shown in Fig. 1B. As explained in the introduction, the N-terminal part (His-2963 to Ser-3011; subdomain A) is conserved across the XendoU/NendoU family and contains the EAV equivalents (His-2963, His-2978, and Lys-3007) of the putative catalytic residues identified in XendoU (His-162, His-178, and Lys-224) (10) and HCoV-229E nsp15 (His-6345, His-6360, and Lys-6401) (13). However, the C-terminal 30 residues (subdomain B) of the NendoU domain appear to be specific for nidoviruses and include two conserved Asp residues (Asp-3014 and Asp-3038), which were found to be equally indispensable for in vitro endoribonuclease activity (13). Moreover, a reverse genetics experiment showed that an Asp-6408→Ala substitution completely blocked HcoV-229E RNA synthesis (13).
As an initial test of its importance for EAV replication, in-frame deletion mutants of the EAV NendoU domain were engineered and transferred to the infectious cDNA clone (29). Either the complete NendoU-coding sequence was deleted from the nsp11-coding region or the sequences roughly corresponding to subdomain A (His-2963 to Lys-3007) or subdomain B (Ser-3011 to Asp-3038) were deleted. Electroporation into BHK-21 cells of in vitro-transcribed RNA from these constructs and subsequent immunostaining showed that, in all three cases, the virus was no longer viable (data not shown), suggesting that the complete NendoU domain is essential for EAV replication. Formally, detrimental effects of these deletions (and other lethal mutations; see below) at the level of pp1ab proteolysis cannot be excluded even though their C-terminal border was relatively distant from the nsp11/12 cleavage site. Unfortunately, straightforward analysis in a trans-cleavage assay of the replicase processing event at the nsp11/12 junction was previously found to be impossible (30).
On the basis of the alignment presented in Fig. 1B, both absolutely and less-conserved residues of EAV NendoU were targeted by site-directed mutagenesis and reverse genetics (Table 1). Each of the five invariant residues was replaced by Ala and one or two additional conservative substitutions. Also some less conserved and variable residues were replaced as a control for the alignment and to confirm that amino acid substitutions in general could be tolerated in this part of the polyprotein. Mutations were introduced into an EAV infectious cDNA clone and, after electroporation of in vitro-transcribed RNA, virus viability was analyzed at different time points after transfection. The initial screening was done on the basis of a double immunofluorescence assay for replicase subunit nsp3 and the nucleocapsid (N) protein, which can be used as markers for the synthesis of genomic and sg mRNA7, respectively (31). Cell culture supernatants were harvested around the end of the first replication cycle (19 h posttransfection) and tested in a plaque assay for the production of infectious virus progeny. Table 1 summarizes the mutations that were tested and the results of the first-cycle analyses that were obtained from two independent transfection experiments.
Both conserved Asp residues in NendoU subdomain B are critically involved in arterivirus RNA synthesis.
We first probed the importance of the pair of Asp residues (Asp-3014 and Asp-3038) in subdomain B that are fully conserved in the NendoU domain of all known nidoviruses. Both conservative (Asp→ Asn or Asp→ Glu) and nonconservative (Asp→ Ala) substitutions were tested, but none of these were tolerated. In a first-cycle analysis, no sign of nsp3 or N protein synthesis was detected in immunofluorescence assays of transfected cells and the production of infectious progeny virus could not be demonstrated in plaque assays (Table 1). Ser-3030 of EAV NendoU is the third subdomain B residue that is fully conserved in all nidoviruses. Also, this amino acid was replaced with Ala and, more conservatively, with Thr and Cys. In all three cases, replication of the mutant virus was significantly reduced, with a plaque size that was 30 to 60% of that for the wt control (termed “intermediate size” in Table 1) and virus titers being 10- to 100-fold reduced. Another Ser residue that was probed, Ser-3011 of EAV NendoU, is located at the border between subdomains A and B. A Thr residue is found at this position in some cellular proteins of the XendoU family and in all nidovirus homologs, with the exception of SARS-CoV (Fig. 1B). To probe the importance of this residue and to corroborate the quality of the alignment in Fig. 1B, Ser-3011 was replaced by Thr. This indeed produced a mutant virus with a phenotype that could not be discriminated from that of the wt control both in terms of the virus titer that was obtained and in terms of plaque size. In contrast, substitutions of the Ser-3011 residue by Ala or Cys were less well tolerated: replication of these mutant viruses was delayed, plaques were 60 to 80% of the wt size, and virus titers were ∼100-fold reduced. Taken together, our data confirmed and extended the general importance for nidovirus RNA synthesis of NendoU subdomain B and, in particular, its pair of conserved Asp residues.
Substitution of putative catalytic residues in NendoU subdomain A is not lethal.
Obviously, the EAV equivalents (His-2963, His-2978, and Lys-3007) of the three residues previously implicated in XendoU/NendoU catalysis (see above) were primary targets for site-directed mutagenesis of subdomain A. Both His residues were replaced with Ala or Gln, whereas Lys-3007 was substituted with Ala, His, or Arg. However, none of these seven substitutions was lethal, although all mutant viruses were severely impaired relative to the wt control. Irrespective of the replacement used, plaque sizes were reduced to approximately 20% of the wt plaque size and virus titers in the order of 102 to 103 PFU/ml were measured in transfected cell culture supernatant harvested at 19 h posttransfection, a reduction of 4 to 5 log units compared to that of the wt virus. The reduced plaque size is illustrated in Fig. 2 for mutants His-2963→Gln and His-2978→Ala. There was no apparent synergistic effect of combining the Ala substitutions of both conserved His residues into a double mutant (mutant H2963A/H2978A): this virus displayed a similar small plaque size phenotype and also produced ∼102 PFU/ml by 19 h posttransfection.
FIG. 2.
Plaque morphology of selected EAV NendoU mutants. The plaque sizes from mutants carrying mutations at the position of the putative catalytic residues His-2963 and His-2978 were approximately fivefold smaller than those of the wt virus. Plaque sizes of mutants carrying other substitutions at the positions of His-2963 and His-2978 or replacements of Lys-3007 were comparable to the plaque sizes depicted in this figure.
To verify the presence of the original mutations in the progeny of the mutants, fresh BHK-21 cells were infected with transfection supernatants harvested after 46 to 72 h. Intracellular RNA was isolated from these infected cells after 24 h, and the direct sequence analysis of RT-PCR products that was derived from this material confirmed that reversion to the wt residue had not occurred for any of the mutants with small plaque sizes. In fact, the two His→Ala mutations in this double H2963A/H2978A mutant were found to be stable for up to 10 passages in BHK-21 cells (data not shown). In conclusion, although the substitution of equivalent residues in both XendoU and coronavirus NendoU reduced in vitro endoribonuclease activity to undetectable levels (10, 13), our in vivo data revealed that EAV mutants carrying such mutations are capable of a consistently low but significant level of replication.
In addition to the replacement of the conserved subdomain A residues, several additional EAV NendoU residues were targeted, mainly as controls for the alignment and the substitutions described above. Replacement of Lys-3004, serving as a control for Lys-3007 mutagenesis, did not have any effect on virus viability, irrespective of whether Ala, His, or Asn was introduced. Also, substitution of a Ser residue (Ser-2982→Ala) that is fully conserved in Arteriviridae but not in Coronaviridae proved to be neutral with regard to virus replication. A final control substitution, replacement of the charged Asp-2992 by Ala, resulted in a nonviable virus. However, upon closer inspection of the alignment (Fig. 1B), we decided to also replace this residue with Gly (present in the mouse arterivirus lactate dehydrogenase-elevating virus) and Asn (which is more conservative compared to the Glu or Gln present at this position in most other nidoviruses). Both the Asp-2992→Gly and the Asp-2992→Asn mutants proved to have wt phenotype. Since, in contrast to Ala, both Gly and Asn frequently reside in β-turns, this result suggests that this property may be important for the residue at this position.
In summary, the EAV system was found to tolerate the substitution of three (putative) active site residues of NendoU which have been absolutely conserved throughout nidovirus evolution. Although replication of these mutants was profoundly impaired, as evident from reduced progeny titers and smaller plaque sizes, this phenotype was in clear contrast with that described above for the mutants carrying substitutions of the pair of conserved Asp residues in subdomain B.
Selective reduction of subgenomic RNA synthesis in mutants with a reduced plaque size.
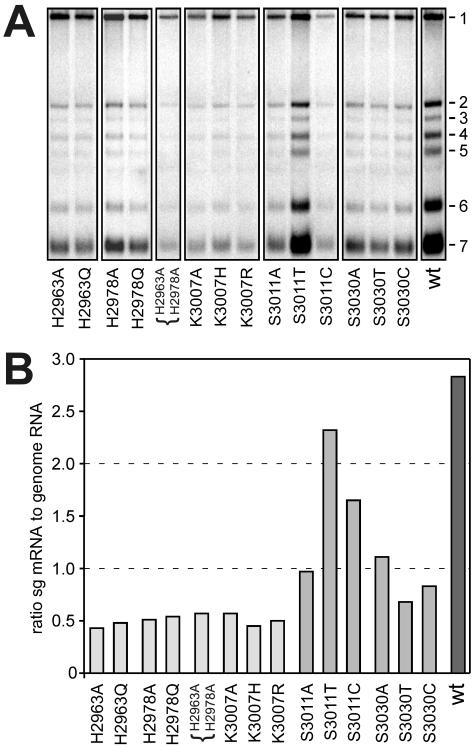
The intracellular RNA synthesis of selected mutants was analyzed by isolating total RNA from transfected BHK-21 cells at 13 h posttransfection, which, in electroporation experiments, approximately equals the peak of RNA synthesis during the first cycle of replication. The viral genome (RNA1) and sg mRNAs (RNA2 to RNA7) were separated in agarose gels and visualized by hybridization. No virus-specific RNAs were detected for the nonviable Asp-3014 and Asp-3038 mutants (data not shown). In line with its previously described wt phenotype, an RNA pattern very similar to that of wt virus was observed for the Ser-3011→Thr mutant. All other mutants analyzed, including those with substitutions at the positions of His-2963, His-2978, and Lys-3007, showed a modest but significant reduction of viral RNA synthesis compared to that of the wt control (Fig. 3A).
FIG. 3.
Analysis of intracellular RNA synthesis for selected EAV NendoU mutants. (A) Hybridization analysis of intracellular RNA isolated from transfected BHK-21 cells at 13 h posttransfection. The RNA was hybridized to an oligonucleotide complementary to the 3′ end of the genome, recognizing both the genomic (RNA1) and sg mRNAs 2 to 7. (B) Reduced sg mRNA synthesis by nsp11 mutants with a small or intermediate plaque size phenotype. Using the gel depicted in Fig. 3A, the signals for the genomic RNA and the most abundant sg mRNAs (2, 6, and 7) in the same lane were quantified. The graph shows the ratio (RNA2 + RNA6 + RNA7)/RNA1 for each mutant and the wt control and illustrates the selective reduction of sg mRNA synthesis in NendoU mutants with a small or intermediate plaque size phenotype.
The relative amounts of genomic RNA and the three most abundant sg mRNAs (mRNAs 2, 6, and 7) were quantified by phosphorimager analysis. When the amount of genome RNA in each lane was set at 100%, the usual value of between 2:1 and 3:1 was obtained for the ratio of sg mRNAs versus genome RNA in the wt control and mutant S3011T, which has a wt phenotype (Fig. 3B). However, in all other mutants, a specific reduction of sg mRNA synthesis was detected. This effect was most prominent in the mutants that produced the smallest plaques, i.e., those with substitutions of His-2963, His-2978, Lys-3007, and the His-2963/His-2978 double mutant. The reduction of sg mRNA synthesis was less striking but still consistently observed for the Ser-3011 and Ser-3030 mutants, which had intermediate plaque sizes (Table 1).
To make it possible to monitor viral RNA synthesis beyond the end of the first replication cycle and without virus spreading to initially untransfected cells, we engineered mutants that were negative for virus production. These constructs were based on the previously described ORF6 knockout mutant (ΔORF6) (16), which lacks membrane (M) protein expression and therefore is unable to produce progeny virus (36, 38). We transferred the NendoU His-2963→Ala/His-2978→Ala double mutation and the Lys-3007→Ala single mutation to the ΔORF6 backbone and compared RNA synthesis to that of the parental ΔORF6 mutant, which contains a wt replicase gene. Viral RNA synthesis was analyzed at 15, 18, 21, and 24 h posttransfection, and a selective reduction in sg mRNA synthesis was again observed for both NendoU mutants at all four time points (data not shown). This indicated that the earlier measurements (Fig. 3B) were not likely to be compromised by the possibility that sg RNA synthesis of these generally impaired NendoU mutants simply lagged behind. Relative to the wt control, these mutants consistently underproduced sg mRNAs, suggesting the direct or indirect involvement of NendoU in a process that is relatively more important for sg mRNA production than for genomic RNA synthesis.
Structural protein synthesis was studied in more detail for a selection of mutants (Fig. 4). Immunofluorescence analysis confirmed that N protein expression of mutants with small and intermediate plaque sizes was reduced and lagged behind. Whereas all cells in the wild-type control were double positive for replicase and N protein at 13 h posttransfection, many cells transfected with the mutants were fully positive for replicase but either still negative or only weakly positive for the N protein (Fig. 4). Only at 20 to 24 h posttransfection had all cells become clearly double positive for both marker proteins (data not shown). In a combined RNA and immunoprecipitation analysis at 13 h posttransfection (Fig. 4) using samples from equal numbers of transfected cells, the amounts of sg RNA6 and RNA7 accumulated by the small plaque size mutants His-2963→Ala and Lys-3007→Arg and intermediate plaque size mutant Ser-3011→Ala were compared to the synthesis of the corresponding structural proteins M and N. Proteins were 35S-labeled from 9 to 13 h posttransfection and immunoprecipitated with monospecific rabbit antisera (7). Compared to the wild-type control, the structural protein synthesis of the three mutants was clearly reduced but not to an extent that would readily explain the large differences (up to 5 log units) in infectious progeny titers. When comparing mutants with small and intermediate plaque sizes (showing differences of 2 to 3 log units in infectious progeny titers), sg RNA and structural protein levels of the latter were found to be threefold higher at most. Finally, an anti-nsp11 serum was used to confirm that mutants of small and intermediate plaque sizes produced similar amounts of cleaved nsp11 compared to that for the wild-type control (data not shown), making it unlikely that an nsp11 processing defect is the explanation for the phenotypes of these mutants.
FIG. 4.
Analysis of structural protein expression of selected EAV NendoU mutants. The top panel shows a hybridization analysis of sg RNA6 and RNA7 isolated from transfected BHK-21 cells at 13 h posttransfection (also see Fig. 3). On the right side, the corresponding immunoprecipitation data are shown for the viral structural proteins M and N, which were 35S-labeled from 9 to 13 h posttransfection. The bottom panel shows an immunofluorescence analysis (also at 13 h posttransfection) of cells transfected with small plaque size mutant Lys-3007→Arg, intermediate plaque size mutant Ser-3011→Ala, or the wild-type control. Cells were double-labeled for replicase subunit nsp3 and structural protein N. All images were recorded with the same exposure time and contrast/brightness settings. Whereas cells transfected with the wild-type control were double positive for replicase and N protein, many cells transfected with the mutants were fully positive for replicase but either still negative or only weakly positive for the N protein.
DISCUSSION
Pleiotropic effects upon EAV NendoU mutagenesis.
In the wake of the 2003 SARS outbreak, the increased interest in the giant and poorly characterized coronavirus replicase triggered an in-depth reanalysis of the nonstructural proteins of SARS-CoV, other coronaviruses, and other nidoviruses. As a direct result, the domain previously referred to as “conserved C-terminal nidovirus-specific domain” (5) was predicted and proven to have endoribonuclease activity (1, 13, 23). Using an HCoV-229E reverse genetics system, Ivanov and coworkers (13) showed that a single Asp→Ala mutation in the nidovirus-specific subdomain B of NendoU completely blocked viral RNA synthesis. Thus, they provided the first experimental evidence for the expected key role of the highly conserved, though nidovirus-specific, NendoU domain in the viral life cycle.
In the present study, using the reverse genetics system for the arterivirus EAV, we have considerably extended and refined the in vivo analysis of NendoU function. As previously concluded by Ivanov and colleagues (13), the results obtained with NendoU deletion mutants and certain subdomain B point mutants indicated that NendoU is critically involved in general nidovirus RNA synthesis. Although the replacement of one of three putative catalytic residues in subdomain A also exerted a major effect on general virus viability, it was somewhat surprising that only a modest effects on viral genome and sg mRNA synthesis were observed for these mutants (Fig. 3), raising new questions on the role of the NendoU domain in the nidovirus life cycle.
Formally, we cannot exclude (fundamental) differences between the role of NendoU in coronaviruses and arteriviruses and the possibility that equivalent mutations may produce different phenotypes in different systems. Reverse genetics data for coronavirus NendoU mutants carrying replacements of the conserved His and Lys residues in subdomain A have not yet been described. Nevertheless, the result obtained with the single HCoV-229E Asp mutant that was tested (13) was identical to that obtained with the corresponding mutant in the EAV system. On the arterivirus side, it is unfortunate that we have not yet succeeded in establishing an in vitro NendoU assay for EAV. This is due to complications during protein expression in Escherichia coli, which apparently tolerates the expression of large amounts of an Asp-3014→Ala mutant protein but not of the wt EAV nsp11 (data not shown). Although the experimental proof that EAV nsp11 indeed possesses endoribonuclease activity is lacking, all residues reported to be important for the in vitro NendoU activity of its coronavirus ortholog are conserved in arteriviruses (Fig. 1B). Thus, we consider it unlikely that this activity would ultimately turn out to be lacking in arterivirus nsp11.
Is the function of NendoU uniform in the life cycle of different nidoviruses?
It is perhaps remarkable that replacements of the most conserved residues in the XendoU/NendoU protein family fully blocked the in vitro endoribonuclease activity but were not lethal for EAV in the reverse genetics approach used in this study. Arguably, this apparent contradiction may reflect the higher sensitivity of the in vivo assay used in this study compared to that of the in vitro system employed by Ivanov et al. (13). In the latter assay, replacement of the pair of His residues and the Lys in subdomain A and the pair of Asp residues in subdomain B yielded equally inactive enzymes. However, it is highly unlikely that NendoU (or XendoU) mutants with very low but different residual activities (e.g., 1% or less of the wt activity) can be discriminated using this type of experiment. In contrast, our biological assays and, in particular, the quantification of infectious progeny virus produced by the mutants, allowed us to establish differences of up to 5 log units (Table 1) and can thus be considered to be more sensitive. Consequently, one explanation for the phenotypic differences between the Asp mutants and the His/Lys mutants would be a higher residual endoribonuclease activity of the latter, which would suffice to support a certain reduced level of virus replication. This explanation would imply that the Asp residues in subdomain B, which are lacking in the cellular branch of the XendoU/NendoU family, play a more important role in NendoU function than the fully conserved His and Lys residues in subdomain A, which were proposed to have a catalytic role on the basis of experimental data and theoretical considerations relating to the reaction mechanisms of other ribonucleases (10).
Despite the common ancestry of arterivirus and coronavirus replicases, the polyprotein of coronaviruses is considerably larger and in addition to NendoU harbors a series of other putative RNA-processing activities with possibly interconnected functions (23). It should also be noted, as observed previously by de Vries and colleagues (8), that the NendoU domain is associated with a different N-terminal domain when coronavirus nsp15 and arterivirus nsp11 are compared. Within the currently defined NendoU domain, the role of subdomain B and its pair of conserved Asp residues is intriguing. Gioia et al. (10) suggested that the two NendoU Asp residues might be the equivalents of two XendoU Glu residues (Glu-161 and Glu-167) which were proposed to participate in catalysis on the basis of mutagenesis studies. However, these two amino acids more or less flank the first conserved His residue (His-162 in XendoU) and thus occupy a completely different position in the primary structure of the protein compared to the pair of conserved Asp residues in NendoU subdomain B. Clearly, structural information for both cellular and nidoviral representatives of this novel endoribonuclease family will be required to understand the domain composition and catalytic mechanism of the enzyme and the specific role of subdomain B in the viral subgroup. At present, it should even be considered that the latter domain, although structurally adjacent to NendoU, could be functionally separate and fulfill a different role that might be the basis for some of the phenotypic differences observed in our collection of EAV mutants.
Dissecting the role of NendoU in the EAV life cycle.
The pleiotropic effects observed upon EAV NendoU mutagenesis and their analysis using the complex system of a replicating RNA virus in its host cell make it difficult to assess at how many levels NendoU may actually influence EAV replication. Clearly, specific deletions and mutations in NendoU can abolish all viral RNA synthesis, supporting a general and critical role in this process. Furthermore, we observed a stronger reduction of sg mRNA synthesis than genomic synthesis in a subset of the mutants, but these findings could also be attributed to the former process being more sensitive to a general debilitating effect of NendoU mutagenesis on virus viability. Still, it appears that the His-2963/His-2978/Lys-3007 mutants form a separate group and that these mutations exert a consistently stronger effect on sg RNA synthesis than, e.g., substitutions of Ser-3011 or Ser-3030 (Fig. 3B). Also, in terms of the production of infectious virus progeny (virus titers and plaque size), these mutants appear to segregate into two clusters (Table 1).
When comparing the wt virus, the intermediate plaque size mutants (replacements of Ser-3011 and Ser-3030), and the small plaque size mutants (replacements of His-2963, His-2978, and Lys-3007), it appears that relatively small differences in RNA synthesis (certainly not more than 10-fold) (Fig. 3) are translated into unexpectedly large differences in virus production (up to 5 log units) (Table 1). These findings could be interpreted to imply that NendoU has an additional function downstream of viral RNA synthesis, for example, in a pathway leading to virion assembly. However, it should also be noted that little is known about the process in which the seven EAV structural proteins, expressed from six sg mRNAs that are each made in constant but different quantities, interact to encapsidate the viral genome and form a virus particle (36). In particular, it is unclear whether the availability of any of these proteins is a rate-limiting factor for the assembly process. Therefore, it certainly cannot be excluded that a relatively small reduction in the synthesis of one or multiple sg mRNAs and a corresponding change in structural protein expression (Fig. 4) could actually cause a disproportional, much larger reduction in the production of infectious progeny.
In view of its conservation in and specificity for the order Nidovirales, the NendoU domain was proposed to be involved in a nidovirus-specific, conserved function. The synthesis of sg mRNAs, a hallmark of nidoviruses, is an example of such a function (4, 17-19) and, based on the consensus sequence of the regulatory TRS motif, Ivanov and colleagues (13) proposed a role for the enzyme in the attenuation step during discontinuous sg minus-strand RNA synthesis. However, the EAV data presented in this study do not support a critical role for NendoU in sg RNA synthesis. Only a relatively small effect on RNA synthesis was observed for the putative active site mutants of EAV NendoU (Fig. 3B). Although, admittedly, sg RNA synthesis was affected to a greater extent than genome synthesis (see above), a much more pronounced effect would have been expected if NendoU were a key factor in nidovirus sg RNA production. Previous studies on other EAV replicase functions have shown that such specific effects can indeed be induced. For instance, deletion of the N-terminal replicase subunit nsp1 completely abolished EAV sg mRNA production without affecting genome replication (25). Also, a Ser-2429→ Pro mutation in the so-called “hinge region” of EAV nsp10, which connects the N-terminal (putative) zinc finger domain and the C-terminal helicase, resulted in an approximately 100-fold reduction of sg mRNA synthesis, while, again, genome replication was not affected (29, 33). Compared to these examples, the defects in RNA synthesis observed for the viable NendoU mutants described in this study should be classified as relatively minor and less specific for the process of sg mRNA synthesis.
In conclusion, notwithstanding the viability of many mutants, the EAV NendoU motif in nsp11 is an important replicase domain, although the consequences of replacement of some of its core residues were less black and white than expected beforehand. Since specific NendoU mutants were found to be replication competent and genetically stable, it will be interesting to analyze their long-term evolution both in BHK-21 cells and in more natural settings like equine cell lines, in which such mutants may have a more pronounced phenotype. In addition, the identification of the natural substrates of coronavirus and arterivirus NendoU and structural information on members of the XendoU/NendoU family will be essential for a better understanding of the mutant phenotypes described in this study.
Acknowledgments
We thank Cheng Kao, Konstantin Ivanov, John Ziebuhr, and Willy Spaan for helpful discussions and/or sharing data prior to publication. We also thank John Ziebuhr for critical reading of the manuscript and acknowledge the technical assistance of Erwin van den Born.
REFERENCES
- 1.Bhardwaj, K., L. Guarino, and C. C. Kao. 2004. The severe acute respiratory syndrome coronavirus Nsp15 protein is an endoribonuclease that prefers manganese as a cofactor. J. Virol. 78:12218-12224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cavanagh, D. 1997. Nidovirales: a new order comprising Coronaviridae and Arteriviridae. Arch. Virol. 142:629-633. [PubMed] [Google Scholar]
- 3.Cowley, J. A., C. M. Dimmock, K. M. Spann, and P. J. Walker. 2000. Gill-associated virus of Penaeus monodon prawns: an invertebrate virus with ORF1a and ORF1b genes related to arteri- and coronaviruses. J. Gen. Virol. 81:1473-1484. [DOI] [PubMed] [Google Scholar]
- 4.Cowley, J. A., C. M. Dimmock, and P. J. Walker. 2002. Gill-associated nidovirus of Penaeus monodon prawns transcribes 3′-coterminal subgenomic mRNAs that do not possess 5′-leader sequences. J. Gen. Virol. 83:927-935. [DOI] [PubMed] [Google Scholar]
- 5.den Boon, J. A., E. J. Snijder, E. D. Chirnside, A. A. F. de Vries, M. C. Horzinek, and W. J. M. Spaan. 1991. Equine arteritis virus is not a togavirus but belongs to the coronaviruslike superfamily. J. Virol. 65:2910-2920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.deVries, A. A. F., E. D. Chirnside, P. J. Bredenbeek, L. A. Gravestein, M. C. Horzinek, and W. J. M. Spaan. 1990. All subgenomic mRNAs of equine arteritis virus contain a common leader sequence. Nucleic Acids Res. 18:3241-3247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.de Vries, A. A. F., E. D. Chirnside, M. C. Horzinek, and P. J. M. Rottier. 1992. Structural proteins of equine arteritis virus. J. Virol. 66:6294-6303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.de Vries, A. A. F., M. C. Horzinek, P. J. M. Rottier, and R. J. de Groot. 1997. The genome organization of the Nidovirales: similarities and differences between arteri-, toro-, and coronaviruses. Semin. Virol. 8:33-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Feder, M., J. Pas, L. S. Wyrwicz, and J. M. Bujnicki. 2003. Molecular phylogenetics of the RrmJ/fibrillarin superfamily of ribose 2′-O-methyltransferases. Gene 302:129-138. [DOI] [PubMed] [Google Scholar]
- 10.Gioia, U., P. Laneve, M. Dlakic, M. Arceci, I. Bozzoni, and E. Caffarelli. 2005. Functional characterization of XendoU, the endoribonuclease involved in small nucleolar RNA biosynthesis. J. Biol. Chem. 280:18996-19002. [DOI] [PubMed] [Google Scholar]
- 11.Gorbalenya, A. E. 2001. Big nidovirus genome: when count and order of domains matter. Adv. Exp. Biol. Med. 494:1-17. [PubMed] [Google Scholar]
- 12.Gorbalenya, A. E., E. V. Koonin, A. P. Donchenko, and V. M. Blinov. 1989. Coronavirus genome: prediction of putative functional domains in the non-structural polyprotein by comparative amino acid sequence analysis. Nucleic Acids Res. 17:4847-4861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ivanov, K. A., T. Hertzig, M. Rozanov, S. Bayer, V. Thiel, A. E. Gorbalenya, and J. Ziebuhr. 2004. Major genetic marker of nidoviruses encodes a replicative endoribonuclease. Proc. Natl. Acad. Sci. USA 101:12694-12699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Landt, O., H.-P. Grunert, and U. Hahn. 1990. A general method for rapid site-directed mutagenesis using the polymerase chain reaction. Gene 96:125-128. [DOI] [PubMed] [Google Scholar]
- 15.Laneve, P., F. Altieri, M. E. Fiori, A. Scaloni, I. Bozzoni, and E. Caffarelli. 2003. Purification, cloning, and characterization of XendoU, a novel endoribonuclease involved in processing of intron-encoded small nucleolar RNAs in Xenopus laevis. J. Biol. Chem. 278:13026-13032. [DOI] [PubMed] [Google Scholar]
- 16.Molenkamp, R., H. van Tol, B. C. D. Rozier, Y. van der Meer, W. J. M. Spaan, and E. J. Snijder. 2000. The arterivirus replicase is the only viral protein required for genome replication and subgenomic mRNA transcription. J. Gen. Virol. 81:2491-2496. [DOI] [PubMed] [Google Scholar]
- 17.Pasternak, A. O., E. van den Born, W. J. M. Spaan, and E. J. Snijder. 2001. Sequence requirements for RNA strand transfer during nidovirus discontinuous subgenomic RNA synthesis. EMBO J. 20:7220-7228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sawicki, S. G., and D. L. Sawicki. 1995. Coronaviruses use discontinuous extension for synthesis of subgenome-length negative strands. Adv. Exp. Biol. Med. 380:499-506. [DOI] [PubMed] [Google Scholar]
- 19.Sawicki, S. G., and Sawicki, D. L. 2005. Coronavirus transcription: a perspective, p. 31-55. In Enjuanes, L. (ed.), Coronavirus replication and reverse genetics. Springer, Berlin, Germany. [DOI] [PMC free article] [PubMed]
- 20.Seybert, A., C. C. Posthuma, L. C. van Dinten, E. J. Snijder, A. E. Gorbalenya, and J. Ziebuhr. 2005. A complex zinc finger controls the enzymatic activities of nidovirus helicases. J. Virol. 79:696-704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Seybert, A., L. C. van Dinten, E. J. Snijder, and J. Ziebuhr. 2000. Biochemical characterization of the equine arteritis virus helicase suggests a close functional relationship between arterivirus and coronavirus helicases. J. Virol. 74:9586-9593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Siddell, S. G., Ziebuhr, J., and Snijder, E. J. 2005. Coronaviruses, toroviruses, and arteriviruses, 823-856. In Mahy, B. W. and V. ter Meulen (ed.), Topley and Wilson's microbiology and microbial infections, vol. 1: virology. Hodder Arnold, London, England. [Google Scholar]
- 23.Snijder, E. J., P. J. Bredenbeek, J. C. Dobbe, V. Thiel, J. Ziebuhr, L. L. M. Poon, Y. Guan, M. Rozanov, W. J. M. Spaan, and A. E. Gorbalenya. 2003. Unique and conserved features of genome and proteome of SARS-coronavirus, an early split-off from the coronavirus group 2 lineage. J. Mol. Biol. 331:991-1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Snijder, E. J., Siddell, S. G., and Gorbalenya, A. E. 2005. The order Nidovirales, p. 390-404. In Mahy, B. W., and V. ter Meulen (ed.), Topley and Wilson's microbiology and microbial infections, vol. 1: virology. Hodder Arnold, London, England. [Google Scholar]
- 25.Tijms, M. A., L. C. van Dinten, A. E. Gorbalenya, and E. J. Snijder. 2001. A zinc finger-containing papain-like protease couples subgenomic mRNA synthesis to genome translation in a positive-stranded RNA virus. Proc. Natl. Acad. Sci. USA 98:1889-1894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.van den Born, E., A. P. Gultyaev, and E. J. Snijder. 2004. Secondary structure and function of the 5′-proximal region of the equine arteritis virus RNA genome. RNA 10:424-437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.van den Born, E., C. C. Posthuma, A. P. Gultyaev, and E. J. Snijder. 2005. Discontinuous subgenomic RNA synthesis in arteriviruses is guided by an RNA hairpin structure located in the genomic leader region. J. Virol. 79:6312-6324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.van der Meer, Y., H. van Tol, J. Krijnse Locker, and E. J. Snijder. 1998. ORF1a-encoded replicase subunits are involved in the membrane association of the arterivirus replication complex. J. Virol. 72:6689-6698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.van Dinten, L. C., J. A. den Boon, A. L. M. Wassenaar, W. J. M. Spaan, and E. J. Snijder. 1997. An infectious arterivirus cDNA clone: identification of a replicase point mutation which abolishes discontinuous mRNA transcription. Proc. Natl. Acad. Sci. USA 94:991-996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.van Dinten, L. C., S. Rensen, W. J. M. Spaan, A. E. Gorbalenya, and E. J. Snijder. 1999. Proteolytic processing of the open reading frame 1b-encoded part of arterivirus replicase is mediated by nsp4 serine protease and is essential for virus replication. J. Virol. 73:2027-2037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.van Dinten, L. C., H. van Tol, A. E. Gorbalenya, and E. J. Snijder. 2000. The predicted metal-binding region of the arterivirus helicase protein is involved in subgenomic mRNA synthesis, genome replication, and virion biogenesis. J. Virol. 74:5213-5223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.van Marle, G., J. C. Dobbe, A. P. Gultyaev, W. Luytjes, W. J. M. Spaan, and E. J. Snijder. 1999. Arterivirus discontinuous mRNA transcription is guided by base pairing between sense and antisense transcription-regulating sequences. Proc. Natl. Acad. Sci. USA 96:12056-12061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.van Marle, G., L. C. van Dinten, W. Luytjes, W. J. M. Spaan, and E. J. Snijder. 1999. Characterization of an equine arteritis virus replicase mutant defective in subgenomic mRNA synthesis. J. Virol. 73:5274-5281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.van Vliet, A. L., S. L. Smits, P. J. Rottier, and R. J. de Groot. 2002. Discontinuous and non-discontinuous subgenomic RNA transcription in a nidovirus. EMBO J. 21:6571-6580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.von Grotthuss, M., L. S. Wyrwicz, and L. Rychlewski. 2003. mRNA cap-1 methyltransferase in the SARS genome. Cell 113:701-702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wieringa, R., A. A. F. de Vries, J. van der Meulen, G. J. Godeke, J. J. M. Onderwater, H. van Tol, H. K. Koerten, A. M. Mommaas, E. J. Snijder, and P. J. M. Rottier. 2004. Structural protein requirements in equine arteritis virus assembly. J. Virol. 78:13019-13027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yan, L., M. Velikanov, P. Flook, W. J. Zheng, S. Szalma, and S. Kahn. 2003. Assessment of putative protein targets derived from the SARS genome. FEBS Lett. 554:257-263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zevenhoven-Dobbe, J. C., S. Greve, H. van Tol, W. J. M. Spaan, and E. J. Snijder. 2004. Rescue of disabled infectious single-cycle (DISC) equine arteritis virus by using complementing cell lines that express minor structural glycoproteins. J. Gen. Virol. 85:3709-3714. [DOI] [PubMed] [Google Scholar]
- 39.Ziebuhr, J. 2004. Molecular biology of severe acute respiratory syndrome coronavirus. Curr. Opin. Microbiol. 7:412-419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ziebuhr, J., E. J. Snijder, and A. E. Gorbalenya. 2000. Virus-encoded proteinases and proteolytic processing in the Nidovirales. J. Gen. Virol. 81:853-879. [DOI] [PubMed] [Google Scholar]