Abstract
The matrix protein (M1) of influenza virus plays an essential role in viral assembly and has a variety of functions, including association with influenza virus ribonucleoprotein (RNP). Our previous studies show that the association of M1 with viral RNA and nucleoprotein not only promotes formation of helical RNP but also is required for export of RNP from the nucleus during viral replication. The RNA-binding domains of M1 have been mapped to two independent regions: a zinc finger motif at amino acid positions 148 to 162 and a series of basic amino acids (RKLKR) at amino acid positions 101 to 105, which is also involved in RNP-binding activity. To further understand the role of the RNP-binding domain of M1 in viral assembly and replication, mutations in the coding sequences of RKLKR and the zinc finger motif of M1 were constructed using a PCR technique and introduced into wild-type influenza virus by reverse genetics. Altering the zinc finger motif of M1 only slightly affected viral growth. Substitution of Arg with Ser at position 101 or 105 of RKLKR did not have a major impact on nuclear export of RNP or viral replication. In contrast, deletion of RKLKR or substitution of Lys with Asn at position 102 or 104 of RKLKR resulted in a lethal mutation. These results indicate that the RKLKR domain of M1 protein plays an important role in viral replication.
The genome of influenza A virus consists of eight distinct segments of negative-sense RNA coding for at least 10 viral proteins, of which 3 are known to function as polymerases. The viral RNA, nucleoprotein (NP), and polymerases are closely associated in the ribonucleoprotein (RNP) (11, 18, 22). Matrix protein (M1) is located between the RNP and the inner surface of the lipid envelope in the intact virion (1, 3, 33). Two major external glycoproteins, hemagglutinin (HA) and neuraminidase (NA), and a small protein, M2, that serves as a transmembrane channel are anchored in the viral envelope (20, 36).
M1 is not only an essential structural component of the virion but also participates in other steps during the replication of influenza virus. During early viral infection, dissociation of M1 from RNP is required for entry of viral RNP into the cytoplasm of the host cell (4, 12, 21). Dissociation is triggered by transport of H+ ions across the viral membrane by M2 (12, 19, 36). Subsequently, newly synthesized M1 migrates to the nucleus of the influenza virus-infected cell, where it associates with newly synthesized RNP (4, 25). It has been shown also that M1 is transported from the cytoplasm into the nucleus during early viral replication (27). Later in the replication cycle, the accumulation of M1 in the cytoplasm is concomitant with the nuclear export of RNP (4, 5, 13, 16, 35). Binding of RNP by M1 is required for the transport of M1-RNP complexes from the nucleus to the cytoplasm (15, 21) and prevents RNP from reentering the nucleus (21). In the cytoplasm, the interaction of M1 with HA, NA, M2, and lipid membranes also indicates a role for M1 in the budding of virions from the cell surface (3, 10, 9, 18, 20, 28, 30, 32, 42). In the maturation of viral particles, the M1/NP ratio of viral particles influences the virion's morphological features and the infectivity of the released viruses (29).
The interactions of M1 with RNP have been studied extensively (2, 6, 26, 31, 33, 41). Two domains in M1 have been shown to affect its association with RNA (41, 42). One RNA-binding domain, containing a zinc finger motif (148C-C----H-H162), has been shown to associate with zinc ions (7) and to inhibit viral replication (23). The other domain, residing in a palindromic stretch of basic amino acids (101-RKLKR-105), has been shown to bind viral RNA (8, 37, 42), fulfilling a prediction based on X-ray crystallographic studies (34). This domain also serves as a nuclear localization signal (NLS) for M1 (40, 43), but its role in viral replication is less certain.
Although it has been reported that under acidic conditions, M1 dissociates from RNP, resulting in a reduction in transport of RNP from the nucleus to the cytosol (4), the biological significance of the binding of M1 to RNP has not been studied extensively. Our recent studies demonstrate that viral RNP is not assembled in the absence of M1 (15). In the present work, we studied the impact of the RNA- and RNP-binding domains of M1 on viral assembly and replication by introducing mutations into the RNA- and RNP-binding domains of the influenza virus M gene.
Generation of M1 mutants corresponding to RNP- and RNA-binding domains.
To determine whether the RKLKR sequence and the zinc finger motif of M1 affect viral replication, the nucleotide sequences of these two elements in M1 were altered and introduced into plasmid expressing viral RNA (pPol I-WSN-M, a gift from Y. Kawaoka, University of Wisconsin—Madison) by site-directed mutagenesis. Figure 1A is a schematic diagram of the RNA- and RNP-binding domains of M1. The RKLKR sequence is located between amino acids 101 and 105, and the zinc finger motif is located between amino acids 148 and 162, of M1 (40, 41, 42). Figure 1B shows that plasmid Wt expressed the wild-type (wt) M gene; plasmid Del-101-RKLKR-105 contained an altered M gene expressing M1 with a deletion of RKLKR; plasmid M101-SNLNS-105 expressed an M protein with amino acids RKLKR replaced with SNLNS; plasmids M-R101S, M-K102N, M-K104N, and M-R105S each contained an altered M gene expressing a single amino acid substitution of Arg with Ser or of Lys with Asn; and plasmid M-C148S contained a DNA sequence coding for an alteration predicted to disrupt the zinc finger motif (7). The wt and altered M genes were also subcloned into the pCR3.1 vector (Invitrogen, Carlsbad, Calif.), which contains a T7 promoter that can express its downstream protein in vitro. The mutated M1's were expressed in vitro by using a coupled reticulocyte lysate system (Promega, Madison, Wis.) and were analyzed by autoradiographic densitometry after sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Analysis of the proteins indicated that the migration of the mutated M1's was comparable to that of WSN Wt M1 (Fig. 1C).
FIG. 1.
(A) Schematic model of an RNP- and RNA-binding domain (RKLKR) at amino acids 101 to 105 and a zinc finger RNA-binding domain at amino acids 148 to 162 of the WSN M1 protein. (B) Amino acid sequences of WSN Wt and mutant M1 constructs. (C) SDS-PAGE and autoradiography of in vitro-translated and 35S-labeled M1 proteins from cDNAs coding for WSN wt and mutant M1 proteins.
Transfectant influenza viruses carrying the M gene mutation were generated by reverse-genetics techniques described by Hoffmann et al. (14) and Neumann et al. (24), with minor modifications. Briefly, 1 day before transfection, confluent 293T cells were subcultured into 12-well plates with a dilution of approximately 1:10 in Opti-MEM I medium (Invitrogen, Gaithersburg, Md.). For transfection, 1 μl of TransIT LT-1 (Panvera, Madison, Wis.) per 0.5 μg of pHW181-PB2, pHW182-PB1, pHW183-PA, pHW184-HA, pHW185-NP, pHW186-NA, or pHW188-NS DNA (gifts from R. Webster, St. Jude Children's Research Hospital, Memphis, Tenn.) or pPol I-WSN-M expressing the relevant individual M1 mutation was used per well of cell culture. Because M1 is a major structural protein, mutation of M1 might impair viral particle assembly. pCAGGS-WSN-M (a gift from Y. Kawaoka), coding for wt M1 protein, was also included in the transfection system, allowing formation of complete virions expressing the mutant M1 proteins. The DNA-TransIT LT-1 mixture was incubated at room temperature for 45 min and added to the cells. Six hours later, the DNA-transfection mixture was replaced with 0.5 ml of Opti-MEM I. Twenty-four hours after transfection, 0.5 ml of Opti-MEM I containing 1 μg of tosylsulfonyl phenylalanyl chloromethyl ketone (TPCK)-trypsin per ml was added to the cells. At different time points posttransfection, samples were harvested. The titer of the virus was determined by HA units (HAU) or PFU assays. The virus particles generated by reverse-genetics were purified three times by plaque formation in MDCK cells and amplified in the allantoic cavities of 9-day-old embryonated eggs. The altered M gene in the virus was confirmed by sequencing analysis (data not shown).
Deletion of RKLKR or substitution of Lys with Asn in RKLKR was a lethal mutation.
To determine the effect of deletion or substitution of RKLKR in M1 on viral replication, mutated viral particles were recovered from the supernatants of 293T cells 48 h after transfection with the seven-plasmid pol I-pol II system (carrying the PB2, PB1, PA, NP, HA, NA, and NS genes) plus plasmids expressing wt M1 protein and the relevant individual M1 mutation. Virus titers in the supernatants, 48 h posttransfection, were analyzed by measurement of HAU and PFU in MDCK cells (Table 1). Approximately 16 HAU/ml and 6 × 106 PFU/ml were rescued from the transfection mixture containing the wt M gene. Deletion of RKLKR or substitution of RKLKR with SNLNS resulted in undetectable HA and undetectable infectious particles by plaque assay. A single amino acid substitution at position 102 (K102N) or position 104 (K104N) was also a lethal mutation for the M gene; there was no detectable infectious virus in the supernatants of transfected 293T cells by plaque assay (Table 1). Prolonging the incubation period to 72 h after transfection did not result in recovery of infectious virus particles of the M1 mutants (data not shown). Transfection of lethal M mutants was also repeated by using mixed 293T and MDCK cells and incubating up to 7 days. There was no recoverable virus for the K102N or K104N mutation (data not shown). Although the Del101-105, 101SNLNS105, K102N, and K104N mutants failed to generate replicable virus particles, the phenotypic wt viral particle could be generated by expression of wt M1 protein in our transfection system. The yield of replication-incompetent particles (RIPs) from 293T cells was measured by counting NP- or M1-positive cells. At 48 h postinfection, the titer of infectious RIPs was approximately 103/ml for Del101-105, 101SNLNS105, K102N, or K104N. RIPs were also confirmed by negative HAU after passage of the supernatants of transfected 293T cells in MDCK cells or in eggs (data not shown).
TABLE 1.
Virus titers in the supernatants of 293T cells after plasmid transfectiona
| Transfectant virus | Amino acid | HAU/ml | PFU/ml |
|---|---|---|---|
| wt | 101-RKLKR-105 | 16 | 6 × 106 |
| Del101-105 | DelRKLKR | NDb | ND |
| 101SNLNS105 | SNLNS | ND | ND |
| R101S | SKLKR | 4 | 1 × 104 |
| K102N | RNLKR | ND | ND |
| K104N | RKLNR | ND | ND |
| R105S | RKLKS | 4 | 4 × 104 |
| C148S | 148-S-C—H-H-162 | 8 | 2 × 106 |
293T cells were transfected with a modified reverse-genetics system to rescue M gene mutants, as described in Materials and Methods. The infectious particles in the supernatants of 293T cells were harvested 48 h posttranfection and titrated in MDCK cells.
ND, none detectable.
M1 proteins with mutations in amino acid position 102 or 104 remained in the cytoplasm, and RNPs of the same mutants remained in the nucleus.
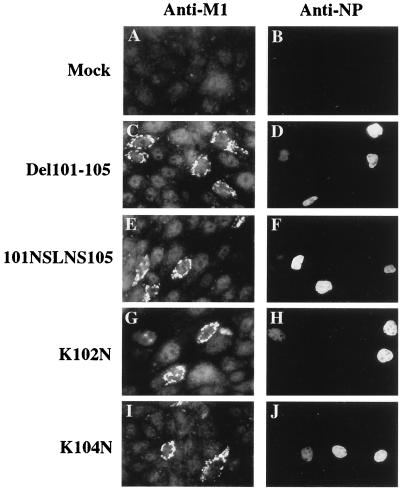
Replication-incompetent influenza viruses in the supernatants of 293T cells, transfected with plasmids expressing the Del101-105, 101SNLNS105, K102N, or K104N M1 mutant, were collected 48 h after transfection and transferred to MDCK cells. The cellular distributions of M1 and RNP in these cells were visualized by indirect-immunofluorescence staining with monoclonal antibodies to M1 and NP proteins. Briefly, cultures of MDCK cells grown on glass coverslips were infected with influenza virus at a multiplicity of infection (MOI) of 1 PFU per cell and were subsequently incubated in minimal essential medium (MEM) containing 2% fetal bovine serum at 33°C for 16 h. Infected MDCK cells were fixed with freshly prepared 4% formaldehyde in phosphate-buffered saline (PBS) for 20 min at room temperature. Background staining was blocked with 3% powdered skim milk in PBS for 1 h. Cells were then incubated at room temperature for 40 min with a monoclonal antibody to either M1 or to NP. The cells were incubated with donkey anti-mouse immunoglobulin G (IgG) conjugated with fluorescein and incubated for 40 min at room temperature. Washed coverslips were mounted in 90% glycerol and 10% PBS in 3,4,5-trihydroxybenzoic acid N-propylester to prevent photobleaching. The cellular distribution of immunofluorescence was visualized using an epifluorescent UV microscope. As shown in Fig. 2, newly synthesized M1 proteins were located in the cytoplasm of the cells infected with the Del101-105, 101SNLNS105, K102N, or K104N mutant. In contrast, newly synthesized NPs of these RIP-infected cells were retained in the nucleus, and no NP was detectable in the cytoplasm by indirect-immunofluorescence staining.
FIG. 2.
Immunofluorescence staining of MDCK cells infected with RIPs from the supernatants of transfected 293T cells. MDCK cells were infected for 16 h with defective M gene mutant particles in the supernatants of plasmid-transfected 293T cells. The cells were incubated with a mouse anti-M monoclonal antibody (A, C, E, G, and I) or a mouse anti-NP monoclonal antibody (B, D, F, H, and J) and with donkey anti-mouse IgG conjugated with fluorescein. The cellular distribution of immunofluorescence was determined with a transmission microscope under an epifluorescent UV light source. Magnification, ×472.
Mutation at amino acid position 101 or 105 of RKLKR or in the zinc finger motif did not affect RNP nuclear export.
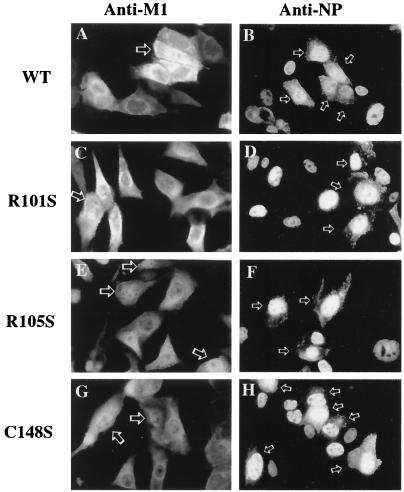
Because none of the R101S, R105S, and C148S substitutions resulted in a lethal mutation of influenza virus (Table 1), the cellular distributions of M1 and RNP in the resulting viruses were studied by infection of MDCK cells with mutant viruses at 0.01 PFU/cell followed by indirect-immunofluorescence staining of NP and M1 proteins 8 h postinfection. As shown in Fig. 3, wt M1 resides mostly in the cytoplasm of infected cells (Fig. 3A). Figure 3A shows the nuclear localization of the M1 protein in the early replication stage of the virus, in which the majority of M1 was in the nucleus of the infected cell. The cytoplasmic distribution of wt RNP was also identified by immunofluorescence staining with an antibody to NP (Fig. 3B). Figure 3B demonstrates that at the early stage of viral infection of MDCK cells, RNP was predominantly located in the nucleus, with little or no cytoplasmic staining. Similar cellular distributions of NP and M1 proteins of mutants with substitutions in the RKLKR domain (R101S and R105S) are shown in Fig. 3C to F. However, the amounts of NP in the cytoplasm of cells infected with mutants were slightly reduced (Fig. 3D and F). The cellular distributions of M1 and NP in cells infected by the substitution mutant virus C148S (Fig. 3G and H) were basically the same as those in cells infected with wt virus (Fig. 3A and B). These results indicate that substitution of the Arg residues at positions 101 and 105 of RKLKR did not have a major impact on nuclear export of RNPs and that mutation of the zinc finger motif did not affect nuclear export of NP at all.
FIG. 3.
MDCK cells were infected for 8 h with viable M gene mutant viruses in the undiluted supernatant produced from plasmid-transfected 293T cells. The cells were incubated with a mouse anti-M monoclonal antibody (A, C, E, and G) or a mouse anti-NP monoclonal antibody (B, D, F, and H) and with donkey anti-mouse IgG conjugated with fluorescein. Cellular distribution of immunofluorescence was determined with a transmission microscope under an epifluorescent UV light source. Arrows in panels A, C, E, and G indicate the localization of M1; arrows in panels B, D, F, and H indicate that of RNP. Magnification, ×472.
Growth properties of M1 mutant viruses in cell culture under restricted conditions.
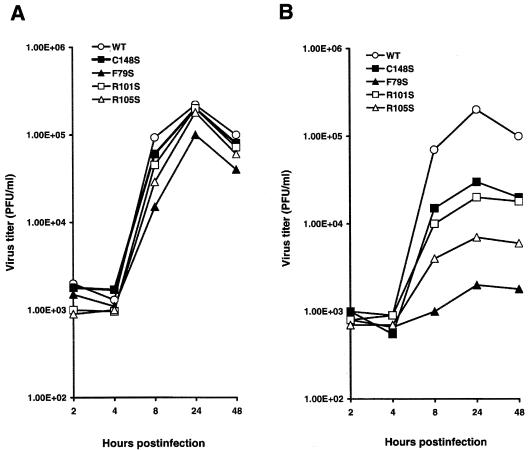
Since neither substitution of Arg at position 101 or 105 of RKLKR nor mutation of the zinc finger domain had a major impact on nuclear export of RNPs, the viral growth characteristics of the R101S, R105S, and C148S mutants were studied by comparison of growth rates under restricted conditions. As a temperature-sensitive mutant control, a temperature-sensitive mutant virus was generated by substituting amino acid Phe with Ser at position 79 (F79S) of M1 of the wt virus. Substitution of amino acid Phe with Ser at position 79 has been identified as the only mutation in the M gene of the ts51 strain, a naturally occurring temperature-sensitive mutant with a cutoff temperature of 39.5°C (38). A one-step growth experiment was performed by infection of MDCK cells with transfectants at an MOI of 5 PFU/cell at 33 or 39.5°C. Figure 4A shows viral growth at 33°C assayed by plaque titration of samples harvested at 2, 4, 8, 24, and 48 h postinfection. The data demonstrate that the M gene mutants grow at a rate similar to that of the wt at 33°C, except for the F79S transfectant virus, which had a slightly lower titer at the same temperature. Infectious particles were first detectable at 8 h postinfection, the peak of the growth was at 24 h postinfection, and virus titers of all mutants, including wt virus, declined 24 h postinfection. Comparison of growth of wt with mutant viruses at 39.5°C is shown in Fig. 4B. The F79S mutant had the slowest growth among the M gene mutants. The R105S mutant had a titer higher than that of the F79S mutant but lower than those of the rest of the mutants. The C148S and R101S mutants had moderate growth rates, between those of the wt and R105S viruses. The slower growth of mutants at 39.5°C suggests that replication of the R105S, R101S, and C148S M1 mutants may be temperature labile at 39.5°C. Virus yields were also studied by measurement of HAU; growth rates were similar to the growth rate measured by PFU, except that the virus titers reached a plateau 48 h postinfection (data not shown).
FIG. 4.
Growth curves of the mutant viruses in MDCK cells. MDCK cells were infected with plaque-purified transfectants (WT, C148S, F79S, R101S, and R105S) at an MOI of 5 PFU/cell at 33°C (A) or 39°C (B) in a 12-well culture (22-mm-diameter) plate. The supernatants of infected MDCK cells were harvested at 2, 4, 8, 24, and 48 h postinfection. Infectious virus particles in the supernatants were determined by plaque assay on MDCK cells at 33°C. The standard deviation of three independent experimental points was below 10% (data not shown).
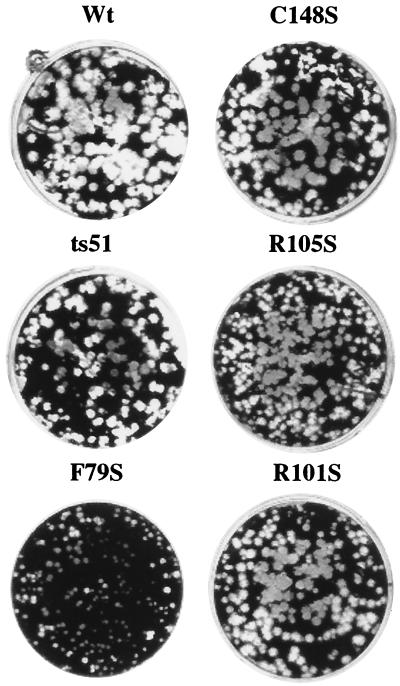
The plaque sizes of M1 mutant viruses are correlated with their growth rates.
Because mutated M gene transfectant viruses had different growth rates in MDCK cells, we determined the plaque formation of these viruses in MDCK cells. To study the plaque morphology of the mutant viruses, MDCK cells in 6-well-plates (diameter, 35 mm) were infected with serial 10-fold dilutions of plaque-purified transfectant viruses. The medium in the dish was replaced with 0.75% agarose in Eagle's MEM (E-MEM) containing 2 μg of trypsin/ml. Plaques were formed by incubation for 3 days at 33°C. Cells were fixed with 100% methanol and stained with 1% crystal violent. Figure 5 shows the plaque formation of transfectant viruses in MDCK cells at 33°C. Transfectant wt and C148S viruses, which had higher growth rates, formed large plaques (2.6 and 2.4 mm in diameter, respectively). In contrast, the F79S transfectant virus, which had a lower growth rate, formed small plaques (0.7 mm). The R105S and R101S mutants formed intermediate-sized plaques (1.8 and 2.0 mm, respectively). However, the ts51 virus, which had the same M gene mutation as the F79S virus, also formed large plaques (diameter, 2.3 mm). The plaque formation of the WSN and ts51 viruses has been studied extensively by Yasuda et al. (39). Their observations indicate that the ts51 virus, with a single mutation at amino acid 79 (from F to S) in M1, forms large plaques. However, substitution of Phe with Ser at amino acid 79 in M1 of the transfectant virus in our study showed that this substitution resulted in small plaques and a somewhat attenuated phenotype compared with those of naturally occurring ts51. At this point, we do not have a satisfactory explanation for the difference between the transfectant F79S virus and the naturally occurring ts51 mutant virus. The difference may be due to the gene constellation effect between transfectant virus and ts51.
FIG. 5.
Plaque morphology of the mutant viruses. MDCK cells were infected with the mutant viruses in 6-well plates at 33°C in the presence of 2 μg of trypsin/ml. After 2 days of incubation, the cells were fixed and stained with crystal violet.
Our data demonstrate that the RNA- and RNP-binding domains of M1 are involved in viral replication. Although the zinc finger domain has inhibitory activity against viral replication when a peptide containing this domain is used in cell culture (23), mutation of the zinc finger domain in the M gene did not have a major impact on viral replication based on our results. Deletion of RKLKR, substitution of RKLKR with SNLNS, and substitution of K's with N′s in RKLKR were all lethal mutations. However, substitution of Arg with Ser in RKLKR in M1 had a minimal effect on viral replication. The different consequences of mutation at Arg versus Lys in the RKLKR sequence on viral replication may be due to the fact that mutation of Lys in RKLKR not only impairs RNP-binding activity but also disrupts the core consensus sequence of the NLS, K(R/K)X(R/K) (17). Table 2 summarizes the comparative sequences of the NLS (amino acids 101 to 105) for influenza A viruses. Unlike the typical NLS, K(R/K)X(R/K), the first Arg of RKLKR in M1 is less conservative; it can be Arg (R), Lys (K), Gln (Q), Thr (T), or Gly (G). The rest of the amino acid sequences are strictly conserved. The consensus sequence of the NLS of M1's in influenza A viruses, such as H5N1, H3N2, and H1N1 viruses, is XKLKR. However, from our results, substitution of Arg at amino acid position 105 had a minimal effect on viral replication, indicating that Arg at position 105 is also less important.
TABLE 2.
Summary of comparative sequence data for NLSs of influenza A virus M1's
| Virus | Amino acidsa | GenBank accession no. |
|---|---|---|
| A/Swine/Iowa/15/30(H1N1) | GKLKR | M33045 |
| A/Hong Kong/498/97(H3N2) | TKLKR | AF255370 |
| A/aquatic bird/Hong Kong/399/99(H3N8) | RKLKR | AJ427299 |
| A/Hong Kong/491/97(H5N1) | QKLKR | AH010698 |
| A/Hong Kong/488/97(H5N1) | KKLKR | AF255377 |
| A/Hong Kong/1073/99(H9N2) | KKLKR | AJ278646 |
M1 protein sequences are from GenBank.
Our previous research (41) indicated that the RKLKR sequence is involved not only in the RNA and RNP association but also in the nuclear translocation of M1, whereas the zinc finger motif is involved only in RNA binding (summarized in Table 3). Our present studies indicate that the RNA-binding activity of the zinc finger motif may not play an important role in viral replication. The lack of nuclear localization of M1 in RIP-infected cells (Del101-105, 101NSLNS105, K102N, and K104N mutants) and the lack of cytoplasmic translocation of RNP might result from disruption of the NLS alone or of the NLS and RNP-binding activities of RKLKR. The lack of cytoplasmic translocation of RNP might result from reductions in levels of the NS2 protein, another nuclear export protein, recruited by M1. On the other hand, nonlethal M1 mutations (R101S and R105S) may still result in M1 proteins with relatively functional NLS and RNP-binding activities. The reduced viral replication of nonlethal M gene mutants at 39.5°C, compared with that of wt virus at the same temperature, may be due to the inefficiency of nuclear localization and the lack of RNP binding of mutated M1's. Reductions in viral titers, however, may not result from decreasing amounts of M proteins in infected cells at 39°C. As determined by immunofluorescence staining of MDCK cells infected with the R101S, R105S, or C148S transfectant influenza virus, the expression level of the mutated M1 protein in infected cells was not significantly lower at 39.5°C than at 33°C (data not shown). Nuclear localization and RNP-binding activities need to be studied in order to confirm whether a single mutation of RKLKR such as R101S or R105S will result in reduction of nuclear localization of M1 and/or reduction of RNP-binding activities of M1.
TABLE 3.
RNA-binding, RNP-binding, and nuclear localization activities of M1
| Protein or peptide | Functional domain
|
RNA-binding activitya | RNP-binding activityb | Nuclear localizationc | |
|---|---|---|---|---|---|
| RKLKR | Zinc finger | ||||
| wt | Yes | Yes | + | + | + |
| C148S | Yes | No | ± | + | + |
| 101SNLNS105 | No | Yes | ± | ± | − |
32P-labeled RNA bound to M1. +, ≥80%; ±, 50 to 70%; −, ≤20%.
RNP association quantified by comparison to the total input 35S-labeled M1 proteins at pH 7.0 and 0.15 M NaCl.
Immunofluorescence staining of nuclear RNP using an anti-NP monoclonal antibody.
M1 protein plays an important role in particle assembly and viral replication. Both the dissociation of M1 from RNP in the early phase of infection and the association of M1 and RNP in the late phase of the infection are required for sufficient viral replication. It is believed that the association of M1with RNP leads to translocation of RNP from the nucleus to the cytoplasm. The basic amino acid sequence (RKLKR) of M1 is important for the function of M1 protein in viral replication by translocation of M1 from the cytoplasm into the nucleus and for interaction with RNP. Our recent studies (15) have demonstrated that viral RNA and M1 together promote the self-assembly of influenza virus NP into the quaternary helical structure similar to the typical viral RNP. In addition to acting as an essential component to promote the transport of RNP out of the nucleus, the RNP binding of M1 has a structural role in final viral assembly. Further studies of specific M1 functional domains may help us to complete the understanding of the role of this viral protein in influenza virus virulence and pathogenesis in a variety of host systems.
Acknowledgments
We thank Ronald Lundquist, Barry Falgout, and Gary Kikuchi for critically reading and improving the manuscript and Michael Klutch of CBER, FDA, for DNA sequencing. We are especially grateful to Roland Levandowski for valuable discussions.
REFERENCES
- 1.Allen, H., J. McCauley, M. Waterfield, and M. J. Gething. 1980. Influenza virus RNA segment 7 has the coding for two polypeptides. Virology 107:548-551. [DOI] [PubMed] [Google Scholar]
- 2.Baudin, F., I. Petit, W. Weissenhorn, and R. W. Ruigrok. 2001. In vitro dissection of the membrane and RNP binding activities of influenza virus M1 protein. Virology 281:102-108. [DOI] [PubMed] [Google Scholar]
- 3.Bucher, D. J., I. G. Kharitonenkov, J. A. Zakomirdin, V. B. Grigoriev, S. M. Klimenko, and J. F. Davis. 1980. Incorporation of influenza virus M-protein into liposomes. J. Virol. 36:586-590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bui, M., G. Whittaker, and A. Helenius. 1996. Effect of M1 protein and low pH on nuclear transport of influenza virus ribonucleproteins. J. Virol. 70:8391-8401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bui, M., E. G. Wills, A. Helenius, and G. R. Whittaker. 2000. Role of the influenza virus M1 protein in nuclear export of viral ribonucleoproteins. J. Virol. 74:1781-1786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Compans, R. W., and H. D. Klenk. 1979. Viral membranes, p. 293-407. In H. Fraenkel-Conrat and R. R. Wagner (ed.), Comprehensive virology, vol. 13. Plenum Publishing Corp., New York, N.Y.
- 7.Elster, C., E. Fourest, F. Baudin, K. Larsen, S. Cusack, and R. W. Ruigrok. 1994. A small percentage of influenza virus M1 protein contains zinc but zinc does not influence in vitro M1-RNA interaction. J. Gen. Virol. 75:37-42. [DOI] [PubMed] [Google Scholar]
- 8.Elster, C., K. Larsen, J. Gagnon, R. W. H. Ruigrok, and F. Baudin. 1997. Influenza virus M1 protein binds to RNA through its nuclear localization signal. J. Gen. Virol. 78:1589-1596. [DOI] [PubMed] [Google Scholar]
- 9.Enami, M., and K. Enami. 1996. Influenza virus hemagglutinin and neuraminidase glycoproteins stimulate the membrane association of the matrix protein. J. Virol. 70:6653-6657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gregoriades, A. 1980. Interaction of influenza M protein with viral lipid and phosphatidylcholine vesicles. J. Virol. 36:470-479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Heggeness, M. H., P. R. Smith, L. Ulmanen, R. M. Krug, and P. W. Choppin. 1982. Studies on the helical nucleocapsid of influenza virus. Virology 118:466-470. [DOI] [PubMed] [Google Scholar]
- 12.Helenius, A. 1992. Unpacking of the incoming influenza virus. Cell 69:577-578. [DOI] [PubMed] [Google Scholar]
- 13.Herz, C., E. Stavnezer, and R. M. Krug. 1981. Influenza virus, an RNA virus, synthesizes its messenger RNA in the nucleus of influenza cells. Cell 26:391-400. [DOI] [PubMed] [Google Scholar]
- 14.Hoffmann, E., G. Neumann, Y. Kawaoka, G. Hobom, and R. G. Webster. 2000. A DNA transfection system for generation of influenza A virus from eight plasmids. Proc. Natl. Acad. Sci. USA 97:6108-6113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Huang, X., T. Liu, J. Muller, R. A. Levandowski, and Z. Ye. 2001. Effect of influenza virus matrix protein and viral RNA on ribonucleoprotein formation and nuclear export. Virology 287:405-416. [DOI] [PubMed] [Google Scholar]
- 16.Krug, R. M., R. V. Alonso-Coplen, L. Julkumen, and M. G. Katze. 1989. Expression and replication of the influenza virus genome, p. 89-162. In R. M. Krug (ed.). The influenza viruses. Plenum Press, New York, N.Y.
- 17.LaCasse, E. C., and Y. A. Lefebvre. 1995. Nuclear localization signals overlap DNA- or RNA-binding domains in nucleic acid-binding proteins. Nucleic Acids Res. 23:1647-1656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lamb, R. A., and P. W. Choppin. 1983. The structure and replication of influenza virus. Annu. Rev. Biochem. 52:467-506. [DOI] [PubMed] [Google Scholar]
- 19.Lamb, R. A., L. J. Holsinger, and L. H. Pinto. 1994. The influenza A virus M2 ion channel protein and its role in the influenza virus life cycle, p. 303-321. In E. Wimmer (ed.), Receptor-mediated virus entry into cells. Cold Spring Harbor Press, Cold Spring Harbor, N.Y.
- 20.Lamb, R. A., and S. L. Zebedee. 1985. Influenza virus M2 protein is an integral membrane protein expressed on the infected-cell surface. Cell 40:627-633. [DOI] [PubMed] [Google Scholar]
- 21.Martin, K., and A. Helenius. 1991. Nuclear transport of influenza virus ribonucleoproteins: the viral matrix protein (M1) promotes export and inhibits import. Cell 67:117-130. [DOI] [PubMed] [Google Scholar]
- 22.Murti, K. G., R. G. Webster, and I. M. Jones. 1988. Localization of RNP polymerases on influenza viral ribonucleoprotein by immunogold labeling. Virology 164:562-566. [DOI] [PubMed] [Google Scholar]
- 23.Nasser, E. H., A. K. Judd, A. Sanchez, D. Anastasiou, and D. J. Bucher. 1996. Antiviral activity of influenza virus M1 zinc finger peptides. J. Virol. 70:8639-8644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Neumann, G., T. Watanabe, H. Ito, S. Watanabe, H. Goto, P. Gao, M. Hughes, D. R. Perez, R. Donis, E. Hoffmann, G. Hobom, and Y. Kawaoka. 1999. Generation of influenza A viruses entirely from cloned cDNAs. Proc. Natl. Acad. Sci. USA 96:9345-9350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Patterson, S., J. Gross, and J. S. Oxford. 1988. The intercellular distribution of influenza virus matrix protein and nucleoprotein in infected cells and their relationship to haemagglutinin in the plasma membrane. J. Gen. Virol. 69:1859-1872. [DOI] [PubMed] [Google Scholar]
- 26.Rees, P. J., and N. J. Dimmock. 1982. Kinetics of synthesis of influenza virus ribonucleoprotein structures. J. Gen. Virol. 59:403-408. [DOI] [PubMed] [Google Scholar]
- 27.Rey, O., and D. P. Nayak. 1992. Nuclear retention of M1 protein in a temperature-sensitive mutant of influenza (A/WSN/33) virus does not affect nuclear export of viral ribonucleoproteins. J. Virol. 66:5815-5824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Roberts, P. C., and R. W. Compans. 1998. Host cell dependence of viral morphology. Proc. Natl. Acad. Sci. USA 95:5746-5751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Roberts, P. C., R. A. Lamb, and R. W. Compans. 1998. The M1 and M2 proteins of influenza A virus are important determinants in filamentous particle formation. Virology 240:127-137. [DOI] [PubMed] [Google Scholar]
- 30.Robertson, B. H., J. C. Bennett, and R. W. Compans. 1982. Selective dansylation of M protein within intact influenza virions. J. Virol. 44:871-876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ruigrok, R. W. H., and F. Baudin. 1995. Structure of influenza virus ribonucleoprotein particles. II. Purified RNA-free influenza virus ribonucleoprotein forms structures that are indistinguishable from the intact influenza virus ribonucleoprotein particles. J. Gen. Virol. 76:1009-1014. [DOI] [PubMed] [Google Scholar]
- 32.Ruigrok, R. W. H., P. C. J. Krijgsman, F. M. de Roude-Verloop, and J. C. de Jong. 1985. Natural heterogenicity of shape, infectivity and protein composition in an influenza A (H3N2) virus preparation. Virus Res. 3:69-76. [DOI] [PubMed] [Google Scholar]
- 33.Schulze, I. T. 1972. The structure of influenza virus. II. A model based on the morphology and composition of subviral particles. Virology 47:181-196. [DOI] [PubMed] [Google Scholar]
- 34.Sha, B., and M. Luo. 1997. Structure of a bifunctional membrane-RNA binding protein, influenza virus matrix protein M1. Nat. Struct. Biol. 4:239-244. [DOI] [PubMed] [Google Scholar]
- 35.Silver, P. A. 1991. How proteins enter the nucleus. Cell 64:489-497. [DOI] [PubMed] [Google Scholar]
- 36.Sugrue, R. J., and A. J. Hay. 1991. Structural characteristics of the M2 protein of the influenza A viruses: evidence that it forms a tetrameric channel. Virology 180:617-624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wakefield, L., and G. G. Brownlee. 1989. RNA-binding properties of influenza A virus matrix protein M1. Nucleic Acids Res. 17:8569-8680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Whittaker, G., I. Kemler, and A. Helenius. 1995. Hyperphosphorylation of mutant influenza virus matrix protein, M1, causes its retention in the nucleus. J. Virol. 69:439-445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yasuda, J., D. J. Bucher, and A. Ishihama. 1994. Growth control of influenza A virus by M1 protein: analysis of transfectant viruses carrying the chimeric M gene. Virology 68:8141-8146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ye, Z., N. W. Baylor, and R. R. Wagner. 1989. Transcription-inhibition and RNA-binding domains of influenza virus matrix protein mapped with anti-idiotype antibodies and synthetic peptides. J. Virol. 63:3586-3594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ye, Z., T. Liu, D. P. Offringa, J. McInnis, and R. A. Levandowski,. 1999. Association of influenza virus matrix protein with ribonucleoproteins. J. Virol. 73:7467-7473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ye, Z., R. Pal, J. W. Fox, and R. R. Wagner. 1987. Functional and antigenic domains of the matrix (M1) protein of influenza virus. J. Virol. 61:239-246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ye, Z., D. Robinson, and R. R. Wagner. 1995. Nucleus-targeting domain of the matrix protein (M1) of influenza virus. J. Virol. 69:1964-1970. [DOI] [PMC free article] [PubMed] [Google Scholar]