Abstract
Presently marketed vaginal barrier methods are cytotoxic and damaging to the vaginal epithelium and natural vaginal flora when used frequently. Novel noncytotoxic agents are needed to protect men and women from sexually transmitted diseases. One novel candidate is a mandelic acid condensation polymer, designated SAMMA. The spectrum and mechanism of antiviral activity were explored using clinical isolates and laboratory-adapted strains of human immunodeficiency virus (HIV) and herpes simplex virus (HSV). SAMMA is highly effective against all CCR5 and CXCR4 isolates of HIV in primary human macrophages and peripheral blood mononuclear cells. SAMMA also inhibits infection of cervical epithelial cells by HSV. Moreover, it exhibits little or no cytotoxicity and has an excellent selectivity index. SAMMA, although not a sulfonated or sulfated polymer, blocks the binding of HIV and HSV to cells by targeting the envelope glycoproteins gp120 and gB-2, respectively, and also inhibits HSV entry postattachment. SAMMA is an excellent, structurally novel candidate microbicide that warrants further preclinical evaluation.
There are now more than 40 million adults and 2.7 million children worldwide living with human immunodeficiency virus (HIV) infection, according to estimates from the Joint United Nations Program on HIV/AIDS (UNAIDS) for 2001 (38). Approximately 90% of these infections are acquired through sexual contact. Women are increasingly infected via heterosexual contact, which has emerged as the principal route of transmission worldwide (38). While physical barrier methods (condoms) remain the most effective protection against sexual transmission of HIV, this modality requires partner initiation and/or consent. Although renewed efforts in vaccine development have provided valuable insight, a protective vaccine may not be available in the near future. Thus, there is an urgent need for novel strategies to block HIV-1 transmission. Topical (vaginal or rectal) microbicides that block the establishment of infection at the mucosal surface provide a method of intervention that can be initiated by the individual at risk independent of the sexual partner.
The ideal microbicide should fulfill a number of criteria that include not only an acceptable antimicrobial and toxicity profile but also features that would lead to extensive use worldwide (21). The compound must have broad activity against transmission of primary HIV isolates, inhibit transmission to relevant cell types within the mucosa, be easy to use without side effects, be essentially odorless and colorless, and be inexpensive to allow worldwide distribution. Ideally, the compound should be active against other sexually transmitted pathogens, especially those causing ulcerative disease such as herpes simplex virus (HSV).
There are several reasons to also focus on genital herpes in the development of topical microbicides. Epidemiological studies have shown that genital ulcer disease, which is caused primarily by HSV-2 (9), enhances the transmission and acquisition of HIV infection (7). HSV may potentiate HIV acquisition by disrupting or activating epithelial cells, which produce proinflammatory cytokines, and may activate or recruit HIV target cells. Additionally, heat-inactivated HSV particles enhance HIV replication in macrophages in vitro, by activating cellular pathways involving NF-κB (24). HSV-infected epithelial cells may release noninfectious viral particles, which, in turn, may enhance HIV replication within macrophages or peripheral blood mononuclear cells (PBMC) (36). Thus, topical microbicides that block both HSV and HIV transmission offer distinct advantages.
The earliest approach to microbicide development focused on spermicidal detergents. The surfactant nonoxynol-9 (N-9), the most commonly used spermicide worldwide, was initially thought to be a potentially safe microbicide. However, N-9 was highly cytotoxic in primary human cervical or vaginal epithelial cells (16). The results of a recently completed randomized, placebo-controlled study are consistent with these in vitro observations: women who used N-9 gel became infected with HIV at a 50% higher rate than did women who used a placebo gel, and had significantly higher incidence of inflammatory lesions than did the placebo group (5). One possibility is that the break in the critical epithelial barrier contributed to the higher transmission rate. This experience highlights the need for novel candidate topical microbicides and for a rigorous preclinical evaluation of candidate agents.
Recently, a novel compound, a polymer derived from sulfuric acid treatment of mandelic acid (mandelic acid condensation product, SAMMA), has been identified as a candidate topical contraceptive (developed by the Topical Prevention for Conception and Disease, Rush University, Chicago, Ill.) (40). Mandelic acid has been used effectively as a urinary antiseptic, particularly as a bladder irrigant during urological procedures (19, 32). In preliminary work, SAMMA exhibited an excellent safety profile and was active against HIV, HSV, bovine papillomavirus, and Neisseria gonorrhoeae (40). Importantly, SAMMA has no deleterious effects on lactobacilli (40). Its simple chemical structure, solubility in water, apparent stability, and characteristics of being colorless and odorless make it attractive as a candidate microbicide. Moreover, unlike other compounds being developed as topical microbicides, it does not contain sulfur, i.e., is not sulfated or sulfonated, and is not a surfactant. These studies were undertaken, therefore, to examine the spectrum and mechanisms of antiviral activity of this unique compound against HIV and HSV by using cells most likely to be early targets during sexual transmission.
MATERIALS AND METHODS
Primary and permanent cell cultures.
CaSki (human cervical cell line) and 293 cells were obtained from the American Type Culture Collection (ATCC: Manassas, Va.) and maintained as previously described (17). Immortalized human epithelial cells derived from normal human endocervix were obtained from R. N. Fichorova and D. J. Anderson and maintained as previously described (12). Primary macrophage cultures were prepared from Ficoll-Hypaque-purified PBMC and purified by adherence after 14 days in culture in RPMI containing 20% fetal calf serum. They were cultured in 96-well plates at a concentration of 1.5 × 105 (200 μl/well). Phytohemagglutinin-activated (5 μg/ml) CD4+ T cells were cultured in 96-well plates at a concentration of 105 cells/ml (200 μl/well) in RPMI supplemented with interleukin-2 (50 U/ml). The astrocytoma cell line U87MG, as well as derivatives expressing CD4 (U87CD4) or CD4 and CCR5 or CXCR4 coreceptors (U87CD4CCR5 and U87CD4CXCR4, respectively), was obtained from the AIDS Research Reference Reagent Program (ARRRP), National Institute of Allergy and Infectious Diseases, (NIAID), and grown as previously described (10).
Viral strains and glycoproteins.
Laboratory-adapted HIV-1 strains included the R5 laboratory isolate HIV-1BAL and the X4 laboratory isolate HIV-1IIIB (Advanced Biotechnologies Inc, Columbia, Md.). Primary isolates included the R5 isolate BR/92/003 and the X4 isolate UG021 (ARRRP and UNAIDS Network for HIV Isolation and Characterization and the Division of AIDS, NIAID). For the single-cycle experiments, replication-defective virus was used. Pseudotyped recombinant viruses were produced in 293T cells by using a three-plasmid cotransfection system described previously (26). The packaging construct pCMVΔR8.2 was previously described (26). The HIV-1 reporter construct pNL4-3.Luc.R-E (obtained from ARRRP) contains the firefly luciferase gene inserted into the pNL4-3 nef gene. Two frameshift mutations (in 5′ Env and Vpr at amino acid 26) render this clone replication defective. Pseudotyped replication-defective viruses were generated by cotransfection of the packaging and reporter constructs with either of two envelope-expressing constructs, pJR-FL and pHXB2, expressing an R5 and X4 envelope, respectively (donated by D. Littman, Skirball Institute, New York University, New York, N.Y.) (14). Transfections were performed in 10-cm2 tissue culture dishes using Lipofectamine 2000 (Invitrogen, Carlsbad, Calif.). Viral supernatants were harvested 48 h posttransfection and treated with 29 U of DNase I (Invitrogen) per ml for 1 h at 37°C to remove any residual plasmid DNA.
The HSV-2 strains used were the laboratory wild-type isolates 333 and G. The clinical isolates MMA and MTWTA were obtained from patients attending a sexually transmitted disease clinic (gift from P. Spear, Northwestern University) and have been partially sequenced (37). DT-1 and DT-2 are acyclovir-sensitive and -resistant strains, respectively, isolated from a neonate (28). Adenovirus type 5 (Ad5) was obtained from ATCC.
Reagents.
Recombinant envelope glycoprotein 120 (gp120) from an X4 (MN) and R5 (YU2) virus, as well as oligomeric IIIB gp120, was obtained from Immunodiagnostics Inc., Woburn, Mass. HSV-2 recombinant glycoprotein B (gB-2) was generated using the Bac-to-Bac system (Gibco) and purified by heparin-affinity chromatography. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis and silver staining confirmed purity. Details of its synthesis will be provided elsewhere (N. Cheshenko and B. C. Herold, unpublished data).
Heparin, N-9, and zidovudine (AZT) were purchased from Sigma (St. Louis, Mo.). Acyclovir was obtained from Glaxo-Wellcome, Research Triangle Park, N.C. TAK-779 was obtained from the ARRRP. The SAMMA polymer was prepared by condensation of methyl mandelate in concentrated H2SO4 and precipitated with ethanol. The product was converted to the disodium salt by treatment with aqueous NaOH. Elemental analyses, nuclear magnetic resonance spectroscopy, high-performance liquid chromatography NMR and HPLC identified the product as 2,2′-diphenyl-2,2′-oxy-diacetic acid with a molecular weight of 256 (330 as the disodium salt) (40).
HIV in vitro assays. (i) Infectivity assays.
Macrophages and phytohemagglutinin-stimulated CD4+ T cells were preincubated with either SAMMA (concentration, 0.1 to 1,000 μg/ml) or control medium for 1 h at 37°C and then subjected to a 2-h infection with virus at 37°C. Laboratory strains were used at a multiplicity of infection (MOI) of 0.1 to 0.25, and primary isolates were used at a final concentration of 34 to 78 ng of p24/ml. Following infection, the cells were washed twice with phosphate-buffered saline (PBS) to remove unbound virus and compound and incubated in fresh media. Viral replication was monitored using a p24 (Gag) protein enzyme-linked immunosorbent assay (ELISA) (Du-Pont, Wilmington, Del).
(ii) Time course assays.
In the single-cycle infection time course assays, U87 cells were cultured in 96-well plates at a concentration of 105 cells/ml (200 μl/well) and allowed to adhere and grow until confluent. They were preincubated with compound or control medium for 1 h at 37°C (t = −1) and then subjected to a 2-h infection with the replication-defective virus. The cells were washed twice with PBS to remove unbound virus.
Alternatively, compound was added immediately, 6, 12, or 24 h after the wash (e.g., t = 2, 8, 14, or 26 h postinfection [p.i.], respectively). The reverse transcriptase inhibitor AZT and the CCR5-specific inhibitor TAK-779, which prevents fusion of HIV with the host cell, were used as added controls (1, 11). The cells were lysed in luciferase lysis buffer after 48 h, and a luciferase assay was performed using a luciferase assay system (Promega, Madison, Wis.). Values were standardized with respect to total protein concentration.
(iii) Preincubation of virus or cells with SAMMA.
To examine whether SAMMA interacts primarily and/or irreversibly with HIV, cells, or both, the compound was preincubated with concentrated HIV-1IIIB for 1 h at 37°C and the mixture was diluted 50-fold so that the final concentration of the virus was an MOI of 0.1. For comparison, diluted virus (MOI ≈ 0.1) was preincubated for 1 h with various concentrations of SAMMA and the mixture was plated (without diluting) on U87 CD4+ CXCR4+ cells. Alternatively, SAMMA (or Dulbecco minimal essential medium [DMEM] as a control) was preincubated with the cells for 1 h at 37°C and then the cells were either washed extensively or not prior to inoculation with HIV-1IIIB at an MOI of 0.1.
(iv) Soluble-gp120 assays.
To more specifically examine whether SAMMA blocks the binding of gp120 to cells, U87 MG, U87 CD4+, U87 CD4+ CCR5+, and U87 CD4+ CXCR4+ were grown to approximately 75% confluency, trypsinized, washed once in DMEM, and resuspended at a concentration of 2 × 106/ml. Cell aliquots of 1 ml each were preincubated for 15 min with SAMMA (10 μg/ml to 1 mg/ml) or DMEM as control. Then 50 ng of either MN (X4) or YU2 (R5) gp120 (ImmunoDiagnostics, Inc., Woburn, Mass.) was added, and the mixture was incubated at 37°C with constant rotation. The cells were washed with chilled PBS, and the pellets were resuspended in 80 μl of distilled H2O and further incubated with 20 μl of 5× sodium dodecyl sulfate sample buffer. The lysate was boiled for 10 min, and an aliquot was analyzed on an 8% Tris-glycine gel and then subjected to Western transfer probing for gp120 using a 1:1,000 dilution of the 1169 polyclonal antiserum (a gift from R. Doms, University of Pennsylvania, Philadelphia, Pa.) and colorimetric detection using an alkaline phosphatase-conjugated anti-rabbit immunoglobulin G and Western Blue stabilized substrate (Promega).
HSV in vitro assays. (i) Infectivity assays.
Plaque assays were conducted to evaluate the effect of SAMMA on HSV or Ad5 infection (13, 16). Serial dilutions of SAMMA were mixed with virus immediately before inoculation of cell monolayers. HSV plaques were counted after 48 h by a blue-plaque immunoassay (17). Adenovirus plaques on 293 cells were counted after 14 days by visualization (13).
(ii) Time course assays.
Time course plaque assays were conducted to determine which steps in viral infection (attachment, entry, or postentry) were inhibited by SAMMA (15). Heparin, a competitive inhibitor of viral binding, and acyclovir, an inhibitor of viral DNA polymerase activity, were included for comparison. Confluent monolayers of CaSki cells were precooled to 4°C and inoculated with ∼200 to 1,000 PFU of virus/well at 4°C for 3 h. Unbound virus was removed by washing the cells three times, and the cells were then shifted to 37°C to allow penetration for 1 h. Unpenetrated virus was inactivated by washing the cell monolayer with a low-pH citrate buffer. The cells were then overlaid with medium. The drugs were added during the 4°C binding period, at the time the cells were transferred to 37°C, or 4 h after citrate treatment for 1 h, followed by a wash, to determine if the compounds act at the level of viral binding, entry, or postentry, respectively. In addition, to determine if the drug could remove bound virus after the 4°C adsorption period, cells were washed three times (5 min, 5 min, and 1 h) with various dilutions of drug and then the plates were shifted to 37°C.
(iii) Preincubation of virus or target cells with SAMMA.
To examine whether SAMMA interacts primarily and/or irreversibly with HSV, epithelial cells, or both, SAMMA was preincubated with ∼104 PFU of HSV-2 (G) per ml for 1 h at 37°C and the mixture was diluted 50-fold to yield ∼200 PFU/well on control plates and inoculated onto monolayers of CaSki cells in duplicate on six-well dishes. For comparison, diluted virus (∼200 PFU) was preincubated for 1 h with various concentrations of SAMMA and the mixture was plated (without dilution) on cells. Alternatively, SAMMA (or DMEM as a control) was preincubated with epithelial cells for 1 h at 37°C and the cells were either washed extensively or not prior to inoculation with HSV-2 (G).
(iv) Soluble-glycoprotein assay.
To more specifically examine whether SAMMA targets HSV-2 gB-2, CaSki cells were exposed to various concentrations of recombinant glycoprotein in the absence or presence of SAMMA (or heparin for comparison) for 1 h at 37°C. The cells were washed, cell lysates were prepared, and the bound gB-2 was quantified by optical density scanning of Western blots using monoclonal antibody 1123(Goodwin Institute, Plantation, Fla.).
Cytotoxicity assays.
The cytotoxic effect of SAMMA (dose ranging from 0.1 to 10 mg/ml) on cervical epithelial cells, CD4+ T cells, or macrophages was determined by quantitating cell viability using an MTS assay (CellTiter96; Promega). Cytotoxicity was examined following a 24-h exposure to drug (acute) or a 1-h daily exposure for seven consecutive days (chronic).
Statistical analysis.
The data presented are means with standard deviation. Student's unpaired, two-tailed t tests were performed as indicated in the figure legends.
RESULTS
Spectrum of anti-HIV and anti-HSV activity.
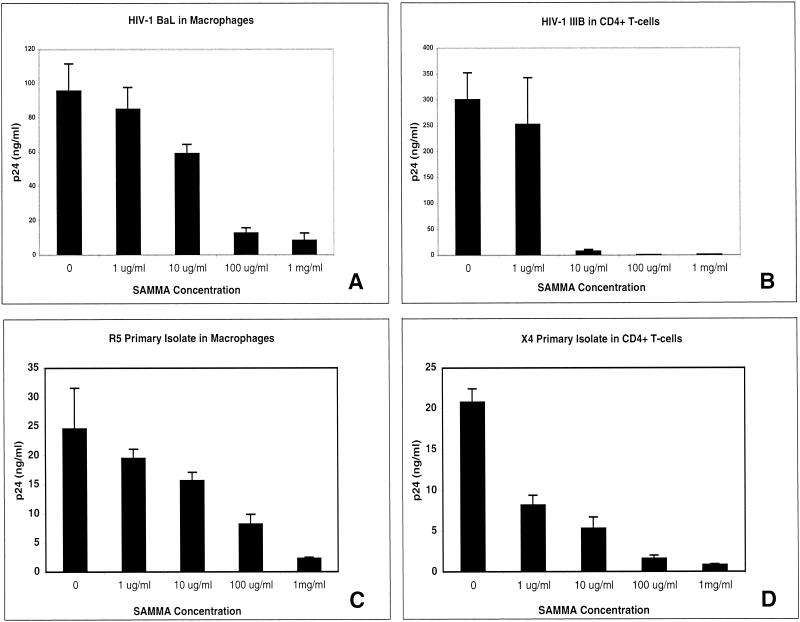
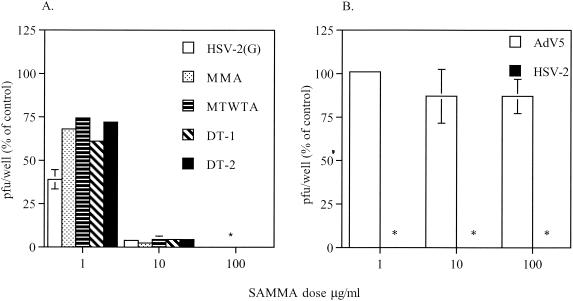
SAMMA was active against laboratory (BaL) and primary R5 isolates of HIV-1 in primary macrophages (Fig. 1A and C, respectively) as well as laboratory (IIIB) and primary X4 isolates in CD4+ T cells (Fig. 1B and D, respectively). SAMMA also inhibited infection of CD4+ T cells by HIV BaL (data not shown). The activity against X4 isolates in CD4+ T cells tended to be higher than against R5 isolates in either CD4+ T cells or macrophages, but both X4 and R5 viruses were significantly inhibited in all experiments by SAMMA at a concentration of 100 μg/ml. SAMMA also inhibited laboratory [HSV-2(G)] and clinical (MMA, MTWTA, and DT-1) isolates of HSV-2 on human cervical epithelial cells (CaSki), as well as an acyclovir-resistant strain (DT2) (Fig. 2A). Similar results were obtained with laboratory strains of HSV-1 and primary or immortalized human endocervical cells (data not shown). In contrast, SAMMA had no inhibitory activity against Ad5 infection of 293 cells but inhibited HSV-2 on these cells, even when present at concentrations as low as 1 μg/ml (Fig. 2B).
FIG. 1.
Effects of SAMMA on HIV infection of macrophages and PBMC. Primary macrophages were infected with the R5 laboratory isolate BaL (A) or the R5 clinical isolate BR/92/003 (C) in the presence of the indicated concentrations of SAMMA. Viral replication was monitored by determining supernatant HIV-1 p24 (Gag) protein by ELISA on day 7. (C and D) Primary CD4+ T cells were infected with the X4 laboratory isolate IIIB (B) or the X4 clinical isolate UG021 (D) in the presence or absence of SAMMA, and p24 was quantified on day 5. Results represent the average p24 concentrations from triplicate wells (error bars indicate standard deviation) and are representative of at least two independent experiments.
FIG. 2.
Effects of SAMMA on HSV or Ad5 infection. (A) Duplicate wells of CaSki cells were exposed to a laboratory isolate (G), clinical isolates (MMA, MTWTA, or DT-1), or an acyclovir-resistant isolate (DT-2) of HSV-2 in the absence or presence of 1, 10, or 100 μg of SAMMA per ml. (B) For comparison, 293 cells were exposed to HSV-2(G) or Ad5 in the absence or presence of SAMMA. Results are presented as PFU formed in the presence of drug as a percentage of PFU formed in the absence of drug. Asterisks indicate that no HSV plaques were observed by immunoassay. Results are means of three independent experiments conducted in duplicate, and error bars indicate standard deviations.
Cytotoxicity of SAMMA.
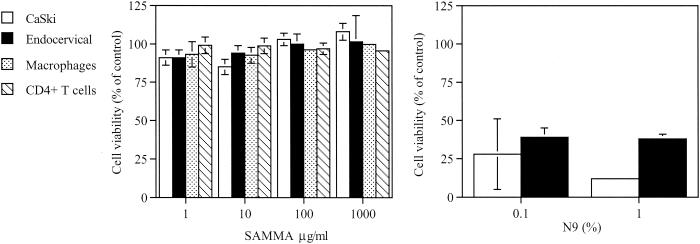
SAMMA had little or no effect on cell viability following a 24-h exposure in cervical epithelial cells (CaSki or immortalized endocervical cells), primary macrophages, or primary human CD4+ lymphocytes (Fig. 3A). Moreover, no toxicity was observed in the setting of repeated exposure of primary macrophages and lymphocytes to the drug (1 h/day for 7 days) (results not shown). The calculated selectivity index of SAMMA toward HIV (R5 and X4) viruses and HSV infection in primary human cell cultures is greater than 1,000. In contrast, N-9 is highly toxic for cervical cells (Fig. 3B) and, as previously reported, has a calculated SI of <0.3 toward HSV-2 (16).
FIG. 3.
Cytotoxicity was quantified using an MTS assay following a 24-h exposure of CaSki, immortalized human endocervical, primary macrophage, or CD4+ T cells to the indicated concentrations of SAMMA (left) or N-9 (right). Results are presented as the number of viable cells as a percentage of control cells incubated in the medium. Each value is the mean of at least two independent experiments conducted in triplicate; error bars indicate standard deviation.
Mechanism of anti-HIV activity.
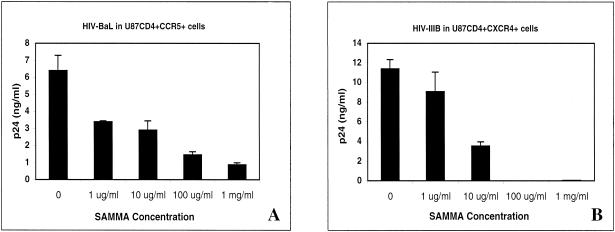
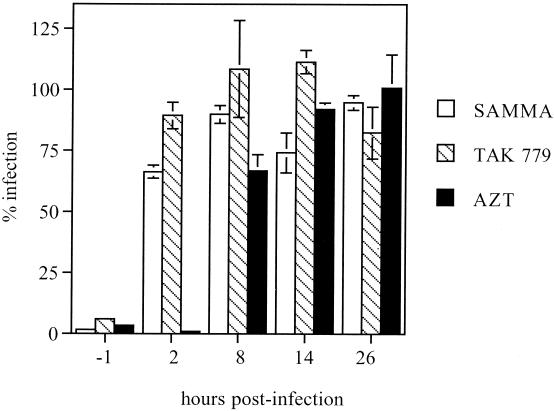
Because SAMMA inhibits HIV infection by both R5 and X4 viruses and exhibits no cytotoxicity in culture, we explored the mechanism of antiviral activity of this novel compound. SAMMA inhibits infection of U87 cells expressing CD4 as well as the appropriate coreceptor (Fig. 4). To determine the part of the virus life cycle inhibited by SAMMA, single-cycle infection experiments with U87 CD4+ CCR5+ cells and the JR-FL (R5) pseudotyped virus were performed while varying the time of introduction of SAMMA or the control inhibitors, TAK-799 and AZT (Fig. 5). SAMMA was active only if added before and present at the time of infection (t = −1), similar to the coreceptor inhibitor TAK-799. If it was added after viral entry (2 h p.i.), there was complete loss of inhibitory activity, consistent with the compound working at the point of attachment and/or entry. In contrast, the reverse transcriptase inhibitor AZT retains activity when added 2 h p.i., but its activity is lost following the completion of reverse transcription 8 h p.i. None of the compounds appeared to work at steps following reverse transcription (14 and 26 h). Similar results were obtaining using U87 CD4+ CXCR4+ cells and the HXB2 (X4) pseudotyped virus.
FIG. 4.
Anti-HIV activity of SAMMA in U87 CD4+ CCR5+ cells infected with the R5 virus virus HIV-BaL (A) and in U87 CD4+ CXCR4+ cells infected with the X4 isolate IIIB (B). U87 cells were preincubated with the indicated SAMMA concentrations or control medium for 1 h at 37°C and then subjected to a 2-h infection with virus. After 2 h, the cells were washed twice with PBS to remove unbound virus and compound. Viral replication was monitored by measuring p24 protein in culture supernatants by ELISA 48 h postinfection. Results are the mean p24 values from triplicate wells and are representative of at least two independent experiments; error bars indicate standard deviation.
FIG. 5.
Anti-HIV activity of SAMMA, AZT, or TAK-779 added at different times to U87 CD4+ CCR5+ cells infected with the R5 replication-defective virus JR-FL. Cells were preincubated with compound or control medium for 1 h at 37°C (t = −1) and then subjected to a 2-h infection with the replication-defective virus. The cells were washed twice with PBS to remove unbound virus and overlaid with fresh medium. Alternatively, the compounds were added immediately (2 h) or 8, 14, and 26 h after initiation of infection. The cells were lysed in luciferase lysis buffer after 48 h. Luciferase values were standardized with respect to total protein concentration. Results are the mean p24 values from triplicate wells and are representative of two independent experiments; error bars indicate standard deviation.
Pretreatment of U87 cells expressing CD4 and either CCR5 or CXCR4 coreceptor with SAMMA followed by washout prior to infection with IIIB resulted in loss of activity at SAMMA concentrations of 10 and 100 μg/ml (Table 1). In contrast, compound activity was enhanced if HIV particles were pretreated with SAMMA for 1 h before being plated, even if this was followed by a 50-fold dilution of the virus-drug mixture prior to infection (Table 1). Together, these data suggest that SAMMA targets primarily the virus, not the cell, and blocks attachment and/or entry.
TABLE 1.
Viral infection following preincubation of cells or virus for 1 h with SAMMA or control media
| SAMMA concin (μg/ml) | Viral activity after following treatmenta
|
|||
|---|---|---|---|---|
| Pretreated cells (no wash) | Pretreated cells (wash) | Pretreated virus (no dilution) | Pretreated virus (50-fold dilution) (Final [SAMMA] after dilution) | |
| HIV-1IIIB on U87 CD4+ CXCR4+ cells | ||||
| 1 | 99 ± 19 | 113 ± 7 | *11 ± 11 | 86 ± 13 (0.02) |
| 10 | *48 ± 8 | 99 ± 9 | *0 | *44 ± 5 (0.2) |
| 100 | *11 ± 1.4 | 91 ± 10 | *0 | *3 ± 2 (2) |
| HSV-2(G) on CaSki cells | ||||
| 1 | 95 ± 1.4 | 71 ± 1.4 | 44 ± 31 | 128 ± 2 (0.02) |
| 10 | *5 ± 0 | 78 ± 8.5 | *0 | *60 ± 6 (0.2) |
| 100 | *0 | 71 ± 1.4 | *0 | *0 (2) |
Results are presented as p24 (HIV) or PFU formed (HSV) in the presence of SAMMA as a percentage of p24 or PFU in the absence of drug and are the mean ± standard deviation of at least two independent experiments conducted in triplicate (HIV) or duplicate (HSV). For each experimental condition, the effects of SAMMA were compared to those of control medium by using the Student unpaired t test. Asterisks indicate a significant reduction in viral infection (P ≤ 0.001).
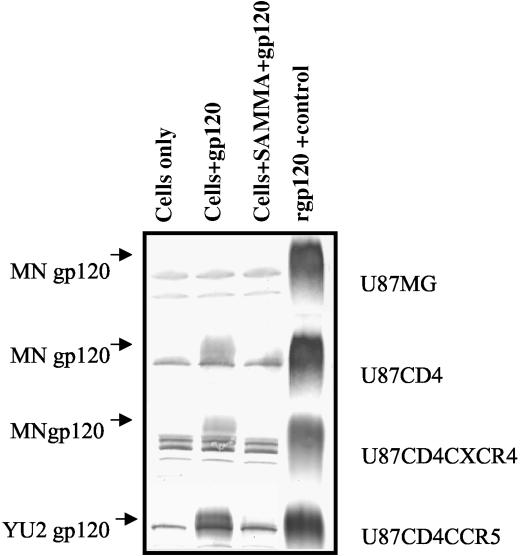
To further test this notion, we examined the effects of SAMMA on the binding of purified recombinant envelope gp120 to U87 cells. As expected, no recombinant gp120 binds to U87 MG cells but the protein binds to U87 cells expressing CD4 in both the absence and presence of coreceptors. Preincubation of either X4 or R5 gp120 with SAMMA results in complete inhibition of binding to U87 CD4+ cells in the presence and absence of the appropriate coreceptor. Representative gels are shown in Fig. 6. Together, these results suggest that SAMMA interacts directly with gp120 and prevents binding to CD4.
FIG. 6.
Effects of SAMMA on HIV-1 gp120-cell membrane interactions. U87MG control, CD4+, CD4+CXCR4+, or CD4+CCR5+ cells were preincubated with medium or 500 μg of SAMMA per ml for 15 min. Then 50 ng of MN gp120 (MG, CD4+, and CD4+CXC4+ cells) or YU2 gp120 (CD4+CCR5+ cells) was added, and the mixture was incubated at 37°C with constant rotation for 1 h and washed extensively. The cell lysates were analyzed on an 8% Tris-glycine gel and then subjected to Western transfer and probing for gp120 using a 1:1,000 dilution of the 1169 polyclonal antiserum. Colorimetric detection was performed using an alkaline phosphatase-conjugated anti-rabbit IgG and Western Blue stabilized substrate. Controls include cells alone (lane 1) and recombinant gp120 alone (lane 4). Data are representative of three independent experiments.
Mechanism of anti-HSV activity.
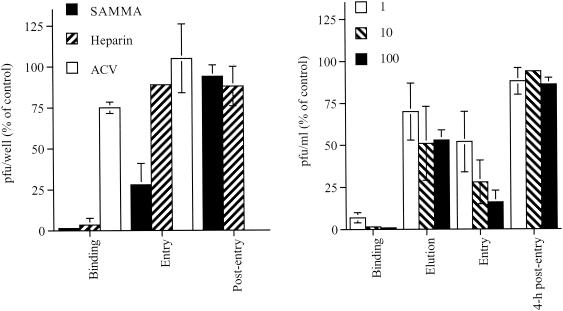
To examine which steps in HSV-2 infection are inhibited by SAMMA, parallel types of experiments were conducted by varying the time of introduction of SAMMA or the control inhibitors, heparin and acyclovir. In addition, by varying the temperature during the experiment, HSV attachment, which occurs at 4°C, can be differentiated from viral penetration, which occurs following the shift to 37°C. The results for a single concentration of each drug are shown in Fig. 7A, and a dose-response curve for SAMMA is shown in Fig. 7B. SAMMA was as effective as heparin and inhibited HSV binding to cells at concentrations <1 μg/ml. Notably, SAMMA retained antiviral activity when added at the time when cells were transferred to 37°C but completely lost activity if added 4 h postentry (1-h exposure). Notably, if SAMMA was added 4 h postentry and maintained in culture medium for the 48-h incubation period, both the number and size of infectious plaques were reduced, presumably because the compound blocks cell-to-cell spread (data not shown) Heparin, in contrast, had little or no effect postbinding, whereas acyclovir was active only if present postentry.
FIG. 7.
Mechanism of anti-HSV activity of SAMMA compared with heparin and ACV. Time course experiments were conducted as described in Materials and Methods. (A) Results when each of the drugs (SAMMA [10 μg/ml], heparin [10 μg/ml], or acyclovir [10 μg/ml]) was added either during the 4°C binding period, at the time of the temperature shift, or 4 h postentry. (B) Dose response to SAMMA added during the 4°C binding period (binding), during the postbinding washes to remove bound virus (elution), at the time of temperature shift (entry), and 4 h postentry (cells were exposed to compounds for 1 h for each of the postbinding treatments and then washed and overlaid with fresh medium). Results are presented as PFU formed in the presence of drug as a percentage of PFU formed in the presence of medium and are means of three independent experiments performed in duplicate; error bars indicate standard deviation.
Some of the antiviral effects postbinding could be explained if SAMMA removes bound virus. This was assessed by washing the cells three times (5 min, 5 min, and 1 h) at 4°C with the drug after the binding period but before shifting the plates to 37°C. At the highest concentration tested, 100 μg/ml, washing with SAMMA resulted in only a ∼50% reduction in viral infection, suggesting that only part of the postbinding effect is explained by elution of bound virus (Fig. 7B). This suggests that SAMMA may directly block entry. Consistent with this notion is the observation that addition of SAMMA to culture plates at the time of the temperature shift (during entry) prevents the expression of viral immediate-early gene products (data not shown).
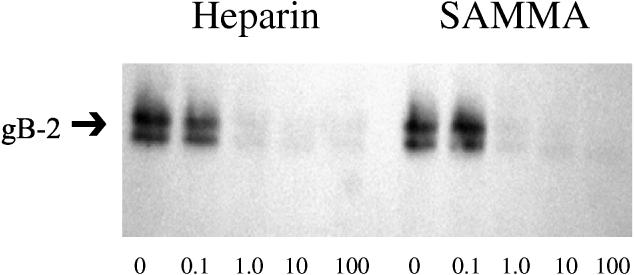
To further assess the effects of SAMMA on HSV binding and to assess whether the drug targets gB-2, we examined the effects of SAMMA on the binding of recombinant gB-2 to cells. gB-2 plays the key role in mediating the binding of HSV-2 to cells and is also required for viral entry (6). SAMMA inhibited the binding of recombinant gB-2 (Fig. 8).
FIG. 8.
Effects of SAMMA on binding of recombinant gB-2 to CaSki cells. Cells were exposed to recombinant gB-2 (10−5 μg/cell for 1 h at 37°C) in the presence or absence of the indicated concentrations of SAMMA or heparin. Bound gB-2 was detected by analyzing Western blots of cell lysates. Results are representative of three independent experiments.
Pretreatment of CaSki cells for 1 h with 10 μg of SAMMA per ml prior to infection essentially abolished HSV infection (Table 1). Consistent with the HIV results, the effects did not persist if the drug was washed out prior to infection (Table 1), suggesting that SAMMA does not irreversibly affect cells and must be present at the time of infection. SAMMA activity was enhanced if HSV particles were pretreated with SAMMA for 1 h, even if this was followed by dilution of the mixture 50-fold compared with adding SAMMA directly to cells prior to infection (Table 1). Together, these results suggest that SAMMA binds directly to the HSV envelope, preventing viral binding and entry.
DISCUSSION
SAMMA is a unique candidate topical microbicide that inhibits primary and laboratory-adapted strains of HIV and HSV at doses that are not toxic to primary cells. A number of other candidate microbicides are under development. These include detergents or surfactants that disrupt virion membranes, such as sodium dodecyl sulfate (18) and C31G (18); natural antimicrobial peptides, magainins, and protegrins (8); and sulfated or sulfonated polysaccharides (SPs) (2, 15, 17, 31). The SPs, including dextran sulfate, carageenan, polystyrene sulfonate, cellulose sulfate, and the naphthalene sulfonated polymer PRO 2000, have similar chemical features (sulfation or sulfonation) and anionic charge. Preliminary studies suggest that SPs act primarily by binding to the V3 loop of gp120 and also to a highly conserved binding region in gp120 involved in coreceptor recognition (25). Similarly, the SPs competitively inhibit the binding of HSV to cell surface heparan sulfate (HS), although the specific binding site for HSV envelope glycoproteins has not been determined (15).
SAMMA is unique because, unlike these other compounds, it has no surfactant properties and does not contain sulfur groups. However, the studies described here suggest that SAMMA may function at a similar point in the virus life cycle to the point at which SPs act, by binding to viral envelope glycoproteins during attachment and entry. The cell surface glycosaminoglycan HS forms a point of initial attachment for a number of organisms including HIV and HSV. HS is the primary attachment receptor for HSV (33, 39). Although HS is not a primary receptor, several studies have shown that HIV also binds to HS and that the quantity of HS on a cell predicts in vitro infectivity (23, 27, 29, 30). HIV infection is significantly reduced in some cell types if binding to HS is blocked either by enzymatic treatment of cells with heparin lyases or by soluble heparin or other competitive inhibitors (23, 27, 29, 30). Interactions between gp120 and HS may concentrate the virus at a cell surface and facilitate subsequent interactions with CD4 and coreceptors. Although SAMMA is not sulfated, it is negatively charged and thus may competitively block viral glycoprotein-HS interactions. This may account for the antiviral activity of SAMMA against both HIV and HSV. Notably, SAMMA showed no activity against Ad5, which binds primarily to CAR and enters via endocytic vesicles (3).
SAMMA inhibits both R5 and X4 HIV infection; however, activity in the lower dose range appears to be greater against X4 viruses. An even greater differential activity for R5 and X4 viruses has been observed with dextran sulfate and was attributed to the higher cationic charge associated with the X4 V3 loop (22). It is important to note that dextran sulfate inhibits X4 gp120 binding to coreceptors and not to CD4 (25) whereas SAMMA blocks binding to CD4. Importantly, SAMMA, at concentrations that are not cytotoxic, efficiently inhibits the entry of R5 viruses, the viral type that predominates early in infection.
SAMMA is also highly effective against HSV-2 and appears to target gB-2, inhibiting both attachment and entry. gB-2 plays the predominant role in mediating HSV-2 binding to cell surface HS but is also required (along with gD and the heterodimeric complex of gH and gL) for viral entry (6, 34). Although the domains of gB-2 required for HS binding have not been defined, a lysine-rich region at the amino acid terminus is presumed to mediate viral binding to HS (4, 35). This region (gB-2 residues 60 to 84) may be a target for SAMMA. Whether an interaction with gB-2 is also the mechanism responsible for the inhibition of HSV entry is not clear. Notably, heparin, which also competes with cell surface HS for gB-2 binding, has little or no antiviral activity if added after binding. Possibly SAMMA, but not heparin, interacts with additional domains of gB-2 or with the other glycoproteins required for viral entry. Alternatively, SAMMA may bind more avidly to gB-2 than does heparin.
SAMMA also exhibits an excellent safety profile in vitro. In addition to its activity against HIV and HSV described here, preliminary studies show that SAMMA is active against N. gonorrhoeae and Chlamydia trachomatis, although the mechanisms have not been explored (40). Importantly, SAMMA has no deleterious effects on lactobacilli. It is also a very effective inhibitor of sperm function and is contraceptive in the rabbit model (40).
The results obtained in these and other preliminary studies suggest that SAMMA warrants further investigation. First, SAMMA inhibits HIV infection of both primary CD4+ T cells and macrophages. Notably, it inhibits isolates that use either CCR5 or CXCR4 coreceptors and does so at concentrations that are readily achievable in a formulated compound. Second, SAMMA contains no sulfur and is not a surfactant or detergent; these features distinguish it from other microbicides in preclinical development. Third, SAMMA exhibits no cytotoxicity in tissue cultures using primary cervical cells, human macrophages, or PBMC. These results are in contrast with those obtained for N-9, sodium dodecyl sulfate, or C31G, which are toxic to primary vaginal cells (16, 20). Fourth, SAMMA is soluble in water and saline, probably inexpensive to manufacture, and likely to be easily formulated. In addition, it is essentially colorless and odorless. These characteristics, combined with its activity in culture against HIV, HSV, and other sexually transmitted disease pathogens, render it an optimal candidate ingredient for preclinical development.
Acknowledgments
This work was supported by Public Health Service grants AI37940 and HD41763.
We thank R. Fichorova and D. Anderson (Harvard Medical School, Boston, Mass.) for the generous gift of the immortalized human endocervical cells.
REFERENCES
- 1.Baba, M., O. Nishimura, N. Kanzaki, M. Okamoto, H. Sawada, Y. Iizawa, M. Shiraishi, Y. Aramaki, K. Okonogi, Y. Ogawa, K. Meguro, and M. Fujino. 1999. A small-molecule, nonpeptide CCR5 antagonist with highly potent and selective anti-HIV-1 activity Proc. Natl. Acad. Sci. USA 96:5698-5703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Baba, M., D. Schols, E. De Clercq, R. Pauwels, M. Nagy, J. Gyorgyi-Edelenyi, M. Low, and S. Gorog. 1990. Novel sulfated polymers as highly potent and selective inhibitors of human immunodeficiency virus replication and giant cell formation. Antimicrob. Agents Chemother. 34:134-138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bergelson, J. M., J. A. Cunningham, G. Droguett, E. A. Kurt-Jones, A. Krithivas, J. S. Hong, M. S. Horwitz, R. L. Crowell, and R. W. Finberg. 1997. Isolation of a common receptor for coxsackie B viruses and adenoviruses 2 and 5. Science 275:1320-1323. [DOI] [PubMed] [Google Scholar]
- 4.Bzik, D. J., C. Debroy, B. A. Fox, N. E. Pederson, and S. Person. 1986. The nucleotide sequence of the gB glycoprotein gene of HSV-2 and comparison with the corresponding gene of HSV-1. Virology 155:322-333. [DOI] [PubMed] [Google Scholar]
- 5.Centers for Disease Control and Prevention. 2000. CDC statement on study results of products containing nonoxynol-9. JAMA 284:1376. [DOI] [PubMed] [Google Scholar]
- 6.Cheshenko, N., and B. C. Herold. 2002. Glycoprotein B plays predominant role in mediating herpes simplex virus type 2 attachment and is required for entry and cell-cell spread. J. Gen. Virol. 83:2247-2252. [DOI] [PubMed] [Google Scholar]
- 7.Cohen, M. S. 1998. Sexually transmitted diseases enhance HIV transmission: no longer a hypothesis. Lancet 351:5-7. [DOI] [PubMed] [Google Scholar]
- 8.Cole, A. M., T. Hong, L. M. Boo, T. Nguyen, C. Zhao, G. Bristol, J. A. Zack, A. J. Waring, O. O. Yang, and R. I. Lehrer. 2002. Retrocyclin: a primate peptide that protects cells from infection by T- and M-tropic strains of HIV-1 Proc. Natl. Acad. Sci. USA 99:1813-1818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Corey, L., H. G. Adams, Z. A. Brown, and K. K. Holmes. 1983. Genital herpes simplex virus infections: clinical manifestations, course, and complications. Ann. Intern. Med. 98:958-972. [DOI] [PubMed] [Google Scholar]
- 10.Dragic, T., and M. Alizon. 1993. Different requirements for membrane fusion mediated by the envelopes of human immunodeficiency virus types 1 and 2. J. Virol. 67:2355-2359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dragic, T., A. Trkola, D. A. Thompson, E. G. Cormier, F. A. Kajumo, E. Maxwell, S. W. Lin, W. Ying, S. O. Smith, T. P. Sakmar, and J. P. Moore. 2000. A binding pocket for a small molecule inhibitor of HIV-1 entry within the transmembrane helices of CCR5. Proc. Natl. Acad. Sci. USA 97:5639-5644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fichorova, R. N., L. D. Tucker, and D. J. Anderson. 2001. The molecular basis of nonoxynol-9-induced vaginal inflammation and its possible relevance to human immunodeficiency virus-1 transmission. J. Infect. Dis. 184:418-428. [DOI] [PubMed] [Google Scholar]
- 13.Graham, F. L., and L. Prevec. 1995. Methods for construction of adenovirus vectors. Mol. Biotechnol. 3:207-220. [DOI] [PubMed] [Google Scholar]
- 14.He, J., S. Choe, R. Walker, P. Di Marzio, D. O. Morgan, and N. R. Landau. 1995. Human immunodeficiency virus type 1 viral protein R (Vpr) arrests cells in the G2 phase of the cell cycle by inhibiting p34cdc2 activity J. Virol. 69:6705-6711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Herold, B. C., N. Bourne, D. Marcellino, R. Kirkpatrick, D. M. Strauss, L. J. Zaneveld, D. P. Waller, R. A. Anderson, C. J. Chany, B. J. Barham, L. R. Stanberry, and M. D. Cooper. 2000. Poly(sodium 4-styrene sulfonate): an effective candidate topical antimicrobial for the prevention of sexually transmitted diseases. J. Infect. Dis. 181:770-773. [DOI] [PubMed] [Google Scholar]
- 16.Herold, B. C., R. Kirkpatrick, D. Marcellino, A. Travelstead, V. Pilipenko, H. Krasa, J. Bremer, L. J. Dong, and M. D. Cooper. 1999. Bile salts: natural detergents for the prevention of sexually transmitted diseases. Antimicrob. Agents Chemother. 43:745-751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Herold, B. C., A. Siston, J. Bremer, R. Kirkpatrick, G. Wilbanks, P. Fugedi, C. Peto, and M. Cooper. 1997. Sulfated carbohydrate compounds prevent microbial adherence by sexually transmitted disease pathogens Antimicrob. Agents Chemother. 41:2776-2780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Howett, M. K., E. B. Neely, N. D. Christensen, B. Wigdahl, F. C. Krebs, D. Malamud, S. D. Patrick, M. D. Pickel, P. A. Welsh, C. A. Reed, M. G. Ward, L. R. Budgeon, and J. W. Kreider. 1999. A broad-spectrum microbicide with virucidal activity against sexually transmitted viruses. Antimicrob. Agents Chemother. 43:314-321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.King, J. B., and D. J. Stickler. 1991. An assessment of antiseptic bladder washout solutions using a physical model of the catheterized bladder. J. Hosp. Infect. 18:179-190. [DOI] [PubMed] [Google Scholar]
- 20.Krebs, F. C., S. R. Miller, B. J. Catalone, P. A. Welsh, D. Malamud, M. K. Howett, and B. Wigdahl. 2000. Sodium dodecyl sulfate and C31G as microbicidal alternatives to nonoxynol 9: comparative sensitivity of primary human vaginal keratinocytes. Antimicrob. Agents Chemother. 44:1954-1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mauck, C., Z. Rosenberg, and L. Van Damme. 2001. Recommendations for the clinical development of topical microbicides: an update. AIDS 15:857-868. [DOI] [PubMed] [Google Scholar]
- 22.Meylan, P. R., R. S. Kornbluth, I. Zbinden, and D. D. Richman. 1994. Influence of host cell type and V3 loop of the surface glycoprotein on susceptibility of human immunodeficiency virus type 1 to polyanion compounds Antimicrob. Agents Chemother. 38:2910-2916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mondor, I., S. Ugolini, and Q. J. Sattentau. 1998. Human immunodeficiency virus type 1 attachment to HeLa CD4 cells is CD4 independent and gp120 dependent and requires cell surface heparans. J. Virol. 72:3623-3634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Moriuchi, M., H. Moriuchi, R. Williams, and S. E. Straus. 2000. Herpes simplex virus infection induces replication of human immunodeficiency virus type 1. Virology 278:534-540. [DOI] [PubMed] [Google Scholar]
- 25.Moulard, M., H. Lortat-Jacob, I. Mondor, G. Roca, R. Wyatt, J. Sodroski, L. Zhao, W. Olson, P. D. Kwong, and Q. J. Sattentau 2000. Selective interactions of polyanions with basic surfaces on human immunodeficiency virus type 1 gp120. J. Virol. 74:1948-1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Naldini, L., U. Blomer, P. Gallay, D. Ory, R. Mulligan, F. H. Gage, I. M. Verma, and D. Trono. 1996. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 272:263-267. [DOI] [PubMed] [Google Scholar]
- 27.Ohshiro, Y., T. Murakami, K. Matsuda, K. Nishioka, K. Yoshida, and N. Yamamoto. 1996. Role of cell surface glycosaminoglycans of human T cells in human immunodeficiency virus type-1 (HIV-1) infection. Microbiol. Immunol. 40:827-835. [DOI] [PubMed] [Google Scholar]
- 28.Oram, R. J., D. Marcellino, D. Strauss, E. Gustafson, C. L. Talarico, A. K. Root, P. L. Sharma, K. Thompson, J. D. Fingeroth, C. Crumpacker, and B. C. Herold. 2000. Characterization of an acyclovir-resistant herpes simplex virus type 2 strain isolated from a premature neonate. J. Infect. Dis. 181:1458-1461. [DOI] [PubMed] [Google Scholar]
- 29.Oravecz, T., M. Pall, J. Wang, G. Roderiquez, M. Ditto, and M. A. Norcross. 1997. Regulation of anti-HIV-1 activity of RANTES by heparan sulfate proteoglycans. J. Immunol. 159:4587-4592. [PubMed] [Google Scholar]
- 30.Patel, M., M. Yanagishita, G. Roderiquez, D. C. Bou-Habib, T. Oravecz, V. C. Hascall, and M. A. Norcross. 1993. Cell-surface heparan sulfate proteoglycan mediates HIV-1 infection of T-cell lines. AIDS Res. Hum. Retroviruses 9:167-174. [DOI] [PubMed] [Google Scholar]
- 31.Pearce-Pratt, R., and D. M. Phillips. 1996. Sulfated polysaccharides inhibit lymphocyte-to-epithelial transmission of human immunodeficiency virus-1. Biol. Reprod. 54:173-182. [DOI] [PubMed] [Google Scholar]
- 32.Robertson, M. H., and M. S. Norton. 1990. Effect of 1% mandelic acid as a bladder irrigation fluid in patients with in-dwelling catheters. Br. J. Clin. Pract. 44:142-144. [PubMed] [Google Scholar]
- 33.Shieh, M. T., D. WuDunn, R. I. Montgomery, J. D. Esko, and P. G. Spear. 1992. Cell surface receptors for herpes simplex virus are heparan sulfate proteoglycans. J. Cell Biol. 116:1273-1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Spear, P. 1993. Entry of alphaherpesviruses into cells. Semin. Virol. 4:167-180. [Google Scholar]
- 35.Stuve, L. L., S. Brown-Shimer, C. Pachl, R. Najarian, D. Dina, and R. L. Burke. 1987. Structure and expression of the herpes simplex virus type 2 glycoprotein gB gene. J. Virol. 61:326-335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Szilagyi, J. F., and C. Cunningham. 1991. Identification and characterization of a novel non-infectious herpes simplex virus-related particle. J. Gen. Virol. 72:661-668. [DOI] [PubMed] [Google Scholar]
- 37.Terhune, S. S., K. T. Coleman, R. Sekulovich, R. L. Burke, and P. G. Spear. 1998. Limited variability of glycoprotein gene sequences and neutralizing targets in herpes simplex virus type 2 isolates and stability on passage in cell culture. J. Infect. Dis. 178:8-15. [DOI] [PubMed] [Google Scholar]
- 38.UNAIDS. 2001. Report on the global HIV/AIDS epidemic—December 2001 update. United Nations, Geneva, Switzerland.
- 39.WuDunn, D., and P. G. Spear. 1989. Initial interaction of herpes simplex virus with cells is binding to heparan sulfate. J. Virol. 63:52-58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zaneveld, L. J., R. A. Anderson, D. Kim, S.-H. Diao, D. P. Waller, C. Chany, K. Feathergill, G. F. Doncel, M. D. Cooper, P. Hermonat, and B. C. Herold. A new antimicrobial contraceptive agent for vaginal prophylactic use: dimandelic acid ether. Fertil. Steril., in press. [DOI] [PubMed]