Abstract
The five-stranded coiled-coil domain of cartilage oligomeric matrix protein (COMPcc) forms a continuous axial pore with binding capacities for hydrophobic compounds, including prominent cell signalling molecules. Here, we report the X-ray structure of the COMPcc domain in complex with vitamin D3 at 1.7 Å resolution. The COMPcc pentamer harbours two molecules of the steroid hormone precursor in a planar s-trans conformation of the conjugated triene, with the aliphatic tails lying along the molecule axis. A hydrophilic ring of five Gln54 side chains divides the channel into two hydrophobic compartments in which the bound vitamin D3 pair is fixed in a head-to-head orientation. Vitamin D3 binding induces a volumetric increase of the cavities of ∼30% while the main chain distances of the pentamer are retained. This adaptation to the bulky ring systems of the ligands is accomplished by a rotamer re-orientation of β-branched side chains that form the knobs into holes of the coiled-coil structure. Compared with binding of vitamin D and retinoic acid by their classical receptors, COMP exerts a distinct mechanism of interaction mainly defined by the pattern of hydrophobic core residues.
Keywords: hydrophobic pore/vitamin D3 storage/X-ray crystallography
Introduction
Cartilage oligomeric matrix protein (COMP) (Oldberg et al., 1992) is a non-collageneous glycoprotein expressed predominantly in articular cartilage, tendon and ligament (Hedbom et al., 1992; DiCesare et al., 1994; Smith et al., 1997; Müller et al., 1998). It is a member of the thrombospondin (TSP) gene family of extracellular glycoproteins (Adams, 2001) and, like TSP-3 and TSP-4 (Mörgelin et al., 1992; Qabar et al., 1995; Shen et al., 1995; Ekman et al., 1997), COMP is assembled into a pentameric bouquet-like structure (Mörgelin et al., 1992), which is stabilized by interchain disulfide bonds in the N-terminal coiled-coil domain (residues 20–83) (Efimov et al., 1994; Malashkevich et al., 1996). The other two known TSPs, TSP-1 and TSP-2, are assembled into three-stranded oligomers (O’Rourke et al., 1992). All TSPs are characterized by a multidomain architecture, which in the case of COMP comprises the N-terminal heptad repeat region (cc) followed by four epidermal growth factor (EGF)-like domains (EF), seven calcium-binding domains (T3) and a C-terminal globular domain (TC). Mutations in the COMP gene shown to affect the conformation of the calcium-binding domains are connected with hereditary skeletal disorders, pseudoachondroplasia (PSACH) and multiple epiphyseal dysplasia (MED), which involve short stature and joint laxity (Hecht et al., 1995, 1998; Briggs et al., 1998; Thur et al., 2001). It has been shown recently that COMP-null mice do not exhibit any altered phenotype of skeletal development, indicating that the pentameric TSPs may be functionally redundant in the respective tissues and that PSACH and MED are not caused by a reduction of COMP in the extracellular matrix, but instead by the effects of a mutant COMP (Svensson et al., 2002).
The seco steroid vitamin D3 is the primary precursor needed for cartilage and bone mineralization, and the regulation of calcium and phosphate homeostasis (Bouillon et al., 1995). It is produced in the skin via rupture of the 9,10 carbon bond of 7-dehydrocholesterol after exposure to sunlight. The active steroid hormone 1α,25(OH)2D3 is then generated by a sequential two-step metabolism of vitamin D3 by the liver and kidney. Its classical function is the stimulation of transmembrane calcium transport in the intestine, bone and kidney. At the transcriptional level, the actions of 1α,25(OH)2D3 are mediated via high affinity binding (KD = 0.55 nM) to the vitamin D nuclear receptor (VDR) (Rochel et al., 2000). This complex forms a functional heterodimer with the retinoid X receptor and then induces the expression of target genes by binding to vitamin D-responsive elements (VDRE) (Haussler et al., 1997). Serum transport of the essentially insoluble vitamin D3 is facilitated by hydrophobic interaction with vitamin D-binding protein (VDB) (Ray, 1996; Verboven et al., 2002). Several candidates, like annexin II (Baran et al., 2000) and a putative 64.5 kDa protein (Nemere et al., 2000b), have been proposed as membrane receptors for 1α,25(OH)2D3, but their characterization is as yet limited to immunochemical and functional studies. Recently, a novel VDR-independent activity of 1α,25(OH)2D3 has been defined that involves regulation of matrix metalloproteinase function via protein kinase C signalling (Maeda et al., 2001). This process is believed to be mediated by rapid membrane-associated signalling, which results in extracellular matrix remodelling of growth plate cartilage. Interestingly, COMP expression in this tissue has been shown to be predominantly located in the proliferative region (Shen et al., 1995; Ekman et al., 1997), indicating a role for COMP in the developing cartilage.
The crystal structure of the recombinant coiled-coil domain of COMP (COMPcc) (Efimov et al., 1996; Malashkevich et al., 1996) revealed a 73 Å long hydro phobic channel with a diameter of 2–6 Å inside of the pentameric α-helical bundle. We could demonstrate binding of different hydrophobic compounds to the recombinantly expressed COMPcc, and have presented an X-ray structure of the COMPcc–all-trans retinol and COMPcc–benzene complexes (Guo et al., 1998). This approach suggested a putative role for COMP as a storage and delivery protein for regulatory molecules in bone metabolism and led us to study in detail COMPcc–vitamin D3 binding by determining the X-ray structure of this complex.
Our data present the coiled-coil domain of COMP as the first example of a structural motif with binding capacities for both vitamin A and D3. A detailed structural comparison with the ligand binding domain (LBD) of the VDR and the vitamin A binding protein (CRBP) reveals novel features of protein–ligand interaction in the coiled-coil structure.
Results
Preparation and overall X-ray structure of the COMPcc–vitamin D3 complex
The coiled-coil domain of COMP comprising residues 20–72 was obtained by recombinant expression in Escherichia coli as described previously (Efimov et al., 1996). After purification in a totally reduced state, reoxidation to the disulfide-linked pentamer was facilitated by addition of oxidized and reduced glutathione at a 10:1 ratio. Pentamer formation of the coiled-coil domain is independent of disulfide linkage, and renatured COMPcc, irrespective of its oxidative state, shows a single species in native PAGE and in sedimentation experiments using analytical ultracentrifugation. After an incubation period of several weeks, the oxidative state of the COMPcc was analysed by non-reducing SDS–PAGE, which, beside the fully oxidized pentamer band, also showed bands corresponding to the tetramer and the unclosed pentamer that runs slightly higher than the closed pentamer (data not shown) (Efimov et al., 1996). We have observed incomplete oxidation of the bacterially expressed COMPcc frequently in our laboratory, indicating that in vitro reoxidation is a methodologically limiting step in the generation of a fully disulfide-linked COMPcc pentamer. Non-reducing SDS–PAGE of solubilized COMPcc– vitamin D3 crystals revealed that the tetrameric and unclosed pentameric forms had been the preferred species during complex formation with vitamin D3, reflecting the situation in the presented solution structure of the complex.
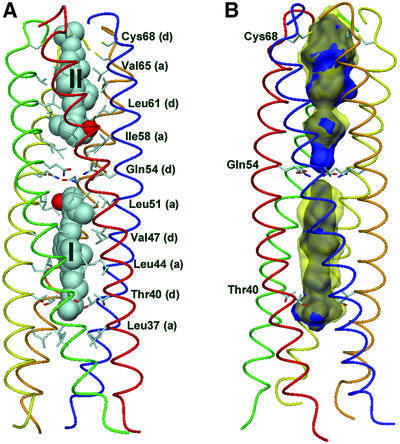
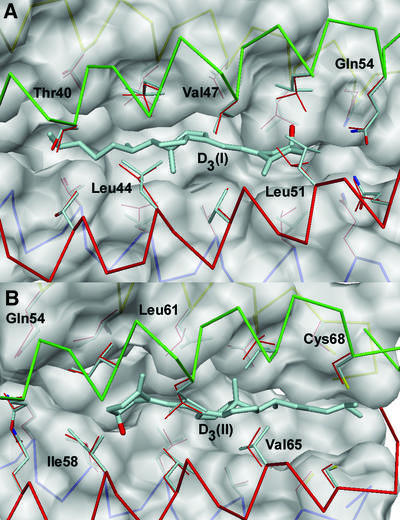
The crystal structure of the COMPcc–vitamin D3 complex was solved at 1.7 Å resolution by molecular replacement using native COMPcc (Protein Data Bank code 1vdf) (Efimov et al., 1996; Malashkevich et al., 1996) as search template (Table I). The COMPcc chain fragment forms a parallel left-handed coiled-coil pentamer with an average length of 70 Å and an average outer diameter of ∼30 Å. The axial pore of the pentamer is divided by the hydrophilic Gln54 ring system into two hydrophobic cavities exclusively lined with aliphatic side chains (Malashkevich et al., 1996). In the COMPcc–vitamin D3 complex structure, one molecule of vitamin D3 is bound inside of each hydrophobic compartment in an elongated conformation, with the length axis of the ligands paralleling the channel symmetry (Figure 1A). Vitamin D3 binding leads to an increase of both cavity volumes by ∼30%, as demonstrated by the overlay of the areas in the occupied (yellow) and unoccupied (blue) (Malashkevich et al., 1996) caverns in Figure 1B. The vitamin D3 molecules are located on different sides of the Gln54 ring system in a head-to-head configuration with the 3-hydroxy groups of the A rings (steroid notation) pointing towards the central hydrophilic narrowing. The N-terminal ligand molecule (I) is located between Thr40 and Leu51, and the second vitamin D3 (II) is positioned between Ile58 and Cys68 at the C-terminus (Figure 1A). The electron density for the protein is well defined with exceptions in chain D showing disordering of the last seven C-terminal residues (66–72). Accordingly, the ring of interchain disulfide bridges between cysteines 71 and 68 of two neighbour chains is disrupted at this position leaving chain D disconnected in the otherwise fully oxidized pentamer. We believe that for the entrance of ligand II via the C-terminus, the state of interchain disulfide bridging is critical, considering the existence of only one ligand in the COMPcc–all-trans retinol complex located at the N-terminus (Guo et al., 1998). A diffusion via the N-terminus can be excluded for molecule II because of the diameter limitation at the Gln54 ring system with 2 Å. Likewise, a fully oxidized pentamer would not be accessible for a vitamin D3 molecule via the C-terminus due to size restrictions. A diffusion via the N-terminus for ligand I, however, seems to be plausible. This assumption is underlined by changes within the helical backbone at the very N-terminus and re-orientations at the Thr40 ring to accommodate the dimethyl group fixation. These facts taken together suggest that the N-terminal position of ligand I is the primary binding site in the COMPcc for hydrophobic compounds, although data concerning the oxidative state of native COMP in cartilage are still lacking.
Table I. Statistics for data collection and structure refinement.
| Data collection | |
|---|---|
| Resolution (Å) | 1.7 |
| Observed reflections | 300.112 |
| Unique reflections | 22.133 |
| Completeness | 100 |
| Redundancy | 4.1 |
| Rsyma | 5.9 |
| Unit cell dimensions (Å, °) | |
| a | 38.29 |
| b | 49.29 |
| c | 55.26 |
| β |
103.87 |
| Refinement statistics |
|
| R-factorb (%) | 23.1 |
| Rfree (%) | 25.4 |
| Mean B-factor (Å2) | |
| Protein | 27.3 |
| Water | 41.5 |
| Ligands I/II | 55.9/71.8 |
| Bonds (Å)c | 0.005 |
| Angle (°)c | 0.9 |
| Dihedrals (°)c | 15.9 |
| Improper (°)c | 0.81 |
aRsym = Σ|I – <I>|/ΣI.
bR-factor = Σ||Fobs| – |Fcalc||/Σ|Fobs|.
cRoot-mean-square error.
Fig. 1. Overall structure of COMPcc in complex with vitamin D3. (A) Side view of the pentameric channel with bound ligand molecules I and II shown by van der Waals spheres. The N-terminus is at the bottom and the helical backbone is shown in ribbon representation with different colours for each chain. Amino acid residues forming knobs into holes according the heptad repeat pattern are shown in atom type with a and d positions indicated in parentheses. The required volume occupied by each vitamin D3 is 482 Å3. (B) Side view with helical backbone in ribbon representation and chains of emphasized amino acids shown in atom type. Two large cavities of the uncomplexed structure (blue) of volumes ranging from 450 Å3 (N-terminal) to 515 Å3 (C-terminal) can be seen. In the complexed structure, the volume inside the central channel is enlarged up so that the cavity volumes (yellow transparent) are increased markedly during diffusion of vitamin D3 into the channels. The volumes are 667 Å3 (N-terminal) and 693 Å3 (C-terminal), respectively. The cavities have been determined using native COMPcc (1vdf) and COMPcc from the complex without vitamin D3, performing MSMS with a sphere radius of 1.5 Å (Sanner et al., 1996).
Conformation of the bound vitamin D3 molecules and interactions with the COMPcc channel
The ligand molecules in the COMPcc–vitamin D3 complex adopt a linear conformation with a length of ∼17 Å separating the 3-OH group of the A ring (steroid notation) and the dimethyl group at carbon C25 (Figure 2). Their conformations resemble that of vitamin D3 in the X-ray structure of the free molecule (Figure 3B) (Suwinska and Kutner, 1996). Both ligands are rather tightly bound, with the A rings adopting a chair B conformation and the 3-hydroxy groups in axial orientation (Bouillon et al., 1995), directed from opposite sides towards the Gln54 ring system. The Cl– ion described as being bound at this position in the uncomplexed COMPcc structure (Malashkevich et al., 1996) is missing, although polar interactions of the 3-hydroxy groups with the proposed dipol system can be excluded due to disfavoured force field parameters.
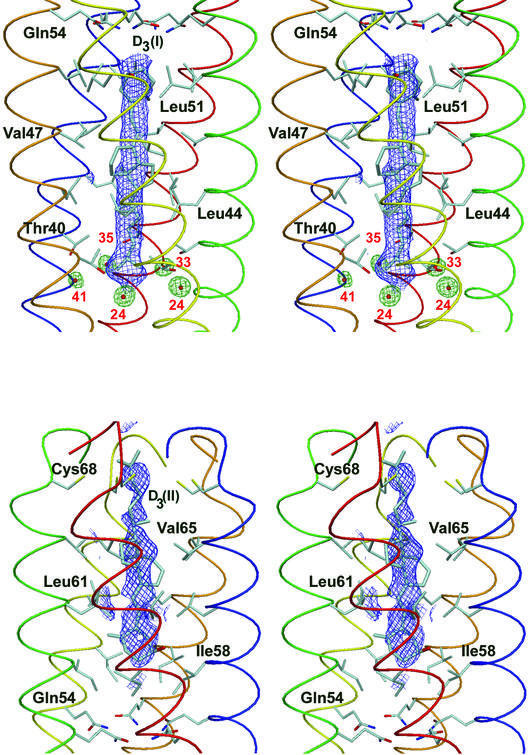
Fig. 2. Stereo view of the 1.7 Å resolution 2Fo – Fc omit map (1.2σ contour level) superimposed on both vitamin D3 molecules (molecule I at the top and II at the bottom). The five water molecules surrounding the dimethyl group of molecule I are shown as red balls and green electron density maps. The B values of the finally refined structure are shown in red.
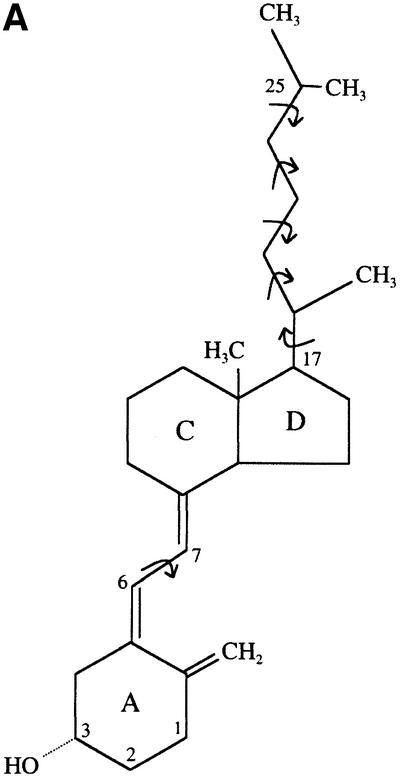
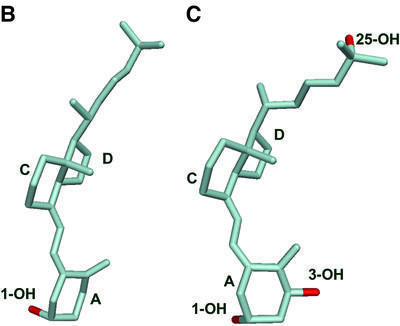
Fig. 3. Conformational flexibility of the vitamin D molecule. (A) Chemical formula of the vitamin D3 molecule used for co-crystallization with COMPcc. The dynamic rotation around the single carbon– carbon bonds is indicated by curved arrows. The molecule is drawn in an elongated conformation, as in the COMPcc. (B) Atomic model of vitamin D3 as a free molecule (Suwinska and Kutner, 1996). (C) Atomic model of 1α,25(OH)2D3 bound to the VDR LBD (Rochel et al., 2000; PDB code 1DB1). The bound molecule shows a more bent overall shape with a dihedral angle of –149°. Ring systems and hydroxyl groups are labelled according to steroid notation.
The aliphatic side chains inside of the coiled coil cause 10 regular constrictions to the diameter of the pore, varying in size between 2 and 6 Å as defined by the van der Waals radii. The space-filling A and C–D ring systems (steroid notation) of the ligands fit into cavities flanked by core residues in heptad repeat positions a and d, Leu51(a)/Val47(d) and Val47(d)/Leu44(a) for molecule I, and Ile58(a)/Leu61(d) and Leu61(d)/Val65(a) for molecule II. The opening of the unliganded channel had been described to be maximal at positions Val47 and Val65 (Malashkevich et al., 1996), which here serve to accommodate the bulky C–D ring systems of the vitamin D3 molecules. The seco B ring with the conjugated triene is tightly fitted into a hydrophobic ring of β-branched residues in heptad position d, Val47 for ligand I and Leu61 for ligand II. This interaction imposes a planar 6-s-trans conformation on the vitamin D3 molecules in the COMPcc. The C6–C7 bond exhibits a torsion angle (C5–C6–C7–C8) of 178° that coincides with the α face of the A ring. The methyl C18 on the β face of the C ring points towards Leu44 (I) and Val65 (II) in close van der Waals contacts to the side chain atoms of three of the five chains (distances between are 3.3 and 3.6 Å). The 17β aliphatic side chains are located in the hydrophobic cavities between Leu44/Thr40 (I) and Val65/Cys68 (II), and the remaining pocket of the channel is large enough to accommodate different variations of the long tail. The terminal dimethyl groups are fixed by residues Thr40 (d) for molecule I and Cys68 (d) for II.
The most remarkable change caused by the incorporation of the ligands is the increase in the cavity volumes within both channels. Analysis of the overall coiled-coil parameter for COMPcc revealed no significant change either in terms of the coiled-coil radius (8.57 Å for wild type in contrast to 8.59 Å for the complexed structure) or in terms of the supercoil pitch length of ∼204 Å for both molecules. The atomic model, when compared with native COMPcc, shows r.m.s.ds on Cα atoms of 0.69 Å. A further comparison revealed an increase of 0.2–0.3 Å in Cα positions for Thr40–Leu51 and Ile58–Val65, respectively. This rather small enlargement for each individual amino acid adds synergistically to a cumulative effect. In addition, the adaptation of the protein core is realized more exclusively by a rotamer re-orientation of β-branched side chains forming knobs into holes (Figure 4). Basically, Leu44, Leu51 for molecule I and Ala65 for molecule II show different κ1 and κ2 angles of their rotamers.
Fig. 4. Orientation of aliphatic side chains for both parts of the COMPcc channel in complex with vitamin D3 molecules I and II in comparison with ligand-free COMPcc. Side views of the pentameric channel are shown with the N-termini on the left. In the protein part, three helices are shown by van der Waals spheres, and ribbon presentation is used for the main chain of the fourth (green) and fifth (red) helix. (A) Coordinates shown for ligand molecule I in the N-terminal channel. (B) Coordinates shown for ligand II at the C-terminus. Ligand molecules are highlighted and represent the predominant orientations resulting from an extensive refinement cycle (see Materials and methods). The side chains of the complexed structure are coloured according to atom type, native COMPcc (1vdf) side chains are shown in red. The most remarkable change occurs at Leu51, where all five side chains have to be spatially re-oriented to accommodate the A ring of vitamin D3.
Discussion
Biological implications
The steroid hormone 1α,25(OH)2D3 is the main stimulator of skeletal development and bone mineralization (Bouillon et al., 1995; Malloy et al., 1999), but it also induces cell differentiation and proliferation in various tissues (Walters, 1992). Potential therapeutic applications of 1α,25(OH)2D3 are treatment of renal osteodystrophy (Gal-Moscovici et al., 2000), osteoporosis (Kleerekoper and Schein, 2001) and psoriasis (van De Kerkhof, 2001). 1α,25(OH)2D3 is also used in numerous cancer therapy models that include the systemic or oral administration of its activated form (Beer et al., 2001; Hara et al., 2001). The incidence of hypercalcaemia as a severe side-effect of such treatments initiated the search for analogues of calcitriol, which differ in their cellular responses. These efforts are often hampered by the poor uptake of the hydrophobic compounds.
In this study we report the structural features of a vitamin D3 binding motif that is part of an extracellular matrix protein predominantly expressed in bone-morphogenetic tissue. The property of the COMPcc structure of adapting to different ligands, including vitamin D3 and all-trans retinol, suggests a storage and delivery function of the COMP protein for signalling molecules relevant in cartilage tissue. Interestingly, it has been shown that the accumulation of vitamin D3 is highest in non-cellular components of bone, including extracellular matrix proteins (Neville and DeLuca, 1966). In a previous study, we demonstrated binding of vitamin D3 to COMPcc by an increase in thermal stability of the coiled-coil pentamer (Guo et al., 1998). The stabilizing effect of the hydrophobic compounds assayed in these experiments was more pronounced for larger molecules like vitamin D3, with a change of the transition midpoint temperature of 8°C, than for smaller compounds such as benzene or cyclohexane (ΔTM = 2°C). We concluded from this that binding of a prolate hydrophobic ligand inside the COMPcc channel should be obligatory because it is energetically favourable. Guo et al. (1998) reported an average binding constant of all-trans retinol to the COMPcc of ∼6 µM. Regarding the higher TM value of the COMPcc–vitamin D3 complex, it can also be concluded that the binding affinity of vitamin D3 to the N-terminal cavity of the COMPcc is even higher.
Both vitamin-D3 and all-trans retinol are poorly soluble in aqueous media, and their delivery to target cells has been shown to be dependent on various transport proteins (Blomhoff et al., 1990; Bouillon et al., 1995). For the cellular uptake of vitamin D metabolites, different pathways are discussed (Willnow and Nykjaer, 2002). Besides free diffusion through the plasma membrane, recent studies have revealed carrier-dependent endo cytic pathways playing a role in renal uptake of 25-hydroxyvitamin D3 (Nykjaer et al., 1999, 2001). A second type of interaction of vitamin D metabolites with the cell surface is defined by direct binding to membrane proteins leading to rapid activation of intracellular second messengers (Rosner et al., 1999; Nemere et al., 2000a). The expression pattern of COMP, which is restricted to growth regions of cartilage while being absent in resting and hypertrophic zones (Smith et al., 1997), would point to a function of COMP in enhancing the availability of hydrophobic growth factors by increasing their local concentration in an avascular tissue. Farquharson et al. (1998) could demonstrate that the promotion of chondrocyte matrix production by ascorbic acid is connected with the synthesis of calcitriol and the upregulation of VDR expression. Also, some phenotypic features in the clinical manifestations of PSACH and MED, like growth retardation, resemble those in patients with hereditary 1α,25(OH)2D3-resistant rickets (HVDRR), a disease caused by defective intestinal calcium absorption as a result of mutations in the VDR gene (Malloy et al., 1999). A lack of 1α,25(OH)2D3 function caused by mutated COMP in PSACH and MED might therefore serve as an explanation for this observation.
Comparison of COMPcc with other vitamin A and D binding proteins
Vitamin D and its numerous metabolites exhibit an unusual conformational flexibility (Figure 3A), which obviously gave rise to an array of differently shaped LBDs in receptor molecules and metabolizing enzymes (Norman et al., 2001). Binding of 1α,25(OH)2D3 and all-trans retinoic acid to the nuclear vitamin D receptor and the retinoid X receptor (RXR) is defined both by electrostatic interactions via fixation of the respective hydroxy functions and hydrophobic contacts that stabilize the different ring systems and aliphatic tail regions. All LBD–vitamin D X-ray structures known so far exhibit the s-trans conformation of the conjugated triene as a basic conformation. Deviations from this, which involve rotations around the C6–C7 bond and the five side chains, serve in adapting to a biologically active conformation as in the VDR/LBD. The ligands in the COMPcc–vitamin D3 complex exhibit a conformation with a planar diene C5=C6–C7=C8 moiety (Figure 4), according to the lowest energy form displayed in the structure of free vitamin D3 (Suwinska and Kutner, 1996) (Figure 3B). This is in contrast to the curved shape (‘twisted bowl’) of 1α,25(OH)2D3 in the VDR (Rochel et al., 2000; Tocchini-Valentini et al., 2001). The more bent geometry of 1α,25(OH)2D3 in VDR (C6–C7 torsion angle of –149°) would cause steric clashes inside the tight COMPcc channel (Figure 3C).
Remarkably, the electron density for both ligand molecules in the COMPcc–vitamin D3 complex is less resolved than for the protein part of the structure (Figure 2). The C–D ring systems are somewhat diffused, suggesting that orientation is not unique. Despite this disordering of vitamin D3, only one predominant binding conformer exists in both cases. The appearance of five water molecules between Leu37 and Thr40 showing slightly different B values and shape of the electron density map suggests an incompatibility with the 5-fold symmetry of COMPcc. This only partial breakdown of local symmetry can explain the ambiguous orientation of vitamin D3 towards COMPcc. As COMPcc is proposed as a storage system for vitamin D, there is no need to fix the ligand molecule in a predominant orientation.
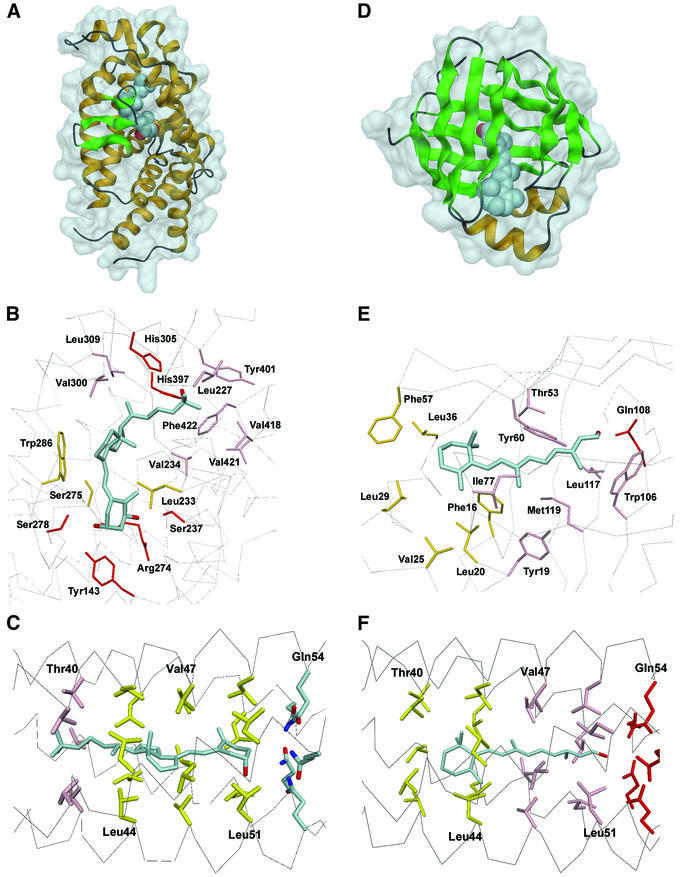
The overall structure of the VDR LBD matches the pattern of other nuclear receptor LBDs with specific deviations due to the widening of the ligand binding pocket (Rochel et al., 2000). The ligand is tightly wrapped by 13 α-helices arranged in three layers and a three-stranded β-sheet (Figure 5A). Each of the three hydroxyl groups forms two hydrogen bonds: 1-OH with Ser237 and Arg274, 3-OH with Tyr143 and Ser278, and the 25-OH with His305 and His397 (Figure 5B). The conjugated triene, connecting the A and C rings, is fitted in a hydrophobic channel sandwiched between Ser275 and Trp286 on one side and Leu233 on the other side. Specific interactions involve the hydrophobic contacts of the 17β-aliphatic chains with a cluster around Val418 on one side and Val300 on the opposite side.
Fig. 5. Structural comparison of binding modes for vitamin D metabolites and all-trans retinol by their respective receptors and COMPcc. (A) Overall view of the VDR–1α,25(OH)2D3 complex (Rochel et al., 2000). (B) Detailed view of the molecular binding mechanism of the VDR LBD. (C) Binding of vitamin D3 by the N-terminal part of the COMPcc channel. (D) Overall view of the CRBP–all-trans retinol complex. (E) Detailed view of retinol binding in CRBP (Cowan et al., 1993; PDB code 1CRB). (F) Vitamin A binding in COMPcc (Guo et al., 1998; PDB code 1FBM). In the detailed views, residues involved in electrostatic interactions are in red, those in van der Waals distance to tail regions in pink, and residues in ring fixation in yellow. The ligand molecules are highlighted in render mode.
In serum, vitamin D3 and its various metabolites, including 1α,25(OH)2D3, are delivered in a complex with vitamin D binding protein (DBP). The solution structure of this carrier protein revealed a partly solvent-exposed cleft functioning as the vitamin D binding site (Verboven et al., 2002), which is in contrast to the closed binding pocket provided by the VDR and the COMPcc presented in this study. This circumstance might also reflect the moderate binding affinity of DBP for 25-hydroxyvitamin D3 (Ka = 107–109 M–1) (Head et al., 2002).
CRBP reveals a 10-stranded antiparallel β-barrel that encapsulates the ligand inside of a lipophilic cavity (Cowan et al., 1993). All-trans retinol is bound along the barrel axis with the hydroxyl group lying in the centre of the barrel (Figure 5D). The retinol molecule is almost planar with the β-ionone ring double bond in trans position to the isoprene tail. The hydroxyl group is positioned within hydrogen-bonding distance of the Gln108 side chain (Figure 5E). The β-ionone ring fits into a hydrophobic niche formed in particular by the side chains of Phe16, L20, Val25, Leu29, Leu36 and Phe57. The isoprene tail is flanked by residues around Tyr119 and Tyr60, and fixed by Leu117 together with Trp106.
A critical feature is the hydrogen bonding of terminal hydroxy functions in both structures. Both the three frontal (1-OH, 3-OH and 25-OH) OH groups in 1α,25(OH)2D3 and the terminal OH group in retinoic acid are fixed via electrostatic interactions. In COMPcc, the Gln54 ring system can be discussed to determine the orientation of the ligands, but it does not add significantly to binding. Rather, the fixation of both ligand molecules is enforced by the spatial arrangement of the cavities between core layers in a and d positions (Figure 5C). We have demonstrated earlier complex formation of COMP with all-trans retinol by the corresponding X-ray crystal structure, which showed a positioning of the β-ionone ring in a hydrophobic environment near Thr40 at the N-terminus and a fixation of the terminal hydroxyl group within the Gln54 ion trap (Figure 5F) (Guo et al., 1998). A mutant COMPcc in which Gln54 is replaced by Ile was found to bind all-trans retinol with similar affinity as the wild-type molecule (Guo et al., 1998). We conclude from this that the alternating pattern of hydrophobic core residues is the driving force for the fixation of both ligand molecules within the channel. The position of ligands in both binding compartments is therefore adjusted by core residues in heptad positions a and d.
Conclusion and perspective
In the present study as well as in earlier works, by the presentation of binding data and X-ray solution structures of the respective complexes, we have demonstrated the association of hydrophobic signalling molecules with the recombinant coiled-coil domain of COMP. In regard to the importance that the assayed morphogens have in limb formation, the possibility of an extracellular matrix protein to mediate their cellular delivery is an attractive model. To assess this proposed functional property of COMP in cartilage, we intend to extract and purify the protein from bovine articular cartilage in native condition. The separation of the lipid phase from this COMP sample and its analysis by mass spectroscopy should help to reveal the physiological role of COMP as suggested by our data.
Materials and methods
Expression and purification of recombinant COMPcc
The coiled-coil domain of rat COMP comprising residues 27–72 was prepared as described previously (Efimov et al., 1996; Guo et al., 1998). Formation of the pentameric complex after purification was facilitated by disulfide reshuffling using a glutathione redox system and checked by non-reducing SDS–PAGE. Oxidized COMPcc was dialysed against PBS pH 7.4 and adjusted to 1 mg/ml. Solid vitamin D3, obtained from Fluka, was added to 1 mg/ml protein solution and the mixture was shaken for 24 h at 4°C in the dark to facilitate equilibration of binding. Unbound vitamin D3 was removed by ultracentrifugation and the solution was concentrated to 10 mg/ml COMPcc with a Vivaspin (Vivascience) device.
Crystallization
Crystallization experiments were performed at room temperature employing the vapour diffusion technique. Hanging droplets were made by mixing 2 µl of protein solution (10 mg/ml) with 0.2 M sodium acetate, 0.1 M Tris–HCl pH 8.5 and 30% PEG 4000. The crystals belong to space group P21 and contain one molecule of the pentameric COMPcc within the asymmetric unit.
Data collection and processing
The high resolution data set was collected at synchrotron DESY (BW7B) on a MAR research imaging plate detector. Diffraction images were processed using program suite MOSFLM (Leslie, 1994) and the structure factors were scaled and reduced using SCALA from the CCP4 package (CCP4, 1994). Statistics of the merged data are given.
Structure determination and refinement
Molecular replacement was performed using the AMoRE program of the CCP4 package (CCP4, 1994). A poly-serine model of native COMPcc structure (1vdf) was used as search template. Positional refinement was performed with CNS using the maximum likelihood method (Brunger et al., 1998). Ten percent of the reflections were excluded for use in a cross-validation set. Refinement with CNS was alternated with manual electron density refitting of side chains and terminal regions using MAIN (Turk, 1992). After including all native amino acid residues, the Rfree/R value fell to 32.8/29.7. At this stage, the ligand molecules have been fitted into a 3.0σ contoured Fo – Fc difference map using the coordinates of the free vitamin D3 molecule (Suwinska and Kutner, 1996). To determine the favoured axial orientation of the ligands within the pentameric channel, a 2° stepwise refinement (conjugated gradient minimization together with individual B-factor refinement) along the 5-fold local symmetry axis was performed. Interpretation of the electron density maps for each solution together with monitoring of the Rfree/R value ratio revealed that one orientation is preferred, which is shown in Figures 2–5. In further refinement, overall anisotropic B-factor and bulk solvent corrections were utilized. Simulated annealing omit maps confirmed the correctness of the protein and ligand structures. At R values of 27.1/25.0, water molecules were added, chosen by distance criteria and hydrogen bonding geometry, and were tested for position in spherical density, reasonable temperature factors, real-space R values and improvemment of the R-factors. The final structure contains 245 water molecules. The final R-factor and Rfree-factor are rather high (25.4 and 23.1%, respectively), which can be explained by the fact that the C-terminal part of chain D was found to be disordered and that both ligand molecules could be refined only in one predominant orientation within the channel.
The co-ordinates for the structure have been deposited in the Protein Data Bank under accession code 1MZ9.
Acknowledgments
Acknowledgements
We thank Martine Schwager for technical assistance and Andrzej Kutner for providing us with the three-dimensional coordinates of the free vitamin D3 molecule. This work was supported by the Swiss National Science Foundation (Grant 3100-049281.96).
References
- Adams J.C. (2001) Thrombospondins: multifunctional regulators of cell interactions. Annu. Rev. Cell Dev. Biol., 17, 25–51. [DOI] [PubMed] [Google Scholar]
- Baran D.T., Quail,J.M., Ray,R., Leszyk,J. and Honeyman,T. (2000) Annexin II is the membrane receptor that mediates the rapid actions of 1α,25-dihydroxyvitamin D(3). J. Cell. Biochem., 78, 34–46. [PubMed] [Google Scholar]
- Beer T.M., Munar,M. and Henner,W.D. (2001) A Phase I trial of pulse calcitriol in patients with refractory malignancies. Cancer, 91, 2431–2439. [PubMed] [Google Scholar]
- Blomhoff R., Green,M.H., Berg,T. and Norum,K.R. (1990) Transport and storage of vitamin A. Science, 250, 399–404. [DOI] [PubMed] [Google Scholar]
- Bouillon R., Okamura,W.H. and Norman,A.W. (1995) Structure– function relationships in the vitamin D endocrine system. Endocr. Rev., 16, 200–257. [DOI] [PubMed] [Google Scholar]
- Briggs M.D. et al. (1998) Diverse mutations in the gene for cartilage oligomeric matrix protein in the pseudoachondroplasia–multiple epiphyseal dysplasia disease spectrum. Am. J. Hum. Genet., 62, 311–319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunger A.T. et al. (1998) Crystallography & NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D, 54, 905–921. [DOI] [PubMed] [Google Scholar]
- CCP4 (1994) The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D, 50, 760–763. [DOI] [PubMed] [Google Scholar]
- Cowan S.W., Newcomer,M.E. and Jones,T.A. (1993) Crystallographic studies on a family of cellular lipophilic transport proteins. Refinement of P2 myelin protein and the structure determination and refinement of cellular retinol-binding protein in complex with all-trans-retinol. J. Mol. Biol., 230, 1225–1246. [DOI] [PubMed] [Google Scholar]
- DiCesare P., Hauser,N., Lehman,D., Pasumarti,S. and Paulsson,M. (1994) Cartilage oligomeric matrix protein (COMP) is an abundant component of tendon. FEBS Lett., 354, 237–240. [DOI] [PubMed] [Google Scholar]
- Efimov V.P., Lustig,A. and Engel,J. (1994) The thrombospondin-like chains of cartilage oligomeric matrix protein are assembled by a five-stranded α-helical bundle between residues 20 and 83. FEBS Lett., 341, 54–58. [DOI] [PubMed] [Google Scholar]
- Efimov V.P., Engel,J. and Malashkevich,V.N. (1996) Crystallization and preliminary crystallographic study of the pentamerizing domain from cartilage oligomeric matrix protein: a five-stranded α-helical bundle. Proteins, 24, 259–262. [DOI] [PubMed] [Google Scholar]
- Ekman S., Reinholt,F.P., Hultenby,K. and Heinegard,D. (1997) Ultrastructural immunolocalization of cartilage oligomeric matrix protein (COMP) in porcine growth cartilage. Calcif. Tissue Int., 60, 547–553. [DOI] [PubMed] [Google Scholar]
- Farquharson C., Berry,J.L., Mawer,E.B., Seawright,E. and Whitehead,C.C. (1998) Ascorbic acid-induced chondrocyte terminal differentiation: the role of the extracellular matrix and 1,25-dihydroxyvitamin D. Eur. J. Cell Biol., 76, 110–118. [DOI] [PubMed] [Google Scholar]
- Gal-Moscovici A., Rubinger,D. and Popovtzer,M.M. (2000) 24,25- dihydroxyvitamin D3 in combination with 1,25-dihydroxyvitamin D3 ameliorates renal osteodystrophy in rats with chronic renal failure. Clin. Nephrol., 53, 362–371. [PubMed] [Google Scholar]
- Guo Y., Bozic,D., Malashkevich,V.N., Kammerer,R.A., Schulthess,T. and Engel,J. (1998) All-trans retinol, vitamin D and other hydrophobic compounds bind in the axial pore of the five-stranded coiled-coil domain of cartilage oligomeric matrix protein. EMBO J., 17, 5265–5272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara K., Kusuzaki,K., Takeshita,H., Kuzuhara,A., Tsuji,Y., Ashihara,T. and Hirasawa,Y. (2001) Oral administration of 1-α-hydroxy vitamin D3 inhibits tumor growth and metastasis of a murine osteosarcoma model. Anticancer Res., 21, 321–324. [PubMed] [Google Scholar]
- Haussler M.R., Haussler,C.A., Jurutka,P.W., Thompson,P.D., Hsieh,J.C., Remus,L.S., Selznick,S.H. and Whitfield,G.K. (1997) The vitamin D hormone and its nuclear receptor: molecular actions and disease states. J. Endocrinol., 154, S57–73. [PubMed] [Google Scholar]
- Head J.F., Swamy,N. and Ray,R. (2002) Crystal structure of the complex between actin and human vitamin D-binding protein at 2.5 Å resolution. Biochemistry, 41, 9015–9020. [DOI] [PubMed] [Google Scholar]
- Hecht J.T. et al. (1995) Mutations in exon 17B of cartilage oligomeric matrix protein (COMP) cause pseudoachondroplasia. Nat. Genet., 10, 325–329. [DOI] [PubMed] [Google Scholar]
- Hecht J.T., Montufar-Solis,D., Decker,G., Lawler,J., Daniels,K. and Duke,P.J. (1998) Retention of cartilage oligomeric matrix protein (COMP) and cell death in redifferentiated pseudoachondroplasia chondrocytes. Matrix Biol., 17, 625–633. [DOI] [PubMed] [Google Scholar]
- Hedbom E. et al. (1992) Cartilage matrix proteins. An acidic oligomeric protein (COMP) detected only in cartilage. J. Biol. Chem., 267, 6132–6136. [PubMed] [Google Scholar]
- Kleerekoper M. and Schein,J.R. (2001) Comparative safety of bone remodeling agents with a focus on osteoporosis therapies. J. Clin. Pharmacol., 41, 239–250. [DOI] [PubMed] [Google Scholar]
- Leslie A.G.W. (1994) MOSFLM User’s Guide, Mosflm version 5.20. MRC Laboratory of Molecular Biology, Cambridge, UK.
- Maeda S., Dean,D.D., Sylvia,V.L., Boyan,B.D. and Schwartz,Z. (2001) Metalloproteinase activity in growth plate chondrocyte cultures is regulated by 1,25-(OH)2D3 and 24,25-(OH)2D3 and mediated through protein kinase C. Matrix Biol., 20, 87–97. [DOI] [PubMed] [Google Scholar]
- Malashkevich V.N., Kammerer,R.A., Efimov,V.P., Schulthess,T. and Engel,J. (1996) The crystal structure of a five-stranded coiled coil in COMP: a prototype ion channel? Science, 274, 761–765. [DOI] [PubMed] [Google Scholar]
- Malloy P.J., Pike,J.W. and Feldman,D. (1999) The vitamin D receptor and the syndrome of hereditary 1,25-dihydroxyvitamin D-resistant rickets. Endocr. Rev., 20, 156–188. [DOI] [PubMed] [Google Scholar]
- Mörgelin M., Heinegard,D., Engel,J. and Paulsson,M. (1992) Electron microscopy of native cartilage oligomeric matrix protein purified from the Swarm rat chondrosarcoma reveals a five-armed structure. J. Biol.Chem., 267, 6137–6141. [PubMed] [Google Scholar]
- Müller G., Michel,A. and Altenburg,E. (1998) COMP (cartilage oligomeric matrix protein) is synthesized in ligament, tendon, meniscus, and articular cartilage. Connect. Tissue Res., 39, 233–244. [DOI] [PubMed] [Google Scholar]
- Nemere I., Larsson,D. and Sundell,K. (2000a) A specific binding moiety for 1,25-dihydroxyvitamin D3 in basal lateral membranes of carp enterocytes. Am. J. Physiol. Endocrinol. Metab., 279, E614–E621. [DOI] [PubMed] [Google Scholar]
- Nemere I., Ray,R. and McManus,W. (2000b) Immunochemical studies on the putative plasmalemmal receptor for 1,25(OH)2D3. I. Chick intestine. Am. J. Physiol. Endocrinol. Metab., 278, E1104–E1114. [DOI] [PubMed] [Google Scholar]
- Neville P.F. and DeLuca,H.F. (1966) The synthesis of [1,2-3H]vitamin D3 and tissue localization of a 0.25-µg (10 IU) dose per rat. Biochemistry, 5, 2201–2207. [DOI] [PubMed] [Google Scholar]
- Norman A.W., Ishizuka,S. and Okamura,W.H. (2001) Ligands for the vitamin D endocrine system: different shapes function as agonists and antagonists for genomic and rapid response receptors or as a ligand for the plasma vitamin D binding protein. J. Steroid Biochem. Mol. Biol., 76, 49–59. [DOI] [PubMed] [Google Scholar]
- Nykjaer A., Dragun,D., Walther,D., Vorum,H., Jacobsen,C., Herz,J., Melsen,F., Christensen,E.I. and Willnow,T.E. (1999) An endocytic pathway essential for renal uptake and activation of the steroid 25-(OH) vitamin D3. Cell, 96, 507–515. [DOI] [PubMed] [Google Scholar]
- Nykjaer A. et al. (2001) Cubilin dysfunction causes abnormal metabolism of the steroid hormone 25(OH) vitamin D3. Proc. Natl Acad. Sci. USA, 98, 13895–13900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldberg A., Antonsson,P., Lindblom,K. and Heinegard,D. (1992) COMP (cartilage oligomeric matrix protein) is structurally related to the thrombospondins. J. Biol. Chem., 267, 22346–22350. [PubMed] [Google Scholar]
- O’Rourke K.M., Laherty,C.D. and Dixit,V.M. (1992) Thrombospondin 1 and thrombospondin 2 are expressed as both homo- and heterotrimers. J. Biol. Chem., 267, 24921–24924. [PubMed] [Google Scholar]
- Qabar A., Derick,L., Lawler,J. and Dixit,V. (1995) Thrombospondin 3 is a pentameric molecule held together by interchain disulfide linkage involving two cysteine residues. J. Biol. Chem., 270, 12725–12729. [DOI] [PubMed] [Google Scholar]
- Ray R. (1996) Molecular recognition in vitamin D-binding protein. Proc. Soc. Exp. Biol. Med., 212, 305–312. [DOI] [PubMed] [Google Scholar]
- Rochel N., Wurtz,J.M., Mitschler,A., Klaholz,B. and Moras,D. (2000) The crystal structure of the nuclear receptor for vitamin D bound to its natural ligand. Mol. Cell, 5, 173–179. [DOI] [PubMed] [Google Scholar]
- Rosner W., Hryb,D.J., Khan,M.S., Nakhla,A.M. and Romas,N.A. (1999) Sex hormone-binding globulin mediates steroid hormone signal transduction at the plasma membrane. J. Steroid Biochem. Mol. Biol., 69, 481–485. [DOI] [PubMed] [Google Scholar]
- Sanner M.F., Olson,A.J. and Spehner,J.C. (1996) Reduced surface: an efficient way to compute molecular surfaces. Biopolymers, 38, 305–320. [DOI] [PubMed] [Google Scholar]
- Shen Z., Heinegard,D. and Sommarin,Y. (1995) Distribution and expression of cartilage oligomeric matrix protein and bone sialoprotein show marked changes during rat femoral head development. Matrix Biol., 14, 773–781. [DOI] [PubMed] [Google Scholar]
- Smith R.K., Zunino,L., Webbon,P.M. and Heinegard,D. (1997) The distribution of cartilage oligomeric matrix protein (COMP) in tendon and its variation with tendon site, age and load. Matrix Biol., 16, 255–271. [DOI] [PubMed] [Google Scholar]
- Suwinska K. and Kutner,A. (1996) Crystal and molecular structure of 1,25-dihydroxycholecalciferol. Acta Crystallogr. B, 52, 550–554. [Google Scholar]
- Svensson L., Aszodi,A., Heinegard,D., Hunziker,E.B., Reinholt,F.P., Fassler,R. and Oldberg,A. (2002) Cartilage oligomeric matrix protein-deficient mice have normal skeletal development. Mol. Cell. Biol., 22, 4366–4371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thur J., Rosenberg,K., Nitsche,D.P., Pihlajamaa,T., Ala-Kokko,L., Heinegard,D., Paulsson,M. and Maurer,P. (2001) Mutations in cartilage oligomeric matrix protein causing pseudoachondroplasia and multiple epiphyseal dysplasia affect binding of calcium and collagen I, II, and IX. J. Biol. Chem., 276, 6083–6092. [DOI] [PubMed] [Google Scholar]
- Tocchini-Valentini G., Rochel,N., Wurtz,J.M., Mitschler,A. and Moras,D. (2001) Crystal structures of the vitamin D receptor complexed to superagonist 20-epi ligands. Proc. Natl Acad. Sci. USA, 98, 5491–5496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turk D.C. (1992) Weiterentwicklung eines Programmes für Molekülgraphik und Elektronendichte-Manipulation und seine Anwendung auf verschiedene Protein-Strukturaufklärungen. PhD thesis, Technische Universität München, Munich, Germany.
- van De Kerkhof C. (2001) New developments in the treatment of psoriasis. Skin Pharmacol. Appl. Skin Physiol., 14, 129–135. [DOI] [PubMed] [Google Scholar]
- Verboven C., Rabijns,A., De Maeyer,M., Van Baelen,H., Bouillon,R. and De Ranter,C. (2002) A structural basis for the unique binding features of the human vitamin D-binding protein. Nat. Struct. Biol., 9, 131–136. [DOI] [PubMed] [Google Scholar]
- Walters M.R. (1992) Newly identified actions of the vitamin D endocrine system. Endocr. Rev., 13, 719–764. [DOI] [PubMed] [Google Scholar]
- Willnow T.E. and Nykjaer,A. (2002) Pathways for kidney-specific uptake of the steroid hormone 25-hydroxyvitamin D3. Curr. Opin. Lipidol., 13, 255–260. [DOI] [PubMed] [Google Scholar]