Abstract
Actin is an abundant component of nerve terminals that has been implicated at multiple steps of the synaptic vesicle cycle, including reversible anchoring, exocytosis, and recycling of synaptic vesicles. In the present study we used the lamprey reticulospinal synapse to examine the role of actin at the site of synaptic vesicle recycling, the endocytic zone. Compounds interfering with actin function, including phalloidin, the catalytic subunit of Clostridium botulinum C2 toxin, and N-ethylmaleimide-treated myosin S1 fragments were microinjected into the axon. In unstimulated, phalloidin-injected axons actin filaments formed a thin cytomatrix adjacent to the plasma membrane around the synaptic vesicle cluster. The filaments proliferated after stimulation and extended toward the vesicle cluster. Synaptic vesicles were tethered along the filaments. Injection of N-ethylmaleimide-treated myosin S1 fragments caused accumulation of aggregates of synaptic vesicles between the endocytic zone and the vesicle cluster, suggesting that vesicle transport was inhibited. Phalloidin, as well as C2 toxin, also caused changes in the structure of clathrin-coated pits in stimulated synapses. Our data provide evidence for a critical role of actin in recycling of synaptic vesicles, which seems to involve functions both in endocytosis and in the transport of recycled vesicles to the synaptic vesicle cluster.
Synaptic communication depends on local recycling of synaptic vesicles in nerve terminals. A major recycling pathway involves retrieval by means of clathrin-coated pits (1–3). The basic features of this process are shared with that of clathrin-dependent endocytosis in nonneuronal cells (4). It involves recruitment of clathrin by adaptors to the membrane followed by polymerization of the clathrin coat. A clathrin-coated pit grows progressively along with an invagination of the coated membrane. After a deeply invaginated coated pit has formed, the base of the pit is cut off resulting in formation of a free coated vesicle. A range of accessory proteins have been identified, which are thought to regulate different aspects of the endocytic process (2). Many of these proteins interact with components of the actin-regulating machinery, which has pointed to a coupling between endocytosis and the actin cytoskeleton (2, 5, 6). Such a coupling is also supported by the observation of actin tails at moving endocytic vesicles (7–10). Disruption of actin function has been shown to inhibit endocytosis in some but not all cell types (11–14).
In nerve terminals clathrin-mediated budding occurs in an endocytic zone of the plasma membrane, which surrounds the release site (1, 3). Several observations indicate that actin filaments are present in this synaptic compartment. Studies of the denervated frog neuromuscular junction have shown that phalloidin labeling accumulates around rather than within release sites (15). In cell-free synaptosome preparations actin was identified by immunogold labeling in regions containing endocytic intermediates associated with filaments (16). Moreover, a filamentous cytomatrix was found to proliferate in the endocytic zone of the lamprey reticulospinal synapse after disruption of synaptojanin function (17). Perturbation of this polyphosphoinositide phosphatase increases the levels of phosphoinositides, which are known to stimulate actin polymerization (18). Although these morphological observations support an involvement of actin filaments in synaptic vesicle recycling, functional evidence is as yet lacking. Extracellular application of actin-depolymerizing compounds at intact synapses has not been found to produce consistent effects on vesicle recycling (19–22). We have now used the lamprey giant reticulospinal synapse to examine the effects of specific disruption of the presynaptic actin cytoskeleton. The reticulospinal axon is large and unbranched, which allows compounds to be microinjected in the vicinity of the release sites. By combining presynaptic microinjections with subsequent microscopic analysis we show that disruption of actin filaments in the endocytic zone is associated with impaired recycling of synaptic vesicles.
Materials and Methods
Preparation and Testing of Reagents.
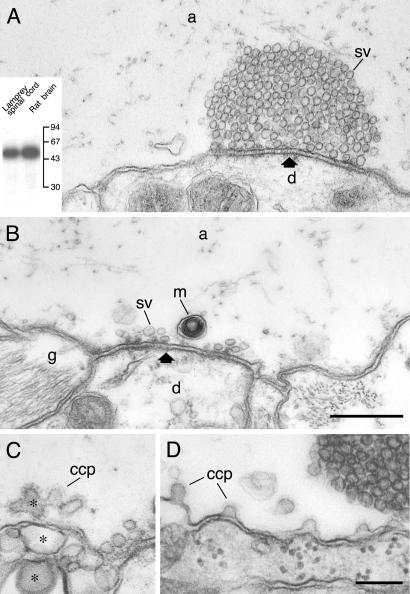
Rhodamine- or Oregon green 488-linked phalloidin (Molecular Probes, R-415; O-7466) was mixed with injection buffer (250 mM K acetate/10 mM Hepes, pH 7.4) at 300 units/250 μl. Synapsin antibodies [G-304 (23)] were labeled with Alexa Fluor 594 (Molecular Probes, A-10239). The catalytic C2I component of Clostridium botulinum C2 toxin was obtained from H. Barth, University of Freiburg, Freiburg, Germany, and tested in an ADP-ribosylation assay (24). In both lamprey spinal cord and rat brain extracts, a single band with a molecular weight corresponding to actin was ADP-ribosylated (see Fig. 4). Rabbit S1 myosin fragments (Sigma) were treated with N-ethylmaleimide (NEM) (Sigma) as described (25). NEM-treated myosin subfragment 1 (NEM-S1) was prepared fresh for each microinjection experiment and tested for binding to β/γ-actin in vitro. Bovine actin was polymerized in 75 mM KCl/2.5 mM MgCl2/10 mM imidazole/1 mM EGTA for 5 min at room temperature (pH 7.2) with or without ATP. Samples were incubated for 15 min after addition of NEM-S1 (1 mM) and then centrifuged at 200,000 × g for 20 min at 15°C. Both supernatant and pellet were analyzed by SDS/PAGE and stained with Coomassie blue. An increased binding of S1 to the pellet was consistently observed in the NEM-S1 + ATP sample. Pellets were also adsorbed on formvar-coated grids and negatively stained with 1% uranyl acetate and examined in a Philips CM12 electron microscope. A consistent decoration of actin filaments with NEM-S1 was observed (not shown).
Fig 4.
Disruption of the actin-containing cytomatrix around synaptic release sites by C. botulinum C2 toxin. (A) Resting control synapse in a reticulospinal axon injected with the C2I subunit of C. botulinum C2 toxin. (Inset) The specific ADP-ribosylation of actin by the C2I subunit in lamprey spinal cord and rat brain extract, respectively. (B and C) Synapses in an axon that was microinjected with the C2I subunit and then stimulated at 5 Hz for 30 min. (C) Expanded portions of the plasma membrane (asterisks), one of which contains clathrin-coated endocytic intermediates. The synapses shown in A–C were located within 200 μm from the injection site. (D) The endocytic zone of a stimulated synapse located ≈500 μm from the injection site in a C2I-injected axon. An increased number of unconstricted coated intermediates was observed. Only occasional vesicles occurred lateral to the vesicle cluster. m, mitochondrion; other designations are as in Figs. 2 and 3. (Bars: A and B, 0.5 μm; C and D, 0.2 μm.)
The C2 toxin and NEM-S1 were dialyzed against injection buffer and then mixed with an injection marker [Cy5-labeled GST, 10:1–5:1 (26)], which had been dialyzed against the same buffer. Control axons were injected with Cy5-GST alone which had no detectable effect on the synaptic structure (not shown; see ref. 26).
Microinjection.
Pressure microinjection into lamprey (Lampetra fluviatilis) giant reticulospinal axons and subsequent stimulation was performed as described (23, 26, 27). The physiological solution contained either a normal Ca2+ level (2.6 mM; stimulated specimens) or a reduced level (0.1 mM Ca2+ with 4 mM Mg2+; specimens maintained at rest). Experiments with phalloidin microinjections were also performed in Ca2+-free solution containing 10 mM EGTA. This condition, which strongly suppresses vesicle cycling (28), was used to ensure that the filaments detected at resting synapses were not due to low levels of vesicle turnover. The specimens were maintained in this solution for at least 30 min before injections.
Action potential stimulation was performed at a rate of 5 Hz. Earlier studies of the reticulospinal synapse have shown that at this rate of stimulation recycling keeps pace with release even after prolonged stimulation, but after perturbation of the synaptic vesicle cycle depletion of vesicles occurs (17, 26). In the present experiments stimulation periods of 2, 5, 15, and 30 min were used. Prolonged exposure with phalloidin (exceeding 30 min) can also induce complex structural changes including actin bundling and proliferation of actin-like filaments outside synapses (not shown). Proliferation of actin filaments in axoplasm after prolonged exposure to phalloidin has been reported (29).
Electron Microscopy.
Stimulation was ended by replacing the physiological solution with 3% glutaraldehyde/0.5% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4), or with 3% glutaraldehyde/4% tannic acid in 0.1 M cacodylate buffer (pH 7.4). Tannic acid treatment was used to enhance the visibility of actin filaments. The specimens were embedded in Durcupan ACM (Fluka), as described (17). For each experimental condition a minimum of five serially cut synapses from at least two different axons were examined. In most experiments both uninjected and GST-Cy5-injected axons were used as controls (17, 26). Endocytic intermediates were quantified as described (17).
Fluorescence Microscopy.
Microinjections were monitored with a charged coupled device detector (Princeton Instruments, Trenton, NJ). Images were used to identify the injected axons and to judge the relative concentration of injected compounds (27). To obtain high-resolution images of fluorescence in living axons (Fig. 1), the spinal cord was removed from the recording chamber, mounted with the ventral side down on a coverslip, and then analyzed with an inverted fluorescence microscope (Nikon Diaphot 300) or a confocal microscope system (Odyssey XL, Noran Instruments, Middleton, WI). To record the intensity of the fluorescent structures in stimulated axons, the injection was performed in normal Ringer's solution. The specimen was then quickly removed from the recording chamber and maintained for 5 min in solution with 30 mM K+. Resting specimens were maintained in Ca2+-free solution with 10 mM EGTA during the injection and image recording. Confocal images were sampled at 0.1-μm steps (×63 objective, numerical aperture 1.4). For each fluorescent structure, the average pixel intensity of the whole ring-like structure was quantified in the focal plane located in the center of the collected image stack and then normalized to the background level measured in an adjacent control region.
Fig 1.
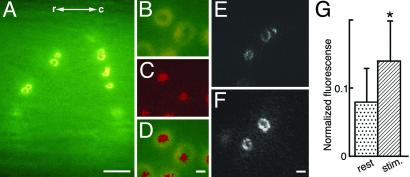
Accumulation of Oregon green-coupled phalloidin at individual synaptic release sites in living reticulospinal axons. (A) Low-power image showing accumulation of phalloidin in ring-like structures after intraaxonal injection. The fluorescent structures remained visible for 15–30 min after the injection, and then gradually dimmed. r-c, rostro-caudal directions. (B–D) Coinjection of Oregon green-coupled phalloidin (B) and Alexa-linked synapsin antibodies (C). Double exposure of both fluorophores is shown in D. (E and F) Middle sections through confocal reconstructions of phalloidin rings in Ca2+-free solution with EGTA (E) and after 5 min of stimulation with high K+ with 2.6 mM Ca2+ (F). The images were obtained from two different preparations in axons injected with similar amounts of Oregon green-coupled phalloidin. (G) Quantitative evaluation of fluorescence recorded in ring-like phalloidin-labeled structures in Ca2+-free solution (n = 18) and during K+ stimulation (n = 22). Data are presented as mean ± SD; *, P < 0.01. (Bars: A, 10 μm; B–D, 1 μm; E and F, 3 μm.)
Results
Phalloidin Identifies a Dynamic Actin Cytomatrix in the Endocytic Zone.
To investigate the distribution of actin filaments in living reticulospinal axons, we first used phalloidin, which binds to and stabilizes actin filaments. Microinjection of fluorophore-coupled phalloidin resulted in accumulation of ring-shaped fluorescent structures at the inner surface of the living axon (Fig. 1 A–F). The location of the fluorescent rings relative to synapses was studied by coinjecting Oregon green-coupled phalloidin with Alexa-linked synapsin antibodies. These antibodies have been shown to bind to synaptic vesicle clusters (23). The synapsin antibodies decorated the center of the rings (Fig. 1 B–D) confirming that the phalloidin-labeled structures surrounded synaptic release sites. We then tested whether the phalloidin rings were affected by synaptic activity. Microinjections were performed either in resting axons maintained in Ca2+-free solution or in axons stimulated with 30 mM K+, respectively (see Materials and Methods). Fluorescent rings of similar shape were observed under both conditions, although they seemed somewhat dimmer under resting conditions (Fig. 1E) than during stimulation (Fig. 1F). Comparison of the averaged fluorescence intensity of the phalloidin-labeled rings showed that it was higher during stimulation as compared with rest (P < 0.01, t test; Fig. 1G).
We next studied the effects of phalloidin at synapses with electron microscopy. In resting axons (Fig. 2A) synapses contained dense synaptic vesicle clusters surrounded by vesicle-free cytoplasm. Endocytic intermediates were only occasionally observed and were absent in most of the synapses studied in serial ultrathin sections. A thin filamentous cytomatrix could be detected in the cytoplasm of the endocytic zone adjacent to the plasma membrane (Fig. 2 A and B), consistent with phalloidin labeling under these conditions. The lateral extension from the margin of the active zone did not exceed 1 μm (Fig. 2 A and C). A similar cytomatrix seemed to be present also in resting uninjected axons, although it was often much less prominent or below the limits of detection (see Fig. 5A).
Fig 2.
Actin-like filaments in the endocytic zone of a resting synapse. (A) Electron micrograph of a synapse in a reticulospinal axon injected with phalloidin. The specimen was maintained in Ca2+-free solution with EGTA. (B and C) The framed regions in A shown at high magnification. sv, synaptic vesicle cluster; a, axoplasmic matrix; d, postsynaptic dendrite; g, glia. Thick arrow indicates the active zone. Thin arrows point to the filamentous cytomatrix. (Bars: A, 0.5 μm; B and C, 0.2 μm.)
Fig 5.
Effects of microinjection of NEM-treated myosin S1 fragments. (A) Resting control synapse in an axon microinjected with NEM-S1. Arrow indicates thin filamentous matrix. (B) A synapse in an axon microinjected with NEM-S1 and then stimulated at 5 Hz for 30 min. Note the presence of synaptic vesicles in the lateral cytomatrix. The active zone is distinguished by the triangular dense bodies facing the postsynaptic density. The synapses in A and B (and D below) were located within 200 μm from the injection site. (C) A synapse located at a distance of ≈500 μm from the injection site in an axon injected and stimulated as in B. The synaptic vesicle cluster at the active zone is partly depleted, and an aggregate of synaptic vesicles is present above the endocytic zone. (D) A synapse in an NEM-S1-injected axon subjected to stimulation at 5 Hz for 2 min before fixation. Thin arrows in B–D indicate the proliferated filamentous cytomatrix. Open arrows indicate synaptic vesicles associated with the filaments. Clathrin-coated pits (ccp) are visible at the plasma membrane in stimulated axons. Other designations are as in Fig. 2. (Bar: 0.5 μm.)
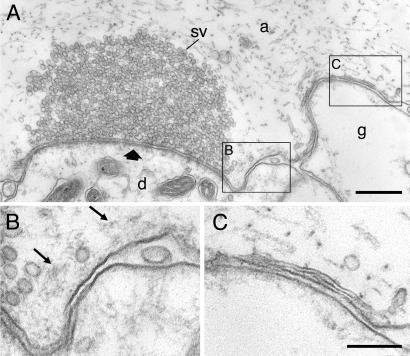
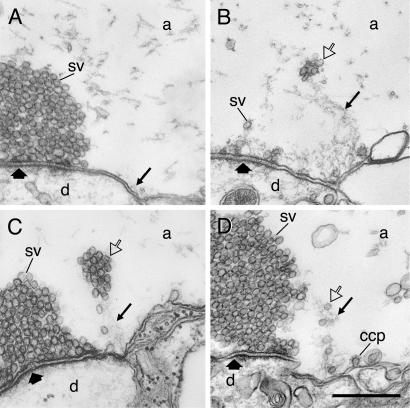
After action potential stimulation, several striking changes occurred in the endocytic zone of phalloidin-injected axons (Fig. 3). First, the filamentous cytomatrix was expanded (Fig. 3 A–C) consistent with the increase in phalloidin fluorescence observed by light microscopy. The filaments also showed a change in organization. Distinct strands of filaments were now seen instead of an unstructured filamentous material (small arrows in Fig. 3 B–E; for images of stimulated control synapses see ref. 27). The filaments could be traced from the endocytic zone to the periphery of the vesicle cluster. Second, synaptic vesicles were tethered to the filaments (Fig. 3 A–D). Their distribution apart from the vesicle cluster suggested that they could represent recycled vesicles moving from the endocytic zone to the cluster. Moreover, an effect on clathrin-coated pits was observed in the endocytic zone (Fig. 3 D and E). In addition to coated pits with a normal morphology, a proportion of the coated pits had very long unconstricted necks (Fig. 3 D and E). The occurrence of such coated pits depended on the concentration of phalloidin in the axon and could reach in single synapses up to 50% of the total amount of coated intermediates found at the endocytic zone. Because coated pits with this morphology have not been observed at this synapse previously, we assume that they were specifically induced by phalloidin (3). Clathrin-coated pits, including those with extended necks, were sometimes seen to be contacting actin-like filaments (Fig. 3E).
Fig 3.
Stimulus-evoked reorganization of filaments after microinjection of phalloidin. (A–C) Electron micrographs from two different synapses in axons that were microinjected with phalloidin and then stimulated at 5 Hz for 30 (A and B) and 15 min (C), respectively, before fixation. Synaptic vesicles (open arrows) are tethered along filaments (thin arrows) extending from the endocytic zone toward the margin of the vesicle cluster. (D and E) Sections from a different synapse in the same specimen as in C. Many clathrin-coated pits (ccp), some of which had an elongated neck, occurred at the plasma membrane of the endocytic zone. Actin-like filaments attached to the neck of coated pits are indicated by thin arrows. Other designations are as in Fig. 2. (Bars: A, 0.5 μm; B–E, 0.2 μm.)
Perturbation of Actin Filaments with C2 Toxin and Myosin Fragments Impairs Vesicle Recycling.
The effects of phalloidin indicated that the actin-containing cytomatrix participates in synaptic vesicle recycling. To examine this possibility further, we tested the effect of disrupting actin polymerization with the enzymatic C2I subunit of C. botulinum C2 toxin (24). The effect of the toxin was studied in synapses either maintained at rest or after 30 min stimulation at 5 Hz. In both conditions, the filamentous cytomatrix could not be detected in the endocytic zone after microinjection of the C2I subunit (Fig. 4). Moreover, the synaptic vesicle clusters were greatly reduced in stimulated (Fig. 4B), but not in unstimulated (Fig. 4A) axons. At least part of the vesicle depletion seemed to be due to an impaired retrieval of vesicle membrane, because the presynaptic plasma membrane in the endocytic zone was expanded (Fig. 4 B and C). Clathrin-coated pits with a distorted morphology were observed on expanded portions of the plasma membrane (Fig. 4C). An effect of the C2 toxin on membrane retrieval was further supported by analysis of stimulated synapses far from the injection site [>500 μm, where the concentration of C2 toxin was lower (27)]. Under these conditions (Fig. 4D) vesicle clusters were not depleted, but the number of clathrin-coated pits was increased approximately 4-fold as compared with synapses from uninjected axons from the same specimen (P < 0.001; t test; n = 6 serially sectioned synapses). Moreover, the proportion of coated pits with wide necks (“stage 2,” see ref. 28, was increased from 26% in stimulated uninjected control axons to 67% in injected axons (P < 0.001; n = 6; t test).
Involvement of the actin cytomatrix in synaptic vesicle recycling was also probed with an actin filament-binding compound. The subfragment 1 (S1) of myosin was pretreated with NEM, which inhibits dissociation of the bound fragment (25). The ability of the fragments to bind actin filaments was verified in in vitro assays and at the ultrastructural level by negative staining (see Materials and Methods). When NEM-S1 fragments were microinjected into axons maintained at rest, the synaptic structure was similar to that in uninjected unstimulated synapses (Fig. 5A). After stimulation at 5 Hz (30 min), the filamentous cytomatrix lateral to the active zone was clearly visible (Fig. 5B, thin arrow). Vesicles seemed to be associated with filaments in the cytomatrix, and the cluster was depleted almost completely (Fig. 5B). Scattered synaptic vesicles could also be detected in the cytoplasm outside the synaptic area (not shown). Effects of NEM-S1 on clathrin-coated pits were not observed. Clathrin-coated pits were similar in structure and number to those in stimulated control synapses. We also examined synapses located far from the injection site (>500 μm). At these synapses the vesicle cluster was only partly depleted (Fig. 5C), whereas large aggregates of synaptic vesicles occurred lateral to the active zones. Axons injected with NEM-S1 were then subjected to only brief stimulation (2 min). Under these conditions, synapses located close to the injection site contained large vesicle clusters (Fig. 5D). Lateral to the cluster synaptic vesicles occurred in strands extending between the endocytic zone and the periphery of the vesicle cluster. Such strands of vesicles were observed consistently in serial sections of synapses in NEM-S1-injected axons, but not in adjacent control axons. In the axons injected with NEM-S1 and stimulated for 2 min, the number of synaptic vesicles lateral to the vesicle cluster was more than three times higher than in synapses in adjacent uninjected giant axons (n = 6 in each case; P < 0.001). We did not, however, observe the regular “arrowhead” decoration of filaments (evident in vitro; see Materials and Methods) in the intact synapse, most likely because of lower concentrations of NEM-S1 at synapses after microinjection.
Discussion
The coupling between synaptic activity and the occurrence of clathrin-coated intermediates at the plasma membrane around synaptic release sites has been documented at a variety of central and peripheral synapses (1, 3, 30, 31). This region has been defined as a distinct synaptic compartment, the endocytic zone, on the basis of light microscopic studies in Drosophila. The dynamin-binding protein Dap160/intersectin was shown to be accumulated in this region (32), and other endocytic proteins including dynamin and endophilin have been localized to this area in stimulated Drosophila synapses (33, 34). Our recent immunogold studies in lamprey synapses have shown that dynamin can be detected in the endocytic zone after stimulation (E. Evergren, N.T., H. Gad, P.L., L.B., and O.S., unpublished observations). Here we localized actin to this compartment at a central synapse and showed that it is linked with synaptic vesicle recycling. The three different compounds used, phalloidin, C2I toxin, and NEM-S1, are all known to affect actin specifically by different mechanisms. Our observation that each compound affected the trafficking of vesicles at endocytic zones strongly indicates that actin is a component of the endocytic protein machinery around synaptic release sites.
Actin-like filaments could be detected in the endocytic zone in resting synapses even after suppression of vesicle cycling in Ca2+-free solution. Under these conditions the cytomatrix appeared as a thin layer adjacent to the plasma membrane. Stimulation induced proliferation of the filamentous cytomatrix toward the synaptic vesicle cluster. We therefore suggest that actin filaments grow out from the endocytic zone during synaptic activity (Fig. 6). Although we did not determine the exact time course of filament proliferation, they were already detectable after 2 min of stimulation, suggesting that it is a rapid process.
Fig 6.
Schematic drawing showing organization of the endocytic zone in a central synapse at rest and during synaptic activity. Recycling of synaptic vesicles is associated with expansion of a cytoskeletal matrix containing actin filaments at the sites of endocytosis.
Synaptic vesicles were associated with the proliferated filaments in stimulated synapses both after NEM-S1 and phalloidin injection. In unstimulated synapses vesicles were not detected in this region (17, 27). These observations suggest that the synaptic vesicles detected lateral to the vesicle cluster represent recycled vesicles trapped during migration from the endocytic zone to the cluster. This possibility is supported by the observation of aggregates of synaptic vesicles adjacent to synaptic vesicle clusters after NEM-S1 injection. The close association between vesicles and actin filaments is suggestive of an actin-based mechanism of vesicle transport in the synapse. Such a mechanism could share features with the actin tails implicated in intracellular movement of different types of vesicles and infectious agents (7, 8, 35, 36). Both actin treadmilling and actin–myosin interactions may potentially be involved in this mechanism of movement (36). Recently, dynamin was shown to be present in actin tails at endocytic vesicles suggesting that these tails may be functionally coupled with the endocytic machinery (9, 10).
The involvement of actin in synaptic vesicle recycling is most likely not restricted solely to vesicle transport. In experiments with C2 toxin and phalloidin we observed an inhibition of endocytosis and changes in the relative proportion of unconstricted clathrin-coated pits. These observations are consistent with the proposed role of actin at the constriction and/or fission steps of endocytosis (2, 5, 6). Our observations of actin filaments attached to the neck of coated pits and the presence of some coated pits with elongated necks could be related to such a function.
Acknowledgments
We thank Dr. Holger Barth for providing C2 toxin and Prof. Uno Lindberg, Stockholm University for providing actin. This work was supported by Swedish Medical Research Council Grants 11287 (to L.B.) and 13473 (to O.S.) and Å. Wibergs Stiftelse (to O.S.), National Institute of Mental Health Grant MH 39327 (to P.G.), National Institutes of Health Grant GM 07982 (to O.B.), the Rockefeller University–Karolinska Institutet Exchange Program (to O.B. and O.S.), and Rockefeller University funds (to O.B.). V.A.P. received support from National Institutes of Health Grant R01 NS037823. O.K. was supported by the European Commission as a Marie Curie Research Fellow.
Abbreviations
NEM, N-ethylmaleimide
NEM-S1, NEM-treated myosin subfragment 1
This paper was submitted directly (Track II) to the PNAS office.
References
- 1.Jarousse N. & Kelly, R. B. (2001) Curr. Opin. Cell Biol. 13, 461-469. [DOI] [PubMed] [Google Scholar]
- 2.Slepnev V. I. & De Camilli, P. (2000) Nat. Rev. Neurosci. 1, 161-172. [DOI] [PubMed] [Google Scholar]
- 3.Brodin L., Löw, P. & Shupliakov, O. (2000) Curr. Opin. Neurobiol. 10, 312-320. [DOI] [PubMed] [Google Scholar]
- 4.Schmid S. L. (1997) Annu. Rev. Biochem. 66, 511-548. [DOI] [PubMed] [Google Scholar]
- 5.Qualmann B., Kessels, M. M. & Kelly, R. B. (2000) J. Cell Biol. 150, 111-116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jeng R. L. & Welch, M. D. (2001) Curr. Biol. 11, R691-R694. [DOI] [PubMed] [Google Scholar]
- 7.Merrifield C. J., Moss, S. E., Ballestrem, C., Imhof, B. A., Giese, G., Wunderlich, I. & Almers, W. (1999) Nat. Cell Biol. 1, 72-74. [DOI] [PubMed] [Google Scholar]
- 8.Rozelle A. L., Machesky, L. M., Yamamoto, M., Driessens, M. H., Insall, R. H., Roth, M. G., Luby-Phelps, K., Marriott, G., Hall, A. & Yin, H. L. (2000) Curr. Biol. 10, 311-320. [DOI] [PubMed] [Google Scholar]
- 9.Lee E. & De Camilli, P. (2002) Proc. Natl. Acad. Sci. USA 99, 161-166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Orth J. D., Krueger, E. W., Cao, H. & McNiven, M. A. (2002) Proc. Natl. Acad. Sci. USA 99, 167-172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gottlieb T. A., Ivanov, I. E., Adesnik, M. & Sabatini, D. D. (1993) J. Cell Biol. 120, 695-710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Geli M. I. & Riezman, H. (1998) J. Cell Sci. 111, Part 8, 1031-1037. [DOI] [PubMed] [Google Scholar]
- 13.Lamaze C., Fujimoto, L. M., Yin, H. L. & Schmid, S. L. (1997) J. Biol. Chem. 272, 20332-20335. [DOI] [PubMed] [Google Scholar]
- 14.Fujimoto L. M., Roth, R., Heuser, J. E. & Schmid, S. L. (2000) Traffic 1, 161-171. [DOI] [PubMed] [Google Scholar]
- 15.Dunaevsky A. & Connor, E. A. (2000) J. Neurosci. 20, 6007-6012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wenk M. R., Pellegrini, L., Klenchin, V. A., Di Paolo, G., Chang, S., Daniell, L., Arioka, M., Martin, T. F. & De Camilli, P. (2001) Neuron 32, 79-88. [DOI] [PubMed] [Google Scholar]
- 17.Gad H., Ringstad, N., Löw, P., Kjaerulff, O., Gustafsson, J., Wenk, M., Di Paolo, G., Nemoto, Y., Crum, J., Ellisman, M. H., et al. (2000) Neuron 27, 301-312. [DOI] [PubMed] [Google Scholar]
- 18.Cremona O., Di Paolo, G., Wenk, M. R., Luthi, A., Kim, W. T., Takei, K., Daniell, L., Nemoto, Y., Shears, S. B., Flavell, R. A., et al. (1999) Cell 99, 179-188. [DOI] [PubMed] [Google Scholar]
- 19.Betz W. J. & Henkel, A. W. (1994) J. Cell Biol. 124, 843-854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kuromi H. & Kidokoro, Y. (1998) Neuron 20, 917-925. [DOI] [PubMed] [Google Scholar]
- 21.Job C. & Lagnado, L. (1998) J. Cell Biol. 143, 1661-1672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Doussau F. & Augustine, G. J. (2000) Biochimie 82, 353-363. [DOI] [PubMed] [Google Scholar]
- 23.Pieribone V. A., Shupliakov, O., Brodin, L., Hilfiker-Rothenfluh, S., Czernik, A. J. & Greengard, P. (1995) Nature 375, 493-497. [DOI] [PubMed] [Google Scholar]
- 24.Barth H., Preiss, J. C., Hofmann, F. & Aktories, K. (1998) J. Biol. Chem. 273, 29506-29511. [DOI] [PubMed] [Google Scholar]
- 25.Lin C. H., Espreafico, E. M., Mooseker, M. S. & Forscher, P. (1996) Neuron 16, 769-782. [DOI] [PubMed] [Google Scholar]
- 26.Shupliakov O., Löw, P., Grabs, D., Gad, H., Chen, H., David, C., Takei, K., De Camilli, P. & Brodin, L. (1997) Science 276, 259-263. [DOI] [PubMed] [Google Scholar]
- 27.Ringstad N., Gad, H., Löw, P., Di Paolo, G., Brodin, L., Shupliakov, O. & De Camilli, P. (1999) Neuron 24, 143-154. [DOI] [PubMed] [Google Scholar]
- 28.Gad H., Löw, P., Zotova, E., Brodin, L. & Shupliakov, O. (1998) Neuron 21, 607-616. [DOI] [PubMed] [Google Scholar]
- 29.Kuznetsov S. A., Langford, G. M. & Weiss, D. G. (1992) Nature 356, 722-725. [DOI] [PubMed] [Google Scholar]
- 30.Heuser J. (1989) Cell Biol. Int. Rep. 13, 1063-1076. [DOI] [PubMed] [Google Scholar]
- 31.Takei K., Mundigl, O., Daniell, L. & De Camilli, P. (1996) J. Cell Biol. 133, 1237-1250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Roos J. & Kelly, R. B. (1999) Curr. Biol. 9, 1411-1414. [DOI] [PubMed] [Google Scholar]
- 33.Estes P. S., Roos, J., van der Bliek, A., Kelly, R. B., Krishnan, K. S. & Ramaswami, M. (1996) J. Neurosci. 16, 5443-5456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Guichet A., Wucherpfennig, T., Dudu, V., Etter, S., Wilsch-Brauniger, M., Hellwig, A., Gonzalez-Gaitan, M., Huttner, W. B. & Schmidt, A. A. (2002) EMBO J. 21, 1661-1672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Higgs H. N. & Pollard, T. D. (2001) Annu. Rev. Biochem. 70, 649-676. [DOI] [PubMed] [Google Scholar]
- 36.Taunton J. (2001) Curr. Opin. Cell Biol. 13, 85-91. [DOI] [PubMed] [Google Scholar]