Abstract
Miyoshi et al. compared the role of the prolactin receptor (PrlR) and its downstream mediator, the signal transducer and activator of transcription 5 (Stat5), in mammary epithelial cells in vivo by studying PrlR-/- and Stat5ab-/- mouse mammary epithelial transplants during pregnancy. At first glance, the two mutant epithelia appear to have similar defects in the differentiation of the alveolar epithelium. However, a closer examination by Miyoshi et al. revealed defects in the epithelial architecture of the smallest ducts of Stat5ab-/- transplants not apparent in the PrlR-/- transplants, suggesting that Stat5 is more than a simple mediator of PrlR action.
Keywords: cell adhesion, mammary epithelium, prolactin receptor, Stat5, steroid hormones
Introduction
Pituitary prolactin is a key regulator of breast development [1-3]. Some prolactin is also produced by the breast epithelium itself, and local prolactin signaling can be deregulated during breast carcinogenesis [4,5]. Prolactin acts via the prolactin receptor (PrlR), a member of the cytokine receptor family [6], and its associated kinase Jak2 [7]. One of the key signaling molecules activated by the PrlR is the signal transducer and activator of transcription 5 (Stat5) [8,9].
A genetic approach to address and compare the contribution of PrlR and its key signaling molecule Stat5 for mammary gland differentiation during pregnancy, by deletion of their genes, is complicated by two instances. First, two closely related Stat5 genes exist, namely Stat5a and Stat5b. They share 92% of their amino acid sequence, are partially functionally redundant, and differ mainly in their carboxy-terminal region [10]. The establishment of Stat5a and Stat5b double knockout mice is therefore mandatory. Second, because PrlR-defective mice [11] or mice carrying inactivated Stat5a and Stat5b (Stat5ab-/-) are infertile [12], it is not possible to study the development of their mammary glands during pregnancy. To overcome the second problem, powerful transplantation techniques have been used (for a review, see [13]). In particular, mammary epithelium from infertile mice can be engrafted to mammary fat pads of wild-type prepubertal mice that are surgically cleared of the endogenous epithelium [14].
PrlR-/- mammary epithelial cells (MECs) engrafted to cleared mammary fat pads of a wild-type host, and thus exposed to a normal endocrine milieu, undergo normal ductal development during puberty but fail to form alveoli and to differentiate into milk-secreting cells during pregnancy [15]. This indicates that the PrlR expressed in MECs is required for alveologenesis and the associated differentiation process.
Miyoshi et al. used mammary epithelial transplants of Stat5ab-/- epithelium and compared them directly with PrlR-/- epithelial transplants [16]. At first glance, the expectations that the phenotypes of Stat5ab-/- and PrlR-/- epithelia are similar are met. But as Miyoshi et al. looked more closely, they uncovered thought-provoking differences that tell us we still have some significant lessons to learn about the biology of these signaling molecules.
Phenotypical similarities of Stat5ab-/- and PrlR-/- epithelia
Whole mount microscopy of mammary glands engrafted with Stat5ab-/- or PrlR-/- epithelium confirms that both mutants form a normal ductal system in the adult virgin host. At the end of pregnancy, however, when the wild-type epithelium has fully expanded and the alveoli are distended by secretion, there is no alveolar development in the PrlR-/- MEC and little development in the Stat5ab-/- MEC. Expression of the milk proteins is substantially reduced in both of the mutant tissues. Together, these data confirm that Stat5 and the PrlR play an essential role in alveolar morphogenesis and differentiation.
Unique defects in the differentiation of the Stat5ab-/- epithelium
As Miyoshi et al. examined the structural organization of the epithelium at the end of pregnancy in more detail, a Stat5ab-/- epithelial-specific phenotype became apparent. The intraductal space, clearly discernable even in the smallest ducts of the PrlR-/- epithelium, is partially obliterated in the Stat5ab-/- epithelium. The epithelial cells lining in the smallest branches are of irregular shape and appear disorganized. There are multiple layers of luminal epithelial cells in the Stat5ab-/- transplants and, consequently, crowded lumina.
Electron micrography reveals that microvilli on the apical surface and tight junctions are difficult to find in Stat5ab-/- epithelia, and that the intercellular spaces are disrupted. It remains most puzzling that the unique Stat5ab-/- defect becomes apparent specifically when alveolar morphogenesis is to take place. This suggests an essential and nonredundant role of Stat5 in the induction or repression of genes in the epithelial cells forming the alveoli versus those forming the ducts. A putative function of such genes is the control of intercellular adhesion in the nascent alveoli.
Proliferative response to estrogen and progesterone is more profoundly inhibited in the PrlR-/- MEC compared with the Stat5ab-/- MEC
Miyoshi et al. assessed the proliferative response of the mutant engrafted epithelia by injecting animals with estra-diol and progesterone, and measuring 5'-bromo-2' deoxyuridine incorporation 48 hours later. In this assay, both mutant epithelia exhibited a reduced epithelial proliferation when compared with an engrafted wild-type epithelium. Remarkably, the inhibition of the PrlR-/--deficient MEC was approximately twofold higher than the inhibition of the Stat5ab-/- MEC.
Since at the end of pregnancy both mutant epithelia have perfectly completed ductal morphogenesis, and this process is under the control of estrogen and progesterone [1], it is at first sight surprising that the mutant epithelia respond differently to estradiol and progesterone in the proliferation assay. Possible explanations for the failure to detect a proliferation phenotype at the end of pregnancy are that the time of hormonal stimulation is longer or because there is compensation through many additional stimuli during pregnancy, or both. Indeed, there are other examples of transient phenotypes that disappear by the end of pregnancy (e.g. wnt-4) [17].
Implications for the role of the PrlR-Jak2-Stat5 pathway in MEC differentiation
The similarities in the defects of PrlR-/- and Stat5ab-/- epithelia confirm an important contribution of the PrlR-Jak2-Stat5 pathway to estrogen/progesterone-induced proliferation and alveologenesis. There is still much to learn regarding which genes are controlled by this pathway in the mammary epithelium.
Stat5 has anti-apoptotic activity in hematopoietic tissues [18]. Whether this also applies to the mammary epithelium remains to be established.
By searching for genes controlled by this pathway, Miyoshi et al. analyzed the expression of genes that have been previously implicated in cellular adhesion and secretory differentiation. They determined Cx32 as a putative target gene of the PrlR-Jak2-Stat5 pathway. Cx32 is a connexin induced at lactation that has a potential role in the establishment of the secretory phenotype. Cx32 expression is absent in the mutant epithelia. Whether the failure of expression is due to its direct regulation by Stat5 or to a more indirect dependence on the secretory phenotype remains to be established.
The observed subtle differences in the phenotype of PrlR-/- and Stat5ab-/- epithelia indicate that, in addition to the classical linear PrlR-Jak2-Stat5 pathway, Stat5-independent pathways triggered by the activated PrlR and its associated Jak2 contribute to the proliferative response of the epithelium. Other pathways that activate Stat5 but do not involve the PrlR are also mandatory for alveologenesis (Fig. 1).
Figure 1.
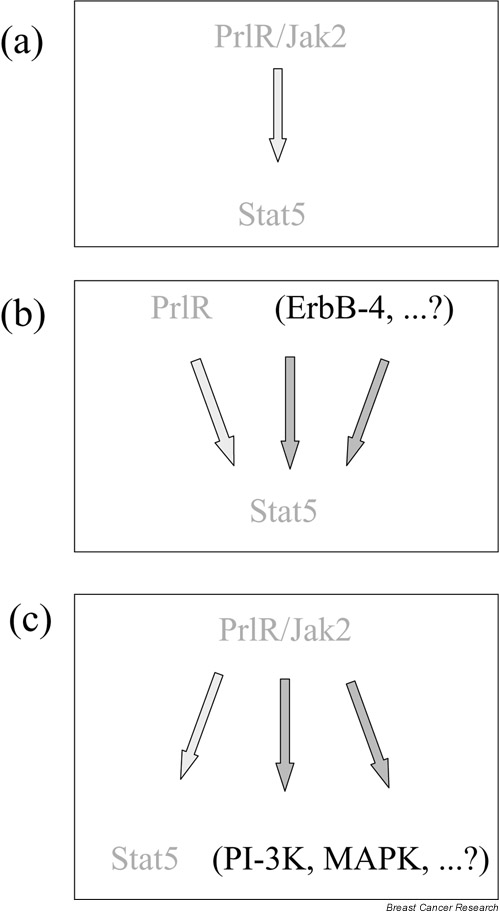
Interdependence of prolactin receptor (PrlR) and signal transducer and activator of transcription 5 (Stat5) signaling. (a) The 'classical' PrlR-Jak2-Stat5 pathway contributes to alveolar development. (b) Alternative routes to activate Stat5 are required for alveologenesis. (c) Potential contribution of multiple PrlR-dependent pathways to the proliferative response to estrogen and progesterone. PI-3K, PI 3-kinase; MAPK, mitogen-activated protein kinase.
PrlR-independent activation of Stat5
The unique defect of the Stat5ab-/- mutant epithelium indicates a role of additional pathways not dependent on the PrlR for the induction of Stat5. This is supported by the reported activation of Stat5 in the mammary epithelium by epidermal growth factor [19] and by the requirement for ErbB-4-triggered activation of Stat5 to trigger lobuloalveolar development during pregnancy [20].
Stat5-independent pathways triggered by the activated PrlR
As observed with other cytokine receptors with associated Jak kinases, activation of the PrlR/Jak2 by prolactin leads to the induction of other signaling pathways, such as the mitogen-activated protein kinase (MAPK) and PI 3-kinase (PI3K) pathways. The relative signaling output of these different pathways appears to be crucial for the biological effect, indicating that the regulation of the balance between these pathways is of utmost importance for development and differentiation.
A recent example for a specific regulator of this balance is the suppressor of cytokine signaling SOCS-3, which has been shown to lead to the selective inhibition of Stat5 signaling but not of the MAPK pathway after IL-2 stimulation [21]. SOCS-3 might have a similar function in the mammary gland, where it is differentially regulated during development [22].
A further point to consider is that extensive crosstalk occurs between prolactin and other hormonal signaling pathways; that is, estrogen induces expression of the progesterone receptor [23] and PrlR [24], prolactin signaling induces the expression of the estrogen receptor ERα and ERβ in granulosa cells [25,26], and prolactin has been shown to upregulate estrogen receptor expression in cultured mammary epithelial cells [27]. It is therefore important to examine the expression of the estrogen receptor and the progesterone receptor in the mutant epithelia to assess whether the number of cells, which are responsive to the hormones, is reduced in the mutantepithelia.
Finally, another possibility is that local prolactin signaling is important to the proliferative response to estrogen and progesterone. Both estrogen and progesterone act by paracrine mechanisms to induce proliferation, and it is conceivable that locally secreted prolactin is one of the mediators of these effects.
Conclusion
Stat5 was originally cloned as the 'mammary gland factor' and functionally characterized as a mediator of prolactin-induced β-casein expression. The literature suggested that the basic role of Stat5 in the mammary gland was to mediate prolactin signaling, while the PrlR in turn relied heavily on Stat5 to mediate its effects.
Miyoshi et al. provide evidence that there is more than this simple mutual relationship between PrlR and Stat5. For example, inactivation of Stat5 leads to much more severe defects in the intercellular adhesion of epithelial cells than PrlR deletion, whereas PrlR deletion has a more dramatic effect on proliferation than a deletion of Stat5. Further analysis of the role of these signaling pathways will provide important insight into mammary gland morphogenesis and differentiation.
Abbreviations
IL = interleukin; MAPK = mitogen-activated protein kinase; MEC = mammary epithelial cell; PI3K = PI 3-kinase; PrlR = prolactin receptor; Stat5 = signal transducer and activator of transcription 5.
Acknowledgments
Acknowledgements
The authors thank G. Paolo Dotto for critical reading of the manuscript and Gillian Kalter for secretarial support.
References
- Nandi S. Endocrine control of mammary-gland development in the C3H/He Crgl mouse. J Natl Cancer Inst. 1958;21:1039–1063. [PubMed] [Google Scholar]
- Lyons WR. Hormonal synergism in mammary growth. Proc R Soc Lond Ser B. 1958;149:303–325. doi: 10.1098/rspb.1958.0071. [DOI] [PubMed] [Google Scholar]
- Horseman ND. Prolactin. In: DeGroot LJ, editor. Endocrinology. 4th. Vol. 1. WB Saunders; 2001. pp. 209–220. [Google Scholar]
- Ginsburg E, Vonderhaar B. Prolactin synthesis and secretion by human breast cancer cells. Cancer Res. 1995;55:2591–2595. [PubMed] [Google Scholar]
- Gill S, Peston D, Vonderhaar BK, Shousha S. Expression of pro-lactin receptors in normal, benign, and malignant breast tissue: an immunohistological study. J Clin Pathol. 2001;54:956–960. doi: 10.1136/jcp.54.12.956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goffin V, Kelly PN. The prolactin/growth hormone receptor family: structure/function relationships. J Mammary Gland Biol Neoplasia. 1997;2:7–17. doi: 10.1023/a:1026313211704. [DOI] [PubMed] [Google Scholar]
- Rui H, Kirken RA, Farrar WL. Activation of receptor-associated tyrosine kinase JAK2 by prolactin. J Biol Chem. 1994;269:5364–5368. [PubMed] [Google Scholar]
- Groner B, Gouilleux F. Prolactin-mediated gene activation in mammary epithelial cells. Curr Opin Genet Dev. 1995;5:587–594. doi: 10.1016/0959-437X(95)80027-1. [DOI] [PubMed] [Google Scholar]
- Ihle JN, Stravapodis D, Parganas E, Thierfelder W, Feng J, Wang D, Teglund S. The roles of Jaks and Stats in cytokine signaling. Cancer J Sci Am. 1998;4(suppl 1):S84–S91. [PubMed] [Google Scholar]
- Liu X, Robinson GW, Gouilleux F, Groner B, Hennighausen L. Cloning and expression of Stat5 and an additional homologue (Stat5b) involved in prolactin signal transduction in mouse mammary tissue. Proc Natl Acad Sci USA. 1995;92:8831–8835. doi: 10.1073/pnas.92.19.8831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ormandy CJ, Camus A, Barra J, Damotte D, Lucas B, Buteau H, Edery M, Brousse N, Babinet C, Binart N, Kelly PA. Null mutation of the prolactin receptor gene produces multiple reproductive defects in the mouse. Genes Dev. 1997;11:167–178. doi: 10.1101/gad.11.2.167. [DOI] [PubMed] [Google Scholar]
- Teglund S, McKay C, Schuetz E, van Deursen JM, Stravopodis D, Wang D, Brown M, Bodner S, Grosveld G, Ihle JN. Stat5a and Stat5b proteins have essential and nonessential, or redundant, roles in cytokine responses. Cell. 1998;93:841–850. doi: 10.1016/s0092-8674(00)81444-0. [DOI] [PubMed] [Google Scholar]
- Robinson GW, Hennighausen L, Johnson PF. Side-branching in the mammary gland: the progesterone-Wnt connection. Genes Dev. 2000;14:889–894. [PubMed] [Google Scholar]
- DeOme KB, Faulkin LJ, Jr, Bern HA, Blair PB. Development of mammary tumors from hyperplastic alveolar nodules transplanted into gland-free mammary fat pads of female C3H mice. Cancer Res. 1959;19:511–520. [PubMed] [Google Scholar]
- Brisken C, Kaur S, Chavarria T, Binart N, Sutherland R, Weinberg R, Kelly P, Ormandy C. Prolactin controls mammary gland development via direct and indirect mechanisms. Dev Biol. 1999;210:96–106. doi: 10.1006/dbio.1999.9271. [DOI] [PubMed] [Google Scholar]
- Miyoshi K, Shillingford JM, Smith GH, Grimm SL, Wagner KU, Oka T, Rosen JM, Robinson GW, Hennighausen L. Signal transducer and activator of transcription (Stat) 5 controls the proliferation and differentiation of mammary alveolar epithelium. J Cell Biol. 2001;155:531–542. doi: 10.1083/jcb.200107065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brisken C, Heineman A, Chavarria T, Elenbaas B, Tan J, Dey S, McMahon J, McMahon A, Weinberg R. Essential function of Wnt-4 in mammary gland development downstream of progesterone signaling. Genes Dev. 2000;14:650–654. [PMC free article] [PubMed] [Google Scholar]
- Kiesling M, Woldman I, Moriggl R, Hofmann J, Marine J-C, Ihle JN, Beug H, Decker T. Antiapoptotic activity of Stat5 required during terminal stages of myeloid differentiation. Genes Dev. 2000;14:232–244. [PMC free article] [PubMed] [Google Scholar]
- Gallego MI, Binart N, Robinson GW, Okagaki R, Coschigano KT, Perry J, Kopchick JJ, Oka T, Kelly PA, Hennighausen L. Prolactin, growth hormone, and epidermal growth factor activate Stat5 in different compartments of mammary tissue and exert different and overlapping developmental effect. Dev Biol. 2001;229:163–175. doi: 10.1006/dbio.2000.9961. [DOI] [PubMed] [Google Scholar]
- Jones FE, Welte T, Fu X-Y, Stern DF. ErbB4 signaling in the mammary gland is required for lobuloalveolar development and Stat5 activation during lactation. J Cell Biol. 1999;147:77–88. doi: 10.1083/jcb.147.1.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cacalano NA, Sanden D, Johnston JA. Tyrosine-phosphorylated SOCS-3 inhibits STAT activation but binds to p120 RasGAP and activates Ras. Nat Cell Biol. 2001;3:460–465. doi: 10.1038/35074525. [DOI] [PubMed] [Google Scholar]
- Tonko-Geymayer S, Goupille O, Tonko M, Soratroi C, Yoshimura A, Streuli C, Ziemiecki A, Kofler R, Doppler W. Regulation and function of the cytokine-inducible SH-2 domain proteins, CIS and SOCS3, in mammary epithelial cells. Mol Endocrinol. 2002;16:1680–1695. doi: 10.1210/mend.16.7.0872. [DOI] [PubMed] [Google Scholar]
- Haslam SZ, Shyamala G. Effect of oestradiol on progesterone receptors in normal mammary glands and its relationship with lactation. Biochem J. 1979;182:127–131. doi: 10.1042/bj1820127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizoguchi Y, Kim JY, Enami J, Sakai S. The regulation of the prolactin receptor gene expression in the mammary gland of early pregnant mouse. Endocr J. 1997;44:53–58. doi: 10.1507/endocrj.44.53. [DOI] [PubMed] [Google Scholar]
- Gibori G, Richards JS, Keyes PL. Synergistic effects of pro-lactin and estradiol in the luteotropic process in the pregnant rat: regulation of estradiol receptor by prolactin. Biol Reprod. 1979;21:419–423. doi: 10.1095/biolreprod21.2.419. [DOI] [PubMed] [Google Scholar]
- Telleria CM, Zhong L, Deb S, Srivastava RK, Park KS, Sugino N, Park-Sarge OK, Gibori G. Differential expression of the estrogen receptors alpha and beta in the rat corpus luteum of pregnancy: regulation by prolactin and placental lactogens. Endocrinology. 1998;139:2432–2442. doi: 10.1210/endo.139.5.5974. [DOI] [PubMed] [Google Scholar]
- Edery M, Imagawa W, Larson L, Nandi S. Regulation of estrogen and progesterone receptor levels in mouse mammary epithelial cells grown in serum-free collagen gel cultures. Endocrinology. 1985;116:105–112. doi: 10.1210/endo-116-1-105. [DOI] [PubMed] [Google Scholar]


