Abstract
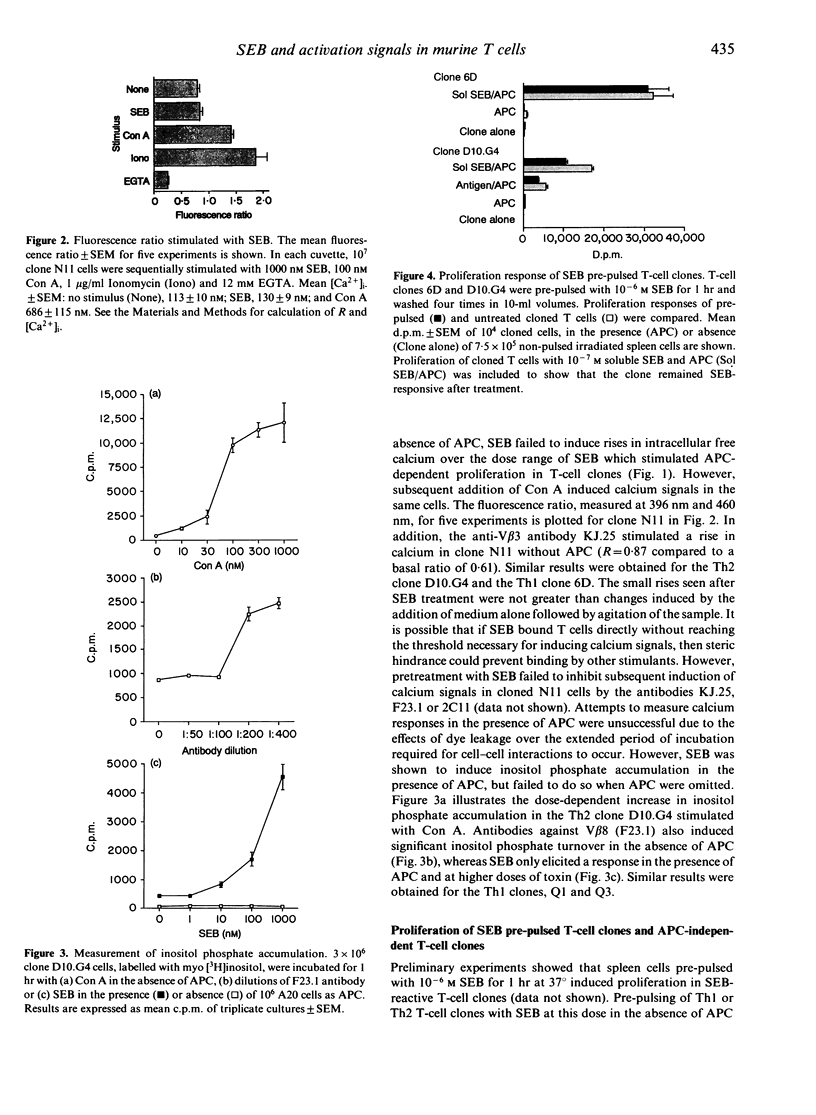
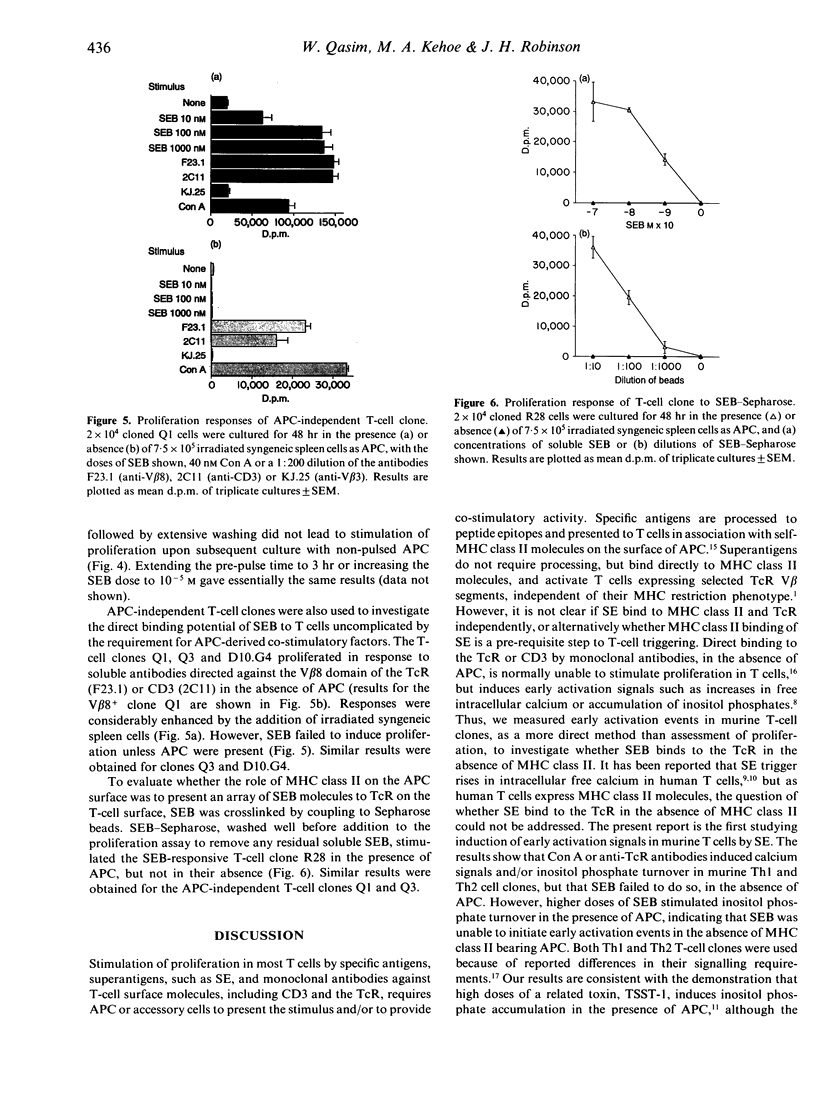
We have investigated the binding potential of staphylococcal enterotoxin B (SEB) to murine T cells using the induction of early activation events in Th1 and Th2 T-cell clones in the absence of antigen-presenting cells (APC) as indicators of direct interactions between SEB and the T-cell receptor (TcR). We consistently found that concanavalin A (Con A) induced rises in intracellular free calcium as well as inositol phosphate accumulation in APC-free T-cell clones. However, SEB uniformly failed to induce either calcium fluxes or inositol phosphate turnover in Th1 and Th2 T-cell clones in the absence of APC. In addition, we have used proliferation assays to show that (i) T-cell clones prepulsed with SEB did not respond when APC were added, (ii) APC-independent T-cell clones responded to soluble anti-TcR antibodies but not to SEB in the absence of APC, and (iii) SEB coupled to Sepharose beads did not stimulate T-cell clones in the absence of APC. Taken together our results argue against SEB binding to the TcR without the participation of MHC class II molecules.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berridge M. J., Dawson R. M., Downes C. P., Heslop J. P., Irvine R. F. Changes in the levels of inositol phosphates after agonist-dependent hydrolysis of membrane phosphoinositides. Biochem J. 1983 May 15;212(2):473–482. doi: 10.1042/bj2120473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bijsterbosch M. K., Rigley K. P., Klaus G. G. Cross-linking of surface immunoglobulin on B lymphocytes induces both intracellular Ca2+ release and Ca2+ influx: analysis with indo-1. Biochem Biophys Res Commun. 1986 May 29;137(1):500–506. doi: 10.1016/0006-291x(86)91238-6. [DOI] [PubMed] [Google Scholar]
- Fleischer B., Schrezenmeier H., Conradt P. T lymphocyte activation by staphylococcal enterotoxins: role of class II molecules and T cell surface structures. Cell Immunol. 1989 Apr 15;120(1):92–101. doi: 10.1016/0008-8749(89)90177-9. [DOI] [PubMed] [Google Scholar]
- Fleischer B., Schrezenmeier H. T cell stimulation by staphylococcal enterotoxins. Clonally variable response and requirement for major histocompatibility complex class II molecules on accessory or target cells. J Exp Med. 1988 May 1;167(5):1697–1707. doi: 10.1084/jem.167.5.1697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fraser J. D. High-affinity binding of staphylococcal enterotoxins A and B to HLA-DR. Nature. 1989 May 18;339(6221):221–223. doi: 10.1038/339221a0. [DOI] [PubMed] [Google Scholar]
- Gajewski T. F., Schell S. R., Nau G., Fitch F. W. Regulation of T-cell activation: differences among T-cell subsets. Immunol Rev. 1989 Oct;111:79–110. doi: 10.1111/j.1600-065x.1989.tb00543.x. [DOI] [PubMed] [Google Scholar]
- Gascoigne N. R., Ames K. T. Direct binding of secreted T-cell receptor beta chain to superantigen associated with class II major histocompatibility complex protein. Proc Natl Acad Sci U S A. 1991 Jan 15;88(2):613–616. doi: 10.1073/pnas.88.2.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grynkiewicz G., Poenie M., Tsien R. Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985 Mar 25;260(6):3440–3450. [PubMed] [Google Scholar]
- Janeway C. A., Jr, Yagi J., Conrad P. J., Katz M. E., Jones B., Vroegop S., Buxser S. T-cell responses to Mls and to bacterial proteins that mimic its behavior. Immunol Rev. 1989 Feb;107:61–88. doi: 10.1111/j.1600-065x.1989.tb00003.x. [DOI] [PubMed] [Google Scholar]
- Marrack P., Kappler J. The staphylococcal enterotoxins and their relatives. Science. 1990 May 11;248(4956):705–711. doi: 10.1126/science.2185544. [DOI] [PubMed] [Google Scholar]
- Meuer S. C., Hodgdon J. C., Hussey R. E., Protentis J. P., Schlossman S. F., Reinherz E. L. Antigen-like effects of monoclonal antibodies directed at receptors on human T cell clones. J Exp Med. 1983 Sep 1;158(3):988–993. doi: 10.1084/jem.158.3.988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mueller D. L., Jenkins M. K., Schwartz R. H. Clonal expansion versus functional clonal inactivation: a costimulatory signalling pathway determines the outcome of T cell antigen receptor occupancy. Annu Rev Immunol. 1989;7:445–480. doi: 10.1146/annurev.iy.07.040189.002305. [DOI] [PubMed] [Google Scholar]
- Norton S. D., Schlievert P. M., Novick R. P., Jenkins M. K. Molecular requirements for T cell activation by the staphylococcal toxic shock syndrome toxin-1. J Immunol. 1990 Mar 15;144(6):2089–2095. [PubMed] [Google Scholar]
- Scholl P. R., Diez A., Karr R., Sekaly R. P., Trowsdale J., Geha R. S. Effect of isotypes and allelic polymorphism on the binding of staphylococcal exotoxins to MHC class II molecules. J Immunol. 1990 Jan 1;144(1):226–230. [PubMed] [Google Scholar]
- Schwartz R. H. T-lymphocyte recognition of antigen in association with gene products of the major histocompatibility complex. Annu Rev Immunol. 1985;3:237–261. doi: 10.1146/annurev.iy.03.040185.001321. [DOI] [PubMed] [Google Scholar]
- Yagi J., Baron J., Buxser S., Janeway C. A., Jr Bacterial proteins that mediate the association of a defined subset of T cell receptor:CD4 complexes with class II MHC. J Immunol. 1990 Feb 1;144(3):892–901. [PubMed] [Google Scholar]




