Abstract
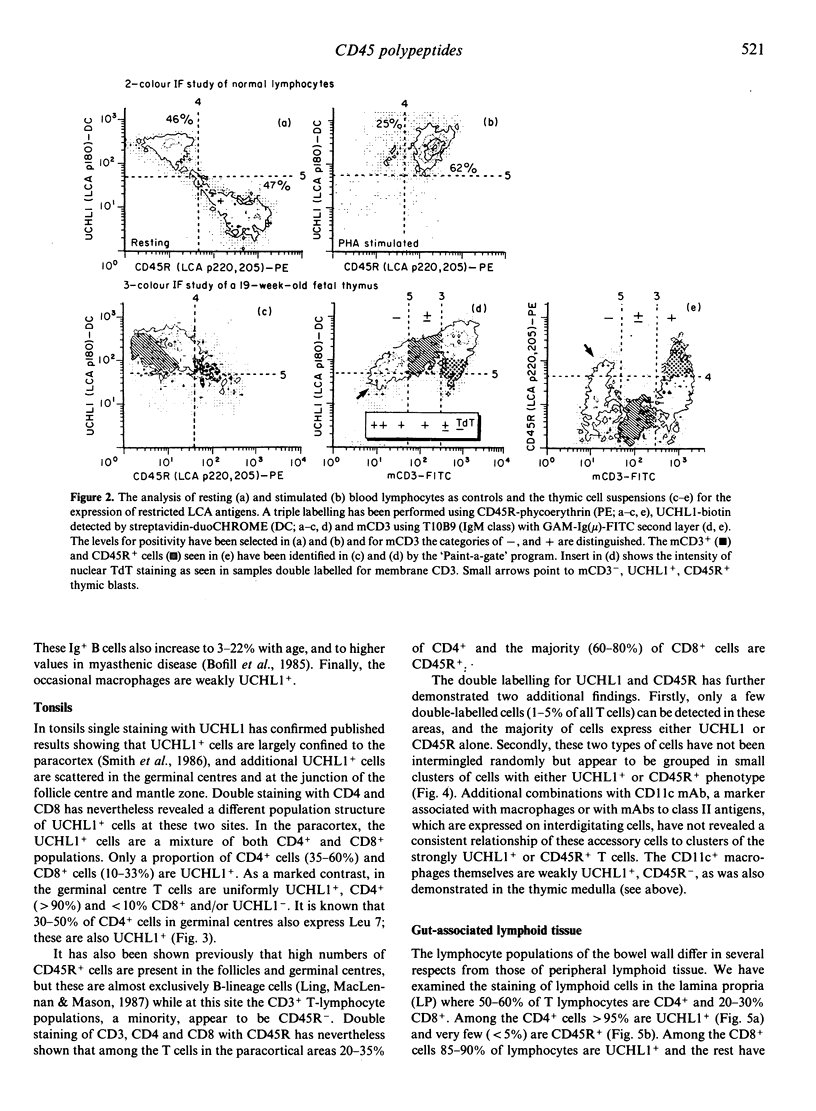
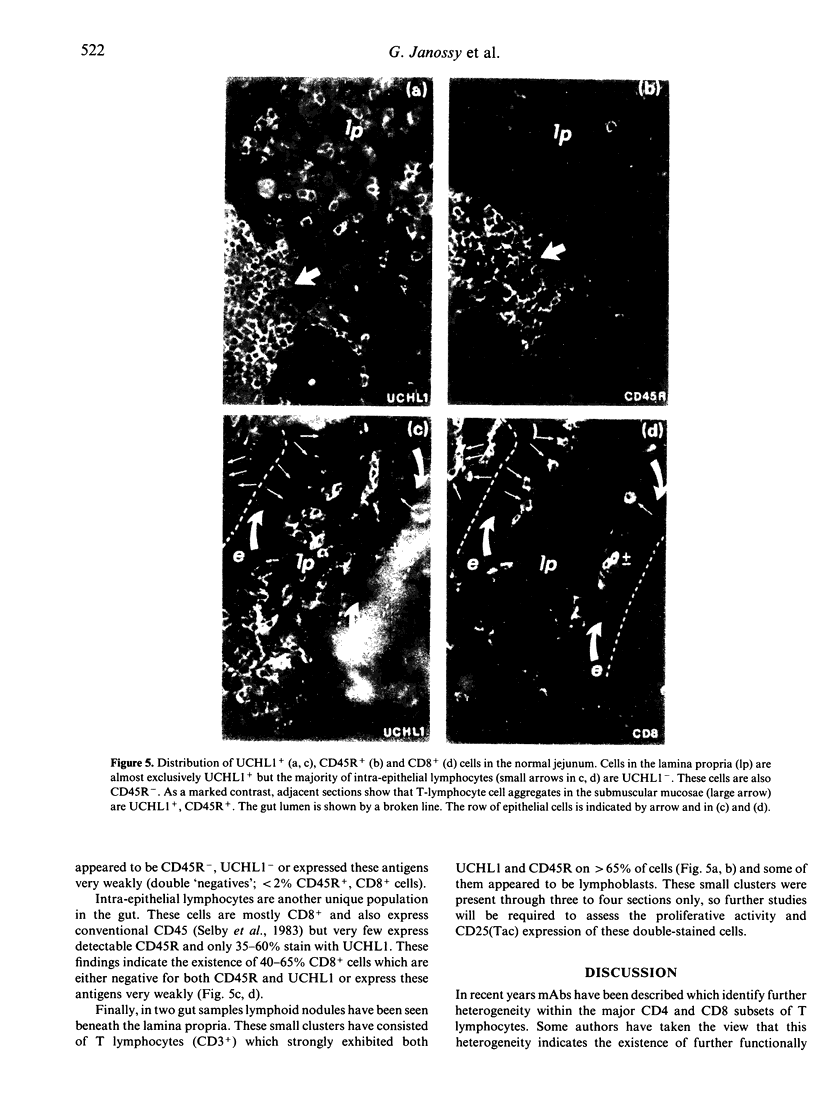
The distribution of T lymphocytes expressing the different polypeptides of the leucocyte common antigen (LCA) family detected by CD45R and UCHL1 antibodies has been studied in normal lymphoid tissues. In the thymus most cortical thymocytes express UCHL1 and co-express CD4 and CD8. The more mature membrane CD3+ (mainly medullary) T cells are heterogenous and may express both UCHL1 and CD45R weakly or be restricted to display CD45R or UCHL1 alone. In the medulla both the CD45R+ and UCHL1+ subpopulations contain single positive CD4 and CD8 cells. In tonsils, germinal centre T cells are almost exclusively UCHL1+, CD4+ and a proportion also express HNK-1 (Leu 7) antigen. In the paracortical areas approximately equal numbers of CD45R+ and UCHL1+ cells are found but these separately occupy nests of cells containing one or the other type. Again, both CD45R+ and UCHL1+ cells include single CD4+ and CD8+ lymphocytes. A small proportion (less than 5%) of strongly CD45R+, UCHL1+ double-stained cells are also seen, and these probably represent recently activated lymphocytes. In the gut, small clusters of such strongly double-labelled cells are in the submuscular mucosae while cells of the lamina propria are almost exclusively UCHL1+. Many intra-epithelial lymphocytes are only weakly positive or negative for UCHL1 and appear to be CD45R-. These results are consistent with the view that expression of different CD45 polypeptides identifies successive stages of thymocyte-T-cell maturation and that following their thymic education, unprimed T lymphocytes are CD45R+, while primed memory T cells are UCHL1+. These populations occupy different microenvironments.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akbar A. N., Terry L., Timms A., Beverley P. C., Janossy G. Loss of CD45R and gain of UCHL1 reactivity is a feature of primed T cells. J Immunol. 1988 Apr 1;140(7):2171–2178. [PubMed] [Google Scholar]
- Akbar A. N., Timms A., Janossy G. Cellular events during memory T-cell activation in vitro: the UCHL1 (180,000 MW) determinant is newly synthesized after mitosis. Immunology. 1989 Feb;66(2):213–218. [PMC free article] [PubMed] [Google Scholar]
- Arthur R. P., Mason D. T cells that help B cell responses to soluble antigen are distinguishable from those producing interleukin 2 on mitogenic or allogeneic stimulation. J Exp Med. 1986 Apr 1;163(4):774–786. doi: 10.1084/jem.163.4.774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beverley P. C. Human T cell subsets. Immunol Lett. 1987 Apr;14(4):263–267. doi: 10.1016/0165-2478(87)90001-0. [DOI] [PubMed] [Google Scholar]
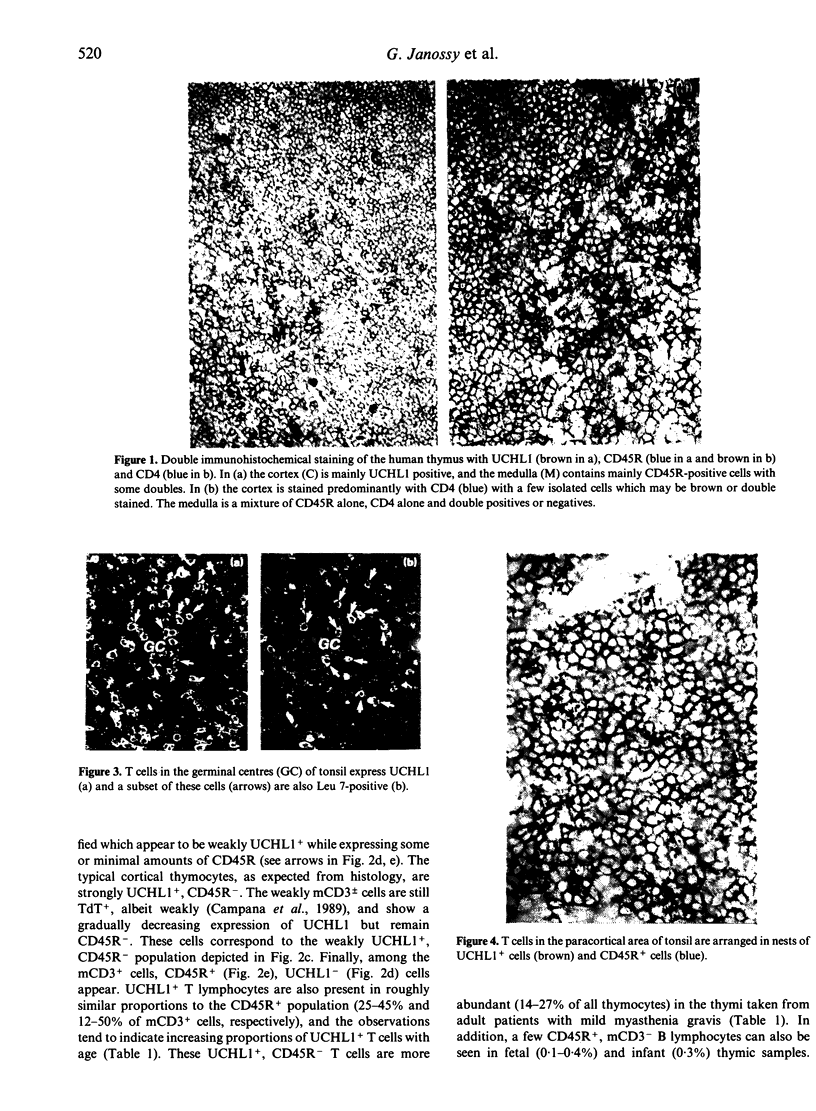
- Bofill M., Janossy G., Willcox N., Chilosi M., Trejdosiewicz L. K., Newsom-Davis J. Microenvironments in the normal thymus and the thymus in myasthenia gravis. Am J Pathol. 1985 Jun;119(3):462–473. [PMC free article] [PubMed] [Google Scholar]
- Campana D., Coustan-Smith E., Janossy G. Double and triple staining methods for studying the proliferative activity of human B and T lymphoid cells. J Immunol Methods. 1988 Feb 24;107(1):79–88. doi: 10.1016/0022-1759(88)90012-9. [DOI] [PubMed] [Google Scholar]
- Corte G., Mingari M. C., Moretta A., Damiani G., Moretta L., Bargellesi A. Human T cell subpopulations defined by a monoclonal antibody. I. A small subset is responsible for proliferation to allogeneic cells or to soluble antigens and for helper activity for B cell differentiation. J Immunol. 1982 Jan;128(1):16–19. [PubMed] [Google Scholar]
- Doyle C., Strominger J. L. Interaction between CD4 and class II MHC molecules mediates cell adhesion. Nature. 1987 Nov 19;330(6145):256–259. doi: 10.1038/330256a0. [DOI] [PubMed] [Google Scholar]
- Hogg N., Takacs L., Palmer D. G., Selvendran Y., Allen C. The p150,95 molecule is a marker of human mononuclear phagocytes: comparison with expression of class II molecules. Eur J Immunol. 1986 Mar;16(3):240–248. doi: 10.1002/eji.1830160306. [DOI] [PubMed] [Google Scholar]
- Isaacson P. G., Norton A. J., Addis B. J. The human thymus contains a novel population of B lymphocytes. Lancet. 1987 Dec 26;2(8574):1488–1491. doi: 10.1016/s0140-6736(87)92622-5. [DOI] [PubMed] [Google Scholar]
- Kincade P. W., Lee G., Watanabe T., Sun L., Scheid M. P. Antigens displayed on murine B lymphocyte precursors. J Immunol. 1981 Dec;127(6):2262–2268. [PubMed] [Google Scholar]
- Morimoto C., Letvin N. L., Boyd A. W., Hagan M., Brown H. M., Kornacki M. M., Schlossman S. F. The isolation and characterization of the human helper inducer T cell subset. J Immunol. 1985 Jun;134(6):3762–3769. [PubMed] [Google Scholar]
- Morimoto C., Letvin N. L., Distaso J. A., Aldrich W. R., Schlossman S. F. The isolation and characterization of the human suppressor inducer T cell subset. J Immunol. 1985 Mar;134(3):1508–1515. [PubMed] [Google Scholar]
- Neiburger J. B., Neiburger R. G., Richardson S. T., Grosfeld J. L., Baehner R. L. Distribution of T and B lymphocytes in lymphoid tissue of infants and children. Infect Immun. 1976 Jul;14(1):118–121. doi: 10.1128/iai.14.1.118-121.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powrie F., Mason D. Phenotypic and functional heterogeneity of CD4+ T cells. Immunol Today. 1988 Sep;9(9):274–277. doi: 10.1016/0167-5699(88)91309-6. [DOI] [PubMed] [Google Scholar]
- Ralph S. J., Thomas M. L., Morton C. C., Trowbridge I. S. Structural variants of human T200 glycoprotein (leukocyte-common antigen). EMBO J. 1987 May;6(5):1251–1257. doi: 10.1002/j.1460-2075.1987.tb02361.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanders M. E., Makgoba M. W., Sharrow S. O., Stephany D., Springer T. A., Young H. A., Shaw S. Human memory T lymphocytes express increased levels of three cell adhesion molecules (LFA-3, CD2, and LFA-1) and three other molecules (UCHL1, CDw29, and Pgp-1) and have enhanced IFN-gamma production. J Immunol. 1988 Mar 1;140(5):1401–1407. [PubMed] [Google Scholar]
- Selby W. S., Janossy G., Bofill M., Jewell D. P. Lymphocyte subpopulations in the human small intestine. The findings in normal mucosa and in the mucosa of patients with adult coeliac disease. Clin Exp Immunol. 1983 Apr;52(1):219–228. [PMC free article] [PubMed] [Google Scholar]
- Serra H. M., Krowka J. F., Ledbetter J. A., Pilarski L. M. Loss of CD45R (Lp220) represents a post-thymic T cell differentiation event. J Immunol. 1988 Mar 1;140(5):1435–1441. [PubMed] [Google Scholar]
- Tedder T. F., Cooper M. D., Clement L. T. Human lymphocyte differentiation antigens HB-10 and HB-11. II. Differential production of B cell growth and differentiation factors by distinct helper T cell subpopulations. J Immunol. 1985 May;134(5):2989–2994. [PubMed] [Google Scholar]
- Terry L. A., Brown M. H., Beverley P. C. The monoclonal antibody, UCHL1, recognizes a 180,000 MW component of the human leucocyte-common antigen, CD45. Immunology. 1988 Jun;64(2):331–336. [PMC free article] [PubMed] [Google Scholar]
- Thomas M. L., Lefrançois L. Differential expression of the leucocyte-common antigen family. Immunol Today. 1988 Oct;9(10):320–326. doi: 10.1016/0167-5699(88)91326-6. [DOI] [PubMed] [Google Scholar]