Abstract
The biochemical basis of life-history tradeoffs is a poorly studied aspect of life-history evolution. We used radiotracer and endocrine approaches to investigate the extent to which morphs of a wing-polymorphic insect differ in the biosynthesis of lipid classes important for dispersal capability vs. reproduction (ovarian growth). The flight-capable genotype of Gryllus firmus biosynthesized a greater amount of total lipid and triglyceride (main flight fuel), which was preferentially allocated to somatic tissue during early adulthood. By contrast, the flightless genotype biosynthesized a significantly greater amount of phospholipid (important in egg development), which was preferentially allocated to ovaries. Topical application of a juvenile-hormone mimic to the flight-capable morph caused it to express all aspects of lipid metabolism seen in the flightless morph. Differences in biosynthesis between morphs (i) occur coincident with 100–400% greater ovarian growth in the flightless morph, (ii) result from alterations of both de novo biosynthesis of fatty acid and downstream partitioning of fatty acids into triglyceride vs. phospholipid, and (iii) possibly result from genetically polymorphic hormonal regulators with negative pleiotropic effects on lipid biosynthesis and ovarian growth. The present study provides direct documentation of genetically based alterations of in vivo flux through pathways of intermediary metabolism leading to the differential production of end products central to the specialization of phenotypes for alternate life histories.
Keywords: life history, biosynthesis, intermediary metabolism, juvenile hormone
Many life-history traits such as longevity and early-age reproduction are negatively associated with each other (1–3). During the past four decades, numerous studies have attempted to identify the functional causes of these tradeoffs, which play a central role in life-history evolution (4–7). Although most work has focused on the differential allocation of internal resources to competing life-history traits, the specific mechanisms underlying tradeoffs at the physiological level remain poorly understood (6, 7), and biochemical (metabolic) aspects of internal tradeoffs have rarely been investigated. This lack of information constitutes a major roadblock to attaining a deep understanding of the mechanisms of life-history evolution (7).
Accumulation of lipid reserves seems to play a pivotal role in many life-history tradeoffs (7–11). In insects, lipid is typically more abundant in individuals adapted to live longer or to withstand stressful or energy-demanding conditions such as starvation or long-duration flight. By contrast, lipid is reduced in individuals with enhanced early-age fecundity or faster rate of juvenile development, traits that often tradeoff with the life-history traits mentioned above (7, 10, 12–16). These associations suggest that the evolution of many life-history traits requires modifications of lipid metabolism to increase energy reserves, and that these modifications, in turn, negatively impact reproductive output or other fitness components. However, the specific mechanisms involved are not well understood.
Recent in vitro enzymological studies suggest that increased lipid reserves in life-history phenotypes result from increased lipid biosynthesis (17–19). However, it is not possible to draw strong inferences about in vivo processes solely from in vitro investigations (20, 21). Ultimately, direct in vivo studies are required to establish a strong causal link between variation in some aspect of metabolism and a life-history tradeoff. Such in vivo studies are rare (19, 22, 23), and only one phenotypic study has reported on variation in any aspect of lipid metabolism in relation to a life-history tradeoff (19). In short, intermediary metabolism largely remains a black box with respect to life-history evolution.
Wing polymorphism in insects has emerged recently as a powerful experimental model in functional studies of life-history tradeoffs (7, 24). Gryllus firmus, one of the most intensively studied species in this regard, exists in natural populations as two genetically specified morphs. One morph is capable of flight but delays ovarian growth, whereas the other morph is flightless but exhibits enhanced (100–400%) ovarian growth. The flight-capable morph contains a substantially greater amount of total lipid and triglyceride [the main flight fuel of this species (25)] than the flightless morph, whereas the reproductive morph contains a greater amount of phospholipid, a lipid class that is an important component of eggs (26). These data suggest that key aspects of morph specialization may involve not only genetically based modifications of total lipid production but also tradeoffs in the production of specific lipid classes important in flight capability vs. egg production.
In the present study we directly quantified in vivo rates of triglyceride and phospholipid biosynthesis and the allocation of these lipid components to somatic and reproductive organs in flight-capable and flightless morphs of G. firmus. Because of the importance of genetic information in evolutionary studies, biosynthetic studies were undertaken in replicate lines selected to produce the flight-capable or flightless morphs. Finally, we used endocrine manipulation to determine whether differences between morphs in lipid biosynthesis might result from morph-specific differences in hormonal regulation.
Materials and Methods
Backgound on G. firmus: Morphs, Selected Stocks, and Rearing Conditions.
G. firmus exists in natural populations as an obligately flightless short-winged (SW) morph or as a long-winged (LW) morph, some of which can fly (27, 28). All LW females have fully developed (pink) flight muscles at or shortly after the adult molt [denoted LW(f)]. Some LW(f) females subsequently degenerate their flight muscles and become flightless. In artificially selected lines used in the present study, >95% of LW females retained their flight muscles, and only these LW(f) individuals are considered here. Ovarian growth in SW females is 100–400% greater than in the LW(f) females during the first week of adulthood (29).
The G. firmus females used in the present study were taken from lines artificially selected for wing morph during the 18th generation of selection. Lines were grouped into three blocks (independent selection trials), each of which contained a line selected for the LW morph, a line selected for the SW morph, and a control line (see figure 1 of ref. 29). Only selected lines are considered here, which are close to being pure breeding (i.e., they produce >90% of the selected morph). Only LW(f) females from the LW lines and SW females from the SW lines were studied. All lines were raised under the same environmental conditions (e.g., 28°C under a 16-h light/8-h dark photoperiod; see ref. 29 for additional details). Eight to 12 newly molted (within 24 h) females and an equal number of males were placed in 3-gallon plastic boxes without oviposition material. Essentially no oviposition occurs during the first 5 days of adulthood (19, 29), the period investigated in the present study. LW(f) individuals from the LW lines and SW individuals from the SW lines differ genetically in the size of wings and the masses of flight muscles, ovaries, and total lipid and triglyceride (16, 29).
Radiotracer studies were conducted on days 0 and 5 of adulthood. These days bracket the period during which there is a substantially greater accumulation of total lipid and triglyceride in LW(f) vs. SW females but substantially lower ovarian growth in the LW(f) morph (16, 19). Lipid biosynthesis was compared between morphs fed one of three diets (standard, low-nutrient, and high-sucrose) that had been used in prior studies and are known to influence lipid metabolism (see ref. 16 for diet composition and rational for use of these specific diets). This was done to identify general differences in lipid metabolism between morphs that were not contingent on a specific diet as well as metabolic aspects dependent on specific diets.
Quantification of Lipid Biosynthesis Using [1-14C]Acetate and [1-14C]Palmitate.
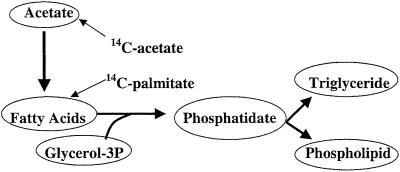
Rates of lipid biosynthesis were quantified in flight-capable and flightless morphs by standard techniques in which the amount of radiolabeled sodium acetate incorporated into various lipid classes during a given period was measured (30). This technique measures both the (de novo) biosynthesis of fatty acid and the incorporation of fatty acid into various lipid classes (Fig. 1). Two microliters of sterile saline (0.9% NaCl) containing ≈6 × 105 dpm of sodium-[1-14C]acetate [2.0 GBq/mmol, 54.7 mCi/mmol (1 Ci = 37 GBq), New England Nuclear] were injected into the abdominal hemocoel of adult female crickets that had been unfed for 4 h before injection. After injection, crickets were kept singly in containers with a small piece of artificial diet for 7 h at 28°C. Incorporation of radiolabel into total lipid, triglyceride, or phospholipid is linear up to 8 h after injection (r2 > 0.89 in all linear regressions). After incubation, crickets were homogenized in 2:1 chloroform/methanol (CHCl3/MeOH), and lipids were extracted by using standard protocols (31) as described in ref. 19. Greater than 90% of total lipid in adult G. firmus females consists of triglyceride and phospholipid (16), as is the case for other crickets (26). Background experiments verified that >95% of the total radiolabel incorporated into lipid was due to incorporation into triglyceride and phospholipid. These two lipid classes were separated cleanly by elution from silica gel columns (0.8 cm3) by using chloroform/methanol solutions of increasing methanol concentration. The identity of eluted lipid classes was verified by standard TLC using triglyceride and phospholipid standards (31). Solvent was removed, and radioactivity (dpm) was quantified by liquid-scintillation spectrometry.
Fig 1.
Simplified biosynthetic pathway from acetate to triglyceride and phospholipid, indicating where radiolabeled sodium acetate and palmitic acid enter the pathway.
As an independent measure of lipid biosynthesis by morphs of G. firmus, the aforementioned experiments were repeated by using [1-14C]palmitic acid, which enters the pathway of lipid biosynthesis at a different point than acetate, after the de novo biosynthesis of fatty acid but before the partitioning of fatty acids into triglyceride and phospholipid (Fig. 1). [1-14C]Palmitic acid [1 × 106 dpm (2.1 GBq/mmol, 56 mCi/mmol)] was injected in 2 μl of PBS (pH 7.0). Crickets were incubated for 4 h, because incorporation of [1-14C]palmitate into lipid was nonlinear after that time. The column-chromatographic method described above did not cleanly separate fatty acids (containing unincorporated [1-14C]palmitate) from radiolabeled triglycerides. Hence biosynthesized triglyceride, phospholipid, and unincorporated palmitic acid were separated by TLC (silica gel plates sequentially eluted with a solution of 65% CHCl3/30% MeOH/5% H20 followed by a solution of 78% hexane/20% petroleum ether/2% acetic acid; see ref. 31). Zones of the TLC plate corresponding to triglyceride and phospholipid were cut out, and dpm were determined by liquid-scintillation spectrometry. Background studies documented that zones of the TLC plate corresponding to biosynthesized lipids other than triglyceride or phospholipid (e.g., cholesterol esters and mono- and diglyceride) contained <5% of total lipid dpm.
Induction of the SW Phenocopy by Hormone Manipulation.
Ten micrograms of the juvenile-hormone (JH) agonist, methoprene, in 2 μl of acetone was applied to the abdomen of LW(f) female G. firmus on days 1 and 3 of adulthood (day 0 = day of molt). This concentration of methoprene causes the expression of SW morphological and reproductive phenotypes (e.g., enhanced ovarian growth) when applied to LW(f) female Gryllus (29). The lipid biosynthetic studies described above were performed on hormone-treated and control LW(f) crickets.
Statistics.
Absolute differences between morphs in incorporation of radiolabel into various lipid classes was assessed by ANOVA, whereas proportional incorporation into triglyceride vs. phospholipid was determined by analysis of covariance (ANCOVA), where the independent variable was dpm of the lipid class in question, and the covariate was total lipid dpm. In preliminary tests, morph × diet interactions did not differ significantly (P > 0.1) for any biosynthetic study, indicating that differences of similar magnitude were observed between the morphs on the three diets. Thus, only data pooled across the diet treatments are reported here. Genetic differences between morphs in absolute or relative lipid levels should be manifest as consistent differences between means of LW(f)- and SW-selected lines across blocks, which were assessed by paired t tests (29, 32).
Results
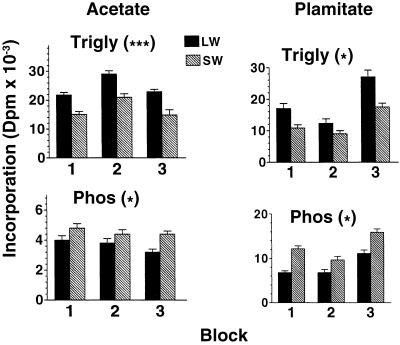
Incorporation of radiolabeled acetate into total lipid did not differ between morphs on the day of molt to adulthood (day 0) but was significantly higher in LW(f) vs. SW females within each block and across blocks on day 5 (Table 1). These results are consistent with a genetically based elevation in the rate of de novo lipid biosynthesis from acetate in the LW(f) vs. SW morphs during the first 5 days of adulthood. The higher overall rate of lipid biosynthesis was primarily due to the higher rate of triglyceride biosynthesis in the LW(f) morph. Incorporation of radiolabeled acetate into triglyceride was significantly higher in LW(f) vs. SW females within each of the three blocks on day 5 (P < 0.005 in each of three ANOVAs), whereas mean incorporation was higher in LW(f) vs. SW females across the three blocks (paired t test, t(2) = 16.94, P < 0.005; Fig. 2). By contrast, mean [1-14C]acetate incorporation into phospholipid, the other major lipid component in G. firmus, was significantly lower in LW(f) vs. SW females across blocks on day 5 (paired t test, t(2) = 4.61, P < 0.05; Fig. 2), although values did not differ significantly within any block (P > 0.1 in all ANOVAs). Similar results were obtained when dpm incorporated into lipid classes were analyzed by ANCOVA with total cricket wet mass as the covariate (data not shown). Similar to the situation for total lipid, no difference in either triglyceride or phospholipid was seen between morphs in any block or across blocks on day 0 (see Fig. 2 legend).
Table 1.
Rate of total lipid biosynthesis in flight-capable [LW(f)] and flightless (SW) morphs of G. firmus on the day of adult molt and day 5 of adulthood
| Morph
|
Block | Results of paired t tests of morph means
|
||
|---|---|---|---|---|
| 1 | 2 | 3 | ||
| Day 0 (day of molt to adult) | ||||
| LW (f) | 9,310 ± 667, n.s. | 13,801 ± 1,024, n.s. | 14,798 ± 1,407, n.s. | t(2) = 0.80, P > 0.5 |
| SW | 8,805 ± 921 | 12,192 ± 1,102 | 15,392 ± 3,232 | |
| Day 5 | ||||
| LW(f) | 26,120 ± 1,111 | 32,413 ± 1,288 | 26,603 ± 954 | t(2) = 15.92, P < 0.005 |
| SW | 19,812 ± 1,157 | 25,046 ± 1,265 | 18,774 ± 959 | |
Values are mean (±SEM) dpm of [1-14C]acetate incorporated into total lipid over a 7-h period. Means are based on 7–8 (day 0) or 23–27 (day 5) individuals. Results of t tests of incorporation rates between LW(f) and SW morphs within blocks are given after LW(f) means. n.s., Nonsignificant;
, P < 0.005. Block 1 means have been published (19).
Fig 2.
Amount of radiolabeled [1-14C]acetate or [1-14C]palmitic acid incorporated into triglyceride or phospholipid in LW(f) or SW G. firmus on day 5 of adulthood. Trigly, triglyceride; Phos, phospholipid. Block refers to independent selection trial (see Materials and Methods). The asterisks in parentheses indicate results of paired t tests (***, P < 0.005; *, P < 0.05). Values in all panels are means (±SEM) based on 20–28 individuals. On day 0, mean triglyceride or phospholipid dpm (from [1-14C]acetate) did not differ significantly between morphs within any block (P > 0.1 for all ANOVAs) or across blocks (P > 0.1 for paired t tests). Grand mean dpm (means across the three blocks) are as follows: triglyceride, 9,606 ± 1,814 [LW(f)] and 9,328 ± 1,861 (SW); phospholipid, 3,030 ± 139 [LW(f)] and 2,861 ± 188 (SW).
In addition to the genetically based differences in absolute rates of lipid biosynthesis between the LW(f) and SW morphs on day 5, proportional incorporation of radiolabeled [1-14C]acetate into phospholipid vs. triglyceride also differed genetically between morphs on that day. The percentage of radiolabeled acetate incorporated into total lipid that was due to phospholipid was significantly higher in the SW vs. the LW(f) morph both within and across blocks (Table 2). Thus, the LW(f) morph allocates a greater proportion of total biosynthesized fatty acids to triglyceride, whereas the SW morph allocates a greater proportion to phospholipid. Incorporation studies using [1-14C]palmitate, which enters the pathway of lipid biosynthesis at a different point than acetate (Fig. 1), yielded results very similar to those obtained by using [1-14C]acetate. On day 5 of adulthood, absolute and proportional triglyceride biosynthesis was higher in LW(f) females, whereas absolute and proportional phospholipid biosynthesis was higher in SW females (Fig. 2 and Table 2).
Table 2.
Difference between LW(f) and SW G. firmus in percentage biosynthesized lipid that was phospholipid
| Morph
|
Block | Results of paired t test of morph means
|
||
|---|---|---|---|---|
| 1 | 2 | 3 | ||
| Acetate | ||||
| LW(f) | 15.8 ± 0.8 (26)** | 11.7 ± 0.5 (23)** | 13.7 ± 0.8 (24)** | t(2) = 7.38 |
| SW | 24.0 ± 1.3 (23) | 17.6 ± 1.9 (24) | 20.9 ± 1.1 (24) | P < 0.02 |
| Palmitate | ||||
| LW(f) | 30.3 ± 1.7 (20)** | 36.5 ± 2.4 (21)** | 32.3 ± 3.3 (22)** | t(2) = 4.90 |
| SW | 54.7 ± 2.0 (20) | 53.1 ± 2.2 (23) | 48.4 ± 2.4 (23) | P < 0.04 |
t tests were performed on adjusted mean phospholipid dpm derived from ANCOVA of phospholipid dpm with total lipid dpm as the covariate.
Percentages differ between morphs within a block (**, P < 0.005) as determined by ANCOVA of phospholipid dpm (numerator) with total lipid dpm as the covariate (denominator).
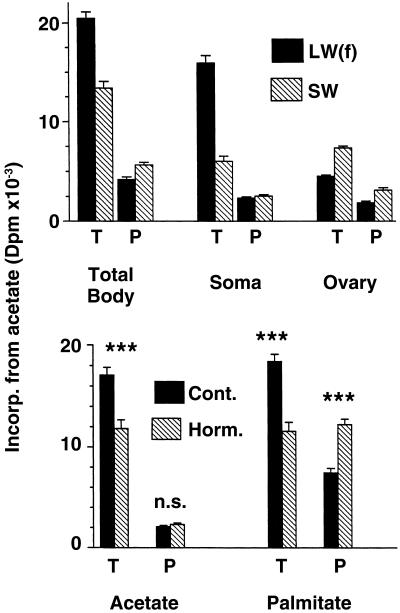
In a separate experiment, morph-specific allocation of newly biosynthesized lipid classes to somatic (body minus ovaries) vs. reproductive (ovarian) tissues was investigated in LW(f) and SW crickets from block 1 on day 5 (Fig. 3). Similar to results presented above, incorporation of [1-14C]acetate into whole-body triglyceride was significantly (53%) greater in the LW(f) vs. SW morph (P < 0.005, ANOVA), whereas incorporation into whole-body phospholipid was significantly (26%) lower in the LW(f) vs. the SW morph (P < 0.005, ANOVA). In addition, the magnitude of differences between morphs in both triglyceride and phospholipid depended on body compartment (Fig. 3). Radiolabeled triglyceride was elevated to a significantly greater degree in the LW(f) vs. the SW morph in somatic (total body minus ovaries) vs. ovarian compartments (ANOVA, interaction between morph and body compartment, P < 0.01). By contrast, radiolabeled phospholipid was elevated to a significantly greater degree in SW than LW(f) morphs in ovarian vs. somatic compartments (ANOVA, interaction between morph and body compartment, P < 0.01). Thus, the LW(f) morph both biosynthesized a greater amount of triglyceride than the SW morph and allocated a greater amount of this lipid type to somatic tissue, whereas the SW morph biosynthesized a greater amount of phospholipid and allocated a greater amount of this lipid type to the ovaries.
Fig 3.
(Upper) Amounts of biosynthesized triglyceride (T) and phospholipid (P) found in both ovaries (Ovary), whole body minus ovaries (Soma), and whole bodies (Total Body = Soma plus Ovary) of morphs of G. firmus from block 1. [1-14C]Acetate was the radiolabel used. (Lower) Effect of methoprene, a JH agonist, on the biosynthesis of triglyceride and phospholipid from [1-14C]acetate or [1-14C]palmitic acid in the LW(f) morph of G. firmus from block 1. Values are means (±SEM) based on 7–8 individuals (Upper) or 22–26 individuals (Lower) measured on day 5 of adulthood.
The JH agonist methoprene strongly effected lipid metabolism (Fig. 3), transforming the LW(f) morph into a SW phenocopy with respect to all aspects of lipid metabolism studied (compare Fig. 2 with 3). Hormone application to LW(f) females on day 5 of adulthood caused a significant reduction in the amount of injected [1-14C]acetate that was incorporated into total lipid [control = 19,177 ± 843 dpm (n = 26), hormone = 14,175 ± 912 dpm (n = 22); P < 0.005, ANOVA]. The reduced total lipid biosynthesis was primarily due to reduced triglyceride biosynthesis (Fig. 3). Absolute incorporation of [1-14C]acetate into phospholipid did not differ significantly between control and hormone-treated crickets (Fig. 3). However, the proportion of total radiolabel incorporated into lipid that was due to phospholipid was substantially higher in hormone-treated [16.9 ± 0.6% (n = 22)] than control crickets [11.2 ± 0.6% (n = 26); results of analysis of covariance (ANCOVA), P < 0.005]. Similar results were obtained when [1-14C]palmitate was used as the radiolabel except that a significantly greater absolute as well as relative amount of radiolabel was incorporated into phospholipid in hormone-treated crickets (Fig. 3). Thus, similar to the situation for unmanipulated morphs from the selected lines, the hormonally induced flightless morph [i.e., methoprene-treated LW(f) crickets] biosynthesized a smaller amount of total lipid than flight-capable (control) crickets but allocated a significantly greater proportion of total lipid biosynthesis to phospholipid.
Discussion
Intermediary metabolism has largely been treated as a black box with respect to the mechanisms of internal resource allocation underlying life-history tradeoffs. Rarely (19, 22, 23) has information been reported on the existence or functional significance of variation in in vivo flux through pathways of intermediary metabolism that underlie life-history tradeoffs. Here we present evidence for a variety of large-magnitude, genetically based alterations of lipid metabolism that contribute significantly to the tradeoff between flight capability and reproduction in the wing-polymorphic cricket, G. firmus.
The LW(f) morph is genetically programmed to biosynthesize a greater amount of total lipid than the SW morph during early adulthood (Table 1). The greater increase in total lipid biosynthesis in the LW(f) vs. SW morphs during the first 5 days of adulthood (Table 1) is correlated with a 40% greater accumulation of total lipid but a 50–75% lower rate of ovarian growth in the LW(f) morph during this time (16). These data suggest that increased biosynthesis and accumulation of total lipid in the LW(f) morph negatively impact ovarian growth and thus are important causes of the genetically based tradeoff between flight capability and ovarian growth in G. firmus. This hypothesis is supported also by the increased ovarian growth that accompanied decreased lipid biosynthesis, when the JH agonist methoprene was applied to the LW(f) morph (discussed below).
The underlying basis of the tradeoff between total lipid biosynthesis and ovarian growth might be due to any of several factors such as limited internal nutrients that necessitate decreased yolk production when an increased amount of nutrients are allocated to lipid biosynthesis. Alternatively, nonenergetic constraints are possible such as limited space in the abdomen, which may not be able to accommodate large ovaries and large lipid stores, or regulation of lipid metabolism by factors that have antagonistic effects on (yolk) protein biosynthesis (discussed below).
The LW(f) morph does not acquire (consume and assimilate) more nutrients or lipid from the diet than the SW morph during the first week of adulthood (ref. 33, and A.J.Z. and T. Rooneem, unpublished data). Thus, the greater accumulation of total lipid in the LW(f) morph must be caused exclusively by morph-specific differences in internal metabolic processes (i.e., an internal tradeoff). To our knowledge, G. firmus is the only model species used in functional studies of genetically based life-history tradeoffs for which both external nutrient acquisition and internal allocation have been quantified. Hence, this is the only species for which strongly supported conclusions can be drawn concerning the extent to which genetically based differences in energy reserves between life-history phenotypes result from an internal tradeoff as opposed to differential resource acquisition, a central issue in life-history tradeoffs (7).
In addition to the tradeoff between total lipid biosynthesis and ovarian growth, we also found a second genetically based tradeoff between lipid classes that are important to flight capability vs. egg production. During early adulthood, the flight-capable LW(f) morph is genetically programmed to biosynthesize both a greater absolute and relative amount of triglyceride (Fig. 2 and Table 2), the major fuel used for flight in this species (25). By contrast, the SW morph biosynthesizes a greater absolute and relative amount of phospholipid than the LW(f) morph (Fig. 2 and Table 2). This lipid component is in high concentration in insect eggs and is used during embryogenesis (26, 34). Thus, morphs of G. firmus differ genetically in two key aspects of lipid biosynthesis. First, the LW(f) morph exhibits greater flux through the pathway of de novo fatty acid biosynthesis (i.e., from acetate to fatty acid; see Fig. 1). This results in a greater production of fatty acids used to construct triglycerides and phospholipid (both of which are primarily composed of fatty acids) and hence a greater production of total lipid in the LW(f) morph (Table 1). Greater de novo biosynthesis of fatty acids in LW(f) vs. SW morphs is independently supported by the substantially higher specific activities of each of five lipogenic enzymes (e.g., fatty acid synthase) in the LW(f) morph on day 5 (19, 24). Second, morphs also differ in more downstream portions of lipid biosynthesis, which determine the degree of partitioning of fatty acids into triglyceride vs. phospholipid (Table 2 and Fig. 1). Differences between the morphs in the biosynthesis of triglyceride and phospholipid seem to be functionally significant. These biosynthetic differences occur during the first week of adulthood, precisely when the morphs first exhibit differences in the concentrations of triglyceride and phospholipid (16).
The LW(f) morph preferentially allocated newly biosynthesized triglyceride to somatic (nonovarian) organs, whereas the SW morph primarily allocated newly biosynthesized phospholipid to the ovaries (Fig. 3). These differing patterns of allocation provide additional support for the hypothesis that increased triglyceride production in the LW(f) morph primarily involves increased lipid allocation to somatic function, whereas increased phospholipid production in the SW morph primarily involves increased allocation to reproduction.
Most previous studies of energy reserves in life-history tradeoffs in insects have measured whole-body lipid, considered as a somatic energy reserve, and have focused on the conflict between lipid accumulation and ovarian growth (6, 12–15). By quantifying individual lipid classes in individual body compartments, we have shown that the relationship between lipid metabolism and life histories is more complex. In some cases, enhanced biosynthesis of a specific lipid type (e.g., triglyceride) seems to impact ovarian growth negatively, whereas enhanced biosynthesis of other types (e.g., phospholipid) seems to be required for increased ovarian growth. Analyses of lipid classes in individual organs also provide a more accurate accounting of the energetic costs of traits linked in a life-history tradeoff. For example, in the studies listed above, triglyceride is considered a somatic energy store. Yet in the SW morph, the majority of triglyceride is found in the ovaries (Fig. 3), where it presumably functions in a reproductive capacity as an energy reserve for oviposited eggs.
Although hormones have long been suspected to be key regulators of life-history tradeoffs, the endocrine control of these tradeoffs remains an understudied aspect of life-history evolution (7, 35). The extent to which tradeoffs result from changes in the hormonal regulation of flux through pathways of metabolism has not been reported previously. An important finding of the present study was the production of a remarkable SW biochemical phenocopy when a JH agonist (methoprene) was applied to the LW(f) morph (see Fig. 3 and Results). That is, methoprene caused the LW(f) morph to express nearly all aspects of lipid biosynthesis seen in the SW morph (compare Fig. 2 with Fig. 3). A previous study showed that this JH agonist also caused enhanced ovarian growth in the LW(f) morph similar to that seen in unmanipulated SW females (29). These results collectively suggest that tradeoffs in G. firmus between overall lipid biosynthesis and ovarian growth, and between the biosynthesis of triglyceride and phospholipid, may be influenced strongly by genetically based variation in endocrine regulation. Numerous studies have shown that JH positively affects ovarian growth but negatively affects total lipid accumulation in insects (30) and thus is predisposed to mediate a tradeoff between these two characters. This hypothesis also suggests that regulatory constraints may be an important underlying cause of tradeoffs that involve the differential accumulation of energy reserves.
Results obtained in the present study constitute direct documentation of genetically based, in vivo differences in flux through pathways of intermediary metabolism that seem to be important components of a life-history tradeoff. Such information is essential to identify the functional causes of life-history tradeoffs that emanate from the structure and regulation of intermediary metabolism (23). Such biochemical information also will be indispensable in assessing the extent and mechanisms by which variation in gene expression is translated into variation in whole-organism physiology and life histories.
Acknowledgments
We thank L. G. Harshman and A. Joern for their comments on an earlier draft of this manuscript. This study was supported by National Science Foundation Grant IBN 9808249.
Abbreviations
SW, short-winged
LW, long-winged
JH, juvenile hormone
This paper was submitted directly (Track II) to the PNAS office.
References
- 1.Reznick D. N. (1985) Oikos 44, 257-267. [Google Scholar]
- 2.Stearns S. C., (1992) The Evolution of Life Histories (Oxford Univ. Press, Oxford).
- 3.Roff D. A., (2002) Life-History Evolution (Sinauer, Sunderland, MA).
- 4.Tinkle D. W. & Hadley, N. F. (1975) Ecology 56, 427-434. [Google Scholar]
- 5.Pianka E. R. (1981) in Physiological Ecology: An Evolutionary Approach to Resource Use, eds. Townsend, C. R. & Calow, P. (Blackwell, Oxford), pp. 300–314.
- 6.Rose M. R. & Bradley, T. J. (1998) Oikos 83, 443-451. [Google Scholar]
- 7.Zera A. J. & Harshman, L. G. (2001) Annu. Rev. Ecol. Syst. 32, 95-126. [Google Scholar]
- 8.Pond C. M. (1981) in Physiological Ecology: An Evolutionary Approach to Resource Use, eds. Towsend, C. R. & Calow, P. (Blackwell, Oxford), pp. 190–219.
- 9.Rogers C. M. & Smith, J. N. M. (1993) Ecology 74, 419-426. [Google Scholar]
- 10.Dingle H., (1996) Migration: The Biology of Life on the Move (Oxford Univ. Press, Oxford).
- 11.Carey C., (1996) Avian Energetics and Nutritional Ecology (Chapman & Hall, New York).
- 12.Service P. M. (1987) Physiol. Zool. 60, 321-326. [Google Scholar]
- 13.Zwann B., Biljsma, R. & Hoekstra, R. F. (1995) Evolution (Lawrence, Kans.) 49, 649-659. [DOI] [PubMed] [Google Scholar]
- 14.Chippindale A. K., Chu, T. J. & Rose, M. R. (1996) Evolution (Lawrence, Kans.) 50, 753-766. [DOI] [PubMed] [Google Scholar]
- 15.Djawdan M., Chippendale, A., Rose, M. & Bradley, T. J. (1998) Physiol. Biochem. Zool. 71, 584-594. [DOI] [PubMed] [Google Scholar]
- 16.Zera A. J. & Larsen, A. (2001) J. Insect Physiol. 47, 1147-1160. [DOI] [PubMed] [Google Scholar]
- 17.Harshman L. G. & Schmidt, J. L. (1998) Evolution (Lawrence, Kans.) 52, 1679-1685. [DOI] [PubMed] [Google Scholar]
- 18.Harshman L. G., Hoffmann, A. A. & Clark, A. G. (1999) J. Evol. Biol. 12, 370-379. [Google Scholar]
- 19.Zhao Z. & Zera, A. J. (2001) J. Insect Physiol. 47, 1337-1347. [DOI] [PubMed] [Google Scholar]
- 20.Kascer H. & Burnes, J. M. (1979) Biochem. Soc. Trans. 7, 1149-1160. [DOI] [PubMed] [Google Scholar]
- 21.Dykhuizen D. E. & Dean, A. M. (1990) Trends Ecol. Evol. 5, 257-262. [DOI] [PubMed] [Google Scholar]
- 22.Boggs C. (1997) Ecology 78, 192-202. [Google Scholar]
- 23.O'Brien D. M., Marilyn, M. L. & Boggs, C. (2002) Proc. Natl. Acad. Sci. USA 99, 4413-4418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zera A. J. & Zhao, Z., (2003) Evolution (Lawrence, Kans.), in press.
- 25.Zera A. J., Sall, J. & Otto, K. (1999) J. Insect Physiol. 45, 275-285. [DOI] [PubMed] [Google Scholar]
- 26.Grapes M., Whiting, P. & Dinan, L. (1989) Insect Biochem. 19, 767-774. [Google Scholar]
- 27.Veazy J. N., Kay, C. A. R., Walker, T. J. & Whitcomb, W. H. (1976) Ann. Entomol. Soc. Am. 69, 374-380. [Google Scholar]
- 28.Zera A. J., Sall, J. & Grudzinski, K. (1997) Physiol. Zool. 70, 519-529. [DOI] [PubMed] [Google Scholar]
- 29.Zera A. J. & Cisper, G. (2001) Physiol. Biochem. Zool. 74, 293-306. [DOI] [PubMed] [Google Scholar]
- 30.Downer R. G. H. (1985) in Comprehensive Insect Physiology, Biochemistry and Pharmacology, eds. Kerkut, G. A. & Gilbert, L. I. (Pergamon, Oxford), Vol. 10, pp. 77–114. [Google Scholar]
- 31.Christie W. W., (1982) Lipid Analysis (Pergamon, Oxford).
- 32.Rose M. R., Nusbaum, T. J. & Chippindale, A. K. (1996) in Adaptation, eds. Rose, M. R. & Lauder, G. V. (Academic, San Diego), pp. 221–241.
- 33.Zera A. J. & Brink, T. (2000) J. Insect Physiol. 46, 1207-1218. [DOI] [PubMed] [Google Scholar]
- 34.Lipsitz E. Y. & McFarlane, J. E. (1970) Comp. Biochem. Physiol. 34, 699-704. [Google Scholar]
- 35.Ketterson E. D. & Nolan, V. (1999) Am. Nat. 154, S4-S25. [DOI] [PubMed] [Google Scholar]