Abstract
MRSA ID was evaluated for its ability to identify methicillin-resistant Staphylococcus aureus. A well-defined collection of staphylococci was used (n = 998). The sensitivity after 24 h was 96.4%, increasing to 98.8% after 48 h. The specificity was 98.2% after 24 h and decreased to 89.7% after 48 h.
Methicillin-resistant Staphylococcus aureus (MRSA) has emerged worldwide as a nosocomial pathogen of major importance, and the incidence of infections caused by MRSA continues to increase (1, 14). The recent emergence of community-acquired MRSA poses an additional challenge for microbiological laboratories to improve their screening strategies (6). Laboratory-based screening for MRSA colonization of patients and health care workers remains a cornerstone of infection control measures to limit the spread of this organism (5). Methods to detect MRSA in clinical samples ideally should have high sensitivity and specificity combined with a short time to reporting of the results. To identify S. aureus from contaminated samples more easily and reliably, selective media have been developed. Ideally, selective media achieve isolation of S. aureus and detection of methicillin resistance in one single step (4).
The purpose of this study is to evaluate the in vitro sensitivity and specificity of a recently developed medium called MRSA ID for the identification of MRSA using a well-defined collection of strains.
A collection consisting of 271 MRSA strains, 249 methicillin-susceptible Staphylococcus aureus (MSSA) strains, and 478 coagulase-negative staphylococci (CNS) was used. The collection was described previously (2, 3). In short, the MRSA isolates were collected in The Netherlands between 1989 and 1998 and are part of the MRSA strain collection of the National Institute of Public Health and Environmental Protection, Bilthoven, The Netherlands. Identification of strains as S. aureus and as being methicillin resistant was performed by duplex PCR for the mecA gene and the coagulase gene (11). Strains were selected on the basis of their different phage types (8, 13; J. A. Rost, unpublished data). The MSSA strains were isolated from cultures of blood collected between May 1996 and January 2005 from patients at the following five hospitals: St. Elisabeth Hospital and Tweesteden Hospital, Tilburg, The Netherlands; Pasteur Hospital, Oosterhout, The Netherlands; Tweesteden Hospital, Waalwijk, The Netherlands; and Amphia Hospital, Breda, The Netherlands. Only one isolate was included per patient per admission period. Isolates were identified by a latex agglutination test (Staphaurex Plus; Murex Diagnostics Ltd., Dartford, England), by the detection of free coagulase by the tube coagulase test with rabbit plasma, and by the detection of DNase (DNase agar; Oxoid Ltd., Basingstoke, England). If the results of these tests were discordant, an AccuProbe culture identification test (Gen-Probe, San Diego, Calif.) was performed according to the manufacturer's instructions, and this was considered the “gold standard.” The blood culture isolates were classified as methicillin susceptible (MIC of oxacillin, ≤2 μg/ml) at the time of collection by broth microdilution susceptibility testing performed according to CLSI (formerly NCCLS) standards (7). The isolates were stored at −70°C until they were tested.
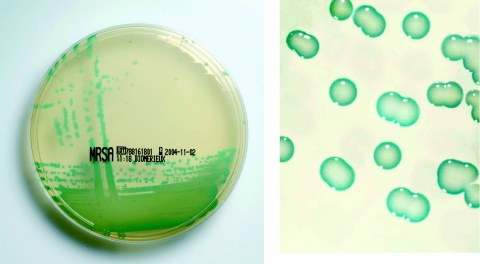
MRSA ID is a new chromogenic medium for identification of MRSA in human specimens and was supplied as prepoured culture plates from bioMérieux, La Balme Les Grottes, France. The composition of the chromogenic and selective mix is proprietary. On MRSA ID, MRSA forms distinctive green colonies (Fig. 1). The selective mixture inhibits MSSA strains, most bacteria not belonging to the genus Staphylococcus, and yeasts. The isolates were inoculated onto Columbia agar plates with 5% sheep blood and incubated for 24 h at 35°C. From the resulting cultures, a suspension with a 0.5 McFarland standard was made, and subsequently, 10 μl was streaked onto an MRSA ID plate with a sterile loop using a three-streak dilution method. The results were read after 24 and 48 h of incubation at 35°C. Growth of colonies showing green coloration was considered to be positive (indicating MRSA). No growth or colonies with colors other than green were considered negative. The procedure was performed as recommended by the manufacturer.
FIG. 1.
MRSA on MRSA ID medium.
Results for the detection of MRSA were compared to the performance of a conventional mannitol salt agar (MSA) plate with an added concentration of 4 μg/ml oxacillin (Oxoid, Haarlem, The Netherlands). Discordant results (no growth of MRSA on MRSA ID or MSA or growth of MSSA on MRSA ID and MSA) were confirmed by mecA gene PCR.
The results obtained with MRSA ID are shown in Table 1. In the current evaluation, 29 MRSA strains gave discordant results, and a PCR for the mecA gene was performed on these isolates. A total of 20 (68%) of the MRSA strains had a negative result with the mecA PCR. These strains were removed from the analysis. The sensitivity of MRSA ID was 96.4% after 24 h and 98.8% after 48 h (P = 0.14). The specificity was 98.2% after 24 h and 89.7% after 48 h (P < 0.001). The sensitivity of MSA was statistically significant lower at both time intervals. The specificity of MSA was significantly lower at 24 h and lower but not statistically significant at 48 h (P = 0.09).
TABLE 1.
Results for MRSA ID medium and MSA medium after 24 and 48 h of incubation
| Isolate | Medium | No. of strains with a positive test result/total no. of strains (%) after:
|
|
|---|---|---|---|
| 24 h | 48 h | ||
| MSSA | MRSA ID | 9/249 (3.6) | 54/249 (21.7) |
| MSA | 16/249 (6.4) | 70/249 (28.1) | |
| CNS | MRSA ID | 4/478 (0.8) | 21/478 (4.4) |
| MSA | 23/478 (4.8) | 27/478 (5.6) | |
| Total (CNS + MSSA) | MRSA ID | 13/727 (1.8) | 75/727 (10.3) |
| MSA | 39/727 (5.3) | 97/727 (13.3) | |
| MRSA | MRSA ID | 242/251 (96.4) | 248/251 (98.8) |
| MSA | 224/251 (89.2) | 237/251 (94.4) | |
In a recent study, the loss of the mecA gene in 36 (14.4%) of 250 methicillin-resistant Staphylococcus aureus isolates after 2 years of storage at −80°C was described (12). In the current evaluation, 29 MRSA strains gave discordant results, and a PCR for the mecA gene was performed on these isolates. A total of 20 (68%) of the MRSA strains no longer harbored the mecA gene. These findings have important implications for the management of strain collections as well as the use of strain collections for an in vitro evaluation. In order to avoid an underestimation of the sensitivity of the MRSA screening test under evaluation, discordant test results must be checked for the presence of the mecA gene at that moment in time.
We evaluated the in vitro sensitivity and specificity of CHROMagar MRSA (CHROMagar Microbiology, Paris, France) using the same collection of strains (2). A total of 216 MRSA strains and 241 MSSA strains were tested. MRSA ID medium used in the present evaluation has a comparable sensitivity at both 24 and 48 h compared to CHROMagar MRSA medium (206/216 [95.4%] and 216/216 [100%] after 24 h and 48 h, respectively). The specificity of MRSA ID medium was significantly lower (P < 0.01), at both 24 and 48 h, than that of CHROMagar MRSA medium (0/241 [100%] at both 24 and 48 h). Overall, the accuracy of detection of MRSA at 24 h of incubation is comparable between MRSA ID and CHROMagar MRSA (18/500 and 10/457, respectively).
In another study, we evaluated the in vitro sensitivities and specificities of oxacillin resistance screening agar base (ORSAB; Oxoid Ltd.) and CHROMagar Staph aureus (CSA; CHROMagar Microbiology) (3). For screening of MRSA strains, antibiotic supplements were added to both media. The sensitivity was low after 24 h of incubation (CSA, 156/266 [58.6%]; ORSAB, 224/266 [84.2%]) but increased significantly after 48 h of incubation (CSA, 206/266 [77.5%]; ORSAB, 243/266 [91.4%]). The specificity for detection of MRSA (measured with MSSA) was high for both media after 24 h (CSA, 358/358 [100%]; ORSAB, 355/358 [99.2%]). After 48 h, it was lower for ORSAB (339/358 [94.7%]) but did not change for CSA. The MRSA ID medium used in the present evaluation has a significantly higher sensitivity than those determined for CSA and ORSAB at both 24 and 48 h in the previous study (P < 0.01). The specificity of MRSA ID is comparable to those determined for ORSAB and CSA at 24 h and significantly lower at 48 h (P < 0.001).
An important limitation of this study is that a high inoculum of MSSA and MRSA isolates in pure culture was used, in contrast to the low concentration usually present in clinical samples. This could lead to more false-negative results in a clinical evaluation, compared to our results. Also, the presence of species other than CNS was not evaluated. This may influence both sensitivity and specificity when MRSA ID medium is used on clinical samples.
In a recent evaluation by Reverdy et al. (10), MRSA ID was compared to ORSAB medium for the identification of MRSA using nasal swabs. Isolation of MRSA on Columbia CNA blood agar was used as the reference method. Suspected colonies were confirmed by mecA gene PCR. A total of 278 nasal swabs were inoculated, and 45 MRSA strains were isolated. After 24 h of incubation, 42 (93.3%) of the 45 strains were isolated on MRSA ID, and 38 (84.4%) were isolated on ORSAB medium. After 48 h, the sensitivity for both media reached 95.6%.
In a clinical evaluation, Perry et al. evaluated MRSA ID, CHROMagar MRSA, ORSAB, and selective mannitol broth for the detection of MRSA (9). A total of 747 swabs from various clinical sites were cultured. A total of 85 MRSA strains were isolated by a combination of all methods. After 48 h of incubation, 89%, 72%, and 78% of the MRSA strains were isolated on MRSA ID, CHROMagar MRSA, and ORSAB, respectively. There was little difference between CHROMagar MRSA, MRSA ID, and ORSAB for the isolation of MRSA from nasal swabs and axillae after 48 h of incubation. However, CHROMagar MRSA appeared to be inferior to all other media for the isolation of MRSA from perineal swabs, possibly due to the impact of the competing flora.
The current evaluation is different from local, clinical studies using consecutive clinical MRSA isolates in an endemic setting, where only a limited number of clonal types will be tested. This leads to an over- or underestimation of the true value of a test under evaluation in other settings. In the current study, a specific type is included only once and many different strains are tested. Therefore, this study provides results representative of a broad array of strains.
In conclusion, MRSA ID is sensitive and specific for differentiation between MSSA and MRSA in vitro. MRSA ID is able to detect a large number of different MRSA strains. The performance of MRSA ID was significantly better after 24 h than after 48 h of incubation. Therefore, with MRSA-ID, optimal results can be obtained within a day. Further clinical studies are required to determine the utility of MRSA ID for the detection of MRSA directly from clinical samples.
REFERENCES
- 1.Archer, G. L. 1998. Staphylococcus aureus: a well-armed pathogen. Clin. Infect. Dis. 26:1179-1181. [DOI] [PubMed] [Google Scholar]
- 2.Diederen, B., I. van Duijn, P. Willemse, P. van Keulen, and J. Kluytmans. 2005. Performance of CHROMagar MRSA medium for detection of methicillin-resistant Staphylococcus aureus. J. Clin. Microbiol. 43:1925-1927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kluytmans, J., A. Van Griethuysen, P. Willemse, and P. Van Keulen. 2002. Performance of CHROMagar selective medium and oxacillin resistance screening agar base for identifying Staphylococcus aureus and detecting methicillin resistance. J. Clin. Microbiol. 40:2480-2482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Merlino, J., M. Leroi, R. Bradbury, D. Veal, and C. Harbour. 2000. New chromogenic identification and detection of Staphylococcus aureus and methicillin-resistant S. aureus. J. Clin. Microbiol. 38:2378-2380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Muto, C. A., J. A. Jernigan, B. E. Ostrowsky, H. M. Richet, W. R. Jarvis, J. M. Boyce, and B. M. Farr. 2003. SHEA guideline for preventing nosocomial transmission of multidrug-resistant strains of Staphylococcus aureus and enterococcus. Infect. Control Hosp. Epidemiol. 24:362. [DOI] [PubMed] [Google Scholar]
- 6.Naimi, T. S., K. H. LeDell, K. Como-Sabetti, S. M. Borchardt, D. J. Boxrud, J. Etienne, S. K. Johnson, F. Vandenesch. S. Fridkin, C. O'Boyle, R. N. Danila, and R. Lynfield. 2003. Comparison of community- and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA 22:2976-2984. [DOI] [PubMed] [Google Scholar]
- 7.National Committee for Clinical Laboratory Standards. 1997. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically, 4th ed. Approved standard M7-A4. National Committee for Clinical Laboratory Standards, Wayne, Pa.
- 8.Parker, M. T. 1983. The significance of phage-typing patterns in Staphylococcus aureus, p. 33-62. In C. S. F. Easmon and C. Adlam (ed.), Staphylococci and staphylococcal infections. Academic Press, Inc., New York, N.Y.
- 9.Perry, J. D., A. Davies, L. A. Butterworth, A. L. Hopley, A. Nicholson, and F. K. Gould. 2004. Development and evaluation of a chromogenic agar medium for methicillin-resistant Staphylococcus aureus. J. Clin. Microbiol. 42:5419-5423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Reverdy, M. E., S. Orenga, J. M. Roche, S. Delorme, and J. Etienne. 2005. Multiresistant bacteria screening: clinical evaluation of MRSA ID, a new chromogenic medium for the screening of methicillin-resistant Staphylococcus aureus, abstr. p1383. Abstr. 15th Eur. Cong. Clin. Microbiol. Infect. Dis. 2005. ECCMID, Copenhagen, Denmark.
- 11.van Griethuysen, A. J., M. Pouw, N. van Leeuwen, M. Heck, P. Willemse, A. Buiting, and J. Kluytmans. 1999. Rapid slide latex agglutination test for detection of methicillin resistance in Staphylococcus aureus. J. Clin. Microbiol. 37:2789-2792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.van Griethuysen, A., I. van Loo, A. van Belkum, C. Vandenbroucke-Grauls, W. Wannet, P. van Keulen, and J. Kluytmans. 2005. Loss of the mecA gene during storage of methicillin-resistant Staphylococcus aureus strains. J. Clin. Microbiol. 43:1361-1365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Van Leeuwen, W. J., and J. A. Rost. 1976. An additional set of phages for the typing of Staphylococcus aureus strains of human origin, non-typable with international basic set of phages. Zentbl. Bacteriol. Parasitenkd. Infectionskr. Hyg. Suppl. 5:1013-1019. [Google Scholar]
- 14.Voss, A., and B. N. Doebbeling. 1995. The worldwide prevalence of methicillin-resistant Staphylococcus aureus. Int. J. Antimicrob. Agents 5:101-106. [DOI] [PubMed] [Google Scholar]