Abstract
Given the increasing commercial and clinical relevance of probiotic cultures, improving the technological robustness of what are often process-sensitive cultures is an important biological goal. The nisin-controlled expression system was used to direct the heterologous expression of the listerial betaine uptake system BetL in the probiotic strain Lactobacillus salivarius UCC118. Following nisin induction, strains expressing betL exhibited a significant increase in resistance to several stresses, including elevated osmo-, cryo-, baro-, and chill tolerance, as well as increased resistance to spray- and freeze-drying. The ability to confer additional stress tolerance on a probiotic culture may be an important step in delivering viable cultures for maximal efficacy.
Probiotic microorganisms, defined as “live microbial feed supplements which beneficially affect the host by improving the intestinal microbial balance” (11), have become the focus of considerable research efforts in recent times (18, 36, 38, 39). Probiotic foods and supplements, because of their potential therapeutic and prophylactic attributes (13, 14, 19, 21, 26, 30), command a growing global market, with recent estimates indicating up to a $6 billion annual worldwide market (18). The introduction of probiotic cultures as part of daily dietary intake may provide a health-promoting, immunomodulatory challenge to the host (9), possibly by stimulating the growth of beneficial bacteria or by lowering levels of potentially harmful microorganisms, thereby reinforcing the body's natural defenses.
One probiotic culture exhibiting promising therapeutic potential is Lactobacillus salivarius subsp. salivarius UCC118. Trials with IL-10 knockout mice revealed that consumption of L.salivarius UCC118 can successfully influence the gastrointestinal flora, attenuate inflammation, and reduce neoplastic lesions (31, 33). Furthermore, human trials have shown that L.salivarius UCC118 can survive gastrointestinal transit, persist within the human gastrointestinal tract for 3 weeks after cessation of administration, stimulate the mucosal immune system, and induce significant alterations in the levels of excreted clostridia and enterococci (9). However, despite the potential health and nutritional benefits associated with L. salivarius UCC118, it is not a particularly robust culture, as it is relatively sensitive to stresses, such as those encountered during spray-drying (low aw and high temperatures), compared to other probiotic microbes (12). Therefore, if L. salivarius UCC118 is to be exploited as an effective probiotic, it is important to protect its viability during processing.
The ability of certain bacterial species to tolerate environmental stress can be attributed, at least in part, to the accumulation of protective compounds termed compatible solutes (41). The preferred compatible solute for the majority of bacteria is the trimethylammonium compound glycine betaine. Listeria monocytogenes contains a secondary glycine betaine transporter (BetL) which is highly characterized at both transcriptional (44) and posttranslational levels (46). While Lactobacillus plantarum and Lactococcus lactis also contain hyperosmotic stress-activated transport systems, QacT and BusA (OpuA), respectively (15, 32, 55), previous analysis in our laboratory has shown that betaine uptake via BetL by L. monocytogenes significantly improves osmo-, cryo-, and barotolerance, as well as increasing survival in certain foods (40, 43, 45, 49). On the basis of these observations, we suggest that an increased ability to accumulate betaine via BetL can improve the ability of an organism to prevail in diverse, stressful environments. To test this hypothesis, we report the first use of the nisin-controlled expression (NICE) system in L. salivarius and demonstrate that nisin-controlled heterologous expression of the listerial betaine uptake system BetL significantly improves the stress tolerance of the probiotic strain L. salivarius UCC118.
MATERIALS AND METHODS
Bacterial strains, plasmids, and growth conditions.
Bacterial strains and plasmids used in this study are listed in Table 1. Lactobacillus salivarius UCC118 was originally isolated from the ileal-cecal region of a human adult undergoing reconstructive surgery. L. salivarius UCC118 was routinely cultured in de Man-Rogosa-Sharpe (MRS) broth. Listeria monocytogenes was grown with aeration in brain heart infusion broth. Both cultures were grown at 37°C. All media were supplied by Oxoid (Basingstoke, United Kingdom). Reconstituted-skim-milk powder (Dairy Products Research Centre, Teagasc-Moorepark, Fermoy, Ireland), at a concentration of 20% (wt/vol), was heat treated at 90°C for 30 min and was used as the carrier medium during both freeze- and spray-drying. Concentrated stocks of the antibiotics erythromycin and chloramphenicol were prepared as described by Maniatis et al. (29) and used at a concentration of 3 μg/ml. Nisin was obtained by filter sterilizing the supernatant of an overnight culture of the nisin-producing strain Lactococcus lactis NZ9700 grown in MRS broth at 30°C. Also, a standardized solution of commercial nisin (Sigma Chemical Co., St. Louis, Mo.) was prepared in 0.05% acetic acid (100 mg/ml) and diluted 1:10 in dimethyl sulfoxide (46).
TABLE 1.
Bacterial strains and plasmids
| Strain or plasmid | Relevant genotype or characteristics | Source or reference |
|---|---|---|
| Strains | ||
| L. salivarius | ||
| UCC118 | Wild-type parent strain | UCC culture collection |
| UCC118n | PnisA expression host harboring pNZ9530 | This study |
| UCC118-BetL+ | UCC118n containing pCPL17 | This study |
| UCC118-BetL− | UCC118n containing the cloning vector pNZ8048 with no insert | This study |
| Listeria monocytogenes LO28nΔBG(pCPL17) | LO28nΔBG containing pCPL17 | 46 |
| Lactococcus lactis NZ9700 | Nisin-producing transconjugant containing Tn5276 | 27 |
| Plasmids | ||
| pNZ8048 | Cmr, carries the nisin-inducible promoter PnisA | 8 |
| pNZ9530 | Eryr, nisRK cloned in pIL252, expression of nisRK driven by transcriptional read-through from the promoter of the preceding rep gene; low copy number | 24 |
| pCPL17 | pNZ8048 plus a promoterless copy of betL | 46 |
Plasmid construction.
The helper plasmid pNZ9530, encoding the nisRK two-component system, was initially transformed into L. salivarius UCC118, forming UCC118n. Plasmid pCPL17 (encoding betL under the transcriptional control of nisA) was subsequently transformed into UCC118n to form UCC118-BetL+. Plasmid pNZ8048, lacking the betL insert was also introduced into UCC118n, forming UCC118-BetL−, for use as a negative control.
Genetic manipulations.
Plasmid DNA isolation was performed using the QIAprep spin miniprep kit (QIAGEN, Crawley, West Sussex, United Kingdom). PCR was performed by using a Hybaid (Middlesex, United Kingdom) PCR Express system. Oligonucleotide primers for PCR were synthesized by Sigma-Genosys Biotechnologies and Taq DNA polymerase (Biotaq; Bioline, London, United Kingdom) was used for all reactions. Colony PCR was carried out following lysis of cells at 96°C for 10 min with IGEPAL CA-630 (Sigma Chemical Co., St. Louis, Mo.). Electrotransformation of L. salivarius UCC118 was achieved by the protocol described by Wei et al. (53). RNA isolation and reverse transcription-PCR (RT-PCR) were carried out as previously described (44). For studies on transcriptional response, to determine the levels of betL expression, an overnight culture of UCC118-BetL+ grown in MRS broth with antibiotic selection at 37°C was used to inoculate three 10-ml volumes of fresh media at a level of 5%. Following 1-h incubation at 37°C, one sample was induced with 0.01% (vol/vol) concentration of the supernatant from an overnight culture of Lactococcus lactis NZ9700; the second was induced with 25 ng of nisin powder/ml. The third sample was not induced with nisin but acted as a control. All samples were incubated at 37°C for a further 5 h before RNA was isolated. Following RT, the PCR primers betL Fnis and betL Rnis (46) were used to amplify the resulting cDNA. In all cases, control PCRs using betL Fnis and betL Rnis primers were used to confirm the complete removal of DNA from non-reverse-transcribed RNA preparations and subsequently following the RT reaction to ensure that levels of cDNA for samples that were to be compared were equal.
Transport assays.
Radiolabeled-betaine uptake studies were carried out as described by Sleator et al. (46), with some minor modifications. Essentially, log-phase cells grown in MRS broth were harvested by centrifugation, washed twice, and resuspended in 50 mM potassium phosphate buffer (pH 6.8) to an optical density at 600 nm (OD600) of 1.0. Glucose was added to a final concentration of 5 mM to energize the cells. After 5 min of incubation at 25°C, assays were initiated by the addition of [14C]glycine betaine (at a final concentration of 40 μM and a specific radioactivity of 5 Ci/mol). Cells were collected on 0.45-μm-pore-size cellulose nitrate filters (Schleicher & Schuell, GmbH, Dassell, Germany, and Millipore Canada Ltd.) under a vacuum. Filters were then washed with 5 ml of buffer and the radioactivity trapped in the cells was measured by liquid scintillation counting.
Bacterial enumeration.
CFU were determined prior to and after treatments using either the spread or pour plate technique. Appropriate serial dilutions were prepared in one-quarter-strength Ringer's solution before surface plating onto MRS agar. Enumeration of probiotics in the inoculated milk preparations was assessed before freeze- and spray-drying and in the resulting powders by examining MRS broth pour plates. To 1 ml of milk or 1 g of powder, 9 ml of one-quarter-strength Ringer's solution was added before diluting further with one-quarter-strength Ringer's solution and pour plating the appropriate dilutions. All agar plates were incubated at 37°C for 48 h in an anaerobic system (Anaerocult A, Merck Darmstadt, Germany). Percentage cell survival was calculated by determining the number of CFU after challenge, expressed as a percentage of CFU at time zero, i.e., immediately prior to treatment.
Osmotolerance.
Overnight cultures of UCC118-BetL+ and UCC118-BetL− were used to inoculate fresh MRS broth supplemented with 0 to 10% (wt/vol) NaCl, both in the presence and absence of 0.01% (vol/vol) supernatant from an overnight culture of Lactococcus lactis NZ9700. OD595 readings were taken every hour for 25 h using the Tecan GENios microplate reader (Alpha Technologies, Ireland). CFU were also monitored at regular intervals throughout the experiment.
Induction of the NICE system in L. salivarius UCC118.
Nisin induction was carried out essentially as described by Pavan et al. (34). Overnight cultures of UCC118-BetL+ and UCC118-BetL− grown in MRS broth supplemented with appropriate antibiotics were inoculated at a level of 5% into fresh media. Following 1 h growth at 37°C, cultures were induced with 0.01% (vol/vol) of the supernatant from an overnight culture of the nisin-producing strain Lactococcus lactis NZ9700, and subsequently incubated for a further 5 h at 37°C. Following nisin induction of UCC118-BetL+ and UCC118-BetL−, various physiological assays, as outlined below, were completed to compare the performances of the strains.
Low-temperature viability.
UCC118-BetL+ and UCC118-BetL− were inoculated at a concentration of 1% into 100 ml MRS broth supplemented with 0.01% (vol/vol) of cell-free supernatant from Lactococcus lactis NZ9700. Both cultures were incubated at 4°C and 10°C for 3 weeks. For freeze-thaw assays, separate 1.5-ml aliquots of MRS broth containing either UCC118-BetL+ or UCC118-BetL− were dispensed into Eppendorf tubes which were frozen at −20°C (slow cooling) and −70°C (fast cooling). Cell counts were recorded before freezing and subsequently following thawing for seven freeze-thaw cycles. Samples were allowed to thaw at room temperature before immediately replacing them in the freezer.
Freeze- and spray-drying.
Following nisin induction, UCC118-BetL+ and UCC118-BetL− were harvested by centrifugation (1,600 × g, 10 min at 4°C using a Beckman J2-21 centrifuge). The supernatant was discarded and the pellets washed once with one-quarter-strength Ringer's solution before resuspending the pellets in reconstituted skim milk. Samples were prepared for freeze-drying by freezing the milk suspensions overnight at −70°C before freeze-drying in an Edwards Modulyo (Mason Technologies, Dublin, Ireland) freeze-dryer for 24 h. Spray-drying was undertaken in a laboratory-scale spray drier (mini spray dryer B-191; Buchi, Flawil, Switzerland). The feed solution was pneumatically atomized into a vertical cocurrent drying chamber using a two-fluid nozzle. The inlet temperature was maintained at a constant 170°C. The outlet-air temperature was controlled by adjusting the flow rate of the feed solution and ranged from 80 to 85°C. Dried powder samples were collected from the base of the cyclone.
High-pressure processing.
Following nisin induction, separate Eppendorf tubes containing log-phase cells of either UCC118-BetL+ or UCC118-BetL− in MRS broth were completely filled. Tubes were sealed with Parafilm and vacuum packaged with 10 ml water in polyamide/polyethylene bags (Alcom, Campogalliano 9MO, Italy) by using a Webomatic vacuum packaging system (Mausegatt 59, type D463; Werner Bonk, Bochum, Germany). These packs were again vacuum packaged to ensure no contact between the pressure-transmitting medium and the Eppendorf tubes. Samples were high-pressure treated in a Iso-Lab 900 high-pressure food processor (Stansted Fluid Power, Stansted, Essex, United Kingdom), using a 9:1 mixture of ethanol and castor oil as pressurizing fluid. This pressure vessel has a 2-liter capacity and an internal diameter of 100 mm. Treatments of 300 MPa and 350 MPa were examined. Pressure was increased at a rate of 300 MPa min−1, maintained at the desired pressure for 5 min, and released at a rate of 300 MPa min−1. The temperature of the vessel of the high-pressure unit was thermostatically controlled at 20°C throughout treatment.
RESULTS
Heterologous expression of betL in Lactobacillus salivarius UCC118.
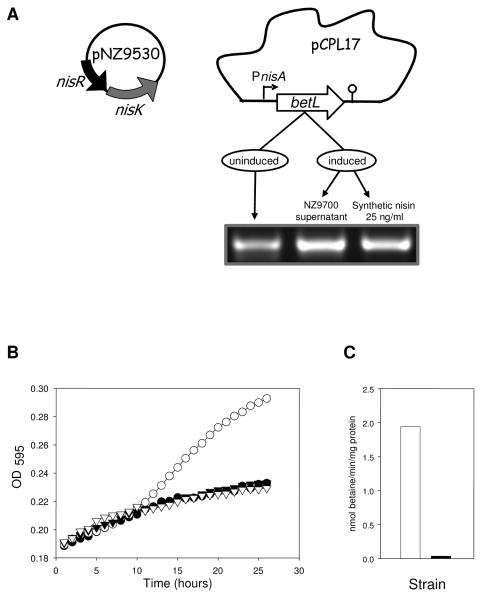
The NICE system was originally developed for use with lactic acid bacteria (LAB) (24) and was subsequently demonstrated to function in a variety of other gram-positive bacteria (10). The system consists of two compatible replicons: an essential helper plasmid, pNZ9530, encoding the nisRK regulatory genes, and the expression vector pNZ8048, bearing the inducible nisA promoter (PnisA) (Fig. 1A). Previously, we demonstrated that a promoterless copy of betL (encoding the secondary betaine uptake system BetL of Listeria monocytogenes) cloned downstream of PnisA in pNZ8048 can be transcribed in a dose-dependent manner in response to added nisin (46). In the present study, this plasmid construct (designated pCPL17) was transformed into L. salivarius UCC118 harboring pNZ9530 (designated UCC118n), forming UCC118-BetL+. Plasmid pNZ8048 lacking the betL insert was also introduced into UCC118n for use as a negative control in subsequent experiments and was designated UCC118-BetL−. Confirmation that the NICE system is functional in L. salivarius UCC118 was obtained from RT-PCR (Fig. 1A). Densitometric analysis following nisin induction of UCC118-BetL+ revealed a three- to fourfold increase in the betL transcript levels when nisin induction was performed using the supernatant of Lactococcus lactis NZ9700 and a two to threefold increase when the culture was induced with synthetic nisin compared to the uninduced culture. Therefore, because the highest transcription levels of the betL gene were obtained when UCC118-BetL+ was induced with supernatant from the nisin-producing strain, this was the source of nisin used in subsequent experiments. Interestingly, as described previously by Sleator et al. (46), a low level of transcription relative to the induced state was observed in the absence of nisin. This “leaky” transcription most likely results from the presence of lactose or galactose in the media which have previously been shown to induce expression of PnisA independent of nisin concentrations (5). Evidence that the BetL protein is functional against the L. salivarius UCC118 background was obtained using radiolabeled betaine uptake studies. Initial betaine uptake rates were determined both for UCC-BetL+ and UCC-BetL− (Fig. 1C). Within the time frame tested, UCC-BetL+ had an uptake rate of 1.95 nmol/min/mg protein, as opposed to 0.03 nmol/min/mg protein for the control strain UCC-BetL−.
FIG. 1.
(A) RT-PCRs showing nisin induced transcriptional control of the betL gene in UCC118-BetL+. Each lane represents the level of betL transcript resulting from 30 PCR cycles with primers XbaIKO (TAAGCGCCACTCTAGACC) and EcoRIKO (GCACGAATTCACCAAGTA) on cDNA generated from total RNA isolated from cells after 30 min of exposure to nonstress conditions in MRS broth (first lane), 0.01% (vol/vol) cell-free supernatant from the nisin-producing strain Lactococcus lactis NZ9700 (second lane) and 25 ng of nisin powder/ml (third lane). Bent arrow, promoter; lollipop, terminator. (B) Growth of L. salivarius UCC118-BetL+ (○ and Δ) and UCC118-BetL− (• and ▴) in MRS broth with 7% added NaCl in the presence (circles) and absence (triangles) of added nisin. Each point represents the mean of at least three independent experiments. (C) [14C]glycine betaine uptake rates in L. salivarius UCC118-BetL+ (white bar) and UCC118-BetL− (black bar).
Osmotolerance.
One of the most common features associated with betaine uptake is osmotolerance. To determine whether heterologous expression of the listerial betaine uptake system, BetL, would improve the osmotolerance of L. salivarius UCC118, we first examined the intrinsic osmotolerance of the control strain, UCC118-BetL−. In line with expectations, replication of the probiotic was significantly affected at NaCl concentrations above 5% (data not shown), and UCC118-BetL− was essentially unable to increase in size at 7% NaCl (Fig. 1B). In MRS broth supplemented with 0 to 10% NaCl, no significant difference was observed between the growth of UCC118-BetL+ and UCC118-BetL− in the absence of nisin, suggesting that the level of BetL expression in the absence of nisin is unable to confer any additional osmotolerance on UCC118 (Fig. 1B). However, nisin induction of betL in the UCC118 background resulted in a significantly higher growth rate and a higher final optical density when grown at 7% NaCl than those of the control, which failed to grow under similar conditions. CFU were consistent with the OD595 values. This significant increase in osmotolerance suggests that BetL is not only expressed but functional in the Lactobacillus background.
Low-temperature viability.
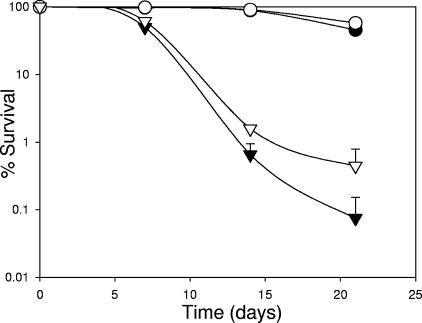
Given that betaine has been shown to enhance growth and survival at low temperatures in Listeria (3, 54), we investigated the survival of UCC118-BetL+ and the control strain UCC118-BetL− at 4°C and 10°C over a period of 21 days (Fig. 2). At 4°C, CFU remained relatively stable, with no significant (P > 0.05) decline recorded. However, during storage at 10°C, while survival of both cultures remained relatively unchanged over the initial 7 days, CFU decreased considerably thereafter. Notably, UCC118-BetL− declined at a faster rate than UCC118-BetL+, which at day 21 gave ∼0.5-log higher counts.
FIG. 2.
Viability of L. salivarius UCC118-BetL+ (○ and Δ) and UCC118-BetL− (• and ▴) during storage at 4°C (circles) and 10°C (triangles) in MRS broth. Results are means of four independent experiments.
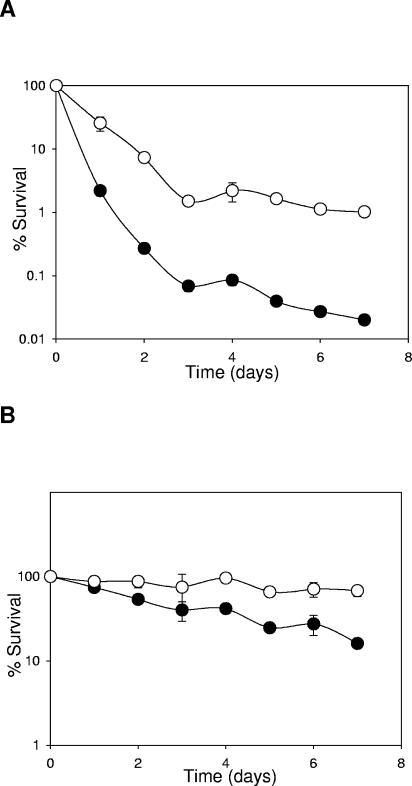
In addition to chill stress, industrially valuable bacterial cultures are commonly exposed to subzero temperatures. After seven repeated freeze-thaw cycles, UCC118-BetL+ was recovered at significantly higher levels than UCC118-BetL− following freezing at −20°C (P ≤ 0.001) (Fig. 3A) and −70°C (P < 0.02) (Fig. 3B). The survival of UCC118-BetL+ was 2 logs greater than the control at −20°C and 0.5 logs greater at −70°C.
FIG. 3.
Survival of L. salivarius UCC118-BetL+ (○) and UCC118-BetL− (•) following repeated freeze-thaw cycles at −20°C (A) and at −70°C (B). Results are means of four independent experiments.
Freeze- and spray-drying.
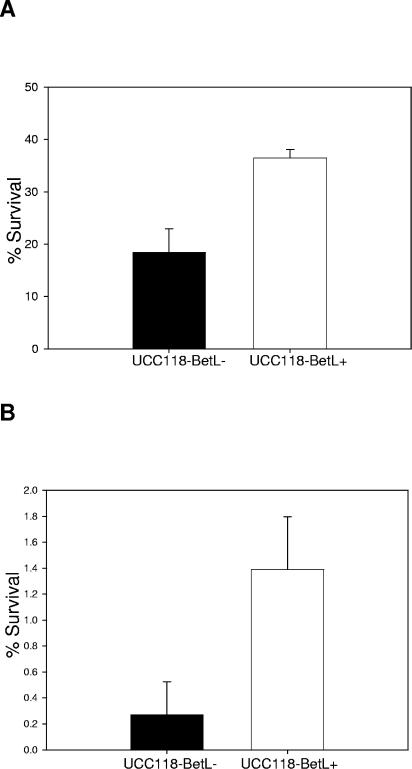
Given that betaine has previously been shown to protect LAB from desiccation (22, 23, 41), we examined whether BetL is capable of protecting L. salivarius UCC118 during freeze- and spray-drying, both of which expose cells to extremes of temperature and low aW. UCC118-BetL+ survived freeze-drying (P ≤ 0.01) and spray-drying (P ≤ 0.05) significantly better than UCC118-BetL−. The percent survival of UCC118-BetL+ during freeze-drying was 36%, compared to 18% for UCC118-BetL− (Fig. 4A). Viability of both strains was, however, severely affected following spray-drying with survival rates of 1.4% and 0.3% obtained for UCC118-BetL+ and UCC118-BetL−, respectively (Fig. 4B).
FIG. 4.
Survival of L. salivarius UCC118-BetL+ and UCC118-BetL− following (A) freeze-drying and (B) spray-drying. Results are means of experiments performed in triplicate.
Barotolerance.
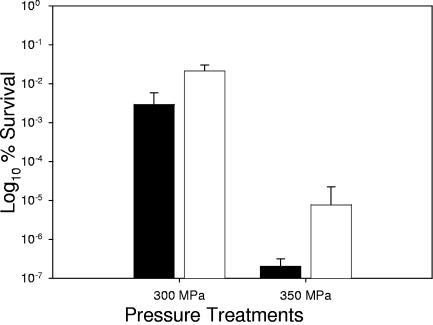
Previously, Smiddy et al. (49) demonstrated a possible role for compatible solutes, specifically betaine, inbacterial barotolerance. When exposure of L. salivarius UCC118 to high pressure was examined, the nisin-induced derivative UCC118-BetL+ survived 300 MPa (P ≤ 0.05) and 350 MPa (P ≤ 0.001) significantly better than the control strain UCC118-BetL− (Fig. 5). While viable counts were dramatically reduced following exposure to both treatments, UCC118-BetL+ was recovered at levels ∼1 log greater than the control at 300 MPa and ∼2 logs greater following exposure to 350 MPa. Pressure treatments were chosen based on previous unpublished data from our laboratory, which revealed that L. salivarius UCC118 is unable to endure pressures greater than 350 MPa.
FIG. 5.
High-pressure-induced inactivation of L. salivarius UCC118-BetL+ (white bars) and UCC118-BetL− (black bars). All treatments were carried out for 5 min at 20°C. Results are means of experiments performed in triplicate.
DISCUSSION
Bacteria, particularly pathogenic and/or commensal species alternating between host and external environments, have evolved complex stress management strategies to ensure their continued survival in diverse ecological niches (1, 17, 47). Among these adaptations is the ability to accumulate compatible solutes, such as betaine. Indeed, compatible solute uptake in the food-borne pathogen Listeria monocytogenes has previously been shown to protect the pathogen from the detrimental affects of low aw, reduced temperature, and extremes of pressure, as well as prolonging gastrointestinal persistence (43, 45, 48, 49), all potentially desirable traits in selecting and designing versatile probiotic strains.
Compatible solute uptake in Listeria is mediated by three transport systems, the simplest (in genetic terms) of which is the secondary betaine transporter BetL. Previously, we described the use of the NICE system to control betL expression in L. monocytogenes in response to nisin in a dose-dependent manner (46). In the present study, we report the first use of the NICE system in L. salivarius and demonstrate that heterologously expressed BetL can significantly improve betaine uptake rates, with a resulting increase in the stress tolerance profile of the probiotic strain L. salivarius UCC118.
RT-PCR analysis confirmed that the NICE system functions in L. salivarius UCC118, with betL transcript levels increasing in response to added nisin. A low level of transcription was observed in the absence of nisin, possibly a consequence of galactose- or lactose-mediated induction of PnisA (5). Interestingly, this low level of expression did not confer any significant increase in osmotolerance. In the presence of nisin, sufficient BetL was produced to confer increased salt tolerance, with growth of the transformed construct at significantly higher salt levels than is possible for the parent strain or for other lactobacilli. For comparative purposes, Listeria (containing BetL under the control of its native promoter) is capable of growth at 9% NaCl (40). The inability to reach the listerial osmotolerance of 9% is not surprising, given the presence of at least two other additional compatible solute uptake systems, Gbu and OpuC, in Listeria.
Betaine uptake has also been shown to play a role in chill tolerance (3, 43, 54). While storage at 4°C had little effect on the viability of L. salivarius UCC118 derivatives, a significant decrease in CFU was observed during storage at 10°C. The increased bactericidal effects observed for the higher storage temperature correlate with previous observations in bifidobacteria by Dave and Shah (7). The detrimental effects observed at the higher storage temperature were suggested to result from increased acidity and subsequent pH decrease (7). Despite the significant reduction in viability of both strains at 10°C, the probiotic expressing BetL displayed a slower death rate and was recovered in higher numbers than the control strain.
In addition to chill tolerance, compatible solutes have also been suggested to improve cryotolerance (6, 25). Indeed, betL expression in L. salivarius UCC118 derivatives leads to a significant increase in viability following repeated freeze-thaw cycles during storage at −20°C and −70°C. Interestingly, for both strains, −20°C appeared significantly more bactericidal than −70°C, a pattern also observed for other lactobacilli (51). It is possible that L. salivarius UCC118 derivatives are more susceptible to damage by external osmotic stress to the cell, a characteristic of slow cooling as opposed to intracellular ice crystal formation, a consequence of fast cooling, a pattern also observed for other lactobacilli (51, 56). The improved viability of UCC118-BetL+, most likely reflects the cryoprotective properties of accumulated betaine which stabilizes protein structure and function, while also maintaining membrane fluidity (1, 41, 49).
Another important role of compatible solutes is to counteract the damaging effects of water loss due to drying (6, 22, 23). Freeze- and spray-drying, both commonly used practices for the preparation and storage of concentrated stocks of LAB, expose cells to the undesirable attenuating effects of extreme temperatures, dehydration, protein denaturation, and membrane damage (2, 12, 28, 50). Consistent with previous observations (20, 52) spray-drying was found to be significantly more detrimental to cell survival than freeze-drying. However, for both drying processes, expression of betL conferred enhanced viability on the probiotic strain, a finding which is not altogether surprising, given that compatible solutes have previously been shown to protect LAB during drying (6, 22, 23). Furthermore, the dramatic difference in survival of the strain expressing betL during freeze-drying as opposed to spray-drying most likely reflects the increased cryotolerance phenotype observed for this strain.
Recently, Smiddy et al. (49) identified a novel role for compatible solutes in contributing to listerial barotolerance. L. salivarius UCC118 expressing betL also survives significantly better than the control strain when exposed to pressures in excess of 300 MPa. While the exact mechanism by which compatible solutes protect the bacterial cell from the damaging effects of high pressure have yet to be determined, it is possible that the preferential exclusion of accumulated betaine from the surface of proteins, forming a hydration shell, may prevent essential enzymes unfolding during high-pressure treatment (41). Furthermore, betaine has also been suggested to contribute to increased barotolerance by maintaining membrane fluidity (49). In any case, induced betaine uptake via BetL significantly improves the pressure resistance profile of L. salivarius UCC118.
The concept of “inverse pathogenesis,” a term used to describe the bioengineering of “generally recognized as safe” organisms with genes from more versatile pathogenic species, has become a growing area of interest in recent times (4, 42). Indeed, Guimarães et al. (16) recently described the construction of a Lactococcus lactis strain expressing inlA, encoding internalin A, a known virulence factor in L. monocytogenes. In this instance, the transformed L. lactis strain is capable of invading the small intestine and delivering DNA into mammalian epithelial cells, making it a safer and more attractive alternative to attenuated L. monocytogenes as an antigen delivery vehicle. While this approach may find favor in clinical applications such as vaccine delivery, it is unlikely that the introduction of genes from pathogens into otherwise harmless probiotic cultures would meet with the approval of regulatory authorities, irrespective of the potential benefits. Nonetheless, this study illustrates that the introduction of a single gene can significantly improve the robustness of a potentially important yet physiologically fragile probiotic culture. It is also likely that BetL homologues from “generally recognized as safe” organisms (e.g., BetP from Corynebacterium glutamicum [35] or BusA [OpuA] from L. lactis [37]), will also offer similar protective effects. Furthermore, natural selection of probiotic cultures with elevated betaine uptake capabilities could dispense with the necessity for recombinant DNA approaches. In conclusion, we suggest that this study demonstrates how the stress response mechanisms of food-borne pathogens can be employed to develop probiotics with improved technological efficacies.
Acknowledgments
We acknowledge the financial assistance of the Irish Government under the National Development Plan 2000-2006, the financial support of Enterprise Ireland through the Advanced Technology Research Programme, and funding of the Alimentary Pharmabiotic Centre by Science Foundation Ireland. Roy D. Sleator is funded by an Embark Postdoctoral Fellowship.
We are grateful to Dave Waldron for his expert advice and help with the spray-drying procedures.
REFERENCES
- 1.Abee, T., and J. A. Wouters. 1999. Microbial stress response in minimal processing. Int. J. Food Microbiol. 50:65-91. [DOI] [PubMed] [Google Scholar]
- 2.Ananta, E., M. Volkert, and D. Knorr. 2005. Cellular injuries and storage stability of spray-dried Lactobacillus rhamnosus GG. Int. Dairy J. 15:399-409. [Google Scholar]
- 3.Angelidis, A. S., and G. M. Smith. 2003. Role of the glycine betaine and carnitine transporters in adaptation of Listeria monocytogenes to chill stress in defined medium. Appl. Environ. Microbiol. 69:7492-7498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Boscari, A., K. Mandon, M. C. Poggi, and D. le Rudulier. 2004. Functional expression of Sinorhizobium meliloti BetS, a high-affinity betaine transporter, in Bradyrhizobium japonicum USDA110. Appl. Environ. Microbiol. 70:5916-5922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chandrapati, S., and D. J. O'Sullivan. 1999. Nisin independent induction of the nisA promoter in Lactococcus lactis during growth in lactose or galactose. FEMS Microbiol. Lett. 170:191-198. [DOI] [PubMed] [Google Scholar]
- 6.Cleland, D., P. Krader, C. McCree, J. Tang, and D. Emerson. 2004. Glycine betaine as a cryoprotectant for prokaryotes. J. Microbiol. Methods 58:31-38. [DOI] [PubMed] [Google Scholar]
- 7.Dave, R. I., and N. P. Shah. 1997. Viability of yoghurt and probiotic bacteria in yoghurts made from commercial starter cultures. Int. Dairy J. 7:31-41. [Google Scholar]
- 8.De Ruyter, P. G. G. A., O. P. Kuipers, and W. M. de Vos. 1996. Controlled gene expression systems for Lactococcus lactis with the food-grade inducer nisin. Appl. Environ. Microbiol. 62:3662-3667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dunne, C., L. Murphy, S. Flynn, L. O'Mahony, S. O'Halloran, M. Feeney, D. Morrissey, G. Thorton, G. Fitzgerald, C. Daly, B. Kiely, E. M. M. Quigley, G. C. O'Sullivan, F. Shanahan, and J. K. Collins. 1999. Probiotics: from myth to reality. Demonstration of functionality in animal models of disease and in human clinical trials. Antonie Leeuwenhoek 76:279-292. [PubMed] [Google Scholar]
- 10.Eichenbaum, Z., M. J. Federle, D. Marra, W. M. de Vos, O. P. Kuipers, M. Kleerebezem, and J. R. Scott. 1998. Use of the lactococcal nisA promoter to regulate gene expression in gram-positive bacteria: comparison of induction level and promoter strength. Appl. Environ. Microbiol. 64:2763-2769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fuller, R. 1989. Probiotics in man and animals. J. Appl. Bacteriol. 66:365-378. [PubMed] [Google Scholar]
- 12.Gardiner, G. E., E. O'Sullivan, J. Kelly, M. A. E. Auty, G. F. Fitzgerald, J. K. Collins, R. P. Ross, and C. Stanton. 2000. Comparative survival rates of human-derived probiotic Lactobacillus paracasei and L. salivarius strains during heat treatment and spray drying. Appl. Environ. Microbiol. 66:2605-2612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gill, H. S. 2003. Probiotics to enhance anti-infective defences in the gastrointestinal tract. Best Pract. Res. Clin. Gastroenterol. 17:755-773. [DOI] [PubMed] [Google Scholar]
- 14.Gismondo, M. R., L. Drago, and A. Lombardi. 1999. Review of probiotics available to modify gastrointestinal flora. Int. J. Antimicrob. Agents 12:287-292. [DOI] [PubMed] [Google Scholar]
- 15.Glaasker, E., E. H. M. L. Heuberger, W. N. Konings, and B. Poolman. 1998. Mechanism of osmotic activation of the quaternary ammonium compound transporter (QacT) of Lactobacillus plantarum. J. Bacteriol. 180:5540-5546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Guimarães, V. D., J. E. Gabriel, F. Lefèvre, D. Cabanes, A. Gruss, P. Cossart, V. Azevedo, and P. Langella. 2005. Internalin-expressing Lactococcus lactis is able to invade small intestine of guinea pigs and deliver DNA into mammalian epithelial cells. Microbes Infect. 7:836-844. [DOI] [PubMed] [Google Scholar]
- 17.Hill, C., P. D. Cotter, R. D. Sleator, and C. G. M. Gahan. 2002. Bacterial stress response in Listeria monocytogenes: jumping the hurdles imposed by minimal processing. Int. Dairy J. 12:273-283. [Google Scholar]
- 18.Hoesl, C. E., and J. E. Altwein. 2005. The probiotic approach: an alternative treatment option in urology. Eur. Urol. 47:288-296. [DOI] [PubMed] [Google Scholar]
- 19.Hoyos, A. B. 1999. Reduced incidence of necrotizing enterocolitis associated with eternal administration of Lactobacillus acidophilus and Bifidobacterium infantis to neonates in an intensive care unit. Int. J. Infect. Dis. 3:197-202. [DOI] [PubMed] [Google Scholar]
- 20.Johnson, J. A. C., and M. R. Etzel. 1995. Properties of Lactobacillus helveticus CNRZ-32 attenuated by spray-drying, freezing-drying, or freezing. J. Dairy Sci. 78:761-768. [Google Scholar]
- 21.Kailasapathy, K., and J. Chin. 2000. Survival and therapeutic potential of probiotic organisms with reference to Lactobacillus acidophilus and Bifidobacterium spp. Immunol. Cell Biol. 78:80-88. [DOI] [PubMed] [Google Scholar]
- 22.Kets, E. P. W., and J. A. M. de Bont. 1994. Protective effect of betaine on survival of Lactobacillus plantarum subjected to drying. FEMS Microbiol. Lett. 116:251-256. [Google Scholar]
- 23.Kets, E. P. W., P. J. M. Teunissen, and J. A. M. de Bont. 1996. Effect of compatible solutes on survival of lactic acid bacteria subjected to drying. Appl. Environ. Microbiol. 62:259-261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kleerebezem, M., M. M. Beerthuyzen, E. E. Vaughan, W. M. de Vos, and O. P. Kuipers. 1997. Controlled gene expression systems for lactic acid bacteria: transferable nisin-inducible expression cassettes for Lactococcus, Leuconostoc, and Lactobacillus spp. Appl. Environ. Microbiol. 63:4581-4584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ko, R., L. T. Smith, and G. M. Smith. 1994. Glycine beatine confers enhanced osmotolerance and cryotolerance on Listeria monocytogenes. J. Bacteriol. 176:426-431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kopp-Hoolihan, L. 2001. Prophylactic and therapeutic uses of probiotics: a review. J. Am. Diet. Assoc. 101:229-241. [DOI] [PubMed] [Google Scholar]
- 27.Kuipers, O. P., M. M. Beerthuizen, R. J. Siezen, and W. M. de Vos. 1993. Characterization of the nisin gene cluster nisABTCIPR of Lactococcus lactis requirement of expression of nisA and nisI genes for development of immunity. Eur. J. Biochem. 216:281-291. [DOI] [PubMed] [Google Scholar]
- 28.Leslie, S. B., E. Israeli, B. Lighthart, J. H. Crowe, and L. M. Crowe. 1995. Trehalose and sucrose protect both membranes and proteins in intact bacteria during drying. Appl. Environ. Microbiol. 61:3592-3597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Maniatis, T., E. F. Fritisch, and J. Sambrook. 1982. Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.
- 30.Mattila-Sandholm, T., S. Blum, J. K. Collins, R. Crittenden, W. de Vos, C. Dunne, R. Fonden, G. Grenov, E. Isolauri, B. Kiely, P. Marteau, L. Morelli, A. Ouwehand, R. Reniero, M. Saarela, S. Salminen, M. Saxelin, E. Schiffrin, F. Shanahan, E. Vaughan, and A. von Wright. 1999. Probiotics: towards demonstrating efficacy. Trends Food Sci. Technol. 10:393-399. [Google Scholar]
- 31.McCarthy, J., L. O'Mahony, L. O'Callaghan, B. Sheil, E. E. Vaughan, N. Fitzsimons, J. Fitzgibbon, G. C. O'Sullivan, B. Kiely, J. K. Collins, and F. Shanahan. 2003. Double blind, placebo controlled trial of two probiotic strains in interleukin 10 knockout mice and mechanistic link with cytokine balance. Gut 52:975-980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Obis, D., A. Guillot, J. C. Gripon, P. Renault, A. Bolotin, and M. Y. Mistou. 1999. Genetic and biochemical characterization of a high-affinity betaine uptake system (BusA) in Lactococcus lactis reveals a new functional organization within bacterial ABC transporters. J. Bacteriol. 181:6238-6246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.O'Mahony, L., M. Feeney, S. O'Halloran, L. Murphy, B. Kiely, J. Fitzgibbon, G. Lee, G. O'Sullivan, F. Shanahan, and J. K. Collins. 2001. Probiotic impact on microbial flora, inflammation and tumour development in IL-10 knockout mice. Aliment. Pharmacol. Ther. 15:1219-1225. [DOI] [PubMed] [Google Scholar]
- 34.Pavan, S., P. Hols, J. Delcour, M. C. Geoffroy, C. Grangette, M. Kleerebezem, and A. Mercenier. 2000. Adaptation of the nisin-controlled expression system in Lactobacillus plantarum: a tool to study in vivo biological effects. Appl. Environ. Microbiol. 66:4427-4432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Peter, H., A. Burkovski, and R. Krämer. 1996. Isolation, characterization, and expression of the Corynebacterium glutamicum betP gene, encoding the transport system for the compatible solute glycine betaine. J. Bacteriol. 178:5229-5234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rautava, S., M. Kalliomaki, and E. Isolauri. 2005. New therapeutic strategy for combating the increasing burden of allergic disease: probiotics—a Nutrition, Allergy, Mucosal Immunology and Intestinal Microbiota (NAMI) Research Group report. J. Allergy Clin. Immunol. 116:31-37. [DOI] [PubMed] [Google Scholar]
- 37.Romeo, Y., D. Obis, J. Bouvier, A. Guillot, A. Fourçans, I. Bouvier, C. Gutierrez, and M. Y. Mistou. 2003. Osmoregulation in Lactococcus lactis: BusR, a transcriptional repressor of the glycine betaine uptake system BusA. Mol. Microbiol. 47:1135-1147. [DOI] [PubMed] [Google Scholar]
- 38.Salminen, S., A. von Wright, L. Morelli, P. Marteau, D. Brassart, W. M. de Vos, R. Fonden, M. Saxelin, K. Collins, G. Mogensen, S. E. Birkeland, and T. Mattila-Sandholm. 1998. Demonstration of safety of probiotics— a review. Int. J. Food Microbiol. 44:93-106. [DOI] [PubMed] [Google Scholar]
- 39.Sanders, M. E. 1998. Overview of functional foods: emphasis on probiotic bacteria. Int. Dairy J. 8:341-347. [Google Scholar]
- 40.Sleator, R. D., C. G. M. Gahan, and C. Hill. 2003. A postgenomic appraisal of osmotolerance in Listeria monocytogenes. Appl. Environ. Microbiol. 69:1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sleator, R. D., and C. Hill. 2001. Bacterial osmoadaptation: the role of osmolytes in bacterial stress and virulence. FEMS Microbiol. Rev. 26:49- 71. [DOI] [PubMed] [Google Scholar]
- 42.Sleator, R. D., and C. Hill. 2006. Patho-biotechnology: using bad bugs to do good things. Curr. Opin. Biotechnol. 17:1-6. [DOI] [PubMed]
- 43.Sleator, R. D., G. A. Francis, D. O'Beirne, C. G. M. Gahan, and C. Hill. 2003. Betaine and carnitine uptake systems in Listeria monocytogenes affect growth and survival in foods and during infection. J. Appl. Microbiol. 95:839-846. [DOI] [PubMed] [Google Scholar]
- 44.Sleator, R. D., C. G. M. Gahan, B. O'Driscoll, and C. Hill. 2000. Analysis of the role of betL in contributing to the growth and survival of Listeria monocytogenes LO28. Int. J. Food Microbiol. 60:261-268. [DOI] [PubMed] [Google Scholar]
- 45.Sleator, R. D., C. G. M. Gahan, T. Abee, and C. Hill. 1999. Identification and disruption of BetL, a secondary glycine betaine transport system linked to the salt tolerance of Listeria monocytogenes LO28. Appl. Environ. Microbiol. 65:2078-2083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sleator, R. D., J. M. Wood, and C. Hill. 2003. Transcriptional regulation and posttranslational activity of the betaine transporter BetL in Listeria monocytogenes are controlled by environmental salinity. J. Bacteriol. 185:7140-7144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sleator, R. D., H. H. Wemekamp-Kamphuis, C. G. M. Gahan, T. Abee, and C. Hill. 2005. A PrfA-regulated bile exclusion system (BilE) is a novel virulence factor in Listeria monocytogenes. Mol. Microbiol. 55:1183-1195. [DOI] [PubMed] [Google Scholar]
- 48.Sleator, R. D., J. Wouters, C. G. M. Gahan, T. Abee, and C. Hill. 2001. Analysis of the role of OpuC, an osmolyte transport system, in salt tolerance and virulence potential of Listeria monocytogenes. Appl. Environ. Microbiol. 67:2692-2698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Smiddy, M., R. D. Sleator, M. F. Patterson, C. Hill, and A. L. Kelly. 2004. Role for compatible solutes glycine betaine and l-carnitine in listerial barotolerance. Appl. Environ. Microbiol. 70:7555-7557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Teixeira, P., H. Castro, and R. Kirby. 1995. Spray drying as a method for preparing concentrated cultures of Lactobacillus bulgaricus. J. Appl. Bacteriol. 78:456-462. [Google Scholar]
- 51.Tomás, M. S. J., V. S. Ocaña, and M. E. Nader-Macías. 2004. Viability of vaginal probiotic lactobacilli during refrigerated and frozen storage. Anaerobe 10:1-5. [DOI] [PubMed] [Google Scholar]
- 52.Wang, Y. C., R. C. Yu, and C. C. Chou. 2004. Viability of lactic acid bacteria and bifidobacteria in fermented soymilk after drying, subsequent rehydration and storage. Int. J. Food Microbiol. 93:209-217. [DOI] [PubMed] [Google Scholar]
- 53.Wei, M. Q., C. M. Rush, J. M. Norman, L. M. Hafner, R. J. Epping, and P. Timms. 1995. An improved method for the transformation of Lactobacillus strains using electroporation. J. Microbiol. Methods 21:97-109. [Google Scholar]
- 54.Wemekamp-Kamphuis, H. H., R. D. Sleator, J. A. Wouters, C. Hill, and T. Abee. 2004. Molecular and physiological analysis of the role of osmolyte transporters BetL, Gbu, and OpuC in growth of Listeria monocytogenes at low temperatures. Appl. Environ. Microbiol. 70:2912-2918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wood, J. M., E. Bremer, L. N. Csonka, R. Kraemer, B. Poolman, T. van der Heide, and L. T. Smith. 2001. Osmosensing and osmoregulatory compatible solute accumulation by bacteria. Comp. Biochem. Physiol. A 130:437-460. [DOI] [PubMed] [Google Scholar]
- 56.Zhao, G., and G. Zhang. 2005. Effect of protective agents, freezing temperature, rehydration media on viability of malolactic bacteria subjected to freeze-drying. J. Appl. Microbiol. 99:333-338. [DOI] [PubMed] [Google Scholar]