Abstract
CXCR4 expression on feline peripheral blood mononuclear cells (PBMC) was analyzed. While monocytes and B lymphocytes expressed CXCR4, no CXCR4 was detected on T lymphocytes, in stark contrast to the expression pattern on T lymphocytes from humans. In spite of the important role that CXCR4 plays in infection with feline immunodeficiency virus, expression on PBMC in vivo was unaffected by infection with either a primary or a cell culture-adapted virus strain.
The major pathway of infection with the primate lentiviruses involves attachment of the virus via a high-affinity interaction with CD4, followed by binding to a seven-transmembrane domain molecule, principally CXCR4 or CCR5. Consequently, viruses that utilize CXCR4 as a receptor preferentially target cell types expressing CXCR4 while those using CCR5 target CCR5-expressing cells. In humans, expression of CCR5 on peripheral blood T cells is restricted largely to CD4+/CD45RO+ cells (memory cells) while CXCR4 is expressed on both naive and memory T cells, albeit with higher levels on CD4+/CD45RA+ cells (naive cells) (3, 13, 14, 16, 18). Moreover, CXCR4 expression is not restricted to T cells; significant amounts of CXCR4 are found on B cells, while NK cells are effectively negative for expression (3, 13). Conversely, CCR5 is expressed on NK cells and not B cells. The differential expression of CXCR4 and CCR5 in the human immune system may provide a basis for the specific targeting of memory T cells early in infection (when CCR5-dependent strains predominate) and the wider range of cell types targeted with the emergence of CXCR4-dependent strains in the later stages of infection (3, 23). Previous studies demonstrated that feline immunodeficiency virus (FIV) utilizes the feline homologue of CXCR4 as a receptor (21, 22). The envelope glycoprotein from cell culture-adapted strains of FIV binds with high affinity to CXCR4-expressing cells (11), and infection with both cell culture-adapted and primary strains of FIV is blocked by CXCR4 antagonists (9-11, 17, 19). Given the restricted expression of CXCR4 and CCR5 in the human immune system and how this affects the in vivo tropism of human immunodeficiency virus (HIV), it is important to determine the expression pattern of CXCR4 in the domestic cat in order to elucidate the in vivo cell tropism of FIV and ultimately the pathogenesis of FIV-related immunodeficiency.
CXCR4 expression in vivo.
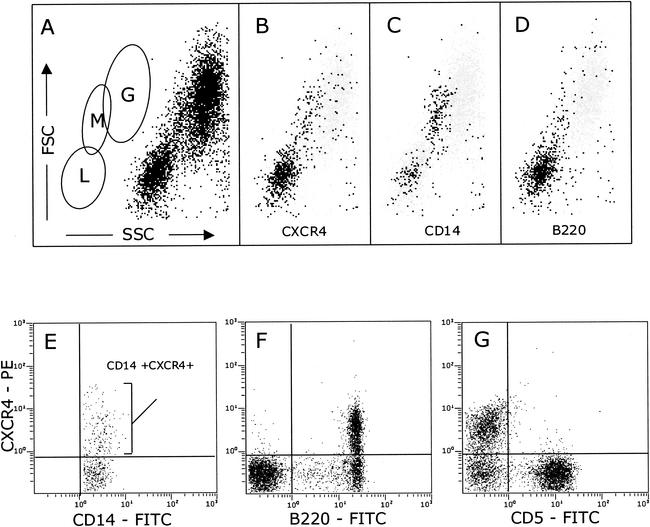
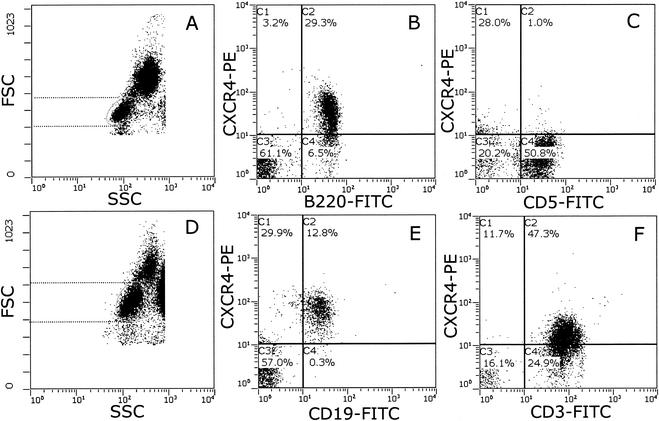
We used cross-species-reactive anti-human CXCR4 monoclonal antibodies (44701, 44717, and 44718; provided by M. Tsang, R&D Systems, Minneapolis, Minn.) that we identified previously (11) and two-color flow cytometry to examine CXCR4 expression on feline peripheral blood mononuclear cells (PBMC) (Fig. 1). Feline leukocytes were prepared by whole-blood lysis (20) and analyzed on an EPICS Elite flow cytometer (Beckman Coulter, Sheffield, United Kingdom). Lymphocyte, monocyte, and granulocyte populations were identified on the basis of their forward scatter (FSC) versus their 90° side scatter (SSC) characteristics (Fig. 1A). CXCR4 expression resided largely within the lymphoid gate (L), with a distinct population clustering in the region (M) consisting predominantly of monocytes (Fig. 1B). In order to confirm the identity of the monocyte population, the cells were dually labeled with antibodies recognizing CXCR4 (44718) and CD14 (Tük4; Dako Ltd., Ely, United Kingdom), a marker expressed at high levels on both murine and human monocytes but also to a lesser degree on a subpopulation of human B cells (12). CD14 expression resided largely within the expected monocyte gate (Fig. 1C), with a minor population of lymphocytes expressing CD14, consistent with feline CD14 having an expression pattern similar to that of human CD14. To further confirm the validity of the monocyte gate, a parallel sample was stained with the B-cell marker B220 (CD45R; Becton Dickinson UK Ltd., Cowley, United Kingdom). Expression of B220 was restricted predominantly to the lymphoid gate (Fig. 1D) and did not correlate with the monocyte population identified by CD14 expression. Thus, monocytes could be reliably identified on the basis of FSC versus SSC characteristics (monocyte [M] gate, Fig. 1A) in conjunction with CD14 expression. The expression of CXCR4 on CD14+ monocytes was variable; however, expression was detected consistently on monocytes from all of the cats tested (Fig. 1E). We next examined the expression of CXCR4 on lymphoid cells. Most of the CXCR4 expression resided in the B220+ population (B cells, Fig. 1F). While the intensity of CXCR4 expression varied among cats, the level of expression was consistent among analyses. CD5+ lymphocytes (in the domestic cat, CD5 expression is restricted to T cells [1, 4]) were ostensibly negative for CXCR4 (Fig. 1G). Similar findings were obtained by using antibodies against feline CD4, CD8, and CD3 (data not shown). Anti-CXCR4 antibodies 44701 and 44717 (R&D Systems) yielded findings similar to those obtained with antibody 44718 (data not shown), suggesting that the low level of reactivity with feline T cells is unlikely to result from the expression of a distinct antigenic conformation of CXCR4 on these cells. In order to compare the intensities of CXCR4 expression on feline and human lymphocytes, we processed samples of feline and human blood in parallel for flow cytometry (Fig. 2). While CXCR4 expression was detected on both feline (Fig. 2B, B220+) and human (Fig. 2E, CD19+) B cells, only human T cells (Fig. 2F, CD3+), and not feline T cells (Fig. 2C, CD5+), expressed detectable levels of CXCR4. Although human lymphocytes (Fig. 2D) were observed to be larger than feline lymphocytes (Fig. 2A), the smaller surface area of the feline cells was insufficient to account for the failure to detect CXCR4 expression on feline T cells. Thus, while the CXCR4 expression pattern on both B cells and monocytes in cats similar to the expression pattern in humans, it differs markedly in that CXCR4 is undetectable on peripheral T cells. It is intriguing that the human and feline viruses induce such similar disease conditions while the major (co)receptor distribution varies between the two species.
FIG. 1.
CXCR4 expression on feline PBMC. (A) Discrimination of lymphocytes (L), monocytes (M), and granulocytes (G) by FSC versus 90° SSC. CXCR4+ (B), CD14+ (C), and B220+ (D) cells were back gated on a plot of FSC versus SSC. The results of a two-color analysis of CXCR4 expression (phycoerythrin [PE]) versus CD14 (E), B220 (F), or CD5 (G) coupled to fluorescein isothiocyanate (FITC) is shown. Each panel represents 10,000 events. Data were analyzed with Expo ADC software (Applied Cytometry Systems, Sheffield, United Kingdom).
FIG. 2.
Comparison of CXCR4 expression on feline T cells with that on human T cells. Feline and human PBMC were processed for flow cytometry by whole-blood lysis. Lymphocytes were identified on the basis of FSC versus 90° SSC characteristics (dotted lines illustrate the respective sizes of feline [A] and human [D] lymphocytes, and the ellipse delineates the analysis gate). The results of a two-color analysis of CXCR4 expression (phycoerythrin [PE]) versus B220 (B) and CD5 (C) on feline lymphocytes and CD19 (E) and CD3 (F) on human lymphocytes is shown. Each panel represents 10,000 events. Data are presented with no baseline offset. FITC, fluorescein isothiocyanate.
CXCR4 expression in FIV-infected cats.
CXCR4 expression is down-modulated on some T-cell subsets from HIV-infected humans (15). Given that CXCR4 expression is down-regulated on FIV-infected cells in vitro (11), we investigated whether FIV infection affects CXCR4 expression in vivo. CXCR4 expression was examined in three groups of three age-matched animals that were uninfected (control) or had been infected for 2 years with either a primary strain of FIV (GL8414) or a cell culture-adapted strain of FIV (PETF14). Mean CXCR4 expression (percent positive or fluorescence intensity) was remarkably consistent among the three study groups (Table 1, group 1), although a wide range of values was observed, in agreement with chemokine receptor expression on human PBMC (13). Further, CXCR4 expression was consistent among three separate analyses (data not shown). Given that it was not possible to analyze CXCR4 expression prior to infection in these animals, we monitored CXCR4 expression on T and B cells in a second group of four cats (vaccine study control group) during the early phase of infection with GL8414. CXCR4 expression was analyzed at 1 week prior to a challenge and at 2, 6, and 13 weeks postinfection (Table 1, group 2). CXCR4 expression did not vary significantly either CD5+ T cells or on B220+ B cells during the course of the analyses. Taken together, the data indicate that FIV infection does not induce gross alterations in the level of CXCR4 expression on feline PBMC.
TABLE 1.
CXCR4 expression on monocytes, B cells, and T cells in the domestic cata
| Cats | CXCR4+ lymphocytes
|
CD14+/CXCR4+ monocytes
|
||||
|---|---|---|---|---|---|---|
| CD5+
|
B220+
|
|||||
| % (range) | MFId (range) | % (range) | MFI (range) | % (range) | MFI (range) | |
| Group 1 | ||||||
| Control | 1.9 (0.9-3.7) | NAb | 28.1 (19.7-37.7) | 6.3 (3.0-11.1) | 11.0 (3.8-18.1) | 2.7 (2.2-3.1) |
| GL8414 infected | 1.4 (0.7-2.1) | NA | 30.8 (23.6-40.2) | 4.4 (3.7-4.8) | 22.0 (3.1-49.2) | 3.0 (2.7-3.4) |
| PETF14 infected | 2.1 (1.2-1.7) | NA | 31.4 (25.6-38.1) | 3.5 (2.8-4.0) | 16.4 (11.6-19.4) | 2.9 (2.8-3.0) |
| Group 2 | ||||||
| Preinfection | 2.2 (1.4-3.9) | NA | 29.8 (21.2-49.1) | 5.5 (5.1-6.3) | NDc | ND |
| 2 wk p.i.e | 0.3 (0.1-0.6) | NA | 29.2 (22.9-32.2) | 6.4 (5.3-8.4) | ND | ND |
| 6 wk p.i. | 2.2 (0.6-4.8) | NA | 33.6 (30.1-38.4) | 7.2 (5.4-8.9) | ND | ND |
| 13 wk p.i. | 0.9 (0.7-1.3) | NA | 28.9 (26.6-30.0) | 6.6 (5.1-9.4) | ND | ND |
The results shown are means (range).
NA, not applicable because of the low number of positives.
ND, not determined.
MFI, mean fluorescence intensity.
p.i., postinfection.
Infection with FIV is CXCR4 dependent (9, 11, 17, 22); thus, CXCR4 expression on feline monocytes is consistent with the notion that FIV is a monocyte-tropic virus (8). Similarly, the high level of CXCR4 expression on B cells is consistent the notion that B cells are a major reservoir of proviral DNA in chronic infection (5). However, the failure to detect CXCR4 expression on the majority of T lymphocytes in peripheral blood stands in contrast to the distribution of CXCR4 expression in humans (3, 13). While it is possible that CXCR4 may be expressed on resting feline T cells in an antigenic conformation that is not recognized by the CXCR4 antibodies used in this study, previous data have demonstrated that the same anti-CXCR4 antibodies used in this study (44701, 44717, and 44718) recognize human CXCR4 on both T and B cells (2) and feline CXCR4 on a range of cell types (11). Moreover, by using the same antibodies, we were able to detect CXCR4 expression on freshly isolated feline thymocytes and on a proportion of T cells within lymph nodes (data not shown), in support of a previous study showing CXCR4 expression on mitogen-activated feline T cells (17). It was shown recently that HIV-specific CD4+ T cells are infected preferentially by HIV in vivo (7), and given that the major reservoir of FIV in early infection is CD4+ lymphocytes, it is possible that in the early stages of infection, FIV infection is restricted to activated T cells within the lymphatic system. Thus, if the majority of CXCR4-expressing T cells within the lymph nodes are CD4+, then CD4+ T cells would be targeted selectively in early infection. Alternatively, early infection may require an interaction with either a non-CXCR4 receptor (6) or a primary receptor in addition to CXCR4, the expression of which is restricted to activated CD4+ lymphocytes. Further elucidation of the mechanism of FIV infection in vivo may reveal conservation of important host and viral factors between this animal lentivirus and its human counterpart.
Acknowledgments
This study was funded by The Wellcome Trust and National Institutes of Health grant AI049765.
We thank M. Tsang for providing anti-CXCR4 antibody.
REFERENCES
- 1.Ackley, C. D., and M. D. Cooper. 1992. Characterization of a feline T-cell-specific monoclonal antibody reactive with a CD5-like molecule. Am. J. Vet. Res. 53:466-471. [PubMed] [Google Scholar]
- 2.Baribaud, F., T. G. Edwards, M. Sharron, A. Brelot, N. Heveker, K. Price, F. Mortari, M. Alizon, M. Tsang, and R. W. Doms. 2001. Antigenically distinct conformations of CXCR4. J. Virol. 75:8957-8967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bleul, C. C., L. J. Wu, J. A. Hoxie, T. A. Springer, and C. R. Mackay. 1997. The HIV coreceptors CXCR4 and CCR5 are differentially expressed and regulated on human T lymphocytes. Proc. Natl. Acad. Sci. USA 94:1925-1930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brown, A. L., T. H. Dunsford, O. Jarrett, B. J. Willett, and M. J. Hosie. 2002. Demonstration of biological activity of CD40 ligand (CD154) in the domestic cat. Cytokine 17:140-148. [DOI] [PubMed] [Google Scholar]
- 5.Dean, G. A., G. H. Reubel, P. F. Moore, and N. C. Pedersen. 1996. Proviral burden and infection kinetics of feline immunodeficiency virus in lymphocyte subsets of blood and lymph node. J. Virol. 70:5165-5169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.deParseval, A., and J. H. Elder. 2001. Binding of recombinant feline immunodeficiency virus surface glycoprotein to feline cells: role of CXCR4, cell-surface heparans, and an unidentified non-CXCR4 receptor. J. Virol. 75:4528-4539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Douek, D. C., J. M. Brenchley, M. R. Betts, D. R. Ambrozak, B. J. Hill, Y. Okamoto, J. P. Casazza, J. Kuruppu, K. Kunstman, S. Wolinsky, Z. Grossman, M. Dybul, A. Oxenius, D. A. Price, M. Connors, and R. A. Koup. 2002. HIV preferentially infects HIV-specific CD4+ T cells. Nature 417:95-98. [DOI] [PubMed] [Google Scholar]
- 8.Dow, S. W., C. K. Mathiason, and E. A. Hoover. 1999. In vivo monocyte tropism of pathogenic feline immunodeficiency viruses. J. Virol. 73:6852-6861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Egberink, H. F., E. De Clercq, A. L. W. Van Vliet, J. Balzarini, G. J. Bridger, G. Henson, M. C. Horzinek, and D. Schols. 1999. Bicyclams, selective antagonists of the human chemokine receptor CXCR4, potently inhibit feline immunodeficiency virus replication. J. Virol. 73:6346-6352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Endo, Y., Y. Goto, Y. Nishimura, T. Mizuno, T. Watari, A. Hasegawa, T. Hohdatsu, H. Koyama, and H. Tsujimoto. 2000. Inhibitory effect of stromal cell derived factor-1 on the replication of divergent strains of feline immunodeficiency virus in a feline T-lymphoid cell line. Vet. Immunol. Immunopathol. 74:303-314. [DOI] [PubMed] [Google Scholar]
- 11.Hosie, M. J., N. Broere, J. Hesselgesser, J. D. Turner, J. A. Hoxie, J. C. Neil, and B. J. Willett. 1998. Modulation of feline immunodeficiency virus infection by stromal cell-derived factor. J. Virol. 72:2097-2104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Labeta, M. O., R. Landmann, J. P. Obrecht, and R. Obrist. 1991. Human B cells express membrane-bound and soluble forms of the CD14 myeloid antigen. Mol. Immunol. 28:115-122. [DOI] [PubMed] [Google Scholar]
- 13.Lee, B., M. Sharron, L. J. Montaner, D. Weissman, and R. W. Doms. 1999. Quantification of CD4, CCR5, and CXCR4 levels on lymphocyte subsets, dendritic cells, and differentially conditioned monocyte-derived macrophages. Proc. Natl. Acad. Sci. USA 96:5215-5220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mackay, C. R. 1999. Dual personality of memory T cells. Nature 401:659-660. [DOI] [PubMed] [Google Scholar]
- 15.Nicholson, J. K., S. W. Browning, R. L. Hengel, E. Lew, L. E. Gallagher, D. Rimland, and J. S. McDougal. 2001. CCR5 and CXCR4 expression on memory and naive T cells in HIV-1 infection and response to highly active antiretroviral therapy. J. Acquir. Immune Defic. Syndr. 27:105-115. [DOI] [PubMed] [Google Scholar]
- 16.Qin, S., J. B. Rottman, P. Myers, N. Kassam, M. Weinblatt, M. Loetscher, A. E. Koch, B. Moser, and C. R. Mackay. 1998. The chemokine receptors CXCR3 and CCR5 mark subsets of T cells associated with certain inflammatory reactions. J. Clin. Investig. 101:746-754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Richardson, J., G. Pancino, T. Leste-Lasserre, J. Schneider-Mergener, M. Alizon, P. Sonigo, and N. Heveker. 1999. Shared usage of the chemokine receptor CXCR4 by primary and laboratory-adapted strains of feline immunodeficiency virus. J. Virol. 73:3661-3671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sallusto, F., D. Lenig, C. R. Mackay, and A. Lanzavecchia. 1998. Flexible programs of chemokine receptor expression on human polarized T helper 1 and 2 lymphocytes. J. Exp. Med. 187:875-883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Willett, B. J., and M. J. Hosie. 1999. The role of the chemokine receptor CXCR4 in infection with feline immunodeficiency virus. Mol. Membr. Biol. 16:67-72. [DOI] [PubMed] [Google Scholar]
- 20.Willett, B. J., M. J. Hosie, J. J. Callanan, J. C. Neil, and O. Jarrett. 1993. Infection with feline immunodeficiency virus is followed by the rapid expansion of a CD8+ lymphocyte subset. Immunology 78:1-6. [PMC free article] [PubMed] [Google Scholar]
- 21.Willett, B. J., M. J. Hosie, J. C. Neil, J. D. Turner, and J. A. Hoxie. 1997. Common mechanism of infection by lentiviruses. Nature 385:587. [DOI] [PubMed] [Google Scholar]
- 22.Willett, B. J., L. Picard, M. J. Hosie, J. D. Turner, K. Adema, and P. R. Clapham. 1997. Shared usage of the chemokine receptor CXCR4 by the feline and human immunodeficiency viruses. J. Virol. 71:6407-6415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wu, L. J., W. A. Paxton, N. Kassam, N. Ruffing, J. B. Rottman, N. Sullivan, H. Choe, J. Sodroski, W. Newman, R. A. Koup, and C. R. Mackay. 1997. CCR5 levels and expression pattern correlate with infectability by macrophage-tropic HIV-1, in vitro. J. Exp. Med. 185:1681-1691. [DOI] [PMC free article] [PubMed] [Google Scholar]