Abstract
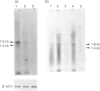
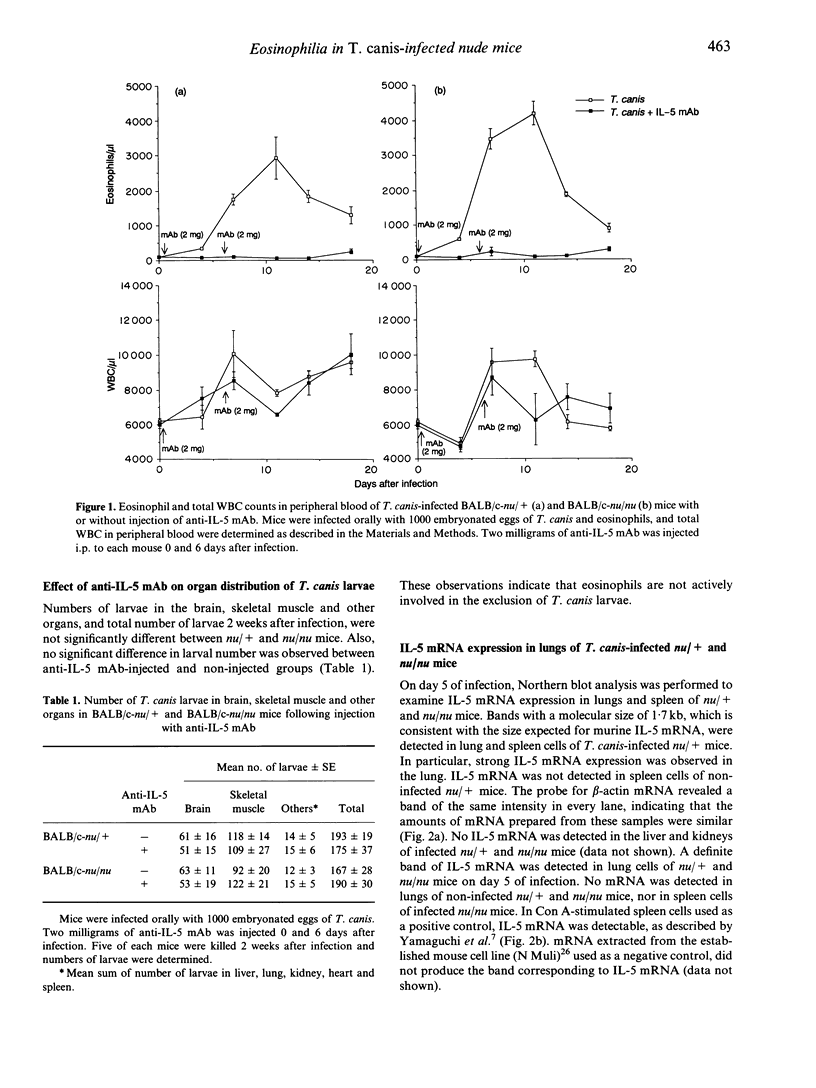
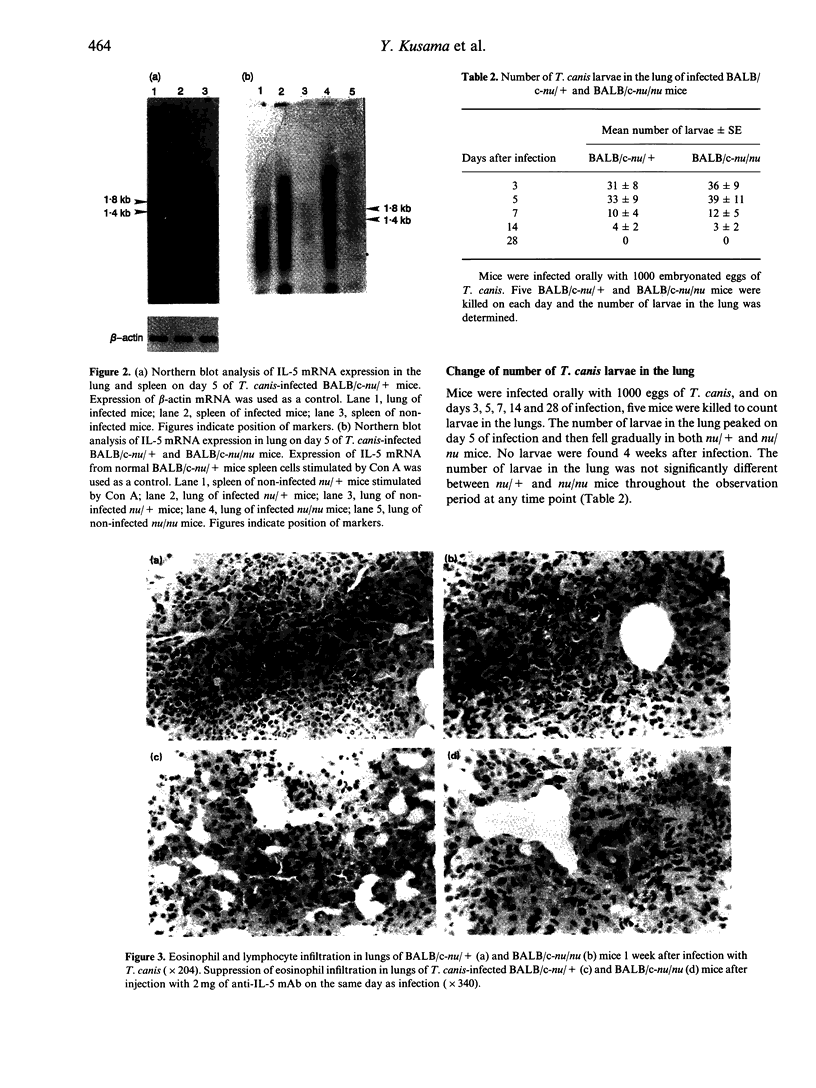
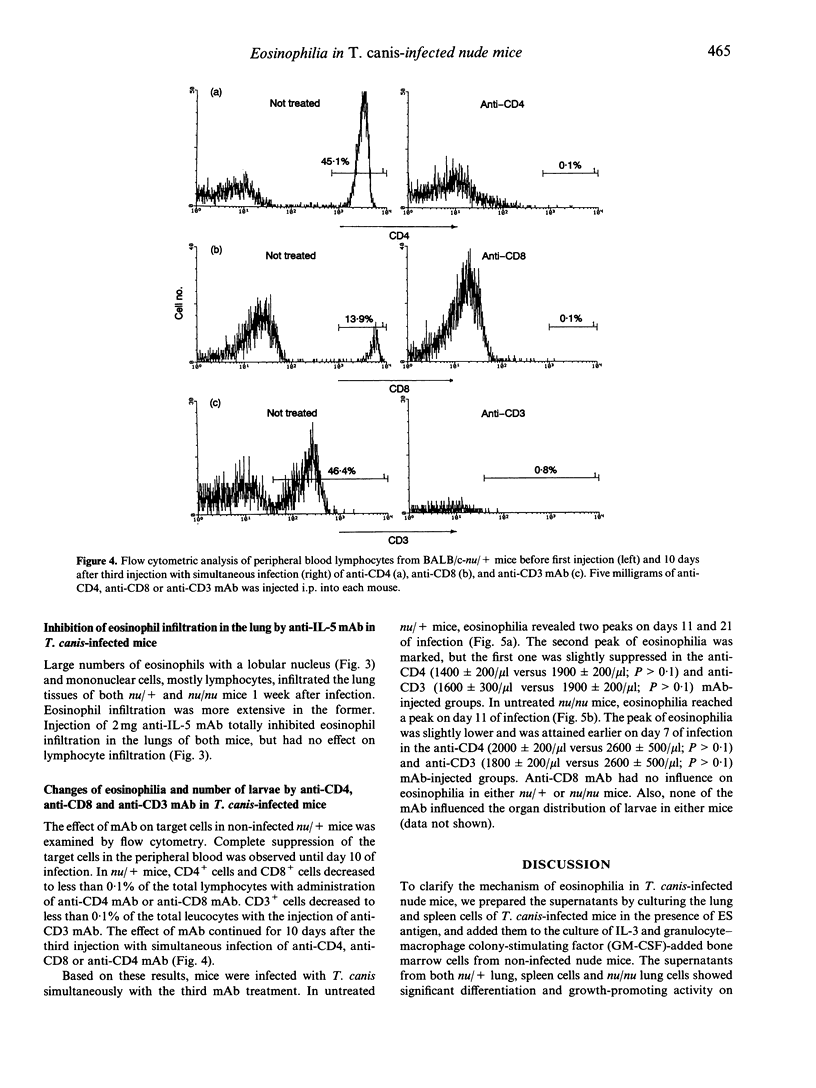
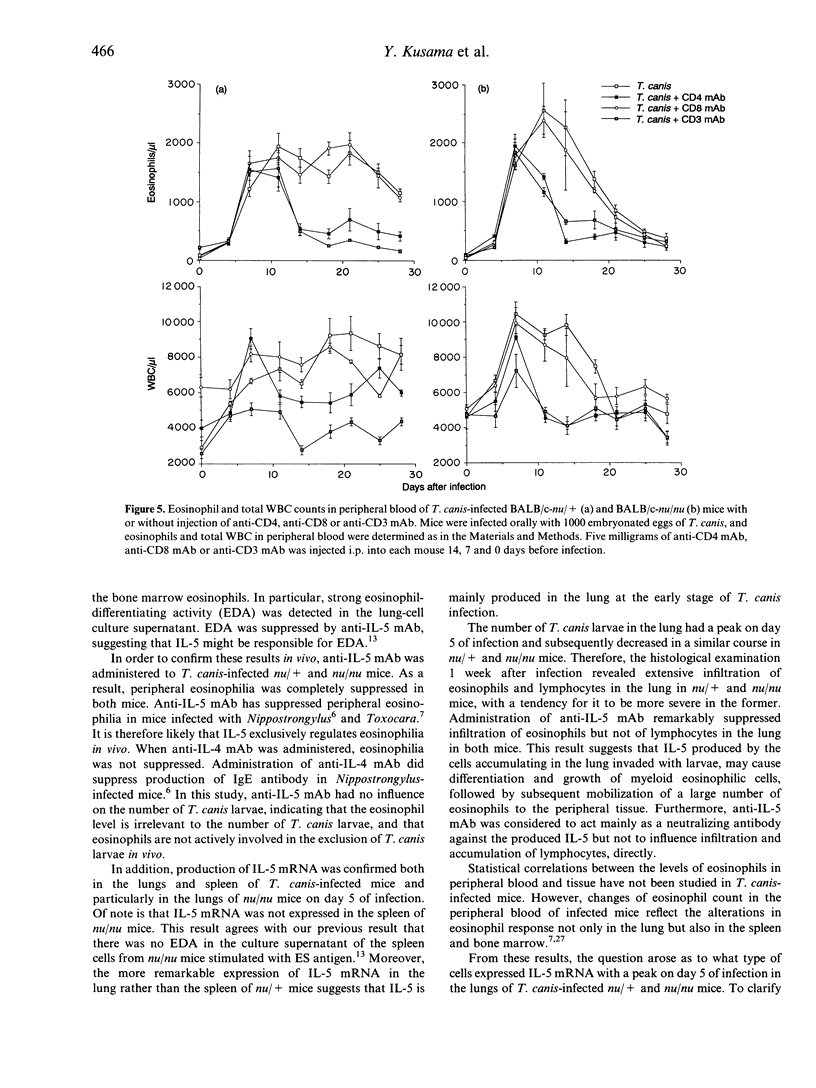
We studied the mechanism of eosinophilia in BALB/c-nu/+ (nu/+) and BALB/c-nu/nu (nu/nu) mice infected with Toxocara canis. Eosinophilia with two peaks on days 11 and 21 of infection was observed in infected nu/+ mice, and with a peak on day 11 in nu/nu mice. Interleukin-5 (IL-5) mRNA was expressed on day 5 of infection in the lung and spleen of nu/+ mice and in the lung of nu/nu mice, but not in the spleen of nu/nu mice. Large numbers of eosinophils and lymphocytes infiltrated the lung of both mice 1 week after infection. The number of larvae in the lung was the largest on day 5. Anti-IL-5 monoclonal antibody (mAb) treatment completely inhibited eosinophilia of both mice, with no change of larval distribution. Administration of anti-CD4 or anti-CD3 mAb markedly reduced the second peak of eosinophilia on day 21 of infection in nu/+ mice, and slightly reduced the first peak of eosinophilia on day 11 in both mice. Anti-CD8 mAb had no effect on the eosinophilia. These results suggest that eosinophilia in both mice is caused by IL-5, and that IL-5 is produced by cells other than CD4+ T cells, in addition to CD4+ T cells.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherwinski H. M., Schumacher J. H., Brown K. D., Mosmann T. R. Two types of mouse helper T cell clone. III. Further differences in lymphokine synthesis between Th1 and Th2 clones revealed by RNA hybridization, functionally monospecific bioassays, and monoclonal antibodies. J Exp Med. 1987 Nov 1;166(5):1229–1244. doi: 10.1084/jem.166.5.1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chomczynski P., Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987 Apr;162(1):156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- Cobbold S. P., Jayasuriya A., Nash A., Prospero T. D., Waldmann H. Therapy with monoclonal antibodies by elimination of T-cell subsets in vivo. Nature. 1984 Dec 6;312(5994):548–551. doi: 10.1038/312548a0. [DOI] [PubMed] [Google Scholar]
- Coffman R. L., Seymour B. W., Hudak S., Jackson J., Rennick D. Antibody to interleukin-5 inhibits helminth-induced eosinophilia in mice. Science. 1989 Jul 21;245(4915):308–310. doi: 10.1126/science.2787531. [DOI] [PubMed] [Google Scholar]
- Coffman R. L., Seymour B. W., Lebman D. A., Hiraki D. D., Christiansen J. A., Shrader B., Cherwinski H. M., Savelkoul H. F., Finkelman F. D., Bond M. W. The role of helper T cell products in mouse B cell differentiation and isotype regulation. Immunol Rev. 1988 Feb;102:5–28. doi: 10.1111/j.1600-065x.1988.tb00739.x. [DOI] [PubMed] [Google Scholar]
- Colley D. G. Variations in peripheral blood eosinophil levels in normal and Schistosoma mansoni-infected mice. J Lab Clin Med. 1974 Jun;83(6):871–876. [PubMed] [Google Scholar]
- Desreumaux P., Janin A., Colombel J. F., Prin L., Plumas J., Emilie D., Torpier G., Capron A., Capron M. Interleukin 5 messenger RNA expression by eosinophils in the intestinal mucosa of patients with coeliac disease. J Exp Med. 1992 Jan 1;175(1):293–296. doi: 10.1084/jem.175.1.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dialynas D. P., Quan Z. S., Wall K. A., Pierres A., Quintáns J., Loken M. R., Pierres M., Fitch F. W. Characterization of the murine T cell surface molecule, designated L3T4, identified by monoclonal antibody GK1.5: similarity of L3T4 to the human Leu-3/T4 molecule. J Immunol. 1983 Nov;131(5):2445–2451. [PubMed] [Google Scholar]
- Harada N., Takahashi T., Matsumoto M., Kinashi T., Ohara J., Kikuchi Y., Koyama N., Severinson E., Yaoita Y., Honjo T. Production of a monoclonal antibody useful in the molecular characterization of murine T-cell-replacing factor/B-cell growth factor II. Proc Natl Acad Sci U S A. 1987 Jul;84(13):4581–4585. doi: 10.1073/pnas.84.13.4581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy J. D., Pierce C. W., Lake J. P. Extrathymic T cell maturation. Phenotypic analysis of T cell subsets in nude mice as a function of age. J Immunol. 1992 Mar 15;148(6):1620–1629. [PubMed] [Google Scholar]
- Kinashi T., Harada N., Severinson E., Tanabe T., Sideras P., Konishi M., Azuma C., Tominaga A., Bergstedt-Lindqvist S., Takahashi M. Cloning of complementary DNA encoding T-cell replacing factor and identity with B-cell growth factor II. Nature. 1986 Nov 6;324(6092):70–73. doi: 10.1038/324070a0. [DOI] [PubMed] [Google Scholar]
- Ledbetter J. A., Rouse R. V., Micklem H. S., Herzenberg L. A. T cell subsets defined by expression of Lyt-1,2,3 and Thy-1 antigens. Two-parameter immunofluorescence and cytotoxicity analysis with monoclonal antibodies modifies current views. J Exp Med. 1980 Aug 1;152(2):280–295. doi: 10.1084/jem.152.2.280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leo O., Foo M., Sachs D. H., Samelson L. E., Bluestone J. A. Identification of a monoclonal antibody specific for a murine T3 polypeptide. Proc Natl Acad Sci U S A. 1987 Mar;84(5):1374–1378. doi: 10.1073/pnas.84.5.1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Limaye A. P., Abrams J. S., Silver J. E., Ottesen E. A., Nutman T. B. Regulation of parasite-induced eosinophilia: selectively increased interleukin 5 production in helminth-infected patients. J Exp Med. 1990 Jul 1;172(1):399–402. doi: 10.1084/jem.172.1.399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosmann T. R., Moore K. W. The role of IL-10 in crossregulation of TH1 and TH2 responses. Immunol Today. 1991 Mar;12(3):A49–A53. doi: 10.1016/S0167-5699(05)80015-5. [DOI] [PubMed] [Google Scholar]
- Nielsen K., Fogh L., Andersen S. Eosinophil response to migrating Ascaris suum larvae in normal and congenitally thymus-less mice. Acta Pathol Microbiol Scand B Microbiol Immunol. 1974 Dec;82(6):919–920. doi: 10.1111/j.1699-0463.1974.tb02394.x. [DOI] [PubMed] [Google Scholar]
- Ohara J., Paul W. E. Production of a monoclonal antibody to and molecular characterization of B-cell stimulatory factor-1. Nature. 1985 May 23;315(6017):333–336. doi: 10.1038/315333a0. [DOI] [PubMed] [Google Scholar]
- Owens R. B. Glandular epithelial cells from mice: a method for selective cultivation. J Natl Cancer Inst. 1974 Apr;52(4):1375–1378. doi: 10.1093/jnci/52.4.1375. [DOI] [PubMed] [Google Scholar]
- Phillips S. M., DiConza J. J., Gold J. A., Reid W. A. Schistosomiasis in the congenitally athymic (nude) mouse. I. Thymic dependency of eosinophilia, granuloma formation, and host morbidity. J Immunol. 1977 Feb;118(2):594–599. [PubMed] [Google Scholar]
- Ruitenberg E. J., Elgersma A., Kruizinga W., Leenstra F. Trichinella spiralis infection in congenitally athymic (nude) mice. Parasitological, serological and haematological studies with observations on intestinal pathology. Immunology. 1977 Oct;33(4):581–587. [PMC free article] [PubMed] [Google Scholar]
- Sugane K., Oshima T. Eosinophilia, granuloma formation and migratory behaviour of larvae in the congenitally athymic mouse infected with Toxocara canis. Parasite Immunol. 1982 Sep;4(5):307–318. doi: 10.1111/j.1365-3024.1982.tb00442.x. [DOI] [PubMed] [Google Scholar]
- Sugane K., Oshima T. Induction of a marked eosinophilia by cyclophosphamide in Toxocara canis infected SJL mice. Parasite Immunol. 1985 May;7(3):255–263. doi: 10.1111/j.1365-3024.1985.tb00074.x. [DOI] [PubMed] [Google Scholar]
- Svetić A., Madden K. B., Zhou X. D., Lu P., Katona I. M., Finkelman F. D., Urban J. F., Jr, Gause W. C. A primary intestinal helminthic infection rapidly induces a gut-associated elevation of Th2-associated cytokines and IL-3. J Immunol. 1993 Apr 15;150(8 Pt 1):3434–3441. [PubMed] [Google Scholar]
- Takamoto M., Sugane K. Mechanisms of eosinophilia in Toxocara canis infected mice: in vitro production of interleukin 5 by lung cells of both normal and congenitally athymic nude mice. Parasite Immunol. 1993 Sep;15(9):493–500. doi: 10.1111/j.1365-3024.1993.tb00636.x. [DOI] [PubMed] [Google Scholar]
- Tominaga A., Matsumoto M., Harada N., Takahashi T., Kikuchi Y., Takatsu K. Molecular properties and regulation of mRNA expression for murine T cell-replacing factor/IL-5. J Immunol. 1988 Feb 15;140(4):1175–1181. [PubMed] [Google Scholar]
- Yamaguchi Y., Matsui T., Kasahara T., Etoh S., Tominaga A., Takatsu K., Miura Y., Suda T. In vivo changes of hemopoietic progenitors and the expression of the interleukin 5 gene in eosinophilic mice infected with Toxocara canis. Exp Hematol. 1990 Dec;18(11):1152–1157. [PubMed] [Google Scholar]