Abstract
Objective
To describe the surgical techniques and early results of inferior vena cava (IVC) resection in patients with advanced liver tumors.
Summary Background Data
Involvement of the IVC by hepatic tumors, although rare, is considered inoperable by standard resection techniques. Concomitant hepatic and IVC resection is required to achieve adequate tumor clearance.
Methods
Between February 1995 and February 1999, 158 patients underwent hepatic resection for colorectal metastases in the authors’ unit. Eight patients, aged 42 to 80 years (mean 62 years), with hepatic metastases from colorectal cancer underwent concomitant resection of the IVC and four to six hepatic segments. Resections were carried out under total hepatic vascular exclusion in four patients and ex vivo in four patients. Between 30° and 360° of the retrohepatic IVC was resected and replaced with an autogenous vein patch (n = 1), a ringed Gore-Tex tube graft (n = 2), a Dacron tube graft (n = 1), or a patch (n = 3) or was repaired by primary suturing (n = 1).
Results
There were two early deaths from multiple organ failure. One patient survived 30 months after ex vivo resection but died of renal cell carcinoma, and another died with recurrent disease at 9 months. The remaining four patients remained alive 5 to 12 months after surgery, with no hepatic failure or venous obstruction; tumor recurrence was present in two. Nonthrombotic occlusion of the neocava occurred in one patient and was stented successfully.
Conclusions
Although concomitant hepatic and IVC resection is associated with a considerable surgical risk, this aggressive surgical approach offers hope for patients with hepatic tumors involving the IVC, who would otherwise have a dismal prognosis. This procedure can be performed under total hepatic vascular exclusion, with or without venovenous bypass, and by ex vivo bench resection.
Untreated, patients with hepatic metastases from colorectal carcinoma have a poor prognosis, with a median survival of less than 12 months. 1–3 Although chemotherapy may have a beneficial effect on the natural history of unresected hepatic metastases, 4 5-year survival is difficult to achieve by chemotherapy alone. 5 Hepatic resection, however, can achieve prolonged survival with an acceptable death rate in selected patients. The actuarial 5-year survival rate for patients undergoing resection is 25% to 50%, with a surgical death rate of approximately 5%. 6–11
Involvement of the hepatocaval confluence or inferior vena cava (IVC) by liver metastases is often considered a contraindication for hepatic resection and is associated with a dismal prognosis. However, the adoption of innovative surgical techniques, such as total hepatic vascular exclusion (HVE), 12–17 venovenous bypass, 18 and ex vivo hepatic resection, 19,20 has rendered such tumors operable. The resected IVC may then be replaced with an autogenous vein graft 21,22 or a prosthetic material. 23–25
This article reports our experience with concomitant hepatic and IVC resection in eight patients with locally advanced hepatic metastases of colorectal origin. The aforementioned surgical techniques were used to achieve resectability.
METHODS
From February 1995 to February 1999, 158 patients with colorectal liver metastases underwent liver resection at our hepatobiliary unit. All surgical procedures were carried out or supervised by the senior author (JPAL). In 18 patients (11%), IVC involvement by tumor was suspected from the preoperative imaging. In 10 of these patients, such suspicion was unwarranted: the tumor was adequately cleared off the IVC after liver mobilization using standard surgical techniques. In the remaining eight patients, initial mobilization confirmed the involvement of the IVC, and the patients underwent extended resections with IVC replacement.
Patient details are summarized in Table 1. There were six men and two women, with a mean age of 62 years (range 42–80 years). The primary adenocarcinoma was of rectosigmoid origin in five patients and right colonic in three patients. All tumors involved the muscle wall but had no detectable regional nodal metastases. The TNM staging was T2N0M0 in five patients and T2N0M1 in the three patients in whom liver metastasis was detected at the time of primary bowel surgery. Liver metastasis was diagnosed during follow-up (1–14 years) in six patients. One patient received adjuvant chemotherapy (5-fluorouracil and folinic acid) before the diagnosis of liver metastasis. Another patient received chemotherapy for bulky liver disease involving the IVC before undergoing hepatic resection, with a partial response. None of the eight patients received radiation therapy.
Table 1. DETAILS OF PATIENTS
CRC, colorectal cancer.
Preoperative Assessment
The location and extent of liver disease and involvement of the hepatic veins and IVC (Table 2) were assessed before surgery with intravenous contrast-enhanced CT (Fig. 1) and MRI. Absence of extrahepatic disease was assessed by colonoscopy and CT scanning of the abdomen, pelvis, and chest in all patients. Cardiovascular and pulmonary fitness for surgery was evaluated with an exercise ECG, chest radiography, and pulmonary function testing in all patients, and with coronary angiography in one patient with noninsulin-dependent diabetes mellitus and borderline ECG changes.
Table 2. DETAILS OF LIVER METASTASES AND SURGICAL PROCEDURES
IVC, inferior vena cava; PSVBP, portosystemic (portal vein & IVC → internal jugular) venovenous bypass; SVBP, systemic (IVC → internal jugular) venovenous bypass; HVE, hepatic vascular exclusion; MHV, middle hepatic vein; RV, renal vein

Figure 1. Preoperative CT scan (patient 7) demonstrated a large colorectal metastatic tumor that destroyed the right portal structures, abutted the left portal triad structures, and was in close apposition to the inferior vena cava.
The number of metastatic liver lesions was one in four patients, two in three patients, and three in one patient. Metastatic disease involved seven hepatic segments in two patients, six segments in two patients, five segments in two patients, four segments in one patient, and two segments in one patient. Liver lesions involved all three hepatic veins in four patients, the right and middle hepatic veins in two patients, and the right hepatic vein in one patient. Involvement of the IVC extended from 30° to 360° of its circumference in eight patients. Portal structures were involved in five patients.
Anesthetic Technique
All patients were anesthetized using an air-oxygen-desflurane–based technique to minimize derangement of postoperative liver function. 26,27 N-acetylcysteine and antioxidants were infused to confer hepatic protection. 28 The antifibrinolytic drug tranexamic acid was administered to maintain platelet function and prevent fibrinolysis resulting from the “anhepatic phase.”29–31 Venovenous bypass was used to maintain hemodynamic stability in the four patients in whom ex vivo liver and cava resection were performed. 18 In the remaining four patients, low right atrial and caval pressures were maintained immediately before and after vascular exclusion and hepatocaval resection to minimize venous backbleeding. This was facilitated by intraoperative use of thoracic epidural blockade and head-up tilt. Mean arterial pressure was maintained at more than 50 mmHg by phenylephrine infusion. After reperfusion of the liver, dopexamine was infused to optimize hepatosplanchnic blood flow. 32,33
Surgical Procedures
Surgery was performed through a transverse upper abdominal incision with upper midline extension to allow access using a mechanical retraction system (Omni-Tract Surgical, Division of Minnesota Scientific Inc., Minneapolis, MN). Biopsies from lymph nodes at the porta hepatis were obtained for frozen section examination; nodal metastases were excluded in all patients. Intraoperative ultrasound of the liver identified two additional lesions in already involved segments 7 and 8 in one patient (patient 1). Standard liver surgery techniques were used, with parenchymal transection using a Cavi-Pulse Ultrasonic Surgical Aspirator (CUSA, Model 200T, Valleylab, Boulder, CO).
The surgical procedures performed are summarized in Table 2. Right hepatectomy (resection of segments 5 through 8) was performed in three patients, with additional segment 3 metastasectomy in one patient. Right hepatic trisegmentectomy with caudate lobectomy (extended right hepatectomy; resection of segments 1 and 4 through 8) was carried out in four patients, with extension of resection across the falciform ligament to remove approximately 30% of segments 2 and 3 in three patients. One of the patients (patient 2) underwent a total hepatectomy with an ex vivo bench dissection and IVC excision and reconstruction with an autologous saphenous vein–IVC composite graft and subsequent reimplantation of parts of segments 2 and 3 using a flop technique. 34 Two patients (patients 7 and 8) underwent ex vivo resection of the involved hepatic segments and the IVC with reimplantation of the remaining parts of segments 2 and 3 on a 20-mm ringed polytetrafluoroethylene (PTFE) tube graft (Gore-Tex, Flagstaff, AZ) (Fig. 2). One patient (patient 1) underwent an ex vivo bench hepatectomy and IVC resection with preservation and reimplantation of segments 4b, 5, and 6 after reattachment of the right and middle hepatic veins to a 22-mm Dacron tube graft (Hemashield, Meadox Medical Inc., Oakland, NJ).
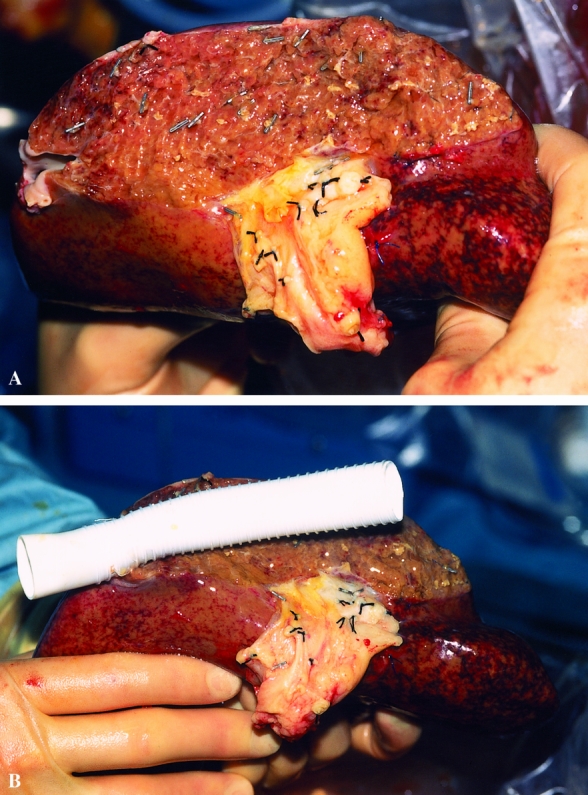
Figure 2. (A) The line of this ex vivo hepatic resection (patient 7) was to the left of the falciform ligament. Parts of segments 2 and 3 were removed en bloc with segments 1 and 4 through 8. Part of the left hepatic vein has been resected, and the left portal vein and hepatic artery have been divided at the division of segments 2 and 3. (B) The resected inferior vena cava was replaced with a 20-mm ringed polytetrafluoroethylene graft that was anastomosed to the left hepatic vein remnant before implantation.
IVC Reconstruction
A complete segment of the suprarenal IVC was resected in four patients and was reconstructed with either a synthetic interposition graft (three patients) or an autologous saphenous vein–IVC composite graft (one patient). In this instance, although all the hepatic veins were infiltrated by tumor, most of the IVC was spared. During the bench dissection, it was possible to remove the IVC and reuse some of it to create a tube that was augmented by a saphenous vein patch. The left hepatic vein remnant was reimplanted onto the side of the tube using continuous prolene (4-0) suture. Between 120° and 270° of the circumference of the suprarenal (hepatic) IVC was excised in three patients and was reconstructed using a Dacron patch. In one patient (patient 6), only 30° of the circumference of the IVC was resected; it was repaired with a simple suture technique using 3-0 prolene suture, without resorting to a patch repair.
The six patients with synthetic graft repair or replacement of the IVC received anticoagulation after surgery with warfarin (dicoumarin) to reduce the risk of vascular graft thrombosis. Most recently (patient 8), we constructed an arteriovenous fistula between the common femoral artery and vein using a 4-mm PTFE graft (IMPRA, Tempe, AZ) to increase caval blood flow. Although it may be possible to discontinue warfarin administration in this situation at approximately 3 months, we decided to continue it indefinitely if the patient suffers no side effects.
Vascular Exclusion Technique
Six of the eight procedures were carried out under total HVE. This involved a Pringle maneuver 35 with clamping of the IVC above and below the liver. Systemic venovenous (femoral to internal jugular vein) bypass became necessary in two patients who did not tolerate IVC clamping and became markedly hypotensive. Portosystemic venovenous bypass with arterial occlusion was instituted in the remaining four patients who underwent total hepatectomy with ex vivo resection of the liver and IVC, as for liver transplantation procedures (Fig. 3) . The removed liver was perfused with University of Wisconsin (UW) solution (ViaSpan, DuPont Merck, Wilmington, DE) and was maintained ex vivo in cold UW solution packed with ice for optimal preservation during bench dissection. Venovenous bypass was not necessary in one patient (patient 3) who underwent right hepatectomy with segment 3 metastasectomy and excision of the IVC. Although total HVE was not necessary in the latter patient, a Pringle maneuver was needed for 15 minutes during hepatic parenchymal dissection. Also, the IVC was clamped above the renal veins and below the preserved middle and left hepatic veins for 15 minutes to enable resection and reconstruction of part of the IVC. Hepatic blood flow was restored before the IVC reconstruction, with venous outflow reestablished via the left and middle hepatic veins (patient 3). HVE was also unnecessary in patient 6, in whom a side clamp was used on the IVC; nonetheless, the Pringle maneuver was necessary for 12 minutes during the parenchymal dissection.
Figure 3. Vascular isolation techniques. This diagram demonstrates the placement of vascular clamps and the extent of inferior vena cava resection (shaded area) in each case. Portosystemic venovenous bypass was used during ex vivo resections (patients 1, 2, 7, and 8). Total hepatic vascular isolation was used in patients 4 and 5. IVC, inferior vena cava; RHV, right hepatic vein; LHV, left hepatic vein; AV, adrenal vein; RV, renal vein; PV, portal vein; CHA, common hepatic artery.
Biliary Reconstruction
In patients who underwent ex vivo major liver resection (n = 4) or in situ radical right hepatic trisegmentectomy (n = 1), biliary reconstruction was performed with a Roux-en-Y hepaticojejunostomy because of tumor involvement of the biliary tree or to avoid tension and the potential for biliary stricture. The anastomoses were fashioned with a single layer of interrupted 3-0 or 4-0 Vicryl sutures to the common hepatic duct (n = 1) or the left hepatic duct (n = 4).
Follow-Up
The mean follow-up period after discharge from the hospital was 11 months (range 3–30 months). CT scanning of the abdomen for the detection of disease recurrence was performed every 6 months, or as dictated by the development of symptoms or by an increase in carcinoembryonic antigen blood levels.
RESULTS
Table 3 summarizes the main outcomes of surgery.
Table 3. OUTCOMES OF SURGERY
* A scalp metastasis was completely excised at 3 months; no evidence of disease elsewhere at 9 months.
Complications
The mean blood transfusion requirement was 8 units (range 0–25). Pleural effusions developed in four patients and required drainage under ultrasound guidance. Hepatocellular jaundice developed in two patients after right hepatectomy and right hepatic trisegmentectomy, with resolution within 2 to 3 weeks. Nonthrombotic obstruction of the IVC as a result of compression by regenerating liver developed in one patient 6 months after IVC reconstruction with a Dacron tube graft (patient 1). This was associated with the development of massive lower limb edema and ascites and was treated with percutaneous endoluminal stenting, with rapid resolution of all symptoms and clinical signs.
Death
Two patients (25%) died within 30 days or during the hospital admission for surgery. A 42-year-old woman who underwent total hepatectomy with ex vivo hepatic resection and reimplantation of segment 2 and part of segment 3 with IVC reconstruction experienced a left hemopneumothorax and rapid onset of respiratory and renal failure on the second postoperative day, requiring ventilation and hemodialysis. Doppler ultrasound suggested portal vein thrombosis. This was not substantiated, however, at intraoperative Doppler examination performed during a repeat laparotomy on the third postoperative day. Despite intensive support, the patient died on postoperative day 15. The second death occurred 39 days after right trisegmentectomy with IVC reconstruction in a 68-year-old man who received intensive preoperative chemotherapy. Despite a negative relaparotomy during the second postoperative week, the patient died of unexplained sepsis and multiple organ failure.
Histology
The IVC was histologically involved in six patients. It was adherent to, but not infiltrated with, tumor in two patients. In two cases where an ex vivo approach was used, in part because of bilateral portal triad structure involvement, histopathology analysis demonstrated the tumor mass to be 17×15 and 17×13 cm and the specimens weighed 1900 and 2040 g. In these cases, there was bile duct invasion in both the left and right hepatic ducts, but clear bile duct resection margins were obtained. Hepatic parenchymal resection margin involvement was seen in one ex vivo resection patient who subsequently received postoperative adjuvant chemotherapy (5-FU and folinic acid).
Disease Recurrence
All patients had undergone surgery for colorectal metastases. There have been two late deaths. Patient 1 developed a right renal adenocarcinoma (confirmed histologically) with infiltration of the IVC and pulmonary and spinal metastases 24 months postoperatively. There was no evidence of disease recurrence in the hepatic remnant. Radiologically guided fine-needle aspirates of lung lesions revealed poorly differentiated carcinoma cells, the origin of which could not be determined. The patient was treated with progesterone and chemotherapy, but died 30 months after liver resection. In patient 3, lung and peritoneal metastases developed 7 months after surgery and resulted in death at 11 months. His course was complicated by portal vein thrombosis at 5 months from celiac lymph node enlargement, and he was never fit enough for chemotherapy. The remaining four patients are alive, but two have evidence of recurrent disease in lungs (patient 4) and bone (patient 7). A small scalp lesion developed in patient 6 and was completely excised at 3 months. The latter patient showed no evidence of disease recurrence on CT scanning of the chest, abdomen, and pelvis at 6 months, and serum carcinoembryonic antigen concentration remained low at the 9-month follow-up.
Outcome of Patients With Suspected IVC Involvement Treated by Hepatectomy Without IVC Resection
Ten patients with colorectal hepatic metastases and suspected IVC involvement were found to have tumors that could be mobilized from the IVC and cleared by standard hepatectomy techniques; these procedures were also performed by the senior author (JPAL). Resections in this group were as follows: left trisegmentectomy (segments 2–5, 8 ± 1) in three patients; right trisegmentectomy (segments 4–8 ± 1) in four (three of these four also had segment 2 or 3 metastasectomy); right hepatectomy (segments 5–8) with segment 3 metastasectomy in one; left hepatectomy (segments 1–4) in one; and segment 1 and 7 resection in one. Of these, five have now died; the causes of death were peritoneal metastases at 2 months, lung metastases at 7 and 31 months, and liver metastases at 22 and 30 months (mean survival 18 months). Five patients remain alive: two disease-free at 9 and 32 months, one disease-free after lung resection at 32 months, one disease-free after a further liver resection at 12 months, and one with lung metastases not amenable to surgery at 25 months (mean follow-up 22 months). In 1 of the 10 patients (currently alive), the liver recurrence was in close proximity to the IVC but was excised without IVC resection.
DISCUSSION
Hepatic tumors in the central or posterior segments may involve the hepatic veins or may extend to involve the IVC, rendering liver resection by conventional techniques difficult. Attempted resection may result in substantial hemorrhage or air embolism, with considerable compromise to safety, adequacy of resection, and curability. Control of blood flow through the IVC is therefore essential to facilitate resection and reconstruction. When involvement of the IVC is minimal (≤60° circumferentially and ≤2 cm longitudinally), control may be simply achieved by applying a side clamp to the retrohepatic cava (as in patient 6 of this series). More extensive involvement of the IVC, however, requires the use of total HVE. 12–14,17 This may be achieved by applying vascular clamps to the IVC below and above the liver (usually just above the right adrenal vein), with concomitant interruption of hepatic blood inflow using a Pringle maneuver. 35 Attention should be directed to the presence of an accessory left hepatic artery; if this is left unclamped, hepatic congestion or excessive blood loss may result. HVE may thus enable the resection to proceed in a bloodless surgical field. This approach is further facilitated by the tolerable prolonged periods (60–90 minutes) of continuous warm hepatic ischemia characteristic of the otherwise healthy livers in most patients with metastatic disease. 14,16,36–38
HVE, however, may significantly reduce cardiac output as a result of decreased venous return, possibly resulting in hemodynamic instability. 39 It was not tolerated in two of the four patients in this series who underwent in situ hepatectomy with IVC resection and repair. Although persistent hemodynamic intolerance may be overcome by clamping the supraceliac aorta, 14,40 we have avoided this maneuver because it may result in renal, intestinal, and spinal cord ischemic injury. Systemic venovenous bypass, however, may restore venous return and cardiac output; it was successfully used in the above two patients. We routinely place cannulas in the internal jugular and common femoral veins percutaneously after induction of anesthesia in patients who may require IVC clamping and possible systemic venovenous bypass.
Although total HVE may allow a bloodless surgical field, access and exposure remain limited in patients with extensive hepatic tumor infiltration. The limited access may compromise resection margins and impair the extent of surgery. The duration of normothermic ischemia imposed by total HVE that can be tolerated is a further limiting factor when an extended and complex resection is planned. These limitations may be overcome by the use of hypothermic liver perfusion. This enables liver resection to proceed in situ, 17 antesitum, 41,42 or ex vivo. 19,43 Hypothermic (4°C) hepatic perfusion using UW solution optimizes liver preservation 44,45 and increases the period of tolerable liver ischemia to 4 hours. 46 Use of these techniques may be hemodynamically tolerable and safe with portal and systemic venovenous bypass, 18 as in liver transplantation. Extensive liver resections may thus be performed under controlled conditions. Cardiopulmonary bypass with hypothermic circulatory arrest has also been used for resection of hepatic tumors that involve the hepatic veins or vena cava. 47 However, the radical and safe resections achievable with the techniques we described call into question the need for thoracotomy, cardioplegia, and systemic hypothermia in these patients, with their attendant complications. 48,49
Although in situ hypothermic perfusion and the antesitum technique are applicable to hepatic tumors involving the hepatocaval confluence or the retrohepatic vena cava, we used the ex vivo bench dissection technique in four patients for the following reasons:
• Tumor encroaching on the portal pedicle of the liver segments to be preserved (two patients)
• Resection and reconstruction of more than one hepatic vein (one patient)
• Extensive IVC replacement (three patients).
Our experience in liver transplantation has undoubtedly influenced our preferences. However, in two of the four patients, the use of in situ techniques may have compromised the resection margins at the portal pedicle. In fact, portal triad resection and reconstruction were required in these two patients, whether the procedure was performed in situ or ex vivo. This was confirmed on histopathologic examination. The combination of both portal and IVC resection and reconstruction negates the argument for any in situ approach in these situations, where there is a theoretical advantage of avoiding hepatic arterial and biliary anastomoses and their inherent risks.
Extension of hepatic tumors to involve the IVC may be suspected before surgery in the presence of lower extremity swelling and venous engorgement. 50 Involvement of the IVC in our patients, however, was suspected or demonstrated on preoperative CT scans and was more clearly delineated by MRI. MRI is currently our gold standard for the preoperative assessment of liver metastases and possible involvement of adjacent major vascular structures. 51 Theoretically, MRI may be a noninvasive alternative that may replace cavography 22,24 in the evaluation of involvement of the IVC. However (in our hands, at least), MRI has proved to have a relatively low specificity: in 10 of the 18 patients, we found that IVC resection was unnecessary. The absence of local recurrence of tumor in the IVC region in all but 1 of the 10 patients suggests that our approach of obtaining a surgically clear margin by peeling the tumor from the IVC whenever possible is correct. Further, in the patient with recurrence, repeat resection was possible at 1 year, and again IVC resection was not needed.
Although replacement of the resected IVC with an autogenous vein graft 21,22 carries advantages with regard to the risk of infection or thrombosis, this option may not be technically feasible, particularly if a relatively long segment of IVC is to be replaced. Synthetic vascular grafts are a useful alternative 24,25,52 and were used as tube grafts in three of our patients and as a patch in three others. Ring-enforced PTFE may be the material of choice 24,25,52 and is an alternative to Dacron; it is our current graft of choice. Although the risk of graft thrombosis may be reduced by postoperative anticoagulation, the value of anticoagulation remains questionable, 25 and the required duration of therapy is hard to determine. Various maneuvers have been suggested to avert this complication, including a groin arteriovenous fistula, 52 which we have used recently, and the placement of a graft smaller than the surrounding native cava 25 to increase IVC blood flow. These maneuvers, however, may not prevent graft compression and subsequent venous obstruction during liver regeneration. Percutaneous endovascular stent placement was effective in restoring the patency of the compressed Dacron caval graft in our patient. The use of a ring-enforced PTFE graft might have prevented this complication.
Despite the availability of innovative surgical techniques that render extensive hepatic resection and concomitant IVC replacement feasible, the surgical death and complication rates associated with this type of surgery remain considerable. Pichlmayr et al 19 reported a 33% death rate in a series of nine patients who underwent ex vivo hepatic resection. The death rate in our series was 25%. Nonetheless, in a recent series of aggressive surgical resection of hepatic metastases involving the IVC, Miyazaki et al 53 reported a 5-year survival rate of 22% after IVC resection, compared with a 27% survival rate in patients without IVC involvement. Ex vivo and in situ resection of the IVC for hepatic colorectal metastases offers an improved quality of life and a chance of survival in patients with hepatic tumors considered inoperable by standard resection techniques. Further improvements in long-term survival will depend on the use of effective adjuvant therapies.
Footnotes
Correspondence: J. Peter A. Lodge, MD, FRCS, St. James’s University Hospital, Beckett St., Leeds LS9 7TF, United Kingdom.
Accepted for publication November 10, 1999.
References
- 1.Bengtsson G, Carlsson G, Hafstrom L, Jonsson P. Natural history of patients with untreated liver metastases from colorectal cancer. Am J Surg 1981; 141:586–589. [DOI] [PubMed] [Google Scholar]
- 2.Lahr CJ, Soong S-J, Cloud G, et al. A multifactorial analysis of prognostic factors in patients with liver metastases from colorectal carcinoma. J Clin Oncol 1983; 1:720–726. [DOI] [PubMed] [Google Scholar]
- 3.Rougier P, Milan C, Lazoethes F, et al. Prospective study of prognostic factors in patients with unresected hepatic metastases from colorectal cancer. Br J Surg 1995; 82:1397–1400. [DOI] [PubMed] [Google Scholar]
- 4.Wagner JS, Adson MA, van Heerden JA, et al. The natural history of hepatic metastases from colorectal cancer: a comparison with resective treatment. Ann Surg 1984; 199:502–508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Isenberg J, Fischbach R, Kruger I, Keller HW. Treatment of liver metastases from colorectal cancer. Anticancer Res 1996; 16:1291–1295. [PubMed] [Google Scholar]
- 6.Iwatsuki S, Esquivel CO, Gordon RD, Starzl TE. Liver resection for metastatic colorectal cancer. Surgery 1986; 100:804–810. [PMC free article] [PubMed] [Google Scholar]
- 7.Nordlinger B, Quilichini MA, Parc R, et al. Hepatic resection from colorectal liver metastases: influence on survival of preoperative factors and surgery for recurrences in 80 cases. Ann Surg 1987; 205:256–263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fortner JG. Recurrence of colorectal cancer after hepatic resection. Am J Surg 1988; 155:378–382. [DOI] [PubMed] [Google Scholar]
- 9.Petrelli N, Gupta B, Piedmonte M, Herrera L. Morbidity and survival of liver resection for colorectal adenocarcinoma. Dis Colon Rectum 1991; 34:899–904. [DOI] [PubMed] [Google Scholar]
- 10.Rosen CB, Nagorney DM, Taswell HF, et al. Perioperative blood transfusion and determinant of survival after liver resection for metastatic colorectal carcinoma. Ann Surg 1992; 216:493–504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Scheele J, Stang R, Altendorf-Hofmann A, Paul M. Resection of colorectal liver metastases. World J Surg 1995; 19:59–71. [DOI] [PubMed] [Google Scholar]
- 12.Heaney JP, Stanton WK, Halbert DS, et al. An improved technique for vascular isolation of the liver. Experimental study and case reports. Ann Surg 1966; 163:237–241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fortner JG, Shiu MH, Kinne DW, et al. Major hepatic resection using vascular isolation and hypothermic perfusion. Ann Surg 1974; 180:644–652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huguet C, Nordlinger B, Galopin JJ, et al. Normothermic hepatic vascular exclusion for extensive hepatectomy. Surg Gynecol Obstet 1978; 147:689–693. [PubMed] [Google Scholar]
- 15.Bismuth H, Castaing D, Garden OJ. Major hepatic resection under total vascular exclusion. Ann Surg 1989; 210:13–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huguet C, Addario-Chieco P, Gavelli A, et al. Technique of hepatic vascular exclusion for extensive liver resection. Am J Surg 1992; 163:602–605. [DOI] [PubMed] [Google Scholar]
- 17.Berney T, Mentha G, Morel P. Total vascular exclusion of the liver for the resection of lesions in contact with the vena cava or the hepatic veins. Br J Surg 1998; 85:485–488. [DOI] [PubMed] [Google Scholar]
- 18.Shaw BW Jr, Martin DJ, Marquez JM, et al. Venous bypass in clinical liver transplantation. Ann Surg 1984; 200:524–534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pichlmayr R, Grosse H, Hauss J, et al. Technique and preliminary results of extracorporeal liver surgery (bench procedure) and of surgery on the in situ perfused liver. Br J Surg 1990; 77:21–26. [DOI] [PubMed] [Google Scholar]
- 20.Yanaga K, Kishikawa K, Shimada M, et al. Extracorporeal hepatic resection for previously unresectable neoplasms. Surgery 1993; 113:637–643. [PubMed] [Google Scholar]
- 21.Miller CM, Schwartz ME, Nishizaki T. Combined hepatic and vena caval resection with autogenous caval graft replacement. Arch Surg 1991; 126:106–108. [DOI] [PubMed] [Google Scholar]
- 22.Yamamoto H, Hayakawa N, Ogawa A, et al. Segmental resection and reconstruction of the inferior vena cava with an autogenous vein graft. Br J Surg 1997; 84:51. [PubMed] [Google Scholar]
- 23.Yagyu T, Shimizu R, Nishida M, et al. Reconstruction of the hepatic vein to the prosthetic inferior vena cava in right extended hemihepatectomy with ex situ procedure. Surgery 1994; 115:740–744. [PubMed] [Google Scholar]
- 24.Huguet C, Ferri M, Gavelli A. Resection of the suprarenal inferior vena cava. The role of prosthetic replacement. Arch Surg 1995; 130:793–797. [DOI] [PubMed] [Google Scholar]
- 25.Sarkar R, Eilber FR, Gelabert HA, Quinones-Baldrich WJ. Prosthetic replacement of the inferior vena cava for malignancy. J Vasc Surg 1998; 28:75–83. [DOI] [PubMed] [Google Scholar]
- 26.O’Beirne HA, Young Y, Thornton J, Bellamy MC. Desflurane versus isoflurane in liver transplantation: a comparison of outcomes. Br J Anaesth 1997; 79:132. [Google Scholar]
- 27.O’Riordan J, O’Beirne HA, Young Y, Bellamy MC. Effects of desflurane and isoflurane on splanchnic microsurgery during major surgery. Br J Anaesth 1997; 78:95–96. [DOI] [PubMed] [Google Scholar]
- 28.El-Din HH, Bellamy MC, Webster NR, Goode H. The effects of antioxidants on haemodynamics and coagulation following reperfusion in orthotopic liver transplantation (OLT). Liver Transplant Surg 1995; 1:418. [Google Scholar]
- 29.Kang YG, Lewis JH, Navalgund A, et al. Epsilon-aminocaproic acid for treatment of fibrinolysis during liver transplantation. Anesthesiology 1987; 66:766–773. [PMC free article] [PubMed] [Google Scholar]
- 30.Yassen KA, Bellamy MC, Sadek SA, Webster NR. Tranexamic acid reduces blood loss during orthotopic liver transplantation. Clin Transplant 1993; 7:453–458. [Google Scholar]
- 31.Cheema SP, Webster NR, Dunn F, Bellamy MC. Mediators of fibrinolysis in orthotopic liver transplantation. Clin Transplant 1996; 10:24–27. [PubMed] [Google Scholar]
- 32.Smithies M, Tai Hwei Yee, Jackson L, et al. Protecting the gut and liver in the critically ill: effects of dopexamine. Crit Care Med 1994; 22:789–795. [DOI] [PubMed] [Google Scholar]
- 33.Bellamy MC, Young Y, Guillou PJ, O’Beirne H. Effects of dopamine and dopexamine on splanchnic microcirculation during major surgery in humans. Br J Anaesth 1996; 76:587P. [DOI] [PubMed] [Google Scholar]
- 34.Dunn SP, Langham MR Jr, Marmon LM. A new approach to the left-lateral segment hepatic transplant. The flop. Transplantation 1990; 49:660–662. [DOI] [PubMed] [Google Scholar]
- 35.Pringle JH. Notes on the arrest of hepatic hemorrhage due to trauma. Ann Surg 1908; 48:541–549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nagasue N, Yukaya H, Ogawa Y, et al. Segmental and subsegmental resections of the cirrhotic liver under hepatic inflow and outflow occlusion. Br J Surg 1985; 72:565–568. [DOI] [PubMed] [Google Scholar]
- 37.Delva E, Camus Y, Nordlinger B, et al. Vascular occlusion for liver resections. Operative management and tolerance to hepatic ischemia: 142 cases. Ann Surg 1989; 209:211–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Huguet C, Gavelli A, Bona S. Hepatic resection with ischemia of the liver exceeding one hour. J Am Coll Surg 1994; 178:454–458. [PubMed] [Google Scholar]
- 39.Delva E, Barberousse JP, Nordlinger B, et al. Hemodynamic and biochemical monitoring during major liver resections with use of hepatic vascular exclusion. Surgery 1984; 95:309–318. [PubMed] [Google Scholar]
- 40.Stephen MS, Sheil AG, Thompson JF, et al. Aortic occlusion and vascular isolation allowing avascular hepatic resection. Arch Surg 1990; 125:482–485. [DOI] [PubMed] [Google Scholar]
- 41.Belghiti J, Dousset B, Sauvanet A, et al. Resultats preliminaires de l’exerese “ex situ” des tumeurs hepatiques: une place entre les traitements palliatifs et la transplantation? Gastroenterol Clin Biol 1991; 15:449–453. [PubMed] [Google Scholar]
- 42.Hannoun L, Delriviere L, Gibbs P, et al. Major extended hepatic resections in diseased livers using hypothermic protection: preliminary results from the first 12 patients treated with this new technique. J Am Coll Surg 1996; 183:597–605. [PubMed] [Google Scholar]
- 43.Forni E, Meriggi F. Bench surgery and liver autotransplantation. Personal experience and technical considerations. G Chir 1995; 16:407–413. [PubMed] [Google Scholar]
- 44.Kalayoglu M, Sollinger HW, Stratta RJ, et al. Extended preservation of the liver for clinical transplantation. Lancet 1988; 1:617–619. [DOI] [PubMed] [Google Scholar]
- 45.Todo S, Nery J, Yanag K, et al. Extended preservation of human liver grafts with UW solution. JAMA 1989; 261:711–714. [PMC free article] [PubMed] [Google Scholar]
- 46.Hannoun T, Borie D, Balladur P, et al. Resections hepatiques ex situ-in vivo. Technique et resultats preliminaires. Chirurgie 1992; 118:292–296. [PubMed] [Google Scholar]
- 47.Sener SF, Arentzen CE, O’Connor B, et al. Hepatic and vena cava resection using cardiopulmonary bypass with hypothermic circulatory arrest. Am Surg 1996; 62:525–528. [PubMed] [Google Scholar]
- 48.Utley JR. Cardiopulmonary bypass surgery. Curr Opin Cardiol 1992; 7:267–275. [DOI] [PubMed] [Google Scholar]
- 49.Taylor KM. Central nervous system effects of cardiopulmonary bypass. Ann Thorac Surg 1998; 66:S20–S24. [DOI] [PubMed] [Google Scholar]
- 50.Kieffer E, Bahnini A, Koskas F. Nonthrombotic disease of the inferior vena cava: surgical management of 24 patients. In: Bergan JJ, Yao JST, eds. Venous Disorders. Philadelphia: WB Saunders; 1991: 501–516.
- 51.Kane PA, Ayton V, Walters HL, et al. MnDPDP-enhanced MR imaging of the liver. Correlation with surgical findings. Acta Radiol 1997; 38:650–654. [DOI] [PubMed] [Google Scholar]
- 52.Bower TC, Nagorney DM, Toomey BJ, et al. Vena cava replacement for malignant disease: is there a role? Ann Vasc Surg 1993; 7:51–62. [DOI] [PubMed] [Google Scholar]
- 53.Miyazaki M, Ito H, Nakagawa K, et al. Aggressive surgical resection for hepatic metastases involving the inferior vena cava. Am J Surg 1999; 177:294–298. [DOI] [PubMed] [Google Scholar]






