Abstract
Cooperative induction of secondary anti-hapten antibody responses was studied by using non-cross-reactive carrier proteins, bacterial α-amylase (BαA), Taka-amylase A (TAA) and keyhole-limpet haemocyanin (KLH), and the 2,4-dinitrophenyl (DNP) and benzylpenicilloyl (BPO) groups as haptenic determinants.
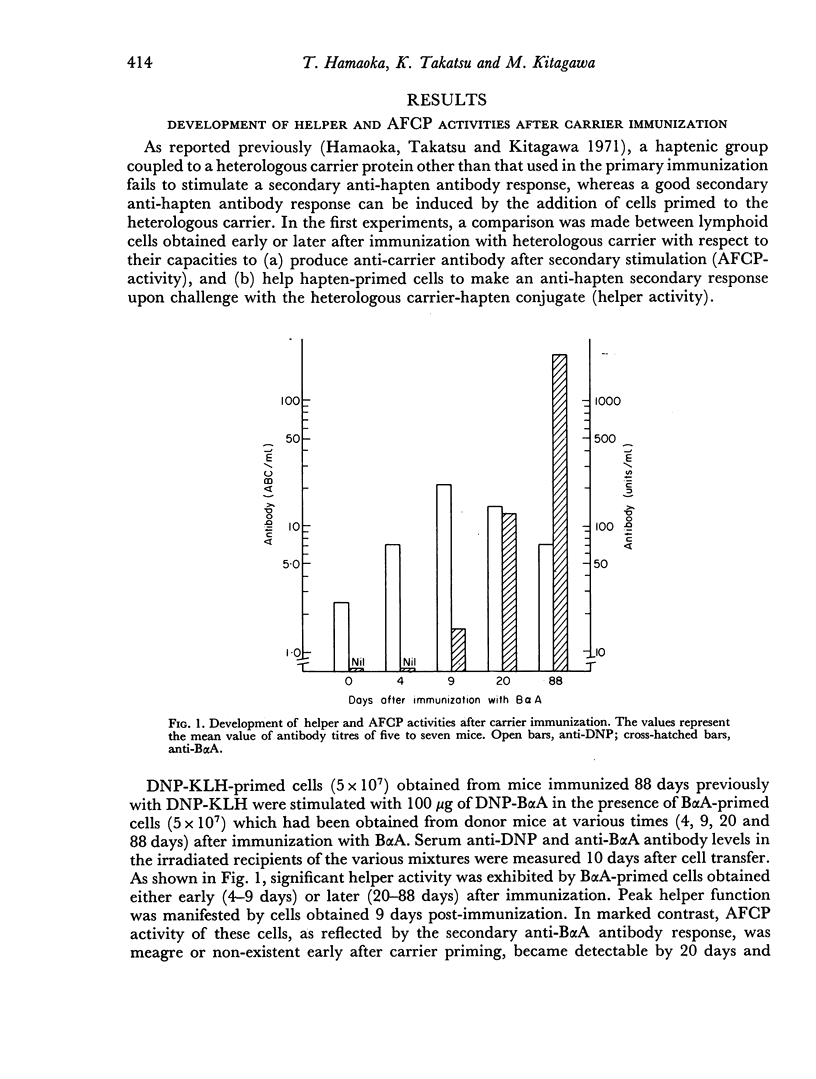
Lymphoid cells obtained from mice primed with a hapten-carrier conjugate could be effectively stimulated with a hapten-heterologous carrier conjugate, provided that lymphoid cells primed to the heterologous carrier were also present. In the carrier-primed lymphoid cell population, helper activity of thymus-derived cells developed earlier following carrier immunization than did the capacity of antibody-forming cell precursors (AFCP) to produce an effective anti-carrier antibody response upon secondary stimulation. Attempts to generate hapten-specific helper cell activity were unsuccessful. Thus, cells primed to one haptenic determinant failed to exert a helper function with cells primed to a second hapten upon subsequent administration to the two haptens together on the same heterologous carrier molecule.
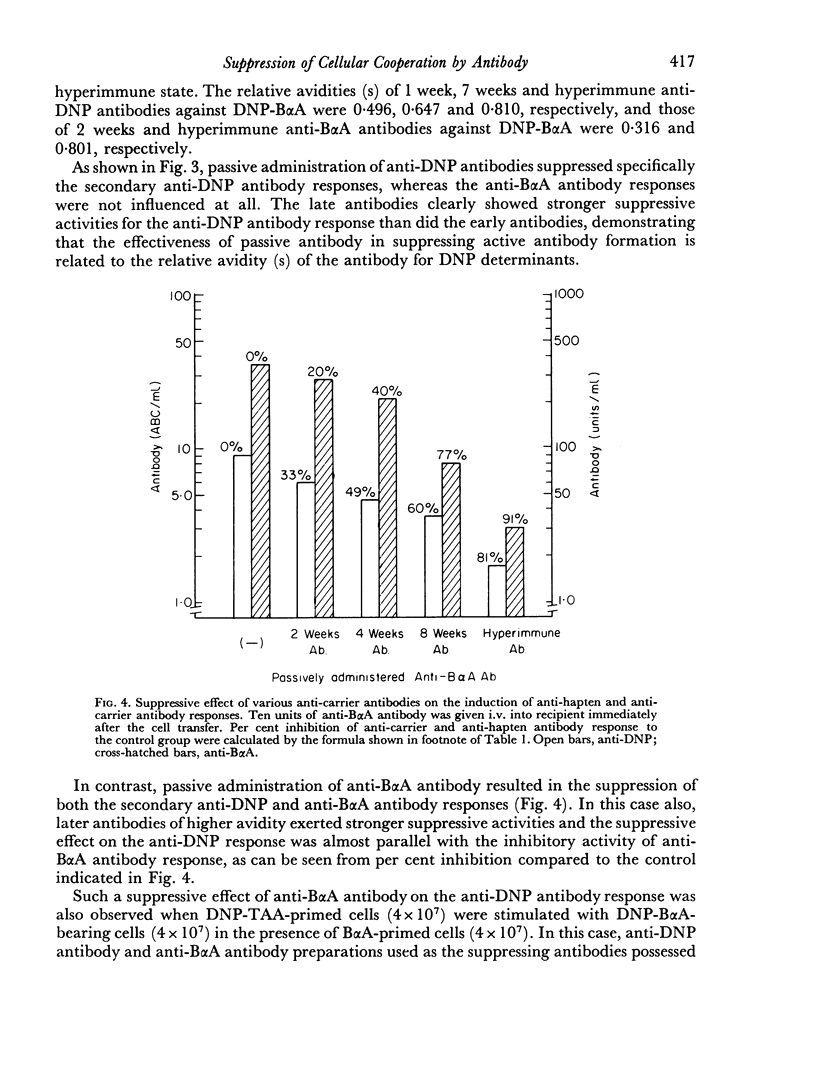
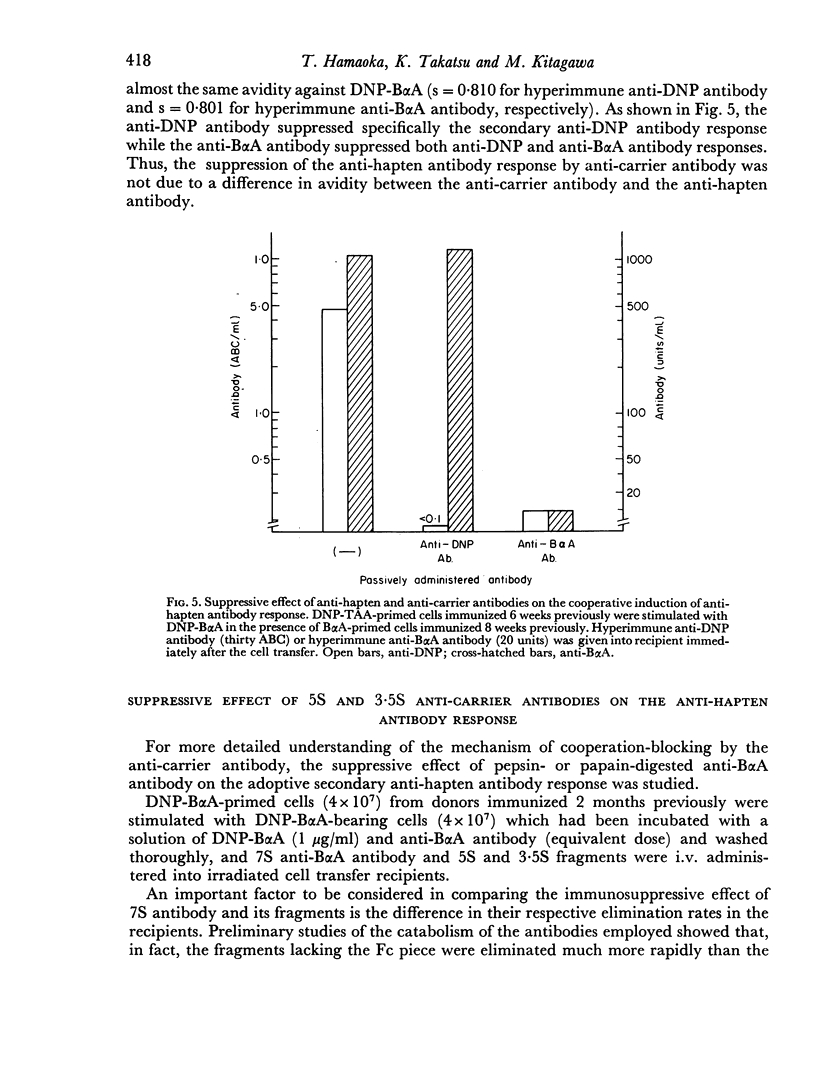
In order to distinguish among carrier determinant specificities which react with thymus-derived helper cells from those which react with bone marrow-derived AFCP, the capacity of various anti-carrier antibodies or antibody fragments to suppress either anti-carrier antibody production alone or together with helper cell function in adoptive secondary anti-hapten antibody responses was tested. In this system, it was found that 7S anti-carrier antibody suppressed the reaction of helper cells and carrier-specific AFCP such that both the anti-hapten and anti-carrier antibody responses were abrogated. By contrast, passively administered 3.5S fragments of anti-carrier antibodies selectively prevented the stimulation of carrier-specific AFCP to produce anti-carrier antibodies, but had no effect on the capacity of carrier-specific helper cells to facilitate the secondary anti-hapten antibody response. As expected, passively administered 7S anti-hapten antibodies selectively abrogated the production of anti-hapten, but not anti-carrier antibodies. These data are discussed in the context of suggesting that distinct determinant sites on carrier molecules are recognized independently by thymus-derived helper cells and by bone marrow-derived AFCP.
Full text
PDF












Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ada G. L. Antigen binding cells in tolerance and immunity. Transplant Rev. 1970;5:105–129. doi: 10.1111/j.1600-065x.1970.tb00358.x. [DOI] [PubMed] [Google Scholar]
- Basten A., Miller J. F., Warner N. L., Pye J. Specific inactivation of thymus-derived (T) and non-thymus-derived (B) lymphocytes by 125I-labelled antigen. Nat New Biol. 1971 May 26;231(21):104–106. doi: 10.1038/newbio231104a0. [DOI] [PubMed] [Google Scholar]
- Celada F., Schmidt D., Strom R. Determination of avidity of anti-albumin antibodies in the mouse. Influence of the number of cells transferred on the quality of the secondary adoptive response. Immunology. 1969 Aug;17(2):189–198. [PMC free article] [PubMed] [Google Scholar]
- Claman H. N., Chaperon E. A. Immunologic complementation between thymus and marrow cells--a model for the two-cell theory of immunocompetence. Transplant Rev. 1969;1:92–113. doi: 10.1111/j.1600-065x.1969.tb00137.x. [DOI] [PubMed] [Google Scholar]
- Feldmann M., Easten A. The relationship between antigenic structure and the requirement for thymus-derived cells in the immune response. J Exp Med. 1971 Jul 1;134(1):103–119. doi: 10.1084/jem.134.1.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greaves M. F. Biological effects of anti-immunoglobulins: evidence for immunoglobulin receptors on 'T' and 'B' lymphocytes. Transplant Rev. 1970;5:45–75. doi: 10.1111/j.1600-065x.1970.tb00356.x. [DOI] [PubMed] [Google Scholar]
- Hamaoka T., Kitagawa M. Antibody production in mice. II. The mechanism of antigenic stimulation in the secondary immune response. Immunology. 1971 Feb;20(2):191–203. [PMC free article] [PubMed] [Google Scholar]
- Hamaoka T., Kitagawa M., Matsuoka Y., Yamamura Y. Antibody production in mice. I. The analysis of immunological memory. Immunology. 1969 Jul;17(1):55–69. [PMC free article] [PubMed] [Google Scholar]
- Hamaoka T., Takatsu K., Masaki H., Matsuoka Y., Kitagawa M. Antibody production in mice. 3. The suppressive effect of antibody on the initiation of secondary immune response. Immunology. 1971 Jun;20(6):871–881. [PMC free article] [PubMed] [Google Scholar]
- Katz D. H., Benacerraf B. The regulatory influence of activated T cells on B cell responses to antigen. Adv Immunol. 1972;15:1–94. doi: 10.1016/s0065-2776(08)60683-5. [DOI] [PubMed] [Google Scholar]
- Katz D. H., Paul W. E., Goidl E. A., Benacerraf B. Carrier function in anti-hapten immune responses. I. Enhancement of primary and secondary anti-hapten antibody responses by carrier preimmunization. J Exp Med. 1970 Aug 1;132(2):261–282. doi: 10.1084/jem.132.2.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller J. F., Mitchell G. F. Thymus and antigen-reactive cells. Transplant Rev. 1969;1:3–42. doi: 10.1111/j.1600-065x.1969.tb00135.x. [DOI] [PubMed] [Google Scholar]
- Mitchison N. A. The carrier effect in the secondary response to hapten-protein conjugates. II. Cellular cooperation. Eur J Immunol. 1971 Jan;1(1):18–27. doi: 10.1002/eji.1830010104. [DOI] [PubMed] [Google Scholar]
- Mitchison N. A. The carrier effect in the secondary response to hapten-protein conjugates. V. Use of antilymphocyte serum to deplete animals of helper cells. Eur J Immunol. 1971 Apr;1(2):68–75. doi: 10.1002/eji.1830010204. [DOI] [PubMed] [Google Scholar]
- OVARY Z., BENACERRAF B. IMMUNOLOGICAL SPECIFICITY OF THE SECONDARY RESPONSE WITH DINITROPHENYLATED PROTEINS. Proc Soc Exp Biol Med. 1963 Oct;114:72–76. doi: 10.3181/00379727-114-28589. [DOI] [PubMed] [Google Scholar]
- Paul W. E. Functional specificity of antigen-binding receptors of lymphocytes. Transplant Rev. 1970;5:130–166. doi: 10.1111/j.1600-065x.1970.tb00359.x. [DOI] [PubMed] [Google Scholar]
- Paul W. E., Katz D. H., Goidl E. A., Benacerraf B. Carrier function in anti-hapten immune responses. II. Specific properties of carrier cells capable of enhancing anti-hapten antibody responses. J Exp Med. 1970 Aug 1;132(2):283–299. doi: 10.1084/jem.132.2.283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raff M. C. Role of thymus-derived lymphocytes in the secondary humoral immune response in mice. Nature. 1970 Jun 27;226(5252):1257–1258. doi: 10.1038/2261257a0. [DOI] [PubMed] [Google Scholar]
- Rajewsky K., Schirrmacher V., Nase S., Jerne N. K. The requirement of more than one antigenic determinant for immunogenicity. J Exp Med. 1969 Jun 1;129(6):1131–1143. doi: 10.1084/jem.129.6.1131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roelants G. E., Askonas B. A. Cell cooperation in antibody induction. The susceptibility of helper cells to specific lethal radioactive antigen. Eur J Immunol. 1971 Jun;1(3):151–157. doi: 10.1002/eji.1830010302. [DOI] [PubMed] [Google Scholar]
- Senyk G., Nitecki D., Goodman J. W. Immunogenicity of glucagon: determinants responsible for antibody binding and lymphocyte stimulation. Science. 1971 Jan 29;171(3969):407–408. doi: 10.1126/science.171.3969.407. [DOI] [PubMed] [Google Scholar]
- Taylor R. B. Cellular cooperation in the antibody response of mice to two serum albumins: specific function of thymus cells. Transplant Rev. 1969;1:114–149. doi: 10.1111/j.1600-065x.1969.tb00138.x. [DOI] [PubMed] [Google Scholar]
- Wigzell H. Specific fractionation of immunocompetent cells. Transplant Rev. 1970;5:76–104. doi: 10.1111/j.1600-065x.1970.tb00357.x. [DOI] [PubMed] [Google Scholar]



