Abstract
In vitro interaction of fluconazole and berberine chloride was investigated against 40 fluconazole-resistant clinical isolates of Candida albicans. Synergism in fungistatic activity was found with the checkerboard microdilution assay. The findings of agar diffusion tests and time-kill curves confirmed the synergistic interaction, but no antagonistic action was observed.
Candida albicans is the most common candidal pathogen, causing mucosal and invasive infections (7, 9, 23, 29). With the increasing clinical use of fluconazole, a choice for the treatment of C. albicans infections (3, 5, 12), fluconazole-resistant isolates are occurring more frequently (2, 19, 28). Attempts have been made to cope with treatment failures by using combination therapy (14). However, contradictory results of either synergic or antagonistic actions in various antifungal combinations have been reported (8, 11, 16, 27). As for fluconazole-resistant C. albicans, few data are available about the synergism of fluconazole with other antifungal agents (1, 4, 25).
Berberine, a bioactive herbal ingredient, was demonstrated to have weak activity against C. albicans and C. glabrata (17, 21, 26). Recently, berberine was combined with amphotericin B to treat disseminated candidiasis in mice (10). To seek a novel combination therapy, we investigated the in vitro interaction of fluconazole and berberine chloride (BBR) against fluconazole-resistant clinical isolates of C. albicans.
Strains and agents.
Forty clinical isolates of fluconazole-resistant C. albicans were used in this study, and C. albicans ATCC 90028 was used as a quality control. Drugs prepared in dimethyl sulfoxide (DMSO) included fluconazole (Pfizer-Roerig Pharmaceuticals, New York, NY) and BBR (Sigma-Aldrich, St. Louis, MO).
Checkerboard microdilution assay.
Assays were performed on all 40 isolates according to methods of the CLSI (formerly NCCLS) (M27-A) (6, 18). The initial concentration of fungal suspension in RPMI 1640 medium was 103 CFU/ml, and the final concentrations ranged from 0.125 to 64 μg/ml for fluconazole and 1 to 32 μg/ml for BBR. Plates were incubated at 35°C for 24 h. Optical density was measured at 630 nm, and background optical densities were subtracted from that of each well. Each isolate was tested in triplicate. MIC80 and MIC50 were determined as the lowest concentration of the drugs (alone or in combination) that inhibited growth by 80% or 50%, respectively, compared with that of drug-free wells. The fractional inhibitory concentration (FIC) index is defined as the sum of the MIC of each drug when used in combination divided by the MIC of the drug used alone. Synergy and antagonism were defined by FIC indices of ≤0.5 and >4, respectively. An FIC index result of >0.5 but ≤4 was considered indifferent (20).
Agar diffusion test.
C. albicans 0304103 (one fluconazole-resistant isolate with a MIC of >32 μg/ml for BBR) was tested by agar diffusion assay. A 100-μl aliquot of 106-CFU/ml suspension was spread uniformly onto the yeast extract-peptone-dextrose agar plate with or without 64 μg/ml fluconazole. Then, 6-mm paper disks impregnated with BBR and fluconazole alone or in combination were placed onto the agar surface. There was 5 μl of DMSO in control disks. Inhibition zones were measured after incubation at 35°C for 48 h. Assays were performed in duplicate.
Time-kill curves.
C. albicans 0304103 in RPMI 1640 medium was prepared at the starting inoculum of 103 CFU/ml or 105 CFU/ml (13). The concentrations were 16 μg/ml for BBR (a concentration of BBR without effect on the growth curve) and 10 μg/ml for fluconazole (in vivo achievable concentration of fluconazole) (16). DMSO comprised <1% of the total test volume. At predetermined time points (0, 12, 24, 36, and 48 h after incubation with agitation at 35°C), a 100-μl aliquot was removed from every solution and serially diluted 10-fold in sterile water. A 100-μl aliquot from each dilution was streaked on the Sabouraud dextrose agar plate. Colony counts were determined after incubation at 35°C for 48 h. The experiment was performed in triplicate. Synergism and antagonism were defined as a respective increase or decrease of ≥2 log10 CFU/ml in antifungal activity produced by the combination compared with that by the more active agent alone after 24 h, while a change of <2 log10 CFU/ml was considered indifferent (6).
The results of the checkerboard analysis are summarized in Table 1. The fluconazole-BBR combination markedly reduced MICs, especially the MIC80s of either individual agent. Synergism was observed in all 40 isolates (100%) in terms of MIC80s. The corresponding median FIC index was 0.034 (range, 0.017 to 0.127). When analyzed with MIC50s instead of MIC80s, the FIC index was generally higher (median, 0.148; range, 0.033 to 1.063). Of the 40 isolates, synergism was observed in 32 (80%), and indifference was observed in 8 (20%). Regardless of MIC endpoints, antagonism was not observed in the combination.
TABLE 1.
Interaction of fluconazole and BBR against 40 clinical isolates of C. albicans resistant to fluconazole by checkerboard microdilution assaya
| Parameter | MIC50 (μg/ml)
|
MIC80 (μg/ml)
|
||
|---|---|---|---|---|
| Median | Range | Median | Range | |
| Fluconazole | 8 | 0.5->64 | 128 | ≥64 |
| BBR | 64 | ≥32 | 64 | >32 |
| Fluconazole + BBRb | 0.5/1 | ≤0.125-1/1-4 | 1/2 | ≤0.125-2/1-8 |
| FIC index | 0.148 | 0.033-1.063 | 0.034 | 0.017-0.127 |
When analyzed with MIC50s, synergism was observed in 32 of 40 isolates (80%) and indifference was observed in 8 of the 40 isolates (20%); at MIC80, synergy was observed in all of the isolates.
MICs in combination are expressed as [fluconazole]/[BBR]. High off-scale MICs were converted to the next highest concentrations.
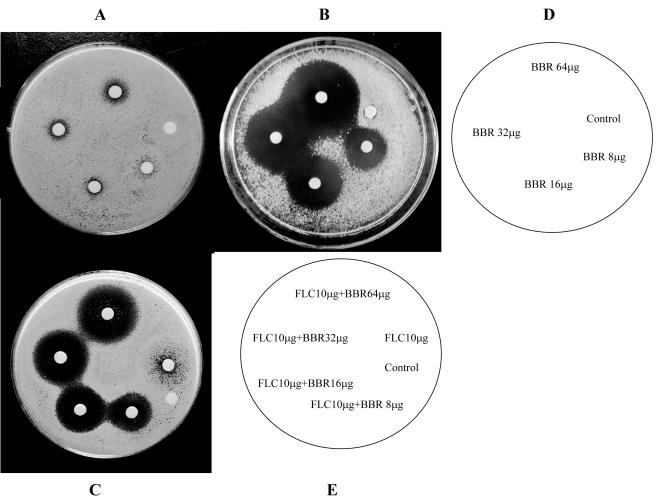
Agar diffusion tests visualized this synergistic interaction. BBR had no antifungal activity in smaller amounts and showed a weak antifungal activity at 64 μg (Fig. 1A). Fluconazole at 10 μg showed only weak inhibition (Fig. 1C). In contrast, BBR showed a powerful fungistatic effect on the agar plate containing 64 μg/ml fluconazole (Fig. 1B). The mean diameters of the inhibitory zones for 8, 16, 32, and 64 μg BBR increased to 15, 21, 28, and 36 mm, respectively. In addition, the combination of fluconazole and BBR yielded significantly clearer and larger zones than those of either drug alone on the plain agar plate (Fig. 1C). The sizes of the inhibition zones increased to 12, 17, 19, and 22 mm around the disks impregnated with 10 μg fluconazole plus different amounts of BBR (8, 16, 32, and 64 μg), respectively.
FIG. 1.
Agar disk diffusion assay of fluconazole (FLC) combined with BBR against C. albicans 0304103. Panels A and C show plain agar plates, and panel B shows an agar plate containing 64 μg/ml of fluconazole. Panel D describes the images for panels A and B, and panel E describes the image for panel C.
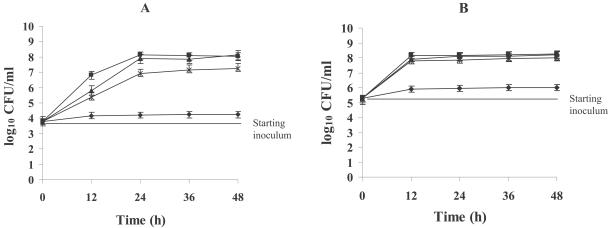
Synergism was confirmed in time-kill curves (Fig. 2). BBR did not affect the growth curve at 16 μg/ml after 24 h, regardless of the initial inoculum, and the fungistatic activity of fluconazole was dramatically enhanced by addition of BBR. Given the initial inoculum of 103 CFU/ml, the combination yielded a 2.7-log10-CFU/ml decrease compared with 10 μg/ml fluconazole alone at 24 h (Fig. 2A). No appreciable antifungal activity of fluconazole was observed under the starting inoculum of 105 CFU/ml (Fig. 2B). However, the fluconazole-BBR combination produced a 2.0-log10-CFU/ml decrease compared with the number of CFU produced by fluconazole alone at 48 h.
FIG. 2.
Time-kill curves of C. albicans 0304103 that were obtained by using initial inoculums of 103 CFU/ml (A) and 105 CFU/ml (B). ▪, growth control; ▴, BBR (16 μg/ml); *, fluconazole (10 μg/ml); ⧫, fluconazole (10 μg/ml) combined with BBR (16 μg/ml).
Our findings suggested that the combination of fluconazole and BBR produced potently synergistic action against fluconazole-resistant C. albicans in vitro. This interaction may be due to the inhibition of sterol 24-methyl transferase by BBR (21). In addition, the mechanism for resistance to azoles was reported to include mainly active efflux of drugs (with overexpressed CDR, MDR1, or FLU1) and target enzyme alterations (15, 22, 24). We postulated that BBR may exert the synergistic interaction with fluconazole by affecting some factor on resistance.
In conclusion, when used in combination with fluconazole, BBR is a promising and safe agent against fluconazole-resistant C. albicans in vitro but further study is needed to determine the underlying mechanism of the synergistic action. The potentiality of using this combination therapy in vivo warrants investigation.
Acknowledgments
This work was supported by grant 30300442 from the National Natural Science Foundation of China.
We thank Jun Gu of Changhai Hospital (Shanghai, China) for providing C. albicans strains.
REFERENCES
- 1.Barchiesi, F., L. Falconi Di Francesco, and G. Scalise. 1997. In vitro activities of terbinafine in combination with fluconazole and itraconazole against isolates of Candida albicans with reduced susceptibility to azoles. Antimicrob. Agents Chemother. 41:1812-1814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boken, D. J., S. Swindells, and M. G. Rinaldi. 1993. Fluconazole-resistant Candida albicans. Clin. Infect. Dis. 17:1018-1021. [DOI] [PubMed] [Google Scholar]
- 3.Brammer, K. W., P. R. Farrow, and J. K. Faulkner. 1990. Pharmacokinetics and tissue penetration of fluconazole in humans. Rev. Infect. Dis. 12(Suppl. 3):S318-S326. [DOI] [PubMed] [Google Scholar]
- 4.D'Auria, F. D., M. Tecca, R. Strippoli, and N. Simonetti. 2001. In vitro activity of propyl gallate-azole drug combination against fluconazole- and itraconazole-resistant Candida albicans strains. Lett. Appl. Microbiol. 32:220-223. [DOI] [PubMed] [Google Scholar]
- 5.Edwards, J. E., Jr., G. P. Bodey, R. A. Bowden, T. Buchner, B. E. de Pauw, S. G. Filler, M. A. Ghannoum, M. Glauser, R. Herbrecht, C. A. Kauffman, S. Kohno, P. Martino, F. Meunier, T. Mori, M. A. Pfaller, J. H. Rex, T. R. Rogers, R. H. Rubin, J. Solomkin, C. Viscoli, T. J. Walsh, and M. White. 1997. International conference for the development of a consensus on the management and prevention of severe candidal infections. Clin. Infect. Dis. 25:43-59. [DOI] [PubMed] [Google Scholar]
- 6.Eliopoulos, G. M., and R. C. Moellering, Jr. 1996. Antimicrobial combinations, p. 330-396. In V. Lorian (ed.), Antibiotics in laboratory medicine, 4th ed. The Williams & Wilkins Co., Baltimore, Md.
- 7.Fridkin, S. K., and W. R. Jarvis. 1996. Epidemiology of nosocomial fungal infections. Clin. Microbiol. Rev. 9:499-511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gil-Lamaignere, C., and F. M. Muller. 2004. Differential effects of the combination of caspofungin and terbinafine against Candida albicans, Candida dubliniensis and Candida kefyr. Int. J. Antimicrob. Agents 23:520-523. [DOI] [PubMed] [Google Scholar]
- 9.Gudlaugsson, O., S. Gillespie, K. Lee, J. Vande Berg, J. Hu, S. Messer, L. Herwaldt, M. Pfaller, and D. Diekema. 2003. Attributable mortality of nosocomial candidemia, revisited. Clin. Infect. Dis. 37:1172-1177. [DOI] [PubMed] [Google Scholar]
- 10.Han, Y., and J. H. Lee. 2005. Berberine synergy with amphotericin B against disseminated candidiasis in mice. Biol. Pharm. Bull. 28:541-544. [DOI] [PubMed] [Google Scholar]
- 11.Henry, K. W., M. C. Cruz, S. K. Katiyar, and T. D. Edlind. 1999. Antagonism of azole activity against Candida albicans following induction of multidrug resistance genes by selected antimicrobial agents. Antimicrob. Agents Chemother. 43:1968-1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kelly, S. L., A. Arnoldi, and D. E. Kelly. 1993. Molecular genetic analysis of azole antifungal mode of action. Biochem. Soc. Trans. 21:1034-1038. [DOI] [PubMed] [Google Scholar]
- 13.Klepser, M. E., E. J. Wolfe, R. N. Jones, C. H. Nightingale, and M. A. Pfaller. 1997. Antifungal pharmacodynamic characteristics of fluconazole and amphotericin B tested against Candida albicans. Antimicrob. Agents Chemother. 41:1392-1395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lewis, R. E., and D. P. Kontoyiannis 2001. Rationale for combination antifungal therapy. Pharmacotherapy 21:149S-164S. [DOI] [PubMed] [Google Scholar]
- 15.Lupetti, A., R. Danesi, M. Campa, M. Del Tacca, and S. Kelly. 2002. Molecular basis of resistance to azole antifungals. Trends Mol. Med. 8:76-81. [DOI] [PubMed] [Google Scholar]
- 16.Marchetti, O., P. Moreillon, M. P. Glauser, J. Bille, and D. Sanglard. 2000. Potent synergism of the combination of fluconazole and cyclosporine in Candida albicans. Antimicrob. Agents Chemother. 44:2373-2381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nakamoto, K., S. Sadamori, and T. Hamada. 1990. Effects of crude drugs and berberine hydrochloride on the activities of fungi. J. Prosthet. Dent. 64:691-694. [DOI] [PubMed] [Google Scholar]
- 18.National Committee for Clinical and Laboratory Standards. 1997. Reference method for broth dilution antifungal susceptibility testing of yeasts, vol. 17, no. 9. Approved standard M27-A. National Committee for Clinical and Laboratory Standards, Wayne, Pa.
- 19.Ng, T. T., and D. W. Denning. 1993. Fluconazole resistance in Candida in patients with AIDS—a therapeutic approach. J. Infect. 26:117-125. [DOI] [PubMed] [Google Scholar]
- 20.Odds, F. C. 2003. Synergy, antagonism, and what the chequerboard puts between them. J. Antimicrob. Chemother. 52:1. [DOI] [PubMed] [Google Scholar]
- 21.Park, K. S., K. C. Kang, J. H. Kim, D. J. Adams, T. N. Johng, and Y. K. Paik. 1999. Differential inhibitory effects of protoberberines on sterol and chitin biosyntheses in Candida albicans. J. Antimicrob. Chemother. 43:667-674. [DOI] [PubMed] [Google Scholar]
- 22.Perea, S., J. L. Lopez-Ribot, W. R. Kirkpatrick, R. K. McAtee, R. A. Santillan, M. Martinez, D. Calabrese, D. Sanglard, and T. F. Patterson. 2001. Prevalence of molecular mechanisms of resistance to azole antifungal agents in Candida albicans strains displaying high-level fluconazole resistance isolated from human immunodeficiency virus-infected patients. Antimicrob. Agents Chemother. 45:2676-2684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pfaller, M. A. 1996. Nosocomial candidiasis: emerging species, reservoirs, and modes of transmission. Clin. Infect. Dis. 22(Suppl. 2):S89-S94. [DOI] [PubMed] [Google Scholar]
- 24.Sanglard, D., F. Ischer, L. Koymans, and J. Bille. 1998. Amino acid substitutions in the cytochrome P-450 lanosterol 14α-demethylase (CYP51A1) from azole-resistant Candida albicans clinical isolates contribute to resistance to azole antifungal agents. Antimicrob. Agents Chemother. 42:241-253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sasaki, E., S. Maesaki, Y. Miyazaki, K. Yanagihara, K. Tomono, T. Tashiro, and S. Kohno. 2000. Synergistic effect of ofloxacin and fluconazole against azole-resistant Candida albicans. J. Infect. Chemother. 6:151-154. [DOI] [PubMed] [Google Scholar]
- 26.Vollekova, A., D. Kost'alova, V. Kettmann, and J. Toth. 2003. Antifungal activity of Mahonia aquifolium extract and its major protoberberine alkaloids. Phytother. Res. 17:834-837. [DOI] [PubMed] [Google Scholar]
- 27.Weig, M., and F. M. Muller. 2001. Synergism of voriconazole and terbinafine against Candida albicans isolates from human immunodeficiency virus-infected patients with oropharyngeal candidiasis. Antimicrob. Agents Chemother. 45:966-968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.White, T. C., K. A. Marr, and R. A. Bowden. 1998. Clinical, cellular, and molecular factors that contribute to antifungal drug resistance. Clin. Microbiol. Rev. 11:382-402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wilson, L. S., C. M. Reyes, M. Stolpman, J. Speckman, K. Allen, and J. Beney. 2002. The direct cost and incidence of systemic fungal infections. Value Health 5:26-34. [DOI] [PubMed] [Google Scholar]