Abstract
PPI-0903 is a new cephalosporin with broad-spectrum activity, including beta-lactam-resistant Streptococcus pneumoniae and Staphylococcus aureus. We used the neutropenic murine thigh and lung infection models to examine the pharmacodynamic characteristics of PPI-0903. Serum drug levels following four fourfold-escalating single doses of PPI-0903 were measured by microbiologic assay. In vivo postantibiotic effects (PAEs) were determined after doses of 1.56, 6.25, 25, and 100 mg/kg of body weight in mice infected with S. pneumoniae ATCC 10813, S. aureus ATCC 29213, or Escherichia coli ATCC 25922. Dose fractionation studies over a 24-h dose range of 0.39 to 1,600 mg/kg were administered every 3, 6, 12, or 24 hours. Nonlinear regression analysis was used to determine which pharmacokinetic-pharmacodynamic (PK-PD) index (total and free 65% drug) best correlated with CFU/thigh at 24 h. Similar to other beta-lactam antibiotics, PPI-0903 produced short to modest in vivo PAEs with either S. pneumoniae or E. coli. The percent time that serum concentrations were above the MIC (%T>MIC) was the PK-PD index that best correlated with efficacy (R2 = 84 to 88% for the three organisms, compared with 9 to 41% for peak/MIC and 30 to 82% for the area under the concentration-time curve/MIC). In subsequent studies we used the neutropenic murine thigh infection model to determine if the magnitude of the free-drug %T>MIC needed for efficacy of PPI-0903 varied among pathogens (including resistant strains). Mice infected with one of five isolates of S. pneumoniae, four isolates of S. aureus, or four gram-negative bacilli were treated for 24 h with 0.10 to 400 mg/kg of PPI-0903 every 6 h. A sigmoid dose-response model was used to estimate the doses (mg/kg/24 h) required to achieve a net bacteriostatic affect over 24 h and to produce a reduction in the burden of organisms from the start of therapy by 1 and 2 log10 CFU/thigh. MICs ranged from 0.008 to 1 μg/ml. Mean free-drug %T>MICs ± the standard deviation associated with the static effect endpoint for S. pneumoniae, S. aureus, and gram-negative isolates were 39 ± 9, 26 ± 8, and 32 ± 6, respectively. Methicillin and penicillin resistance did not alter the magnitude of free-drug %T>MIC required for efficacy. The free-drug %T>MIC necessary for efficacy was slightly reduced in animals with normal neutrophil counts. Treatment effect was similar in both the thigh and lung infection models. The pharmacodynamic characteristics of PPI-0903 are similar to those of other compounds within the cephalosporin class.
PPI-0903 is a new cephalosporin with broad-spectrum antimicrobial activity and is in early clinical development. PPI-0903 has enhanced potency against gram-positive cocci, including multiply drug-resistant Streptococcus pneumoniae and methicillin-resistant Staphylococcus aureus (10).
The goals of our experiments were to characterize the in vivo time course antimicrobial activity of PPI-0903 and determine the pharmacokinetic-pharmacodynamic (PK-PD) index and index magnitude predictive of efficacy to provide guidance for dosing regimen design in human studies.
MATERIALS AND METHODS
Bacteria, media, and antibiotics.
Five strains of Streptococcus pneumoniae (one penicillin susceptible, one penicillin intermediate, and three penicillin resistant), four strains of Staphylococcus aureus (two methicillin susceptible and two methicillin resistant), and four gram-negative bacilli (two Escherichia coli and two Klebsiella pneumoniae strains) were used for these experiments. All organisms except S. pneumoniae were grown, subcultured, and quantified in Mueller-Hinton (MH) broth (Difco Laboratories, Detroit, MI) and MH agar (Difco Laboratories, Detroit, MI). Sheep blood agar plates (Remel, Milwaukee, WI) were utilized for S. pneumoniae. The lower limit of organism quantification in these studies was 100 CFU/thigh or lung. Penicillin, methicillin, and cefotaxime were obtained from Sigma (St. Louis, MO). PPI-0903 (lot no. M599-007) and PPI-0903 M (bioactive metabolite of PPI-0903; lot no. M599-R10501) were manufactured by Takeda Chemical Industries (Japan).
In vitro susceptibility studies.
The MICs of PPI-0903 M, penicillin, methicillin, and cefotaxime for the various isolates were determined by standard Clinical Laboratory Standards Institute broth microdilution methods (15).
Murine thigh infection model.
Animals were maintained in accordance with the American Association for Accreditation of Laboratory Animal Care criteria (16). All animal studies were approved by the Animal Research Committee of the William S. Middleton Memorial VA Hospital.
Six-week-old, specific-pathogen-free, female ICR/Swiss mice weighing 23 to 27 g were used for all studies (Harlan Sprague-Dawley, Indianapolis, IN). Mice were rendered neutropenic (neutrophils < 100/mm3) by injecting cyclophosphamide (Mead Johnson Pharmaceuticals, Evansville, IN) intraperitoneally 4 days (150 mg/kg) and 1 day (100 mg/kg) before thigh infection. Previous studies have shown that this regimen produces neutropenia in this model for 5 days (4). A small group of mice were not treated with cyclophosphamide, in order to determine the impact of normal neutrophil counts on efficacy. Broth cultures of freshly plated bacteria were grown to logarithmic phase to an absorbance of 0.3 at 580 nm (Spectronic 88; Bausch and Lomb, Rochester, N.Y.). After a 1:10 dilution into fresh MH broth, bacterial counts of the inoculum ranged from 106.6 to 108.7 CFU/ml. Thigh infections with each of the isolates were produced by injection of 0.1 ml of inoculum into the thighs of halothane-anesthetized mice 2 h before therapy with PPI-0903.
Murine lung infection model.
Stationary-phase broth cultures of Klebsiella pneumoniae ATCC 43816 were obtained by overnight incubation. Cultures were centrifuged at 10,000 × g for 20 min and washed twice with 0.85% NaCl before being resuspending in 100 ml of 0.85% NaCl. Diffuse pneumonia in mice was produced by a 45-min exposure to an aerosol of 108.0 CFU/ml produced by a Collison nebulizer set at a rate of 4 to 5 liter/min. Antimicrobial therapy was initiated 14 h after the infection procedure. Two mice were used for each treatment regimen.
Drug pharmacokinetics.
Single-dose serum pharmacokinetic studies were performed in mice in the thigh infection model given subcutaneous doses (0.2 ml/dose) of PPI-0903 (1.56, 6.25, 25, and 100 mg/kg of body weight). For each of the doses examined, two groups of three mice were sampled by retroorbital puncture at 0.25- to 1-h intervals over 6 h (sample times included 0.25, 0.5, 1, 2, 4, and 6 h). Individual animals were sampled three or four times. The total volume collected from individual animals was less than 10% of the total blood volume. Samples were then centrifuged for 5 min at 10,000 × g, and serum was removed. Serum PPI-0903 concentrations were determined by microbiologic assay using E. coli NIH-J as the test organism and noble agar as the diffusion medium. The lower limit of detection of this assay was 0.12 μg/ml. Intraday variation was less than 7%. All pharmacokinetic studies were performed on the same day. Pharmacokinetic indices, including elimination half-life, area under the concentration-time curve (AUC), and peak level, were calculated using a noncompartmental model. For doses used in treatments for which actual kinetic measurements were not made, estimates were based upon linear extrapolation from the four studied dose levels. Protein binding in the serum of neutropenic infected mice was analyzed using ultrafiltration methods (9, 12). The degree of binding was measured using PPI-0903 concentrations of 10 and 100 μg/ml.
Treatment protocols. (i) In vivo time kill and PAEs.
Two hours after infection in the thigh with S. pneumoniae ATCC 10813, S. aureus ATCC 29213, or E. coli ATCC 25922, neutropenic mice were treated with single subcutaneous doses of PPI-0903 (1.56, 6.25, 25, or 100 mg/kg). Control growth was determined at four sampling times (two mice per time point) over 12 h (at 0, 2, 6, and 12 h). The treated groups (two mice per time point) were sampled nine times over 24 h (at 0, 1, 2, 4, 6, 9, 12, 18, and 24 h). The thighs were removed at each time point and processed immediately for CFU determination. The times that the levels of PPI-0903 (free drug) in the serum remained above the MIC (%T>MICs) for the organisms were calculated from the pharmacokinetic studies based upon a linear extrapolation from the peak serum level. The postantibiotic effect (PAE) was calculated by subtracting the time it took for organisms to increase 1 log in the thighs of saline-treated animals from the time it took organisms to grow the same amount in treated animals after serum drug levels fell below the MIC for the infecting organism (7). The formula for calculating the PAE was as follows: PAE = T − C, where C is the time for 1 log10 of control growth and T is the time for 1 log10 of treatment growth after levels had fallen below the MIC.
(ii) PK-PD index determination.
Neutropenic mice were infected with either penicillin-susceptible S. pneumoniae ATCC 10813, methicillin-resistant S. aureus ATCC 33591, or K. pneumoniae ATCC 43816. Treatment with PPI-0903 was initiated 2 h after infection. Groups of two mice were treated for 24 h with 20 different dosing regimens using twofold-increasing total doses divided into one, two, four, or eight doses. Total doses of PPI-0903 ranged more than 256-fold against each organism (0.39 to 100 mg/kg/24 h for S. pneumoniae and S. aureus and 6.25 to 1,600 mg/kg/24 h for K. pneumoniae). Drug doses were administered subcutaneously in 0.2-ml volumes. The mice were sacrificed after 24 h of therapy, and the thighs were removed and processed for CFU determination. Untreated control mice were sacrificed just before treatment and after 24 h.
(iii) PK-PD index magnitude studies.
Similar dosing studies using six fourfold-increasing PPI-0903 doses administered every 6 h were utilized to treat neutropenic animals infected in the thigh with five strains of S. pneumoniae (one penicillin susceptible, one with intermediate sensitivity, and three penicillin resistant), four strains of S. aureus (two methicillin susceptible and two methicillin-resistant [MRSA]), and four gram-negative strains (two Escherichia coli and two Klebsiella pneumoniae strains). The PPI-0903 MICs for the organisms studied varied 250-fold. The total daily dose of PPI-0903 used in these studies varied from 0.40 to 1,600 mg/kg/24 h. In treatment against the gram-negative organisms, the starting dose level was 1.56 mg/kg. For all other organisms, the starting dose level was 0.10 mg/kg.
Data analysis.
The results of these studies were analyzed using the sigmoid dose-effect model. The model is derived from the Hill equation: E = (Emax × DN)/(ED50N + DN), where E is the effect or in this case the log change in CFU per thigh or lung, comparing treated mice and untreated controls after the 24-hour period of study. Emax is the maximum effect. D is the 24-hour total dose. ED50 is the dose required to achieve 50% of the Emax, and N is the slope of the dose-effect curve. The indices Emax, ED50, and N were calculated using nonlinear least-squares regression. The correlation between efficacy and each of the three PK-PD indices studied (total and free-drug T>MIC, AUC/MIC, and peak/MIC) was determined by nonlinear least-squares multivariate regression (Sigma Stat; Jandel Scientific Software, San Rafael, CA). The coefficient of determination, or R2, was used to estimate the variance that could be due to regression with each of the PK-PD indices.
To allow a comparison of the potencies of PPI-0903 against a variety of organisms, we utilized the 24-h static dose and doses necessary to achieve 1 and 2 log10 kills. The magnitude of the PK-PD index associated with each endpoint dose was calculated from the following equation: log10 D = {log10 [E/(Emax − E)] + log ED50}/N, where E is control growth (D equals the dose), E = control growth plus 1 log when D = 1 log kill, or E = control plus 2 log for D = 2 log kill.
RESULTS
In vitro susceptibility testing.
The MICs of PPI-0903 M, penicillin, methicillin, and cefotaxime for the 13 study strains are shown in Table 1. PPI-0903 M MICs varied 250-fold (range, 0.008 to 2.0 μg/ml).
TABLE 1.
PPI-0903, penicillin, cefotaxime, and methicillin in vitro activities against selected isolates of S. pneumoniae, S. aureus, and Enterobacteriaceae
| Organism | MIC (mg/liter)
|
|||
|---|---|---|---|---|
| PPI-0903 | Penicillin | Cefotaxime | Methicillin | |
| S. pneumoniae 10813 | 0.008 | 0.008 | 0.12 | |
| S. pneumoniae 1199 | 0.06 | 1.0 | 0.25 | |
| S. pneumoniae 1329 | 0.06 | 2.0 | 0.5 | |
| S. pneumoniae 1325 | 0.06 | 2.0 | 0.5 | |
| S. pneumoniae 1293 | 0.12 | 2.0 | 0.5 | |
| S. aureus 25923 | 0.12 | 0.5 | 0.25 | |
| S. aureus 29213 | 0.25 | 1.0 | 0.5 | |
| MRSA | 0.50 | 8.0 | >8.0 | |
| S. aureus 33591 | 1.0 | >64 | >8.0 | |
| E. coli 25922 | 1.0 | 0.03 | ||
| E. coli 1894-1 | 2.0 | 0.03 | ||
| K. pneumoniae 43816 | 2.0 | 0.03 | ||
| K. pneumoniae 216 | 2.0 | 0.03 | ||
Pharmacokinetics.
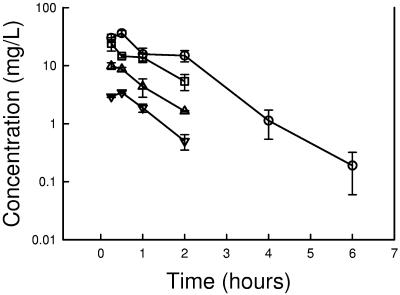
The time course of serum drug levels of PPI-0903 in infected neutropenic mice following subcutaneous doses of 1.56, 6.25, 25, and 100 mg/kg are shown in Fig. 1 and Table 2. Over the dose range studied, kinetics were somewhat nonlinear, with the elimination half-life increasing 1.6-fold with dose escalation. The elimination half-life ranged from 0.54 to 0.88 h. The AUC and peak values for the escalating single doses ranged from 4.1 to 58 mg · h/liter and 3.4 to 36 μg/ml, respectively. PPI-0903 binding in mouse serum was 63 to 66% at drug concentrations of 10 and 100 μg/ml. Both free and total drug levels were considered in pharmacokinetic calculations throughout the study.
FIG. 1.
Serum PPI-0903 concentrations (total drug) after administration of single doses of 1.56, 6.25, 25, and 100 mg/kg in neutropenic infected mice. Each symbol represents the mean ± standard deviation of the levels in the sera of three mice.
TABLE 2.
Total-drug serum pharmacokinetics values following single-dose subcutaneous administration of PPI-0903
| Parameter | 100 mg/kg | 25 mg/kg | 6.25 mg/kg | 1.56 mg/kg |
|---|---|---|---|---|
| Cmax (mg/liter) | 36.3 | 24.1 | 9.89 | 3.44 |
| AUC (mg · h/liter) | 58 | 31.4 | 11.4 | 4.08 |
| t1/2 (h) | 0.73 | 0.88 | 0.65 | 0.54 |
In vivo PAE.
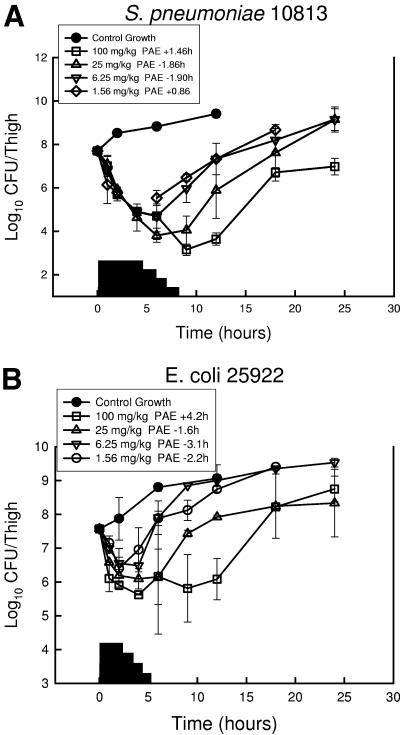
At the start of therapy, neutropenic mice had 107.6 to 107.7 CFU of S. pneumoniae ATCC 10813, S. aureus ATCC 29213, or E. coli ATCC 25922/thigh. The growth of organisms in the thighs of saline-treated mice ranged between 1.71 ± 0.16 and 1.50 ± 0.25 log10 CFU/thigh over 12 h. Growth of 1 log10 CFU/thigh in saline-treated animals occurred in 4.2, 3.4, and 5.1 h in S. pneumoniae-, S. aureus-, and E. coli-infected animals, respectively. Free-drug serum PPI-0903 levels following the single doses of 1.56, 6.25, 25, and 100 mg/kg were estimated to remain above the MIC of S. pneumoniae (0.008 mg/liter) for 4.3, 5.9, 8.0, and 8.2 h, respectively. Following the same drug doses, levels were estimated to remain above the MIC for E. coli (1.0 mg/liter) for 0.6, 1.3, 2.8, and 3.1 h. The estimated times above the MIC for S. aureus (MIC, 0.25 mg/liter) were 0.17, 0.84, 4.2, and 4.6 h. These PPI-0903 doses produced marked reductions in organism burden ranging from 2.2 ± 0.15 to 4.6 ± 0.21 log10 CFU/thigh at the 6- to 9-h time points with S. pneumoniae compared to values at the start of therapy. These doses also produced killing of S. aureus and E. coli ranging from 1.2 ± 0.28 and 2.0 ± 0.61 log10 CFU/thigh in 4 to 9 h. The time-kill curves for each of the three studies are shown in Fig. 2. Against S. pneumoniae, escalating doses produced small to no free-drug PAEs ranging from −1.9 to 1.5 h. A similar study with E. coli produced free-drug PAEs ranging from −0.33 to 5.7 h. Against S. aureus the duration of the PAEs was 0.8 to 7.2 h. No detectable drug carryover was observed in any of the treatment groups.
FIG. 2.
In vivo PAE of PPI-0903 after administration of single doses of 1.56, 6.25, 25, and 100 mg/kg against S. pneumoniae ATCC 10813 (a), S. aureus ATCC 29213 (b), and E. coli ATCC 25922 (c). Each symbol represents the mean ± standard deviation for two mice. Widths of the solid bars represent the duration of time free-drug serum levels exceeded the MIC of the infecting pathogen.
PK-PD index determination.
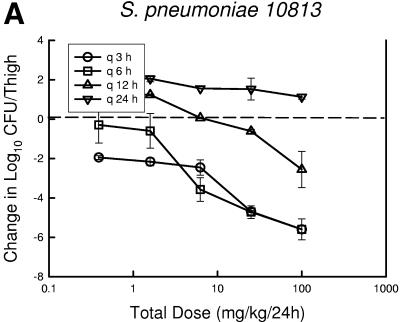
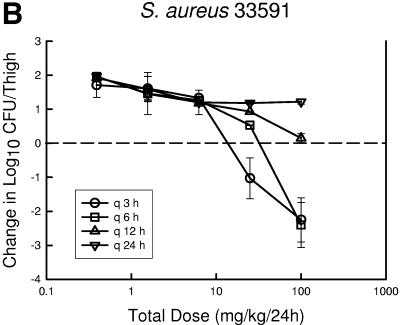
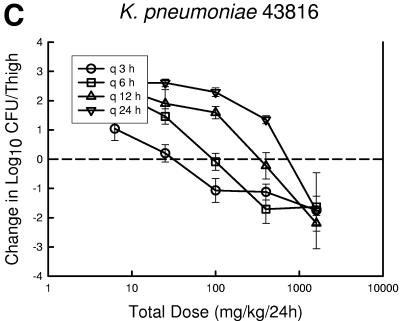
At the start of therapy mice had 7.6 ± 0.05, 6.6 ± 0.19, and 7.2 ± 0.25 log10 CFU of S. pneumoniae ATCC 10813, S. aureus ATCC 33591, and K. pneumoniae ATCC 43816/thigh, respectively. The organisms grew at rates of 2.1 ± 0.21, 2.0 ± 0.31, and 2.5 ± 0.30 log10 CFU/thigh after 24 h in saline-treated control mice, respectively. Escalating doses of PPI-0903 again resulted in marked killing of the strains. The highest doses using the dosing interval of every 3 or 6 h reduced organism burden from 1.80 ± 0.49 to 5.40 ± 0.54 log10 CFU/thigh. Less killing was observed with the more widely spaced dosing intervals (12- and 24-h regimens). The dose-response relationships for the four dosing intervals against S. pneumoniae, S. aureus, and K. pneumoniae are shown in Fig. 3a, b, and c, respectively. The dose-response curves are generally shifted to the right as the dosing interval is lengthened from every 3 h to every 24 h, indicating reduced in vivo potency. In the study with S. aureus, even the highest dose levels did not achieve a net bacteriostatic effect with the intervals of every 12 and 24 h. Similarly, the 24-hour regimen was fairly ineffective in the study with S. pneumoniae. These dose regimen relationships indicate that treatment efficacy was dependent upon the dosing intervals studied and suggest that efficacy was driven by the %T>MIC index.
FIG.3.
Relationship between PPI-0903 dosing interval and efficacy against S. pneumoniae ATCC 10813 (a), S. aureus ATCC 33591 (b), and K. pneumoniae ATCC 43816 (c) in a murine thigh infection model. Each symbol represents the mean data from two mice (four thighs). The dashed horizontal line represents the burden of organisms at the start of therapy. Data below the line represent killing, and data above the line show growth.
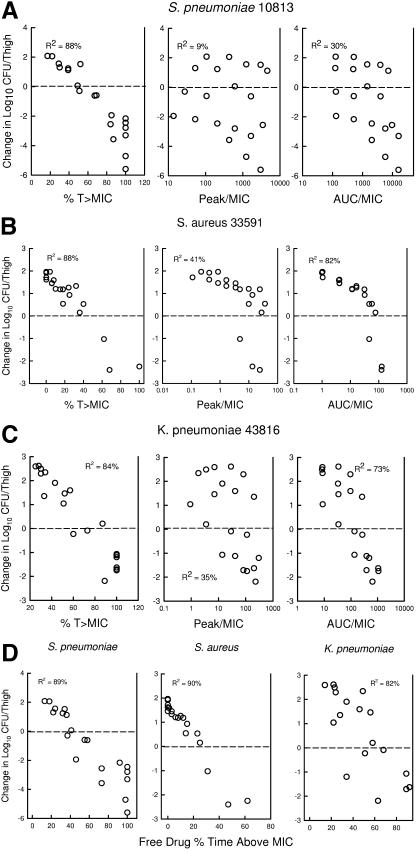
The relationships between microbiologic effect and each of the pharmacodynamic indices (based on total drug), percent time above the MIC, 24-h AUC/MIC, and peak/MIC against S. pneumoniae ATCC 10813 are shown in Fig. 4a. As with other beta-lactam antibiotics, the strongest relationships were observed when results were correlated with the %T>MIC, with an R2 value of 88% (AUC/MIC R2, 30%; peak/MIC R2, 9%). The pharmacodynamic response relationships against S. aureus and K. pneumoniae were similarly best described by the %T>MIC index (S. aureus %T>MIC R2 of 88%, AUC/MIC R2 of 82%, and peak/MIC R2 of 41%; K. pneumoniae %T>MIC R2 of 83%, AUC/MIC R2 of 35%, and peak/MIC R2 of 65%). Consideration of bound or unbound drug levels did not appreciably impact the relationship between efficacy and %T>MIC (not shown).
FIG. 4.
Relationships of the PPI-0903 total drug level, %T>MIC, peak/MIC, and 24-h AUC/MIC for S. pneumoniae ATCC 10813 (a), S. aureus 33591 (b), and K. pneumoniae (c) with the change in log10 CFU/thigh after 24 h of therapy. Each symbol represents the mean data from two mice (four thighs). The dashed horizontal line represents the burden of organisms at the start of therapy. Data above the line represent growth, and data below the line represent killing. R2 is the coefficient of determination.
Magnitude of the PK-PD index associated with efficacy.
Calculations of the doses necessary to achieve a static effect, as well as 1- and 2-log10 reductions against multiple organisms, are shown in Table 3. The growth curves of the five pneumococcal, four staphylococcal, and four gram-negative strains in thighs of control animals were similar. At the start of therapy mice had 7.2 ± 0.1 (range, 7.0 to 7.3) log10 CFU/thigh of pneumococci. Organism burden at therapy start was similar for the staphylococci and gram-negative bacilli, ranging from 6.6 to 7.3 log10 CFU/thigh (mean, 6.9 ± 0.3 log10 CFU/thigh). The organisms grew 2.1 ± 0.5 log10 CFU/thigh (range, 1.2 to 3.4) in saline-treated control mice. The maximal reduction in S. pneumoniae with PPI-0903-treated mice compared to untreated controls ranged from 3.8 ± 0.3 to 4.9 ± 0.2 log10 CFU/thigh (mean, 4.3 ± 0.5). Less killing was observed against the S. aureus and gram-negative bacilli. The organ burden reductions compared to therapy start for S. aureus ranged from 2.6 to 3.4 log10 CFU/thigh (mean, 3.0 ± 0.3). Similar killing was observed against the four gram-negative bacilli (range, 2.0 to 2.9; mean, 2.5 ± 0.3 log10 CFU/thigh).
TABLE 3.
PPP-0903 in vivo activity in murine thigh and lung infection models against multiple organisms
| Organism | MIC (mg/liter) | Static
|
1 log kill
|
2 log kill
|
|||
|---|---|---|---|---|---|---|---|
| Dose (mg/kg/24 h) | %T>MIC | Dose (mg/kg/24 h) | %T>MIC | Dose (mg/kg/24 h) | %T>MIC | ||
| S. pneumoniae 10813 (neutropenic) | 0.008 | 0.32 | 34 | 0.45 | 37 | 0.61 | 43 |
| S. pneumoniae 10813 (normal) | 0.20 | 29 | 0.29 | 33 | 0.41 | 36 | |
| S. pneumoniae 1199 | 0.06 | 1.8 | 35 | 3.42 | 44 | 6.03 | 51 |
| S. pneumoniae 1329 | 0.06 | 1.15 | 30 | 2.04 | 37 | 3.72 | 45 |
| S. pneumoniae 1325 | 0.06 | 6.5 | 52 | 8.46 | 53 | 10.65 | 60 |
| S. pneumoniae 1293 | 0.12 | 14.3 | 50 | 22.2 | 59 | 34.0 | 64 |
| Mean ± SD | 39 ± 9 | 43 ± 9 | 50 ± 10 | ||||
| S. aureus 25923 | 0.12 | 3.75 | 36 | 7.23 | 44 | 14.8 | 51 |
| S. aureus 29213 | 0.25 | 2.57 | 30 | 7.63 | 37 | 26.9 | 56 |
| MRSA | 0.50 | 9.88 | 21 | 22.0 | 34 | 64.1 | 50 |
| S. aureus 33591 | 1.0 | 6.19 | 15 | 9.98 | 18 | 18.0 | 23 |
| Mean ± SD | 26 ± 8 | 33 ± 9 | 45 ± 13 | ||||
| E. coli 25922 | 1.0 | 56 | 32 | 89.2 | 44 | 154 | 50 |
| E. coli 1894-1 | 2.0 | 234 | 37 | 1,040 | 57 | ||
| K. pneumoniae 43816 (thigh) | 2.0 | 22 | 15 | 51 | 24 | 992 | 58 |
| K. pneumoniae 43816 (lung) | 38 | 22 | 117 | 35 | 808 | 53 | |
| K. pneumoniae 216 | 2.0 | 125 | 34 | 500 | 44 | ||
| Mean ± SD | 28 ± 9 | 41 ± 11 | 54 ± 3 | ||||
Table 3 shows the 24-hour dose and free-drug %T>MIC necessary to achieve a net static effect. Against the 13 organisms studied, static doses ranged more than 450-fold (0.48 to 230 mg/kg/24 h). However, the free-drug %T>MIC corresponding with these static doses varied from only 15 to 52% (mean, 32 ± 10). Penicillin and methicillin resistance did not alter the magnitude of the %T>MIC necessary for efficacy.
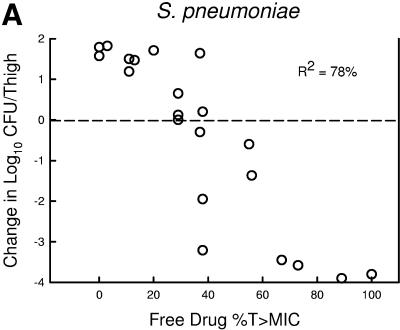
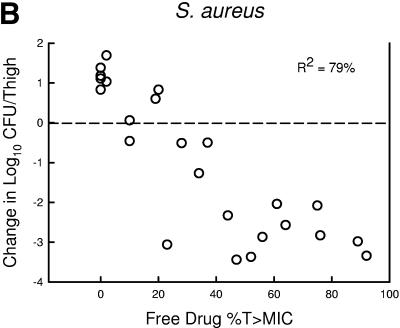
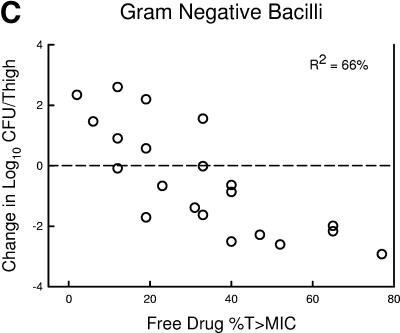
The relationship between the free-drug %T>MIC efficacy against the three organism groups is demonstrated graphically in Fig. 5a, b, and c. The dose-response relationships were relatively strong, with R2 values ranging from 78 to 91%.
FIG.5.
Relationship between the PPI-0903 free-drug %T>MIC and efficacy against five S. pneumoniae (a), four S. aureus (b), and four gram-negative bacilli (c). Each symbol represents the mean data for two mice (four thighs). R2 is the coefficient of determination. The dashed horizontal line represents the burden of organisms at the start of therapy. Data below the line represent killing, and data above the line represent growth.
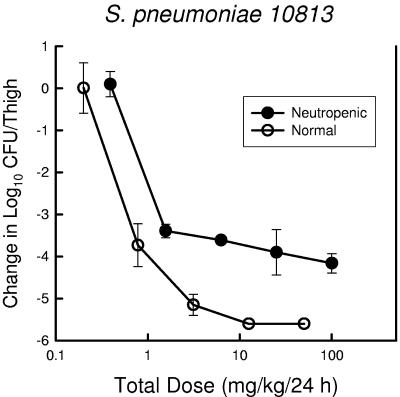
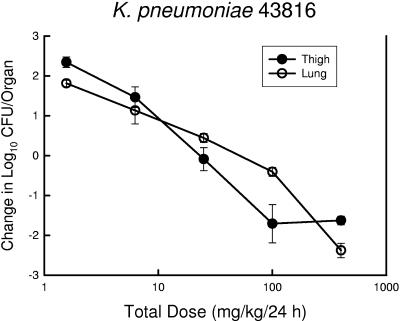
Two substudies were undertaken to determine the impact of infection site and host immunity on the magnitude of the PK-PD index associated with efficacy. Among animals infected in the thigh with S. pneumoniae ATCC 10813, PPI-0903 therapy was examined in both neutropenic and nonneutropenic mice (Fig. 6). The dose-response relationship for the nonneutropenic group was shifted somewhat to the left, suggesting less drug was needed for efficacy. However, the amounts of drug necessary to produce a static effect and 1- and 2-log reductions in organisms were only 1.5- to 1.6-fold higher in neutropenic mice. These differences were not statistically significant. Maximal organism killing over the dose range studied was more than 1 log greater in the nonneutropenic group. In a second study, K. pneumoniae ATCC 43816 was used in both the thigh and pneumonia model to determine if a similar PPI-0903%T>MIC would be necessary to achieve various therapeutic endpoints (Fig. 7). The dose-response curves are remarkably similar for both infection models. Not surprisingly, the %T>MIC associated with a net static effect and 1- and 2-log10 organism reductions were nearly identical (Table 3).
FIG. 6.
Relationship between PPI-093 dose level and change in log10 CFU/thigh in both normal (hollow symbols) and neutropenic (solid symbols) mice infected with S. pneumoniae ATCC 10813. Each symbol represents the mean data for two mice (four thighs).
FIG. 7.
Relationship between PPI-093 dose level and change in log10 CFU/organ in mice infected with K. pneumoniae ATCC 43816 in both the thigh (solid symbols) and lung (hollow symbols) infection models. Each symbol represents the mean data for two mice (four thighs).
DISCUSSION
A variety of in vitro and in vivo studies have demonstrated that the killing activity of beta-lactam antibiotics is not enhanced by exposure to drug concentrations far exceeding the MIC (1, 4, 11, 13, 14, 17, 18). Furthermore, PAE studies have demonstrated minimal persistent effects with drugs from the beta-lactam class with most bacteria. The one exception is with staphylococci, where the duration of the PAE with beta-lactam antibiotics has been longer against S. aureus than either S. pneumoniae or gram-negative bacilli. The efficacy of antibiotics characterized by this pattern of activity is optimized by prolonging the time that serum levels exceed the MIC of the infecting bacteria. Several animal infection models have identified the %T>MIC as the principal PK-PD index predictive of efficacy (1, 3, 5, 17-19).
The current studies characterized the in vivo pharmacodynamic activity of a new anti-MRSA cephalosporin, PPI-0903. The rate and extent of bacterial killing was not remarkably increased despite a 64-fold escalation in dose level, which produced serum drug levels more than 1,000-fold higher than the MIC. However, marked killing was observed when free drug levels exceeded the MIC. The in vivo PAEs were either nonexistent or of modest duration against the S. pneumoniae and E. coli isolates studied. The PAEs were of somewhat longer duration against S. aureus, an observation previously observed with other beta-lactams. One would predict that %T>MIC would be the PK-PD index that most strongly correlated with efficacy of PPI-0903 given this pattern of antimicrobial activity. Data from the current multiple dosing regimen studies confirmed that the %T>MIC is the best PK-PD predictor of efficacy of this cephalosporin.
Numerous in vitro and in vivo models and clinical trials have suggested that the magnitude of the %T>MIC predictive of cephalosporin efficacy ranges from 25 to 70%, depending upon the defined therapeutic endpoint (4, 5, 6). The cephalosporin %T>MIC target to produce a net bacteriostatic effect has been reported most commonly in the range of 30 to 40% as long as free drug levels are considered. A somewhat larger target of 50 to 70% has been required to produce maximal bacteriologic efficacy in animal model studies. In the current investigation, dosing regimens that produced free-drug %T>MICs in a similar range produced a net static effect. The three pneumococcal organisms resistant to penicillin also demonstrated elevated PPI-0903 MICs. However, the relative increase in the MIC did not preclude reaching the therapeutic study endpoints. Similarly, the in vivo activity of this novel cephalosporin was not affected by methicillin resistance in S. aureus. Among the bacterial species studied, the pharmacodynamic target was lowest for S. aureus, which is similar to data from other beta-lactam antibiotics. The reduced %T>MIC is likely related to the longer PAEs against this species compared to others.
Numerous studies have demonstrated that this index value is independent of the dosing interval, animal species, site of infection, or the cephalosporin used as long as free drug levels are considered (1, 2, 5, 11, 17, 18). In these studies as well, the %T>MIC associated with efficacy was similar in the thigh and lung infection models.
While PPI-0903 has not yet undergone extensive clinical investigation, the current studies suggest that the relationship between the pharmacokinetics of this cephalosporin and its efficacy is similar to other cephalosporin antibiotics (5, 8, 14, 17, 18). In vitro susceptibility studies available thus far have found that more than 90% of clinical isolates of S. pneumoniae, S. aureus, and Enterobacteriaceae (including beta-lactam-resistant strains) are inhibited by PPI-0903 concentrations of 0.25, 2.0, and 0.5 mg/liter, respectively (Y. Ge, R. S. Blosser, D. F. Sahm, and J. A. Karlowsky, Abstr. 104th Am. Soc. Microbiol. Gen. Meeting., abstr. A139, 2004). Phase I pharmacokinetic studies should soon be available. The relationship between human pharmacokinetics and the surveillance in vitro susceptibility data should be considered relative to a %T>MIC target ranging from 20 to 50% (based on static dose). These pharmacodynamic targets should be useful for dosing regimen design for future clinical trials. The current studies suggest that this new cephalosporin should be effective in vivo against a wide spectrum of organisms, including MRSA.
REFERENCES
- 1.Ambrose, P. G., R. Quintiliani, and N. Chin. 1997. Continuous vs intermittent infusion of cefuroxime for the treatment of community-acquired pneumonia. Infect. Dis. Clin. Pract. 7:463-470. [Google Scholar]
- 2.Andes, D., and W. A. Craig. 2002. A critical review of animal model pharmacokinetics and pharmacodynamics. Int. J. Antimicrob. Agents 19:261-268. [DOI] [PubMed] [Google Scholar]
- 3.Andes, D., and W. A. Craig. 1998. In vivo activities of amoxicillin and amoxicillin-clavulanate against Streptococcus pneumoniae: application to breakpoint determinations. Antimicrob. Agents Chemother. 42:2375-2379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Craig, W. A. 1998. Pharmacokinetics and pharmacodynamics of antibiotics in mice and men. Clin. Infect. Dis. 26:1-12. [DOI] [PubMed] [Google Scholar]
- 5.Craig, W. A. 1995. Interrelationship between pharmacokinetics and pharmacodynamics in determining dosage regimens for broad-spectrum cephalosporins. Diagn. Microbiol. Infect. Dis. 22:89-96. [DOI] [PubMed] [Google Scholar]
- 6.Craig, W. A. 2003. Basic pharmacodynamics of antibacterials with clinical applications to the use of beta-lactams, glycopeptides, and linezolid. Infect. Dis. Clin. North Am. 17:479-501. [DOI] [PubMed] [Google Scholar]
- 7.Craig, W. A., and S. Gudmundsson. 1996. Postantibiotic effect, p. 296-329. In V. Lorian (ed.), Antibiotics in laboratory medicine, 4th ed. Williams & Wilkins Co., Baltimore, Md.
- 8.Craig, W. A., and D. R. Andes. 1996. Pharmacokinetics and pharmacodynamics of antibiotics in otitis media. Pediatr. Infect. Dis. J. 15:255-259. [DOI] [PubMed] [Google Scholar]
- 9.Craig, W. A., and P. G. Welling. 1977. Protein binding of antimicrobials: clinical pharmacokinetic and therapeutic implications. Clin. Pharmacokinet. 2:252-268. [DOI] [PubMed] [Google Scholar]
- 10.Ishikawa, T., N. Matsunaga, H. Tawada, N. Kuroda, Y. Nakayama, Y. Ishibashi, M. Tomimoto, Y. Ikeda, Y. Tagawa, Y. Iizawa, K. Okonogi, S. Hashiguchi, and A. Miyake. 2003. TAK-599, a novel N-phosphono type prodrug of anti-MRSA cephalosporin T-91825: synthesis, physiocochemical and pharmacological properties. Bioorg. Med. Chem. 11:2427-2437. [DOI] [PubMed] [Google Scholar]
- 11.Leggett, J. E., B. Fantin, S. Ebert, K. Totsuka, B. Vogelman, W. Calamae, H. Mattie, and W. A. Craig. 1989. Comparative antibiotic dose-effect relationships at several dosing intervals in murine pneumonitis and thigh-infection models. J. Infect. Dis. 159:281-292. [DOI] [PubMed] [Google Scholar]
- 12.Kunin, C. M., W. A. Craig, M. Kornguth, and R. Monson. 1973. Influence of binding on the pharmacologic activity of antibiotics. Ann. N. Y. Acad. Sci. 226:214-224. [DOI] [PubMed] [Google Scholar]
- 13.Mouton, J. W., A. Schmitt-Hoffmann, S. Shapiro, N. Nashed, and N. C. Punt. 2004. Use of Monte Carlo simulations to select therapeutic doses and provisional breakpoints of BAL9141. Antimicrob. Agents Chemother. 48:1713-1718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mouton, J. W., and J. G. den Hollander. 1994. Killing of Pseudomonas aeruginosa during continuous and intermittent infusion of ceftazidime in an in vitro pharmacokinetic model. Antimicrob. Agents Chemother. 38:931-936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.National Committee for Clinical Laboratory Standards. 2000. Methods for dilution susceptibility tests for bacteria that grow aerobically. Approved standard, 5th ed. Document M7-A5. National Committee for Clinical Laboratory Standards, Wayne, Pa.
- 16.National Research Council, Committee on the Care and Use of Laboratory Animals, Institute of Laboratory Animal Resources, and Commission on Life Sciences. 1996. Guide for the care and use of laboratory animals. National Academy Press, Washington, D.C.
- 17.Nicolau, D. P., C. O. Onyeji, M. Zhong, P. R. Tessier, M. A. Banevicius, and C. H. Nightingale. 2000. Pharmacodynamic assessment of cefprozil against Streptococcus pneumoniae: implications for breakpoint determinations. Antimicrob. Agents Chemother. 44:1291-1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Roosendaal, R., I. A. J. M. Bakker-Woudenberg, J. C. van den Berghe, and M. F. Michel. 1985. Therapeutic efficacy of continuous versus intermittent administration of ceftazidime in an experimental Klebsiella pneumoniae pneumonia in rats. J. Infect. Dis. 156:373-378. [DOI] [PubMed] [Google Scholar]
- 19.Vogelman, B., S. Gudmundsson, J. Leggett, J. Turnidge, S. Ebert, and W. A. Craig. 1988. Correlation of antimicrobial pharmacokinetic parameters with therapeutic efficacy in an animal model. J. Infect. Dis. 158:831-847. [DOI] [PubMed] [Google Scholar]