Abstract
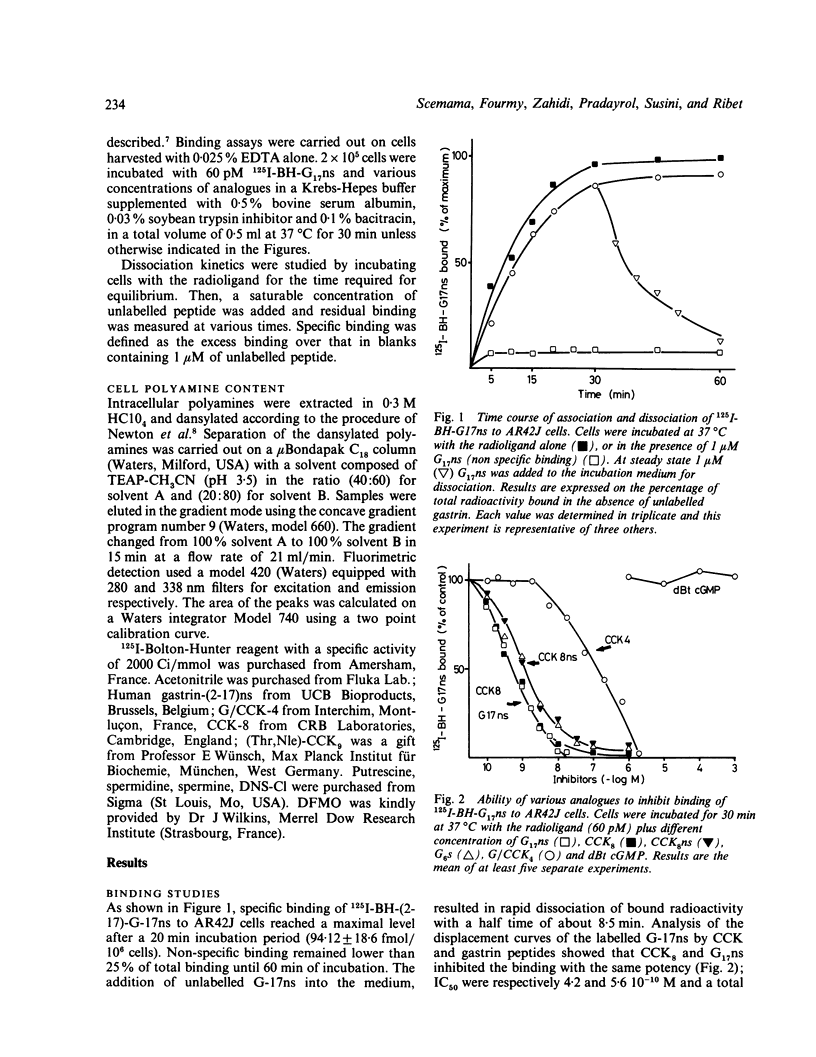
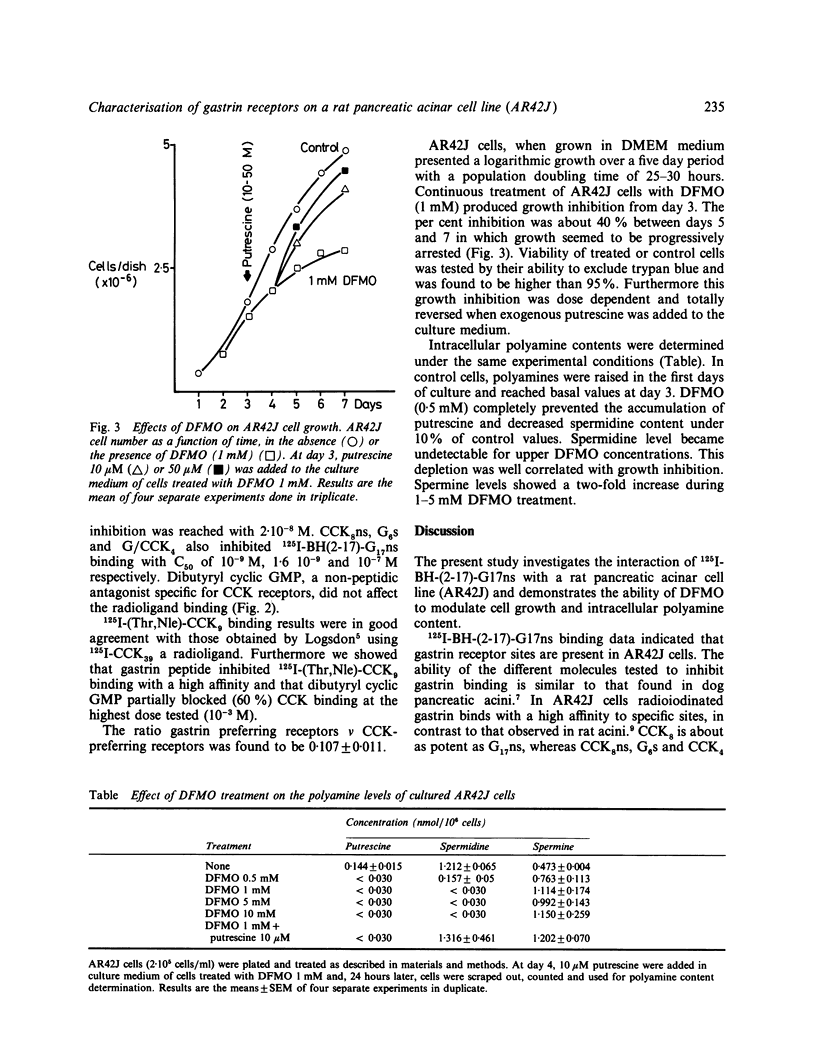
Trophic changes of the exocrine pancreas after in vivo gastrin (G)/CCK treatment are well documented but up to now the study of the mechanisms involved is restricted by the lack of a suitable in vitro model. Nevertheless the in vivo trophic effect induced by gastrin/CCK peptides has been associated with an increase of ornithine decarboxylase (ODC) activity. In the present work, using the AR42J cell line in which CCK receptors and stimulation of amylase release by CCK peptides has already been demonstrated, we investigated the presence of gastrin binding sites and the possible modulation of proliferation by an inhibitor of ODC activity. 125I-BH-G17ns binding is saturable, reversible and specific. Potencies of the different analogues tested are G17ns greater than CCK8 greater than CCK8ns greater than or equal to G6s greater than G/CCK4. Furthermore dBt cGMP, a non-peptide antagonist for CCK receptors, does not compete for gastrin binding. This indicates the existence of a subclass of gastrin binding sites. Difluoromethyl ornithine (DFMO) (1 mM), an irreversible inhibitor of ODC, inhibits cell growth from day 3 up to day 7. This growth inhibition is dose dependent and closely related to an intracellular polyamine modulation. Putrescine and spermidine levels fell under detectable values while spermine levels increased. All these data suggest that this cell line could be a useful in vitro model to study the mechanisms of gastrin induced growth control.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Dembinski A. B., Johnson L. R. Stimulation of pancreatic growth by secretin, caerulein, and pentagastrin. Endocrinology. 1980 Jan;106(1):323–328. doi: 10.1210/endo-106-1-323. [DOI] [PubMed] [Google Scholar]
- Fourmy D., Zahidi A., Pradayrol L., Vayssette J., Ribet A. Relationship of CCK/gastrin receptor binding to amylase release in dog pancreatic acini. Regul Pept. 1984 Dec;10(1):57–68. doi: 10.1016/0167-0115(84)90053-3. [DOI] [PubMed] [Google Scholar]
- Logsdon C. D. Glucocorticoids increase cholecystokinin receptors and amylase secretion in pancreatic acinar AR42J cells. J Biol Chem. 1986 Feb 15;261(5):2096–2101. [PubMed] [Google Scholar]
- Morisset J., Benrezzak O. Reversal of alpha-difluoromethylornithine inhibition of caerulein-induced pancreatic growth by putrescine. Regul Pept. 1985 Jul;11(3):201–208. doi: 10.1016/0167-0115(85)90051-5. [DOI] [PubMed] [Google Scholar]
- Newton N. E., Ohno K., Abdel-monem M. M. Determination of diamines and polyamines in tissues by high-pressure liquid chromatography. J Chromatogr. 1976 Sep 15;124(2):277–285. doi: 10.1016/s0021-9673(00)89743-5. [DOI] [PubMed] [Google Scholar]
- Sankaran H., Goldfine I. D., Deveney C. W., Wong K. Y., Williams J. A. Binding of cholecystokinin to high affinity receptors on isolated rat pancreatic acini. J Biol Chem. 1980 Mar 10;255(5):1849–1853. [PubMed] [Google Scholar]
- Solomon T. E., Vanier M., Morisset J. Cell site and time course of DNA synthesis in pancreas after caerulein and secretin. Am J Physiol. 1983 Jul;245(1):G99–105. doi: 10.1152/ajpgi.1983.245.1.G99. [DOI] [PubMed] [Google Scholar]


