Abstract
The endothelium is a thin monocelular layer that covers all the inner surface of the blood vessels, separating the circulating blood from the tissues. It is not an inactive organ, quite the opposite. It works as a receptor-efector organ and responds to each physical or chemical stimulus with the release of the correct substance with which it may maintain vasomotor balance and vascular-tissue homeostasis. It has the property of producing, independently, both agonistic and antagonistic substances that help to keep homeostasis and its function is not only autocrine, but also paracrine and endocrine. In this way it modulates the vascular smooth muscle cells producing relaxation or contraction, and therefore vasodilatation or vasoconstriction. The endothelium regulating homeostasis by controlling the production of prothrombotic and antithrombotic components, and fibrynolitics and antifibrynolitics. Also intervenes in cell proliferation and migration, in leukocyte adhesion and activation and in immunological and inflammatory processes. Cardiovascular risk factors cause oxidative stress that alters the endothelial cells capacity and leads to the so called endothelial "dysfunction" reducing its capacity to maintain homeostasis and leads to the development of pathological inflammatory processes and vascular disease.
There are different techniques to evaluate the endothelium functional capacity, that depend on the amount of NO produced and the vasodilatation effect. The percentage of vasodilatation with respect to the basal value represents the endothelial functional capacity. Taking into account that shear stress is one of the most important stimulants for the synthesis and release of NO, the non-invasive technique most often used is the transient flow-modulate "endothelium-dependent" post-ischemic vasodilatation, performed on conductance arteries such as the brachial, radial or femoral arteries. This vasodilatation is compared with the vasodilatation produced by drugs that are NO donors, such as nitroglycerine, called "endothelium independent". The vasodilatation is quantified by measuring the arterial diameter with high resolution ultrasonography. Laser-Doppler techniques are now starting to be used that also consider tissue perfusion.
There is so much proof about endothelial dysfunction that it is reasonable to believe that there is diagnostic and prognostic value in its evaluation for the late outcome. There is no doubt that endothelial dysfunction contributes to the initiation and progression of atherosclerotic disease and could be considered an independent vascular risk factor. Although prolonged randomized clinical trials are needed for unequivocal evidence, the data already obtained allows the methods of evaluation of endothelial dysfunction to be considered useful in clinical practice and have overcome the experimental step, being non-invasive increases its value making it use full for follow-up of the progression of the disease and the effects of different treatments.
Ever since the endothelium was discovered by microscopical examination, it has always been considered to be a lining that acted as a barrier stopping intravascular coagulation. Nevertheless, in the last decades, the recognition of its multiple functions has shown it to be a true regulator of blood flow and tissue homeostasis. Although it is a monolayer that covers the inner surface of the entire vascular system, its total weight is more than a liver and has a mass equal to several hearts or, if it is extended, covers a various tennis courts surface area. For these reason, it has been postulated as the biggest and most important gland of the body [1].
Anatomic and functional properties of the endothelial cell
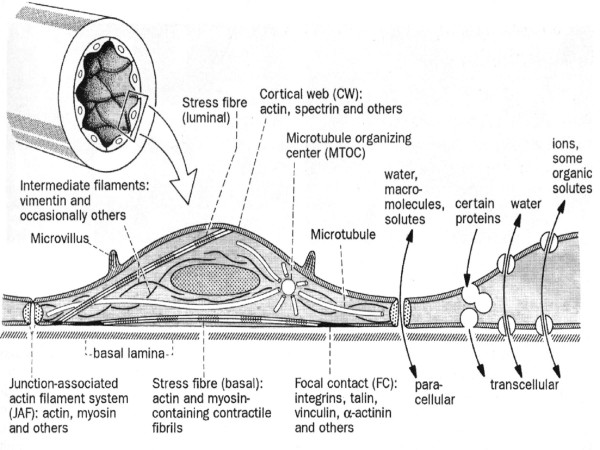
Basically, the endothelial cell has the same characteristics as all the cells of the human body; cytoplasm and organelles surrounding a nucleus and contained by the cellular membrane. The cell membrane is made of a double layer of phospholipids separated by water compartments and crossed by complex proteins that work as receptor or ion channels. Various contractile proteins cross the cytoplasm: actin, myosin, tropomyosin, α-actin and others, that allow motor activities.[2] Some are organized as structures like the cortical web, the junction-associated actin filament system related to the intercellular unions and the striated myofibril-like filament bundles or stress fibers (Figure 1).
Figure 1.
General organization of the cytoskeleton in vascular endothelial cells. From Drenckham D, Ness W.[2]
The cortical web surrounds the internal surface of the sarcolema and is responsible for the cells shape and elasticity. It is sensitive to changes of the intravascular tension and increases its stiffness with increases of the intravascular pressure. It also anchor's different membrane proteins, among them annexine, which regulates endo- and exocytosis, the E-selectins and cadherine, related to the adhesiveness of leukocyte and platelets. The adherence of these elements and their passage through the endothelial cell depends on the integrity of the cortical membrane.
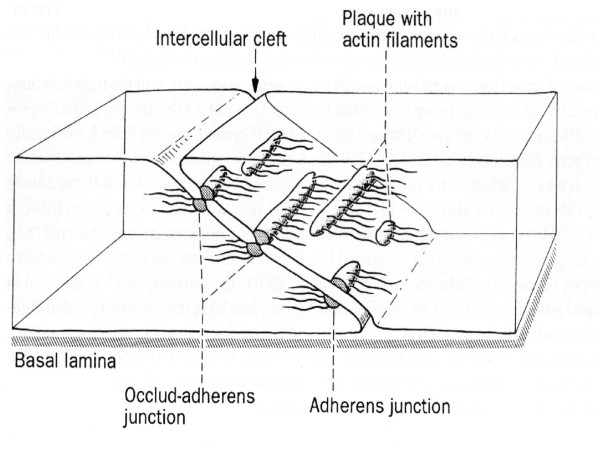
The junction-associated actin filament system, known as FAU system, is found in the intercellular space and its contraction and relaxation controls the dimension of the intercellular space. In this way, it regulates the passage of solutes and macromolecules between the blood and the sub endothelial space. The Ca2+ concentrations, intracellular second messenger and the common factor of the external function of cells with intermittent or cyclic activities activate it, the energy is provided by adenosine tri-phosphate (ATP). Pro-inflammatory cytokines, reactive oxygen species, thrombin, platelet activating factor, an increase of Ca2+ concentration in ischemic conditions, ATP exhaustion and other toxic substances, alter the functions of the junction-associated actin filament system and allow an opening of the intercellular space and, with that, alteration of the endothelial permeability. The FAU system is closely related to the intercellular adhesion molecules, especially with VE-cadherine maintaining a balance between adhesive and contractile forces. Both cyclic adenosine mono-phosphate (cAMP), originated through the adenylate-cyclase, and the cyclic guanine mono-phosphate (cGMP), generated by a Ca2+-nitric oxide guanylate-cyclase dependent pathway, are second messengers that stabilize the FAU system and counteract the induction of intercellular separation, which is done through a Ca2+-dependent calmodulin. Nitrates, behave the same way. Protein-kinase C (PKC) activation has the opposite effect (Figure 2.)
Figure 2.
Intercellular cleft and occludens adherens junction in vascular endothelial cells. From Drenckham D, Ness W.[2]
Stress fibers are myofibril-like straight filament bundles composed of actin filaments interspersed with myosin filaments in a similar way as in striated muscle, and cross the cytoplasm in all directions (Figure 1). The more pressure and friction exerted by the circulating blood, the more abundant they are. As all contracting tissues, their contraction and relaxation depend on the intracellular Ca2+ concentration and the presence of ATP, their principal function is to adapt the shape of the cells to the mechanical forces of blood flow and wall distention, reducing the possibility of cellular lesions. When flow increases, so does shear stress and the cells flatten and align in the direction of blood flow, whereas, when flow decreases shear stress does also, and the cells increase their volume loosing their alignment, looking like cobble stones paving.
Morphological changes acquire importance in capillary flow because they can slow or halt flow, as can be seen under the effects of serotonine, histamine, noradrenaline and trombine, although FAU also acts on this function. In the capillary the blood cells are usually larger than the diameter, but flow through the capillary by two main mechanisms: a) by the flexibility and deforming capacity of both type of cells, blood and endothelial cells, and b) by the negative electrostatic charge both cells have and, therefore, repel each other. Endothelial cells have a negative electrostatic charge because of the high concentration of sialitic acid. If this concentration is diminished for any reason, blood flow is disturbed.
The cell membrane is covered with flask-shaped membrane invaginations, sometimes shaped like a pocket and sometimes protruding out of the membrane, other times flattened, undistinguished from the basic structure of the cell membrane, but all of them very rich in lipids, sphingomyelin, complex protein structures and multiple receptors. These sites have been called "caveolae". They are so abundant that it is estimated that they occupy between 5% and 10% of the total cell surface, and are presumed to be cellular membrane receptor-efector areas.[3] In normal circumstances there are various ways of transporting plasmatic molecules through the endothelial barrier: a) intercellular unions, that generally act as filters controlled by the hydrostatic pressure that allow the passage of water and dissolved substances; b) vesicles formed from the "caveolae" that ease the passage of macromolecules through the cell membrane and cytoplasm; and c) true transcellular channels usually formed from various caveolae that connect opposite sides of the cell membrane. Through them, the endothelial regulates the passage of fluid and macromolecules between the vascular and cellular compartments, when it fails in the venous capillary area edema is produced, toxic and vasoactive substances can cause this.
Endothelial physiology
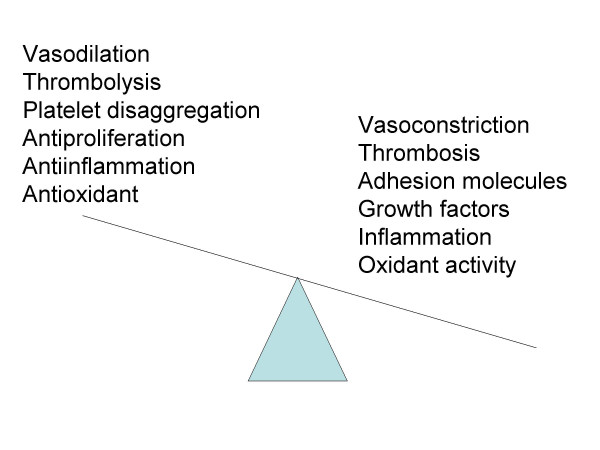
The endothelial cell behaves as a receptor-efector structure, that senses different physical or chemical stimulus that occur inside the vessel, and therefor modify the vessel shape or releases the necessary products to counter act the effect of the stimulus and maintain homeostasis. The endothelium is capable of producing a large variety of different molecules, as agonists as antagonists, therefore balancing the effects in both directions. Endothelium produces vasodilators and vasoconstrictors, procoagulants and anticoagulants, inflammatory and anti-inflammatory, fibrinolytics and antifibrinolytics, oxidizing and antioxidizing, and many others (Figure 3) [1,6]. When endothelial cell lose their ability to maintain this delicate balance, the conditions are right for the endothelium to be invaded by lipids and leukocytes (monocites and T-lymphocytes). The inflammatory response is incited and fatty streaks appear, the first step in the formation of the atheromatous plaque. If the situations persist, fatty streaks progress and the plaque are exposed to rupture and set the condition for thrombogenesis and the vascular occlusion.
Figure 3.
Regulatory functions of the endothelium. Normal or anti-atherogenic vs dysfunction or atherogenic propierties. From Esper RJ, et al.[5]
Nitric oxide
Nearly all stimuli that produce vasodilatation do it through nitric oxide (NO), a volatile gas, biologically active, present practically in all tissue and thanks to its low molecular weight and its lipophilic properties it diffuses easily across cell membranes. The NO crosses the endothelial intima and reaches the smooth muscular tissue of the arterial wall, and, through nitrosilation of the hem from the guanylate-cyclase degrades the GTP releasing cGMP, which in turn regulates the cytosolic Ca2+ and causes smooth muscle fiber relaxation and therefore vasodilatation [7].
NO is produced by the action of nitric oxide synthases (NOS) on L-arginine aminoacid, producing NO and L-citruline, requiring O2 and Nicotinamide Adenine Dinucleotid Phosphate (NADP) coenzyme, essential in Redox process. Tetra-hydro-biopterine accelerates this process, which is favored by other cofactors like flavin-adenine dinucleotide, and thiol groups like cysteine and reduced-glutation. Three NOS isoenzimes are known, two constitutives and low-production, NOS-I from neurological tissue and NOS-III from endothelial cells, both respond to agonist that increase intracellular Ca2+. The other one, inducible NOS-II, is specially expressed in macrophage and endothelial cells due to the effect of pro-inflammatory cytokines and can release several times more NO than the constitutives NOS. Both constitutives and inducible NOS are in the endothelial cells. Constitutives NOS produces NO for short periods when it is induced by vasodilators like acetylcholine or bradikinine. Inducible NOS synthesize NO for longer periods in a constant manner when the stimulus comes from pro-inflammatory cytokines like tumor necrosis factor-α (TNF-α) [7,8].
The most important stimulation for NO release comes from shear stress, that is caused by the increase in blood velocity and leads to a vasodilatation proportional to the amount of NO released by the endothelium [9]. This vasodilatation is called endothelium-dependent. The endothelial cell membranes contains specialized ion channels, such as Ca2+-activated K+ channels, that open in response to shear stress [10]. The effect is to hyperpolarize the endothelial cell, increasing the driving force for Ca2+ entry and activates the enzyme NOS-III and the subsequent generation of NO [10]. Nitrates given in any way are NO donors, unconfined NO into the circulation directly releasing cGMP in the smooth muscle cell and causing a vasodilatation that is not dependent of the endothelial response, for this reason it is called endothelium-independent vasodilatation. Shear stress induces a persistent production of NO that maintains a constant vasodilatation [11].
Shear stress explains the importance of the haemodynamic factor in the formation, localization and plaque fissure. This usually develops in areas were the shear stress is low (<6 din/cm2), oscillating or retrograde, were NO release is diminished and adhesion molecules are increased and chemical and growth factors create a pro-inflammatory atmosphere. On the other hand, a high shear stress (>70 din/cm2) con cause endothelial erosion and provoke platelet aggregation, or cause plaque rupture or damage. That is to say that a low or retrograde shear stress allows plaque formation and progression of the atherosclerotic lesion, and a high a shear stress causes damage to the plaque. Physiological levels of shear stress that protect the endothelium are between these two values (6 to 70 din/cm2) [12-15].
NO apart of being a vasodilator, also reduces vascular permeability and the monocite and lymphocyte adhesion molecules synthesis. NO also reduces platelet aggregation, tissue oxidation, tissue inflammation, activation of thrombogenic factors, cell growth, proliferation and migration, also it inhibits proatherogenic and pro-inflammatory cytokines expression and it favors fibrinolysis. Nuclear factor kappa-B (NFkB) inhibitor (I-kB) is also expressed by NO. All these factors reduce atherogenesis and its complication. For this reason NO is considered the antiatherogenic molecule [16-22].
The endothelial cell releases angiotensin-II (AII) as an antagonist of NO by means of hydrolyzing angiotensin-I by angiotensine converting enzyme (ACE). Through AT1 receptor, AII causes vasoconstriction and prothrombogenic, oxidizing and antifybrinolitic effects, and also favors adhesion molecule expression and leukocyte adhesion. Also it stimulates growth and proliferation factors, activates inflammation and incite expression of pro-inflammatory and proatherogenic cytokines. All these effects allow atherosclerosis to start develop, progress and complicate. Also AII stimulates endothelin converting enzyme that degrades big-endothelin releasing endothelin-I, the most powerful vasoconstrictor on vessel walls [23-25].
As can be seen, depending on the balance of these two substances, NO and AII, a vasodilatation and antiatherosclerotic or vasoconstriction and atherogenic effect will prevail. It is not necessary by increased production of one or the other, the diminished synthesis of one, will make normal amounts of the other prevail (Figure 3). The endothelium should maintain an adequate homeostasis so that the disease does not appear and this depends on the capacity it has of producing the protective molecules. When this function is lost or damaged, we refer to it as "endothelial dysfunction" or "endothelial activation". The classical cardiovascular risk factors (hypercholesterolemia, hypertension, smoking, diabetes, sedentary, etc.) and the so called new risk factors like hiperhomocysteinemia, lipoprotein Lp(a), infections by Chlamydia pneumoniae, Helicobacter Pilorae, Cytomegalovirus, herpes zoster virus or bacteroides gingivalis, all have a common factor which is a state of oxidative stress that directly or through heating proteins (HSP-60), stimulate NF-kB replication that leads to the production of proatherogenic cytokines like TNF-α, interleukins IL-1 and IL-6, adhesion molecules and chemokines, that cause inhibition of ONS-III activity and with that NO production, and favoring AII synthesis and activity [26-30]. The pro-inflammatory cytokines stimulate the replication of NF-kB that leads to more cytokines production. Then inflammation amplify inflammatory response [31].
It is useful to consider the other great source of vasodilatation, via arachidonic acid cascade that ends in prostaciclines, which release cAMP from ATP that regulates cytosolic Ca2+, therefore producing relaxation and vasodilatation. There are other vasodilatation mechanisms, like endothelium derived hyperpolarizing factor that increases the intracellular concentration of K through CP450 cytochrome, and natriuretic peptide-C intervention. It has recently been postulated that some ACE inhibitors stimulate endothelium derived hyperpolarizing factor which would explain some beneficial effects these compounds have that have not been satisfactorily explained by ACE inhibition only. All these alternative mechanisms are important in certain situations to substitute NO deficit.
Endothelium and renin-angiotensin system
The Renin-Angiotensin System (RAS) is a cascade of enzymatic reactions that ends in AII. The renin, produced by the kidney, acts on the angiotensinogen, produced by the liver degrading it to AI, which in turn will be acted on by circulating or tissue ACE and hydrolyzed to AII. Chymase, Carboxypeptidase, Cathepsin G and Tonin can generate AII from AI independently of ACE, and AII can also originate directly from angiotensinogen by non-renin enzymes, such as tissue plasminogen activator (t-AP), Cathepsin G and Tonin. Because of structural similarity ACE also degrades other peptides that are substrates to this enzyme such as P substance, enkephalins, neurotensin, takynine and kininogen, the latter responsible for bradykinin generation. Stimulates ONS-III, inducing ON synthesis and prostacicline production, which in turn have opposite the effects of AII. As can be seen, the interrelationship between agonists and antagonists is very complex and requires a delicate balance which is the endotheliums primary function [26-31]. There are two RAS, one is circulatory and the other is in the tissues, the latter at a cellular level develops approximately 90% of the systems activity.
AII acts by stimulating specific receptors, in the human body we are able to distinguish two different types of receptors: AT1 and AT2. the AT1 receptors are responsible for known effects of AII; vasoconstriction, increase of aldosterone activity, myocardial hypertrophy, vessel wall smooth muscle proliferation, renal sodium reabsortion, increase in peripheral noradrenergic activity, vasopresin release, sympathetic stimulation, decrease in renal blood flow, etc. The effects of AT2 receptor stimulation is not completely known, but there are animal and human data that allow us to assume that they era responsible for apoptosis, a clear inhibition of proliferation, vascular endothelial neogenesis stimulation and vasodilatation, all opposite effects of AT1 [32]. AT2 receptors are found during the last three months of pregnancy in the fetus, and during the first three weeks of life. In adult tissues, they are predominantly expressed in the brain and adrenals, with lower levels expressed elsewhere [33]. They are expressed when there is vascular injury.
There are different angiotensine peptides capable of stimulating the AT1 receptors with varying intensity: AII(2–8), AIV(3–8), AII(1–7). This fact and the different effects of each one when the specific receptors were blocked suggest that there are other specific receptors sensitive to each of these peptides. In experimental animal models different specific receptor variant have been isolated and cloned, for example in the rat AT1A, AT1B, and AT1C. Nevertheless, in human beings, only AT1 and AT2 receptors of ATII have been identified. AII(1–7) has opposite effects of the classic AII, producing vasodilatation, inhibiting proliferation, myocardial hypertrophy and vascular smooth muscle proliferation, and it is very unlikely that it should act through the known AT1 and AT2 receptors. This led to believe that there are specific receptors for it. This has actually been reported recently although they have not yet been cloned [32-34].
Oxidant byproducts and atherosclerosis
Oxidant products, such as superoxide anion (O2-), hydrogen peroxide (H2O2), hydroxyl radical (HO), hypochlorous acid (HOCl) and lipid radicals [33], are produced as a consequence of normal aerobic metabolism. These molecules are highly reactive with other biological molecules and are referred as Reactive Oxygen Species (ROS). Under normal physiological conditions, ROS production is balanced by an efficient system of antioxidants, molecules that are capable to neutralize them and thereby preventing oxidant damage. In the tissues, naturally occurring enzymatic antioxidants such as superoxide dismutase, glutathione peroxidase, and catalase play an important role in the conversion of ROS to oxygen and water. Several nonenzymatic antioxidants, including the lipid-soluble vitamin E and β-carotene and the water-soluble antioxidants vitamin C, which particular protects plasma lipids from peroxidation, scavenges superoxide anion, and plays a role in recycling vitamin E [35]. In pathological states, the ROS may be present in relative excess. This shift of the balance in favor of oxidation termed "oxidative stress", may have detrimental effects on cellular and tissue function. As mentioned before, cardiovascular risk factors generate oxidative stress (Table I).
Table 1.
Normal aerobic metabolism oxidizing and antioxidizing systems
| OXIDIZING SYSTEMS | ANTIOXIDIZING SYSTEMS |
| Oxidant byproducts | Enzymatic antioxidants |
| Superoxide Anion (O2-) | Superoxide dismutase |
| Hydrogen Peroxide (H2 O2) | Glutathione peroxidase |
| Hydroxyl radical (HO) | Catalase |
| Hypochlorous acid (HOCl) | |
| Lipid radicals | |
| Enzimatic oxidants | Lipid-soluble antioxidants |
| Dehidrogenase Xantine | Vitamin E |
| NADPH oxidase | β-Carotene |
| Mieloperoxidase | |
| Monoamino oxidase | Water-soluble antioxidants |
| ON synthetase | Vitamin C |
LDL-cholesterol molecules are easily oxidized in a state of oxidative stress, especially the small and dense molecules. Native LDL molecules are innocuous, they do not produce any inflammatory reaction and do not lead to foam cells when phagocyte by specific native macrophage receptors. Oxidized LDL-cholesterol molecules (LDL-ox), are highly immunogenic, and is associated with up-regulation of pattern-recognition receptors for innate immunity, including scavengers receptors and toll-like receptors [36,37]. They are found in all atherosclerotic lesions and they generate antibodies that are capable of neutralizing them. LDL-ox attack the arterial intima and leads to release of phospholipids that can activate endothelial cells [38], induces the production of endothelium adhesion molecules and monocytes attraction [39], has endothelium cytotoxic effect, increases proinflamatory gene activity and cellular growth factors, it provokes endothelial dysfunction, platelet aggregation, metaloproteinase expression and favors thrombogenesis [40]. LDL-ox molecules are found in the subendothelial layers and help to activate monocytes that are transformed into macrophage, upregulating their scavenger receptors and toll-like receptors that then phagocyte them. With progressive accumulation of LDL-ox macrophage modulate their phenotype turning them into foam cells. Foam cells are the principal component of the fatty streaks, first step in atheromatous plaque formation, and they trigger antigenic reaction in T-lymphocytes that initiate or increase the immunological response [41]. Also TNF-α is activated and endothelial cell apoptosis is induced and has a close relation with the severity of acute ischaemic syndromes [42,43].
HDL cholesterol and apolipoprotein A-1 have direct antiatherogenic and vascular protective effects. They have antioxidant effects attributed to the binding of transition metals and to the presence of paraoxonase, an enzyme carried predominantely by apolipoproteins A-1 and J, containing HDL particles, which has powerful antioxidant effects. Morover, they have been shown anti-inflammatory effects, scavenging of toxic phospholipids, stimulation of reverse cholesterol transport, antithrombotic and profibrinolytic effects, and attenuation of endothelial dysfunction [44].
The excess of ROS, especially superoxide anion, can oxidize NO and transform it into peroxynitrite (ONOO), a inactive molecule that can lead to more oxidation. This situation is usually seen when ONS-II activation is induced by the high concentration of NO it generates. This also happens with high levels of LDL, especially small and dense molecules that are prone to oxidation. Asymmetric dimethyl-amino-arginine (ADMA) exists normally in the body and it inhibits NO synthesis by competing with L-arginine. In this way, reduce NO tissue concentration with all the consequences that this causes, to the extent that many investigators consider it a new atherosclerotic risk factor. Serum levels of ADMA keep a close relationship with LDL-ox concentration and viceversa [45,46]. Peroxinitrite (ONOO) can oxidize tetra-hidro-biopterin, a critical cofactor for ONS [47].
Long-term with most organic nitrates is frequently associated with a progressive reduction of hemodynamic effects. Nitrates activates vascular NADPH oxidase with incremental O2- generations, and this highly reactive molecules oxidize NO to ONOO [48]. Moreover, continuos treatment with nitrates causes ONS-III dysfunction by oxidative stress. Reduced bioavailability of tetra-hydro-biopterin is involved in the pathogenesis of this phenomenon and is prevented by supplemental folic acid administration. ROS transform regular ONS-III function, and produces O2- in place of NO [49]. Oxidative stress may explain the development of tolerance and the impaired endothelial function during continuous organic nitrates administration.
Inflammation and thrombosis
There is a clear relationship between inflammation and thrombosis, each influencing the other. Inflammatory cytokines induce procoagulant molecules in endothelial cells such as von Willebrand factor, tissue factor and plasminogen activating inhibitor factors, PAI-1 and PAI-2. Activated inflammatory cells also produce molecules that contribute to the thrombogenesis such as tissue factor and Thrombin, which in turn generates an intense mitogenic stimulus and platelet activation [50-52]. Interleukin-6 not only increase plasma concentration of C-Reactive Protein (CRP) in the liver but also fibrinogen, PAI-1, and serum-A amyloid protein. On the other hand, CRP amplify immunological response inducing leukocyte adhesion molecules and chemokines production in the endothelial cells, and show synergetic action with bacterial polysaccharides inducing monocyte grow factors [53]. Interleuquine IL-1 provokes synthesis of PAI-1 in the endothelial cells whereas IL-4 induces plasminogen tissue activator (t-PA) by monocytes. Cell surface-based signaling system CD40L ligand (CD154), binding to its receptor CD40 on the leucocyte, can induce tissue factor expression [50]. Platelets can express CD154, the molecule that regulates tissue factor gene expression in the macrophage and smooth muscle cells [51,52]. As can be seen, this process is a complex feed-back in which inflammation favors thrombosis, anti-inflammatory treatment has antithrombotic effects and viceversa [54,55].
Immunological system and atherogenesis
Atherosclerosis is related to activation of the immunological system [56]. The developing atherosclerotic plaque are infiltrated not only by macrophage but by T-lymphocyte (CD-4)Th called helper, and T-lymphocyte CD-8, that suggest a specific immunological response [57]. Never the less investigators have not yet reached an agreement on whether the effect is harmful or beneficial on the developing plaque. T-lymphocytes (CD4)Th1 produce TNF-α, Interferon-γ and IL-6, are all pro-inflammatory compounds that activate macrophage and are responsible for the late hypersensitivity reactions. On the other hand, T-lymphocytes (CD-4)Th2 generate IL-4, IL-5, IL-10 and IL-13, all of these anti-inflammatory molecules that promote antibody responses and energetically inhibit macrophage activity [58]. In the atherosclerotic plaque of experimental animals and in human beings, inflammatory cytokines produced by T-lymphocyte (CD4)Th1, such as Interferon-γ and IL-12, have been found in a pro-inflammatory surrounding similar to reumathoid arthritis [59,62]. In other plaques they have not been detected, so it is suspected that there is a reduction in the inflammatory response which leads to the believe that the balance between T-lymphocytes (CD-4)Th1 and Th2, may play an important role in the progression or regression of the plaque. In this respect statins play a role modulating immunological activity [63-65].
Sphingomyelinase is another immunological mediator produced by macrophage and endothelial cell when stimulated by inflammatory cytokines. It is one of the substances that are responsible for oxidized lipoproteins passing through the endothelium, for foam cells formation and progression, and complication of the atherosclerotic plaque [66,67].
From endothelial dysfunction to acute ischemic syndrome
Atherosclerosis has been considered as a disease of "four concepts". In a substudy of 5.209 patients from the Framingham Study followed for 10 years, it was seen that those patients with peripheral vascular disease had more probabilities of having an Acute Myocardial Infarction (AMI) or a Stroke, while those who had had an AMI had more possibilities of having a Stroke or peripheral vascular disease. Also those who had suffered a Stroke had more chances of having an AMI or peripheral vascular disease. From these observation the first concept, that atherosclerosis is a diffuse disease, was postulated [68,69].
The finding of lesions in different stages of development throughout the body, and inclusive in the same territory, lead to the second concept that "atherosclerosis is a heterogeneous and multiform disease" [69].
In advanced stages of arteriosclerosis, with stage IV and Va by AHA classification or Ross type III lesion, two types of lesions can be distinguished: I) Stable or fibrous plaque, with a small and generally central lipid core protected by a thick and resistant cover with a high content of collagen and without signs of inflammation. These lesions usually obstruct the vessel significantly and are easily seen by arteriography. II) High Risk, unstable or vulnerable plaque, with a large lipid core usually eccentric, covered by a weak and thin fibrous cap with little collagen and large quantities of macrophage and T-lymphocytes that are expression of a great inflammatory reaction that seldomly occlude the vessel significantly and is frequently not appreciated by angiography. This has generated the third concept that the "quality of the plaque is more important than the size", as proved by the fact that these plaques rupture easily and are responsible for the majority of the acute coronary syndromes. According to a metaanalysis by Falk et al, these high risk plaque which obstruct less than 70% and even less than 50% usually are asymptomatic, they are not easily recognized and not considered significant by angiography are responsible for 86% of acute coronary syndromes [69-72].
In atherosclerotic disease classical risk factors play an important part; high cholesterol serum levels, arterial hypertension, smoking, obesity, sedentariness, and the so called new risk factors such as hyperhomocysteinemia, Lp(a) lipoprotein, Cytomegalovirus, Chlamydia pneumoniae, Helicobacter pilorae, Bacteroides gingivalis, genetic factors (gene ECA, gene HLA and others), serum inflammatory markers (CRP, serum-A amyloid protein, and others), and prothrombotic factors (PAI-I, D-dimer, fibrinogen, von Willebrand, etc.), microalbumineamia, all of these factors contribute, in different degrees, to make acute coronary syndromes happen. This suggests the fourth concept, that "atherosclerosis is an inflammatory, immunological, polygenic and multifactorial disease" [72-77].
The evolution of the disease can be slow and patients with risk factors develop chronic or unstable angina or AMI, this form is observed in less than 40%, or it can develop abruptly in patients who had low risk and were asymptomatic when they develop unstable angina, AMI or sudden death. This is found in more than 60% of patients and this fact obliges us to find another explanation based on a new physiopathological model.
All the experience obtained in the last years, suggests that the endothelium's dysfunction is no only the initial stage of the atherosclerotic disease that generates plaque formation, but also can causes plaque growth, and unprotected the high risk plaque leading to develop a vascular event. Between these two extremes endothelial dysfunction is responsible for all the plaque growth, differences in plaque development and plaque characteristics. For all these reasons, endothelial dysfunction is one of the principal mechanisms in atherosclerotic disease [78]. The presence of classical and new risk factors generates a chronic exogenous state of injury to the endothelium that promotes abnormal response, vasoconstriction, accumulation of inflammatory cells, migration of smooth muscle cells, increased cytokine production, etc, all these factors help atheromatous plaque formation and in turn generate a negative feedback that leads to a second injury, this time endogenous, that finally leaves the plaque unprotected allowing it to rupture or erosion and triggering thrombogenic phenomena [79].
Diabetes and endothelium
Both type 1 (insulin-dependent) and type 2 (no insulin-dependent) diabetic patients, have mostly been described under enhanced oxidative stress, and both conditions are known to be powerful and independent risk factors for coronary heart disease, stroke, and peripheral arterial disease.
Hyperglycemia causes glycosylation of proteins and phospholipids, thus increasing intracellular oxidative stress. Nonenzymatic reactive products, known as Maillard or browning reaction, glucose-derived Schiff base, and Amadori products, form chemically reversible early glycosylation products which subsequently rearrange to form more stable products, some of them on long-lived proteins (e.g. vessel wall collagen) and continue undergoing complex series of chemical rearrangements to form advanced glycosylation end products (AGEs). Once formed, AGEs are stable and virtually irreversible. AGEs generate ROS with consequent increased vessel oxidative damage [80].
Phagocytes have specialized receptors for AGEs, their activation leading to oxidation of lipoproteins, especially the phospholipid component in LDL, and stimulating an immune-inflamatory response and a thrombogenic response through Tromboxane A2 release and platelet aggregation induction. Diabetic patients have increased levels of inflammatory markers, including CRP, with proinflammatory and proatherogenic properties.
The impressive correlation between coronary artery disease and alterations in glucose metabolism has raised the hypotesis that atherosclerosis and diabetes may share common antecedents. Large-vessel atherosclerosis can precede the development of diabetes, suggesting that rather than atherosclerosis being a complication of diabetes, both conditions may share genetic and environmental antecedents, a "common soil" [81].
Published data suggest that abnormal endothelial function precedes other evidence of vascular disease and that the progresion of metabolic syndrome to type 2 diabetes parallels the progression of endothelial dysfunction to atherosclerosis. Both type 1 and type 2 diabetes, like metabolic syndrome and other cardiovascular risk factors determine an abnormal endothelium response thought to precede the development of atherosclerosis.
Lipoprotein-associated phospholipase A2 (LP PLA2)
The lipoprotein-associated phospholipase A2 (LP PLA2), alternatively termed as platelet activating factor acetylhydrolase, is a calcium-independent phospholipase which hydrolyzes specifically the short acyl groups at the sn-2 position of the phospholipid substrate. Thus LP PLA2 plays a key role in the degradation of proinflammatory oxidized phospholipids (oxPL) and in the generation of lysophosphatidylcholine (lyso-PC) and oxidized fatty acids. LP PLA2 can also hydrolyze short-chain diacylglycerols, triacylglycerols, and acetylated alkanols, and displays a PLA1 activity [82].
The LP PLA2 has been considered a risk marker for endothelial dysfunction in diabetic patients. LP PLA2 levels have show a significant and positive correlation with CRP levels, reflecting the possible relation of LP PLA2 with inflammatory activity in atherosclerotic arteries [83]. Packard et al confirmed this previous data in a substudy of the West of Scotland Coronary Prevention Study in which they compared different inflammatory markers as cardiovascular risk predictors [84]. They found that in these patients LP PLA2 levels had a strong, positive association with the development of coronary events i.e., myocardial infaction, cardiovascular death or revascularization procedure, which was not confounded by other risk factors. The patients in which LP PLA2 levels were in the highest quintile had almost twice the risk of those in whom they were in the lowest quintile [85].
Myeloperoxidase and Paraoxonase are other enzymes involved in the evolution of endothelial dysfunction associated with type-2 diabetes [86].
Erectile dysfunction, sildenafil and endothelial function
Erectile dysfunction is an important complication of cardiovsascular disease and can work as a marker of endothelial function as well as a predictor of elevated risk of cardiovascular disease. About 30% of diabetic patients are afected by it [87].
Ischaemic heart disease can contribute to erectile dysfunction usually through fear although it can also be the first expression of coronary heart disease. Endothelial dysfunction is often found in patients with erectile dysfunction.
Erection is produced when erotic stimuli are percived by diferent senses and reach the hypothalamus thus inhibiting the simpathetic tone and releasing NO at the non adrenergic and non colinergic nerve terminals and in the endothelial cells of the penile arterioles. The NO reaches the cavernous smooth muscle and the arteriolar wall activating the enzime guanylate cyclase that degrades GTP releasing GMP, which reduces the smooth muscle Ca2+ uptake and produces vasodilatation. Phosphodiesterase-5 (PDE-5) reverts this situation by changing GMP back to GTP. This enzime is found in the corpus cavernosum, blood vessels and trachea, but not in the myocardium, and is inhibited by sildenafil, that in this way prolongs erection. Sildenafil is very highly selective for PDE-5 compared to other PDE, so as to be considered to have a PDE-5/PDE-3 selectivity ratio equal to 4000:1. This fact does not allow sildenafil to have a measurable positive inotropic effect on the myocardium [88].
Assessment endothelial function. Invasive and non-invasive techniques
Endothelial dysfunction, or loss or reduction of its capacity of defense against proatherogenic factors, is obtained evaluating any of the endothelium functions. For example, quantifying circulating adhesion molecules, proatherogenic substances, antifibrinolytics, evaluation of serum markers of inflammation, etc. All these are direct or indirect markers the endothelium capacity to protect against new atherosclerotic lesions or protect existing lesions from causing a vascular event. Because of the ease with which it can be done and its reliability, the most commonly used test in basic science and clinical research is endothelial dependent vasodilatation modulated by flow, considered at this moment the gold standard [78].
The first experiments to evaluate endothelial dependent vasodilatation were performed by invasive techniques catheterizing coronary arteries, were drugs that induced NO release were injected, such as acetylcholine, metacholine, papaverine, P substance, etc, and then measuring the percentage of vasodilatation. Ludmer et al [11], found that injecting acetylcholine in normal coronary arteries produced endothelial dependent vasodilatation, were as in coronary arteries with moderate or severe atherosclerotic lesions a paradoxical vasoconstriction was obtained, indicating that endothelial dysfunction was present. The paradoxical vasoconstriction is due to stimulation of the muscarinic receptors of the smooth muscle cells by direct acetylcholine action. It was also found that injecting nitroglycerine, an NO donor, there was always vasodilatation, in this case considered endothelium independent vasodilatation. Vita et al [88], using the same method found that the amount of coronary vasodilatation obtained by acetylcholine diminished in an inverse ratio with the increase of total cholesterol or LDL-cholesterol levels. They also observed that the presence of cardiovascular risk factors, alone or combined, kept an inverse linear relationship with the endothelium dependent vasodilatation response, evidencing the additive effects on endothelial dysfunction [88].
Later forearm pletismography was used. This technique could be considered partially invasive. It was performed by placing the forearm in a pletismograph for veins impedance and then injects the drug being studied, usually acetylcholine or metacholine, into the brachial artery. Panza et al [89-91], studying hypertensive patients, found them to have less vasodilatation response than normal controls. Also, hypertensive patients had an increase in vascular resistance in comparison with normotensive patients, which was permitted by the endothelial dysfunction. As endothelium maintains a constant vasodilatation level produced by NO, it was considered that in hypertension there was an endothelial dysfunction that reduced NO release, and with that a reduced basal vasodilatation. This led them to postulate that endothelial dysfunction could be one of the causes of hypertension. Later on, other working groups concluded that endothelial dysfunction was probably a consequence and not the cause of hypertension [88-91]. This technique mainly evaluates the resistance arteries response.
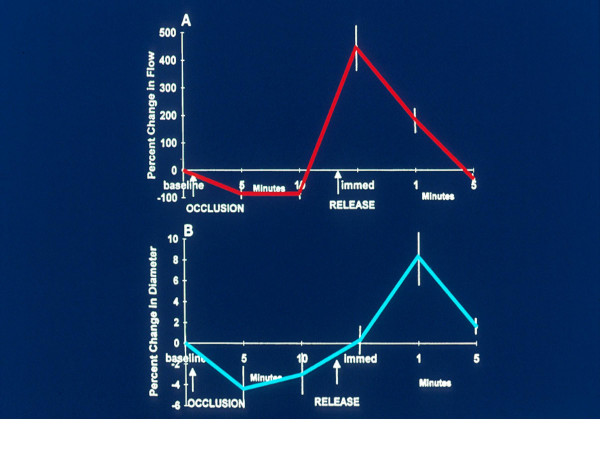
In the last years, Celermajer et al [92], assessed the level of vasodilatation by ultrasonography. This technique, non-invasive and easy to repeat, by which due to the changes that occur through time allow us to learn the natural evolution of the disease or see the changes produced by the different treatments given. Consists in causing forearm ischaemia and observes the amount of post ischaemic vasodilatation. Ischaemia is produced by compressing the forearm by inflating a conventional cuff for measuring blood pressure 30 mmHg above systolic pressure for five minutes. When the pressure is released there is a marked increase of flow to the forearm and this increases shear stress which in turn stimulates NO release that causes vasodilatation. The amount of vasodilatation is directly proportional to the amount of NO released by the endothelium and this allows us to evaluate endothelial function. The increase in flow and vasodilatation is measured by high-resolution ultrasonography of the brachial artery, and is expressed as a percentage of the increase of the basal values [93,94]. It can also be performed on the radial and femoral arteries [92,94]. This technique evaluates mainly the conductance vessels, different from pletismography that mainly evaluates the resistance vessels, although both methods test NO release (Figure 4).
Figure 4.
Flow mediated post-ischemic endothelial dependent vasodilatation. Percent change in flow and diameter after occlusion. From Correti MC, et al.[93]
This method has shown a gradual decrease in the endothelium dependent response depending on age, due to gradual loss of NO synthesis by the endothelial cells [95-97]. This same response is seen in patients with atherosclerotic lesions whether symptomatic or asymptomatic [97-99], in post-menopause women due to lack of estrogen [100,101], and in presence of cardiovascular risk factor such as hypercholesterolemia [102], hypertension [103], active smoking [104,105], passive smoking [106], obesity [107], diabetes [108], sedentariness [109], and hyperhomocysteinemia [110]. Mild infections [111] and increasing levels of CPR may also decrease de endothelial response [112]. The reversal of the cardiovascular risk factors allows a better endothelial function increasing the endothelial dependent vasodilatation which expresses an increase in NO release. The reduction of serum cholesterol levels [113-116], arterial pressure control [103,113], quit smoking [94], weight control [107], diabetes improvement [118], and physical activity [119] improving the endothelial dependent response, indicating more ON released. The administration of L-arginine, a NO precursor, increases vasodilatation in patients with high cholesterol, coronary artery disease and heart failure [120-122]. As has been explained previously, all these risk factors act by a common pathway, oxidative stress, with increased production of ROS [124,125]. It has been observed that giving anti oxidant vitamins C and E diminishes the production of ROS [126-128], including when given before a meal with a high fat content to reduce oxidation of post absorption fatty acids and tryglicerydes [129], it is also effective before smoking [130]. Recently it has been reported that there is an additive effect with the simultaneous administration of a statin and ACE inhibitor in hipercholesterolemic coronary artery disease patients (Figure 5) [131].
Figure 5.
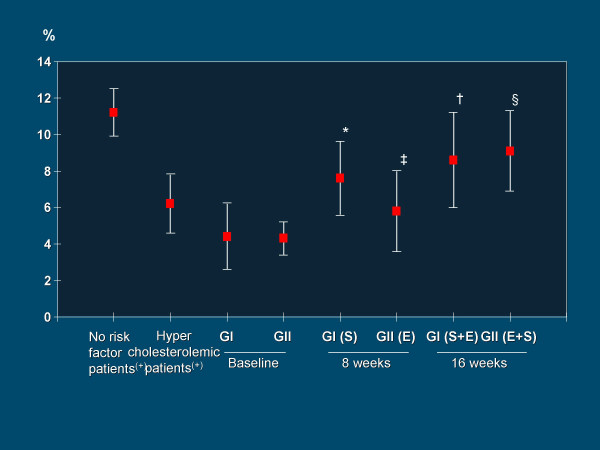
Flow mediated post-ischemic endothelium-dependent vasodilatation expressed as percent increase in arterial diameter (mean ± SD) with respect to baseline values in hypercholesterolemic coronary artery disease patients under the effects of the statin Simvastatin and the ECA inhibitor Enalapril, either separately or combined. E: Enalapril, GI: Group I, GII: Group II, S: Simvastatin. * p < 0.001 vs baseline, ‡ p < 0.01 vs Baseline, † p < 0.05 vs 8 weeks, § p < 0.001 vs 8 weeks. From Esper RJ, et al.[131]
The National Cholesterol Education Program (NCEP) recommends a maximum LDL cholesterol level of 160 mg/dl in healthy population without risk factors, 130 mg/dl for primary prevention, 100 mg/dl for secondary prevention and 70 mg/dl for high risk patients. Nevertheless, Steinberg et al [132], have evaluated endothelial function in healthy young patients with this technique without cardiovascular risk factors and cholesterol levels between 146 and 195 mg/dl and LDL-cholesterol between 87 and 119 mg/dl, and found that the lower the total cholesterol and LDL cholesterol levels, the better the vasodilatation response, as an expression of better endothelial response, which leads to the idea that the lower the cholesterol the better the endothelial response [114].
Although the method of evaluating endothelial response by measurements on the brachial artery is non-invasive and easily repeated, without any risk for the patient, it is time consuming and needs skilled and patient operators [78]. With just a small displacement of the transducer the results are altered. Some automated systems have shown a reduction in the variation between operators [78,133,134]. Some investigators have developed different equipment to maintain the transducer in a constant position. It is a well known fact that the response is different whether the cuff is positioned on the arm or forearm. Recently, an International task force published the guidelines for the techniques and assessment of this method [94].
Looking for simpler methods of performing these procedures laser-Doppler has recently been used. This technique considers the vasodilatation and the increase in blood flow, evaluation tissue perfusion. It has the advantage of being simple with immediate results and does not need skilled operators [135,136].
Prognostic value of post-ischaemic flow mediated endothelium dependent vasodilatation
Although a lot has been published about post-ischemic flow mediated endothelium dependent vasodilatation as a test for endothelial function the same doubts remain: can we compare the brachial artery with the coronary artery?, and, does this technique have a prognostic value?.
The endothelial function of the brachial artery measured by this method has shown a good correlation with the coronary artery response at the same time and in the same patient with acetylcholine and with other methods which has led to consider the brachial artery as a "surrogate" of the coronary artery [137,138]. It has also been shown at necropsy that patients with grade I-III lesions in the left descending coronary artery, had lesions of the same type and severity in their brachial artery, confirming the similarity [138]. Respect the sensibility of the method, using 7.5 Mhz or more transducers, modest changes of 0,1 mm can be measured precisely and are easily reproduced [78,94]. It has been shown that the inter-observer variation, with qualified observers is 0,1 mm [78,94].
With respect of the prognostic value, as this is a new method, not enough time has elapsed for a proper evaluation [139]. Never the less, an excellent correlation has been noted with the severity of the vascular lesions, even when they are asymptomatic and undetectable by the usual clinical methods. It has also been reported that patients without cardiovascular disease but with risk factors, had a brachial artery vasodilatation response diminished when compared with a normal population, and if they had a peripheral artery disease the response was even more diminished compared with the positive risk factor population. This leads to the assumption that as the severity of the lesions increases, so does the endothelial dysfunction [140].
Some authors have related left ventricular mass with endothelial function, and have reported that with normal ventricular geometry, endothelial function was within normal limits, were as in patients with concentric remodeling of the left ventricle showed a depressed endothelial function, and in patients with concentric or eccentric left ventricular hypertrophy the decrease of endothelial function was even greater [141]. The functional class by stress test also has a close relation with endothelial function. Those patients who reached 100 to 150 watts had a normal endothelial dependent vasodilatation similar to controls, but if they reached 75 or less watts, the vasodilatation response was depressed. The response to nitroglycerine showed a similar response in both groups.
Schachinger, et. al. [142] in an average of 82 month follow up, documented 14 major cardiovascular events, sudden death, AMI, unstable angina, stroke, or revascularization procedures, in patients in the inferior tertile of the vasodilatation response, and found 5 events in the superior tertile of the population. Both the endothelial dependent and endothelial independent response and their ratio (EDV/EIV) were strongly related to future cardiovascular events. Neunteufl et. al. [143] in a long follow up, found that the depressed vasodilatation response can predict the cardiovascular event risk for the next 5 years. In patient with normal vasodilatation versus abnormal vasodilatation, with a cut off at >10% in normal and <10% in abnormal response, the need for PTCA or CABG differed significantly, with a greater tendency of AMI in the patients with a poor response. Considering the total event rates, the flow mediated post-ischaemic endothelial vasodilatation response measured by high resolution ultrasonography of the brachial artery showed a sensitivity of 86% with a 51% specificity and a very important negative predictive value of 93%.
Al Suwaidi et. al. [144] studied patients with mild coronary artery disease (<30% stenosis) with quantitative coronary angiography with a follow up of 8 months, and found that patients with severe endothelial dysfunction with acetylcholine had a significant increase of events, 14% when compared to patients with a normal or mildly depressed vasodilatation response.
Schoeder et. al. [145] compared post ischaemic vasodilatation response of the brachial artery with the predictive value of angina, stress test and radioisotope myocardial perfusion. They found that this test had a sensitivity of 71%, a specificity of 81% and a positive predictive value of 95%, similar values to the other three diagnostic tests.
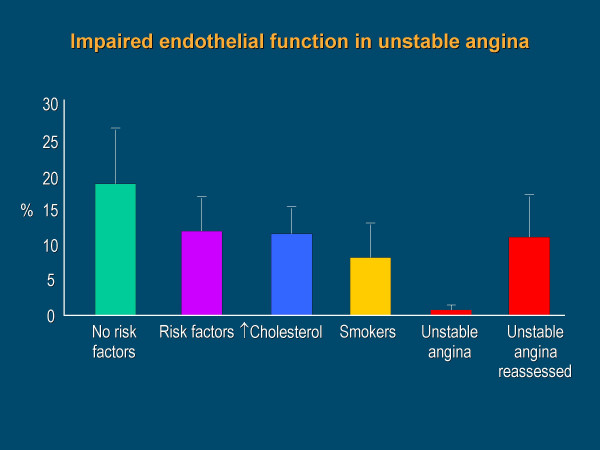
Our group [146], found that endothelial dependant vasodilatation during unstable angina is practically absent, creating a doubt about whether "the endothelial dysfunction is caused by the plaque rupture or if it existed previously and did not protect the high risk atheromatous plaque allowing the acute coronary event". Based on this study and comparing with what happens to the ischaemic myocardium, created the concept of "stunned" endothelium to explain the low vasodilatation response in acute ischaemia and "hibernated" endothelium when the poor response is chronic, as observed with hypercholesterolemia and the presence of other risk factor (Figure 6).
Figure 6.
Endothelium depended vasodilatation in no cardiovascular risk factor subjects and in patients with risk factors, hypercholesterolemia, smokers, within 24 hours of rapidly stabilized unstable angina, and approximately 30 days later, after pharmacological treatment. From Esper RJ, et al.[146]
Cohen et. al. [147] in a subgroup of patients taking part in the CARE trial, studied the brachial artery vasodilatation with ultrasound and found that post AMI patients treated with statins and with cholesterol values considered "normal", had a significantly greater vasodilatation than the group that received placebo. The magnitude of the increase was correlated with the decrease of the LDL cholesterol fraction. It has been considered that the improvement of the vasodilatation response could be the main mechanism of the reduction of recurrent events found in the CARE trial.
It has been reported that non-invasive assessment of endothelial function in the brachial artery predicts perioperative cardiovascular events in patients undergoing vascular surgery [148]. In 1479 subjects attending a routine exam at the Framingham Study, it has been shown that advanced age and systolic blood pressure are strongly associated with decreased endothelial function, and the dilatation was larger in females vs males [149].
Ganz et al. discussed the pronostic value of the post-ischaemic flow mediated endotelial vasodilatation in a revision of the last 10 trials on the subject, and concluded that is a useful diagnosis procedure with a high pronostic value. In the same issue, in editorials by Verma et al, and Willerson et al., arrived at similar conclusions [150-152].
The atherosclerotic lesion always starts with endothelial dysfunction and progresses with the persistence of this dysfunction [78,153-157]. From a practical point of view, the established vascular lesions seen by angiography, Intravascular Ultrasound (IVUS), Magnetic Nuclear Resonance, Computed bean Tomography, Carotid ultrasonography, etc, could be considered like the electrocardiogram in ischemic heart disease, were as the techniques for endothelial function evaluation would be similar to the conclusions reached with stress tests, reassuring the hypothesis that functional alterations precede the anatomic lesions in the development of atherosclerosis and the ischaemic cascade [156,157]. This idea is reinforced by the relation between endothelial dysfunction and microalbuminuria, another risk marker that is not often taken into consideration [158].
Contributor Information
Ricardo J Esper, Email: ricardo.esper@fibertel.com.ar.
Roberto A Nordaby, Email: rnodaby@fibertel.com.ar.
Jorge O Vilariño, Email: jorvil@waycom.com.ar.
Antonio Paragano, Email: antonioparagano@hotmail.com.
José L Cacharrón, Email: cacharron@intramed.net.ar.
Rogelio A Machado, Email: rmachado@uolsinectis.com.ar.
References
- Rubanyi GM. The role of endothelium in cardiovascular homeostasis and diseases. J Cardiovasc Pharmacol. 1993;22:S1–S14. doi: 10.1097/00005344-199322004-00002. [DOI] [PubMed] [Google Scholar]
- Drenckhahan D, Ness W. The endothelial contractile cytoskeleton. In: Born GVR, Schwartz CJ, editor. Vascular Endothelium Physiology, Pathology, and Therapeutic opportunities. Stuttgart, Schattauer-Verlag; 1997. pp. 1–25. [Google Scholar]
- Andersen RGW. Caveolae: where incoming and outgoing messengers meet. Proc Natl Acad Sci USA. 1993;90:10909–10913. doi: 10.1073/pnas.90.23.10909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esper RJ, Vilariño JO. La disfunción endotelial. Consideraciones fisiopatológicas y diagnósticas. In: Esper RJ, Vilariño JO, editor. La placa de alto riesgo. Prous Sciences, Barcelona; 2002. pp. 19–43. [Google Scholar]
- Esper RJ, Vilariño JO. La disfunción endotelial. In: Esper RJ, editor. Aterotrombosis en el tercer milenio. Prous Sciences, Barcelona; 2004. pp. 49–83. [Google Scholar]
- Vanhoutte PM. How to assess endothelial function in human blood vessels. J Hypertension. 1999;17:1047–1058. doi: 10.1097/00004872-199917080-00001. [DOI] [PubMed] [Google Scholar]
- Loscalzo J, Welch G. Nitric oxide and its role in the cardiovascular system. Prog Cardiovasc Dis. 1995;38:87–104. doi: 10.1016/S0033-0620(05)80001-5. [DOI] [PubMed] [Google Scholar]
- Jones CJH, Kuo L, Davis MJ, deFily DV, Chillian WM. Role of nitric oxide in coronary microvascular responses to adenosine and increased metabolic demand. Circulation. 1996;91:1807–1813. doi: 10.1161/01.cir.91.6.1807. [DOI] [PubMed] [Google Scholar]
- Cooke JP, Tsao PS. Go with the flow. Circulation. 2001;103:2773–2775. doi: 10.1161/01.cir.103.23.2773. [DOI] [PubMed] [Google Scholar]
- Miura H, Watchel RE, Liu Y, Loberiza F, Saito T, Miura M, Gutterman DD. Flow-induced dilation of human coronary arterioles: important role of Ca(2+)-activated K(+) channels. Circulation. 2001;103:1992–1998. doi: 10.1161/01.cir.103.15.1992. [DOI] [PubMed] [Google Scholar]
- Ludmer PL, Selwyn AP, Shook TL, Wayne RR, Mufge GH, Alexander RW, Ganz P. Paradoxical vasoconstriction induced by acetylcholine in atherosclerosis coronary arteries. N Engl J Med. 1986;315:1046–1051. doi: 10.1056/NEJM198610233151702. [DOI] [PubMed] [Google Scholar]
- Asakura K, Karino T. Flow patterns and spatial distribution of atherosclerosis lesions in human coronary arteries. Circ Res. 1990;66:1045–1066. doi: 10.1161/01.res.66.4.1045. [DOI] [PubMed] [Google Scholar]
- Makek AM, Alper SL, Izumo S. Hemodynamic shear stress and its role in atherosclerosis. JAMA. 1999;282:2035–2042. doi: 10.1001/jama.282.21.2035. [DOI] [PubMed] [Google Scholar]
- Jalali S, Li YS, Sotoudeh M, Yuan S, Li S, Chien S, Shyy JY. Shear stress activates p60src-Ras-MAPK signalling pathways in vascular endothelial cells. Arterioscler Thromb Vasc Biol. 1998;18:227–234. doi: 10.1161/01.atv.18.2.227. [DOI] [PubMed] [Google Scholar]
- Feldman CL, Stone PH. Intravascular hemodynamic factors responsible for progression of coronary atherosclerosis and development of vulnerable plaque. Curr Opin in Cardiol. 2000;15:430–440. doi: 10.1097/00001573-200011000-00010. [DOI] [PubMed] [Google Scholar]
- Cooke JP, Tsao PS. Is NO an endogenous antiatherogenic molecule? Arterioscler Thromb. 1994;14:753–759. doi: 10.1161/01.atv.14.5.653. [DOI] [PubMed] [Google Scholar]
- Baldwin A. The transcription factor NK-6B and human disease. J Clin Invest. 2001;107:3–6. doi: 10.1172/JCI11891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tak P, Firestein G. NF-6B. A key role in inflammatory disease. J Clin Invest. 2001;107:7–11. doi: 10.1172/JCI11830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Libby P. Current concepts of the pathogenesis of the acute coronary syndrome. Circulation. 2001;104:365–372. doi: 10.1161/01.cir.104.3.365. [DOI] [PubMed] [Google Scholar]
- Thurberg B, Collins T. The nuclear factor-kappa B/inhibitor of kappa B autoregulatory system and atherosclerosis. Curr Opin Lipidol. 1998;9:387–396. doi: 10.1097/00041433-199810000-00002. [DOI] [PubMed] [Google Scholar]
- Marx N, Sukhova GK, Collins T. PPARα activactors inhibitor cytokine-induced vascular cell adhesion molecule-1 expresion in human endothelial cells. Circulation. 1999;99:3125–31. doi: 10.1161/01.cir.99.24.3125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marx N, Mackman N, Schoenbeck U. PPARα activactors inhibit tissue factor expression and activity in human monocytes. Circulation. 2001;103:213–219. doi: 10.1161/01.cir.103.2.213. [DOI] [PubMed] [Google Scholar]
- Dzau VJ. Vascular angiotensin pathways: a new therapeutic target. J Cardiovasc Pharmacol. 1987;10:S9–S16. [PubMed] [Google Scholar]
- Johnston CJ. Renin-angiotensin system: a dual tissue and hormonal system for cardiovascular control. J Cardiovasc Pharmacol. 1992;20:S1–S5. [Google Scholar]
- Kawano H, Do YS, Kawano Y, Starnes V, Barr M, Law RE, Hsueh WA. Angiotensin II has multiple profibrotic effects in human cardiac fibroblast. Circulation. 2000;101:1130–1137. doi: 10.1161/01.cir.101.10.1130. [DOI] [PubMed] [Google Scholar]
- Biasucci LM, Vitelli A, Liuzzo G, Altamura S, Caliguri G, Monaco C, Rabuzzi AG, Ciliberto G, Maseri A. Elevated levels of interleukin-6 in unstable angina. Circulation. 1996;94:874–877. doi: 10.1161/01.cir.94.5.874. [DOI] [PubMed] [Google Scholar]
- Kacimi R, Long CS, Karliner JS. Chronic hypoxia modulates the interleukin-1β-stimulated inducible nitric oxide synthase pathway in cardiac myocites. Circulation. 1997;96:1937–1943. doi: 10.1161/01.cir.96.6.1937. [DOI] [PubMed] [Google Scholar]
- Torre-Amione G, Kapadia S, Lee J, Bies RD, Lebovitz R, Mann DL. Expression and functional significance of tumor necrosis factor receptors in human miocardium. Circulation. 1995;92:1487–1493. doi: 10.1161/01.cir.92.6.1487. [DOI] [PubMed] [Google Scholar]
- Smith SC, Allen PM. Neutralization of endogenous tumor necrosis factor ameliorates the severity of myosin-induced myocarditis. Circ Res. 1992;70:856–863. doi: 10.1161/01.res.70.4.856. [DOI] [PubMed] [Google Scholar]
- Matsumori A, Yamada T, Suzuki H, Matoba Y, Sasayama S. Increased circulating cytokines in patients with myocarditis and cardiomyophaty. Br Heart J. 1994;72:561–566. doi: 10.1136/hrt.72.6.561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valen G, Yan Z, Hansson GK. Nuclear factor Kappa-B and the heart. J Am Coll Cardiol. 2001;38:307–314. doi: 10.1016/S0735-1097(01)01377-8. [DOI] [PubMed] [Google Scholar]
- Siragy H. Angiotensin II receptor bloquers: review of the binding characteristics. Am J Cardiol. 1999;84:3–8. doi: 10.1016/S0002-9149(99)00727-4. [DOI] [PubMed] [Google Scholar]
- Nickening G, Harrison DG. The AT1-type angiotensin receptor in oxidative stress and atherogenesis. Part I: oxidative stress and atherogenesis. Circulation. 2002;105:393–396. doi: 10.1161/hc0302.102618. [DOI] [PubMed] [Google Scholar]
- Benter IF, Ferrario CM, Morris M, Diz DI. Antihypertensive actions of angiotensin(1–7) in spontaneously hypertensive rats. Am J Physiol. 1995;269:H313–H319. doi: 10.1152/ajpheart.1995.269.1.H313. [DOI] [PubMed] [Google Scholar]
- Givertz MM, Sawyer DB, Colucci WS. Antioxidants and myocardial contractility. Illuminating the "dark side" of β-adrenergic receptor activation. Circulation. 2001;103:782–783. doi: 10.1161/01.cir.103.6.782. [DOI] [PubMed] [Google Scholar]
- Peiser L, Mukhopadhyay S, Gordon S. Scavenger receptors in innate immunity. Curr Opin Immunol. 2002;14:123–128. doi: 10.1016/S0952-7915(01)00307-7. [DOI] [PubMed] [Google Scholar]
- Janeway CA, Jr, Medzhitov R. Innate immune recognition. Annu Rev Immunol. 2002;20:197–216. doi: 10.1146/annurev.immunol.20.083001.084359. [DOI] [PubMed] [Google Scholar]
- Leitinger N. Oxidized phospholipids as modulators of inflammation in atherosclerosis. Curr Opin Lipido. 2003;14:421–430. doi: 10.1097/00041433-200310000-00002. [DOI] [PubMed] [Google Scholar]
- Li D, Mehta JL. Antisense to LOX-1 inhibits oxidized LDL-mediated up-regulation of monocyte chemoatractant protein-1 and monocyte adhesion to human coronary artery endothelial cells. Circulation. 2000;101:2889–2895. doi: 10.1161/01.cir.101.25.2889. [DOI] [PubMed] [Google Scholar]
- Tsimikas S, Witztum JL. Measuring circulating oxidized low-density lipoprotein to evaluate coronary risk. Circulation. 2001;103:1930–1932. doi: 10.1161/01.cir.103.15.1930. [DOI] [PubMed] [Google Scholar]
- Libby P. Molecular bases of the acute coronary síndromes. Circulation. 1995;91:2844–2850. doi: 10.1161/01.cir.91.11.2844. [DOI] [PubMed] [Google Scholar]
- Li D, Mehta JL. Up-regulation of endothelial receptor for oxidized LDL (LOX 1) by oxidized and implications in apoptosis of human coronary artery endothelial cells: evidence from use of antisense LOX-1 mRNA and chemical inhibitors. Ateroscler Thromb Vasc Biol. 2000;20:1116–1122. doi: 10.1161/01.atv.20.4.1116. [DOI] [PubMed] [Google Scholar]
- Kadokami T, Frye C, Lemster B, Wagner CL, Feldman AM, McTiernan CF. Anti-Tumor necrosis factor-α antibody limits heart failure in a transgenic model. Circulation. 2001;104:1094–1097. doi: 10.1161/hc3501.096063. [DOI] [PubMed] [Google Scholar]
- Shah P, Kaul S, Nilsson J, Cercek B. Exploiting the vascular protective effects of high-density lipoprotein and its apolipoproteins. An idea whose time for testing is coming. Part I. Circulation. 2001;104:2376–2383. doi: 10.1161/hc4401.098467. [DOI] [PubMed] [Google Scholar]
- Böger R, Bode-Böger S, Szuba A, Tsao PS, Chan JR, Tangphao O, Blaschke TF, Cooke JP. Asymetric Dimethylarginina (ADMA). A novel risk factor for endothelial Dysfunction. Circulation. 1998;98:1842–1847. doi: 10.1161/01.cir.98.18.1842. [DOI] [PubMed] [Google Scholar]
- Ehara S, Ueda M, Naruko T, Haze K. Elevated levels of oxidated low density lipoprotein show a positive relationship with the severity of acute coronary syndrome. Circulation. 2001;103:1955–1960. doi: 10.1161/01.cir.103.15.1955. [DOI] [PubMed] [Google Scholar]
- Landmesser U, Harrison DG. Oxidant stress and marker for cardiovascular events. Ox marks the spots. Circulation. 2001;104:2638–2640. [PubMed] [Google Scholar]
- Sage PR, de la Lande IS, Stafford I, Bennett CL, Phillipov G, Stubberfield J, Horowitz JD. Nitroglycerin tolerance in human vessels. Evidence for impaired nitroglycerin bioconversion. Circulation. 2000;102:2810–2815. doi: 10.1161/01.cir.102.23.2810. [DOI] [PubMed] [Google Scholar]
- Gori T, Burstein JM, Ahmed S, Miner SES, Al-Hesayen A, Kelly S, Parker JD. Folic acid prevents nitroglycerin-induced nitric oxide sinthase dysfunction and nitrate tolerance. A human in vivo study. Circulation. 2001;104:1119–1123. doi: 10.1161/hc3501.095358. [DOI] [PubMed] [Google Scholar]
- March F, Schoembeck U, Bonnefoy JY. Activación the monocyte/macrophage related to acute atheroma complication by ligation of CD-40 induction of collagenase, stromelysin, and tissue factor. Circulation. 1997;96:396–399. doi: 10.1161/01.cir.96.2.396. [DOI] [PubMed] [Google Scholar]
- Henn V, Slupsky JR, Grafe M. CD-40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature. 1998;391:591–594. doi: 10.1038/35393. [DOI] [PubMed] [Google Scholar]
- von Hundelshausen P, Weber KSC, Huo Y. RANTES deposition by platelets triggers monocyte arrest on inflamed and atherosclerotic endothelium. Circulation. 2001;103:1772–1777. doi: 10.1161/01.cir.103.13.1772. [DOI] [PubMed] [Google Scholar]
- Yeh ETH, Anderson V, Pasceri V, Willerson JT. C-Reactive Protein. Linking inflammation to cardiovascular complications. Circulation. 2001;104:974–975. doi: 10.1161/01.cir.104.9.974. [DOI] [PubMed] [Google Scholar]
- Fuster V, Moreno P, Fayad ZA, Corti R, Badimon JJ. Atherotrombosis and high-risk plaque. Part I: Evolving concepts. J Am Coll Cardiol. 2005;46:937–954. doi: 10.1016/j.jacc.2005.03.074. [DOI] [PubMed] [Google Scholar]
- Libby P, Simon DI. Inflammation and trombosis. The clot thickens. Circulation. 2001;103:1718–1720. doi: 10.1161/01.cir.103.13.1718. [DOI] [PubMed] [Google Scholar]
- Nicoletti A, Caliguri G, Paulosson G. Functionality of specific immunity in atherosclerosis. Am Heart J. 1999;138:438–443. doi: 10.1016/S0002-8703(99)70271-1. [DOI] [PubMed] [Google Scholar]
- Mackay IR, Rosen FS. Autoimmune disease. N Engl J Med. 2001;345:340–350. doi: 10.1056/NEJM200108023450506. [DOI] [PubMed] [Google Scholar]
- Mosmann Tr, Cherwinsky H, Bond MW. Two type of murine helper-T cell clone: definition according to profiles of linphokine activities and secreted proteins. J Immunol. 1986;136:2348–2357. [PubMed] [Google Scholar]
- Lee TS, Yen HC, Pann CC. The role of interleukin 12 in the development of the atherosclerosis in apo E-deficient mice. Atherioscler Thromb Vasc Biol. 1999;19:734–742. doi: 10.1161/01.atv.19.3.734. [DOI] [PubMed] [Google Scholar]
- Zhou X, Paulsson G, Stemme S. Hypercholesterolemia is associated with a T-helper Th1/Th2 switch of the immune response in apo E-knockout mice. J Clin Invest. 1998;101:1717–25. doi: 10.1172/JCI1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasceri V, Yeh ET. A tale of two diseases: atherosclerosis and rheumatoid arthritis. Circulation. 1999;100:2124–2126. doi: 10.1161/01.cir.100.21.2124. [DOI] [PubMed] [Google Scholar]
- Laurat E, Poirier B, Tupin E. In vivo downregulation of T-helper cell 1 immune responses reduces atherosclerosis in apolipoprotein E-knockout mice. Circulation. 2001;104:197–202. doi: 10.1161/01.cir.104.2.197. [DOI] [PubMed] [Google Scholar]
- Kak B, Mulhaupt F, Myit S. Statins as newly reconognized type of immunomodulator. Nature Medicine. 2000;6:1399–1402. doi: 10.1038/82219. [DOI] [PubMed] [Google Scholar]
- Palinsky W. Immunomodulation: a New Role for statins? Nature Medine. 2000;6:1311–12. doi: 10.1038/82107. [DOI] [PubMed] [Google Scholar]
- Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352:1685–1695. doi: 10.1056/NEJMra043430. [DOI] [PubMed] [Google Scholar]
- Wong ML, Xie B, Beatini N, Phu P. Acute systemic inflammation up-regulates secretory sphingomyelinase in vivo: A possible link between inflammatory cytokines and atherosclerosis. PNAS. 2000;97:8681–8686. doi: 10.1073/pnas.150098097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xian CJ, Paultre F, Pearson TA, Reed RG. Plasma Sphingomyelin level as a Risk Factor for coronary artery disease. Arterios Throm and Vasc Biolog. 2000;20:2614–2618. doi: 10.1161/01.atv.20.12.2614. [DOI] [PubMed] [Google Scholar]
- Fuster V, Moreno PR, Fayad ZA, Corti R, Badimon JJ. Atherotrombosis and high risk plaque. Part I: Evolving concepts. J Am Coll Cardiol. 2005;46:937–954. doi: 10.1016/j.jacc.2005.03.074. [DOI] [PubMed] [Google Scholar]
- Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352:1685–1695. doi: 10.1056/NEJMra043430. [DOI] [PubMed] [Google Scholar]
- Falk E, Shah PK, Fuster V. Coronary plaque disruption. Circulation. 1995;92:657–671. doi: 10.1161/01.cir.92.3.657. [DOI] [PubMed] [Google Scholar]
- Ziada KM, Vince DG, Nissen SE, Tuzcu EM. Arterial remodeling and coronary artery disease: The concept of "dilated" versus "obstructive" coronary atherosclerosis. J Am Coll Cardiol. 2001;38:297–306. doi: 10.1016/S0735-1097(01)01374-2. [DOI] [PubMed] [Google Scholar]
- Ross R. Atherosclerosis – An inflammatory disease. N Engl J Med. 1999;340:115–126. doi: 10.1056/NEJM199901143400207. [DOI] [PubMed] [Google Scholar]
- Danesch J, Whincup P, Walker M, Lennon L, Thomson A, Appleby P, Rumley A, Lowe GDO. Fibrin D-dimer and coronary heart disease. Prospective study and meta-analysis. Circulation. 2001;103:2323–2327. doi: 10.1161/01.cir.103.19.2323. [DOI] [PubMed] [Google Scholar]
- Manunucci M. Von Willebrand factor. A marker of endothelial damage. Atheros thromb & vasc biol. 1998;18:1359–1362. doi: 10.1161/01.atv.18.9.1359. [DOI] [PubMed] [Google Scholar]
- Thompson SG, Kienast J, Pyke SD, Haverkate F. Hemostatic factors and the risk of miocardial infartion and sudden death in patients with angina pectoris. N Engl J Med. 1995;332:635–641. doi: 10.1056/NEJM199503093321003. [DOI] [PubMed] [Google Scholar]
- Jensen JS, Feldt-Rasmussen BF, Strandgaard S, Schroll M, Borch-Johnson K. Hypertension, microalbuminuria and risk of cardiac ischemic disease. Hypertension. 2000;35:898–903. doi: 10.1161/01.hyp.35.4.898. [DOI] [PubMed] [Google Scholar]
- Fuster V. Remodelado del trombo: un punto clave en la progresión de la aterosclerosis coronaria. Rev Española Cardiol. 2000;53:2–7. [PubMed] [Google Scholar]
- Esper RJ. Interrogando al endotelio. Rev Argent Cardiol. 2000;68:429–439. [Google Scholar]
- Esper RJ. Detección de la placa de alto riesgo. Intercontinental Cardiology. 2001;10:63–70. [Google Scholar]
- Fisman E, Esper RJ, Tenenbaum A. Diabetes y Enfermedad Cardiovascular: Aspectos Fisiopatológícos y Clínicos del Síndrome Metabólico y la Insuficiencia Cardíaca Congestiva. Buenos Aires, Maestría en Ateroesclerosis. 2005;8:1–32. [Google Scholar]
- Stern MP. Diabetes and cardiovascular disease. The "common soil" hypothesis. Diabetes. 1995;44:369–374. doi: 10.2337/diab.44.4.369. [DOI] [PubMed] [Google Scholar]
- Tselepis AD, John CM. Inflammation, bioactive lipids and atherosclerosis: potencial roles of a lipoprotein-associated phospholipase A2, platelet activating factor-acetylhydrolase. Atheroscler. 2002;3:57–68. doi: 10.1016/S1567-5688(02)00045-4. [DOI] [PubMed] [Google Scholar]
- Kaski JC, García-Moll X, Cosin-Sales J. Marcadores serológicos de inflamación y vulnerabilidad de la placa ateroesclerótica. In: Esper RJ, editor. Aterotrombosis en el tercer milenio. Barcelona, Prous Sciences; 2004. pp. 111–136. [Google Scholar]
- Packard CJ, O'Reilly DSJ, Caslake MJ, McMahon AD, Ford I, Cooney J, Macphee CH, Suckling KE, Krishna M, Wilkinson FE, Rumley A, Lowe GDO, Doherty G, Burczak JD. Lipoprotein-associated phopholipase A2 as an independent predictor of coronary heart disease. West of Scotland Coronary Prevention Study Group. N Engl J Med. 2000;343:1148–1155. doi: 10.1056/NEJM200010193431603. [DOI] [PubMed] [Google Scholar]
- Moldoveanu E, Tanaseanu C, Tanaseanu S, Kosaka T, Manea G, Marta DS, Popescu LM. Plasma markers of endotelial dysfunction in type 2 diabetics. Eur J Intern Med. 2006;17:38–42. doi: 10.1016/j.ejim.2005.09.015. [DOI] [PubMed] [Google Scholar]
- Herrmann HC, Chang G, Klugherz BD, Mahoney PD. Haemodynamic effects of sildenafil in men with severe coronary artery disease. N Engl J Med. 2000;342:1622–1626. doi: 10.1056/NEJM200006013422201. [DOI] [PubMed] [Google Scholar]
- Esper RJ, Vilariño JO. Tratamiento de la aterotrombosis y la disfunciòn endothelial. In: Esper RJ, editor. Arterotrombosis en el tercer milenio. Barcelona, Prous Sciences; 2004. pp. 165–222. [Google Scholar]
- Vita JA, Treasure CB, Nabel EG. Coronary vasomotor response to acetylcholine relate to risk factor for coronary artery disease. Circulation. 1990;81:491–498. doi: 10.1161/01.cir.81.2.491. [DOI] [PubMed] [Google Scholar]
- Panza JA, Quyyumi AA, Brush JE, Jr, Epstein SE. Abnormal endothelium-dependent vascular relaxation in patients with essential hypertension. N Engl J Med. 1990;323:22–27. doi: 10.1056/NEJM199007053230105. [DOI] [PubMed] [Google Scholar]
- Panza JA, Casino PR, Kilcoyne CM, Quyyumi AA. Role of the endothelium-derived nitric oxide in the abnormal endothelium-dependent vascular relaxation in patients with essential hypertension. Circulation. 1993;87:1468–1474. doi: 10.1161/01.cir.87.5.1468. [DOI] [PubMed] [Google Scholar]
- Panza JA, Quyyumi AA, Callahan TS, Epstein SE. Effect of antihypertensive treatment of endothelium-dependent vascular relaxation in patients with essential hypertension. J Am Coll Cardiol. 1993;21:1145–1151. doi: 10.1016/0735-1097(93)90238-v. [DOI] [PubMed] [Google Scholar]
- Celermajer DS, Sorensen KE, Gooch VM, Spiegelhalter DJ, Miller OI, Sullivan ID, Lloyd JK, Deanfield JE. Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet. 1992;340:1111–1115. doi: 10.1016/0140-6736(92)93147-F. [DOI] [PubMed] [Google Scholar]
- Corretti MC, Plotnik GD, Vogel RA. Technical aspects of evaluating brachial artery vasodilatation using high-frequency ultrasound. Am J Physiol. 1995;268:H1397–H1404. doi: 10.1152/ajpheart.1995.268.4.H1397. [DOI] [PubMed] [Google Scholar]
- Corretti MC, Todd J, Anderson TJ, Benjamin EJ, Celermajer DS, Charbonneau F, Creager MA, Deanfield JE, Drexler H, Gerhard-Herman M, Herrington D, Vallance P, Vita J, Vogel R. Guidelines for the ultrasound assessment of endotelial-dependent flow-mediated vasodilation of the brachial artery. J Am Coll Cardiol. 2002;39:257–265. doi: 10.1016/S0735-1097(01)01746-6. [DOI] [PubMed] [Google Scholar]
- Zeiher AM, Drexler H, Saurier B, Just H. Endothelium-mediated coronary blood flow modulation in humans: effect of age, atherosclerosis, hypercholesterolemia, and hypertension. J Clin Invest. 1993;92:652–662. doi: 10.1172/JCI116634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Celermajer DS, Sorensen KE, Spiegelhalter DJ, Georgakopoulos D, Robinson J, Deanfield JE. Ageing is associated with endothelial dysfunction in healthy men years before the age-related decline in women. J Am Coll Cardiol. 1994;24:471–476. doi: 10.1016/0735-1097(94)90305-0. [DOI] [PubMed] [Google Scholar]
- Vilariño JO, Cacharrón JL, Suarez DH, Kura M, Machado R, Bolaño AL, Esper RJ. Evaluación de la función endotelial por eco-Doppler. Influencia de la edad, sexo y factores de riesgo. Rev Argent Cardiol. 1998;66:523–532. [Google Scholar]
- Gordon JB, Ganz P, Nabel EG, Fish RD, Zebede J, Mudge GH, Alexander RW, Selwyn AP. Atherosclerosis influence the vasomotor response of epicardial coronary arteries to exercise. J Clin Invest. 1989;83:1946–1952. doi: 10.1172/JCI114103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nabel EG, Selwyn AP, Ganz P. Large coronary arteries in humans are responsive to changing blood flow: an endothelium-dependent mechanism that fail in patients with atherosclerosis. J Am Coll Cardiol. 1990;16:349–356. doi: 10.1016/0735-1097(90)90584-c. [DOI] [PubMed] [Google Scholar]
- Williams JK, Adams MR, Klopfenstein HS. Estrogen modulates responses of atherosclerotic coronary arteries. Circulation. 1990;81:1680–1687. doi: 10.1161/01.cir.81.5.1680. [DOI] [PubMed] [Google Scholar]
- Lieberman EH, Gerhard MD, Uehata A, Walsh BW, Selwyn AP, Ganz P, Yeung AC, Creager MA. Estrogen improves endothelium-dependent, flow-mediated vasodilation in postmenopausal women. Ann Intern Med. 1994;121:936–941. doi: 10.7326/0003-4819-121-12-199412150-00005. [DOI] [PubMed] [Google Scholar]
- Creager MA, Cooke JP, Mendelsohn ME, Gallagher SJ, Coleman SM, Loscalzo J, Dzau VJ. Impaired vasodilatation of forearm resistance vessels in hypercholesterolemic humans. J Clin Invest. 1990;86:228–234. doi: 10.1172/JCI114688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panza JA, Cannon RO, III, Eds . Endothelium, nitric oxide, and atherosclerosis. New York, Futura Publishing Inc; 1999. [Google Scholar]
- Celermajer DS, Sorensen KE, Georgakopuolos D, Bull C, Tomas O, Robinson J, Deanfield JE. Cigarette smoking is associated with dose-related and potentially reversible impairment of endothelium-dependent dilation in healthy young adults. Circulation. 1993;88:2149–2155. doi: 10.1161/01.cir.88.5.2149. [DOI] [PubMed] [Google Scholar]
- Zeiher A, Schächinger V, Minners J. Long-term cigarette smoking impairs endothelium-dependent coronary vasodilator function. Circulation. 1995;92:1094–1100. doi: 10.1161/01.cir.92.5.1094. [DOI] [PubMed] [Google Scholar]
- Celermajer DS, Adams MR, Clarkson P, Robinson J, McCredie R, Donald A, Denfield JE. Passive smoking and impaired endothelium-dependent arterial dilatation in healthy young adults. N Engl J Med. 1996;334:150–154. doi: 10.1056/NEJM199601183340303. [DOI] [PubMed] [Google Scholar]
- Mark AL, Correia M, Morgan DA, Schaffer RA, Haynes WG. Obesity-induced hypertension. New concepts from de emerging biology of obesity. Hypertension. 1999;33:537–541. doi: 10.1161/01.hyp.33.1.537. [DOI] [PubMed] [Google Scholar]
- Johnstone MT, Creager SJ, Scales KM, Cusko JA, Lee BK, Creager MA. Impaired endothelium-dependent vasodilation in patients with insulin-dependent diabetes mellitus. Circulation. 1993;88:2510–2516. doi: 10.1161/01.cir.88.6.2510. [DOI] [PubMed] [Google Scholar]
- Snell PG, Mitchell JH. Physical inactivity. An easily modified risk factor? Circulation. 1999;100:2–4. doi: 10.1161/01.cir.100.1.2. [DOI] [PubMed] [Google Scholar]
- Celermajer DS, Sorensen K, Ryalls M, Robinson J, Thomas O, Leonard JV, Deanfield JE. Impaired endothelial function occurs in the systemic arteries of children with homozygous homocystinuria but not in their heterozygous parents. J Am Coll Cardiol. 1993;22:854–858. doi: 10.1016/0735-1097(93)90203-d. [DOI] [PubMed] [Google Scholar]
- Charakida M., Donald AE, Terese M, Leary S, Halcox JP, Ness A, Smith GD, Golding J, Friberg P, Klein NJ, Deanfield JE. Endothelial dysfunction in childhood infection. Circulation. 2005;111:1660–1665. doi: 10.1161/01.CIR.0000160365.18879.1C. [DOI] [PubMed] [Google Scholar]
- Fichtlscherer S, Rosenberg G, Walter DH, Breuer S, Dimmeler S, Zehier AM. Elevated C Reactive Protein levels impaired endothelial vasoreactivity in patients with coronary artery disease. Circulation. 2000;102:1000–1006. doi: 10.1161/01.cir.102.9.1000. [DOI] [PubMed] [Google Scholar]
- Egashira K, Hirooka Y, Kai H, Sugimachi M, Suzuki S, Inou T, Takeshita A. Reduction in serum cholesterol with pravastatin improves endothelium-dependent vasomotion in patients with hypercholesterolemia. Circulation. 1994;89:2519–2524. doi: 10.1161/01.cir.89.6.2519. [DOI] [PubMed] [Google Scholar]
- Treasure CB, Klein JL, Weintraub WS, Talley JD, Stillabouer ME, Kosinski AS, Zhang J, Bocuzzi SJ, Cedarholm JC, Alexander RW. Beneficial effects of cholesterol-lowering therapy on the coronary endothelium in patients with coronary artery disease. N Engl J Med. 1995;332:481–487. doi: 10.1056/NEJM199502233320801. [DOI] [PubMed] [Google Scholar]
- Waters D. Cholesterol lowering. Should it continue to be the last thing we do. Circulation. 1999;99:3215–3217. doi: 10.1161/01.cir.99.25.3215. [DOI] [PubMed] [Google Scholar]
- Esper RJ. The role of lipid-lowering therapy in multiple risk factor management. Drugs. 1998;56:1–7. doi: 10.2165/00003495-199856001-00001. [DOI] [PubMed] [Google Scholar]
- Webb D, Vallance P, Eds . Endothelial function in hypertension. Berlin, Springer-Verlag; 1997. [Google Scholar]
- Murakami T, Mizuno S, Ohsato K, Moriuchi I, Arai Y, Nio Y, Kaku B, Takahashi Y, Ohnaka M. Effects of troglitazone on frequency of coronary vasoespastic-induced angina pectoris in patients with diabetes mellitus. Am J Cardiol. 1999;84:92–94. doi: 10.1016/S0002-9149(99)00199-X. [DOI] [PubMed] [Google Scholar]
- Horning B, Maier V, Drexler H. Physical training improves endothelial function in patients with chronic heart failure. Circulation. 1996;93:210–214. doi: 10.1161/01.cir.93.2.210. [DOI] [PubMed] [Google Scholar]
- Creager MA, Gallagher SJ, Girerd XJ, Coleman SM, Dzau VJ, Cooke JP. L-Arginine improves endothelium-dependent vasodilation in hypercholesterolemic humans. J Clin Invest. 1992;90:1248–1253. doi: 10.1172/JCI115987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drexler H, Zeiher AM, Meinertz T, Just H. Correcting endothelial dysfunction in coronary microcirculation of hypercholesterolemic patients by L-arginine. Lancet. 1991;338:1546–1550. doi: 10.1016/0140-6736(91)92372-9. [DOI] [PubMed] [Google Scholar]
- Clarkson D, Adams MR, Powe A, et al. Oral L-arginine improves endothelium-dependent dilation in hypercholesterolemic young adults. J Clin Invest. 1996;97:1989–1994. doi: 10.1172/JCI118632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griendling KK, FitzGerald GA. Oxidative stress and cardiovascular injury: Part I: Basic Mechanisms and in vivo monitoring of ROS. Circulation. 2003;108:1912–1916. doi: 10.1161/01.CIR.0000093660.86242.BB. [DOI] [PubMed] [Google Scholar]
- Ridker PM, Cushman M, Stampfer MJ, Tracy RP, Hennekens CH. Inflammation, aspirin and the risk of cardiovascular disease in apparently healthy men. N Engl J Med. 1997;336:973–979. doi: 10.1056/NEJM199704033361401. [DOI] [PubMed] [Google Scholar]
- Ridker PM. Inflammation, infection and cardiovascular risk. How good is the clinical evidence? Circulation. 1998;96:1671–1674. doi: 10.1161/01.cir.97.17.1671. [DOI] [PubMed] [Google Scholar]
- Levine GN, Frei B, Koulouris SN, Gerhard MD, Keaney JF, Vita JA. Ascorbic acid reverses endothelial vasomotor dysfunction in patients with coronary artery disease. Circulation. 1996;93:1107–1113. doi: 10.1161/01.cir.93.6.1107. [DOI] [PubMed] [Google Scholar]
- Gokce N, Keaney JF, Jr, Frei B, et al. Long-term ascorbic acid adminstration reverses endothelial vasomotor dysfunction in patients with coronary artery disease. Circulation. 1999;99:3234–3240. doi: 10.1161/01.cir.99.25.3234. [DOI] [PubMed] [Google Scholar]
- Solzbach U, Hornig B, Jeserich M, Hanjörg J. Vitamin C improves endothelial dysfunction of epicardial coronary arteries in hypertensive patients. Circulation. 1997;96:1513–1519. doi: 10.1161/01.cir.96.5.1513. [DOI] [PubMed] [Google Scholar]
- Plotnick GD, Corretti MC, Vogel RA. Effect of antioxidant vitamins on the transient impair- ment of endothelial-dependent brachial artery vasoactivity following a single high-fat meal. JAMA. 1997;278:1682–1686. doi: 10.1001/jama.278.20.1682. [DOI] [PubMed] [Google Scholar]
- Heitzer T, Just H, Munzel T. Antioxidant vitamin C improves endothelial dysfunction in chronic smokers. Circulation. 1996;94:6–9. doi: 10.1161/01.cir.94.1.6. [DOI] [PubMed] [Google Scholar]
- Esper RJ, Machado R, Vilariño J, Cacharrón JL, Ingino CA, García Guiñazú CA, Bereziuk E, Bolaño LA, Suarez DH. Endothelium-dependent response in hypercholesterolemic coronary artery disease patients under the effects of simvastatin and enalapril, either separately o combined. Am Heart J. 2000;140:684–689. doi: 10.1067/mhj.2000.109649. [DOI] [PubMed] [Google Scholar]
- Steinberg HO, Bayazeed B, Hook G, Johnson A, Cronin J, Baron AD. Endothelial dysfunction is associated with cholesterol levels in the high normal range in humans. Circulation. 1997;96:3287–3293. doi: 10.1161/01.cir.96.10.3287. [DOI] [PubMed] [Google Scholar]
- Hijmering ML, Stroes ES, Pasterkamp G. Variability of flow mediated dilation: consecuences for clinical application. Atherosclerosis. 2001;157:369–373. doi: 10.1016/S0021-9150(00)00748-6. [DOI] [PubMed] [Google Scholar]
- Kuvin JT, Patel AR, Karas RH. Need for standardization of noninvasive assessment of vascular endothelial function. Am Heart J. 2001;141:327–328. doi: 10.1067/mhj.2001.113221. [DOI] [PubMed] [Google Scholar]
- Pergola PE, Kellogg DL, Johson JM, Kosiba WA, Salomon DE. Role de sympathetic nerves in the vascular effects of local temperature in human foream skin. Am J Physiol. 1993;265:H785–H792. doi: 10.1152/ajpheart.1993.265.3.H785. [DOI] [PubMed] [Google Scholar]
- Rauch U, Osende JI, Chesebro JH, Badimon JJ. Statins and cardiovascular diseases: the multiple effects of lipid-lowering therapy by statins. Atherosclerosis. 2000;153:181–189. doi: 10.1016/S0021-9150(00)00397-X. [DOI] [PubMed] [Google Scholar]
- Anderson TJ, Uehata A, Gerhard MD, Meredith IT, Knab S, Delagrande D, Lieberman EH, Ganz P, Creager MA, Yeung AC, Selwyn AP. Close relation of endothelial function in the human coronary and peripheral circulation. J Am Coll Cardiol. 1995;26:1235–1241. doi: 10.1016/0735-1097(95)00327-4. [DOI] [PubMed] [Google Scholar]
- Sorensen KE, Kristensen IB, Celermajer DS. Atherosclerosis in the human brachial artery. J Am Coll Cardiol. 1997;29:318–322. doi: 10.1016/S0735-1097(96)00474-3. [DOI] [PubMed] [Google Scholar]
- Celermajer DS. Endothelial dysfunction: Does it matter? Is it reversible? J Am Coll Cardiol. 1997;30:325–333. doi: 10.1016/S0735-1097(97)00189-7. [DOI] [PubMed] [Google Scholar]
- Yataco R, Corretti M, Gardner AW, Womack CJ, Katzel LI. Endothelial reactivity an cardiac risk factors in older patients with peripheral arterial disease. Am J Cardiol. 1999;83:754–758. doi: 10.1016/S0002-9149(98)00984-9. [DOI] [PubMed] [Google Scholar]
- Perticone F, Maio R, Ceravolo R, Cosco C, Cloro C, Mattioli PL. Relationship between left ventricular mass and endothelium-dependent vasodilation in never treated hypertensive patients. Circulation. 1999;99:1991–1996. doi: 10.1161/01.cir.99.15.1991. [DOI] [PubMed] [Google Scholar]
- Schachinger V, Britten M, Zeiher AM. Prognostic impact of coronary vasodilator dysfunction on adverse long-term outcome of coronary heart disease. Circulation. 2000;101:1899–1906. doi: 10.1161/01.cir.101.16.1899. [DOI] [PubMed] [Google Scholar]
- Neunteufl T, Heher S, Katzenchlager R, Wolfl G. Late prognostic value of flow-mediated dilation in the brachial artery of patients with chest pain. Am J Cardiol. 2000;86:207–210. doi: 10.1016/S0002-9149(00)00857-2. [DOI] [PubMed] [Google Scholar]
- Al Suwaidi J, Hamasaki S, Higano ST, Nishimura RA, Holmes DR, Jr, Lerman A. Long-term follow-up of patients with mild coronary artery disease and endothelial dysfunction. Circulation. 2000;101:948–954. doi: 10.1161/01.cir.101.9.948. [DOI] [PubMed] [Google Scholar]
- Schroeder S, Enderle MD, Ossen R, Meisner C, Baumbach A, Pfohl M, Herdez C, Oberhoff M, Haering HU, Karsch KR. Noninvasive determination of the endothelium-mediated vasodilation as a screening test for coronary artery disease: pilot study to assess the predictive value in comparison with angina pectoris, exercise electrocardiography, and myocardial perfusion imaging. Am Heart J. 1999;138:731–739. doi: 10.1016/S0002-8703(99)70189-4. [DOI] [PubMed] [Google Scholar]
- Esper RJ, Vilariño JO, Cacharron JL, Machado R, Ingino CA, García Guiñazú CA, Bereziuk E, Bolaño AL, Suarez DH, Kura M. Impaired endothelial function in patients with rapidly stabilized unstable angina: assessment by noninvasive braquial artery ultrasonography. Clin Cardiol. 1999;22:699–703. doi: 10.1002/clc.4960221104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boulanger CM, Scoazec A, Ebrahimian T, Henry P, Mathieu E, Tedgui A, Mallat Z. Circulating microparticules from patients with myocardial infarction cause endothelial dysfunction. Circulation. 2001;104:2649–2652. doi: 10.1161/hc4701.100516. [DOI] [PubMed] [Google Scholar]
- Cohen JD, Drury JH, Ostdiek J, Finn J, Babu BR, Flaker G, Belew K, Donohue T, Labovitz A. Benefits of lipid-lowering on vascular reactivity in patients with coronary artery disease and average cholesterol levels: A mechanism for reducing clinical events. Am Heart J. 2000;139:734–738. doi: 10.1016/S0002-8703(00)90057-7. [DOI] [PubMed] [Google Scholar]
- Gokce N, Keaney JF, Menzolan JO, Watkins M, Kahn D, Hunter L, Duffy S, Vita JA. Impaired brachial artery endothelial function predicts perioperative cardiovascular events in patients undergoing vascular surgery. Circulation. 2001;104:II–328. (Abstract) [Google Scholar]
- Ganz P, Vita JA. Testing endothelial vasomotor function. Nitric oxide, a multipotent molecule. Circulation. 2003;108:2049–2053. doi: 10.1161/01.CIR.0000089507.19675.F9. [DOI] [PubMed] [Google Scholar]
- Verma S, Buchanan MR, Anderson TJ. Endothelial function testing as a biomarker of vascular disease. Circulation. 2003;108:2054–2059. doi: 10.1161/01.CIR.0000089191.72957.ED. [DOI] [PubMed] [Google Scholar]
- Willerson JT, Kereiakes DJ. Endothelial disfunction. Circulation. 2003;108:2060–2061. doi: 10.1161/01.CIR.0000099580.72044.83. [DOI] [PubMed] [Google Scholar]
- Benjamin EJ, Larson MG, Kupka MJ, Mitchell GF, Keaney JF, Vasan RS, Lehman B, Fan S, Osypiuk E, Vita JA. Cross-sectional correlates of brachial artery endothelial function in the community: The NHBL'S Framingham Heart Study. Circulation. 2001;104:II–152. (Abstract) [Google Scholar]
- Glagov S, Weisenberg E, Zarins CK, Stankunavicius R, Kolettis GJ. Compensatory enlargement of human atherosclerotic coronary arteries. N Engl J Med. 1987;316:1371–1375. doi: 10.1056/NEJM198705283162204. [DOI] [PubMed] [Google Scholar]
- Stehouwer CDA. Is measurement of endothelial dysfunction clinically useful? Eur J Clin Invest. 1999;29:459–61. doi: 10.1046/j.1365-2362.1999.00497.x. [DOI] [PubMed] [Google Scholar]
- Sica D. Endothelial cell function. New considerations. Eur Heart J. 2000;21:B13–B21. [Google Scholar]
- Pederson SD, Pederson EM, Ringgard S, Falk E. Coronary artery disease: plaque vulnerability, disruption, and trombosis. In: Fuster V, editor. The vulnerable aterosclerotic plaque. New York, Futura publishing Co; 1999. pp. 1–24. [Google Scholar]
- Paredos P, Kek A. Relation of blunted dilation of the brachial artery in insulin-dependent diabetes mellitus to microalbuminuria. Am J Cardiol. 2000;86:364–367. doi: 10.1016/S0002-9149(00)00938-3. [DOI] [PubMed] [Google Scholar]