Abstract
Objective
To report findings regarding health-related quality-of-life outcomes by treatment arm, both overall and within subgroups defined by selected baseline characteristics, among patients who participated in the Submacular Surgery Trials (SST) randomized trial of observation vs surgical removal of subfoveal choroidal neovascular lesions that were either idiopathic or associated with ocular histoplasmosis (SST Group H Trial).
Design and Methods
Eligible patients were 18 years or older and had subfoveal choroidal neovascularization (including a classic component on fluorescein angiography) and visual acuity of 20/50 to 20/800 inclusive in the eye randomly assigned to surgery or observation. Trained interviewers, who were masked to treatment assignment, administered the National Eye Institute Visual Function Questionnaire(NEI-VFQ), the 36-Item Short-Form Health Survey (SF-36), and the Hospital Anxiety and Depression Scale (HADS) by telephone before enrollment and at 6, 12, and 24 months after enrollment; early enrollees also had interviews at 36 and 48 months. Baseline clinical examinations provided data regarding visual acuity, other aspects of vision, and presence of unilateral or bilateral choroidal neovascularization.
Results
Of 225 patients interviewed at baseline and enrolled, 201, 190, and 161 patients (respectively, 89%, 85%, and 88% of those eligible to be interviewed) were interviewed 12, 24, and 36 months later. The NEI-VFQ scores in both treatment arms improved from baseline (median score, 75) to the 24-month interview. Patients in the surgery arm had 4-point larger improvements, on average, than patients in the observation arm (95% confidence interval, 1–8 points). The largest differences between treatment arms for mean 24-month improvements from baseline were in the role difficulties subscale (9 points) and dependency subscale(8 points), with larger mean improvements in the surgery arm. Scores on the SF-36 worsened by approximately 1 point on the physical component summary and improved by 2 points or more on the mental component summary in both treatment arms by 24 months incomparison to baseline. The percentage of HADS-defined “definite cases” of both anxiety and depression declined from baseline in each treatment arm.
Conclusions
Vision-targeted quality of life improved more after submacular surgery than with observation, supporting a possible small overall benefit of surgery suggested by the ophthalmic outcomes reported elsewhere. Ophthalmologists and patients similar to those who participated in the SST Group H Trial should be aware of the effects of submacular surgery on specific aspects of vision-targeted quality of life as well as on ophthalmic outcomes when considering this treatment approach.
Measurement of health-related quality of life has been advocated as an adjunct to clinical measurements in randomized treatment trials and other study designs in which patient outcomes are assessed.1–3 When the condition for which treatment is being evaluated is non–life threatening and when differences between treatments with respect to clinical outcomes may be small, assessment of patients’ perceptions of treatment benefit or harm is particularly important.4 However, patient perceptions of treatment effects may be informative whether similar to or different from clinical outcomes.
Since 1997, the Submacular Surgery Trials (SST) Research Group has been conducting randomized trials of surgical removal of subfoveal choroidal neovascular lesions to evaluate the role of this procedure in the treatment of patients with age-related macular degeneration and other ophthalmic conditions. The first trial to be initiated was for patients who had subfoveal choroidal neovascularization that was either idiopathic or associated with the ocular histoplasmosis syndrome (SST Group H Trial). Data from uncontrolled case series and from a small pilot study suggested that such patients were those most likely to benefit from submacular surgery. Because neovascular lesions of ocular histoplasmosis typically are diagnosed when patients are in midlife, these patients are at risk of loss of employment in their usual occupations. Thus, many aspects of their lives may be affected by their vision problems and by treatments that may be administered.
Measurement of both vision-targeted and general health-related quality of life was included in the design of the SST. These measures were considered important because of the cost of surgery, inconvenience to patients treated surgically who were instructed to maintain a face-down position for 2 days (or longer) after surgery, and risk of postoperative complications that were expected to require further treatment. Furthermore, review of findings from randomized trials of other treatments for subfoveal choroidal neovascularization secondary to age-related macular degeneration5 suggested that differences between active interventions and observation with respect to visual acuity may be small. Thus, it was plausible that surgically treated and observed patients would differ more on health-related quality-of-life scales, particularly vision-targeted scales, than on visual acuity measurements and other clinical outcomes.
The SST Research Group has published data regarding health-related quality of life at the time patients enrolled in the SST Group H Trial.6 Scores on a standard vision-targeted instrument were lower than expected based on the visual acuity of the better eye when compared with published scores from patients who had other ophthalmologic conditions. In particular, patients who had bilateral choroidal neovascularization reported substantial loss of visual function on some scales even after accounting for differences in visual acuity of the better eye and other factors.6 Visual acuity and related findings from the SST Group H Trial have been reported elsewhere.7 For the full group of patients enrolled, visual acuity changes over time suggested a small benefit to surgery, but differences between the surgery and observation arms were largest during the first year after enrollment and declined over time. The prespecified subgroup of patients who had best-corrected visual acuity of the study eye worse than 20/100 (20/125-20/800) at baseline had a large benefit from surgery, based on stabilization or improvement of visual acuity, but patients who had a better baseline visual acuity (20/50-20/100) had no benefit.
The purpose of this report is to summarize health-related quality-of-life findings from the interviews conducted during the follow-up period with patients who participated in the SST Group H Trial and to compare these findings between patients in the randomly assigned treatment arms. Because of the marked difference in baseline scores observed between patients with unilateral and bilateral choroidal neovascular lesions6 and because of the difference in visual acuity outcomes by baseline visual acuity,7 comparisons of findings by treatment arm within these subgroups of patients are of particular interest.
METHODS
The members of an independent Data and Safety Monitoring Committee approved the design and methods proposed for the SST before enrollment of patients was initiated in the SST Group H Trial in April 1997. Institutional review boards at all participating institutions reviewed and approved the study design and the consent forms to be used locally. All patients gave signed consent before enrollment and random treatment assignment.
Many of the methods used in the SST and in the SST Group H Trial have been published.6,7 The SST Manual of Procedures8 and the SST Forms Book9 are available and provide detailed information regarding study design, methods, and policies.
PATIENT ELIGIBILITY AND ENROLLMENT
The details of eligibility requirements for patients and eyes, clinical evaluation procedures, and clinical data collection methods and schedules are given elsewhere.7 Eligible patients were 18 years or older and had new or recurrent subfoveal choroidal neovascular lesions with evidence of classic choroidal neovascularization on fluorescein angiography and best-corrected visual acuity of 20/50 to 20/800, inclusive, in at least 1 eye (study eye). The visual acuity of the fellow (nonstudy) eye had to be light perception or better. Patients had to agree to complete telephone interviews administered prior to enrollment and at scheduled times after enrollment to be eligible for the randomized trial. Enrollment and random treatment assignment did not take place until the baseline interview was completed by telephone with an interviewer located at the SST Coordinating Center, Baltimore, Md.
Patients judged to be eligible by the examining ophthalmologist were invited to participate in the study. The consent and enrollment process are described elsewhere.6,7 Randomization was stratified by enrolling clinical center only. The clinical center staff were notified of the assigned treatment for the study eye, surgery or observation, by means of an automated message sent by telecopier on the day of enrollment. This notice was accompanied by a schedule of the target dates for future interviews and examinations and the permissible intervals during which each one could be completed for the data to be accepted into the study database. The patient specified on the telecopied notice whether future interviews should be initiated by the interviewers or would be initiated by the patient. Whenever the interviewers were asked to initiate the interview contact, the patient provided telephone information, preferred days of the week, and times of day for future interviews before the notice was telecopied back to the coordinating center.
INTERVIEW METHODS
The interview instruments selected for the SST were the National Eye Institute Visual Function Questionnaire (NEI-VFQ),10–12 the 36-Item Short-Form Health Survey (SF-36),13 the Hospital Anxiety and Depression Scale (HADS, 14 items),14 and the 3-item SST Vision Preference Value Scale.15 The NEI-VFQ was designed to be applicable to patients with a number of different vision-limiting or vision-threatening conditions. Initially, the 25-item NEI-VFQ with selected supplemental questions added was used for the interviews. By January 1998, all 14 supplemental questions provided by the developers12 had been incorporated into the interview. The items are scored to yield an overall score and scores on 11 subscales that address different aspects of visual function and a general health subscale. Overall scores on the NEI-VFQ and subscale scores have a range of 0 (worst score) to 100 (best score). The SF-36 measures general health status and health-related functioning. The SF-36 items are combined to create 8 subscales that can be combined further to create 2 summary scales.16 The summary scale developers calibrated scores on the physical and mental component summary scales to have means of 50 points and standard deviations of 10 points in the general US population.16 Higher scores on SF-36 summary scales indicate better perceived health status. The HADS instrument was included in the interview because of both anecdotal and published evidence of depression17 among patients with poor vision and anxiety regarding future vision reported by patients who participated in a pilot study conducted by the SST investigators. The HADS has been used with outpatients with many medical conditions.18 Scores on the 2 HADS scales (anxiety and depression, 7 items each) range from 0 (best score) to 21 (worst score). The SST Vision Preference Value Scale was incorporated into the interview to permit assessment of the value patients placed on current vision and health relative to perfect vision or complete blindness. Baseline findings from the SST Vision Preference Value Scale have been reported elsewhere.15
All interviews were conducted from the Coordinating Center by trained interviewers who were masked to the study eye, the clinical trial, and the treatment arm. However, patients occasionally revealed pertinent information during the course of the interviews despite instructions not to do so. Interviewers used a computer-assisted system that interacted with the primary study database to confirm study identifiers assigned to the patient so that the interview data could be integrated with the clinical data for each patient. Before each interview was concluded, scores for the HADS depression subscale were calculated automatically from the responses given by the patient. Whenever a patient had a score that would result in classification as a “definite case” of depression, based on the recommendations of the HADS developers,14 the interviewer requested the patient’s permission to notify the SST ophthalmologist of the score so that an appropriate referral could be considered.
All patients were scheduled for follow-up interviews at 6, 12, and 24 months after enrollment. Patients who enrolled by September 30, 2000, were scheduled for a 36-month interview and those who enrolled by September 30, 1999, also were scheduled for a 48-month interview. The last study interviews for all patients who had not completed a 48-month interview already were conducted during the final year of patient follow-up that began on October 1, 2002.
BASELINE CLINICAL DATA COLLECTION
Clinical data collection at baseline and during follow-up is described elsewhere.7 Baseline data collection included measurement of the best-corrected visual acuity, reading speed, and contrast threshold of each eye according to standard protocols. Baseline stereoscopic color photographs of the macula and optic disc of each eye and a film-based fluorescein angiogram were taken and forwarded to the SST Photograph Reading Center, Baltimore, Md, for interpretation and documentation of characteristics of the eyes and subfoveal lesions.
Best-corrected visual acuity measurements made before enrollment were used to subgroup patients by the visual acuity of the study eye (20/50-20/100 vs 20/125-20/800), a grouping of interest specified during trial planning. Patients were classified as having bilateral or unilateral neovascular disease primarily on the basis of masked central interpretations of baseline photographs.6
QUALITY ASSURANCE
An initial training session for interviewers was conducted by the chair of the SST Patient-Centered Outcomes Subcommittee before the trial began; additional training was provided during interviewer workshops held at least once each year. The goal of training was to standardize administration of the interview and interactions with trial patients. The interviewers met periodically during the first years of the trial to reach agreement on issues of interview administration and interactions with patients and to recommend improvements in the computer-assisted system. The SST Patient-Centered Outcomes Committee met twice yearly to review data regarding interview administration, to review preliminary data (combined across treatment arms), and to make recommendations regarding methodology.
STATISTICAL DESIGN AND STUDY MONITORING
In the SST Group H Trial, visual acuity was the primary clinical measurement of interest for comparison of the treatment arms. In particular, change in best-corrected visual acuity from baseline to the 24-month examination was the basis for calculating the target sample size.7 In the original study design of the parallel evaluation of quality of life, which was developed before the NEI-VFQ was published, 24-month change in scores on the SF-36 interview was designated the most important secondary outcome for assessing the effect of surgery. Subsequently, the NEI-VFQ interview was given greater weight based on experience with the interview instruments and the perception that it had greater relevance to the problems experienced by study patients and would be more responsive to changes in vision over time. The target sample size of 240 patients was calculated on the basis of the primary clinical outcome of interest to provide 218 patients with visual acuity measured at the 24-month examination.7,8 It was estimated that this sample size would permit detection of 4-point differences between treatment arms in the physical component summary and mental component summary scales of the SF-36.16 Information provided by the developers of the NEI-VFQ indicated that this sample size would permit detection of 10-point or greater differences in scores between treatment arms for most NEI-VFQ scales.12 However, our purpose, and the focus of analyses, is description of findings from the quality-of-life interviews rather than hypothesis testing.
Responsibility for monitoring accumulating data regarding safety and effectiveness of surgery was entrusted to the Data and Safety Monitoring Committee appointed in 1996. However, monitoring focused on clinical outcomes rather than on quality-of-life outcomes. Key recommendations are summarized elsewhere.7
Owing to funding constraints and other considerations,7 accrual halted on September 30, 2001, and scheduled patient follow-up ended on September 30, 2003. Thus, all patients were eligible for interviews 24 months after enrollment, but only patients who enrolled by September 2000 were eligible for 36-month interviews and only those who enrolled by September 1999 were eligible for 48-month interviews.
DATA ANALYSIS AND STATISTICAL METHODS
The NEI-VFQ, SF-36, and HADS scores were calculated using the recommendations of the developers, except that a scale score was not calculated in the few instances in which half the items that make up a scale had not been answered. For the NEI-VFQ driving scale, patients who responded that they had stopped driving because of vision (26 patients at baseline; 30 patients at the 24-month interview) were given a score of 0. The HADS developers proposed scores to classify patients for anxiety and depression: 0 to 7, noncases; 8 to 10, doubtful (unlikely) cases; 11 or greater, definite cases.
Because most patients who participated in this clinical trial had baseline NEI-VFQ scores at or near the ceiling on the color vision, peripheral vision, and ocular pain subscales6 and because the subfoveal neovascular lesions in study eyes were relatively small, the visual acuity of the study eyes was relatively good and the visual acuity of the fellow eyes of most patients was excellent at baseline,7 these subscales were judged to be of less importance than others for comparison of treatment arms during follow-up. Furthermore, these are 1- or 2-item scales that yield scores that have categorical distributions. They have been retained in calculations of overall NEI-VFQ scores but have not been presented separately in this analysis of follow-up findings.
Distributions of baseline interview scores were summarized for display purposes using exploratory data analysis methods.19 Because of the highly skewed nature of the distributions of scores on most NEI-VFQ scales, medians and interquartile ranges or 95% confidence intervals (CIs) on the medians have been used to describe the distributions. For the same reason, distributions of scores were compared between treatment arms using the Wilcoxon rank sum test.20 Although not all patients completed all scheduled interviews, median scores from cross-sectional distributions at each interview time have been connected by lines for display purposes. Changes in scores from baseline to follow-up interviews, which tended to be distributed normally or approximately so, were compared using t tests for means; mean changes in scores also have been connected by lines for display purposes. For comparisons of findings between treatment arms for the full group of patients, P ≤ .01 was deemed “statistically significant.” Otherwise, P values were not adjusted for multiple comparisons. Within subgroups of patients defined by baseline ophthalmic status, P ≤.10 also has been noted.
Continuous-time mixed linear marginal models were used to adjust for potentially important covariates to estimate the overall effect of surgery on NEI-VFQ scores. In addition to treatment arm and interview times, baseline scores and health status, as measured by the SF-36 physical component summary scale, were included as covariates.
Data from all interviews completed by September 30, 2003, were analyzed. Data from each patient were analyzed with the treatment arm to which he or she was assigned randomly at the time of enrollment (“intent-to-treat” analysis approach). All data displays use responses from completed interviews only; no attempt was made to impute scores for missed interviews. Data analysis was performed using SAS software (SAS Inc, Cary, NC) and custom-written programs.
RESULTS
When accrual ended on September 30, 2001, two hundred twenty-five patients had enrolled; 112 patients had been assigned to the surgery arm and 113 patients to the observation arm. Ocular histoplasmosis was judged by personnel at the SST Photograph Reading Center to be the underlying cause of the choroidal neovascular lesion for 192 (85%) of the enrollees. Based on central review of baseline photographs and clinical data, 58 patients (30 [27%] in the observation arm and 28 [25%] in the surgery arm) had neovascular lesions in both eyes and were classified for analysis purposes as bilateral cases. Examples of neo-vascular lesions observed in fellow eyes have been published elsewhere.6 All other patients were classified as unilateral cases. No consideration was given to other causes of vision loss in fellow eyes when classifying patients as unilateral or bilateral cases. Except for 1 patient who refused surgery after enrollment, all patients had the assigned treatment, that is, submacular surgery or observation (no treatment).
CHARACTERISTICS OF PATIENTS AT STUDY ENROLLMENT BY STATUS OF EYES
Sociodemographic characteristics and visual status of patients at baseline are summarized in Table 1 and Table 2 by neovascular status within treatment arms. The treatment arms were well balanced, both overall and within subgroups of unilateral and bilateral patients, for all characteristics examined. Except for 5 study eyes in the observation arm, all study eyes were phakic at baseline. Patients with bilateral choroidal neovascularization were older, less often employed, and more often hypertensive. As expected, the fellow eye was the better-seeing eye among unilateral cases; among bilateral cases, the better-seeing eye was the study eye in almost half the patients (Table 2). Most bilateral cases had deficits in visual acuity, contrast threshold, and reading speed in both eyes, as expected.
Table 1.
Sociodemographic and Health Characteristics of Patients at the Time of Enrollment by Treatment Arm and Choroidal Neovascularization (CNV) Status, SST Group H Trial
|
No. (%) of Patients by Treatment Arm and CNV Status of Eyes |
||||
|---|---|---|---|---|
|
Observation Arm |
Surgery Arm |
|||
| Characteristic | Unilateral Cases (n = 83) | Bilateral Cases (n = 30) | Unilateral Cases (n = 84) | Bilateral Cases (n = 28) |
| Age, y | ||||
| <30 | 9 (11) | 1 (3) | 11 (13) | 0 |
| 30–39 | 16 (19) | 4 (13) | 18 (21) | 4 (14) |
| 40–49 | 28 (34) | 4 (13) | 22 (26) | 3 (11) |
| 50–59 | 15 (18) | 7 (23) | 17 (20) | 9 (32) |
| 60–69 | 10 (12) | 7 (23) | 10 (12) | 8 (29) |
| ≥ 70 | 5 (6) | 7 (23) | 6 (7) | 4 (14) |
| Gender | ||||
| Women | 44 (53) | 13 (43) | 51 (61) | 16 (57) |
| Men | 39 (47) | 17 (57) | 33 (39) | 12 (43) |
| Race/ethnicity* | ||||
| White, not Hispanic | 79 (95) | 30 (100) | 81 (96) | 27 (96) |
| Other | 4 (5) | 0 | 3 (4) | 1 (4) |
| Occupational status | ||||
| Employed | 62 (75) | 12 (40) | 61 (73) | 12 (43) |
| Retired | 12 (14) | 12 (40) | 11 (13) | 10 (36) |
| Housespouse or student | 4 (5) | 3 (10) | 6 (7) | 0 |
| Unemployed or disabled | 5 (6) | 3 (10) | 6 (7) | 6 (21) |
| Smoking history | ||||
| Never smoked | 28 (34) | 8 (27) | 35 (42) | 7 (25) |
| Former smoker | 20 (24) | 9 (30) | 22 (26) | 11 (39) |
| Current smoker | 35 (42) | 13 (43) | 27 (32) | 10 (36) |
| Coexisting conditons | ||||
| Hypertension† | 15 (18) | 15 (50) | 17 (20) | 11 (39) |
| Diabetes mellitus* | 4 (5) | 1 (3) | 4 (5) | 3 (11) |
Abbreviation: SST, Submacular Surgery Trials.
Noted as a self-report by the patient.
Based on diagnosis by a physician, use of antihypertensive medications, or a systolic blood pressure reading of 140 mm Hg or higher or a diastolic blood pressure reading of 90 mm Hg or higher.
Table 2.
Visual Status of Patients at the Time of Enrollment by Treatment Arm and Choroidal Neovascularization (CNV) Status, SST Group H Trial
|
No. (%) of Patients by Treatment Arm and CNV Status of Eyes |
||||
|---|---|---|---|---|
|
Observation |
Surgery |
|||
| Characteristic or Measurement | Unilateral Cases (n = 83) | Bilateral Cases (n = 30) | Unilateral Cases (n = 84) | Bilateral Cases (n = 28) |
| Better-seeing eye* | ||||
| Study eye | 0 | 15 (50) | 0 | 12 (43) |
| Fellow eye | 83 (100) | 15 (50) | 84 (100) | 16 (57) |
| VA, Snellen equivalent | ||||
| ≥20/20 | 73 (88) | 6 (20) | 76 (90) | 9 (32) |
| 20/25-20/40 | 9 (11) | 3 (10) | 8 (10) | 3 (11) |
| 20/50-20/80 | 1 (1) | 10 (33) | 0 | 6 (21) |
| ≤20/100 | 0 | 11 (37) | 0 | 10 (36) |
| Contrast threshold, % contrast required | ||||
| ≤1.6 | 19 (23) | 1 (3) | 11 (13) | 0 |
| >1.6–3.2 | 58 (70) | 18 (60) | 69 (82) | 18 (64) |
| >3.2–4.5 | 6 (7) | 7 (23) | 4 (5) | 5 (18) |
| >4.5 | 0 | 4 (13) | 0 | 5 (18) |
| Reading speed with enlarged text, wpm† | ||||
| ≥160 | 18 (22) | 1 (3) | 20 (24) | 0 |
| 120–159 | 31 (39) | 2 (7) | 32 (39) | 6 (22) |
| 80–119 | 22 (28) | 15 (52) | 21 (26) | 13 (48) |
| 40–79 | 8 (10) | 8 (28) | 8 (10) | 5 (19) |
| <40 | 1 (1) | 3 (10) | 1 (1) | 3 (11) |
| Worse-seeing eye‡ VA, Snellen equivalent | ||||
| 20/50-20/80 | 39 (47) | 7 (23) | 38 (45) | 7 (25) |
| 20/100-20/160 | 21 (25) | 5 (17) | 25 (30) | 5 (18) |
| 20/200-20/320 | 17 (20) | 10 (33) | 15 (18) | 6 (21) |
| ≤20/400 | 6 (7) | 8 (27) | 6 (7) | 10 (36) |
| Contrast threshold, % contrast required | ||||
| >1.6–3.2 | 29 (35) | 6 (20) | 29 (35) | 11 (39) |
| >3.2–4.5 | 25 (30) | 9 (30) | 23 (27) | 3 (11) |
| >4.5–8.9 | 15 (18) | 8 (27) | 19 (23) | 7 (25) |
| >8.9–17.8 | 8 (10) | 4 (13) | 8 (10) | 3 (11) |
| >17.8 | 6 (7) | 3 (10) | 5 (6) | 4 (14) |
| Reading speed with enlarged text, wpm‡ | ||||
| ≥120 | 8 (10) | 0 | 11 (13) | 0 |
| 80–119 | 15 (19) | 3 (10) | 18 (22) | 5 (19) |
| 40–79 | 34 (42) | 13 (45) | 29 (35) | 7 (26) |
| 0–39 | 23 (29) | 13 (45) | 24 (29) | 15 (56) |
Abbreviations: SST, Submacular Surgery Trials; VA, visual acuity; wpm, words per minute.
Better-seeing eye defined as the eye with better visual acuity at baseline, or study eye when equal, for all measurements.
Reading speed not measured at baseline for 7 patients. Percentages based on numbers with measurements.
Worse-seeing eye defined as the eye with the worse visual acuity at baseline, or the fellow eye when equal, for all measurements.
Distributions of baseline scores from the NEI-VFQ are shown in Figure 1 overall (top, left) and for each scale separately and by treatment arm for all patients and for unilateral and bilateral cases separately. Baseline scores on most NEI-VFQ scales differed between patients with unilateral and bilateral choroidal neovascularization. However, treatment arms were well balanced on interview scores at baseline among all patients and within subgroups of bilateral and unilateral cases. None of the comparisons of baseline scores suggested any clinically or statistically meaningful difference between treatment arms (P>.05, Wilcoxon rank sum tests).
Figure 1.
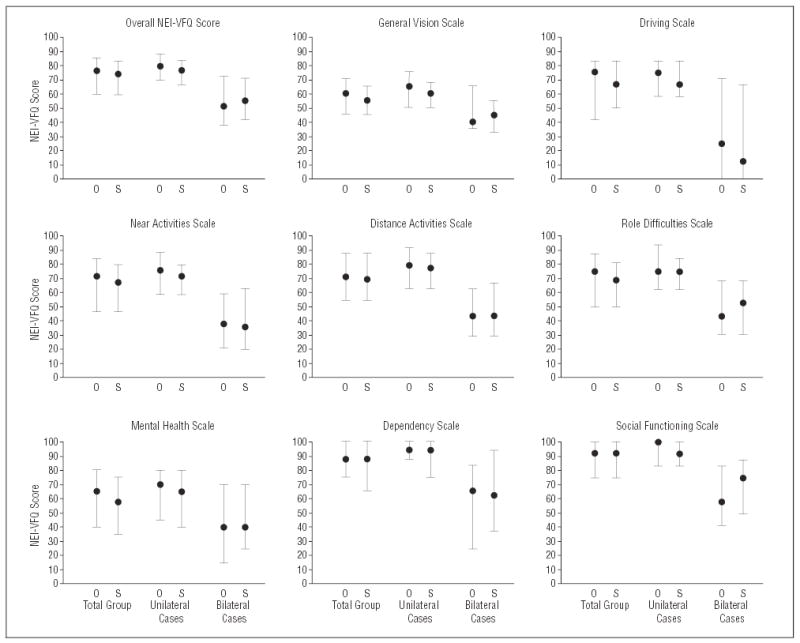
Summaries of distributions of National Eye Institute Visual Function Questionnaire (NEI-VFQ) scores by scale and treatment arm at baseline. The leftmost pair of bars for each scale is for all patients in each treatment arm; the middle pair of vertical bars is for patients in each treatment arm who were classified as unilateral cases; the rightmost pair of bars is for patients in each treatment arm who were classified as bilateral cases. Each vertical bar spans the interquartile range of the distribution. The black dot indicates the median of the distribution. Best possible score on each scale is 100 points. The observation arm (O) was composed of 113 patients (83 unilateral cases, 30 bilateral cases); the surgery arm (S), 112 patients (84 unilateral cases, 28 bilateral cases).
COMPLETION OF FOLLOW-UP INTERVIEWS
A total of 989 follow-up interviews were expected based on vital status and date of enrollment. Of 493 follow-up interviews expected for patients in the observation arm, 421 (85%) were completed. Patients in the surgery arm completed 443 (89%) of 496 interviews expected. Bilateral cases had somewhat better interview completion rates (91% overall) than unilateral cases (86% overall) in both treatment arms. Of 18 patients (16%) in the observation arm and 15 patients (13%) in the surgery arm who missed the 24-month interview, 9 patients in each arm completed either a 36-month or 48-month interview.
At baseline, 106 (94%) of 113 patients in the observation arm and 110 (98%) of 112 patients in the surgery arm answered all of the questions. For the 24-month interviews, 92 (99%) of 93 patients in the observation arm and 96 (99%) of 97 patients in the surgery arm answered all of the questions.
Baseline interviews were conducted by 9 different interviewers; 3 interviewers conducted 176 (78%) of the baseline interviews with the 225 enrollees. Two interviewers administered 713 (83%) of the 864 follow-up interviews.
VISION-TARGETED QUALITY OF LIFE AND CHANGES FROM BASELINE DURING PATIENT FOLLOW-UP
Median overall NEI-VFQ scores increased in both treatment arms from the baseline interview to the 6-month interview, from 77 to 81 among patients in the observation arm, and from 74 to 81 among patients in the surgery arm (Figure 2). At the 24-month interview, median overall scores were 85 (95% CI, 80–88) and 86 (95% CI, 83–87) in the observation and surgery arms, respectively. Distributions of scores in the 2 treatment arms were similar at all interview times.
Figure 2.

Median overall and subscale scores from the National Eye Institute Visual Function Questionnaire (NEI-VFQ) at each scheduled interview time by treatment arm. Best possible score on each subscale is 100 points. Solid lines indicate patients in the observation arm (n=113 at baseline); broken lines, patients in the surgery arm (n=112 at baseline).
At the 24-month interview, only 25 patients (15 [16%] in the observation arm and 10 [10%] in the surgery arm) had NEI-VFQ overall scores that were 5 points or more worse than at baseline. Of the other 166 patients, 13 (14%) in the observation arm and 26 (27%) in the surgery arm had 24-month scores that were 15 points or more better than at baseline. Mean changes in scores from baseline, differences in changes between treatment arms, and 95% CIs are summarized in Table 3. Mean changes were positive (mean scores improved from baseline) in both treatment arms at all times with only 2 exceptions (dependency subscale in the observation arm at 12 and 36 months). The mean difference in 24-month changes in overall scores was 4 (95% CI, 1–8), favoring surgery (P=.01, t test). For individual subscales, the differences in mean 24-month changes ranged from 3 (social functioning) to 9 (role difficulties). The mean difference in changes for both the role difficulties subscale and the dependency subscale met our criterion for statistical significance.
Table 3.
Differences Between Treatment Arms in Changes in NEI-VFQ Scores From Baseline to Follow-Up Interviews, SST Group H Trial*
|
Mean Change†, Difference‡, and 95% CI by Time After Enrollment by Treatment Arm |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
6 mo |
12 mo |
24 mo |
36 mo |
|||||||||
|
Mean Change |
Mean Change |
Mean Change |
Mean Change |
|||||||||
| Variable | Observation | Surgery | Difference (95% CI) | Observation | Surgery | Difference (95% CI) | Observation | Surgery | Difference (95% CI) | Observation | Surgery | Difference (95% CI) |
| NEI-VFQ Scale | ||||||||||||
| Overall | 3.2 | 5.3 | 2.1 (−0.6 to 4.7) | 2.1 | 6.5 | 4.4 (1.4 to 7.5)§ | 4.1 | 8.3 | 4.2 (0.9 to 7.6)§ | 3.0 | 6.0 | 3.0 (−1.2 to 7.2) |
| General vision | 2.4 | 4.4 | 1.9 (−2.0 to 5.9) | 1.7 | 7.7 | 6.0 (1.3 to 10.7)§ | 3.1 | 7.4 | 4.3 (−0.5 to 9.2) | 1.5 | 6.7 | 5.2 (−0.3 to 10.7) |
| Driving | 4.6 | 2.2 | −2.4 (−8.2 to 3.5) | 2.6 | 4.2 | 1.6 (−4.8 to 7.9) | 3.0 | 7.3 | 4.3 (−2.3 to 11.0) | 3.2 | 4.6 | 1.4 (−6.6 to 9.4) |
| Near activities | 3.6 | 7.8 | 4.1 (−0.4 to 8.7) | 3.5 | 9.6 | 6.2 (1.6 to 10.7)§ | 5.3 | 10.9 | 5.6 (0.8 to 10.4) | 5.3 | 7.8 | 2.5 (−3.5 to 8.4) |
| Distance activities | 4.3 | 6.4 | 2.1 (−1.8 to 5.9) | 2.5 | 8.0 | 5.4 (1.4 to 9.4)§ | 5.8 | 11.3 | 5.5 (1.0 to 10.0) | 3.5 | 8.5 | 5.0 (−0.3 to 10.4) |
| Role difficulties | 3.2 | 6.9 | 3.7 (−1.1 to 8.6) | 4.4 | 10.5 | 6.1 (0.4 to 11.7) | 5.1 | 13.7 | 8.7 (3.0 to 14.3)§ | 4.2 | 8.7 | 4.6 (−1.8 to 10.9) |
| Mental health | 7.6 | 9.1 | 1.5 (−3.8 to 6.8) | 7.5 | 13.0 | 5.5 (−0.1 to 11.1) | 11.2 | 17.2 | 6.0 (−0.3 to 12.3) | 8.5 | 13.8 | 5.3 (−1.8 to 12.5) |
| Dependency | 1.8 | 5.9 | 4.1 (−0.6 to 8.8) | −1.7 | 6.4 | 8.1 (2.6 to 13.6)§ | 2.4 | 10.1 | 7.7 (2.1 to 13.2)§ | −2.3 | 3.8 | 6.1 (−0.5 to 12.8) |
| Social functioning | 2.7 | 7.2 | 4.5 (1.3 to 7.8) | 1.4 | 5.2 | 3.8 (0.1 to 7.6) | 2.9 | 5.5 | 2.6 (−1.6 to 6.8) | 2.1 | 5.7 | 3.6 (−1.0 to 8.1) |
| Interviews | ||||||||||||
| Expected | 113 | 112 | NA | 113 | 112 | NA | 111 | 112 | NA | 97 | 97 | NA |
| No. (%)completed | 98 (87) | 104 (93) | NA | 99 (88) | 102 (91) | NA | 94 (85) | 97 (87) | NA | 78 (80) | 83 (86) | NA |
Abbreviations: CI, confidence interval; NA, not applicable; NEI-VFQ, National Eye Institute Visual Function Questionnaire; SST, Submacular Surgery Trials.
Boldfaced values indicate primary outcome time.
Positive change indicates improvement from baseline.
Difference equals mean change in surgery arm minus mean change in observation arm. Positive differences indicate more improvement in scores in surgery arm.
P≤.01, t test.
Adjustment for interview times, baseline score, and SF-36 physical component summary score at each interview using mixed linear models provided marginal estimates of the overall difference between treatment arms in changes in NEI-VFQ scores from baseline. The estimated change from baseline in the overall score was 3.5 points larger in the surgery arm than in the observation arm (P=.005). Estimated differences favored surgery over observation (P<.01) for 5 of the 8 subscales of interest in this analysis: near activities (4.9 points), distance activities (4.3 points), role difficulties (5.0 points), dependency (6.8 points), and social functioning (3.6 points).
CHANGE IN VISION-TARGETED QUALITY OF LIFE IN PATIENT SUBGROUPS
Distributions of NEI-VFQ overall scores and changes in scores from baseline also were compared between treatment arms within the subgroups of 167 unilateral cases and 58 bilateral cases. Median overall scores are displayed in Figure 3. As indicated by separation of the 95% CIs on the median overall scores from the 24-month interviews, median scores for unilateral cases remained at levels substantially better than those of bilateral cases in both treatment arms during follow-up. Median scores on follow-up interviews were slightly higher than on baseline interviews among unilateral cases in both treatment arms. Among bilateral cases, median overall scores in the surgery arm were somewhat higher than in the observation arm through the 24-month interview, with median overall NEI-VFQ scores from the 24-month interview of 69 and 51 for the surgery and observation arms, respectively (P=.08, Wilcoxon rank sum test). For unilateral and bilateral cases, the median scores for most subscales (data not shown) followed the same pattern as the median overall scores displayed in Figure 3, with separation of scores of unilateral and bilateral cases, higher (better) median scores during follow-up than at baseline, and more variability over time among scores for bilateral cases.
Figure 3.

Median overall National Eye Institute Visual Function Questionnaire scores at each interview time for unilateral cases and bilateral cases, as defined by the presence of choroidal neovascularization in the nonstudy (fellow) eye at baseline. Vertical lines at 24 months delimit the 95% confidence intervals of median scores. Best possible score is 100 points. Solid lines indicate patients in the observation arm (83 unilateral cases, 30 bilateral cases); broken lines, patients in the surgery arm (84 unilateral cases, 28 bilateral cases).
Changes in overall NEI-VFQ scores from baseline to follow-up interviews are shown in Figure 4. Among unilateral cases (Figure 4A), median scores in both treatment arms increased from baseline, by 3 to 5 points in the observation arm and by 5 to 8 points in the surgery arm. Mean changes among unilateral cases differed between treatment arms only at the 12-month interview (P=.02, t test). For bilateral cases, the differences in mean changes between the observation and surgery arms were larger than for unilateral cases through the 24-month interviews (Figure 4B). Mean change in overall NEI-VFQ scores from baseline remained close to no change through the 36-month interview for bilateral cases in the observation arm but ranged from 4 to 9 points in the surgery arm. The 24-month interviews yielded the largest difference (9 points, 95% CI, 1–17) between the surgery arm and observation arm (P=.03, t test) for bilateral cases.
Figure 4.
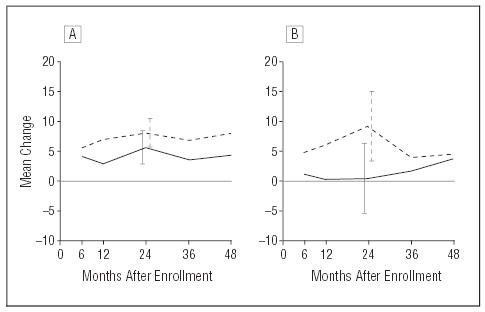
Mean changes in National Eye Institute Visual Function Questionnaire overall scores from baseline to each interview time by treatment arm. Vertical lines at 24 months delimit the 95% confidence intervals on mean changes. Positive changes indicate improvements from baseline scores. A, Unilateral cases. Solid line indicates patients in the observation arm (n=83 at baseline); broken line, patients in the surgery arm (n=84 at baseline). B, Bilateral cases. Solid line indicates patients in the observation arm (n=30 at baseline); broken line, patients in the surgery arm (n=28 at baseline).
Mean changes in NEI-VFQ subscale scores from baseline are displayed by treatment arm for unilateral and bilateral cases in Figure 5. For most scales, the patterns are similar to those seen for changes in overall scores in Figure 4. The largest differences between treatment arms for 24-month changes among unilateral cases were in the role difficulties and dependency subscales (6 points each). Among bilateral cases the largest differences (12–16 points) between treatment arms were observed at the 24-month interviews for the near activities, distance activities, role difficulties, and mental health subscales (P<.05, t tests). Among bilateral cases, score changes to the 36- and 48-month interviews (when fewer patients were eligible for interviews) were similar in the 2 treatment arms on most scales. Changes from baseline to the 24-month interview are summarized in Table 4 for unilateral cases and bilateral cases.
Figure 5.

Mean changes in National Eye Institute Visual Function Questionnaire scale scores from baseline to follow-up interviews at specified times. Vertical lines indicate 95% confidence intervals on mean changes to 24 months. For each subscale, the left panel of the pair presents mean changes among unilateral cases (83 in the observation arm and 84 in the surgery arm at baseline); the right panel, mean changes among bilateral cases (30 in the observation arm and 28 in the surgery arm at baseline). Positive changes indicate improvements from baseline scores. Solid lines indicate patients in the observation arm; broken lines, patients in the surgery arm.
Table 4.
Change in NEI-VFQ Scores From Baseline to 24-Month Interview by Baseline Choroidal Neovascularization (CNV) Status, SST Group H Trial
|
Mean Change and 95% CI by Baseline CNV Status and Treatment Arm |
||||||
|---|---|---|---|---|---|---|
|
Unilateral CNV Cases |
Bilateral CNV Cases |
|||||
| NEI-VFQ Scale | Observation (n = 68) | Surgery (n = 71) | Difference* (95% CI) | Observation (n = 26) | Surgery (n = 26) | Difference* (95% CI) |
| Overall | 5.5 | 8.0 | 2.5 (−1.1 to 6.0) | 0.3 | 9.1 | 8.8 (0.8 to 16.9)† |
| General vision | 4.9 | 7.3 | 2.5 (−2.8 to 7.8) | −1.5 | 7.7 | 9.2 (1.8 to 20.3)‡ |
| Driving | 8.0 | 8.9 | 0.8 (−5.8 to 7.5) | −11.1 | 2.8 | 13.9 (−2.8 to 30.6) |
| Near activities | 6.5 | 9.6 | 3.2 (−2.3 to 8.6) | 2.2 | 14.3 | 12.1 (1.9 to 22.2)† |
| Distance activities | 7.0 | 10.2 | 3.2 (−1.9 to 8.3) | 2.5 | 14.1 | 11.6 (1.9 to 21.3)† |
| Role difficulties | 6.5 | 12.9 | 6.3 (0.2 to 12.5)† | 1.2 | 16.1 | 14.9 (2.0 to 27.8)† |
| Mental health | 13.9 | 16.1 | 2.2 (−5.0 to 9.4) | 4.0 | 20.4 | 16.3 (3.5 to 29.2)§ |
| Dependency | 3.7 | 9.2 | 5.5 (−0.3 to 11.2)‡ | −1.0 | 12.5 | 13.5 (−0.2 to 27.3)‡ |
| Social functioning | 2.9 | 4.5 | 1.5 (−3.2 to 6.3) | 2.9 | 8.3 | 5.4 (−3.7 to 14.6) |
Abbreviations: CI, confidence interval; NEI-VFQ, National Eye Institute Visual Function Questionnaire; SST, Submacular Surgery Trials.
Positive differences indicate more improvement in the surgery arm than in the observation arm. These values are calculated by subtracting the value from the surgery arm from the observation arm.
.01<P≤.05, t test.
.05<P≤.10, t test.
P≤.01, t test.
As noted earlier, changes in visual acuity favored the surgery arm in the predefined subgroup of eyes with a best-corrected baseline visual acuity of 20/125 to 20/800 but did not differ between treatment arms in the subgroup of study eyes with a baseline visual acuity 20/50 to 20/100.7 In both subgroups, the 4-point difference in 24-month changes in overall scores (Table 5) was similar to the change for the full group of patients (Table 3). Among the smaller subgroup of patients who had visual acuity of the study eye worse than 20/100 at baseline, differences in 24-month changes are positive overall and for each subscale except driving, suggesting somewhat more improvement from baseline among patients in the surgery arm than in the observation arm. However, all CIs include 0, except for the general vision subscale, indicating that there may be no meaningful difference between treatment arms. The difference between treatment arms for the general vision subscale met our criterion for statistical significance (P=.01, t test). In the larger subgroup of patients whose study eyes had a baseline visual acuity of 20/50 to 20/100, inclusive, 24-month changes in the overall NEI-VFQ scores and scores on 4 subscales favored surgery (P<.05, t tests). The small number of bilateral cases limited investigation of the combined effects of bilateral choroidal neovascularization and baseline visual acuity of the study eye.
Table 5.
Change in NEI-VFQ Scores From Baseline to 24-Month Interview by Baseline Visual Acuity, SST Group H Trial
|
Mean Change and 95% CI by Baseline Visual Acuity and Treatment Arm |
||||||
|---|---|---|---|---|---|---|
|
Baseline Visual Acuity 20/50-20/100 |
Baseline Visual Acuity 20/125-20/800 |
|||||
| NEI-VFQ Scale | Observation (n = 56) | Surgery (n = 57) | Difference* | Observation (n = 38) | Surgery (n = 40) | Difference* |
| Overall | 2.5 | 6.8 | 4.3 (0.2 to 8.4)† | 6.3 | 10.3 | 4.0 (−1.6 to 9.7) |
| General vision | 2.9 | 4.0 | 1.1 (−5.6 to 7.8) | 3.3 | 12.2 | 9.0 (2.2 to 15.8)† |
| Driving | −0.1 | 7.8 | 7.9 (−1.5 to 17.3)‡ | 7.4 | 6.6 | −0.8 (−10.0 to 8.4) |
| Near activities | 2.4 | 7.6 | 5.2 (−0.4 to 10.7)‡ | 9.5 | 15.6 | 6.0 (−2.3 to 14.4) |
| Distance activities | 3.2 | 9.5 | 6.3 (1.1 to 11.5)† | 9.6 | 13.9 | 4.2 (−3.9 to 12.4) |
| Role difficulties | 2.5 | 10.7 | 8.3 (1.6 to 15.0)† | 8.9 | 18.0 | 9.1 (−0.7 to 18.9)‡ |
| Mental health | 11.8 | 15.3 | 3.5 (−4.5 to 11.4) | 10.3 | 20.0 | 9.7 (−0.7 to 20.2)‡ |
| Dependency | 0.4 | 8.6 | 8.1 (1.2 to 15.1)† | 5.3 | 12.2 | 6.9 (−2.4 to 16.2) |
| Social functioning | 1.6 | 4.2 | 2.6 (−2.5 to 7.7) | 4.8 | 7.3 | 2.5 (−4.9 to 9.8) |
Abbreviations: CI, confidence interval; NEI-VFQ, National Eye Institute Visual Function Questionnaire; SST, Submacular Surgery Trials.
Positive differences indicate more improvement in the surgery arm than in the observation arm. These values are calculated by subtracting the mean 2-year change for the observation arm from the mean 2-year change for the surgery arm.
P≤.05, t test.
.05<P≤.10, t test.
GENERAL HEALTH-RELATED QUALITY OF LIFE AND CHANGES DURING FOLLOW-UP
The mean SF-36 summary scale scores and standard deviations for the baseline and 24-month interviews are given in Table 6 by treatment arm for all patients and separately for unilateral cases and bilateral cases. Scores were similar in the 2 treatment arms at both time points. Mean scores for the physical component summary changed little during the 24-month period; in contrast, scores for the mental component summary increased (improved) during the same period, both overall and within subgroups of unilateral cases and bilateral cases, in both treatment arms. The mean difference in changes in the physical component summary scores for patients in the surgery arm vs patients in the observation arm was −0.4 (95% CI, −2.7 to +2.0). The mean difference in changes in the mental component summary scores was 1.0 (95% CI, −2.2 to +4.1). Neither the 24-month distributions of scores nor changes in scores differed between treatment arms (P>.40, Wilcoxon rank sum tests).
Table 6.
SF-36 Scores From Baseline and 24-Month Interviews by Treatment Arm, SST Group H Trial
|
Mean Score* (SD) |
|||||
|---|---|---|---|---|---|
|
Baseline Interviews |
24-mo Interviews |
||||
| Patient Subgroup | Summary Scale | Observation | Surgery | Observation | Surgery |
| All patients | Physicalcomponent | 49.6 (8.2) | 48.7 (8.0) | 48.7 (8.5) | 47.7 (9.1) |
| Mentalcomponent | 49.6 (12.0) | 49.6 (11.6) | 51.9 (10.7) | 52.7 (10.4) | |
| No. of patients | 113 | 112 | 94 | 97 | |
| Unilateral cases | Physicalcomponent | 50.5 (7.5) | 48.9 (6.6) | 49.8 (7.8) | 48.7 (9.0) |
| Mentalcomponent | 50.7 (11.4) | 50.1 (11.4) | 52.6 (10.6) | 52.4 (10.5) | |
| No. of patients | 83 | 84 | 68 | 71 | |
| Bilateral cases | Physicalcomponent | 47.0 (9.4) | 48.0 (11.3) | 46.0 (9.5) | 45.3 (9.3) |
| Mentalcomponent | 46.5 (13.1) | 48.0 (12.1) | 50.1 (11.0) | 53.5 (10.2) | |
| No. of patients | 30 | 28 | 26 | 26 | |
Abbreviations: SF-36, 36-Item Short Form Health Survey; SST, Submacular Surgery Trials.
Higher scores indicate better health status.
DEPRESSION AND ANXIETY DURING FOLLOW-UP
At the time of enrollment, 10 patients (9%) in the observation arm and 7 (6%) in the surgery arm were classified by the HADS criteria as definite cases of depression. Based on the 24-month interview, 3 patients (3%) in the observation arm and 2 (2%) in the surgery arm among those interviewed were so classified. Baseline prevalence of definite anxiety was higher than depression: 20 patients (18%) in the observation arm and 15 (13%) in the surgery arm. Based on the 24-month interview, only 6 patients (6%) in each treatment arm were classified as definite cases of anxiety using the HADS criteria. Thus, treatment arms did not differ with respect to HADS classification as definite cases of either depression or anxiety.
COMMENT
During follow-up in the SST Group H Trial, patients in the surgery arm reported somewhat better vision-targeted quality of life than patients in the observation arm, as indicated by changes in scores on follow-up interviews compared with baseline. Median scores from follow-up interviews tended to remain near baseline levels or to improve in both treatment arms (Figure 2). Improvements from baseline scores were larger in the surgery arm (Table 3), with the largest effect of surgery noted at 12 months. The modest difference in unadjusted and adjusted 24-month changes in overall NEI-VFQ scores favoring the surgery arm parallels the difference of 1.2 lines (6 letters) in mean change in visual acuity from baseline to the 24-month examination in study eyes among SST Group H Trial patients reported elsewhere.7 Similarly, the difference in change scores on the near activities subscale (Table 3) may reflect the larger proportion of study eyes in the surgery arm than in the observation arm that had faster reading speeds at 24 months than at baseline.7
Most patients who participated in this clinical trial were of working age and employed at baseline (Table 1). Thus, differences favoring surgery for bilateral cases merit consideration when treatment options for subfoveal choroidal neovascularization in the second eye of a patient with the ocular histoplasmosis syndrome are presented. Bilateral cases were expected to demonstrate more marked effects of surgery, whether positive or negative. Some of the differences observed among bilateral cases that favored surgery were quite large at 6 months through 24 months after enrollment (Figure 5). When mean changes from baseline scores were considered (Table 4), differences of 10 points or larger were observed for bilateral cases for at least 2 years on several NEI-VFQ scales. An earlier investigation21 suggested that differences of this size may reflect an appreciable difference in the aspects of visual function captured by these scales. The findings favoring surgery among both unilateral cases and bilateral cases on the role difficulties and dependency sub-scales (Table 4) may be particularly important to patients of working age.
The target sample size for the SST Group H Trial was projected based on visual acuity outcomes of interest and not quality-of-life outcomes. Nevertheless, the power of the trial was sufficient to detect differences between treatment arms for NEI-VFQ scores (Table 3). Furthermore, despite a smaller number of bilateral than unilateral cases enrolled (58 and 167 patients, respectively), this number was sufficient to document poorer scores among bilateral cases than among unilateral cases at baseline (Figure 1) and during follow-up (Figure 3).
The largest improvements in visual acuity after surgery were observed during the first year of clinical follow-up7 while the largest improvements in NEI-VFQ scores were observed at the 24-month interviews (Table 3). Although this comparison and the decline in scores after the 24-month interview suggest a lag in the patient’s perception of the effects of worsening visual acuity when the change is relatively slow, the difference could be due to chance or to factors not yet identified. Further exploration of the independent and joint effects of visual acuity and changes from baseline over time, bilateral choroidal neovascularization, usual occupation, and other factors will be required to understand the net effect on vision-targeted quality of life as measured by the NEI-VFQ.
Several theories have been proposed to explain why scores on most NEI-VFQ scales improved for most patients in both treatment arms. Patients may have adapted to their vision problems or acquired coping skills. Participation in a multicenter clinical trial, in which both eyes were monitored, may have had a positive effect on patients’ perceptions of care received or may have had an altruistic appeal. However, a similar effect on vision-targeted quality of life was not observed in the SST Group B Trial22 or SST Group N Trial.23 Scores may have improved as patients gained familiarity with the interview or with the interviewers, possibly prompting a desire to please. Initially patients may have feared further large losses of visual acuity in the study eye or fellow eye; with mean 2-year visual acuity losses of 2 lines in study eyes in the observation arm, 1 line in study eyes in the surgery arm, and no loss in the fellow eyes of patients in either treatment arm, patients may have been reassured regarding the rate of future visual acuity loss. Finally, patients may have responded positively to the attention to their concerns, expressed by the clinical staff and implied by inclusion of the health-related quality-of-life interview as part of data collection for this trial, or may have had a greater awareness of being part of the research team. If this explanation were correct, the same phenomenon should have been observed in the other SST clinical trials.22,23 These and other factors that may have contributed to improved scores require further investigation and are beyond the scope of this report.
Although the SF-36 has been used widely, either alone or, as in the SST Group H Trial, together with a condition-targeted instrument, it has not proven to be sensitive to changes in vision in patients with subfoveal choroidal neovascularization.24 Thus, this instrument does not appear to be useful as an outcome measure in clinical trials in ophthalmology in which vision is affected by the condition studied. However, the SF-36 has been shown to be useful as a surrogate for other health conditions when adjustment of NEI-VFQ scores for such conditions is desired in either cross-sectional or prospective studies.25
As the first study of vision-targeted and general health-related quality of life among patients with choroidal neovascularization in the ocular histoplasmosis syndrome or idiopathic choroidal neovascularization, the SST Group H Trial has quantified the effect of both unilateral and bilateral choroidal neovascularization6 and provided data regarding changes over time in a well-characterized subset of patients. The database on which these findings are based was quite good, with high rates of completion of expected interviews in both treatment arms and few missing responses. Findings for patients in the observation arm may be particularly useful when designing future evaluations of treatments for similar ophthalmic conditions. As demonstrated by the SST Group H Trial, findings from quality-of-life assessment may support recommendations based on clinical findings but also may prompt further investigation of factors that contribute to the clinical outcomes and effects on the patient. In conclusion, ophthalmologists and patients similar to those who participated in the SST Group H Trial should consider the potential effects of submacular surgery on specific aspects of vision-targeted quality of life as well as on ophthalmic outcomes when considering treatment options.
Footnotes
Writing Committee for SST Group H Trial Report No. 10: Barbara S. Hawkins, PhD; Päivi H. Miskala, PhD; Eric B. Bass, MD, MPH; Neil M. Bressler, MD; Ashley L. Childs, MS; Carol M. Mangione, MD, MSPH; Marta J. Marsh, MS. Documentation of approval of the manuscript submitted for publication by the members of the SST Research Group is on file at the SST Coordinating Center, Baltimore, Md.
Financial Disclosure: Dr Bressler’s employer, The Johns Hopkins University, receives funding from Novartis Pharma AG, Basel, Switzerland; QLT, Inc, Vancouver, British Columbia; and Genentech, South San Francisco, Calif, for consulting services and research efforts by Dr Bressler. The terms of these arrangements are managed by The Johns Hopkins University according to its conflict-of-interest policies.
Funding/Support: The SST is sponsored by the National Eye Institute, National Institutes of Health, US Department of Health and Human Services, Bethesda, Md, through cooperative agreements U10 EY11547, EY11557, and EY11558 with The Johns Hopkins University. Participating clinical centers were supported by contracts with The Johns Hopkins University. Dr Mangione’s participation as chair of the SST Patient-Centered Outcomes Subcommittee is supported by contract between the David Geffen School of Medicine, University of California, Los Angeles, and The Johns Hopkins University.
References
- 1.Ware JE Jr. Evaluating measures of general health concepts for use in clinical trials. In: Furberg C, Schuttinga JA, eds. Quality of Life Assessment. Practice, Problems, and Promise: Proceedings of a Workshop, October 15–17, 1990. Washington, DC: US Dept of Health and Human Services; 1993:51–63. NIH publication 93–3503.
- 2.Patrick DL. Reactions and recommendations: quality of life in NIH-sponsored studies. In: Furberg C, Schuttinga JA, eds. Quality of Life Assessment. Practice, Problems, and Promise. Proceedings of a Workshop, October 15–17, 1990. Washington, DC: US Dept of Health and Human Services; 1993:81–88. NIH publication 93–3503.
- 3.Drummond MF, Ferris FF III. Major themes and conclusions: a strategy for measurement of quality of life in National Eye Institute Trials. In: Drummond MF, ed. Measuring the Quality of Life of People With Visual Impairment: Proceedings of a Workshop. Washington, DC: US Dept of Health and Human Services; 1990:65–66. NIH publication 90–3098.
- 4.McDowell I. General health measurement and quality of life in visual impairment. In: Drummond MF, ed. Measuring the Quality of Life of People With Visual Impairment: Proceedings of a Workshop. Washington, DC: US Dept of Health and Human Services; 1990:29–33. NIH publication 90–3098.
- 5.Submacular Surgery Trials Pilot Study Investigators. Submacular Surgery Trials randomized pilot trial of laser photocoagulation versus surgery for recurrent choroidal neovascularization secondary to age-related macular degeneration, II: Quality-of-life outcomes: Submacular Surgery Trials Pilot Study report No. 2. Am J Ophthalmol. 2000;130:408–418. doi: 10.1016/s0002-9394(00)00730-3. [DOI] [PubMed] [Google Scholar]
- 6.Submacular Surgery Trials Research Group. Health- and vision-targeted quality of life among patients with ocular histoplasmosis or idiopathic choroidal neovascularization at time of enrollment in a randomized trial of submacular surgery: SST report No. 5. Arch Ophthalmol In press. [DOI] [PMC free article] [PubMed]
- 7.Submacular Surgery Trials Research Group. Surgical removal vs observation for subfoveal choroidal neovascularization, either associated with the ocular histoplasmosis syndrome or idiopathic, I: ophthalmic findings from a randomized clinical trial: SST Group H Trial: SST report No. 9. Arch Ophthalmol. 2004;122:1597–1611. doi: 10.1001/archopht.122.11.1597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Submacular Surgery Trials Research Group. Submacular Surgery Trials (SST) Manual of Procedures. Springfield, Va: National Technical Information Service; 1998. NTIS accession No. PB98–166648.
- 9.Submacular Surgery Trials Research Group. Submacular Surgery Trials (SST) Forms Book. Springfield, Va: National Technical Information Service; 1998. NTIS accession No. PB98–159445.
- 10.Mangione CM, Lee PP, Gutierrez PR, Spritzer K, Berry S, Hays RD National Eye Institute Visual Function Questionnaire Field Test Investigators. Development of the 25-item National Eye Institute Visual Function Questionnaire. Arch Ophthalmol. 2001;119:1050–1058. doi: 10.1001/archopht.119.7.1050. [DOI] [PubMed] [Google Scholar]
- 11.Mangione CM, Lee PP, Pitts J, Gutierrez P, Berry S, Hays RD NEI-VFQ Field Test Investigators. Psychometric properties of the National Eye Institute Visual Function Questionnaire (NEI-VFQ) Arch Ophthalmol. 1998;116:1496–1504. doi: 10.1001/archopht.116.11.1496. [DOI] [PubMed] [Google Scholar]
- 12.VFQ-25 Rand Health homepage. Available at: http://www.rand.org/health/surveys/vfq25/ Accessed September 24, 2004.
- 13.Ware JE, Snow KK, Kosinski M, Gaskel B. SF-36 Health Survey Manual and Interpretation Guide. Boston, Mass: The Health Institute, New England Medical Center, 1993.
- 14.Zigmond AS, Snaith RP. The Hospital Anxiety and Depression Scale. Acta Psychiatr Scand. 1983;67:361–370. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]
- 15.Submacular Surgery Trials Research Group. Patients’ perceptions of the value of current vision: assessment of preference values among patients with subfoveal choroidal neovascularization—The Submacular Surgery Trials Vision Preference Value Scale (SST-VPVS). SST report No. 6. Arch Ophthalmol In press. [DOI] [PMC free article] [PubMed]
- 16.Ware JE, Kosinski M, Keller SD. SF-36 Physical and Mental Health Summary Scales: A User’s Manual. Boston, Mass: The Health Institute, New England Medical Center; 1994.
- 17.Williams RA, Brody BL, Thomas RG, Kaplan RM, Brown SJ. The psychological impact of macular degeneration. Arch Ophthalmol. 1998;116:514–520. doi: 10.1001/archopht.116.4.514. [DOI] [PubMed] [Google Scholar]
- 18.Herrmann C. International experiences with the Hospital Anxiety and Depression Scale: a review of validation data and clinical results. J Psychosom Res. 1997;42:17–41. doi: 10.1016/s0022-3999(96)00216-4. [DOI] [PubMed] [Google Scholar]
- 19.Tukey JW. Exploratory Data Analysis. Reading, Mass: Addison-Wesley Publishing Co; 1977:34–41.
- 20.Snedecor GW, Cochran WG. Statistical Methods. 7th ed. Ames: Iowa State University Press; 1980:144–145.
- 21.Submacular Surgery Trials Research Group. Responsiveness of the National Eye Institute Visual Function Questionnaire to changes in visual acuity: findings in patients with subfoveal choroidal neovascularization: SST report No. 1. [published correction appears in Arch Ophthalmol2003:121:1513] Arch Ophthalmol. 2003;121:531–539. doi: 10.1001/archopht.121.4.531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Submacular Surgery Trials Research Group. Surgery for hemorrhagic choroidal neovascular lesions of age-related macular degeneration: quality-of-life findings: SST report No. 14. Ophthalmology. 2004;111:2007–2014. doi: 10.1016/j.ophtha.2004.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Submacular Surgery Trials Research Group. Surgery for subfoveal choroidal neovascularization in age-related macular degeneration: quality-of-life findings: SST report No. 12. Ophthalmology. 2004;111:1981–1992. doi: 10.1016/j.ophtha.2004.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Childs AL the Submacular Surgery Trials Patient-Centered Outcomes Subcommittee for the Submacular Surgery Trials Pilot Study Investigators. Responsiveness of the SF-36 Health Survey to changes in visual acuity among patients with subfoveal choroidal neovascularization. Am J Ophthalmol. 2004;137:373–375. doi: 10.1016/S0002-9394(03)00911-5. [DOI] [PubMed] [Google Scholar]
- 25.Miskala PH, Bressler NM, Meinert CL. Relative contributions of reduced vision and general health to NEI-VFQ scores in patients with neovascular age-related macular degeneration. Arch Ophthalmol. 2004;122:758–766. doi: 10.1001/archopht.122.5.758. [DOI] [PubMed] [Google Scholar]


