Abstract
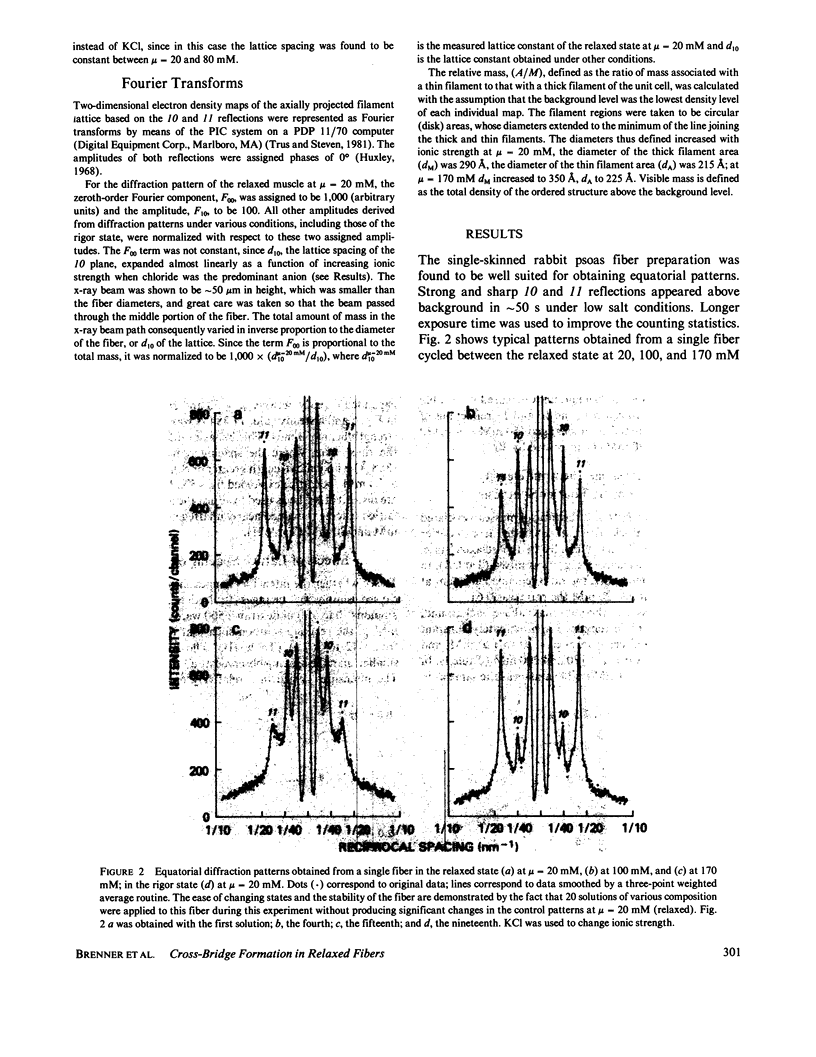
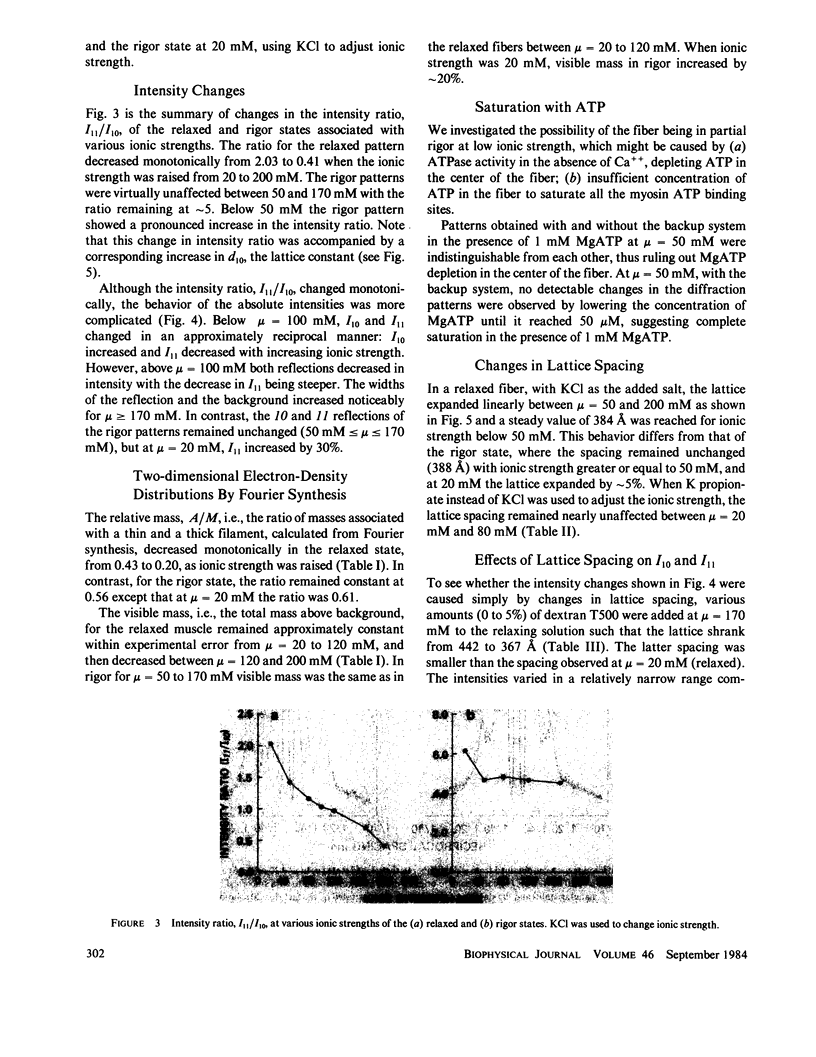
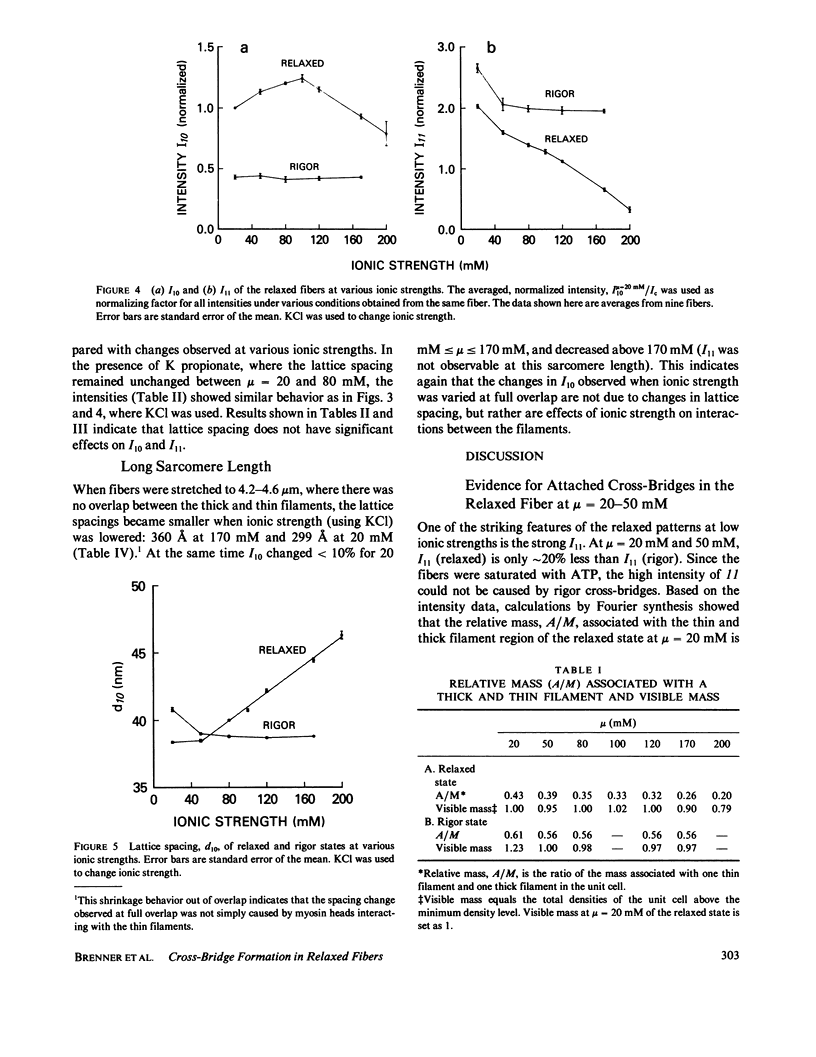
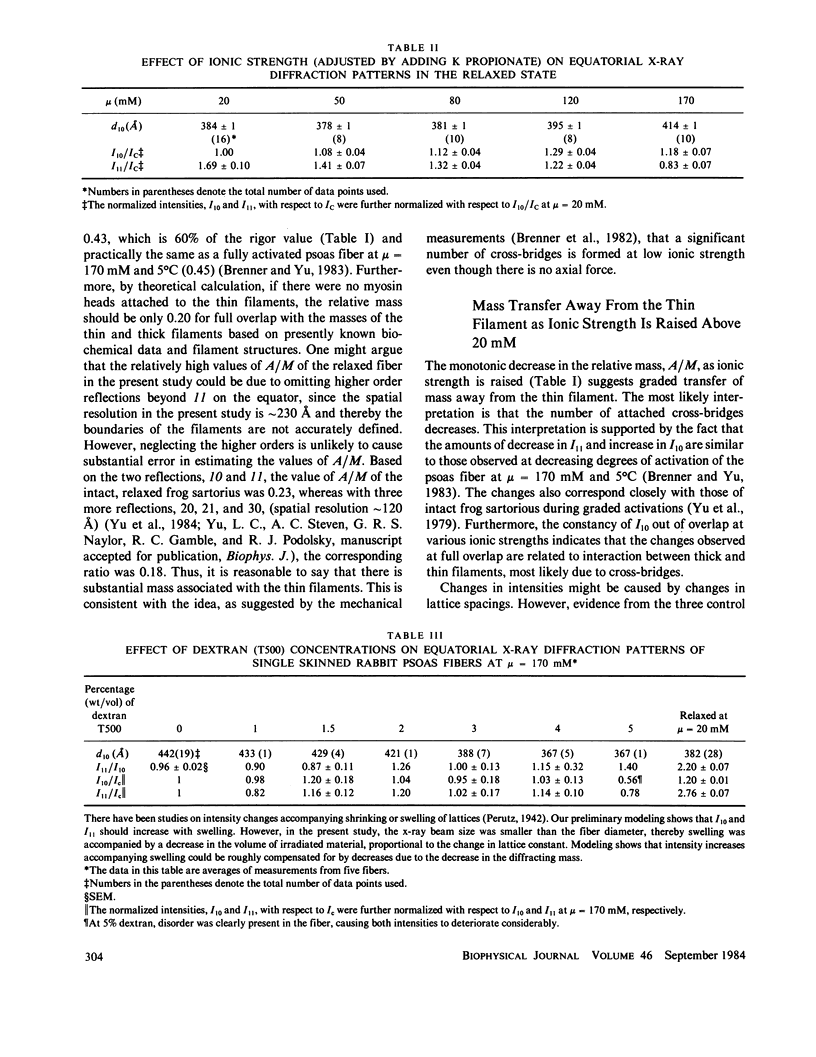
Equatorial x-ray diffraction patterns from single skinned rabbit psoas fibers were studied at various ionic strengths to obtain structural information regarding cross-bridge formation in relaxed muscle fibers. At ionic strengths between 20 and 50 mM, the intensity of the 11 reflection, I11, of the relaxed state was close to that of the rigor state, whereas the intensity of the 10 reflection, I10, was approximately twice that of rigor reflection. Calculations by two-dimensional Fourier synthesis indicated that substantial extra mass was associated with the thin filaments under these conditions. With increasing ionic strength between 20 and 100 mM, I10 increased and I11 decreased in an approximately linear way, indicating net transfer of mass away from the thin filaments towards the thick filaments. These results provided evidence that cross-bridges were formed in a relaxed fiber at low ionic strengths, and that the number of cross-bridges decreased as ionic strength was raised. Above mu = 100 mM, I10 and I11 both decreased, indicating the onset of increasing disorder within the filament lattice.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brenner B., Schoenberg M., Chalovich J. M., Greene L. E., Eisenberg E. Evidence for cross-bridge attachment in relaxed muscle at low ionic strength. Proc Natl Acad Sci U S A. 1982 Dec;79(23):7288–7291. doi: 10.1073/pnas.79.23.7288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenner B. Technique for stabilizing the striation pattern in maximally calcium-activated skinned rabbit psoas fibers. Biophys J. 1983 Jan;41(1):99–102. doi: 10.1016/S0006-3495(83)84411-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chalovich J. M., Chock P. B., Eisenberg E. Mechanism of action of troponin . tropomyosin. Inhibition of actomyosin ATPase activity without inhibition of myosin binding to actin. J Biol Chem. 1981 Jan 25;256(2):575–578. [PMC free article] [PubMed] [Google Scholar]
- Chalovich J. M., Eisenberg E. Inhibition of actomyosin ATPase activity by troponin-tropomyosin without blocking the binding of myosin to actin. J Biol Chem. 1982 Mar 10;257(5):2432–2437. [PMC free article] [PubMed] [Google Scholar]
- Huxley H. E. Structural difference between resting and rigor muscle; evidence from intensity changes in the lowangle equatorial x-ray diagram. J Mol Biol. 1968 Nov 14;37(3):507–520. doi: 10.1016/0022-2836(68)90118-6. [DOI] [PubMed] [Google Scholar]
- Lymn R. W. Myosin subfragment-1 attachment to actin. Expected effect on equatorial reflections. Biophys J. 1978 Jan;21(1):93–98. doi: 10.1016/S0006-3495(78)85510-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuda T., Podolsky R. J. X-ray evidence for two structural states of the actomyosin cross-bridge in muscle fibers. Proc Natl Acad Sci U S A. 1984 Apr;81(8):2364–2368. doi: 10.1073/pnas.81.8.2364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Podolsky R. J., St Onge H., Yu L., Lymn R. W. X-ray diffraction of actively shortening muscle. Proc Natl Acad Sci U S A. 1976 Mar;73(3):813–817. doi: 10.1073/pnas.73.3.813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wray J. S., Vibert P. J., Cohen C. Cross-bridge arrangements in Limulus muscle. J Mol Biol. 1974 Sep 15;88(2):343–348. doi: 10.1016/0022-2836(74)90486-0. [DOI] [PubMed] [Google Scholar]
- Yu L. C., Arata T., Steven A. C., Naylor G. R., Gamble R. C., Podolsky R. J. Structural studies of muscle during force development in various states. Adv Exp Med Biol. 1984;170:207–220. doi: 10.1007/978-1-4684-4703-3_19. [DOI] [PubMed] [Google Scholar]
- Yu L. P., Hartt J. E., Podolsky R. J. Equatorial x-ray intensities and isometric force levels in frog sartorius muscle. J Mol Biol. 1979 Jul 25;132(1):53–67. doi: 10.1016/0022-2836(79)90495-9. [DOI] [PubMed] [Google Scholar]



