Abstract
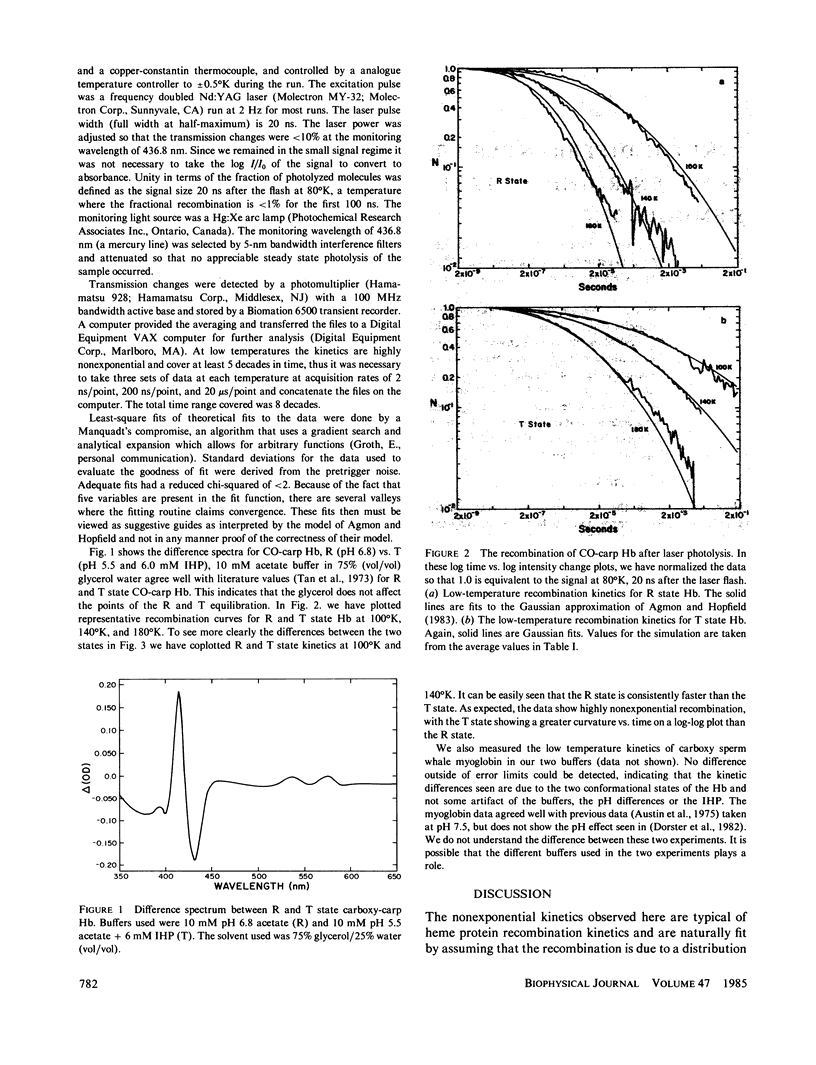
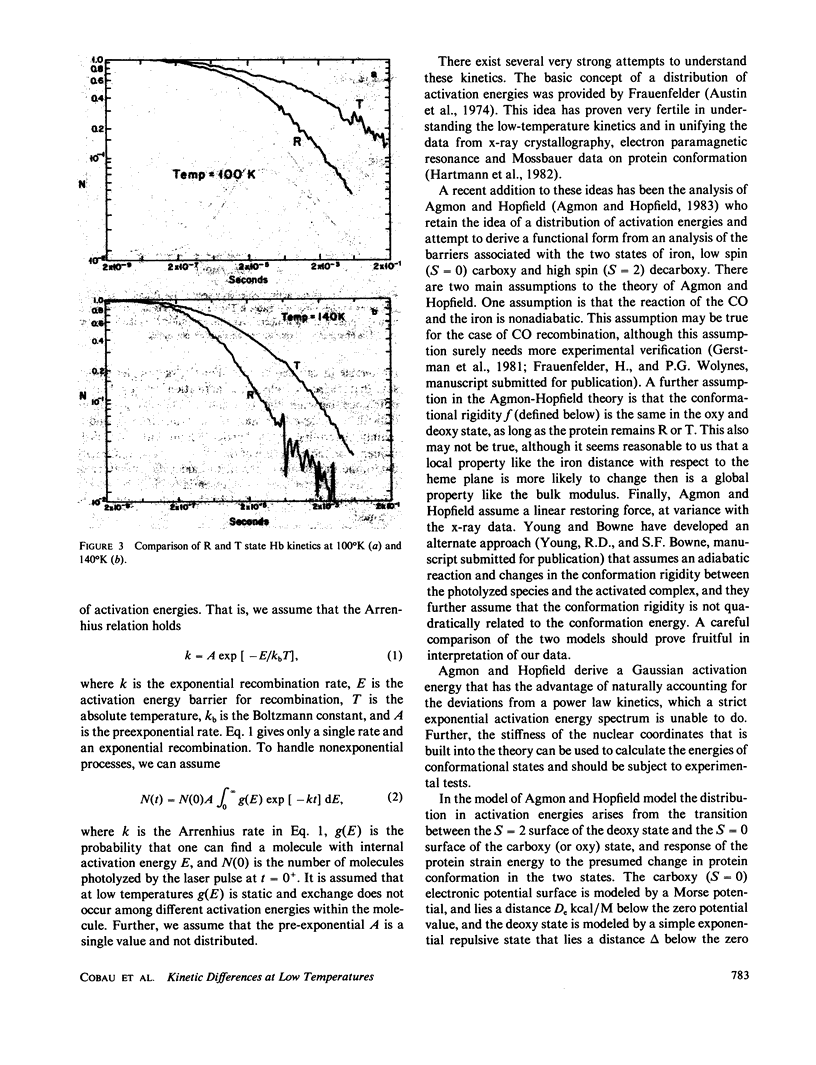
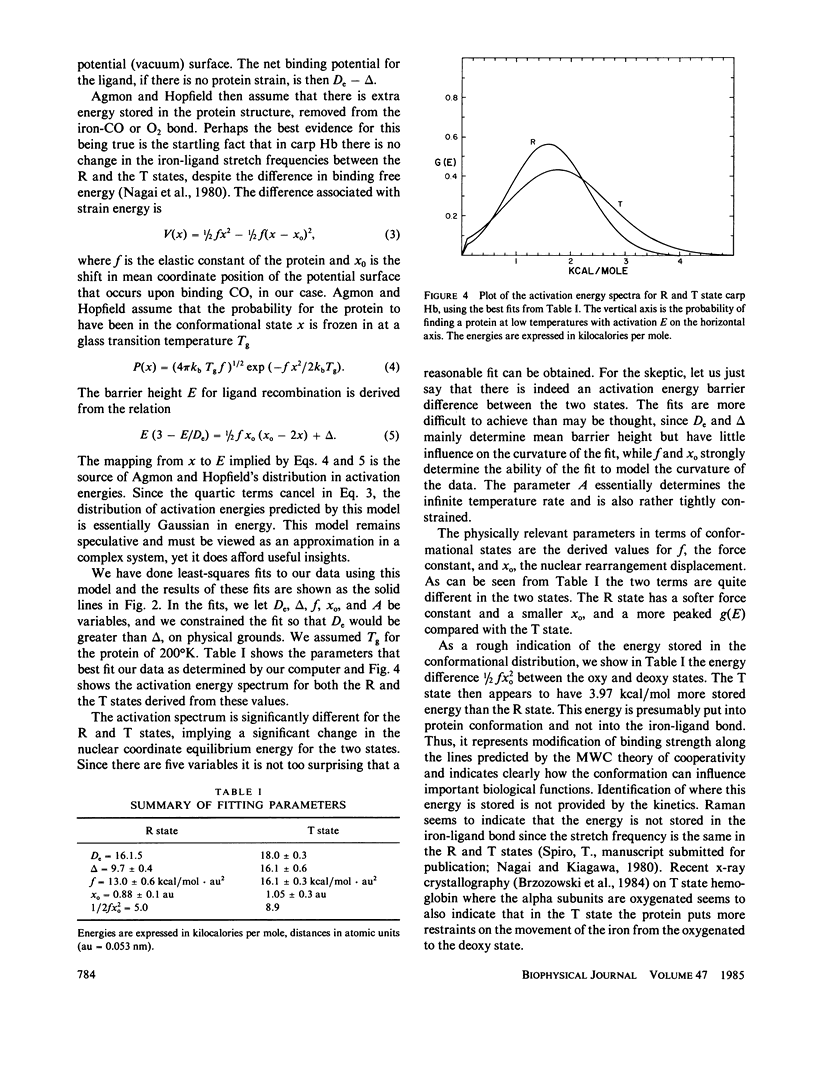
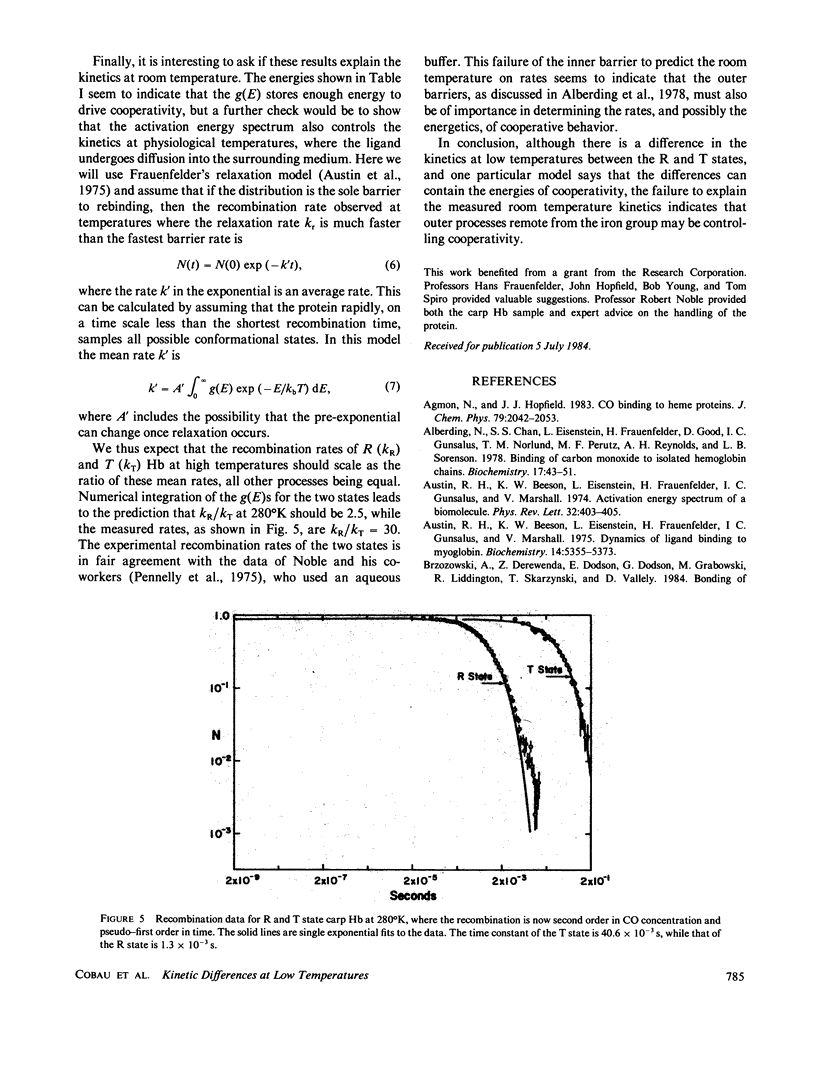
We use the low-temperature recombination kinetics of carbon monoxide with carp hemoglobin to determine that the R and T states of hemoglobin exhibit different low-temperature geminate recombination kinetics. The peak of the fitted Gaussian activation energy spectrum is at 1.5 kcal/mol for R state and 1.8 kcal/mol for T state. The distribution in activation energies is fit well by the Agmon-Hopfield linear strain model. The T state is fit with a stronger elastic constant than R, and has a larger displacement of the protein conformation coordinate than does the R state, indicating that the T state does have a significantly greater rigidity and also stores more strain energy in its conformational states than does R hemoglobin. The pre-exponential in the activation energy spectrum is only a factor of two greater in the R than the T state and the low-temperature activation energy spectrum does not correctly predict the difference in the on rates for R and T states at 300 degrees K, indicating that processes removed from the binding site are important in cooperativity.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alberding N., Chan S. S., Eisenstein L., Frauenfelder H., Good D., Gunsalus I. C., Nordlund T. M., Perutz M. F., Reynolds A. H., Sorensen L. B. Binding of carbon monoxide to isolated hemoglobin chains. Biochemistry. 1978 Jan 10;17(1):43–51. doi: 10.1021/bi00594a007. [DOI] [PubMed] [Google Scholar]
- Austin R. H., Beeson K. W., Eisenstein L., Frauenfelder H., Gunsalus I. C. Dynamics of ligand binding to myoglobin. Biochemistry. 1975 Dec 2;14(24):5355–5373. doi: 10.1021/bi00695a021. [DOI] [PubMed] [Google Scholar]
- Doster W., Beece D., Bowne S. F., DiIorio E. E., Eisenstein L., Frauenfelder H., Reinisch L., Shyamsunder E., Winterhalter K. H., Yue K. T. Control and pH dependence of ligand binding to heme proteins. Biochemistry. 1982 Sep 28;21(20):4831–4839. doi: 10.1021/bi00263a001. [DOI] [PubMed] [Google Scholar]
- Hartmann H., Parak F., Steigemann W., Petsko G. A., Ponzi D. R., Frauenfelder H. Conformational substates in a protein: structure and dynamics of metmyoglobin at 80 K. Proc Natl Acad Sci U S A. 1982 Aug;79(16):4967–4971. doi: 10.1073/pnas.79.16.4967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MONOD J., WYMAN J., CHANGEUX J. P. ON THE NATURE OF ALLOSTERIC TRANSITIONS: A PLAUSIBLE MODEL. J Mol Biol. 1965 May;12:88–118. doi: 10.1016/s0022-2836(65)80285-6. [DOI] [PubMed] [Google Scholar]
- Nagai K., Kitagawa T. Differences in Fe(II)-N epsilon(His-F8) stretching frequencies between deoxyhemoglobins in the two alternative quaternary structures. Proc Natl Acad Sci U S A. 1980 Apr;77(4):2033–2037. doi: 10.1073/pnas.77.4.2033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noble R. W., Parkhurst L. J., Gibson Q. H. The effect of pH on the reactions of oxygen and carbon monoxide with the hemoglobin of the carp, Cyprinus carpio. J Biol Chem. 1970 Dec 25;245(24):6628–6633. [PubMed] [Google Scholar]
- Pennelly R. R., Tan-Wilson A. L., Noble R. W. Structural states and transitions of carp hemoglobin. J Biol Chem. 1975 Sep 25;250(18):7239–7244. [PubMed] [Google Scholar]
- Perutz M. F. Nature of haem-haem interaction. Nature. 1972 Jun 30;237(5357):495–499. doi: 10.1038/237495a0. [DOI] [PubMed] [Google Scholar]
- Tan A. L., De Young A., Noble R. W. The pH dependence of the affinity, kinetics, and cooperativity of ligand binding to carp hemoglobin, Cyprinus carpio. J Biol Chem. 1972 Apr 25;247(8):2493–2498. [PubMed] [Google Scholar]


