Abstract
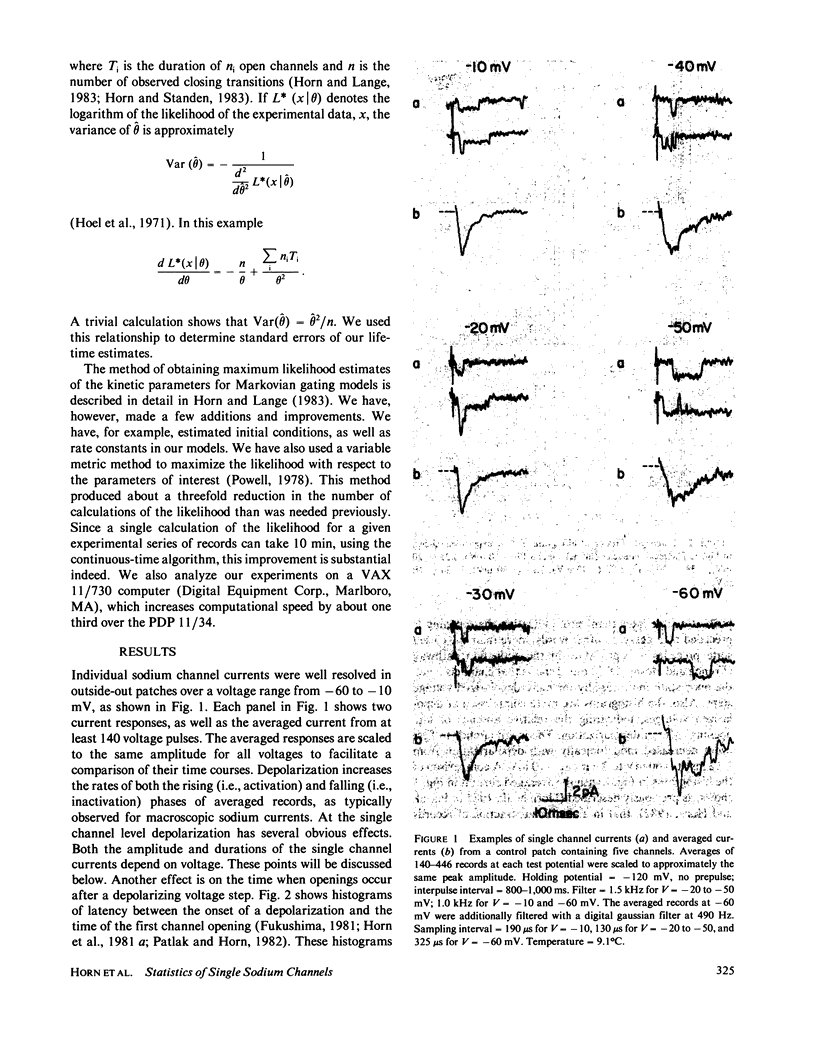
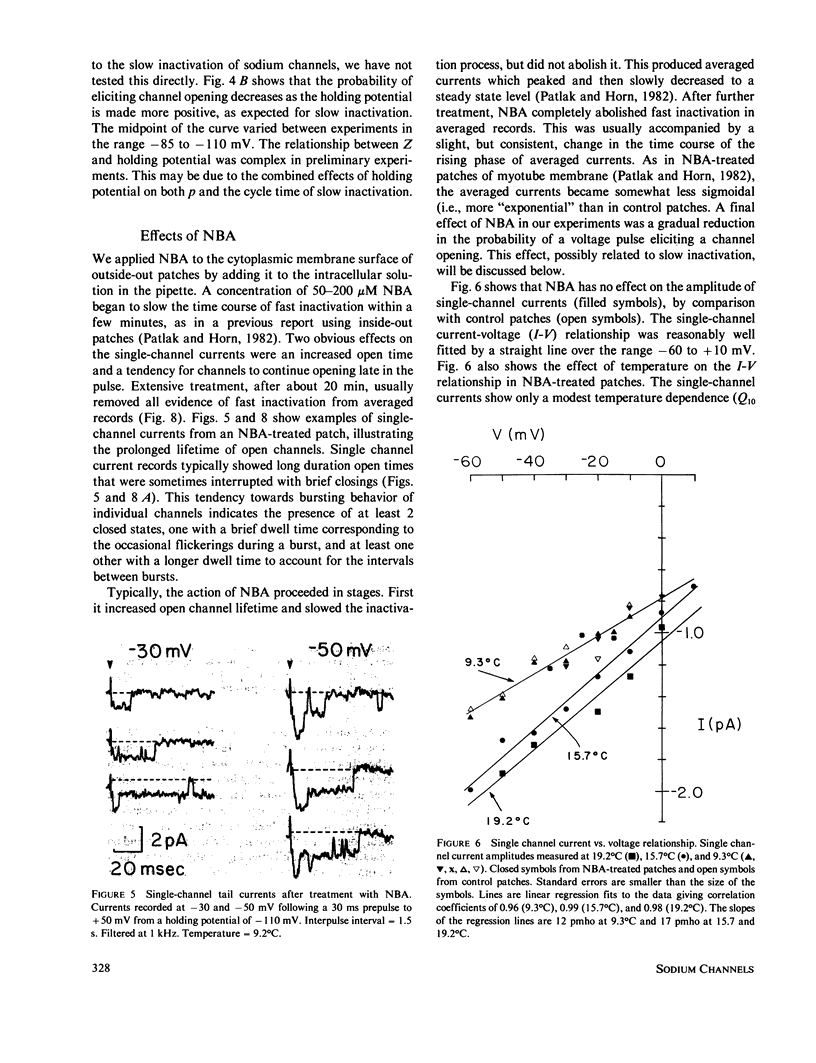
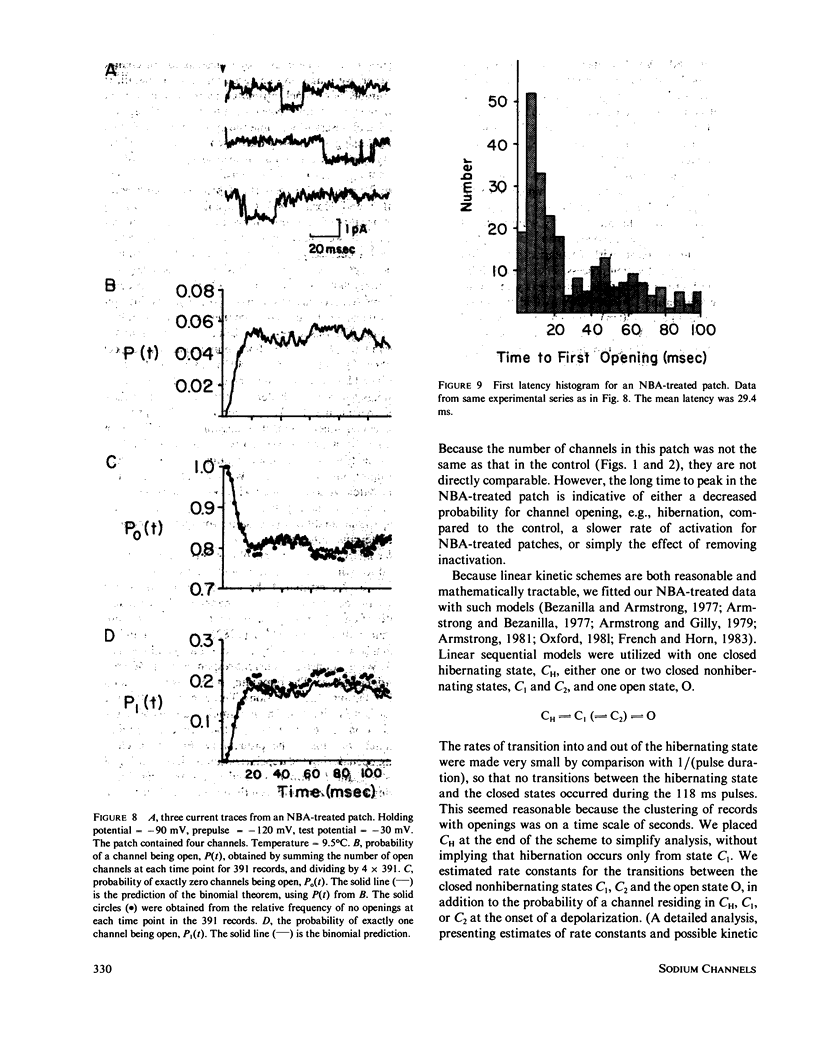
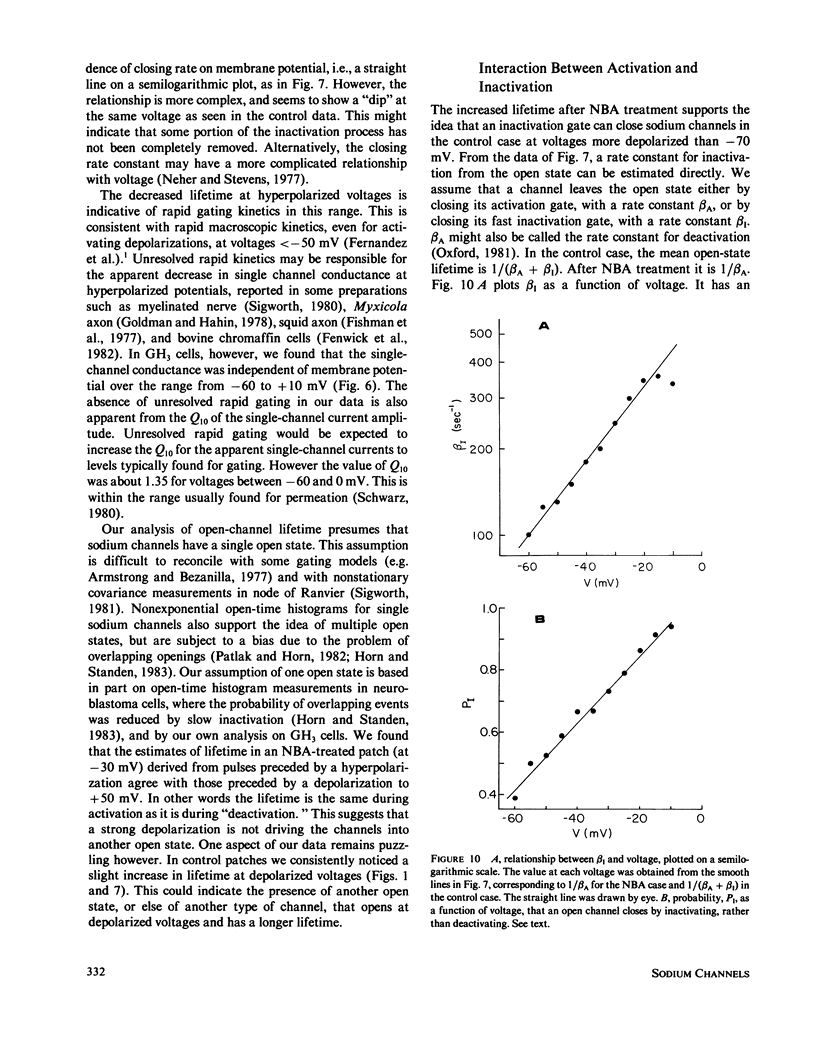
Currents were obtained from single sodium channels in outside-out excised patches of membrane from the cell line GH3. The currents were examined in control patches and in patches treated with N- bromoacetamide ( NBA ) to remove inactivation. The single-channel current-voltage relationship was linear over the range -60 to + 10 mV, and was unaffected by NBA . The slope conductance at 9.3 degrees C was 12 pS, and the Q10 for single channel currents was about 1.35. The currents in both control and NBA -treated patches showed evidence of a slow process similar to desensitization in acetylcholine-receptor channels. This process was especially apparent at rapid rates of stimulation (5 Hz), where openings occurred in clusters of records. The clustering of records with and without openings was analyzed by runs analysis, which showed a statistically significant trend toward nonrandom ordering in the responses of channels to voltage pulses. NBA made this nonrandom pattern more apparent. The probability that an individual channel was "hibernating" during an activating depolarization was estimated by a maximum likelihood method. The lifetime of the open state was also estimated by a maximum likelihood method, and was examined as a function of voltage. In control patches the open time was mildly voltage-dependent, showing a maximum at about -50 mV. In NBA -treated patches the open time was greater than in the control case and increased monotonically with depolarization; it asymptotically approached that of the control patches at hyperpolarized potentials. By comparing channel open times in control and NBA -treated patches, we determined beta A and beta I, the rate constants for closing activation gates and fast inactivation gates. Beta I was an exponential function of voltage, increasing e-fold for 34 mV. beta A had the opposite voltage dependence. The probability of an open channel closing its fast inactivation gate, rather than its activation gate, increased linearly with depolarization from -60 to -10 mV. These results indicate that inactivation is inherently voltage dependent.
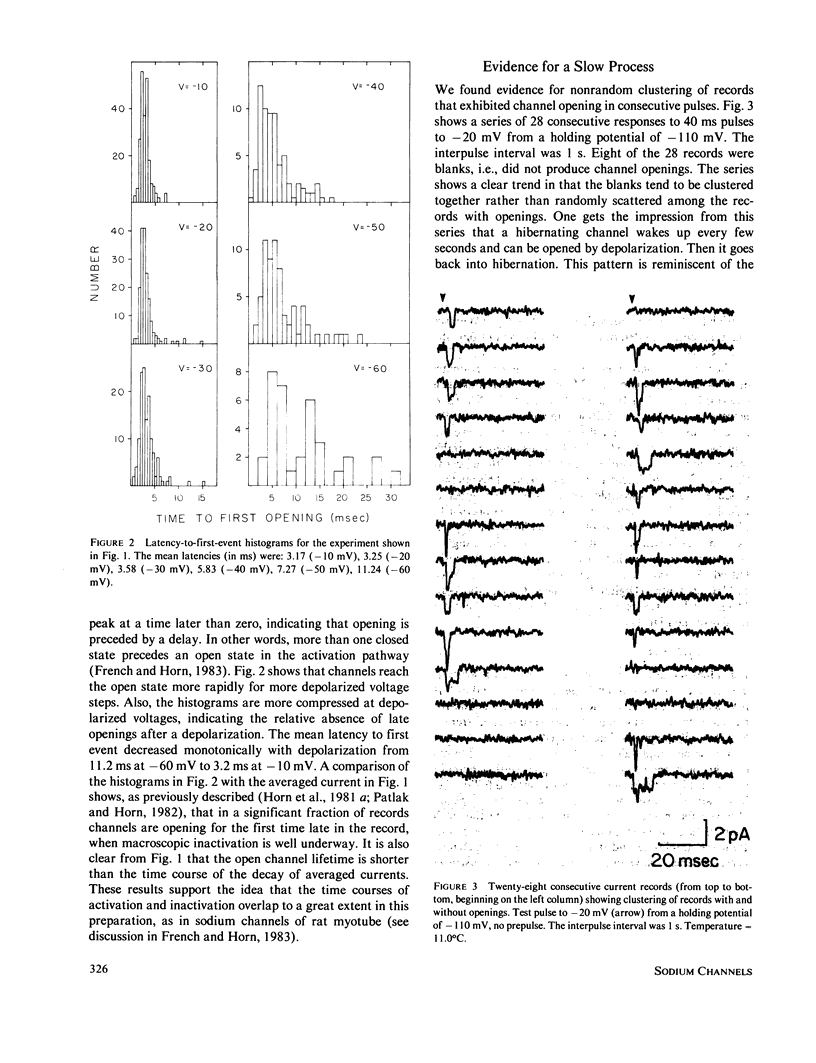
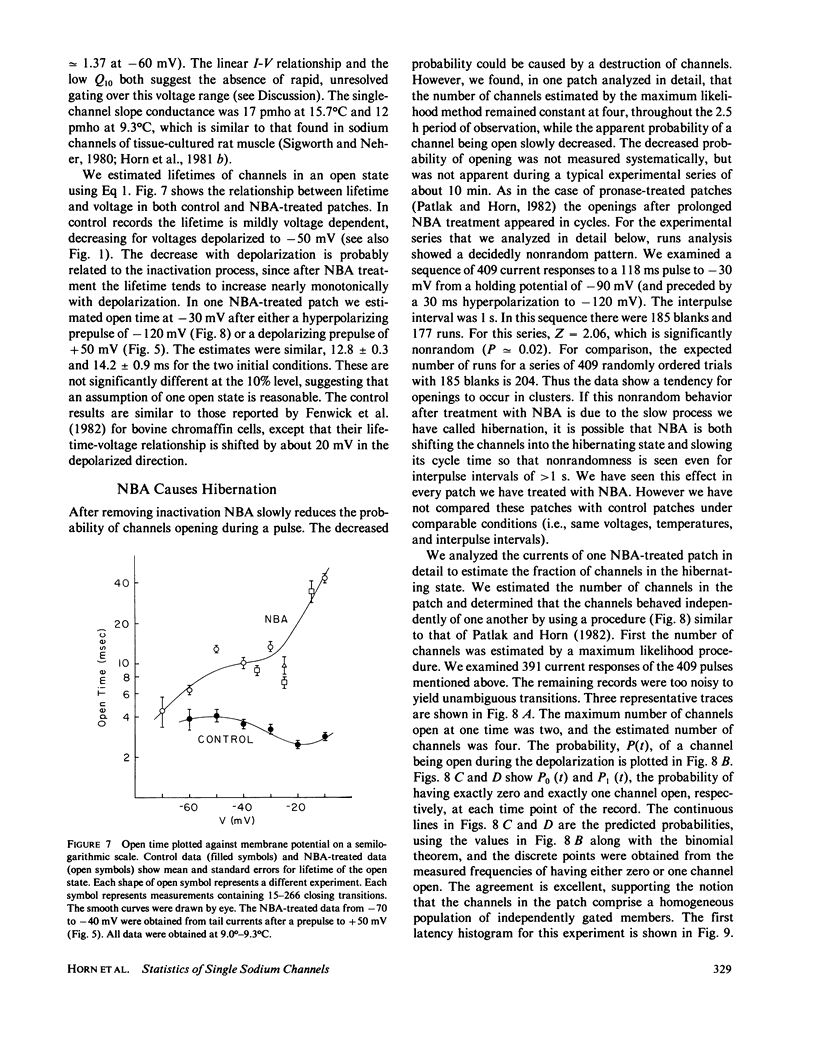
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adelman W. J., Jr, Palti Y. The effects of external potassium and long duration voltage conditioning on the amplitude of sodium currents in the giant axon of the squid, Loligo pealei. J Gen Physiol. 1969 Nov;54(5):589–606. doi: 10.1085/jgp.54.5.589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong C. M., Bezanilla F. Inactivation of the sodium channel. II. Gating current experiments. J Gen Physiol. 1977 Nov;70(5):567–590. doi: 10.1085/jgp.70.5.567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong C. M., Bezanilla F., Rojas E. Destruction of sodium conductance inactivation in squid axons perfused with pronase. J Gen Physiol. 1973 Oct;62(4):375–391. doi: 10.1085/jgp.62.4.375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong C. M., Gilly W. F. Fast and slow steps in the activation of sodium channels. J Gen Physiol. 1979 Dec;74(6):691–711. doi: 10.1085/jgp.74.6.691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong C. M. Sodium channels and gating currents. Physiol Rev. 1981 Jul;61(3):644–683. doi: 10.1152/physrev.1981.61.3.644. [DOI] [PubMed] [Google Scholar]
- Barchi R. L. Biochemical studies of the excitable membrane sodium channel. Int Rev Neurobiol. 1982;23:69–101. doi: 10.1016/s0074-7742(08)60622-4. [DOI] [PubMed] [Google Scholar]
- Bezanilla F., Armstrong C. M. Inactivation of the sodium channel. I. Sodium current experiments. J Gen Physiol. 1977 Nov;70(5):549–566. doi: 10.1085/jgp.70.5.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bezanilla F., Armstrong C. M. Properties of the sodium channel gating current. Cold Spring Harb Symp Quant Biol. 1976;40:297–304. doi: 10.1101/sqb.1976.040.01.030. [DOI] [PubMed] [Google Scholar]
- Bezanilla F., Taylor R. E., Fernández J. M. Distribution and kinetics of membrane dielectric polarization. 1. Long-term inactivation of gating currents. J Gen Physiol. 1982 Jan;79(1):21–40. doi: 10.1085/jgp.79.1.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brodwick M. S., Eaton D. C. Chemical modification of excitable membranes. Prog Clin Biol Res. 1982;79:51–72. [PubMed] [Google Scholar]
- Chandler W. K., Meves H. Slow changes in membrane permeability and long-lasting action potentials in axons perfused with fluoride solutions. J Physiol. 1970 Dec;211(3):707–728. doi: 10.1113/jphysiol.1970.sp009300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenwick E. M., Marty A., Neher E. Sodium and calcium channels in bovine chromaffin cells. J Physiol. 1982 Oct;331:599–635. doi: 10.1113/jphysiol.1982.sp014394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernández J. M., Bezanilla F., Taylor R. E. Distribution and kinetics of membrane dielectric polarization. II. Frequency domain studies of gating currents. J Gen Physiol. 1982 Jan;79(1):41–67. doi: 10.1085/jgp.79.1.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fishman H. M., Moore L. E., Poussart D. Ion movements and kinetics in squid axon II. Spontaneous electrical fluctuations. Ann N Y Acad Sci. 1977 Dec 30;303:399–428. [PubMed] [Google Scholar]
- French R. J., Horn R. Sodium channel gating: models, mimics, and modifiers. Annu Rev Biophys Bioeng. 1983;12:319–356. doi: 10.1146/annurev.bb.12.060183.001535. [DOI] [PubMed] [Google Scholar]
- Fukushima Y. Identification and kinetic properties of the current through a single Na+ channel. Proc Natl Acad Sci U S A. 1981 Feb;78(2):1274–1277. doi: 10.1073/pnas.78.2.1274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman L., Hahin R. Initial conditions and the kinetics of the sodium conductance in Myxicola giant axons. II. Relaxation experiments. J Gen Physiol. 1978 Dec;72(6):879–898. doi: 10.1085/jgp.72.6.879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., HUXLEY A. F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol. 1952 Aug;117(4):500–544. doi: 10.1113/jphysiol.1952.sp004764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamill O. P., Marty A., Neher E., Sakmann B., Sigworth F. J. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981 Aug;391(2):85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Horn R., Lange K. Estimating kinetic constants from single channel data. Biophys J. 1983 Aug;43(2):207–223. doi: 10.1016/S0006-3495(83)84341-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horn R., Patlak J., Stevens C. F. Sodium channels need not open before they inactivate. Nature. 1981 Jun 4;291(5814):426–427. doi: 10.1038/291426a0. [DOI] [PubMed] [Google Scholar]
- Horn R., Patlak J., Stevens C. F. The effect of tetramethylammonium on single sodium channel currents. Biophys J. 1981 Nov;36(2):321–327. doi: 10.1016/S0006-3495(81)84734-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matteson D. R., Armstrong C. M. Evidence for a population of sleepy sodium channels in squid axon at low temperature. J Gen Physiol. 1982 May;79(5):739–758. doi: 10.1085/jgp.79.5.739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neher E., Stevens C. F. Conductance fluctuations and ionic pores in membranes. Annu Rev Biophys Bioeng. 1977;6:345–381. doi: 10.1146/annurev.bb.06.060177.002021. [DOI] [PubMed] [Google Scholar]
- Nonner W. Relations between the inactivation of sodium channels and the immobilization of gating charge in frog myelinated nerve. J Physiol. 1980 Feb;299:573–603. doi: 10.1113/jphysiol.1980.sp013143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nonner W., Spalding B. C., Hille B. Low intracellular pH and chemical agents slow inactivation gating in sodium channels of muscle. Nature. 1980 Mar 27;284(5754):360–363. doi: 10.1038/284360a0. [DOI] [PubMed] [Google Scholar]
- Oxford G. S. Some kinetic and steady-state properties of sodium channels after removal of inactivation. J Gen Physiol. 1981 Jan;77(1):1–22. doi: 10.1085/jgp.77.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oxford G. S., Wu C. H., Narahashi T. Removal of sodium channel inactivation in squid giant axons by n-bromoacetamide. J Gen Physiol. 1978 Mar;71(3):227–247. doi: 10.1085/jgp.71.3.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patlak J., Horn R. Effect of N-bromoacetamide on single sodium channel currents in excised membrane patches. J Gen Physiol. 1982 Mar;79(3):333–351. doi: 10.1085/jgp.79.3.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quandt F. N., Narahashi T. Modification of single Na+ channels by batrachotoxin. Proc Natl Acad Sci U S A. 1982 Nov;79(21):6732–6736. doi: 10.1073/pnas.79.21.6732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rae J. L., Levis R. A. Patch Clamp Recordings from the Epithelium of the Lens Obtained using Glasses Selected for Low Noise and Improved Sealing Properties. Biophys J. 1984 Jan;45(1):144–146. doi: 10.1016/S0006-3495(84)84142-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogart R. Sodium channels in nerve and muscle membrane. Annu Rev Physiol. 1981;43:711–725. doi: 10.1146/annurev.ph.43.030181.003431. [DOI] [PubMed] [Google Scholar]
- Rudy B. Slow inactivation of the sodium conductance in squid giant axons. Pronase resistance. J Physiol. 1978 Oct;283:1–21. doi: 10.1113/jphysiol.1978.sp012485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sachs F., Neil J., Barkakati N. The automated analysis of data from single ionic channels. Pflugers Arch. 1982 Dec;395(4):331–340. doi: 10.1007/BF00580798. [DOI] [PubMed] [Google Scholar]
- Sakmann B., Patlak J., Neher E. Single acetylcholine-activated channels show burst-kinetics in presence of desensitizing concentrations of agonist. Nature. 1980 Jul 3;286(5768):71–73. doi: 10.1038/286071a0. [DOI] [PubMed] [Google Scholar]
- Schwarz W. Temperature experiments on nerve and muscle membranes of frogs. Indications for a phase transition. Pflugers Arch. 1979 Oct;382(1):27–34. doi: 10.1007/BF00585900. [DOI] [PubMed] [Google Scholar]
- Sigworth F. J. Covariance of nonstationary sodium current fluctuations at the node of Ranvier. Biophys J. 1981 Apr;34(1):111–133. doi: 10.1016/S0006-3495(81)84840-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigworth F. J., Neher E. Single Na+ channel currents observed in cultured rat muscle cells. Nature. 1980 Oct 2;287(5781):447–449. doi: 10.1038/287447a0. [DOI] [PubMed] [Google Scholar]
- Sigworth F. J. The variance of sodium current fluctuations at the node of Ranvier. J Physiol. 1980 Oct;307:97–129. doi: 10.1113/jphysiol.1980.sp013426. [DOI] [PMC free article] [PubMed] [Google Scholar]


