Abstract
Cells respond to proliferation with increased accumulation of biotin, suggesting that proliferation enhances biotin demand. Here we determined whether peripheral blood mononuclear cells (PBMC) increase biotin uptake at specific phases of the cell cycle, and whether biotin is utilized to increase biotinylation of carboxylases. Biotin uptake was quantified in human PBMC that were arrested chemically at specific phases of the cell cycle, i.e., biotin uptake increased in the G1 phase of the cycle [658 ± 574 amol biotin/(106 cells × 30 min)] and remained increased during phases S, G2, and M compared with quiescent controls [200 ± 62 amol biotin/(106 cells × 30 min)]. The abundance of the sodium-dependent multivitamin transporter (SMVT, which transports biotin) was similar at all phases of the cell cycle, suggesting that transporters other than SMVT or splicing variants of SMVT may account for the increased biotin uptake observed in proliferating cells. Activities of biotin-dependent 3-methylcrotonyl-CoA carboxylase and propionyl-CoA carboxylase were up to two times greater in proliferating PBMC compared with controls. The abundance of mRNA encoding 3-methylcrotonyl-CoA carboxylase and propionyl-CoA carboxylase paralleled carboxylase activities, suggesting that PBMC respond to proliferation with increased expression of genes encoding carboxylases. Similarly, expression of the gene encoding holocarboxylase synthetase (which catalyzes binding of biotin to carboxylases) increased in response to proliferation, suggesting that cellular capacity to biotinylate carboxylases was increased. In summary, these findings suggest that PBMC respond to proliferation with increased biotin uptake early in the cell cycle, and that biotin is utilized to increase activities of two of the four biotin-requiring carboxylases.
Keywords: biotin, carboxylase, cell cycle, peripheral blood mononuclear cells, transport, humans
In mammals, biotin serves as a covalently bound coenzyme for the following four biotin-dependent carboxylases: acetyl-CoA carboxylases (EC 6.4.1.2); pyruvate carboxylase (EC 6.4.1.1); propionyl-CoA carboxylase (PCC,4 EC 6.4.1.3); and 3-methylcrotonyl-CoA carboxylase (MCC, EC 6.4.1.4) (1). The attachment of biotin to an є-amino group of a lysine residue in the four apocarboxylases is catalyzed by holocarboxylase synthetase (EC 6.3.4.10). Biotin-dependent carboxylases catalyze essential steps in gluconeogenesis, synthesis of fatty acids, metabolism of odd-chain fatty acids and metabolism of some amino acids (1).
Biotin uptake into human cells is mediated by the sodium-dependent multivitamin transporter (SMVT), which binds biotin, pantothenic acid and lipoic acid with similar affinity (2). Circumstantial evidence has been provided that transporters other than SMVT might also account for some cellular biotin uptake (3,4). In previous studies, we showed that peripheral blood mononuclear cells (PBMC) respond to proliferation with a five-fold increase of biotin uptake (5). Theoretically, PBMC may increase biotin uptake to meet increased demand for this coenzyme of the biotin-dependent carboxylases. Currently, whether biotin uptake increases at an early phase of the cell cycle (e.g., G1) or at a late phase (e.g., M) is unknown.
Increased biotin demand of proliferating PBMC is likely to be physiologically important. PBMC represent a heterogeneous population of immune cells (T cells, B cells and various granulocytes), which mediate cellular and humoral immune responses (6). Some PBMC (T and B cells) proliferate rapidly after either antigenic or mitogenic stimulation. If the increased biotin demand of proliferating PBMC is not met, this may reduce proliferation rates. For example, biotin deficiency in mice causes reduced proliferation of splenocytes (7) and incubation of mouse splenocytes in biotin-deficient medium reduces generation of cytotoxic T lymphocytes (8).
In the present study, we addressed the following questions: 1) Does biotin uptake increase in specific phases of the cell cycle? If so, in what phases? 2) Is the increased uptake of biotin mediated by increased expression of the SMVT gene at specific phases of the cell cycle? 3) Is the increased biotin uptake associated with increased biotinylation of carboxylases? 4) Is there increased expression of genes encoding biotin-dependent carboxylases and holocarboxylase synthetase in proliferating PBMC?
SUBJECTS AND METHODS
Subjects
Healthy adults (n = 8 men, 4 women), aged 25–55 y participated in this study. Eleven subjects were Caucasian; one man was African-American. Subjects were nonsmokers and did not take any vitamin supplements for at least 2 wk before the study began. Some drugs (anticonvulsants) may interfere with biotin metabolism (9); thus, individuals undergoing anticonvulsant treatment were not eligible for study participation. This study was approved by the Institutional Review Boards for the Protection of Human Subjects at the University of Nebraska-Lincoln and the University of Arkansas for Medical Sciences. Informed written consent was obtained from each subject.
PBMC culturing for carboxylase activity studies
A heparinized blood sample (~150 mL) was collected from each subject. PBMC were isolated aseptically from blood as previously described (5). PBMC (4 × 109 cells/L) were suspended in RPMI-1640 containing 10% (v/v) autologous plasma, 100,000 IU/L penicillin and 100,000 μg/L streptomycin. After a baseline aliquot of the suspension was collected (denoted “day 0”), concanavalin A (20 mg/L final concentration) or pokeweed lectin (2.0 mg/L) was added to the medium to stimulate proliferation of PBMC. At timed intervals (1, 2 and 3 d after addition of concanavalin A or pokeweed lectin), aliquots were collected and used for determination of activities of biotin-dependent carboxylases as described below. Cell densities (cells/L) were determined by using a hemocytometer (5).
Carboxylase activities
Analysis of PCC and MCC activities in PBMC was based on the incorporation of [14C]bicarbonate into organic acids; unincorporated [14C]bicarbonate was removed by acidification and evaporation. Aliquots of PBMC suspension (2 mL) were centrifuged for 10 min at 2260 μg; the supernatants were discarded and the PBMC pellets were suspended in 90 μL of homogenization buffer as described previously (10). After vortexing, 25 μL of 0.5% Triton X-100 was added to lyse the cells. Complete lysis of cells by this procedure was confirmed by light microscopy.
Activities of PCC and MCC were determined as described previously with the following minor modifications (10). Briefly, lysed PBMC were incubated with carboxylase substrate (propionyl-CoA or 3-methylmalonyl-CoA), [14C]bicarbonate, and cofactors. After incubation, unbound [14C]bicarbonate was volatilized by addition of perchloric acid, and samples were dried. Samples were resuspended in scintillation fluid, and the bound [14C]bicarbonate was quantified by liquid scintillation counting.
Pilot studies provided evidence that activities of two biotin-dependent carboxylases, acetyl-CoA carboxylase and pyruvate carboxylase, do not reach a detectable level in quiescent and proliferating PBMC. Thus, acetyl-CoA carboxylase and pyruvate carboxylase were not studied further.
Proliferation rate
Uptake of [3H]thymidine (specific radioactivity 1.3 TBq/mmol; ICN, Irvine, CA) into PBMC was used to determine proliferation rates; [3H]thymidine uptake was measured as previously described (5).
PBMC culturing for biotin transport studies
Isolation and culture of PBMC was conducted as described above with the following modifications. PBMC were cultured with 20 mg/L concanavalin A at 37°C for 30 h to induce cell proliferation; quiescent controls were incubated without concanavalin A. Then one of the following chemicals was added to the proliferating PBMC and incubation was continued for 16 h to arrest the cells at specific phases of the cell cycle: 100 nmol/L (final concentration) wortmannin to cause arrest in G1 phase (11); 118 μmol/L aphidicolin to cause arrest in S phase (12); 1 μmol/L okadaic acid to cause arrest in G2 phase (13–15); or 5 μmol/L colchicine to cause arrest in M phase (16). Then PBMC were used for biotin transport studies and for analysis of SMVT gene expression.
Biotin transport
Rates of biotin transport into PBMC were determined using [3H]biotin at a physiologic concentration (475 pmol/L) as described previously (3). The abundance of SMVT protein in cells was determined by Western blotting, using a polyclonal antibody to human SMVT as described previously (17). The abundance of mRNA encoding SMVT was determined as described below.
Gene expression analysis by polymerase chain reaction (PCR)
The abundance of mRNA encoding PCC, MCC and holocarboxylase synthetase was determined in PBMC that had been cultured with pokeweed lectin for 3 d; these PBMC represent a mixed population of proliferating cells from various phases of the cell cycle. Quiescent controls were incubated without mitogen. The abundance of mRNA encoding SMVT was determined in PBMC that were arrested chemically at various phases of the cell cycle as described above.
Total RNA was extracted from PBMC by the RNeasy mini-kit according to the manufacturer’s instructions (Qiagen, Valencia, CA). The amount of RNA was quantified spectrophotometrically at 260 nm. RNA from each sample (1μg) was reverse transcribed using the Smart PCR cDNA synthesis kit (Clontech, Palo Alto, CA) and Reverse Transcriptase Superscript II RT (Life Technologies, Gaithersburg, MD). The ssDNA was amplified by PCR using the following oligonucleotide primers for MCC, PCC, holocarboxylase synthetase, SMVT and the housekeeping gene glyceraldehyde-3-phosphate dehydrogenase (Integrated DNA Technologies, Coralville, IA): 5′-GAA TAT ATG CAG AAG ATC CTA GCA ATA-3′ and 5′-ATG TGC CTG AAG AAG TGA AAA GTG TCG-3′ for human 3-methylcrotonyl-CoA carboxylase (18); 5′-TCC GTG AGT GCC ACG ATC CCA GTG ACC-3′ and 5′-GAG CTC ATG ACA TCA TAG GCA CCT CCA-3′ for human propionyl-CoA carboxylase (19); 5′-ATG GAA GAT AGA CTC CAC ATG GAT A-3′ and 5′-CCG CCG TTT GGG GAG GAT GAG GTT T-3′ for human holocarboxylase synthetase (accession number XM 009757); 5′-GTG TTC CAG ACA CTG GTC ATG TTC CTC-3′ and 5′-AGC CAA CAG GCC CAC AAC AGC ACC AGG -3′ for human SMVT (2); and 5′-ACC ACA GTC CAT GCC ATC ACT GCC ACC-3′ and 5′-TCC ACC ACC CTG TTG CTG TAG CCA AAT-3′ for human glyceraldehyde-3-phosphate dehydrogenase (20).
The amount of DNA produced by PCR parallels the abundance of mRNA (template) in the starting material (cells). PCR was performed using the following temperatures and times per cycle: 94°C for 1 min (denaturating), 55°C for 1 min (annealing), and 72°C for 2 min (extending). The PCR-amplified DNA was collected at timed intervals after 15, 18, 21, 25, 30, 35, 40, 50, and 60 cycles. Aliquots (10 μL) of each sample were chromatographed using 1.5% agarose gels. DNA on gels was stained with ethidium bromide, visualized and digitized using FOTO/Analyst PC image, version 2.0 (Fotodyne, Hartland, WI), and quantified using the Molecular Analyst software package, version 1.5 (Bio-Rad Laboratories, Hercules, CA). Only values from within the linear phase of PCR amplification were considered for data analysis.
Statistics
Homogeneity of variances among groups was tested using Bartlett’s test (21). When variances were heterogenous, data were log transformed before further statistical testing. Significance of differences among groups was tested by one-way ANOVA. Fisher’s Protected Least Significant Difference procedure was used for post-hoc testing (21). StatView 5.0.1 (SAS Institute; Cary, NC) was used to perform all calculations. Differences were considered significant if P < 0.05. Data are expressed as means ± 1 SD.
RESULTS
Expression of biotin transporters
Uptake of biotin into PBMC increased early in the cell cycle and remained increased during later phases of the cycle. For example, the rate of biotin uptake into PBMC in G1 phase was 658 ± 574 amol/(106 cells × 30 min), compared with 200 ± 62 amol/(106 cells × 30 min) in quiescent controls (Fig. 1). Rates of biotin uptake were 466 –528 amol/(106 cells × 30 min) in PBMC in phases S, G2 and M of the cycle, respectively. These data are consistent with increased expression of biotin transporters throughout the cell cycle compared with quiescent cells.
FIGURE 1.

Uptake of biotin into proliferating human peripheral blood mononuclear cells (PBMC) increased early in the cell cycle. PBMC were isolated from healthy adults and cultured with 20 mg/L concanavalin A for 30 h to induce proliferation; controls were cultured without concanavalin A. Next, proliferating PBMC were arrested chemically at various phases of the cell cycle (see text) and rates of biotin transport were determined. Data are expressed as means ± sd, n = 5. *Different from quiescent controls, P < 0.01.
Next, we determined whether expression of SMVT (as judged by mRNA abundance) increased at a specific phase of the cell cycle in PBMC; abundance of mRNA encoding SMVT was determined by quantitative PCR. The abundance of mRNA encoding SMVT increased early in the cell cycle and remained increased during later phases of the cell cycle (Fig. 2A). For example, the abundance of mRNA encoding SMVT was 29 ± 44 absorbance units in PBMC in G1 phase of the cycle, compared with 3 ± 1 absorbance units in quiescent controls. The abundance of mRNA encoding SMVT was 26 –32 absorbance units in PBMC in S, G2 and M phase of the cycle. The abundance of mRNA encoding the housekeeping gene glyceraldehyde-3-phosphate dehydrogenase was similar in quiescent and proliferating PBMC, suggesting that differences observed for mRNA encoding SMVT were not an artifact (Fig. 2A; P > 0.05). These data are consistent with increased expression of the SMVT gene in proliferating cells.
FIGURE 2.

Expression of the sodium-dependent multivitamin transporter (SMVT) at various phases of the cell cycle in human peripheral blood mononuclear cells (PBMC). Cells were isolated from healthy adults and cultured with 20 mg/L concanavalin A for 30 h to induce proliferation; controls were cultured without concanavalin A. Next, proliferating PBMC were arrested chemically at various phases of the cell cycle (see text). Panel A: The abundance of mRNA encoding SMVT was determined by polymerase chain reaction and gel densitometry. Data are expressed as means ± sd, n = 5. *,**Different from quiescent controls,*P < 0.05, **P < 0.01. Inserts show representative examples of mRNA encoding SMVT and the housekeeping gene glyceraldehyde-3-phosphate dehydrogenase (G3PDH). Panel B: The abundance of SMVT protein was determined in PBMC at various phases of the cell cycle by using an antibody to human SMVT.
Next we quantified SMVT protein at various phases of the cell cycle. Increased abundance of SMVT mRNA was not associated with increased abundance of SMVT protein, as judged by probing cell extracts with anti-SMVT antibody. The abundance of SMVT protein was similar in quiescent and proliferating PBMC at various phases of the cell cycle (Fig. 2B), suggesting that transporters other than SMVT or splicing variants of SMVT may account for the increased biotin uptake observed in proliferating cells.
Carboxylase activities
PBMC responded to mitogen-induced proliferation with increased carboxylase activities. Three days after addition of pokeweed lectin to the culture medium, activity of MCC was 2.8 ± 0.8 times the activity in quiescent controls (Fig. 3). Similarly, 3 d after addition of concanavalin A to the culture medium, activity of MCC was 3.1 ± 1.0 times control values. Thus, stimulation of carboxylase activity in proliferating PBMC was not restricted to a particular mitogen.
FIGURE 3.

Activity of 3-methylcrotonyl-CoA carboxylase (MCC) in cultured human peripheral blood mononuclear cells (PBMC). Cells were isolated from healthy adults and cultured in mitogen-containing medium (2.0 mg/L pokeweed lectin; 20 mg/L concanavalin A) for up to 3 d (d 0 = before addition of mitogen). At timed intervals, aliquots were collected to measure MCC activity. Data are expressed as means ± sd, n = 7. *Different from unstimulated cells on d 0, P < 0.01.
Activity of PCC in PBMC also increased in response to mitogen-stimulated proliferation. However, the increase of activity was less striking than the increase observed for MCC. Three days after addition of pokeweed lectin to the culture medium, activity of PCC was 1.5 ± 0.5 times the activity in quiescent controls (Fig. 4). Stimulation with concanavalin A did not produce a significant increase; 3 d after addition of concanavalin A to medium, activity of PCC was 1.2 ± 0.2 times control values. These findings are consistent with the hypothesis that proliferating PBMC accumulate biotin to increase activities of biotin-dependent carboxylases.
FIGURE 4.

Activity of propionyl-CoA carboxylase (PCC) in cultured human peripheral blood mononuclear cells (PBMC). Cells were isolated from healthy adults and cultured in mitogen-containing medium (2.0 mg/L pokeweed lectin; 20 mg/L concanavalin A) for up to 3 d (d 0 = before addition of mitogen). At timed intervals, aliquots were collected to measure PCC activity. Data are expressed as means ± sd, n = 7. *Different from unstimulated cells on d 0, P < 0.05.
Thymidine uptake
Proliferation of mitogen-stimulated PBMC was confirmed by measuring cellular [3H]thymidine uptake. PBMC responded to mitogen stimulation with a >100-fold increase of thymidine uptake (Fig. 5). Peak thymidine uptake was achieved 48 –72 h after addition of mitogen to the medium. The time course of thymidine uptake in PBMC approximately paralleled the time course of activities of biotin-dependent carboxylases (Figs. 3, 4).
FIGURE 5.

Uptake of [3H]thymidine into cultured human peripheral blood mononuclear cells (PBMC). Cells were isolated from healthy adults and cultured in mitogen-containing medium (2.0 mg/L pokeweed lectin; 20 mg/L concanavalin A) for up to 3 d (d 0 = before addition of concanavalin A). At timed intervals, aliquots were collected to measure [3H]thymidine uptake. Data are expressed as means ± sd, n = 7. Different from unstimulated cells on d 0, *P < 0.05; **P < 0.01.
mRNA encoding carboxylases and holocarboxylase synthetase
PBMC responded to mitogen-induced proliferation with increased levels of mRNA encoding MCC and PCC. The level of mRNA encoding MCC was ~1.3 times greater in PBMC stimulated with pokeweed lectin compared with quiescent controls (Fig. 6, upper panel). Note that mRNA levels were compared during the linear phase of PCR amplification, i.e., 40 PCR cycles for MCC. The level of mRNA encoding PCC was ~3.2 times greater in PBMC stimulated with pokeweed lectin compared with quiescent controls (Fig. 6, middle panel). It remains uncertain whether the observed increase of mRNA levels was due to increased transcription, or decreased mRNA degradation, or both.
FIGURE 6.
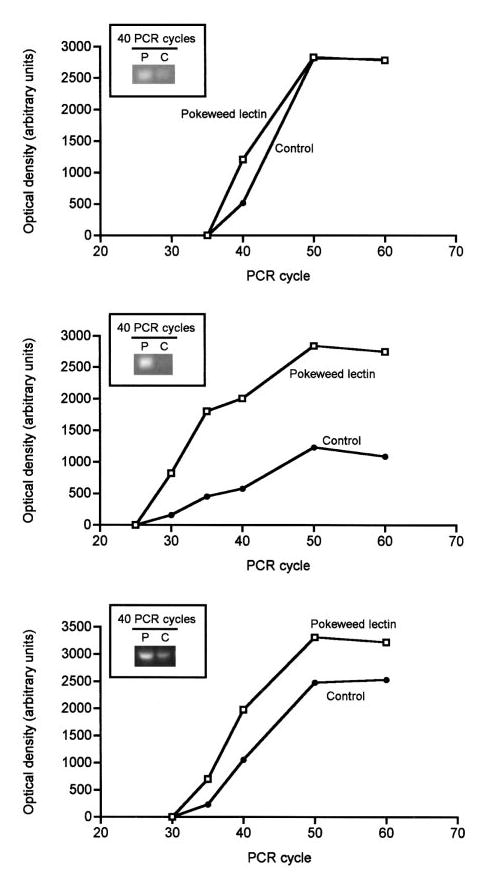
The abundance of mRNA encoding 3-methylcrotonyl-CoA carboxylase, propionyl-CoA carboxylase, and holocarboxylase synthetase in cultured human peripheral blood mononuclear cells (PBMC). Cells were isolated from healthy adults and cultured in mitogen-containing medium (2.0 mg/L pokeweed lectin) for 3 d to induce proliferation; controls were cultured without pokeweed lectin. The abundance of mRNA was determined by polymerase chain reaction; each data point is a single measure. Upper panel: 3-methylcrotonyl-CoA carboxylase; middle panel: propionyl-CoA carboxylase; lower panel: holocarboxylase synthetase. P, pokeweed lectin; C, control.
Holocarboxylase synthetase catalyzes the covalent binding of biotin to apocarboxylases to form holocarboxylases. The level of mRNA encoding holocarboxylase synthetase was ~1.4 times greater in PBMC stimulated with pokeweed lectin compared with quiescent controls during the linear phase of PCR amplification (Fig. 6, lower panel). This finding is consistent with the hypothesis that expression of the holocarboxylase synthetase gene increases in response to cell proliferation, mediating increased binding of biotin to carboxylases.
Finally, we quantified mRNA encoding the housekeeping gene glyceraldehyde-3-phosphate dehydrogenase in proliferating and quiescent PBMC. The level of mRNA encoding glyceraldehyde-3-phosphate dehydrogenase (1.0 ± 0.09 times control) in proliferating PBMC was not different from quiescent controls, suggesting that effects observed for mRNA encoding MCC, PCC and holocarboxylase synthetase were specific for these biotin-dependent pathways.
DISCUSSION
The results of this study are consistent with the hypotheses that human PBMC 1) increase expression of the SMVT gene early in the cell cycle; 2) increase biotin uptake early in the cell cycle; 3) utilize transporters other than SMVT or splicing variants of SMVT to mediate some of the cellular accumulation of biotin; 4) accumulate sufficient biotin to support increased activities of MCC and PCC; and 5) increase expression of genes encoding MCC, PCC and holocarboxylase synthetase.
Transport rates of biotin in proliferating PBMC in G1, S, G2 and M phase of the cell cycle are about three times the transport rates in quiescent controls (G0 phase). This is consistent with previous observations using PBMC that were not synchronized with regard to the phase of the cell cycle, i.e., rates of biotin uptake in proliferating PBMC were three to seven times the rates of biotin uptake in quiescent PBMC (5). The cellular volume of proliferating PBMC is about twice the volume of quiescent controls (22). This suggests that the abundance of biotin transporters per cell might be greater in proliferating PBMC compared with quiescent controls, but that the transporter density (number of transporters/cell surface area) is similar in proliferating and quiescent PBMC due to greater cell surface area in proliferating cells.
The present study suggests that expression of the SMVT gene increases early in the cell cycle (G1) and remains increased during later phases of the cycle (S, G2, and M), as judged by mRNA abundance. In contrast, abundance of SMVT protein was similar in quiescent and proliferating PBMC. To explain these apparently conflicting findings, we propose that splicing variants of SMVT may account for some biotin uptake. We have observed that the antibody that was used to probe SMVT binds at least two proteins, i.e., the 69-kDa SMVT and a smaller protein with a molecular weight of 48 kDa (17). Splicing variants of SMVT remain to be characterized.
This study provides evidence that proliferating PBMC accumulate sufficient biotin to support increased activities of biotin-dependent MCC and PCC. Presumably, some of the carboxylase-dependent metabolic pathways are important for cell proliferation. The percentage increase of enzyme activity in proliferating PBMC was greater for MCC than for PCC. The control mechanisms leading to disproportionate increases in the activities of the two enzymes are unknown. Both MCC and PCC are located in the same cellular compartment, i.e., the mitochondria (23). Hence, the two carboxylases should have equal access to the coenzyme biotin.
It is possible that the greater increase of MCC compared with PCC might be due to different half-lives of the two enzymes, i.e., PCC might be degraded more rapidly than MCC. Studies in rat liver and a mouse preadipocyte cell line suggest that the half-lives of various biotin-dependent carboxylases differ substantially. The half-life of pyruvate carboxylase is 28 –35 h (24), whereas that of acetyl-CoA carboxylase is 48 –59 h (25,26). To our knowledge, no studies have been conducted to determine half-lives of MCC and PCC in human PBMC.
In addition, holocarboxylase synthetase might have a greater affinity for MCC than for PCC, leading to preferential biotinylation of MCC over PCC. To our knowledge, this hypothesis has not yet been tested.
The present study is consistent with the hypothesis that PBMC respond to proliferation with increased synthesis of new apo-carboxylases (followed by biotinylation) rather than with increased biotinylation of existing apo-carboxylases. The abundance of mRNA encoding both MCC and PCC was greater in proliferating PBMC compared with quiescent controls, suggesting that increased expression of carboxylase genes occurs at the level of transcription rather than translation. The abundance of mRNA could be increased either by increased transcription or by decreased degradation of mRNA in response to proliferation.
Expression of the holocarboxylase synthetase gene increased in response to cell proliferation as judged by the abundance of mRNA encoding holocarboxylase synthetase. We speculate that the increased abundance of mRNA leads to increased activity of holocarboxylase synthetase in proliferating PBMC, mediating increased rates of biotinylation of carboxylases.
Biotinylation of proteins other than carboxylases might also generate increased demand for biotin in proliferating cells. For example, Wolf and co-workers have proposed an enzymatic mechanism leading to biotinylation of histones (27,28). We recently demonstrated that histones are biotinylated in vivo in human PBMC and that biotinylation of histones increases in response to proliferation (29).
Are the data presented here pertinent to human health? Cells in certain tissues exhibit high rates of proliferation to fulfill their normal function, e.g., cells in growing fetal tissues, intestinal mucosal cells and immune cells after antigenic stimulation. This study and previous studies (5) suggest that proliferating cells may have an increased demand for biotin. Biotin deficiency leads to decreased intracellular carboxylase activities (30 –32), fetal malformations (33,34) and impairment of immune function (7,35,36). Whether there is a causal link between reduced carboxylase activities, reduced rates of cell proliferation, and abnormal function and development of tissues remains to be investigated.
Footnotes
Presented in part at Experimental Biology, April 2000, San Diego, CA [Zempleni J. & Mock D. M. (2000) Lymphocytes increase biotin uptake during G1 phase of the cell cycle to increase biotinylation of histones. FASEB J. 14: A243 (abs.)].
Supported by National Institutes of Health grants DK 60447 and DK 36823, and the U.S. Department of Agriculture/National Research Initiative Competitive Grants Program project award 2001–35200-10187. This is a contribution of the University of Nebraska Agricultural Research Division, Lincoln, NE 68583, Journal Series No. 13570.
Abbreviations used: MCC, 3-methylcrotonyl-CoA carboxylase; PBMC, peripheral blood mononuclear cells; PCC, propionyl-CoA carboxylase; PCR, polymerase chain reaction; SMVT, sodium-dependent multivitamin transporter.
References
- 1.Zempleni J, Mock DM. Biotin biochemistry and human requirements. J Nutr Biochem. 1999;10:128 –138. doi: 10.1016/s0955-2863(98)00095-3. [DOI] [PubMed] [Google Scholar]
- 2.Wang H, Huang W, Fei YJ, Xia H, Fang-Yeng TL, Leibach FH, Devoe LD, Ganapathy V, Prasad PD. Human placental Na+-dependent multivitamin transporter. J Biol Chem. 1999;274:14875–14883. doi: 10.1074/jbc.274.21.14875. [DOI] [PubMed] [Google Scholar]
- 3.Zempleni J, Mock DM. Uptake and metabolism of biotin by human peripheral blood mononuclear cells. Am J Physiol. 1998;275:C382–C388. doi: 10.1152/ajpcell.1998.275.2.C382. [DOI] [PubMed] [Google Scholar]
- 4.Zempleni J, Mock DM. Human peripheral blood mononuclear cells: inhibition of biotin transport by reversible competition with pantothenic acid is quantitatively minor. J Nutr Biochem. 1999;10:427–432. doi: 10.1016/s0955-2863(99)00024-8. [DOI] [PubMed] [Google Scholar]
- 5.Zempleni J, Mock DM. Mitogen-induced proliferation increases biotin uptake into human peripheral blood mononuclear cells. Am J Physiol. 1999;276:C1079 –C1084. doi: 10.1152/ajpcell.1999.276.5.C1079. [DOI] [PubMed] [Google Scholar]
- 6.Klein, J. & Horejsi, V. (1997) Immunology, 2nd ed. Blackwell Science, Oxford, UK.
- 7.Báez-Saldaña A, Díaz G, Espinoza B, Ortega E. Biotin deficiency induces changes in subpopulations of spleen lymphocytes in mice. Am J Clin Nutr. 1998;67:431–437. doi: 10.1093/ajcn/67.3.431. [DOI] [PubMed] [Google Scholar]
- 8.Kung JT, MacKenzie CG, Talmage DW. The requirement for biotin and fatty acids in the cytotoxic T-cell response. Cell Immunol. 1979;48:100 –110. doi: 10.1016/0008-8749(79)90103-5. [DOI] [PubMed] [Google Scholar]
- 9.Krause KH, Berlit P, Bonjour JP. Vitamin status in patients on chronic anticonvulsant therapy. Int J Vitam Nutr Res. 1982;52:375–385. [PubMed] [Google Scholar]
- 10.Zempleni J, Trusty TA, Mock DM. Lipoic acid reduces the activities of biotin-dependent carboxylases in rat liver. J Nutr. 1997;127:1776 –1781. doi: 10.1093/jn/127.9.1776. [DOI] [PubMed] [Google Scholar]
- 11.Yano H, Nakanishi S, Kimura K, Hanai N, Saitoh Y, Fukui Y, Nonomura Y, Matsuda Y. Inhibition of histamine secretion by wortmannin through the blockade of phosphatidylinositol 3-kinase in RBL-2H3 cells. J Biol Chem. 1993;268:25846 –25856. [PubMed] [Google Scholar]
- 12.Dasso M, Newport JW. Completion of DNA replication is monitored by a feedback system that controls the initiation of mitosis in vitro: studies in Xenopus. Cell. 1990;61:811–823. doi: 10.1016/0092-8674(90)90191-g. [DOI] [PubMed] [Google Scholar]
- 13.Cohen P. The structure and regulation of protein phosphatases. Annu Rev Biochem. 1989;58:453–508. doi: 10.1146/annurev.bi.58.070189.002321. [DOI] [PubMed] [Google Scholar]
- 14.Haavik J, Schelling DL, Campbell DG, Andersson KK, Flatmark T, Cohen P. Identification of protein phosphatase 2A as the major tyrosine hydroxylase phosphatase in adrenal medulla and corpus striatum: evidence from the effects of okadaic acid. FEBS Lett. 1989;251:36 –42. doi: 10.1016/0014-5793(89)81424-3. [DOI] [PubMed] [Google Scholar]
- 15.Cohen P, Klumpp S, Schelling DL. An improved procedure for identifying and quantitating protein phosphatases in mammalian tissues. FEBS Lett. 1989;250:596 –600. doi: 10.1016/0014-5793(89)80803-8. [DOI] [PubMed] [Google Scholar]
- 16.Chalifour LE, Dakshinamurti K. The biotin requirement of human fibroblasts in culture. Biochim Biophys Res Commun. 1982;104:1047–1053. doi: 10.1016/0006-291x(82)91355-9. [DOI] [PubMed] [Google Scholar]
- 17.Griffin, J. B., Stanley, J. S. & Zempleni, J. (2002) Synthesis of a rabbit polyclonal antibody to the human sodium-dependent multivitamin transporter. Int. J. Vitam. Nutr. Res. (in press). [DOI] [PubMed]
- 18.Hillier L, Lennon G, Becker M, Bonaldo MF, Chiapelli B, Chissoe S, Dietrich N, DuBuque T, Favello A, Gish W, Hawkins M, Hultman M, Kucaba T, Lacy M, Le M, Le N, Mardis E, Moore B, Morris M, Parsons J, Prange C, Rifkin L, Rohlfing T, Schellenberg K, Soares MB, Tan F, Thierry-Meg J, Trevaskis E, Underwood K, Wohldmann P, Waterston R, Wilson R, Marra M. Generation and analysis of 280,000 human expressed sequence tags. Genome Res. 1996;6:807–828. doi: 10.1101/gr.6.9.807. [DOI] [PubMed] [Google Scholar]
- 19.Lamhonwah AM, Barankiewicz TJ, Willard HF, Mahuren DJ, Quan F, Gravel RA. Isolation of cDNA clones coding for the alpha and beta chains of human propionyl-CoA carboxylase: chromosomal assignments and DNA polymorphisms associated with PCCA and PCCB genes. Proc Natl Acad Sci USA. 1986;83:4864 –4868. doi: 10.1073/pnas.83.13.4864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Arcari P, Martinelli R, Salvatore F. The complete sequence of a full length cDNA for liver glyceraldehyde-3-phosphate dehydrogenase: evidence for multiple mRNA species. Nucleic Acids Res. 1984;12:9179 –9189. doi: 10.1093/nar/12.23.9179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.SAS Institute Inc. (1999) StatView Reference, 3rd ed. SAS Institute, Cary, NC.
- 22.Williams GT, Lau KMK, Coote JM, Johnstone AP. NAD metabolism and mitogen stimulation of human lymphocytes. Exp Cell Res. 1985;160:419 –426. doi: 10.1016/0014-4827(85)90189-2. [DOI] [PubMed] [Google Scholar]
- 23.Zempleni J. (2001) Biotin. In: Present Knowledge in Nutrition, 8th ed. (Bowman, B. A. & Russell, R. M., eds.), pp. 241–252. ILSI Press, Washington, DC.
- 24.Freytag SO, Utter MF. Regulation of the synthesis and degradation of pyruvate carboxylase in 3T3–L1 cells. J Biol Chem. 1983;258:6307–6312. [PubMed] [Google Scholar]
- 25.Majerus P, Kilburn E. Acetyl coenzyme A carboxylase. The roles of synthesis and degradation in regulation of enzyme levels in rat liver. J Biol Chem. 1969;244:6254 –6262. [PubMed] [Google Scholar]
- 26.Nakanishi S, Numa S. Purification of rat liver acetyl coenzyme A carboxylase and immunochemical studies on its synthesis and degradation. Eur J Biochem. 1970;16:161–173. doi: 10.1111/j.1432-1033.1970.tb01068.x. [DOI] [PubMed] [Google Scholar]
- 27.Hymes J, Fleischhauer K, Wolf B. Biotinylation of biotinidase following incubation with biocytin. Clin Chim Acta. 1995;233:39 –45. doi: 10.1016/0009-8981(94)05965-u. [DOI] [PubMed] [Google Scholar]
- 28.Hymes J, Fleischhauer K, Wolf B. Biotinylation of histones by human serum biotinidase: assessment of biotinyl-transferase activity in sera from normal individuals and children with biotinidase deficiency. Biochem Mol Med. 1995;56:76 –83. doi: 10.1006/bmme.1995.1059. [DOI] [PubMed] [Google Scholar]
- 29.Stanley JS, Griffin JB, Zempleni J. Biotinylation of histones in human cells: effects of cell proliferation. Eur J Biochem. 2001;268:5424 –5429. doi: 10.1046/j.0014-2956.2001.02481.x. [DOI] [PubMed] [Google Scholar]
- 30.Velazquez A, Zamudio S, Baez A, Murguia-Corral R, Rangel-Peniche B, Carrasco A. Indicators of biotin status: a study of patients on prolonged total parenteral nutrition. Eur J Clin Nutr. 1990;44:11–16. [PubMed] [Google Scholar]
- 31.Velazquez A, Teran M, Baez A, Gutierrez J, Rodriguez R. Biotin supplementation affects lymphocyte carboxylases and plasma biotin in severe protein-energy malnutrition. Am J Clin Nutr. 1995;61:385–391. doi: 10.1093/ajcn/61.2.385. [DOI] [PubMed] [Google Scholar]
- 32.Rodriguez-Melendez R, Cano S, Mendez ST, Velazquez A. Biotin regulates the genetic expression of holocarboxylase synthetase and mitochondrial carboxylases in rats. J Nutr. 2001;131:1909 –1913. doi: 10.1093/jn/131.7.1909. [DOI] [PubMed] [Google Scholar]
- 33.Watanabe T. Teratogenic effects of biotin deficiency in mice. J Nutr. 1983;113:574 –581. doi: 10.1093/jn/113.3.574. [DOI] [PubMed] [Google Scholar]
- 34.Watanabe T, Endo A. Teratogenic effects of avidin-induced biotin deficiency in mice. Teratology. 1984;30:91–94. doi: 10.1002/tera.1420300112. [DOI] [PubMed] [Google Scholar]
- 35.Petrelli F, Moretti P, Campanati G. Studies on the relationships between biotin and the behaviour of B and T lymphocytes in the guinea pig. Experientia. 1981;37:1204 –1206. doi: 10.1007/BF01989920. [DOI] [PubMed] [Google Scholar]
- 36.Kumar M, Axelrod AE. Cellular antibody synthesis in thiamin, riboflavin, biotin and folic acid-deficient rats. Proc Soc Exp Biol Med. 1978;157:421–423. doi: 10.3181/00379727-157-40068. [DOI] [PubMed] [Google Scholar]


