Abstract
The Belgian rotavirus strain B4106, isolated from a child with gastroenteritis, was previously found to have VP7 (G3), VP4 (P[14]), and NSP4 (A genotype) genes closely related to those of lapine rotaviruses, suggesting a possible lapine origin or natural reassortment of strain B4106. To investigate the origin of this unusual strain, the gene sequences encoding VP1, VP2, VP3, VP6, NSP1, NSP2, NSP3, and NSP5/6 were also determined. To allow comparison to a lapine strain, the 11 double-stranded RNA segments of a European G3P[14] rabbit rotavirus strain 30/96 were also determined. The complete genome similarity between strains B4106 and 30/96 was 93.4% at the nucleotide level and 96.9% at the amino acid level. All 11 genome segments of strain B4106 were closely related to those of lapine rotaviruses and clustered with the lapine strains in phylogenetic analyses. In addition, sequence analyses of the NSP5 gene of strain B4106 revealed that the altered electrophoretic mobility of NSP5, resulting in a super-short pattern, was due to a gene rearrangement (head-to-tail partial duplication, combined with two short insertions and a deletion). Altogether, these findings confirm that a rotavirus strain with an entirely lapine genome complement was able to infect and cause severe disease in a human child.
Group A rotaviruses (family Reoviridae) are important enteropathogens of humans and of a large variety of mammals and birds (11, 37). The rotavirus genome consists of 11 segments of double-stranded RNA (dsRNA) encoding six structural viral proteins (VP) and six nonstructural proteins (NSP) (37). A mature infectious rotavirus particle is composed of three concentric layers, consisting of a protein core, an inner protein capsid, and an outer protein capsid (84). The outer protein layer consists of VP4 and VP7, the two independent neutralization antigens of the virus, defining 26 P (protease-sensitive) and 15 G (glycoprotein) types, respectively (37, 55, 68, 69, 85). The G and P types are peculiarly distributed across the various animal species (88), suggesting host species barriers and restriction, although a number of unusual G and P types, regarded as animal-like strains, have been identified in humans in different parts of the world (85, 88).
Although rotaviruses infect particular species preferentially for which they have been defined as the homologous strains, heterologous rotavirus infections occur in both natural and experimental circumstances. Studies in the rabbit and mouse models have demonstrated that only homologous virus strains replicate efficiently and spread horizontally (21, 38, 44). Based on a Jennerian approach, animal strains that are naturally attenuated in humans have been exploited for the construction of candidate rotavirus vaccines for humans. In a number of field trials with such candidate rotavirus vaccines, the rhesus rotavirus (RRV) strain MMU18006 and the bovine strains NCDV, UK, and WC3 were shown to replicate to a lower extent in humans than in their homologous hosts but to induce immune responses (24, 25, 56, 98, 99). Conversely, a number of studies have also proven that, under experimental conditions, heterologous rotavirus strains can infect and/or induce diarrhea in a heterologous animal model. Human rotavirus strains (HRVs) have been shown to cause disease in several newborn animals (55). In the piglet model, virulent HRV strains induced diarrhea and viraemia, whereas attenuated HRV strains did not (7). In the rabbit model, RRV has been shown to replicate efficiently and transmit horizontally (21). Also, a pigeon rotavirus strain (PO-13) was shown to infect and cause diarrhea in mice (77).
Several genome segments have been implicated as potential determinants of rotavirus host range restriction and virulence by the analyses of natural or laboratory engineered reassortants (13, 14, 36, 47, 76). The study of naturally occurring heterologous rotavirus strains and of animal-human reassortant strains may provide a unique tool to understand how rotavirus cross the host-species barrier and may help to address the molecular determinants that control rotavirus host-species specificity and pathogenicity. Nevertheless, our understanding of the molecular basis of rotavirus species restriction and virulence is still hampered by the lack of a reverse genetic system for rotaviruses and the lack of sequencing data for entire genomes of rotavirus strains. From this perspective, projects of full-genome sequencing of humans and animal rotavirus strains would be very useful to characterize uncommon HRV strains and to address the role of each genome segment in host range restriction and virulence. Currently, only six rotavirus strains have been completely sequenced. The simian rotavirus strain SA11 (G3P[2]) and the avian rotavirus PO-13 (G7P[17]) have been sequenced and compared to other strains in their entirety (50, 75). From the HRV strains KU (G1P[8]) and TB-Chen (G2P[4]) and the bovine rotavirus strains RF (G6P[1]) and UK (G6P[5]) full genome sequences can be found in the GenBank database.
The HRV strain B4106 was isolated from a Belgian child affected by severe gastroenteritis that required hospital care (31). Strain B4106 displayed a supershort dsRNA electropherotype and G3P[14] specificity, all of which are uncommon features of HRVs. Furthermore, sequence and phylogenetic analyses revealed that strain B4106 possessed VP7, VP4, and NSP4 genes closely related to those of lapine rotavirus (LRV) strains, and thus a possible heterologous rotavirus infection of lapine origin or a natural human-lapine reassortant was suspected (31). In the present study we sequenced and analyzed the entire genomes of the lapine-like HRV strain B4106 and a LRV strain 30/96 (G3P[14]), isolated recently in Italy (69), in order to test whether strain B4106 is entirely lapine or a human-lapine reassortant virus.
MATERIALS AND METHODS
Rotavirus strains.
HRV strain B4106 was isolated from a 6-year-old boy who was admitted to the Gasthuisberg University Hospital in Leuven, Belgium, with nausea and severe diarrhea. The child had no previous history of traveling but lived in a rural environment where rabbits were frequently encountered (31). The source of the LRV strain 30/96 was described previously (69).
RNA extraction.
Viral RNA was extracted from fecal material of the HRV B4106 strain or from a preparation of the tissue culture-adapted LRV 30/96 strain using the QIAamp Viral RNA minikit (QIAGEN/Westburg, Leusden, The Netherlands) according to the manufacturer's instructions.
RT-PCR.
The extracted RNA was denatured at 97°C for 5 min, and reverse transcriptase PCR (RT-PCR) was carried out by using a QIAGEN OneStep RT-PCR kit (QIAGEN/Westburg). Primers for the amplification of gene segments VP1-3, VP6, NSP1-3, and NSP5 were developed based on alignments of sequences retrieved from GenBank (Table 1). The RT-PCR was carried out with an initial reverse transcription step of 30 min at 45°C, followed by PCR activation at 95°C for 15 min, 40 cycles of amplification (45 s at 94°C, 45 s at 45°C, and 2.5 min at 70°C for VP6, NSP1-3, and NSP5 and 45 s at 94°C, 45 s at 45°C, and 6 min at 70°C for VP1-3), with a final extension of 7 min at 70°C.
TABLE 1.
Primers designed for the amplification and sequencing of the VP1, VP2, VP3, VP6, NSP1, NSP2, NSP3, and NSP5 encoding gene segments of B4106 and 30/96a
| Gene | Primer name | Primer sequence |
|---|---|---|
| VP1 | GEN-VP1F | 5′-GGCTATTAAAGCTRTACAATGG-3′ |
| GEN-VP1R | 5′-GGTCACATCTAAGCGYTC-3′ | |
| VP2 | GEN-VP2F | 5′-GGCTATTRAAGGYTCAATGG-3′ |
| GEN-VP2R | 5′-GGTCATATCTCCACARTGG-3′ | |
| VP3 | GEN-VP3F | 5′-GGCTWTTAAAGCAGTACCA-3′ |
| GEN-VP3R | 5′-GGYCACATCATGACTAGTG-3′ | |
| VP6 | GEN-VP6F | 5′-GGCTTTWAAACGAAGTCTTC-3′ |
| GEN-VP6R | 5′-GGTCACATCCTCTCACT-3′ | |
| NSP1 | LAP-NSP1F | 5′-GGGCTTTTTTTTGAAAAGTC-3′ |
| LAP-NSP1R | 5′-GGGTTCACAGTATTTTGC-3′ | |
| NSP2 | GEN-NSP2F | 5′-GGCTTTTAAAGCGTCTCAG-3′ |
| GEN-NSP2R | 5′-GGTCACATAAGCGCTTTC-3′ | |
| NSP3 | GEN-NSP3F | 5′-GGCTTTTAATGCTTTTCAGTG-3′ |
| GEN-NSP3R | 5′-ACATAACGCCCCTATAGC-3′ | |
| NSP5 | GEN-NSP5F | 5′-GGCTTTTAAAGCGCTACAG-3′ |
| GEN-NSP5R | 5′-GGTCACAAAACGGGAGT-3′ |
All of the primers are strictly 5′ and 3′ coterminal, except for the primer GEN-NSP3R, which begins at nucleotide 5 starting from the 3′ end of the NSP3 gene. R = A or G, Y = C or T, and W = A or T.
Nucleotide sequencing.
The PCR amplicons were purified with the QIAquick PCR purification kit (QIAGEN/Westburg) and sequenced with the ABI Prism BigDye terminator cycle sequencing reaction kit (Applied Biosystems Group, Foster City, CA) on an ABI Prism 3100 automated sequencer (Applied Biosystems Group) as described previously (31).
Determination of the 5′ and 3′ terminal sequences.
To obtain the complete nucleotide sequence of strains B4106 and 30/96, the 5′ and 3′ terminal sequences of the 11 gene segments were determined by using the single-primer amplification method (65). Briefly, after RNA extraction, a modified amino-linked oligonucleotide (TGP-Linker [5′-PO4-TTCCTTATGCAGCTGATCACTCTGTGTCA-spacer-NH2-3′]) was ligated to the 3′ ends of both strands of the viral dsRNA with T4 RNA Ligase (Promega, Leiden, The Netherlands). RT-PCR with the primer TGP-3Out (5′-TGACACAGAGTGATCAGC-3′; complementary to the 3′ end of TGP-Linker) and appropriate gene-specific primers (based on the known internal sequences of each segment) was carried out. The following thermal cycling conditions were used: an initial reverse transcription step at 45°C for 30 min, PCR activation at 95°C for 15 min, 45 min during which the temperature was gradually lowered from 83 to 60°C (to allow the newly transcribed cDNA strands to anneal), 10 min at 72°C (to allow the DNA polymerase to repair the partial duplexes), 40 cycles of amplification (45 s at 94°C, 45 s at 45°C, and 1 min at 70°C), with a final extension of 7 min at 70°C. These amplified products were purified and sequenced as described above.
RNA and protein sequence analysis.
The chromatogram sequencing files were analyzed by using Chromas 2.23 (Technelysium, Queensland, Australia), and contigs were prepared by using SeqMan II (DNASTAR, Madison, WI). Nucleotide and protein sequence similarity searches were performed by using the National Center for Biotechnology Information (National Institutes of Health, Bethesda, MD) BLAST (basic local alignment search tool) server on GenBank database release 143.0 (4). Multiple sequence alignments were calculated by using ClustalX 1.81 (93). Sequences were manually edited in the GeneDoc version 2.6.002 alignment editor (80).
Phylogenetic analysis.
Phylogenetic and molecular evolutionary analyses were conducted by using the MEGA version 2.1 software (64), based on the nucleotide and deduced amino acid sequences of the different rotavirus genes segments available in GenBank. Genetic distances on the amino acid level were calculated by using the Poisson correction parameter. The dendrograms were constructed by using the neighbor-joining method.
Dot plot analysis.
The dot plot analysis was done by using the Pairwise FLAG version web 0.3 of the Biomedical Engineering Center at the Industrial Technology Research Institute in Taiwan (http://bioinformatics.itri.org.tw/prflag/prflag.php).
Sequence submission.
The complete nucleotide sequence data of the 11 gene segments of strain B4106 reported in the present study were deposited in GenBank under the accession numbers AY740731 to AY740741, whereas the sequences of the 11 genome segments of strain 30/96 were deposited under accession numbers DQ205221 to DQ205231.
RESULTS
Complete sequencing of the 11 dsRNA segments of the HRV strain B4106 and the LRV strain 30/96.
The partial sequences of the VP7, VP4, and NSP4 genes of the Belgian HRV B4106 and the Italian LRV 30/96 strains were determined previously (31, 69). To further investigate the relationship of HRV B4106 with LRVs, the complete genomes of HRV B4106 and LRV strains 30/96 were determined. The entire genome of B4106 (30/96) contained a total of 18,881 (18,504) nucleotides, encoding a total of 5,797 (5,797) amino acids. The overall identity of B4106 and 30/96 was 93.4% at the nucleotide level and 96.9% at the amino acid level (Table 2). When the individual sequences of B4106 and 30/96 were compared, sequence identities between 88.2 and 96.9% at the nucleotide level and between 91.4 and 99.5% at the amino acid level were found for all of the 11 genome segments. A comparison of these gene segments with two HRV strains (KU and TB-Chen) and other cognate mammalian gene segments is provided in Table 2. Due to the rearranged NSP5 gene, B4106 has a “super-short” electropherotype (31).
TABLE 2.
Number of nucleotides and deduced amino acids of the 11 gene segments of B4106 (30/96)a
| Protein | No. of nt: B4106 (30/96) | No. of aa: B4106 (30/96) | % nt similarity (B4106 to 30/96)b | % aa similarity (B4106 to 30/96) | % aa similarities between B4106 and KU and TB-Chen | % Minimum and maximum similarity between B4106 and other mammalian rotaviruses |
|---|---|---|---|---|---|---|
| VP1 | 3,302 (3,302) | 1,088 (1,088) | 94.5 | 98.1 | 88.8-96.8 | 88.8-98.1 |
| VP2 | 2,687 (2,687) | 880 (880) | 88.2 | 97.9 | 91.6-96.9 | 91.6-98.2 |
| VP3 | 2,591 (2,591) | 835 (835) | 91.5 | 96.1 | 80.8-81.8 | 80.8-96.1 |
| VP4 | 2,361 (2,361) | 776 (776) | 91.5 | 97.2 | 57.2-58.8 | 42.1-97.2 |
| VP6 | 1,356 (1,356) | 397 (397) | 96.9 | 99.5 | 90.4-97.2 | 89.0-99.5 |
| VP7 | 1,062 (1,062) | 326 (326) | 95.4 | 97.5 | 79.5-69.8 | 67.6-97.5 |
| NSP1 | 1,591 (1,590) | 492 (492) | 89.0 | 91.4 | <20 | <20.0-93.0 |
| NSP2 | 1,059 (1,059) | 317 (317) | 95.4 | 98.4 | 88.6-92.4 | 84.2-98.4 |
| NSP3 | 1,078 (1,078) | 313 (313) | 95.7 | 97.7 | 86.2-80.4 | 76.3-97.7 |
| NSP4 | 751 (751) | 175 (175) | 95.4 | 94.0 | 81.9-90.5 | 56.2-98.0 |
| NSP5 | 1,043 (667) | 198 (198) | 95.3* | 95.1 | 87.0-82.1 | 80.8-96.2 |
| Total | 18,881 (18,504) | 5,797 (5,797) | 93.4* | 96.9 |
The percent similarities at the nucleotide (nt) and amino acid (aa) levels between B4106 and 30/96, the percent similarities between B4106 and HRV strains KU and TB-Chen, and the minimum and maximum percent similarities between B4106 and other available mammalian rotavirus strains are presented.
*, For these calculations, the duplication in NSP5 of B4106 was omitted.
Structural viral proteins of B4106 and 30/96.
The structural viral proteins of the core, VP1, VP2, and VP3 of B4106 and 30/96 shared a high degree of amino acid identity (96.1 to 98.1%) with each other (Table 2). The core proteins showed conservation of the four RNA-dependent-RNA-polymerase motifs, including the GDD motif (VP1) (50, 54), and the basic region, two leucine zippers (amino acids 536 to 557 and amino acids 665 to 686), and the five consecutive blocks of conserved acidic and basic amino acid residues found in VP2 (63, 75) are also conserved.
Comparison of the complete B4106 and 30/96 VP4 amino acid sequences with other complete VP4 amino acid sequences showed an identity of 95.5 to 97.2% with P[14] LRV strains BAP-2, ALA, and C-11 isolated from rabbits in the United States (20). Several general features of VP4, such as the two or three conserved trypsin cleavage sites (5, 66), conserved cysteine and proline residues (82), and a proposed α2β1 integrin ligand site which might be involved in viral cell entry (27), are all conserved in B4106 and 30/96.
Analysis of the deduced amino acid sequence of the VP6 gene segment of B4106 and 30/96 in the region known to correlate with subgroup specificity shows conserved residues at positions 305-Ala, 310-Asn, 315-Glu, 339-Ser, 342-Met, and 348-Ser, all characteristic of subgroup I rotaviruses (53, 67, 91).
The VP7 amino acid sequence of strain B4106 is highly similar (95.8 to 97.5%) to only VP7 proteins of lapine G3 rotavirus strains 30/96, 308/01, 160/01, 229/01, and BAP-2 (found in combination with P[14] or P[22] genotypes) isolated in Italy or the United States (20, 69). When the VP7 antigenic regions A, B, C, and F (22, 35, 58) of strain B4106 are compared to strain 30/96, only two amino acid substitutions are found at residues 210 (Asn → Asp) and 211 (Thr → Ala).
Nonstructural viral proteins of HRV B4106 and LRV 30/96 strains.
The nonstructural rotavirus proteins are only found in virus-infected cells and are essential for rotavirus translation, replication, and packaging. The NSP1 protein of B4106 showed 91.1 to 93.0% identity at the amino acid level to other known LRV strains (30/96, C-11, BAP-2 and ALA) (Table 2 and data not shown), and the cysteine-rich putative zinc finger domains were highly conserved as in all mammalian rotaviruses (48, 101).
The amino acid sequences of the NSP2 and NSP3 proteins of HRV B4106 were highly similar (98.4 and 97.7%, respectively) to those of LRV strain 30/96 (Table 2). The NSP2 RNA-binding domain and four cysteines at residues 6, 8, 85, and 285, suggested to be involved in disulfide bonds (83), were also conserved. In NSP3, the consensus sequence (I/L)xxM(I/L)(S/T)xxG (73), also present in the RNA-binding proteins of orbiviruses (NS2) and reoviruses (σNS), and a hydrophobic heptad repeat region, involved in the oligomer forming properties of NSP3 (50, 87, 95), were also conserved. However, the proposed leucine zipper domain, with five leucines at amino acid residues 275, 281, 288, 295, and 302 (73), were absent in the NSP3 of both B4106 and 30/96 strains. Only one Leu at residue 302 was preserved, and the other four positions are, respectively, replaced by Ile, Phe, Val, and Thr for B4106 and Val, Phe, Val, and Thr for 30/96.
At the amino acid level, the viral enterotoxin NSP4 of strain B4106 showed 92.6 to 98.0% identity to LRV rotavirus strains classified in genogroup A, which includes strain 30/96 (23, 69). The three conserved hydrophobic domains (18) and the two glycosylation sites (12) in the N-terminal part of B4106 NSP4 were nearly identical to the homologous regions in the LRV strains ALA, BAP, BAP-2, and 30/96. The domain associated with the formation of NSP4 homotetramers (92), the enterotoxic and membrane destabilizing peptide (8, 94), the VP4 binding domain (6), and the inner capsid particle binding domain (92), all located in the C-terminal part of NSP4, were also highly similar between the two strains and with the homologous regions of LRVs ALA, BAP, and BAP-2.
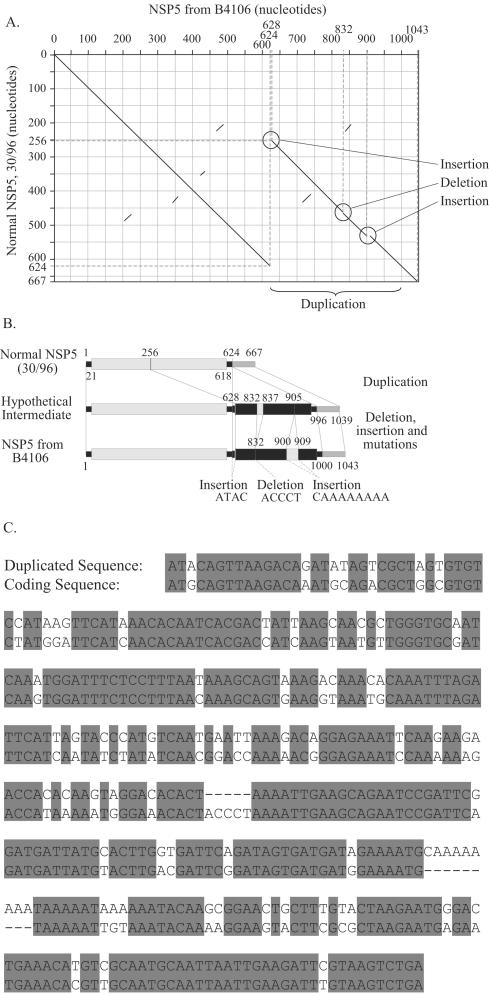
The gene segment encoding NSP5 and NSP6 of B4106 consisted of 1,043 nucleotides and contained two out-of-frame open reading frames (ORFs) of 198 (NSP5) and 96 (NSP6) amino acids, starting at nucleotides 22 and 80, respectively. The reason for this large size compared to the NSP5/NSP6 genes of 30/96 (667 nucleotides), SA11 (667 nucleotides), PO-13 (729 nucleotides), and most other mammalian rotaviruses was a partial duplication in the 3′ noncoding region of the B4106 NSP5 sequence (Fig. 1). To date, the NSP5 sequence has been determined for only one rotavirus (LRV strain ALA) other than B4106 and 30/96. A duplication of the NSP5 gene of ALA (42) was most likely generated by a mechanism similar to the duplication found in strain B4106. The amino acid identity of the NSP5 protein of B4106 was 95.1% identical to that of LRV 30/96 (Table 2).
FIG. 1.
Analysis of rearranged NSP5 gene of HRV strain B4106. (A) Dot plot analysis, comparing the nucleotide sequence of the LRV 30/96 NSP5 gene with the duplicated NSP5 gene of B4106. (B) Schematic representation of the duplication, the hypothetical intermediate, and the subsequent insertion and deletion events. For further details, see the text. (C) Comparison of the duplicated region with the homologous coding sequence of B4106.
Structure of duplicated NSP5 gene.
A dot plot comparison between the LRV 30/96 NSP5 gene segment and the rearranged NSP5 gene of strain B4106 clearly showed a duplication, a deletion, and insertions in the rearranged NSP5 gene of HRV strain B4106 (Fig. 1A). There were probably two major steps in the formation of this rearranged NSP5 gene, depicted in Fig. 1B. First, a region of 368 nucleotides starting at nucleotide 257 and comprising the last part of the NSP5 ORF and the first six nucleotides of the 3′ noncoding region (up to nucleotide 625) was duplicated. Four random nucleotides (ATAC) were inserted between the two copies, leaving the normal ORF intact (hypothetical intermediate). In a second step (most likely spread over time after the initial duplication event and the time of isolation of strain B4106), a deletion of five nucleotides (ACCCT) between nucleotides 832 and 837 and an insertion of nine nucleotides (CAAAAAAAA) between nucleotides 905 and 906 occurred. Many extra mutations have accumulated over time in the duplicated region compared to the original sequence, resulting in an identity of only 81%. A comparison of the duplicated region with the homologous coding sequence of B4106 is shown in Fig. 1C.
Phylogeny.
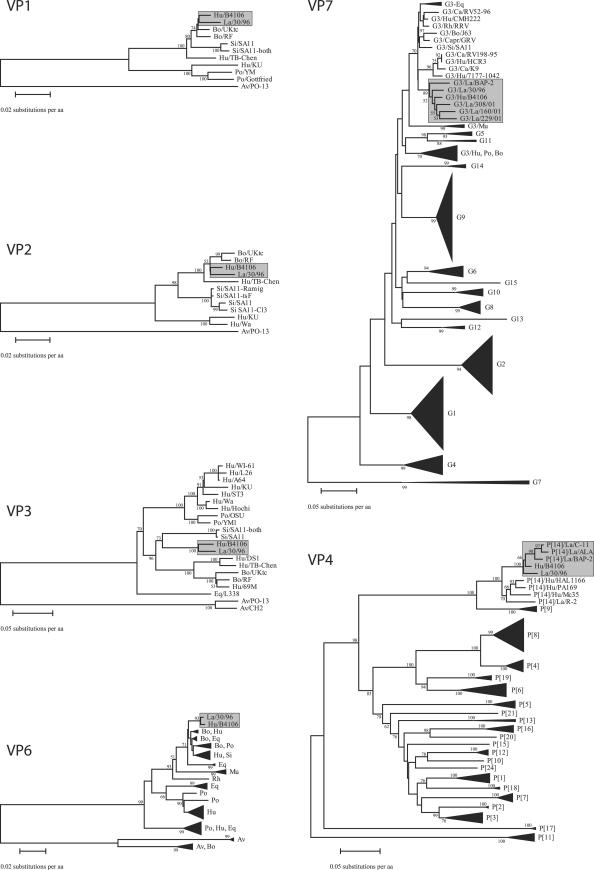
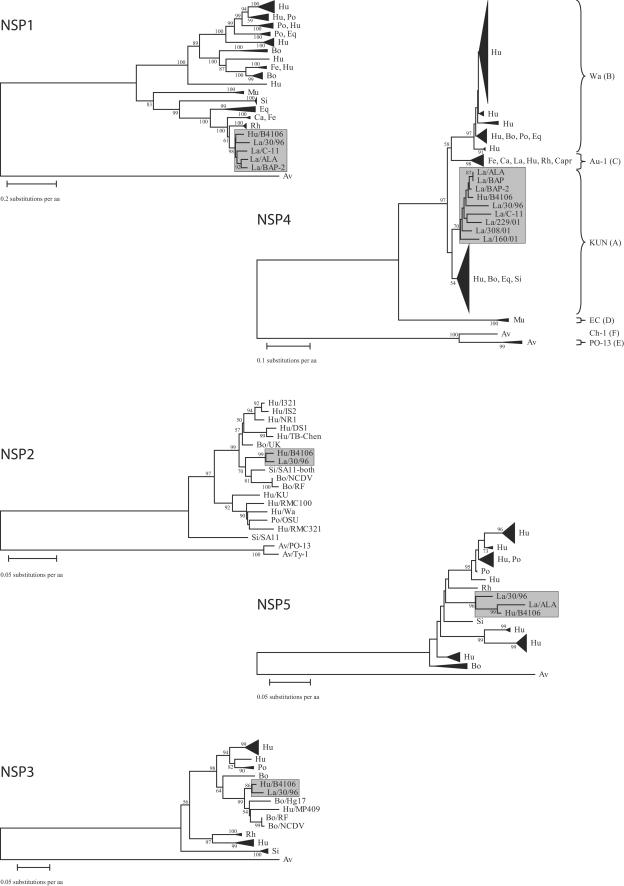
All of the 11 gene segments of the HRV strain B4106 clustered very closely with corresponding segments of LRV strain 30/96 and with several other LRV strains, namely, ALA, BAP, BAP-2, C-11, 308/01, 160/01, and 229/01, isolated in the United States or Italy (20, 69) in separate and exclusive lapine subclusters (Fig. 2 and 3). However, the high VP1 and VP2 amino acid sequence conservation (>97.0% for B4106, 30/96, and the two bovine rotavirus strains RF and Uktc) hampered a clear tree resolution. Accordingly, the phylogenetic trees were reconstructed using the nucleotide alignments of the VP1 and VP2 genes. The nucleotide-based phylogenetic dendrograms were identical to the amino acid-based dendrograms, but the genetic relationship between the human rotavirus B4106 and the lapine rotavirus 30/96 was supported by more robust bootstrap values (100 and 96 in the VP1 and VP2 dendrograms, respectively) (data not shown), suggesting that both gene segments are of lapine origin.
FIG. 2.
Phylogenetic dendrograms constructed with the structural rotaviral protein sequences of VP1, VP2, VP3, VP4, VP6, and VP7. Accession numbers can be found in in the supplemental material. Bootstrap values (2,000 replicates) greater than 50 are shown. The size of the triangles is an indication for the amount of sequences they represent. Hu, human; La, lapine; Bo, bovine; Po, porcine; Si, simian; Eq, equine; Ca, canine; Fe, feline; Capr, caprine; Rh, rhesus; Mu, murine; Av, avian. For the construction of the VP4 amino acid dendrogram representatives of all known P-types were used, except P[22], P[23], and P[25] because no complete amino acid sequences of these P-genotypes are available. For the VP7 amino acid dendrogram, the sequence of B4106, and representatives of all known G-types were used. For simplicity, not all G3 rotaviruses are depicted completely, one G3 subcluster containing only murine rotaviruses was designated G3/Mu, and a second subcluster (G3/Hu-Po-Bo) contained human, porcine, and bovine rotavirus strains. Lapine subclusters, including those containing HRV strain B4106, are boxed.
FIG. 3.
Phylogenetic dendrograms of the nonstructural rotaviral protein sequences of NSP1, NSP2, NSP3, NSP4, and NSP5. Accession numbers can be found in supplemental data. Bootstrap values (2,000 replicates) above 50 are shown. The size of the triangles is an indication for the amount of sequences they represent. For the NSP4 amino acid dendrograms, the sequence of B4106, and strains representing the six (A to F) established genogroups were used. Hu, human, La, lapine; Bo, bovine; Po, porcine; Si, simian; Eq, equine; Ca, canine; Fe, feline; Capr, caprine; Rh, rhesus; Mu, murine; Av, avian. Lapine subclusters, including those containing HRV strain B4106, are boxed.
DISCUSSION
In a previous study (31), we hypothesized that HRV strain B4106, an unusual human G3P[14] strain exhibiting a super-short electropherotype, could have been of lapine origin or a natural human-lapine reassortant. Completion of sequence comparisons of the entire genome of HRV strain B4106 and an LRV strain (30/96) have now confirmed that strain B4106 that caused gastroenteritis and hospitalized a child in Belgium, was of lapine origin, with an overall genome identity between B4106 and 30/96 of 93.4% at the nucleotide level, and 96.9% at the amino acid level. This is an original report supported by complete genome sequences to confirm that a nonhuman rotavirus strain was able to infect and induce disease in humans. The structural features of each of the 11 genome segments of B4106 and 30/96 are strikingly similar, and phylogenetic analysis revealed that the 11 genome segments of B4106 clustered in all-lapine phylogenetic subclusters (Fig. 2 and 3). The evidence that all of the genome segments of B4106 are lapine in origin demonstrates that a rotavirus transmission of an heterologous strain, rather than a reassortant virus, was able to induce gastroenteritis in a human host.
To date, information and understanding of rotavirus species-specificity have been provided by RNA-RNA hybridization analysis (79). By extensive use of RNA-RNA hybridization, it has been shown that rotaviruses recovered from the same animal species constitute a separate genogroup and usually share a high degree of overall genome homology. At least three genogroups have been identified for HRVs, namely, Wa-like, DS-1-like, and AU-1-like. The frequent occurrence of genetic reassortment events for practically all gene segments of HRV strains cocirculating in Finland was shown by partial sequencing of the 11 gene segments of HRV strains representative for several different electropherotypes (74). Natural reassortment between viruses of different genogroups occurs much more rarely than within rotaviruses of the same genogroup (51, 86). Cross-RNA-RNA hybridization, hemagglutination inhibition experiments, and partial sequencing data have suggested interspecies transmissions of rotavirus strains between different host species, such as human, cattle, pigs, dogs, cats, and horses (78, 81). Here, we have confirmed the interspecies transmission of a LRV strain to a human host by systematic comparison of all of their 11 gene segments. Although epidemiological investigation have demonstrated examples of intergenogroup reassortants, there is little evidence of interspecies transmission due to rotavirus completely belonging to another species (1, 79, 81), although a high rate of exposure of humans to animal rotavirus strains is expected to exist in some geographical settings.
Presumably, when rotaviruses cross the host species barrier, they are not naturally able to efficiently infect or spread in a new host. Instead, rotaviruses tend to reassort with another rotavirus inherent in that host species, and the resultant reassortant strains may have more chances to efficiently infect and spread among the population of the new host. Certain genome segments are more likely involved in rotavirus adaptation to new hosts under natural conditions than others. For example, the unusual G6P[9] HRV strains that were recently detected in Hungary (9) may have originated from the human G6P[9] strain PA151, isolated in the early 1980s in Italy (41), in which cross-hybridization analysis demonstrated that at least three genome segments, coding for the VP4 (fourth), NSP1 (fifth), and NSP4 (tenth) were derived from HRVs of the AU-1 genogroup, whereas at least six of the remaining genome segments, including the VP7 gene, were derived from a bovine strain (49). Similarly, the bovine-like G10P[11] HRV strains, which are well established in the human neonatal population in India, may have originated from cattle via reassortment with a human rotavirus strain (2, 52, 97). Complete characterization of the genome of the Indian bovine-like HRV strain I321 (G10P[11]) via hybridization and/or partial sequence analysis indicated that all of the gene segments were very closely related to those derived from bovine strains, whereas the NSP1 and NSP3 genes were of human origin (30, 33, 34, 87). Also, the VP7, NSP1, NSP4, and VP6-encoding gene segments of the neonatal G9P[11] strain 116E have been shown to be of human origin, whereas VP4 was of bovine origin (28, 29, 40). Other successful animal-like HRVs, with a porcine-like VP7 of G5 serotype specificity, have been detected in Brazil, Argentina, and Paraguay since the early 1980s (10, 17, 26, 43, 70) that were shown to be, by either RNA-RNA hybridization or sequence analysis, naturally occurring reassortants between members of the human Wa and porcine rotavirus genogroups (3). Finally, the nearly complete sequence analysis of the unusual G9P[19] HRV strains RMC321, with long electropherotype and SGI specificity, detected during an epidemic of infantile gastroenteritis in Manipur, India, in 1987 and 1998, revealed that at least seven genome segments were clearly of porcine origin (62, 96). Altogether, these data suggest a scenario similar to the one depicted for animal influenza viruses that are able to induce asymptomatic to lethal infections in heterologous host species but cannot be propagated successfully unless they are adapted by acquiring genome segments derived from heterologous strains via reassortment (16, 45, 57, 100).
The molecular basis of rotavirus natural cross-species infectivity and disease is not clear, but several genome segments, including VP3, VP4, VP7, NSP1, NSP2, and NSP4, have been implicated in host range restriction and/or virulence. Outstanding experiments based on generation of reassortants and experimental animal infections, while not providing definitive data, demonstrated that the control of virulence and host species specificity may reside in the complex interactions of multiple genes yet under the influence of the genetic context (13, 15, 21, 36, 46, 47, 60, 76, 101). In spite of the lapine nature of its VP3, VP4, VP7, NSP1, NSP3, and NSP4 genes, strain B4106 caused severe diarrhea in a human host, indicating that direct cross-species lapine-to-human transmission (not involving reassortment) may occur in nature and that the virulence of the lapine strains may be retained in the human host. This finding is particularly intriguing since P[14] HRV strains could only replicate partially in the rabbit host under experimental conditions, presumably due to the absence of more lapine-like genome segments (19). It would be interesting to determine whether the lapine-like HRV B4106 is able to efficiently infect and spread horizontally in rabbits as all LRVs do.
An intriguing finding is the partial duplication found in the 3′ noncoding region of the NSP5 gene of B4106 as depicted in Fig. 1. Most likely this duplication was generated through a mechanism similar to the duplication found in the NSP5 gene of lapine rotavirus strain ALA (42). Head-to-tail partial duplications have been found on several occasions in rotavirus genes VP6, NSP1, NSP3, NSP4, and NSP5 and also in the genes of other members of the family of the Reoviridae, such as the phytoreo- and orbiviruses (32, 72). Several molecular mechanisms for the occurrence of these head-to-tail partial duplications have been proposed (39, 42, 59, 61, 72, 89, 90). The finding that the similarity between the duplicated sequence and the homologous coding sequence of B4106 was only 81% is interesting. The mutations, deletions, and insertions found in the duplicated region probably arose over time since little selective pressure is asserted on the duplicated region to maintain important gene or protein functions. Other previously described duplications showed similarities ranging from 91 to 100% (39, 71). This indicates that LRV strains with a duplication in their NSP5 gene must have been circulating for quite some time before they infected a human host.
Along with the data presented here, a total of eight rotavirus strains have been completely sequenced. Complete genome characterization by sequence analysis of prototype rotavirus strains isolated from several animal species will allow to set up a universally available database to map the gene constellations of human-animal reassortant strains to provide a more in-depth understanding of the natural mechanisms that drive rotavirus evolution and host range restriction.
Supplementary Material
Acknowledgments
We thank all of the coworkers of the Laboratory of Clinical and Epidemiological Virology, Department of Microbiology and Immunology, Rega Institute for Medical Research, University of Leuven, Leuven, Belgium, for helpful comments and discussion.
J.M. was supported by the Institute for the Promotion of Innovation through Science and Technology in Flanders (IWT Vlaanderen).
Footnotes
Supplemental material for this article may be found at http://jvi.asm.org/.
REFERENCES
- 1.Ahmed, K., T. Nakagomi, and O. Nakagomi. 2005. Isolation and molecular characterization of a naturally occurring non-structural protein 5 (NSP5) gene reassortant of group A rotavirus of serotype G2P[4] with a long RNA pattern. J. Med. Virol. 77:323-330. [DOI] [PubMed] [Google Scholar]
- 2.Aijaz, S., K. Gowda, H. V. Jagannath, R. R. Reddy, P. P. Maiya, R. L. Ward, H. B. Greenberg, M. Raju, A. Babu, and C. D. Rao. 1996. Epidemiology of symptomatic human rotaviruses in Bangalore and Mysore, India, from 1988 to 1994 as determined by electropherotype, subgroup, and serotype analysis. Arch. Virol. 141:715-726. [DOI] [PubMed] [Google Scholar]
- 3.Alfieri, A. A., J. P. Leite, O. Nakagomi, E. Kaga, P. A. Woods, R. I. Glass, and J. R. Gentsch. 1996. Characterization of human rotavirus genotype P[8]G5 from Brazil by probe-hybridization and sequence. Arch. Virol. 141:2353-2364. [DOI] [PubMed] [Google Scholar]
- 4.Altschul, S. F., W. Gish, W. Miller, E. W. Myers, and D. J. Lipman. 1990. Basic local alignment search tool. J. Mol. Biol. 215:403-410. [DOI] [PubMed] [Google Scholar]
- 5.Arias, C. F., P. Romero, V. Alvarez, and S. Lopez. 1996. Trypsin activation pathway of rotavirus infectivity. J. Virol. 70:5832-5839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Au, K. S., N. M. Mattion, and M. K. Estes. 1993. A subviral particle binding domain on the rotavirus nonstructural glycoprotein NS28. Virology 194:665-673. [DOI] [PubMed] [Google Scholar]
- 7.Azevedo, M. S., L. Yuan, K. I. Jeong, A. Gonzalez, T. V. Nguyen, S. Pouly, M. Gochnauer, W. Zhang, A. Azevedo, and L. J. Saif. 2005. Viremia and nasal and rectal shedding of rotavirus in gnotobiotic pigs inoculated with Wa human rotavirus. J. Virol. 79:5428-5436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ball, J. M., P. Tian, C. Q. Zeng, A. P. Morris, and M. K. Estes. 1996. Age-dependent diarrhea induced by a rotaviral nonstructural glycoprotein. Science 272:101-104. [DOI] [PubMed] [Google Scholar]
- 9.Banyai, K., J. R. Gentsch, D. D. Griffin, J. L. Holmes, R. I. Glass, and G. Szucs. 2003. Genetic variability among serotype G6 human rotaviruses: identification of a novel lineage isolated in Hungary. J. Med. Virol. 71:124-134. [DOI] [PubMed] [Google Scholar]
- 10.Bok, K., N. Castagnaro, A. Borsa, S. Nates, C. Espul, O. Fay, A. Fabri, S. Grinstein, I. Miceli, D. O. Matson, and J. A. Gomez. 2001. Surveillance for rotavirus in Argentina. J. Med. Virol. 65:190-198. [PubMed] [Google Scholar]
- 11.Both, G. W., A. R. Bellamy, and D. B. Mitchell. 1994. Rotavirus protein structure and function. Curr. Top. Microbiol. Immunol. 185:67-105. [DOI] [PubMed] [Google Scholar]
- 12.Both, G. W., L. J. Siegman, A. R. Bellamy, and P. H. Atkinson. 1983. Coding assignment and nucleotide sequence of simian rotavirus SA11 gene segment 10: location of glycosylation sites suggests that the signal peptide is not cleaved. J. Virol. 48:335-339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bridger, J. C., W. Dhaliwal, M. J. Adamson, and C. R. Howard. 1998. Determinants of rotavirus host range restriction: a heterologous bovine NSP1 gene does not affect replication kinetics in the pig. Virology 245:47-52. [DOI] [PubMed] [Google Scholar]
- 14.Broome, R. L., P. T. Vo, R. L. Ward, H. F. Clark, and H. B. Greenberg. 1993. Murine rotavirus genes encoding outer capsid proteins VP4 and VP7 are not major determinants of host range restriction and virulence. J. Virol. 67:2448-2455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Burke, B., and U. Desselberger. 1996. Rotavirus pathogenicity. Virology 218:299-305. [DOI] [PubMed] [Google Scholar]
- 16.Capua, I., and D. J. Alexander. 2004. Human health implications of avian influenza viruses and paramyxoviruses. Eur. J. Clin. Microbiol. Infect. Dis. 23:1-6. [DOI] [PubMed] [Google Scholar]
- 17.Carmona, R. C., M. C. Timenetsky, F. F. da Silva, and C. F. Granato. 2004. Characterization of rotavirus strains from hospitalized and outpatient children with acute diarrhoea in Sao Paulo, Brazil. J. Med. Virol. 74:166-172. [DOI] [PubMed] [Google Scholar]
- 18.Chan, W. K., K. S. Au, and M. K. Estes. 1988. Topography of the simian rotavirus nonstructural glycoprotein (NS28) in the endoplasmic reticulum membrane. Virology 164:435-442. [DOI] [PubMed] [Google Scholar]
- 19.Ciarlet, M., M. K. Estes, C. Barone, R. F. Ramig, and M. E. Conner. 1998. Analysis of host range restriction determinants in the rabbit model: comparison of homologous and heterologous rotavirus infections. J. Virol. 72:2341-2351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ciarlet, M., M. K. Estes, and M. E. Conner. 1997. Comparative amino acid sequence analysis of the outer capsid protein VP4 from four lapine rotavirus strains reveals identity with genotype P[14] human rotaviruses. Arch. Virol. 142:1059-1069. [DOI] [PubMed] [Google Scholar]
- 21.Ciarlet, M., M. K. Estes, and M. E. Conner. 2000. Simian rhesus rotavirus is a unique heterologous (nonlapine) rotavirus strain capable of productive replication and horizontal transmission in rabbits. J. Gen. Virol. 81:1237-1249. [DOI] [PubMed] [Google Scholar]
- 22.Ciarlet, M., Y. Hoshino, and F. Liprandi. 1997. Single point mutations may affect the serotype reactivity of serotype G11 porcine rotavirus strains: a widening spectrum? J. Virol. 71:8213-8220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ciarlet, M., F. Liprandi, M. E. Conner, and M. K. Estes. 2000. Species specificity and interspecies relatedness of NSP4 genetic groups by comparative NSP4 sequence analyses of animal rotaviruses. Arch. Virol. 145:371-383. [DOI] [PubMed] [Google Scholar]
- 24.Clark, H. F., P. A. Offit, R. W. Ellis, J. J. Eiden, D. Krah, A. R. Shaw, M. Pichichero, J. J. Treanor, F. E. Borian, L. M. Bell, and S. A. Plotkin. 1996. The development of multivalent bovine rotavirus (strain WC3) reassortant vaccine for infants. J. Infect. Dis. 174:S73-S80. [DOI] [PubMed] [Google Scholar]
- 25.Clements-Mann, M. L., R. Dudas, Y. Hoshino, P. Nehring, E. Sperber, M. Wagner, I. Stephens, R. Karron, A. Deforest, and A. Z. Kapikian. 2001. Safety and immunogenicity of live attenuated quadrivalent human-bovine (UK) reassortant rotavirus vaccine administered with childhood vaccines to infants. Vaccine 19:4676-4684. [DOI] [PubMed] [Google Scholar]
- 26.Coluchi, N., V. Munford, J. Manzur, C. Vazquez, M. Escobar, E. Weber, P. Marmol, and M. L. Racz. 2002. Detection, subgroup specificity, and genotype diversity of rotavirus strains in children with acute diarrhea in Paraguay. J. Clin. Microbiol. 40:1709-1714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Coulson, B. S., S. L. Londrigan, and D. J. Lee. 1997. Rotavirus contains integrin ligand sequences and a disintegrin-like domain that are implicated in virus entry into cells. Proc. Natl. Acad. Sci. USA 94:5389-5394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cunliffe, N. A., B. K. Das, M. Ramachandran, M. K. Bhan, R. I. Glass, and J. R. Gentsch. 1997. Sequence analysis demonstrates that VP6, NSP1, and NSP4 genes of Indian neonatal rotavirus strain 116E are of human origin. Virus Genes 15:39-44. [DOI] [PubMed] [Google Scholar]
- 29.Das, B. K., J. R. Gentsch, Y. Hoshino, S. Ishida, O. Nakagomi, M. K. Bhan, R. Kumar, and R. I. Glass. 1993. Characterization of the G serotype and genogroup of New Delhi newborn rotavirus strain 116E. Virology 197:99-107. [DOI] [PubMed] [Google Scholar]
- 30.Das, M., S. J. Dunn, G. N. Woode, H. B. Greenberg, and C. D. Rao. 1993. Both surface proteins (VP4 and VP7) of an asymptomatic neonatal rotavirus strain (I321) have high levels of sequence identity with the homologous proteins of a serotype 10 bovine rotavirus. Virology 194:374-379. [DOI] [PubMed] [Google Scholar]
- 31.De Leener, K., M. Rahman, J. Matthijnssens, L. Van Hoovels, T. Goegebuer, I. van der Donck, and M. Van Ranst. 2004. Human infection with a P[14], G3 lapine rotavirus. Virology 325:11-17. [DOI] [PubMed] [Google Scholar]
- 32.Desselberger, U. 1996. Genome rearrangements of rotaviruses. Arch. Virol. Suppl. 12:37-51. [DOI] [PubMed] [Google Scholar]
- 33.Dunn, S. J., T. L. Cross, and H. B. Greenberg. 1994. Comparison of the rotavirus nonstructural protein NSP1 (NS53) from different species by sequence analysis and northern blot hybridization. Virology 203:178-183. [DOI] [PubMed] [Google Scholar]
- 34.Dunn, S. J., H. B. Greenberg, R. L. Ward, O. Nakagomi, J. W. Burns, P. T. Vo, K. A. Pax, M. Das, K. Gowda, and C. D. Rao. 1993. Serotypic and genotypic characterization of human serotype 10 rotaviruses from asymptomatic neonates. J. Clin. Microbiol. 31:165-169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dyall-Smith, M. L., I. Lazdins, G. W. Tregear, and I. H. Holmes. 1986. Location of the major antigenic sites involved in rotavirus serotype-specific neutralization. Proc. Natl. Acad. Sci. USA 83:3465-3468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.El Attar, L., W. Dhaliwal, C. R. Howard, and J. C. Bridger. 2001. Rotavirus cross-species pathogenicity: molecular characterization of a bovine rotavirus pathogenic for pigs. Virology 291:172-182. [DOI] [PubMed] [Google Scholar]
- 37.Estes, M. K. 2004. Rotaviruses and their replication, p. 1747-1786. In D. M. Knipe, P. M. Howley, D. E. Griffin, R. A. Lamb, M. A. Martin, B. Roizman, and S. E. Straus (ed.), Fields virology, 4th ed. Lippincott/The Williams & Wilkins Co., Philadelphia, Pa.
- 38.Feng, N., J. W. Burns, L. Bracy, and H. B. Greenberg. 1994. Comparison of mucosal and systemic humoral immune responses and subsequent protection in mice orally inoculated with a homologous or a heterologous rotavirus. J. Virol. 68:7766-7773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gault, E., N. Schnepf, D. Poncet, A. Servant, S. Teran, and A. Garbarg-Chenon. 2001. A human rotavirus with rearranged genes 7 and 11 encodes a modified NSP3 protein and suggests an additional mechanism for gene rearrangement. J. Virol. 75:7305-7314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gentsch, J. R., B. K. Das, B. Jiang, M. K. Bhan, and R. I. Glass. 1993. Similarity of the VP4 protein of human rotavirus strain 116E to that of the bovine B223 strain. Virology 194:424-430. [DOI] [PubMed] [Google Scholar]
- 41.Gerna, G., A. Sarasini, M. Parea, S. Arista, P. Miranda, H. Brussow, Y. Hoshino, and J. Flores. 1992. Isolation and characterization of two distinct human rotavirus strains with G6 specificity. J. Clin. Microbiol. 30:9-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gorziglia, M., K. Nishikawa, and N. Fukuhara. 1989. Evidence of duplication and deletion in super short segment 11 of rabbit rotavirus Alabama strain. Virology 170:587-590. [DOI] [PubMed] [Google Scholar]
- 43.Gouvea, V., L. de Castro, M. C. Timenetsky, H. Greenberg, and N. Santos. 1994. Rotavirus serotype G5 associated with diarrhea in Brazilian children. J. Clin. Microbiol. 32:1408-1409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Greenberg, H. B., P. T. Vo, and R. Jones. 1986. Cultivation and characterization of three strains of murine rotavirus. J. Virol. 57:585-590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Holmes, E. C., E. Ghedin, N. Miller, J. Taylor, Y. Bao, K. St George, B. T. Grenfell, S. L. Salzberg, C. M. Fraser, D. J. Lipman, and J. K. Taubenberger. 2005. Whole-genome analysis of human influenza A virus reveals multiple persistent lineages and reassortment among recent H3N2 viruses. PLoS Biol. 3:In press. [DOI] [PMC free article] [PubMed]
- 46.Horie, Y., O. Masamune, and O. Nakagomi. 1997. Three major alleles of rotavirus NSP4 proteins identified by sequence analysis. J. Gen. Virol. 78:2341-2346. [DOI] [PubMed] [Google Scholar]
- 47.Hoshino, Y., L. J. Saif, S. Y. Kang, M. M. Sereno, W. K. Chen, and A. Z. Kapikian. 1995. Identification of group A rotavirus genes associated with virulence of a porcine rotavirus and host range restriction of a human rotavirus in the gnotobiotic piglet model. Virology 209:274-280. [DOI] [PubMed] [Google Scholar]
- 48.Hua, J., E. A. Mansell, and J. T. Patton. 1993. Comparative analysis of the rotavirus NS53 gene: conservation of basic and cysteine-rich regions in the protein and possible stem-loop structures in the RNA. Virology 196:372-378. [DOI] [PubMed] [Google Scholar]
- 49.Iizuka, M., E. Kaga, M. Chiba, O. Masamune, G. Gerna, and O. Nakagomi. 1994. Serotype G6 human rotavirus sharing a conserved genetic constellation with natural reassortants between members of the bovine and AU-1 genogroups. Arch. Virol. 135:427-432. [DOI] [PubMed] [Google Scholar]
- 50.Ito, H., M. Sugiyama, K. Masubuchi, Y. Mori, and N. Minamoto. 2001. Complete nucleotide sequence of a group A avian rotavirus genome and a comparison with its counterparts of mammalian rotaviruses. Virus Res. 75:123-138. [DOI] [PubMed] [Google Scholar]
- 51.Iturriza-Gomara, M., B. Isherwood, U. Desselberger, and J. Gray. 2001. Reassortment in vivo: driving force for diversity of human rotavirus strains isolated in the United Kingdom between 1995 and 1999. J. Virol. 75:3696-3705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Iturriza-Gomara, M., G. Kang, A. Mammen, A. K. Jana, M. Abraham, U. Desselberger, D. Brown, and J. Gray. 2004. Characterization of G10P[11] rotaviruses causing acute gastroenteritis in neonates and infants in Vellore, India. J. Clin. Microbiol. 42:2541-2547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Iturriza-Gomara, M., C. Wong, S. Blome, U. Desselberger, and J. Gray. 2002. Molecular characterization of VP6 genes of human rotavirus isolates: correlation of genogroups with subgroups and evidence of independent segregation. J. Virol. 76:6596-6601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kamer, G., and P. Argos. 1984. Primary structural comparison of RNA-dependent polymerases from plant, animal and bacterial viruses. Nucleic Acids Res. 12:7269-7282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kapikian, A. Z., Y. Hoshino, and R. M. Chanock. 2004. Rotaviruses, p. 1787-1834. In D. M. Knipe, P. M. Howley, D. E. Griffin, R. A. Lamb, M. A. Martin, B. Roizman, and S. E. Straus (ed.), Fields virology, 4th ed. Lippincott/The Williams & Wilkins Co., Philadelphia, Pa.
- 56.Kapikian, A. Z., Y. Hoshino, R. M. Chanock, and I. Perez-Schael. 1996. Efficacy of a quadrivalent rhesus rotavirus-based human rotavirus vaccine aimed at preventing severe rotavirus diarrhea in infants and young children. J. Infect. Dis. 174:S65-S72. [DOI] [PubMed] [Google Scholar]
- 57.Kawaoka, Y., S. Krauss, and R. G. Webster. 1989. Avian-to-human transmission of the PB1 gene of influenza A viruses in the 1957 and 1968 pandemics. J. Virol. 63:4603-4608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kirkwood, C., P. J. Masendycz, and B. S. Coulson. 1993. Characteristics and location of cross-reactive and serotype-specific neutralization sites on VP7 of human G type 9 rotaviruses. Virology 196:79-88. [DOI] [PubMed] [Google Scholar]
- 59.Kojima, K., K. Taniguchi, M. Kawagishi-Kobayashi, S. Matsuno, and S. Urasawa. 2000. Rearrangement generated in double genes, NSP1 and NSP3, of viable progenies from a human rotavirus strain. Virus Res. 67:163-171. [DOI] [PubMed] [Google Scholar]
- 60.Kojima, K., K. Taniguchi, and N. Kobayashi. 1996. Species-specific and interspecies relatedness of NSP1 sequences in human, porcine, bovine, feline, and equine rotavirus strains. Arch. Virol. 141:1-12. [DOI] [PubMed] [Google Scholar]
- 61.Kojima, K., K. Taniguchi, T. Urasawa, and S. Urasawa. 1996. Sequence analysis of normal and rearranged NSP5 genes from human rotavirus strains isolated in nature: implications for the occurrence of the rearrangement at the step of plus strand synthesis. Virology 224:446-452. [DOI] [PubMed] [Google Scholar]
- 62.Krishnan, T., B. Burke, S. Shen, T. N. Naik, and U. Desselberger. 1994. Molecular epidemiology of human rotaviruses in Manipur: genome analysis of rotaviruses of long electropherotype and subgroup I. Arch. Virol. 134:279-292. [DOI] [PubMed] [Google Scholar]
- 63.Kumar, A., A. Charpilienne, and J. Cohen. 1989. Nucleotide sequence of the gene encoding for the RNA binding protein (VP2) of RF bovine rotavirus. Nucleic Acids Res. 17:2126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kumar, S., K. Tamura, I. B. Jakobsen, and M. Nei. 2001. MEGA2: molecular evolutionary genetics analysis software. Bioinformatics 17:1244-1245. [DOI] [PubMed] [Google Scholar]
- 65.Lambden, P. R., S. J. Cooke, E. O. Caul, and I. N. Clarke. 1992. Cloning of noncultivatable human rotavirus by single primer amplification. J. Virol. 66:1817-1822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lopez, S., C. F. Arias, J. R. Bell, J. H. Strauss, and R. T. Espejo. 1985. Primary structure of the cleavage site associated with trypsin enhancement of rotavirus SA11 infectivity. Virology 144:11-19. [DOI] [PubMed] [Google Scholar]
- 67.Lopez, S., R. Espinosa, H. B. Greenberg, and C. F. Arias. 1994. Mapping the subgroup epitopes of rotavirus protein VP6. Virology 204:153-162. [DOI] [PubMed] [Google Scholar]
- 68.Martella, V., M. Ciarlet, K. Banyai, E. Lorusso, A. Cavalli, M. Corrente, G. Elia, S. Arista, M. Camero, C. Desario, N. Decaro, A. Lavazza, and C. Buonavoglia. Identification of a novel VP4 genotype carried by a serotype G5 porcine rotavirus strain. Virology, in press. [DOI] [PubMed]
- 69.Martella, V., M. Ciarlet, A. Camarda, A. Pratelli, M. Tempesta, G. Greco, A. Cavalli, G. Elia, N. Decaro, V. Terio, G. Bozzo, M. Camero, and C. Buonavoglia. 2003. Molecular characterization of the VP4, VP6, VP7, and NSP4 genes of lapine rotaviruses identified in Italy: emergence of a novel VP4 genotype. Virology 314:358-370. [DOI] [PubMed] [Google Scholar]
- 70.Mascarenhas, J. D., A. C. Linhares, Y. B. Gabbay, and J. P. Leite. 2002. Detection and characterization of rotavirus G and P types from children participating in a rotavirus vaccine trial in Belem, Brazil. Mem. Inst. Oswaldo Cruz 97:113-117. [DOI] [PubMed] [Google Scholar]
- 71.Matsui, S. M., E. R. Mackow, S. Matsuno, P. S. Paul, and H. B. Greenberg. 1990. Sequence analysis of gene 11 equivalents from “short” and “super short” strains of rotavirus. J. Virol. 64:120-124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Matthijnssens, J., M. Rahman, and M. Van Ranst. 2006. Loop model: mechanism to explain partial gene duplications in segmented dsRNA viruses. Biochem. Biophys. Res. Commun. 340:140-144. [DOI] [PubMed] [Google Scholar]
- 73.Mattion, N. M., J. Cohen, C. Aponte, and M. K. Estes. 1992. Characterization of an oligomerization domain and RNA-binding properties on rotavirus nonstructural protein NS34. Virology 190:68-83. [DOI] [PubMed] [Google Scholar]
- 74.Maunula, L., and C. H. von Bonsdorff. 2002. Frequent reassortments may explain the genetic heterogeneity of rotaviruses: analysis of Finnish rotavirus strains. J. Virol. 76:11793-11800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Mitchell, D. B., and G. W. Both. 1990. Completion of the genomic sequence of the simian rotavirus SA11: nucleotide sequences of segments 1, 2, and 3. Virology 177:324-331. [DOI] [PubMed] [Google Scholar]
- 76.Mori, Y., M. A. Borgan, M. Takayama, N. Ito, M. Sugiyama, and N. Minamoto. 2003. Roles of outer capsid proteins as determinants of pathogenicity and host range restriction of avian rotaviruses in a suckling mouse model. Virology 316:126-134. [DOI] [PubMed] [Google Scholar]
- 77.Mori, Y., M. Sugiyama, M. Takayama, Y. Atoji, T. Masegi, and N. Minamoto. 2001. Avian-to-mammal transmission of an avian rotavirus: analysis of its pathogenicity in a heterologous mouse model. Virology 288:63-70. [DOI] [PubMed] [Google Scholar]
- 78.Nakagomi, O., M. Mochizuki, Y. Aboudy, I. Shif, I. Silberstein, and T. Nakagomi. 1992. Hemagglutination by a human rotavirus isolate as evidence for transmission of animal rotaviruses to humans. J. Clin. Microbiol. 30:1011-1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Nakagomi, O., and T. Nakagomi. 2002. Genomic relationships among rotaviruses recovered from various animal species as revealed by RNA-RNA hybridization assays. Res. Vet. Sci. 73:207-214. [DOI] [PubMed] [Google Scholar]
- 80.Nicholas, K. B., H. B. Nicholas, and D. W. Deerfield. 1997. GeneDoc: analysis and visualization of genetic variation. Embnet News 4:14. [Google Scholar]
- 81.Palombo, E. A. 2002. Genetic analysis of group A rotaviruses: evidence for interspecies transmission of rotavirus genes. Virus Genes 24:11-20. [DOI] [PubMed] [Google Scholar]
- 82.Patton, J. T., J. Hua, and E. A. Mansell. 1993. Location of intrachain disulfide bonds in the VP5* and VP8* trypsin cleavage fragments of the rhesus rotavirus spike protein VP4. J. Virol. 67:4848-4855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Patton, J. T., L. Salter-Cid, A. Kalbach, E. A. Mansell, and M. Kattoura. 1993. Nucleotide and amino acid sequence analysis of the rotavirus nonstructural RNA-binding protein NS35. Virology 192:438-446. [DOI] [PubMed] [Google Scholar]
- 84.Prasad, B. V., G. J. Wang, J. P. Clerx, and W. Chiu. 1988. Three-dimensional structure of rotavirus. J. Mol. Biol. 199:269-275. [DOI] [PubMed] [Google Scholar]
- 85.Rahman, M., J. Matthijnssens, S. Nahar, G. Podder, D. A. Sack, T. Azim, and M. Van Ranst. 2005. Characterization of a novel P[25],G11 human group A rotavirus. J. Clin. Microbiol. 43:3208-3212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Ramachandran, M., C. D. Kirkwood, L. Unicomb, N. A. Cunliffe, R. L. Ward, M. K. Bhan, H. F. Clark, R. I. Glass, and J. R. Gentsch. 2000. Molecular characterization of serotype G9 rotavirus strains from a global collection. Virology 278:436-444. [DOI] [PubMed] [Google Scholar]
- 87.Rao, C. D., M. Das, P. Ilango, R. Lalwani, B. S. Rao, and K. Gowda. 1995. Comparative nucleotide and amino acid sequence analysis of the sequence-specific RNA-binding rotavirus nonstructural protein NSP3. Virology 207:327-333. [DOI] [PubMed] [Google Scholar]
- 88.Santos, N., and Y. Hoshino. 2005. Global distribution of rotavirus serotypes/genotypes and its implication for the development and implementation of an effective rotavirus vaccine. Rev. Med. Virol. 15:29-56. [DOI] [PubMed] [Google Scholar]
- 89.Scott, G. E., O. Tarlow, and M. A. McCrae. 1989. Detailed structural analysis of a genome rearrangement in bovine rotavirus. Virus Res. 14:119-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Shen, S., B. Burke, and U. Desselberger. 1994. Rearrangement of the VP6 gene of a group A rotavirus in combination with a point mutation affecting trimer stability. J. Virol. 68:1682-1688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Tang, B., J. M. Gilbert, S. M. Matsui, and H. B. Greenberg. 1997. Comparison of the rotavirus gene 6 from different species by sequence analysis and localization of subgroup-specific epitopes using site-directed mutagenesis. Virology 237:89-96. [DOI] [PubMed] [Google Scholar]
- 92.Taylor, J. A., J. A. O'Brien, and M. Yeager. 1996. The cytoplasmic tail of NSP4, the endoplasmic reticulum-localized nonstructural glycoprotein of rotavirus, contains distinct virus binding and coiled coil domains. EMBO J. 15:4469-4476. [PMC free article] [PubMed] [Google Scholar]
- 93.Thompson, J. D., T. J. Gibson, F. Plewniak, F. Jeanmougin, and D. G. Higgins. 1997. The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 25:4876-4882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Tian, P., J. M. Ball, C. Q. Zeng, and M. K. Estes. 1996. The rotavirus nonstructural glycoprotein NSP4 possesses membrane destabilization activity. J. Virol. 70:6973-6981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.van Staden, V., J. Theron, B. J. Greyling, H. Huismans, and L. H. Nel. 1991. A comparison of the nucleotide sequences of cognate NS2 genes of three different orbiviruses. Virology 185:500-504. [DOI] [PubMed] [Google Scholar]
- 96.Varghese, V., S. Das, N. B. Singh, K. Kojima, S. K. Bhattacharya, T. Krishnan, N. Kobayashi, and T. N. Naik. 2004. Molecular characterization of a human rotavirus reveals porcine characteristics in most of the genes including VP6 and NSP4. Arch. Virol. 149:155-172. [DOI] [PubMed] [Google Scholar]
- 97.Varshney, B., M. R. Jagannath, R. R. Vethanayagam, S. Kodhandharaman, H. V. Jagannath, K. Gowda, D. K. Singh, and C. D. Rao. 2002. Prevalence of, and antigenic variation in, serotype G10 rotaviruses and detection of serotype G3 strains in diarrheic calves: implications for the origin of G10P11 or P11 type reassortant asymptomatic strains in newborn children in India. Arch. Virol. 147:143-165. [DOI] [PubMed] [Google Scholar]
- 98.Vesikari, T. 1996. Trials of oral bovine and rhesus rotavirus vaccines in Finland: a historical account and present status. Arch. Virol. 12:177-186. [DOI] [PubMed] [Google Scholar]
- 99.Vesikari, T., E. Isolauri, E. D'Hondt, A. Delem, F. E. Andre, and G. Zissis. 1984. Protection of infants against rotavirus diarrhoea by RIT 4237 attenuated bovine rotavirus strain vaccine. Lancet i:977-981. [DOI] [PubMed] [Google Scholar]
- 100.Webster, R. G., Y. Guan, L. Poon, S. Krauss, R. Webby, E. Govorkovai, and M. Peiris. 2005. The spread of the H5N1 bird flu epidemic in Asia in 2004. Arch. Virol. Suppl. 2005:117-129. [DOI] [PubMed] [Google Scholar]
- 101.Xu, L., Y. Tian, O. Tarlow, D. Harbour, and M. A. McCrae. 1994. Molecular biology of rotaviruses. IX. Conservation and divergence in genome segment 5. J. Gen. Virol. 75:3413-3421. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.