Abstract
Objective:
To investigate the potential immunologic and anti-inflammatory effects of hypertonic saline plus dextran (HSD) in hemorrhagic trauma patients.
Background:
Unbalanced inflammation triggered by shock has been linked to multiorgan dysfunction (MOD) and death. In animal and cellular models, HSD alters the inflammatory response to shock, attenuating MOD and improving outcome. It remains untested whether HSD has similar effects in humans.
Methods:
A single 250-mL dose of either HSD (7.5% NaCl, 6% dextran-70) or placebo (0.9% NaCl) was administered to adult blunt trauma patients in hemorrhagic shock. The primary outcome was to measure changes in immune/inflammatory markers, including neutrophil activation, monocyte subset redistribution, cytokine production, and neuroendocrine changes. Patient demographics, fluid requirements, organ dysfunction, infection, and death were recorded.
Results:
A total of 27 patients were enrolled (13 HSD) with no significant differences in clinical measurements. Hyperosmolarity was modest and transient, whereas the immunologic/anti-inflammatory effects persisted for 24 hours. HSD blunted neutrophil activation by abolishing shock-induced CD11b up-regulation and causing CD62L shedding. HSD altered the shock-induced monocyte redistribution pattern by reducing the drop in “classic” CD14++ and the expansion of the “pro-inflammatory” CD14+CD16+ subsets. In parallel, HSD significantly reduced pro-inflammatory tumor necrosis factor (TNF)-α production while increasing anti-inflammatory IL-1ra and IL-10. HSD prevented shock-induced norepinephrine surge with no effect on adrenal steroids.
Conclusions:
This first human trial evaluating the immunologic/anti-inflammatory effects of hypertonic resuscitation in trauma patients demonstrates that HSD promotes a more balanced inflammatory response to hemorrhagic shock, raising the possibility that similar to experimental models, HSD might also attenuate post-trauma MOD.
This is the first human trial demonstrating that hypertonic saline dextran (HSD) has immunologic and anti-inflammatory effects in trauma patients in hemorrhagic shock. HSD attenuated neutrophil activation, altered monocyte redistribution patterns, and reduced pro-inflammatory and increased anti-inflammatory cytokine generation. Similar to experimental studies, HSD promoted a more balanced inflammatory response to shock.
Hemorrhagic shock following civilian trauma is an important contributor to the morbidity and mortality in this patient population.1 The body's early response to traumatic injury and hemorrhage is characterized by an excessive innate immune activation and by an overwhelming inflammatory reaction.2 Research findings suggest a strong link between immune dysfunction and post-traumatic complications such as multiorgan dysfunction (MOD) and sepsis.3–5 Regulated inflammatory responses are generally considered a beneficial host response to injury,6 while post-traumatic hyperinflammation and ensuing immune incompetence are considered to be maladaptive and often auto-destructive.2
One of the fundamental principles applied to the management of hemorrhagic shock is the early administration of fluid and blood products to correct the deranged hemodynamic status. Crystalloid solutions have generally been considered first-line therapy. However, recent evidence suggests that isotonic crystalloid solutions may actually aggravate the immune dysfunction.7 By contrast, a number of experimental studies reveal that hypertonic solutions have favorable immunomodulatory effects on hemorrhage/resuscitation-induced leukocyte activation.4,8–11 For example, data derived from both in vitro human leukocyte studies as well as animal models of hemorrhagic shock found that hypertonicity decreases neutrophil activation/adherence,12–16 stimulates lymphocyte proliferation,17 inhibits pro-inflammatory but stimulates anti-inflammatory cytokine production by monocyte/macrophages,8,18–22 and reduces hormone secretion.23,24 In the in vivo setting, hypertonic saline resuscitation strategies have been shown to mitigate the development of inflammation. When considered in conjunction with the ability of small-volume (4 mL/kg) infusion of hypertonic saline, to restore mean arterial pressure and microvascular perfusion25,26 as well as its proven safety record in patients,27,28 there has been renewed interest in the use of hypertonic saline solutions, with or without dextran in the management of patients with hemorrhagic shock.29–31 One critical piece of information lacking in the translation of these beneficial effects to the human trauma setting is whether hypertonic saline/hypertonic saline dextran (HS/HSD) is able to exert comparable immunologic effects in humans.
The major objective of the present studies was to investigate the immunomodulatory effects of HSD in patients sustaining hemorrhagic shock following trauma. Without altering standards of treatment, patients were randomized to receive either a single 250-mL bolus of HSD or placebo. Blood drawn at subsequent time points was evaluated for a number of cellular and molecular inflammatory markers known to be altered by shock/resuscitation. We hypothesized that these markers would be altered in resuscitated trauma patients, in a manner previously reported in experimental models. These data were intended to provide “proof of principle” for larger trials that intended to study clinical outcomes with this resuscitation regimen.
MATERIALS AND METHODS
Study Population
This prospective, randomized, double-blinded, placebo-controlled trial of HSD resuscitation for traumatic hemorrhagic shock was conducted at Sunnybrook and Women's College Health Sciences Centre between April 2001 and August 2002. Institutional Research Ethics Board approved the study with provisions for delayed informed consent for the initial intervention. All patients or their next of kin were informed of their participation within 24 hours, and permission was obtained for continued data collection.
Patients were eligible for inclusion in the study if they sustained blunt trauma, were 16 years of age or older, had at least one recorded episode of hypotension (systolic blood pressure ≤90 mm Hg) with clear evidence of blood loss (external or internal including thorax, abdomen, or retroperitoneum). Patients were excluded if they refused to participate, were admitted ≥6 hours after injury, were without vital signs, pregnant, or had stigmata of chronic disease.
Upon arrival in the Emergency Department, eligible patients randomly received a single 250-mL intravenous bolus of either HSD (7.5% NaCl, 6% dextran-70) or placebo (0.9% NaCl) from identical unidentified bags. Since there is little evidence demonstrating the superiority of either HS or HSD, we opted for HSD because dextran prolongs the plasma volume expansion effect of HS and would allow comparison with earlier clinical trials in trauma. Resuscitation protocol otherwise adhered to the Advanced Trauma Life Support guidelines. The crystalloid solutions used during the first 24 hours were normal saline (0.9% NaCl) and Ringers' Lactate. Patients were followed until hospital discharge or death.
Clinical Parameters
Baseline data collected included: age, gender, injury severity score (ISS), mechanism of injury, time from injury to hospital admission, time in the trauma room, lowest blood pressure and heart rate in the trauma room, total amount of fluids and blood administered for resuscitation within the first 24 hours, length of hospital stay, length of ICU stay, number of ventilated days, all surgical interventions, organ dysfunction as measured by ΔMOD score and pneumonia (defined as any 3 of the following: fever, leukocytosis, radiologic infiltrate, and sputum culture). Additional laboratory data collected prospectively included complete blood count, serum sodium, osmolality, chloride, potassium, creatinine, lactate, bicarbonate, PT/PTT, and INR.
Antibodies, Reagents, and Solutions
The following antihuman monoclonal antibodies (mAbs) were purchased from Becton Dickinson Biosciences (BD, San Jose, CA): CD16–fluorescein isothiocyanate (FITC), CD45–peridinin chlorophyll protein (PerCP), CD14–allophycocyanin (APC), CD11b–FITC, CD62L–phycoerythrin (PE), IL-1-PE, IL-1ra-PE, IL-10-PE and TNF-α-PE. Lipopolysaccharide (LPS; Escherichia coli 055:B5), paraformaldehyde, and brefelden A (BFA) were purchased from Sigma Chemical Co (St. Louis, MO). Tissue culture reagents were purchased from GibcoBRL/Life Technologies (Grand Island, NY). Hypertonic saline plus dextran (7.5% NaCl in 6% dextran-70) was prepared by the hospital's pharmacy. Normal saline (0.9% NaCl) from Baxter was used as placebo.
Blood Collection and Stimulation Conditions
Blood samples (15 mL) were collected before infusion, and again at 1, 3, 6, and 24 hours after the intervention in EDTA and sodium heparin sterile glass Vacutainers (BD, Franklin Lakes, NJ). Whole blood aliquots (2 mL) were treated with the Golgi transport inhibitor BFA at a final concentration of 10 μg/mL and used to determine unstimulated (reflecting spontaneous in vivo monocyte activation) and endotoxin-stimulated (LPS, 1 μg/mL) intracellular cytokine expression. Non-BFA-treated blood was processed in parallel for determination of unstimulated and endotoxin-stimulated cellular adhesion molecule expression. All samples were cultured for 22 hours at 37°C in a 5% CO2 humidified atmosphere.
Cell Surface and Intracellular Immunofluorescence Staining
Surface adhesion molecules were detected by staining whole blood with anti-CD11b-FITC and CD62L-PE mAbs. To identify neutrophils and monocyte subsets, samples were further stained with anti-CD45-FITC and CD14-APC mAbs. Class-matched isotype immunoglobulin negative control mAbs were added simultaneously to separate tubes for all samples to detect nonspecific binding. Samples were then treated with FACS Lysing Solution (BD) and centrifuged. The supernatant was washed with CellWASH (BD) and cell pellets resuspended in paraformaldehyde. Thereafter, specimens for intracellular cytokine detection were further treated with FACS Permeabilizing Solution (BD) and incubated with corresponding antihuman-cytokine mAbs. After incubation, cells were washed, resuspended in paraformaldehyde, and analyzed by flow cytometry.
Flow Cytometric Analysis
Stained cell suspensions were acquired on a dual-laser FACSCalibur flow cytometer (BD Biosciences) calibrated for 4-color analysis. An electronic acquisition gate was set on neutrophils and total monocytes according to regionalization on the basis of anti-CD45/CD14 fluorescence emission characteristics using bivariate dotplots in CellQuest Pro software (BD). Further sequential gating was used to identify 2 major subpopulations of monocytes on the basis of their coexpression of CD16 and CD14. Typically, ≥5,000 CD14+ monocyte-gated events were acquired for assessment of intracellular cytokines and cellular adhesion molecule expression. Relative mean fluorescence intensities (MFI) of the sample antigen fluorescence minus the MFI of the isotype control, in arbitrary units scaled from 0 to 10,000 of selected adhesion molecules or intracellular cytokines in unstimulated and LPS-stimulated cultures, were quantified using fluorescence histogram data. Analysis gates and quadrant markers were set to define positive and negative populations according to the nonspecific staining of isotype-matched negative controls. Instrument fluorescence intensity calibration was standardized for each cytometer run using CaliBRITE beads and AutoCOMP software (BD). Absolute cell counts were obtained by multiplying the corresponding percentages of cells derived from FACS analysis by total leukocyte counts obtained from a hematology analyzer (Coulter Electronics, Hialeah, FL).
Circulating Hormone and Adhesion Molecule Analysis
Specimens for catecholamine [epinephrine (Epi) and norepinephrine (NE)] analysis were drawn into EDTA and reduced glutathione vacuum tubes (Amersham, Arlington Heights, IL). Plasma was separated by centrifugation (3000g, 15 minutes) and the supernatant frozen at −80°C until assay. Circulating unbound catecholamine concentrations were quantified by gas chromatography-mass spectrometry (GC-MS). Plasma cortisol and DHEA were measured by IMMULITE automated, solid phase chemiluminescent enzyme immunometric analyzer (Diagnostic Products Corporation, Los Angeles, CA). Soluble CD62L was measured using a sandwich ELISA kit according to the manufacturer's instructions (Bender MedSystems, Burlingame, CA).
Statistical Analysis
Serial changes in cellular adhesion molecules, intracellular cytokines, and hormones were evaluated by 2-way analysis of variance for repeated measures. To isolate specific group and time differences among treatment means, we used the Newman-Keuls post hoc multiple comparison test with Huynh-Feldt correction for multiple comparisons. Data within the groups were compared at each data point by a factorial analysis of variance and Scheffé multiple comparison test. χ2 test was used for intergroup comparisons of baseline characteristics. Data are expressed as mean ± standard error with a 0.05 significance level. Estimation of sample size was based on previous experimental studies where neutrophil exposure to HS consistently altered some of the parameters we propose to investigate (CD62L and CD11b) in all animals and isolated cell assays tested.12,13,15,16 We hypothesize that a similar effect and magnitude would be present in trauma patients and estimated that 10 patients per group would provide sufficient power.
RESULTS
Clinical Outcome
Twenty-seven patients were entered into the trial and received an initial infusion. Two were subsequently excluded from the analysis for failing to meet inclusion criteria, and one other refused to participate in the study. The HSD group included 10 patients and the control group 14.
Both groups had similar characteristics, including demographics and physiologic parameters upon enrollment, injury severity, type of trauma, and preadmission interventions (Table 1). Some differences were noted with respect to fluid use between the 2 groups. Patients randomized to HSD received more crystalloid before arriving in the trauma room but received less crystalloid while in the Emergency Room and about half the amount of blood and colloids during the initial 24 hours (Table 1). This may reflect the longer prehospital time and greater likelihood of referrals from other hospitals in the HSD group (not statistically significant).
TABLE 1. Baseline Characteristics and Outcomes of the Study Patients (mean SD)

All patients were admitted to the ICU with similar length of stay and number of operative interventions. MOD and pneumonia affected both groups equally and ventilation times, despite being 24 hours shorter in the HSD group, did not statistically differ from control. Two control patients died, shortening the group's mean hospital stay and accounting for the longer in-hospital stay observed in the HSD group compared with control patients (P < 0.05). No patients received vasopressors or inotropes throughout the 24-hour study period.
Blood Chemistry and Osmolality
HSD resuscitation caused a modest and transient increase in both serum sodium and osmolality, which normalized by 24 hours (Table 2). No differences in serum chloride, potassium, creatinine, lactate, bicarbonate, PT/PTT, and INR were detected between the 2 groups. HSD did not affect blood typing or cross-matching.
TABLE 2. Changes in Serum Sodium and Osmolality at Baseline and After Resuscitation

Total Leukocyte Counts
Hemorrhage/resuscitation induced a significant overall leukocytosis in all patients as evidenced by elevated baseline cell counts (Table 3). The increase in white cells was mainly due to a marked granulocytosis. All patients experienced a drop in total leukocyte and neutrophil counts postresuscitation as compared with baseline. Although there were no significant differences in cell counts between treatment groups, it is notable that HSD reduced the magnitude of postresuscitation lymphopenia as compared with the control group. This finding is in accordance with previous experimental models indicating that hypertonicity may influence lymphocyte mobilization and proliferation.17
TABLE 3. Leukocyte, Granulocyte, and Lymphocyte Counts at Baseline and Post-Resuscitation

Cell Surface and Soluble Adhesion Molecule Expression
Changes in expression of selected neutrophil adhesion molecules such as CD11b and CD62L are one measure of cellular activation following shock/resuscitation. Hemorrhage/resuscitation caused a progressive increase in the surface expression of CD11b starting as early as the first measurement at 1 hour and persisting up to 24 hours (Fig. 1A). A single dose of HSD prevented this shock-induced CD11b up-regulation observed in the control group. This inhibitory effect on CD11b expression was sustained below baseline for the entire 24-hour observation period.

FIGURE 1. Neutrophil cell-surface and soluble adhesion molecule expression. HSD (7.5% NaCl, 6% Dextran-70) significantly decreased neutrophil surface expression of the adhesion molecules CD11b (A) and CD62L (B) on unstimulated (•, control; ○, HSD) and LPS-stimulated (▪, control; □, HSD) cells up to 24 hours. Data are expressed as percentage change in mean fluorescence intensity (ΔMFI) compared with baseline conditions (set at 100%). C, HSD caused an increase in the soluble form of CD62L that paralleled its reduction on the neutrophil surface. Serum concentrations (ng/mL) were measured by ELISA. Significant differences: *P < 0.05 versus baseline values within a treatment group; †P < 0.05 versus time-matched control values.
Hemorrhage/resuscitation per se caused no change in neutrophil CD62L expression (Fig. 1B). In contrast, HSD caused a progressive reduction in surface CD62L expression, reaching a nadir at 3 hours and remaining low over the entire experimental period. These changes in CD62L surface expression parallel those reported in experimental models where hypertonic inhibition of neutrophil activation was linked to reduced MOD.12,32 LPS stimulation elicited typical changes in both CD11b/CD62L from all patients, attesting to neutrophil viability and functionality (Fig. 1A, B).
The reduction of CD62L surface expression was paralleled by an increase in serum soluble CD62L (Fig. 1C), suggesting that the reduction in the surface expression was due to well-characterized shedding mechanisms.
Expression patterns in monocytic CD11b differed from that observed in neutrophils. Despite an apparent early rise induced by hemorrhage/resuscitation, monocytic CD11b expression did not change significantly over time or between the 2 groups. LPS stimulation again was able to increase CD11b expression in control cells, but this rise was significantly less in monocytes from patients treated with HSD (Fig. 2A).

FIGURE 2. Monocyte cell-surface adhesion molecule expression. HSD (7.5% NaCl, 6% Dextran-70) significantly altered monocyte surface expression of CD11b (A) and CD62L (B) on unstimulated (•, control; ○, HSD) and LPS-stimulated (▪, control; □, HSD) cells up to 24 hours. Data are expressed as percentage change in mean fluorescence intensity (ΔMFI) compared with baseline conditions (set at 100%). Significant differences: *P < 0.05 versus baseline values within a treatment group; †P < 0.05 versus time-matched control values.
In contrast, hemorrhage/resuscitation caused no significant increase in CD62L expression until late, 24 hours. HSD resuscitation not only reduced CD62L expression throughout the study period but also abolished its late increase in expression (Fig. 2B). LPS stimulation elicited a significant loss of surface CD62L that was of similar magnitude in both groups.
Blood Monocyte Subset Redistribution
Multiparameter flow cytometry was used to measure the differential pattern of “classic” (CD14++CD16−) and “pro-inflammatory” (CD14+CD16+) monocyte subset redistribution in response to HSD. The absolute monocyte counts and relative subset distributions are summarized in Table 4. The classic CD14++ subset comprised the majority of circulating monocytes in both HSD and control groups at baseline. Interestingly, the percentage and number of CD14++ monocytes dropped significantly in the control group, while in patients receiving HSD the proportion of these cells increased modestly.
TABLE 4. Whole Blood Monocyte Counts and Subset Distribution by Resuscitation Group

The effect of HSD on monocyte redistribution was even more pronounced for the pro-inflammatory CD14+CD16+ subset. While hemorrhage/resuscitation caused a significant expansion of CD14+CD16+ cells up to 24 hours in control patients, HSD markedly reduced this subset over the same time course (Table 4). Opposing alterations in CD14++ and CD14+CD16+ monocyte subsets were reflected by changes in the monocyte subset ratio, which showed a progressive increase (32% to 57%) in control patients and a decrease (29% to 19%) in HSD resuscitated patients (Table 4).
These results demonstrate that while hemorrhage/resuscitation causes selective expansion of pro-inflammatory CD14+CD16+ monocyte phenotype in the peripheral blood, a single dose of HSD elicits less monocyte redistribution, with a bias toward selective depletion of the pro-inflammatory CD14+CD16+ subpopulation.
Monocytic Intracellular Cytokine Expression
Next we assessed the impact of HSD on spontaneous and LPS-stimulated intracellular expression of pro-(TNF-α, IL-1β) and anti-inflammatory (IL-1ra, IL-10) cytokines in classic and pro-inflammatory monocyte subsets (Fig. 3). Compared with conventional circulating cytokine assay methods, flow cytometric intracellular cytokine detection offers the unique advantages of whole blood analysis, which does not require cellular purification or isolation, allowing detection at a single cell level rather than bulk cytokine release.33 Changes in monocytic intracellular cytokine production were expressed as MFI values (amount of cytokine per positive cell), presented in Figure 3.

FIGURE 3. Monocyte intracellular cytokine expression. HSD (7.5% NaCl, 6% Dextran-70) significantly inhibited tumor necrosis factor (TNF)-α (A, B) but enhanced IL-10 (C, D) and IL-1ra (E, F) production in unstimulated (•, control; ○, HSD) and LPS-stimulated (▪, control; □, HSD) “classic” (CD14++CD16−) and “pro-inflammatory” (CD14+CD16+) monocyte subsets, respectively. Intracellular cytokines were detected by flow cytometry. Data are expressed as percentage change in mean fluorescence intensity (ΔMFI) compared with baseline conditions (set at 100%). Significant differences: *P < 0.05 versus baseline values within a treatment group; †P < 0.05 versus time-matched control values.
TNF-α expression by unstimulated CD14+ monocytes increased significantly over time after hemorrhage/resuscitation (Fig. 3B, solid line). The percentage of TNF-α-positive monocytes more than doubled, whereas the MFI increased by almost 20% above baseline values by 24 hours postresuscitation. In contrast, the increased expression of TNF-α was prevented in cells from patients receiving HSD (Fig. 3B, solid line).
Further detailed analysis revealed that, although both CD14++ and CD14+CD16+ subsets expressed TNF-α (Fig. 3A, B), the expanded pro-inflammatory CD14+CD16+ subset was primarily responsible for both spontaneous and LPS-induced up-regulation of TNF-α production following hemorrhage/resuscitation. Moreover, HSD specifically inhibited TNF-α production capacity of CD14+CD16+ pro-inflammatory monocytes (Fig. 3B). Similar results were observed in LPS-stimulated monocytes (Fig. 3A, B, dotted lines).
There was no statistical difference in the spontaneous or LPS-stimulated expression of IL-1β, although we found a trend toward lower expression of this cytokine in patients that received HSD (data not shown). These results demonstrate that HSD effectively inhibits both the spontaneous and stimulated capacity of pro-inflammatory monocytes to produce TNF-α in response to hemorrhage/resuscitation.
One potential mechanism contributing to the anti-inflammatory effects of HSD is its ability to induce counterinflammatory cytokines. To determine the effect of HSD on the ability of monocytes to promote a counterinflammatory response, we measured spontaneous and LPS-induced intracellular expression of the anti-inflammatory cytokines IL-10 and IL-1ra (Fig. 3C–F). In contrast to the early stimulating effect exerted on pro-inflammatory TNF-α production, hemorrhage/resuscitation had no significant influence on either anti-inflammatory cytokine during the first 6 hours postresuscitation (Fig. 3C, D solid lines). A single dose of HSD, however, led to an early and sustained increase in IL-10 and IL-1ra production (Fig. 3C, E). HSD more than doubled the percentage of spontaneously IL-10-positive CD14+ monocytes by 1 hour, which remained significantly elevated for 24 hours. This response was closely matched by a corresponding increase in both IL-10 and IL-1ra MFI values. Monocyte subset analysis traced the IL-10 production exclusively to the CD14++ subset, a response further potentiated by endotoxin stimulation (Fig. 3C). This confirms an inability of CD14+CD16+ monocytes to produce IL-10.34 IL-1ra expression was observed in both CD14++ and CD14+CD16+ monocytes (Fig. 3E, F, solid lines). Similar results were observed in LPS-stimulated neutrophils (Fig. 3C–F, dotted lines).
Hormonal Responses
Alterations in the hormonal milieu following shock/resuscitation may influence the immune response.35 Hemorrhage/resuscitation elicited a significant rise in circulating NE, and a steady decrease in Epi compared with levels measured at admission (Fig. 4). HSD completely prevented the NE variations elicited by hemorrhage/resuscitation and NE levels were significantly lower than control at 3 and 6 hours (Fig. 4A). HSD caused no significant change in Epi circulating levels compared with control (Fig. 4B). Circulating concentrations of the adrenal steroids DHEA and cortisol did not change significantly over time or between treatment groups, with the exception of a modest decrease in cortisol levels in both groups at 24 hours (data not shown).
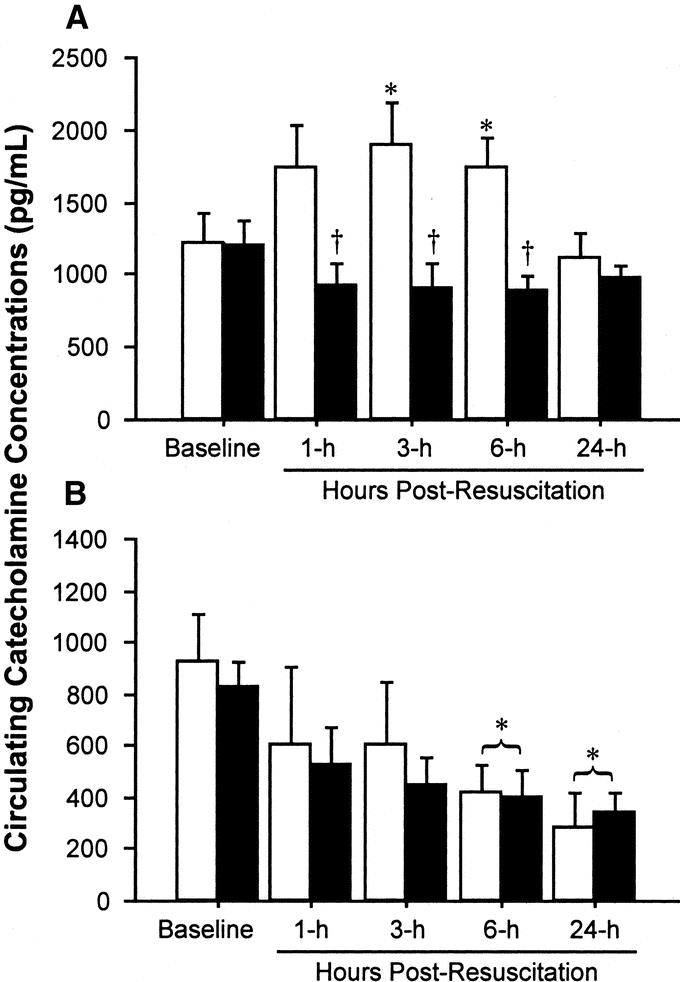
FIGURE 4. Circulating catecholamine concentrations. HSD significantly blunted the rise in circulating norepinephrine concentration (NE; A) compared with control, without altering epinephrine concentrations (Epi; B). Plasma concentrations (mean ± SE, pg/mL) were measured in control (○) and HSD (▪) groups by mass spectroscopy. Significant differences: *P < 0.05 versus baseline values within a treatment group; †P < 0.05 versus time-matched control values.
DISCUSSION
The present studies are the first to investigate the effects of supplemental treatment with a single bolus of HSD on immune parameters in patients with hemorrhagic shock following trauma. Until now, human studies on the immunologic effects of hypertonic saline have been limited to elective surgical patients30 and healthy volunteers.36 Kolsen-Petersen et al found that no significant changes in multiple immune indices following HS administration to women undergoing abdominal hysterectomy.30 Angle et al similarly reported modest alterations in immune function when HS was given to normal human volunteers.36 The mechanisms responsible for the difference between our study and those previously reported are not clear. It might relate to the fact that the effects of hypertonicity are more pronounced when coupled with a second inflammatory stimulus such as hemorrhage/resuscitation. In this regard, Oreopoulos et al reported that the effect of HS on liver cytokine expression was only observed when given in combination with liver ischemia/reperfusion.21 Given the multiple effects of ischemia/reperfusion on cell signaling cascades, these studies suggest future investigation of interactions between the effects of ischemia/reperfusion and those induced by hypertonicity. Another possible explanation is that the properties of HS are only evident in the presence of an intense inflammatory response that might be not present in healthy volunteers and patients with a minor surgical insult. Thus, severely traumatized patients would likely benefit the most from hypertonic saline therapy and should be the focus of future studies on the potential benefits of hypertonicity in attenuating MOD.
This trial was neither designed nor powered to identify any possible benefit in terms of reducing MOD and sepsis. Although we did not find a significant reduction in ventilator time, there was a suggestion that it was shorter after HSD. The literature suggests that the organ-protective effects of hypertonicity are most evident in the lungs. In rodent models, work from several groups, including our own, showed that hypertonic saline mitigated the histologic, cellular, and other markers of acute lung injury.12,14,21,37 Despite no human trial ever been specifically designed to measure them, many have documented similar protective effects on post-trauma lung dysfunction. For example, Simmas et al found that children with severe head injury treated with HS had significantly less acute respiratory distress syndrome than controls,31 whereas Mattox et al reported less pneumonia among trauma patients resuscitated with HS.38
Neutrophils are implicated as key players in the pathogenesis of endothelial cell and tissue injury provoked by hemorrhagic shock, in particular post-traumatic acute lung injury.39 Hypertonic saline-induced inhibition of neutrophil activation and adhesion has been proposed as the main mechanism for its lung-protective properties.14 In experimental models of hemorrhagic shock, one of the best established immunomodulatory effects of hypertonicity is its capacity to down-regulate CD62L/CD11b expression and prevent neutrophil-mediated organ damage.4,12,13,15,16 A major finding of the present study was that HSD significantly inhibited neutrophil activation, as measured by cell surface expression of CD62L/CD11b and soluble CD62L. This strongly suggests that HSD resuscitation may help prevent organ injury by reducing unbalanced neutrophil-endothelial interactions and transmigration into tissues such as the lungs.
Our findings also reveal that HSD blunts CD62L/CD11b expression on circulating monocytes. Given the pivotal role of monocytes as nonspecific effector cells, secreting cytokines, and regulating inflammation, this finding further suggests that HSD may help avert widespread tissue injury via its capacity to limit monocyte activation. This idea is supported by studies of trauma patients showing that acute up-regulation of monocytic CD62L and CD11b after injury is related to the development of post-traumatic organ failure.40,41
An important finding of the present study was that HSD differentially modulates the post-traumatic mobilization and cytokine production of functionally heterogeneous monocyte subsets. Human monocytes comprise various subpopulations with distinct phenotypic and functional properties.42 The main population of “classic” monocytes is strongly CD14-positive but CD16 negative (CD14++ subset). A smaller subset (<10% in healthy individuals) is weakly CD14-positive but coexpresses CD16 (CD14+CD16+ subset).43 These so-called “pro-inflammatory” monocytes readily express TNF-α but fail to produce significant amounts of IL-10.34 Indeed, they are likened to a “circulating macrophage,” which is selectively mobilized (via β-adrenergic mechanisms) from the marginal pool during stress.44
We found that while hemorrhage/resuscitation provoked a drop in classic CD14++ monocytes and expansion of the pro-inflammatory CD14+CD16+ subset (ie, an exaggerated inflammatory response), HSD caused an expansion of CD14++ monocytes and reduction of the CD14+CD16+ subset. This is in accordance with studies showing significant post-traumatic phenotypic alterations in circulating monocytes.35,45,46 Considerable expansion of the CD14+CD16+ subset has been reported in patients with systemic inflammation and sepsis.47 This finding could have further relevance to acute lung injury, since under inflammatory conditions, up to 70% of alveolar macrophages are derived from circulating CD14+CD16+ monocytes.48
Changes in the pattern of monocyte subset redistribution were paralleled by shifts in monocytic cytokine production profiles. HSD increased the production of anti-inflammatory IL-10 and IL-1ra by the CD14++ subset, and at the same time dramatically reduced intracellular TNF-α production by the pro-inflammatory CD14+CD16+ subset. This is consistent with studies demonstrating that the minor population of CD14+CD16+ monocytes have an enhanced capacity for TNF-α generation49 but fail to produce significant amounts of IL-10.34 Our findings also support clinical studies showing increased TNF-α mRNA transcription and serum concentrations immediately following trauma and hemorrhage.50,51
To our knowledge, no other reports or studies directly evaluated the effects of HSD on cytokine production in trauma patients, but experimental data corroborate our findings. Using microarray cDNA analysis, Gushchin et al found that hypertonicity modulates cytokine gene transcription, effectively inhibiting TNF-α expression by organs and human leukocytes after hemorrhage/resuscitation.19 Animal studies using a 2-hit model of shock/resuscitation reported that hypertonic preconditioning inhibits TNF-α generation and up-regulates IL-10 production by isolated alveolar and peritoneal macrophages as well as hepatic tissue.20–22
Another interesting finding of the present study is the suggestion that HSD anti-inflammatory actions may be linked to cross-talk between cytokines and neuroendocrine hormones. We found that HSD prevented the post-traumatic surge of NE, consistent with reports that HSD attenuates sympathetic hormone secretion after hemorrhage.23,24 It is particularly relevant since catecholamines are recognized as key regulators of cytokine production.52 Maddali et al showed that trauma-induced elevations in catecholamines mediate monocyte/macrophage inflammatory cytokine production.35 The immunomodulatory effects of catecholamines on monocytes are mediated via α- and β-adrenoreceptors coupled to intracellular cAMP. NE-induced α-adrenergic stimulation reduces cAMP, inhibiting TNF-α, but enhances IL-10 production.52 Thus, it can be postulated that HSD's differential cytokine induction capacity may be, at least partly, due to down-regulation of α-adrenergic signaling. This concept is supported by findings that hypertonicity triggers rapid cAMP accumulation in human leukocytes, which suppresses their inflammatory activity.53 Further studies are needed to fully elucidate the mechanisms of hypertonicity-induced sympathetic cytokine regulation.
Our results indicate HSD has the capacity to rapidly up-regulate counterinflammatory cytokines IL-10 and IL-1ra that attenuate postinjury hyperinflammatory reactions. The ability of HSD to quickly reestablish inflammatory cytokine balance may be the basis for reduced tissue injury and organ dysfunction observed in experimental models. The beneficial therapeutic effects of HSD may also derive from its ability to prevent extensive delayed counter-regulation by reducing the magnitude of the initial pro-inflammatory response. This idea is supported by evidence that hypertonic saline reduces the risk of delayed immunosuppression and septic complications in animals after hemorrhagic shock by restoring suppressed lymphocyte function.8,17,37 Interestingly, we also found that HSD prevented post-trauma lymphopenia, possibly reflecting less lymphocyte perturbation in these patients. HSD might also reduce septic complications through rapid restoration of gut perfusion, thereby preventing further damage to the enteral barrier and reducing bacterial translocation and associated cytokinemia.54
It is reasonable to expect that the earlier HSD is administered after injury, the greater its subsequent effects. However, in this trial, we found remarkable immunologic changes, even though HSD infusion was delayed and patients were given large amounts of crystalloids, thereby diluting the hypertonic solution and preventing the large increases in serum osmolality reported in experimental models. Similar results, showing that HSD continues to exert its effects a day after infusion and even following repeated doses, have been observed in rodent models.15 This observation may facilitate future trials in that HSD does not necessarily have to be administered in isolation or immediately after trauma.
There are other interesting observations to be made from this study. The immunologic effects were still evident 24 hours postresuscitation in HSD-treated patients, despite relatively modest (up to 10 mmol/L above control) and transient increases in serum osmolality compared with levels reported in animal models.12 This unique finding merits further exploration over a longer observation period. This trial did not address the possible effects of dextran. Dextran prolongs HS intravascular fluid expansion effect and has mild anti-inflammatory properties.25 While current opinion is that the effects observed by adding dextran to HS are primarily due to the slightly better and more sustained plasma volume expansion, dextran's role remains to be investigated.25
The clinical role of HS has still not been fully investigated. The only recent clinical trial by Cooper et al found that HS (without dextran) administered to comatose and hypotensive trauma patients resulted in no neurologic improvement by 6 months.29 Despite not measuring inflammatory parameters or reporting MOD, HS patients had a higher survival (55% versus 47%). Although not statistically significant, this certainly merits further investigation.29
CONCLUSION
This is the first human trial to demonstrate the anti-inflammatory and immunologic properties of hypertonic saline plus dextran in trauma patients in hemorrhagic shock. This study provides evidence that when administered in the early stages of shock resuscitation, hypertonic saline plus dextran exerts profound immunomodulatory effects, promoting a more balanced pro- and anti-inflammatory response. Consistent with experimental models where similar effects were linked to a reduction in post-traumatic complications, including multiorgan dysfunction and sepsis, our findings open the possibility that this may be true in humans as well and reinforces the need for larger randomized control trials.
ACKNOWLEDGMENTS
The authors thank Michele Rizoli and Sheila Petrongolo for the expert editorial and technical assistance provided.
Footnotes
Supported by the Defence Research and Development Canada.
Reprints: Sandro Rizoli, MD, PhD, FRCSC, Sunnybrook and Women's College Health Sciences Centre, University of Toronto, 2075 Bayview Ave, Suite H1-71, Toronto, Ontario M4N 3M5, Canada. E-mail: sandro.rizoli@sw.ca.
REFERENCES
- 1.Moore FA, McKinley BA, Moore EE. The next generation in shock resuscitation. Lancet. 2004;363:1988–1996. [DOI] [PubMed] [Google Scholar]
- 2.Giannoudis PV. Current concepts of the inflammatory response after major trauma: an update. Injury. 2003;34:397–404. [DOI] [PubMed] [Google Scholar]
- 3.Maier RV. Pathogenesis of multiple organ dysfunction syndrome: endotoxin, inflammatory cells, and their mediators. Cytokines and reactive oxygen species. Surg Infect (Larchmt). 2000;1:197–205. [DOI] [PubMed] [Google Scholar]
- 4.Rizoli SB, Rotstein OD, Sibbald WJ. The immunological effects of hypertonic saline. In: Vincent JL, ed. Yearbook of Intensive Care and Emergency Medicine. Berlin: Springer-Verlag, 2002:446–453. [Google Scholar]
- 5.Rotstein OD. Modeling the two-hit hypothesis for evaluating strategies to prevent organ injury after shock/resuscitation. J Trauma. 2003;54(suppl):203–206. [DOI] [PubMed] [Google Scholar]
- 6.Oberholzer A, Oberholzer C, Moldawer LL. Cytokine signaling: regulation of the immune response in normal and critically ill states. Crit Care Med. 2000;28:N3–N12. [DOI] [PubMed] [Google Scholar]
- 7.Rhee P, Wang D, Ruff P, et al. Human neutrophil activation and increased adhesion by various resuscitation fluids. Crit Care Med. 2000;28:74–78. [DOI] [PubMed] [Google Scholar]
- 8.Junger WG, Coimbra R, Liu FC, et al. Hypertonic saline resuscitation: a tool to modulate immune function in trauma patients? Shock. 1997;8:235–241. [PubMed] [Google Scholar]
- 9.Rotstein OD. Novel strategies for immunomodulation after trauma: revisiting hypertonic saline as a resuscitation strategy for hemorrhagic shock. J Trauma. 2000;49:580–583. [DOI] [PubMed] [Google Scholar]
- 10.Kolsen-Petersen JA. Immune effect of hypertonic saline: fact or fiction? Acta Anaesthesiol Scand. 2004;48:667–678. [DOI] [PubMed] [Google Scholar]
- 11.Shukla A, Hashiguchi N, Chen Y, et al. Osmotic regulation of cell function and possible clinical applications. Shock. 2004;21:391–400. [DOI] [PubMed] [Google Scholar]
- 12.Rizoli SB, Kapus A, Fan J, et al. Immunomodulatory effects of hypertonic resuscitation on the development of lung inflammation following hemorrhagic shock. J Immunol. 1998;161:6288–6296. [PubMed] [Google Scholar]
- 13.Rizoli SB, Kapus A, Parodo J, et al. Hypertonicity prevents lipopolysaccharide-stimulated CD11b/CD18 expression in human neutrophils in vitro: role for p38 inhibition. J Trauma. 1999;46:794–798; discussion 798–799. [DOI] [PubMed]
- 14.Angle N, Hoyt DB, Coimbra R, et al. Hypertonic saline resuscitation diminishes lung injury by suppressing neutrophil activation after hemorrhagic shock. Shock. 1998;9:164–170. [DOI] [PubMed] [Google Scholar]
- 15.Rizoli SB, Kapus A, Parodo J, et al. Hypertonic immunomodulation is reversible and accompanied by changes in CD11b expression. J Surg Res. 1999;83:130–135. [DOI] [PubMed] [Google Scholar]
- 16.Rizoli SB, Rotstein OD, Parodo J, et al. Hypertonic inhibition of exocytosis in neutrophils: central role for osmotic actin skeleton remodeling. Am J Physiol Cell Physiol. 2000;279:C619–C633. [DOI] [PubMed] [Google Scholar]
- 17.Loomis WH, Namiki S, Hoyt DB, et al. Hypertonicity rescues T cells from suppression by trauma-induced anti-inflammatory mediators. Am J Physiol Cell Physiol. 2001;281:C840–C848. [DOI] [PubMed] [Google Scholar]
- 18.Cuschieri J, Gourlay D, Garcia I, et al. Hypertonic preconditioning inhibits macrophage responsiveness to endotoxin. J Immunol. 2002;168:1389–1396. [DOI] [PubMed] [Google Scholar]
- 19.Gushchin V, Stegalkina S, Alam HB, et al. Cytokine expression profiling in human leukocytes after exposure to hypertonic and isotonic fluids. J Trauma. 2002;52:867–871. [DOI] [PubMed] [Google Scholar]
- 20.Oreopoulos GD, Bradwell S, Lu Z, et al. Synergistic induction of IL-10 by hypertonic saline solution and lipopolysaccharides in murine peritoneal macrophages. Surgery. 2001;130:157–165. [DOI] [PubMed] [Google Scholar]
- 21.Oreopoulos GD, Wu H, Szaszi K, et al. Hypertonic preconditioning prevents hepatocellular injury following ischemia/reperfusion in mice: a role for interleukin 10. Hepatology. 2004;40:211–220. [DOI] [PubMed] [Google Scholar]
- 22.Powers KA, Woo J, Khadaroo RG, et al. Hypertonic resuscitation of hemorrhagic shock upregulates the anti-inflammatory response by alveolar macrophages. Surgery. 2003;134:312–318. [DOI] [PubMed] [Google Scholar]
- 23.Cross JS, Gruber DP, Gann DS, et al. Hypertonic saline attenuates the hormonal response to injury. Ann Surg. 1989;209:684–691; discussion 691–692. [DOI] [PMC free article] [PubMed]
- 24.Wade CE, Hannon JP, Bossone CA, et al. Neuroendocrine responses to hypertonic saline/dextran resuscitation following hemorrhage. Circ Shock. 1991;35:37–43. [PubMed] [Google Scholar]
- 25.Kramer GC. Hypertonic resuscitation: physiologic mechanisms and recommendations for trauma care. J Trauma. 2003;54(suppl):89–99. [DOI] [PubMed] [Google Scholar]
- 26.Wade CE, Kramer GC, Grady JJ, et al. Efficacy of hypertonic 7.5% saline and 6% dextran-70 in treating trauma: a meta-analysis of controlled clinical studies. Surgery. 1997;122:609–616. [DOI] [PubMed] [Google Scholar]
- 27.Dubick MA, Wade CE. A review of the efficacy and safety of 7.5% NaCl/6% dextran 70 in experimental animals and in humans. J Trauma. 1994;36:323–330. [DOI] [PubMed] [Google Scholar]
- 28.Kreimeier U, Messmer K. Small-volume resuscitation: from experimental evidence to clinical routine: advantages and disadvantages of hypertonic solutions. Acta Anaesthesiol Scand. 2002;46:625–638. [DOI] [PubMed] [Google Scholar]
- 29.Cooper DJ, Myles PS, McDermott FT, et al. Prehospital hypertonic saline resuscitation of patients with hypotension and severe traumatic brain injury: a randomized controlled trial. JAMA. 2004;291:1350–1357. [DOI] [PubMed] [Google Scholar]
- 30.Kolsen-Petersen JA, Nielsen JO, Bendtzen K, et al. Infusion of hypertonic saline (7.5% NaCl) causes minor immunological changes in normovolaemic women. Acta Anaesthesiol Scand. 2004;48:224–233. [DOI] [PubMed] [Google Scholar]
- 31.Simma B, Burger R, Falk M, et al. A prospective, randomized, and controlled study of fluid management in children with severe head injury: lactated Ringer's solution versus hypertonic saline. Crit Care Med. 1998;26:1265–1270. [DOI] [PubMed] [Google Scholar]
- 32.Angle N, Hoyt DB, Cabello-Passini R, et al. Hypertonic saline resuscitation reduces neutrophil margination by suppressing neutrophil L selectin expression. J Trauma. 1998;45:7–12; discussion 12–13. [DOI] [PubMed]
- 33.Ghanekar SA, Maecker HT. Cytokine flow cytometry: multiparametric approach to immune function analysis. Cytotherapy. 2003;5:1–6. [DOI] [PubMed] [Google Scholar]
- 34.Frankenberger M, Sternsdorf T, Pechumer H, et al. Differential cytokine expression in human blood monocyte subpopulations: a polymerase chain reaction analysis. Blood. 1996;87:373–377. [PubMed] [Google Scholar]
- 35.Maddali S, Stapleton PP, Freeman TA, et al. Neuroendocrine responses mediate macrophage function after trauma. Surgery. 2004;136:1038–1046. [DOI] [PubMed] [Google Scholar]
- 36.Angle N, Cabello-Passini R, Hoyt DB, et al. Hypertonic saline infusion: can it regulate human neutrophil function? Shock. 2000;14:503–508. [PubMed] [Google Scholar]
- 37.Coimbra R, Hoyt DB, Junger WG, et al. Hypertonic saline resuscitation decreases susceptibility to sepsis after hemorrhagic shock. J Trauma. 1997;42:602–606; discussion 606–607. [DOI] [PubMed]
- 38.Mattox KL, Maningas PA, Moore EE, et al. Prehospital hypertonic saline/dextran infusion for post-traumatic hypotension: the U.S.A. Multicenter Trial. Ann Surg. 1991;213:482–491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Martinez-Mier G, Toledo-Pereyra LH, Ward PA. Adhesion molecules and hemorrhagic shock. J Trauma. 2001;51:408–415. [DOI] [PubMed] [Google Scholar]
- 40.Maekawa K, Futami S, Nishida M, et al. Effects of trauma and sepsis on soluble L-selectin and cell surface expression of L-selectin and CD11b. J Trauma. 1998;44:460–468. [DOI] [PubMed] [Google Scholar]
- 41.Rainer TH, Ng MH, Lam NY, et al. Role of monocyte L-selectin in the development of post-traumatic organ failure. Resuscitation. 2001;51:139–149. [DOI] [PubMed] [Google Scholar]
- 42.Geissmann F, Jung S, Littman DR. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity. 2003;19:71–82. [DOI] [PubMed] [Google Scholar]
- 43.Ziegler-Heitbrock HW. Definition of human blood monocytes. J Leukoc Biol. 2000;67:603–606. [DOI] [PubMed] [Google Scholar]
- 44.Steppich B, Dayyani F, Gruber R, et al. Selective mobilization of CD14(+)CD16(+) monocytes by exercise. Am J Physiol Cell Physiol. 2000;279:C578–C586. [DOI] [PubMed] [Google Scholar]
- 45.McCarter MD, Mack VE, Daly JM, et al. Trauma-induced alterations in macrophage function. Surgery. 1998;123:96–101. [PubMed] [Google Scholar]
- 46.Kampalath B, Cleveland RP, Chang CC, et al. Monocytes with altered phenotypes in posttrauma patients. Arch Pathol Lab Med. 2003;127:1580–1585. [DOI] [PubMed] [Google Scholar]
- 47.Scherberich JE, Nockher WA. CD14++ monocytes, CD14+/CD16+ subset and soluble CD14 as biological markers of inflammatory systemic diseases and monitoring immunosuppressive therapy. Clin Chem Lab Med. 1999;37:209–213. [DOI] [PubMed] [Google Scholar]
- 48.Yoshioka Y, Ohwada A, Harada N, et al. Increased circulating CD16+ CD14dim monocytes in a patient with pulmonary alveolar proteinosis. Respirology. 2002;7:273–279. [DOI] [PubMed] [Google Scholar]
- 49.Belge KU, Dayyani F, Horelt A, et al. The proinflammatory CD14+CD16+DR++ monocytes are a major source of TNF. J Immunol. 2002;168:3536–3542. [DOI] [PubMed] [Google Scholar]
- 50.DeLong WG Jr, Born CT. Cytokines in patients with polytrauma. Clin Orthop 2004:57–65. [DOI] [PubMed] [Google Scholar]
- 51.Ferguson KL, Taheri P, Rodriguez J, et al. Tumor necrosis factor activity increases in the early response to trauma. Acad Emerg Med. 1997;4:1035–1040. [DOI] [PubMed] [Google Scholar]
- 52.Bergmann M, Sautner T. Immunomodulatory effects of vasoactive catecholamines. Wien Klin Wochenschr. 2002;114:752–761. [PubMed] [Google Scholar]
- 53.Orlic T, Loomis WH, Shreve A, et al. Hypertonicity increases cAMP in PMN and blocks oxidative burst by PKA-dependent and -independent mechanisms. Am J Physiol Cell Physiol. 2002;282:C1261–C1269. [DOI] [PubMed] [Google Scholar]
- 54.Hirsh M, Dyugovskaya L, Bashenko Y, et al. Reduced rate of bacterial translocation and improved variables of natural killer cell and T-cell activity in rats surviving controlled hemorrhagic shock and treated with hypertonic saline. Crit Care Med. 2002;30:861–867. [DOI] [PubMed] [Google Scholar]


