Abstract
We have developed a strategy for multiplex PCR based on PCR suppression. PCR suppression allows DNA target amplification with only one sequence-specific primer per target and a second primer that is common for all targets. Therefore, an n-plex PCR would require only n + 1 primers. We have demonstrated uniform, efficient amplification of targeted sequences in 14-plex PCR. The high specificity of suppression PCR also provides multiplexed amplification with allele specificity. Multiplexed PCR was used to develop assays for genotyping DNA samples from cystic fibrosis-affected individuals. The new approach greatly simplifies primer design, significantly increases the PCR multiplexing level, and decreases the overall primer cost. In addition, this assay is more readily amenable to automation and is therefore suitable for high-throughput genetic diagnostics.
The very high complexity of mammalian genomes requires preamplification of DNA targets before analysis. Most mutation detection techniques use PCR as a necessary first step for complexity reduction. Multiplex PCR (mpxPCR) provides simultaneous amplification of many targets of interest in one reaction (1, 2), thus increasing the assay throughput and allowing more efficient use of each DNA sample. Most multiplex reactions, however, are restricted to amplification of five to ten targets (3). One reason for this is that with each primer set added, the reaction conditions allowing efficient amplification of each DNA fragment become increasingly less flexible. In addition, primer–primer interactions increase the probability of primer-dimer formation. As a result, primer design for mpxPCR is not a trivial task and often fails in experimental trials (2).
Several attempts have been made to overcome some of the difficulties with mpxPCR. One of the suggested methods is based on an additional preamplification step yielding a long DNA fragment, which is then used as a template for reamplification of short amplicons (4–6). This approach was used successfully in several studies where multiple exons of a gene of interest were studied in parallel (6, 7). This method, however, does not offer a general solution for amplification of multiple targets from different parts of complex genomes. Another improvement to mpxPCR, yielding rather uniform amplification of all PCR products, uses two rounds of amplification. The first round is performed with a relatively low concentration of gene-specific primers containing a universal or zip code sequence, whereas the second round uses a high concentration of shorter primers corresponding to the zip code (8, 9). Special primer design has also been proposed to decrease primer–primer interactions (10). Nevertheless, the maximal achievable multiplexing by using conventional PCR remains restricted to about 10 targets and requires tedious optimization of PCR conditions (3, 11).
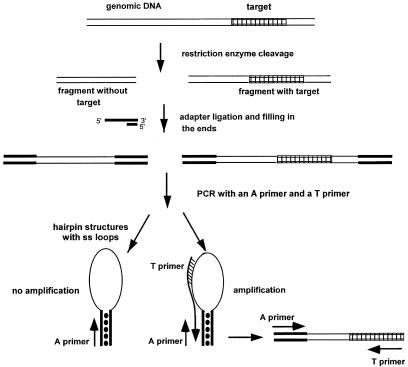
Here we describe a new procedure for increasing the PCR multiplexing level. We use the PCR suppression (PS) effect (12, 13). This allows PCR amplification with only one target-specific primer; another primer is common for all targets and corresponds to an adapter ligated to both ends of all genomic fragments (Fig. 1). In PS-based PCR (PS PCR) the adapter is about 40 bases long and has a high GC content. During PCR, after each denaturation step, self-complementary GC-rich ends of single-stranded (ss) fragments form strong duplexes; thus, all ss DNA fragments adopt hairpin structures. Replication of such DNA fragments using the primer corresponding to the adapter (A primer) is suppressed. However, synthesis can occur from a primer (T primer) complementary to a target located within the ss loop of the hairpin structure, and this product is then amplified efficiently by the A and T primers. As a result, efficient PCR is possible only for target-containing fragments. Because the A primer is the same for all targets, such PCR allows amplification with only one target-specific primer. PS PCR has been used successfully in a variety of applications: genome walking (13), cDNA subtractive hybridization (14–16), and targeted genomic differential display (TGDD; refs. 17 and 18). Here we show that PS PCR also allows efficient multiplex target amplification with allele specificity.
Figure 1.
Outline of the PS PCR procedure.
Materials and Methods
Samples.
Nonphosphorylated oligonucleotides, unlabeled or fluorescently labeled, were purchased as custom synthesis products from Integrated DNA Technologies (Coralville, IA). DNA from peripheral blood lymphocytes from anonymous donors was isolated by using a Qiagen Blood kit according to the manufacturer's protocol (Qiagen, Chatsworth, CA). DNA samples from cystic fibrosis affected individuals were kindly provided by R. Nelson and B. Allitto (Genzyme).
AmpliTaq and AmpliTaq Gold DNA polymerases were from Applied Biosystems, KlenTaq DNA polymerase from Ab peptides (St. Louis), and Taq DNA polymerase from Amersham Pharmacia.
Preparation of adapter-ligated DNA was performed as described in ref. 18. Human genomic DNA was digested with RsaI restriction enzyme (New England Biolabs) and ligated with adapters consisting of two annealed oligonucleotides: TGTAGCGTGAAGACGACAGAAAGGGCGTGGTGCGGACGCGGG and CCCGCGTCCGC. The complementary oligonucleotides were of different lengths to ensure the right polarity of ligation to blunt-ended genomic fragments. The recessed ends of the ligated DNA fragments were automatically filled in during the first round of subsequent PCR in the presence of dNTPs and DNA polymerase.
DNA Amplification.
Adapter-ligated DNA (2–5 ng) was amplified by PCR in a 25 μl reaction volume containing 1 × PCR buffer (depending on the DNA polymerase), 2.5 mM MgCl2, 250 μM of each dNTP, 5–10 pmol of each primer, and 2.5 units of thermostable DNA polymerase. The PCR mixtures containing all components but primers were denatured at 95°C for 3–10 min; the primer mixture (5–10 pmol of each) was added at 95°C; and 38 cycles of PCR (94°C for 10 sec, 68°C for 15 sec, and 72°C for 1 min) were performed. The A primer common for all targets was fluorescein-labeled, and the PCR products were analyzed by electrophoresis on a 2% agarose gel and by 6% denaturing PAGE.
To perform genotyping of cystic fibrosis (CF) DNAs in a mpxPCR, DNA samples were preamplified in a 15 cycle PCR with primers 1, 2, 4 (Table 1), the nested cystic fibrosis transmembrane conductance regulator (CFTR) primer 12 (Table 2), and the A primer, TGTAGCGTGAAGACGACAGAA. Then 1 μl of 500-fold diluted PCR product was reamplified in a 38-cycle PCR with a mixture of four T primers (including CFTR normal or mutant primer, primers 10 or 11; Table 2) and the A primer, GAAAGGGCGTGGTGCGGACGCGG, by using the same PCR conditions described above.
Table 1.
PCR primers targeted at anonymous sequences in human chromosome 7 used in PS mpxPCR
| Primer (5′–3′) | Direction, length (nt), GC content | Product length (bp)
|
|
|---|---|---|---|
| Expected | Obtained | ||
| 1. AATGCCTGCCATGTATAAGCTACCCGGTC | Reverse, 29, 15/29 | 165 | 165 |
| 2. GTCCCGTCCCCATCCTCACAAGCTGTCGC | Reverse, 29, 19/29 | 285 | 290 |
| 3. AGTGCCCATGCCCGTGAGACCTGGAGAAG | Direct, 29, 18/29 | 507 | 510 |
| 4. CCGGAGGAAATTGGAGTAGACTCGGAAGAG | Direct, 30, 16/30 | 212 | 210 |
| 5. GCAGCCCCAAGCACCAAGCTGAGCAAACAG | Direct, 30, 18/30 | 970 | ≈1,000 |
| 150 | |||
Table 2.
PCR primers targeted at bialleleic loci and the CFTR locus used in PS mpxPCR
| Gene | Primer (5′–3′) | Direction, length (nt), GC content | Product length (bp)
|
|
|---|---|---|---|---|
| Expected | Obtained | |||
| 1. Antithrombin III | GGTCCCATCTCCTCTACCTGATACAGACTC | Reverse, 30, 16/30 | 604 | ∼600 |
| 2. Interleukin 1 α (IL-1 α) | n CTGCACTTGTGATCATGGTTTTAGAAATCATC | Direct, 32, 12/32 | 197 | 200 |
| m CTGCACTTGTGATCATGGTTTTAGAAATAATA | ||||
| 3. Neurofibromatosis 1 | n GAGGACCATGGCTGAGTCTCCTTTAGTGTCC | Reverse, 31, 17/31 | 276 | 280 |
| m GAGGACCATGGCTGAGTCTCCTTTAGTATCA | ||||
| 4. Aldolase B | GGCTTGACTTTCCAACACGGAGAAGCATTG | Direct, 30, 15/30 | 449 | 450* |
| 5. Alpha2-macroglobulin | n CCCTTACTCAAGTAATCACTCACCAGTGTTGAG | Reverse, 33, 15/33 | 135 | 135 |
| m CCCTTACTCAAGTAATCACTCACCAGTGTAGAA | ||||
| 6. Insulin-like growth factor II | ACCCTGAAAATTCCCGTGAGAAGGGAGATGGC | Reverse, 32, 17/32 | 468 | 470 |
| 7. Triglyceride lipase, exon 4 | CAACACACTGGACCGCAAAAGGCTTTCATC | Direct, 30, 15/30 | 155 | 155 |
| 8. Integrin B2 subunit | CGGGCGCTGGGCTTCACGGACATAGTGACC | Reverse, 30, 20/30 | 400 | 400 |
| 9. Low-density lipoprotein receptor | CAGAGACAGTGCCCAGGACAGAGTCGGTCC | Reverse, 30, 19/30 | 365 | 365 |
| 10. CFTR (n) | GACGCTTCTGTATCTATATTCATCATAGGAAACACCAAAG | Reverse, 40, 15/40 | 355 | 350 |
| 11. CFTR ΔF508 | GACGCTTCTGTATCTATATTCATCATAGGAAACACCAAT | Reverse, 39, 14/39 | 355 | 350 |
| 12. CFTR, nested | TCTTCTAGTTGGCATGCTTTGATGACGCT | Reverse, 29, 13/29 | 372 | ND |
Primers targeted at the interleukin 1 α gene, neurofibromatosis 1 locus, and α2-macroglobulin were used as a normal (n) and a double mismatch (m) variant. ND, not determined.
This product appeared as a double peak (see Fig. 3).
Display and Analysis of PCR Products.
PCR products (2–3 μl) were denatured for 3 min at 90°C in a stop solution (Amersham Pharmacia), and analyzed by using a 6% denaturing PAGE and an automated laser fluorescence (ALF) sequencing instrument (Amersham Pharmacia) as described (17, 18).
Sequencing of CFTR Fragments.
CFTR DNA fragments were amplified from several DNAs by using direct fluorescein-labeled GGGAGAACTGGAGCCTTCAGAG and reverse GGGTAGTGTGAAGGGTTCATATGC primers. PCR product (5–10 fmol) was sequenced by using a fmol DNA sequencing kit (Promega) according to the manufacturer's protocol and the products were resolved by 6% PAGE in an ALF sequencing instrument (Amersham Pharmacia).
Results
General Considerations.
PS PCR relies on hairpin structures formed by all genomic fragments after DNA is digested with a restriction enzyme, ligated with long GC-rich PS adapters, denatured, and reannealed (Fig. 1). PS PCR usually uses primers that tolerate high primer-annealing temperatures (13). As a result, the specificity of PS PCR is extremely high (13, 17, 18).
The available experimental data suggest that PS PCR is well suited for multiplex PCR. Several issues, however, need to be addressed to optimize this method for multiplex amplification. To perform PS PCR, genomic DNA should be digested with an appropriate restriction enzyme and ligated with PS adapters. Thus, the design of the gene-specific primers will depend on the location of the target and also on the availability of a proximal restriction site. Another problem, which is inherent to all PCR-based methods with multiple targeting, is preferential amplification of certain templates and loss of others during repeated PCR cycles.
14-Plex PCR Using Human Genomic DNA.
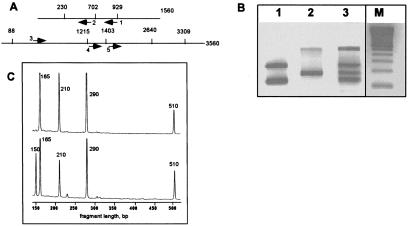
To demonstrate PS-based mpxPCR, we chose two fragments (1560 and 3560 base pairs) from a large sequenced segment on human chromosome 7q22 (accession no. AF053356), and generated their RsaI maps. Five primers were chosen to amplify five corresponding targets (see Fig. 2A for the scheme and Table 1). Human genomic DNA from an anonymous blood donor was digested with RsaI restriction enzyme and ligated with PS-adapters. The specificity of all primers was first tested in single reactions by using the A primer and the corresponding T primer. In all but one case (primer 5 in Table 1), single products of the expected sizes were generated. Primer 5 generated one additional fragment of smaller than expected size (Table 1). The nature of this product was not studied. Next, PCRs with different multiplexing by using equimolar mixtures of T primers and the A primer were performed. In these experiments, the A primer was fluorescently labeled, and 35–38 cycles of PCR were performed to visualize products from 2–5 ng of genomic DNA.
Figure 2.
PS-based mpxPCR targeted at several anonymous sequences in chromosome 7 DNA. 5 ng of RsaI-digested human genomic DNA with ligated adapter was amplified in a 25 μl reaction containing 1 × PCR buffer (PE), 2.5 mM MgCl2, 250 μM dNTP, 2.5 units of thermostable DNA polymerase [a (1:1:1) mixture of Taq, AmpliTaq, and AmpliTaq Gold DNA polymerases] and 5 pmol of each primer. The fluorescently labeled A primer was TGTAGCGTGAAGACGACAGAA, which corresponded to the 5′ outermost part of the ligated adapter. For the T primers see Table 1. (A) RsaI restriction maps of two fragments from human chromosome 7 with the PCR primers. (B) Resolution of mpxPCR products by using 2% agarose gel electrophoresis. (1) PCR with the T primers 1 + 2; (2) PCR with the T primers 3 + 4; (3) PCR with the T primers 1 + 2 + 3 + 4 (see Table 1); (M) 100-bp size marker. (C) Resolution of the 4-plex (Top; primers 1–4 in Table 1) and 5-plex (Bottom; primers 1–5 in Table 1) PCR products by using 6% PAGE and an ALF sequencer. The numbers above the peaks show the amplicon lengths. The 1-kb-long PCR amplicon is beyond the displayed window.
Fig. 2B shows the products of 2-plex and 4-plex PCRs after agarose gel electrophoresis and Fig. 2C presents 4-plex and 5-plex PCR products after resolution on polyacrylamide gels. All targeted amplicons were detected as PCR products, and the amounts of amplicons in the range 100–400 base pairs were similar. However, an ≈500 bp amplicon was synthesized in smaller amounts (see also Fig. 4 and below).
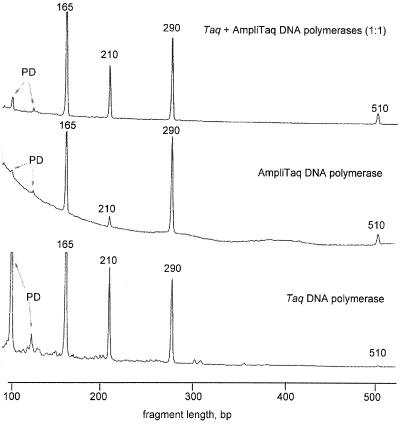
Figure 4.
Different DNA polymerases display different specificity in mpxPCR. 4-plex PCR used primers 1, 2, 3, and 4 in Table 1. Thermostable DNA polymerase (2.5 units) or a mixture of two DNA polymerases (2.5 units) was used in PCR. Other conditions were as in Fig. 2. PD, primer-dimers.
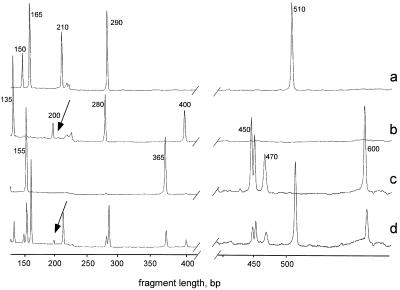
In a different experiment, we chose to amplify portions of 12 genes known to have single base variations at biallelic loci (9). The primers were taken from Belgrader et al. (9) and extended by several bases (for different primers, extension was from different sides or from both sides), so that all gene-specific primers were 30–33 bases long, depending on the GC content. The choice between the direct or reverse primer was determined by the location of the nearest RsaI site. All 12 primers were tested in single reactions, and three primers (cytochrome 450IID gene, C6 complement gene, and S-beta pseudogene) were excluded from our subsequent experiments because of inadequate specificity. The primer for the aldolase B gene (primer 4 in Table 2) generated a PCR product, which appeared as a double peak on a polyacrylamide gel. Multiplex PCR with different combinations of gene-specific primers, including a mixture of all nine T primers, generated bands with sizes that corresponded to the expected ones. Fig. 3 a–c show the patterns obtained in 5-plex PCR (primers 1–5 in Table 1), 4-plex PCR (primers 2, 3, 5 and 8 in Table 2), and 5-plex PCR (primers 1, 4, 6, 7, and 9 in Table 2). Finally, we performed a 14-plex PCR, in which we combined the primers for the five fragments from chromosome 7 (Table 1) and nine gene-specific primers from Table 2 (primers 1–9). In this experiment, all 14 targeted amplicons were detected (Fig. 3d). The relative amounts of the amplicons inversely correlated with the size of the fragments and also depended of the annealing temperature (Tm) of the T primer (Table 3, see also Discussion). For example, the relatively low concentration of the interleukin 1-α amplicon (Fig. 3, marked by arrow) correlated with the lowest Tm of the corresponding primer.
Figure 3.
14-fold mpxPCR. (a) A 5-plex PCR was performed with primers 1–5 (Table 1); (b) 4-plex PCR with primers 2, 3, 5, and 8 (Table 2); (c) 5-plex PCR with primers 1, 4, 6, 7, and 9 (Table 2): (d) 14-plex PCR with primers 1–5 (Table 1) plus primers 1–9 (Table 2). Primers 2, 3, and 5 (Table 2) were normal types. Other conditions were as in Fig. 2. The 450-bp fragment (aldolase B gene; primer 4 in Table 2) appeared as a double peak, as it also did in a uniplex PCR (data not shown). The 1-kb-long fragment (product generated by the primer 5 in Table 1) is beyond the displayed window. The fragment's lengths (bp) are indicated above the peaks. The 200-bp-long IL2 amplicon is marked by an arrow.
Table 3.
Dependence of the amplicon yield on the Tm of the primer in the multiplex PS PCR
| Primer | Tm, °C | Product size, bp | ss loop size, nt | Peak area, arbitrary units |
|---|---|---|---|---|
| no. 2, Table 2 | 60.4 | 200 | 170 | 310 |
| no. 5, Table 2 | 63.4 | 135 | 308 | 1,020 |
| no. 4, Table 1 | 64.7 | 210 | 188 | 2,180 |
| no. 7, Table 2 | 65.7 | 155 | 607 | 2,590 |
| no. 1, Table 1 | 65.7 | 165 | 227 | 4,630 |
The Tm's of the primers were calculated using a nearest-neighbor thermodynamic parameter set (ref. 19, see also http://www.idtdna.com/technotes_facs/Calculating_Tm). Peak areas were determined by analyzing a 14-plex PCR pattern using Fragment manager software provided with the ALF sequencing instrument.
Different DNA Polymerases Display Different Specificity in mpxPCR.
Three parameters are important in evaluating the quality of multiplex amplification reaction: (i) the uniformity of amplification of different targets; (ii) the amount of primer-dimers; and (iii) the nonspecific background or signal to background ratio. Fig. 4 presents the results obtained with two different DNA polymerases, AmpliTaq and Taq DNA polymerase, in an experiment designed to amplify four amplicons (primers 1–4 in Table 1). The data show that Taq DNA polymerase amplifies 100- to 400-bp amplicons more uniformly than AmpliTaq; however, this is accompanied by a greater amount of primer-dimers (Fig. 4). In addition, the 510-bp fragment is amplified by Taq DNA polymerase much less efficiently than by AmpliTaq. At the same time, AmpliTaq DNA polymerase generates much higher nonspecific background and amplifies 100–400 bp amplicons less uniformly that Taq DNA polymerase. To keep a low background and low primer-dimers and still amplify all of the targets, we used a 1:1 mixture of both polymerases, which produced the best result (Fig. 4). We also tested KlenTaq and AmpliTaq Gold DNA polymerases and found that the combination of these DNA polymerases with AmpliTaq or Taq DNA polymerase also generates a good amplification pattern with low background (data not shown). At the moment, we routinely use a mixture (1:1:1) of three different DNA polymerases, AmpliTaq, AmpliTaq Gold, and Taq DNA.
Allele-Specific mpxPCR.
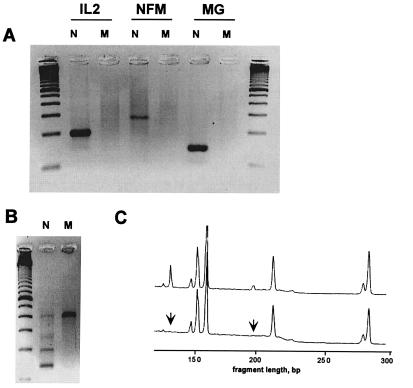
To be useful for mutation detection the PCR should discriminate between alleles, and single-base discrimination should be routinely attainable. We performed mpxPCR with primers containing a 3′-terminal mismatched base, imitating allele-specificity. In addition, a penultimate base was mismatched to increase the impact of the 3′-mismatch (20–22). Fig. 5A shows that uniplex PS PCR, with such mismatched primers targeting interleukin, neurofibromatosis, and α2-macroglobulin genes, perfectly discriminated against a 3′-end mismatch nucleotide. Next, we performed a 4-plex PCR with a mixture of these three primers, either wild or mutated, together with a fourth primer, which was the same in both wild and mutant reactions (it was targeted at an integrin B2 subunit gene fragment, primer 8 in Table 2). Fig. 5B shows an agarose gel where three bands, corresponding to interleukin, neurofibromatosis, and α2-macroglobulin gene fragments, are essentially absent when the corresponding mismatched primers were used for amplification. In addition, Fig. 5C shows the absence of the interleukin and macroglobulin gene fragments amplified in a 14-plex PCR with the corresponding mutated primers. These data suggest that this method can be used for multiplex genotyping of DNA samples.
Figure 5.
Allele-specific mpxPCR. (A) Uniplex reactions with normal (N) and mutated (M) primers targeting the interleukin-2 gene (IL2), neurofibromatosis (NFM) gene, and α2-macroglobulin (MG) gene. (B) A 4-plex PCR with an equimolar mixture of normal type (N) or mutant (M) primers. The fourth target in both reactions was a fragment of the integrin B2 subunit gene (primer 8 in Table 2). The PCR products were resolved on a 2% agarose gel. (C) A window showing the absence of two targets (MG and IL2, marked by arrows) amplified with the mutant primers in a 14-plex PCR. PCR products were resolved by using 6% PAGE in an ALF sequencing instrument.
Genotyping of DNA Samples from Cystic Fibrosis-Affected Individuals By Using the PS-Based mpxPCR.
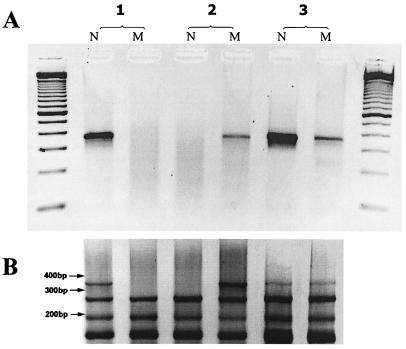
To experimentally test the applicability of mpx PS PCR in disease diagnostics we performed genotyping of DNA samples from CF-affected individuals with the ΔF508 mutation consisting in a deletion of three nucleotides in exon 10 of the CFTR gene (23). The region around the ΔF508 mutation is extremely AT-rich; therefore, 39–40 base-long reverse CFTR primers were designed to survive the 68°C annealing temperature (Table 2). The choice of the reverse primer was determined by the location of an RsaI site about 300 bp upstream of the ΔF508 mutation point. The normal and mutated primers were designed to differ by one nucleotide in length and also by the 3′-terminal nucleotide (Table 2). However, preliminary experiments showed that neither primer provided adequate specificity. Therefore, in this particular case we performed nested PCR and used in the first step a 29-mer nested CFTR primer (primer 12 in Table 2) in combination with the 5′-outmost adapter primer in a 15 cycle PCR. Then, 1 μl of the PCR product was diluted 500-fold and reamplified in a 38 cycle PCR with either normal or mutant CFTR primers (primers 10 or 11, respectively) and 3′ A primer (see Materials and Methods). Fig. 6A shows that only the normal CFTR primer amplifies an expected 350-bp fragment from unaffected DNA; both primers generate products from CFTR-heterozygous DNA; and only a mutant primer amplifies a product from ΔF508 CFTR-homozygous DNA. The lower efficiency of PCR with the CFTR mutant primer (Fig. 6A) is due to the shorter length and, consequently, the lower Tm of the corresponding primer (see Table 2). It is remarkable, however, that the difference between the normal and mutated CFTR primers is practically eliminated in mpxPCR (see below and Fig. 6B).
Figure 6.
Genotyping of CF human DNA samples. (A) Uniplex reactions with primers targeting normal type (1), ΔF508 CFTR-homozygous (2), and ΔF508 CFTR-heterozygous (3) DNA. The PCR products were analyzed on a 2% agarose gel. (B) Genotyping of the same DNA samples in a 4-plex PCR. In this reaction, CFTR normal type (N) and CFTR mutant (M) primers (primers 10 and 11, respectively, in Table 2) were used in a mixture with primers 1, 2, and 4 (Table 1) (see Materials and Methods for the details).
To demonstrate the possibility of multiplex allele-specific PCR, we performed genotyping of the CFTR locus on the background of 3 other loci (primers 1, 2, and 4 in Table 1) in five DNA samples, which supposedly contained three homozygous and two heterozygous ΔF508 CFTR mutations. In these experiments, we performed two-step mpxPCR as described above. Fig. 6B exemplifies the results. ΔF508 CFTR homozygous DNA samples were positive with the mutant CFTR primer and negative with the normal CFTR primer (Fig. 6B). Heterozygous ΔF508 CFTR samples showed the presence of the CFTR fragment with both normal and mutant CFTR primers, thus, confirming ΔF508 heterozygosity. In all DNAs tested by multiplex genotyping, the status of CFTR mutation was identified correctly and was confirmed by direct sequencing.
Discussion
We have developed an approach to mpxPCR based on the PS effect (13, 24). This new approach requires half the number of primers as compared to conventional mpxPCR. This substantially simplifies primer design and brings down primer costs. Primer cost savings are at least 1.5-fold. For example, in conventional PCR aimed to amplify 12 biallelic loci (9) the average length of the sum of the two gene-specific primers was 45 (38–62)-nt, whereas in the PS PCR the same targets were amplified with an average primer that was 31 (30–33)-nt long (see primers 1–9 in Table 2), and only a single labeled primer was required.
By using this method, we have performed 14-plex PCR targeting DNA fragments from various human chromosomes. It is important to emphasize that all T primers efficient in single reactions were efficient also in mpxPCR, and none of the T primers was found to be noncompatible with others. Therefore, we believe that this approach can be adapted for various kinds of studies. We are also positive that the multiplexing level can be increased further.
The preparation of DNA samples for PS PCR includes digestion of genomic DNA with an appropriate restriction enzyme and ligation with the PS-driving adapters. These extra steps, however, would not be burdensome if the DNA samples prepared could be used in multiple experiments. It would be most desirable, therefore, to choose one restriction enzyme for all targets. In our study, we did not have to drop any of the targets because of the lack of a RsaI site in the range accessible to amplification. Therefore, we believe that the choice of a single restriction enzyme for most of the targets should not be a problem, and a suitable site will be available in proximity to practically every target.
The PCR conditions, which we used for mpxPCR, did not require special optimization and should be considered as default conditions applicable for amplification of any target, provided the primer's Tm tolerates 65–68°C. We have found that the PCR cycling conditions have little impact on the amplification result (data not shown). We assume that the hairpin-loop structures acquired by all genomic fragments present a positive factor for efficient target amplification. Indeed, all of the targets are located within the ss regions of the hairpin-loop structures and are, therefore, available to corresponding primers for binding. This may explain the fact that the relative amplification efficiency of similar sized targets in mpxPCR correlates with the Tm of the primers (Table 3). The size of the ss loop seems to be another factor affecting the efficiency of PS PCR. A target residing in a larger ss loop (≈600-nt) is amplified less efficiently than a target located in a smaller ss loop (227-nt; see Table 3). This may occur because larger ss loops form stable secondary structures more easily than shorter ones; this would inhibit annealing of the corresponding T primer and decrease the yield of the amplicon. The size of the ss loop is determined by the distance between the two restriction sites and cannot be regulated by the experimenter. However, the decreased efficiency of amplification of targets with large ss loops can be partly compensated for by increasing the length of the primer and consequently its Tm.
The general trend of decreased PCR amplification efficiency for longer targets is seen also in mpx PS PCR. The necessity of different size targets is determined solely by the use of gel-based methods for resolution and analysis of the amplification products. If the analysis procedure avoids gel-based separation methods [e.g., DNA microarray-based analysis of the PCR products (25) or mass spectrometry (26)], it will be possible to design primers to generate PCR products of very similar size. This should help in more uniform amplification of different targets and might further increase the level of achievable multiplexing.
Different thermostable DNA polymerases displayed different specificity in PS mpxPCR. Mixtures of DNA polymerases generated more uniform amplification patterns than individual DNA polymerases (Fig. 4). We speculate that minor differences in strand displacement activity and processivity of different DNA polymerases may cause differences in specificity. Mixes of DNA polymerases have already been used in PCR for other purposes. For example, a combination of two thermostable DNA polymerases, one of which had 3′–5′ proofreading activity, has been shown to increase the length of PCR amplification (27).
In PS mpxPCR, the criteria for primer selection are, in general, the same as in conventional mpxPCR: the primers should not be self complementary or mutually complementary and should not form homo- and heterodimers (28). Because PS PCR requires only one specific primer, its uniqueness is crucial. Therefore, the primers are usually relatively long oligonucleotides with high GC contents. This allows high annealing temperatures and thereby increases the specificity (13). To increase the specificity further, one can perform nested PCR (13, 18, 24). For the purposes of multiplexing, however, nested PCR may not be required. Our data show that 30- to 32-base-long target-primers combined with the 5′-outmost adapter primer provide specific amplification for most of the targets (see Tables 1 and 2), and, therefore, nested primers are not obligatory for successful multiplexing. During this study, only one target was encountered, which was not amplified with 30- to 32-base-long primers (CFTR gene). The region around the ΔF508 mutation is extremely AT-rich, and to reach the Tm's of 67–68°C it was necessary to use 40-base-long primers and perform nested PCR from the target side. However, in all other cases we successfully generated PCR amplicons by performing one-step PCR. This substantially simplified the protocol compared with two-step procedures.
The necessity of only one gene-specific primer adds much flexibility to primer design and allows the use of primers complementary to either of the two DNA strands, depending on the availability of the closest restriction site. This is especially important for amplification of homologous gene-family members or repetitive sequences, where it is often difficult to choose two distinct specific primers for each gene-family member.
The extremely high specificity of PS PCR allows allele-specific amplification with single-base discrimination. In our experiments we tested G/A and C/A mismatches which display moderate destabilizing effects (29) and correspond to common C → T and G → T variations (30). The fact that allele specificity has been attained in a 14-plex PCR, demonstrates the exquisite fidelity of this approach. Another advantage of this technique is its amenability to automation and development of high-throughput genetic diagnostics. For example, by performing 14-plex PCR in 96-well microtiter plate, one will be able to analyze 192 chromosomes at 14 loci simultaneously. This method can be easily combined with various advanced techniques, e.g., two-color detection and microarray-based analysis of the PCR products, which will increase further the throughput and information content of every experiment.
Acknowledgments
We thank Daniel Little of Sequenom, Rosemary Nelson and Bernice Allitto of Genzyme for the human CF DNA samples, and Vadim Demidov and Joel Graber of Boston University for valuable suggestions and critical reading of the manuscript.
Abbreviations
- mpxPCR
multiplex PCR
- PS
PCR suppression
- ss
single-stranded
- CF
cystic fibrosis
- CFTR
cystic fibrosis transmembrane regulator
- ALF
automated laser fluorescence
- Tm
annealing temperature
References
- 1.Chamberlain J S, Gibbs R A, Ranier J E, Nguyen P N, Caskey C T. Nucleic Acids Res. 1988;16:11141–11156. doi: 10.1093/nar/16.23.11141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Edwards M C, Gibbs R A. PCR Methods Appl. 1994;3:S65–S75. doi: 10.1101/gr.3.4.s65. [DOI] [PubMed] [Google Scholar]
- 3.Hacia J G, Sun B, Hunt N, Edgemon K, Mosbrook D, Robbins C, Fodor S P, Tagle D A, Collins F S. Genome Res. 1998;8:1245–1258. doi: 10.1101/gr.8.12.1245. [DOI] [PubMed] [Google Scholar]
- 4.Li D, Vijg J. Nucleic Acids Res. 1996;24:538–539. doi: 10.1093/nar/24.3.538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stuven T, Griese E U, Kroemer H K, Eichelbaum M, Zanger U M. Pharmacogenetics. 1996;6:417–421. doi: 10.1097/00008571-199610000-00005. [DOI] [PubMed] [Google Scholar]
- 6.van Orsouw N J, Zhang X, Wei J Y, Johns D R, Vijg J. Genomics. 1998;52:27–36. doi: 10.1006/geno.1998.5410. [DOI] [PubMed] [Google Scholar]
- 7.Rines R D, van Orsouw N J, Sigalas I, Li F P, Eng C, Vijg J. Carcinogenesis. 1998;19:979–984. doi: 10.1093/carcin/19.6.979. [DOI] [PubMed] [Google Scholar]
- 8.Shuber A P, Grondin V J, Klinger K W. Genome Res. 1995;5:488–493. doi: 10.1101/gr.5.5.488. [DOI] [PubMed] [Google Scholar]
- 9.Belgrader P, Marino M M, Lubin M, Barany F. Genome Science & Technology. 1996;1:77–87. [Google Scholar]
- 10.Brownie J, Shawcross S, Theaker J, Whitcombe D, Ferrie R, Newton C, Little S. Nucleic Acids Res. 1997;25:3235–3241. doi: 10.1093/nar/25.16.3235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hacia J G, Sun B, Hunt N, Edgemon K, Mosbrook D, Robbins C, Fodor S P, Tagle D A, Collins F S. Genome Res. 1998;8:1245–1258. doi: 10.1101/gr.8.12.1245. [DOI] [PubMed] [Google Scholar]
- 12.Launer G A, Lukyanov K A, Tarabykin V S, Lukyanov S A. Mol Genet Mikrobiol Virusol. 1994;6:38–41. [PubMed] [Google Scholar]
- 13.Siebert P D, Chenchik A, Kellogg D E, Luckyanov K A, Lukyanov S A. Nucleic Acids Res. 1995;23:1087–1088. doi: 10.1093/nar/23.6.1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Diatchenko L, Lau Y F, Campbell A P, Chenchik A, Moqadam F, Huang B, Lukyanov S, Lukyanov K, Gurskaya N, Sverdlov E D, Siebert P D. Proc Natl Acad Sci USA. 1996;93:6025–6030. doi: 10.1073/pnas.93.12.6025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Akopyants N S, Fradkov A, Diatchenko L, Hill J E, Siebert P D, Lukyanov S A, Sverdlov E D, Berg D E. Proc Natl Acad Sci USA. 1998;95:13108–13113. doi: 10.1073/pnas.95.22.13108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Diatchenko L, Lukyanov S, Lau Y F, Siebert P D. Methods Enzymol. 1999;303:349–380. doi: 10.1016/s0076-6879(99)03022-0. [DOI] [PubMed] [Google Scholar]
- 17.Broude N E, Storm N, Malpel S, Graber J H, Lukyanov S, Sverdlov E, Smith C L. Genet Anal. 1999;15:51–63. doi: 10.1016/s1050-3862(98)00038-2. [DOI] [PubMed] [Google Scholar]
- 18.Lavrentieva I, Broude N E, Lebedev Y, Gottesman I I, Lukyanov S A, Smith C L, Sverdlov E D. FEBS Lett. 1999;443:341–347. doi: 10.1016/s0014-5793(99)00004-6. [DOI] [PubMed] [Google Scholar]
- 19.Allawi H T, SantaLucia J., Jr Biochemistry. 1997;36:10581–10594. doi: 10.1021/bi962590c. [DOI] [PubMed] [Google Scholar]
- 20.Norby S, Lestienne P, Nelson I, Rosenberg T. Biochem Biophys Res Commun. 1991;175:631–636. doi: 10.1016/0006-291x(91)91612-g. [DOI] [PubMed] [Google Scholar]
- 21.Cha R S, Zarbl H, Keohavong P, Thilly W G. PCR Methods Appl. 1992;2:14–20. doi: 10.1101/gr.2.1.14. [DOI] [PubMed] [Google Scholar]
- 22.Matsunaga J, Tomita Y, Tagami H. Exp Dermatol. 1995;4:377–381. doi: 10.1111/j.1600-0625.1995.tb00063.x. [DOI] [PubMed] [Google Scholar]
- 23.Zielenski J, Rozmahel R, Bozon D, Kerem B, Grzelczak Z, Riordan J R, Rommens J, Tsui L C. Genomics. 1991;10:214–228. doi: 10.1016/0888-7543(91)90503-7. [DOI] [PubMed] [Google Scholar]
- 24.Lukyanov K A, Launer G A, Tarabykin V S, Zaraisky A G, Lukyanov S A. Anal Biochem. 1995;229:198–202. doi: 10.1006/abio.1995.1402. [DOI] [PubMed] [Google Scholar]
- 25.Gerry N P, Witowski N E, Day J, Hammer R P, Barany G, Barany F. J Mol Biol. 1999;292:251–262. doi: 10.1006/jmbi.1999.3063. [DOI] [PubMed] [Google Scholar]
- 26.Tang K, Fu D-J, Julien D, Braun A, Cantor C R, Koester H. Proc Natl Acad Sci USA. 1999;96:10016–10020. doi: 10.1073/pnas.96.18.10016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Barnes W M. Proc Natl Acad Sci USA. 1994;91:2216–2220. doi: 10.1073/pnas.91.6.2216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rychlik W. Mol Biotechnol. 1995;3:129–134. doi: 10.1007/BF02789108. [DOI] [PubMed] [Google Scholar]
- 29.Cotton R G H. Biochem J. 1989;263:1–10. doi: 10.1042/bj2630001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cargill M, Altshuler D, Ireland J, Sklar P, Ardlie K, Patil N, Shaw N, Lane C R, Lim E P, Kalyanaraman N, et al. Nat Genet. 1999;22:231–238. doi: 10.1038/10290. [DOI] [PubMed] [Google Scholar]