Abstract
p38 mitogen-activated protein kinase (MAPK) regulates cytokines in arthritis and is, in turn, regulated by MAPK kinase (MKK) 3 and MKK6. To modulate p38 function but potentially minimize toxicity, we evaluated the utility of targeting MKK3 by using MKK3−/− mice. These studies showed that TNF-α increased phosphorylation of p38 in WT cultured synoviocytes but that p38 activation, IL-1β, and IL-6 expression were markedly lower in MKK3−/− synoviocytes. In contrast, IL-1β or LPS-stimulated p38 phosphorylation and IL-6 production by MKK3−/− synoviocytes were normal. Detailed signaling studies showed that NF-κB also contributes to IL-6 production and that TNF-α-induced NF-κB activation is MKK3-dependent. In contrast, LPS-mediated activation of NF-κB does not require MKK3. To determine whether this dichotomy occurs in vivo, two inflammation models were studied. In K/BxN passive arthritis, the severity of arthritis was dramatically lower in MKK3−/− mice. Phospho-p38, phospho-MAPK activator protein kinase 2, IL-1β, CXC ligand 1, IL-6, and matrix metalloproteinase (MMP) 3 levels in the joints of MKK3−/− mice were significantly lower than in controls. Exogenous IL-1β administered during the first 4 days of the passive model restored arthritis to the same severity as in WT mice. In the second model, IL-6 production after systemic LPS administration was similar in WT and MKK3−/− mice. Therefore, selective MKK3 deficiency can suppress inflammatory arthritis and cytokine production while Toll-like receptor 4-mediated host defense remains intact.
Keywords: cytokines, inflammation, rheumatoid arthritis, fibroblast, signal transduction
The mitogen-activated protein kinases (MAPKs) are signal transduction enzymes that integrate cellular responses to the environmental stresses. The pathways include parallel and interacting cascades that are initiated by activation of upstream MAPK kinase (MKK) kinases, which, in turn, activate MKKs. These enzymes then phosphorylate the three main MAPK families, extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK), and p38 MAPK. In addition to participating in many normal host functions, the MAPKs have been implicated in inflammatory diseases (1). p38, in particular, is thought to play a role in rheumatoid arthritis (RA). It is expressed and activated in the rheumatoid synovial intimal lining (2), and p38 inhibitors are effective in rodent models of arthritis (3). Therefore, p38 could serve as a therapeutic target (4, 5). Inhibition of this kinase in humans has been complicated by several problems, such as hepatotoxicity and skin rash (6). Additional concerns related to impaired resistance to infection have been raised with p38 inhibitors because of effects on host defense, including suppression of neutrophil killing and enhanced colony counts in bacterial infections (7, 8).
An alternative approach is to target upstream MKKs, such as MKK3 or MKK6, which could potentially regulate some p38 functions while leaving others intact. MKK3 and MKK6 are two closely related kinases that phosphorylate p38 at the Thr-Gly-Tyr site but do not activate ERK1/2 or JNK (9, 10). MKK4 is a JNK-activating kinase but is also able to phosphorylate p38 when it is overexpressed (11). The hierarchy of MKK3 and MKK6 varies considerably depending on the cell lineage and the type of environmental stimulus (12), raising the possibility that one could block an upstream kinase that will have an impact on only a subset of functions that are relevant to disease while leaving other p38-mediated responses intact.
We previously demonstrated that both MKK3 and MKK6 can phosphorylate p38 in cultured fibroblast-like synoviocytes (FLS) and are activated in RA synovium (13). The relative contributions of these two kinases to p38 activation in synoviocytes have also been examined by using dominant negative (DN) MKK3 and MKK6 constructs (14). Among the different MKK subtypes, these data suggested that MKK3 might play a more prominent role in p38-mediated cytokine and MMP expression in FLS. The present study was performed to determine the hierarchy of the p38 MAP kinases in arthritis and assess the potential for MKK3 as a therapeutic target. These experiments demonstrate that MKK3 deficiency markedly decreases synovial inflammation in the passive K/BxN model of arthritis, in part through defective IL-1β expression, while still permitting normal responses to LPS through a NF-κB-dependent mechanism.
Results
MAPK Signaling in TNF-α-Activated FLS.
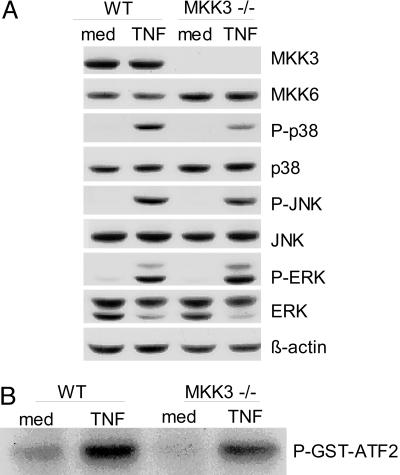
Expression of phospho-p38, phospho-JNK, and phospho-ERK in medium and TNF-α-stimulated cells was initially determined by Western blot analysis. TNF-α increased phosphorylation of p38 in the WT cells in a dose-dependent fashion, with maximum activation occurring at ≈10 ng/ml (data not shown). JNK and ERK phosphorylation were normal in MKK3−/− FLS, although phosphorylation of p38 was significantly lower in MKK3−/− cells (66% inhibition compared with WT; P < 0.05, n = 3) (Fig. 1A). In vitro kinase assays on WT and MKK3−/− FLS lysates were then performed by using GST-activating transcription factor (ATF) 2 as the substrate. As shown in Fig. 1B, marked increases of kinase activity were observed in TNF-α-stimulated FLS. Kinase activity was significantly lower in MKK3−/− cells compared with WT cells (42% inhibition; P < 0.05, n = 3).
Fig. 1.
MAPK signaling in TNF-α-stimulated WT and MKK3−/− FLS. Cultured FLS from WT and MKK3−/− mice were stimulated with medium or TNF-α for 15 min. Cell lysates were evaluated by Western blot analysis. (A) Representative blot shows inhibition of p38 but not JNK or ERK phosphorylation (n = 3). (B) Cell lysates were immunoprecipitated with anti-p38, and kinase activity was measured, using GST-ATF-2 as the substrate (n = 3).
MKK3-Dependent IL-6 and IL-1β Production After TNF-α Stimulation.
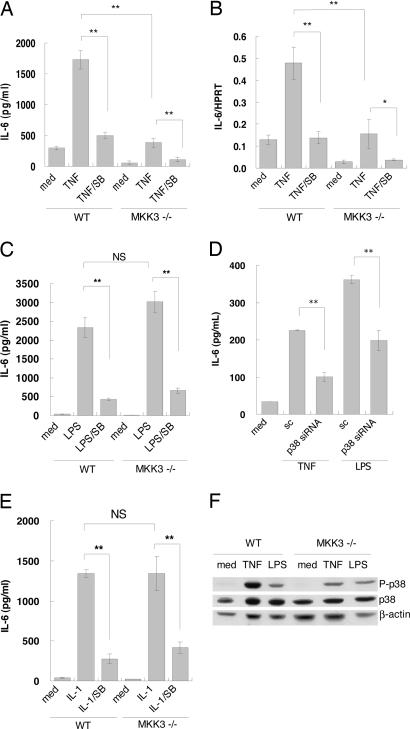
To determine whether MKK3 regulates production of p38-driven cytokines in FLS, WT and MKK3−/− cells were treated with medium or TNF-α, and culture supernatants were collected after 24 h. As shown in Fig. 2A, TNF-α significantly increased IL-6 production by WT FLS. However, IL-6 production by the MKK3−/− FLS after TNF-α stimulation was markedly lower than by WT cells (P < 0.01). The p38 inhibitor SB203580 (10 μM) also blocked IL-6 production by the WT cells, confirming p38 dependence of the TNF-α pathway. SB203580 further decreased IL-6 production in the MKK3−/− cells, suggesting that a small amount of residual p38 activation might still contribute. Quantitative real-time PCR showed that TNF-α-induced IL-6 mRNA accumulation was inhibited in the MKK3-deficient cells (Fig. 2B). As with IL-6, TNF-α-induced IL-1β mRNA induction was suppressed in the MKK3−/− FLS as well as by SB203580 (for WT FLS, TNF-α stimulation resulted in 16.2 ± 5.2-fold change in IL-1β/hypoxanthine phosphoribosyltransferase mRNA expression, and TNF-α plus SB203580 resulted in 0.9 ± 0.5-fold change; for MKK3−/− cells, TNF-α stimulation resulted in 3.0 ± 1.2-fold change, and TNF-α plus SB203580 resulted in 1.1 ± 1.0-fold change; P < 0.01 for WT compared with MKK3−/−, n = 4).
Fig. 2.
Regulation of TNF-α-, IL-1β-, or LPS-induced IL-6 expression by WT and MKK3−/− FLS. Cultured FLS from WT and MKK3−/− mice were stimulated with or without TNF-α, IL-1β, or LPS for 24 h. Culture supernatants and total RNA were collected. IL-6 protein and mRNA levels were measured by ELISA and real-time PCR, respectively. (A and B) TNF-α-induced IL-6 protein and mRNA were significantly decreased in MKK3−/− FLS. (C) LPS-induced IL-6 levels were similar in WT and MKK3−/− FLS. However, SB203580 (10 mM) blocked IL-6 production. (D) Effect of p38α siRNA knock-down. FLS were transfected with either p38α siRNA or scrambled control (sc) and stimulated with TNF-α or LPS, and supernatants were assayed for IL-6 by ELISA (n = 4). Knock-down was confirmed by Western blot analysis (not shown). siRNA blocked IL-6 production, although the presence of other p38 isoforms or small amounts of residual p38α could still affect the results. (E) IL-1β-induced IL-6 levels were not changed in WT and MKK3−/− FLS. However, both are blocked by SB203580 (10 mM). ∗, P < 0.05; ∗∗, P < 0.01 compared with WT, TNF-α, IL-1β, or LPS control. (F) Cultured FLS from WT and MKK3−/− mice were stimulated with TNF-α or LPS for 15 min. Western blot analysis was performed to evaluate p38 activation. TNF-α was a more potent inducer of phospho-p38 than LPS in WT FLS. However, LPS-stimulated p38 phosphorylation was independent of MKK3.
MKK3-Independent IL-6 and IL-1β Production After Toll-Like Receptor (TLR) 4 and IL-1β Stimulation.
To determine the effect of MKK3 deficiency on TLR4-dependent innate immune responses, we examined the contribution of MKK3 to LPS-induced cytokine production by FLS. As shown in Fig. 2C, LPS-induced IL-6 protein production from MKK3−/− FLS was similar to WT FLS and was blocked by SB203580. Similar results were obtained by using another selective p38 inhibitor, SB202190 (data not shown). In addition, p38α small interfering RNA (siRNA) knock-down also inhibited LPS- and TNF-α-induced IL-6 production (Fig. 2D). LPS-induced IL-1β mRNA expression was also similar in WT and MKK3−/− FLS (IL-1β/hypoxanthine phosphoribosyltransferase mRNA induction was 5.9 ± 0.4-fold and 4.7 ± 1.6-fold, respectively; P > 0.10). As shown in Fig. 2E, MKK3 deficiency had no effect on IL-1β-induced IL-6 production, which is consistent with similarity between LPS and IL-1 signaling pathways (15).
Activation of p38 by LPS in MKK3−/− FLS.
Because of the differential effects of TNF-α and LPS in the MKK3−/− FLS, we examined the ability of each ligand to activate p38. Fig. 2F shows that TNF-α is a more potent inducer of p38 phosphorylation than LPS in WT FLS. In MKK3−/− FLS, however, TNF-α was much less effective compared with WT FLS. Although LPS was less potent in the WT cells, there was no difference in p38 activation between WT and MKK3−/− FLS. Therefore, unlike TNF-α, LPS-mediated p38 phosphorylation is independent of MKK3.
Activation of NF-κB by LPS and TNF-α in MKK3−/− FLS.
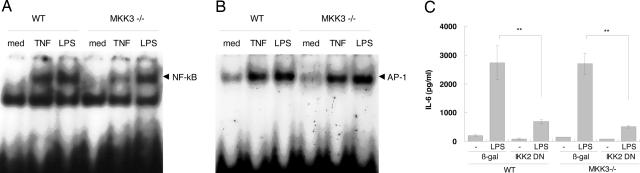
The observation that LPS is a less potent activator of p38 (Fig. 2F) suggests that another pathway might also contribute to the high IL-6 production despite modest LPS-induced phospho-p38 levels. To evaluate a potential role for NF-κB, which has been implicated in IL-6 regulation by FLS, WT and MKK3−/− cells were stimulated with LPS or TNF-α for 1 h, and EMSAs were performed. As shown in Fig. 3A, NF-κB activation was robust in both TNF-α- and LPS-stimulated WT FLS. However, LPS stimulation in MKK3−/− cells had normal NF-κB responses, whereas MKK3 deficiency significantly decreased TNF-α-mediated NF-κB activation (63% inhibition; P < 0.01, n = 3). For comparison, activator protein 1 activation was not affected by the MKK3 status of cells (Fig. 3B). These data suggest that NF-κB is not only required for IL-6 but also that it might account for the ability of LPS to overcome limited p38 activation in WT and MKK3−/− FLS. To prove the role of NF-κB, WT and MKK3−/− FLS were infected with an IκB kinase (IKK) 2 DN adenovirus. As shown in Fig. 3C, LPS-induced IL-6 production was blocked by IKK2 DN treatment in both WT and MKK3−/− FLS.
Fig. 3.
Regulation of NF-κB activation in WT and MKK3−/− FLS. (A) Cultured FLS from WT and MKK3−/− mice were stimulated with TNF-α or LPS for 1 h. EMSA was performed to evaluate NF-κB binding. In WT cells, TNF-α and LPS stimulation led to similar levels of NF-κB activation. However, NF-κB induction was defective in the MKK3−/− FLS after TNF-α stimulation. (B) For comparison, activator protein 1 (AP-1) binding was not affected by MKK3. (C) Cultured FLS from WT and MKK3−/− mice were infected by IKK2 DN adenovirus at a multiplicity of infection of 5,000 or control virus encoding β-gal and were stimulated with LPS for 24 h. IL-6 was measured by ELISA. LPS-induced IL-6 production was blocked by IKK2 DN treatment in both WT and MKK3−/− FLS (n = 3). ∗, P < 0.05; ∗∗, P < 0.01 compared with β-gal control.
Role of MKK3 in Passive K/BxN Arthritis.
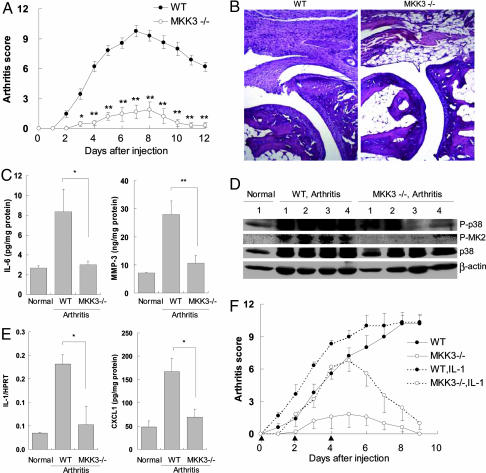
Because p38 activation and cytokine production in TNF-α-stimulated FLS depend on MKK3, passive K/BxN arthritis was studied in WT and MKK3−/− mice. The severity of arthritis was dramatically lower in the MKK3−/− mice (Fig. 4A). Histopathological analysis showed markedly reduced inflammatory cell infiltration and joint destruction in MKK3−/− mice (histologic scores were 0.43 ± 0.28 and 6.0 ± 0.65 for MKK3−/− and WT, respectively; P < 0.01) (see Fig. 4B for representative sections). Western blot analysis of the joint extracts showed that phospho-p38 and phospho-MAPK activator protein kinase (APK) 2 induction was significantly inhibited in MKK3−/− mice (P < 0.01; Fig. 4D), as were joint levels of IL-6 and MMP-3 at the conclusion of the experiment (P < 0.05; Fig. 4C). Because the mechanisms of disease initiation could be distinct from chronic inflammation, we also looked at relevant mediators 2 days after the second serum injection. CXC ligand 1 and IL-1β expression were suppressed in early arthritis (P < 0.05; Fig. 4E). Because the passive K/BxN model is IL-1-dependent (16, 17), we determined whether administration of exogenous IL-1 restores arthritis severity in the MKK3−/− mice. As shown in Fig. 4F, IL-1β treatment led to arthritis severity that was identical to WT mice in the early phases of the disease. Arthritis rapidly decreased to control levels after the IL-1β was discontinued.
Fig. 4.
Effect of MKK3 on passive K/BxN arthritis in vivo. (A) WT or MKK3−/− mice (n = 10 WT; n = 9 MKK3−/−) were administered K/BxN serum i.p. on days 0 and 2, and arthritis scores were evaluated until day 12. (B) Effect of MKK3 on histopathological evaluation in passive K/BxN arthritis on day 12. Representative examples of ankle histology demonstrate markedly less inflammation in the MKK3−/− mice compared with WT. (C and E) Effect of MKK3 deficiency on synovial IL-6 and MMP-3 (day 12) expression and IL-1β and CXC ligand 1 (day 4) expression. Real-time PCR or ELISAs were performed on joint extracts of WT and MKK3−/− mice. ∗, P < 0.05; ∗∗, P < 0.01 compared with WT arthritis control (n = 4). (D) Effect of MKK3 on synovial p38 and MAPKAPK-2 activation. Western blot analysis was performed on the joint. Note the lower levels of phospho-p38 and phospho-MAPKAPK-2 (P-MK2) in the MKK3−/− mice. (F) WT or MKK3−/− mice (n = 5 except for WT plus IL-1, where n = 3) were administered K/BxN serum i.p. on days 0 and 2. They were treated with IL-1β (1 μg i.p.) or vehicle on days 0, 2, and 4. IL-1 treatment (see arrows) restored arthritis to normal levels in the MKK3−/− mice.
Role of MKK3 in LPS-Induced Cytokine Production in Vivo.
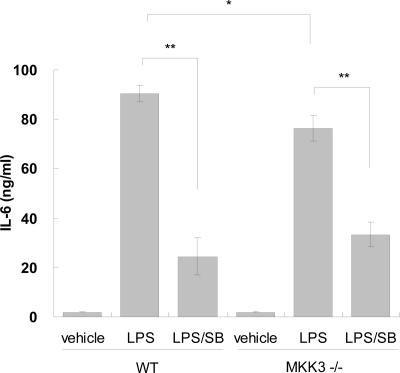
In vitro studies suggested that activation of TLR4 did not require MKK3 for IL-6 expression. Therefore, WT and MKK3−/− mice were administered LPS, and cytokine levels in the serum were determined. IL-6 levels in the MKK3−/− mice were only slightly lower than in WT mice, demonstrating that the TLR4-mediated innate immune responses are largely intact in vivo. In contrast, pretreatment with SB203580 (50 mg/kg) blocked LPS-stimulated cytokine production (Fig. 5).
Fig. 5.
Effect of MKK3 on LPS-induced cytokines production in vivo. LPS (400 μg per mouse) was injected i.p., and plasma was collected 3 h later. IL-6 was measured by ELISA. Note that LPS-induced IL-6 production was only modestly lower in MKK3−/− mouse sera compared with WT. However, SB203580 (50 mg/kg orally) inhibited IL-6 production in both genotypes. ∗, P < 0.05; ∗∗, P < 0.01 compared with WT or LPS control (n = 5).
Discussion
RA is a chronic inflammatory disease marked by synovial hyperplasia and local invasion into the extracellular matrix. Synovitis is mediated, in part, by cytokines that activate a broad array of cell signaling mechanisms and lead to the release of destructive enzymes (18). MAPKs are especially important because they control the production of cytokines such as IL-1, IL-6, IL-8, and MMPs that participate in destruction of the extracellular matrix. The three MAPK families, ERK, JNK, and p38, modulate gene expression by phosphorylating numerous key transcription factors, such as activator protein 1 and ATF-1/2, as well as the downstream kinase MAPKAPK-2 (1, 19).
Several MAPK members are activated in the rheumatoid synovium and have been implicated in the pathogenesis of RA. Among them, p38 is thought to be crucial because selective p38 inhibitors block joint inflammation and destruction in several animal models of arthritis (3). Of the p38 isoforms, p38α is particularly relevant for cytokine regulation in macrophages and is blocked by pyridinylimidazole compounds, such as SB203580, which compete for the ATP-binding pocket (20). Phospho-p38α is present in rheumatoid synovial blood vessels, perivascular mononuclear cell infiltrates, and the intimal lining (2). p38δ is also activated in RA, especially at sites of synovial invasion into the extracellular matrix (21).
The two main upstream kinases that regulate p38, MKK3 and MKK6, are also expressed and activated in RA synovium, especially in the intimal lining FLS (13). Using DNA constructs, we showed that both MKK3 and MKK6 contribute to p38 phosphorylation in cytokine-stimulated FLS (14). However, MKK3 blockade more effectively inhibited p38-mediated induction of IL-6, IL-8, and MMP-3. p38 can also serve as a substrate for a third MKK, MKK4, which is normally considered a JNK kinase. However, siRNA knock-down studies suggest that MKK4 does not play a significant role in p38 activation or IL-6 production in FLS after exposure to TNF-α (data not shown). These data suggest that MKK3 might be the dominant MKK that controls p38 in FLS after TNF-α stimulation.
To test this hypothesis, we evaluated p38 function in cultured FLS from MKK3−/− mice. These studies showed that p38 requires MKK3 in primary murine FLS for normal phosphorylation after TNF-α stimulation. In contrast, MKK3 deficiency had no effect on JNK or ERK activation. Although p38 phosphorylation was modestly decreased in MKK3−/− FLS, much more profound suppression of IL-6 production was observed. The impact of MKK3 deficiency on IL-6 production was mimicked by SB203580 or p38α knock-down in WT cells and is similar to MKK3−/− murine embryonic fibroblasts (22). FLS are also a primary source of IL-6 in RA synovium (23); these studies suggest that selective MKK3 blockade might effectively modulate the cytokine profile in inflammatory arthritis. The mechanism of IL-6 inhibition is complex and can be caused by changes in transcriptional, translational, and mRNA stability effects as previously demonstrated for p38 in cultured FLS (24).
The possibility that TLR responses or other pathways to p38 activation and cytokine production remain intact despite MKK3 deficiency was tested in vitro. First, the TLR4 ligand LPS was used to stimulate the murine FLS to assess cytokine production. Even though LPS is not a particularly potent inducer of phospho-p38 compared with TNF-α, the absence of MKK3 had no effect on its ability to activate p38. Surprisingly, MKK3 plays no role in this pathway, even though TNF-α-mediated IL-6 production is almost completely dependent on this kinase. Similarly, IL-1β stimulation, which shares signaling pathways with TLR4 (15), also was MKK3-independent. Of particular interest, SB203580 blocked LPS as well as IL-1β-induced IL-6 production, suggesting that p38 activation still participates through other upstream kinases.
The relatively low level of phospho-p38 after LPS stimulation compared with TNF-α also suggested that the TLR ligand might activate alternative pathways that contribute to IL-6 production. NF-κB is an attractive candidate because it can induce IL-6 expression in cultured FLS (25). Both LPS and TNF-α increased NF-κB binding in FLS, but MKK3−/− cells had a significant defect in NF-κB activation only after TNF-α stimulation. Therefore, both MKK3 and p38 are required for full NF-κB activation after TNF receptor ligation, but MKK3 plays no role in NF-κB activation induced by LPS. The requirement for NF-κB in LPS-mediated cytokine expression was confirmed by using a DN IKK2 construct, which significantly decreased IL-6 production.
These data suggest that LPS-induced IL-6 production depends on NF-κB and p38, without a contribution from MKK3. TNF-α-induced IL-6 production requires NF-κB, MKK3, and p38. Furthermore, NF-κB induction by TNF-α also requires MKK3, and, as a result, NF-κB cannot rescue IL-6 production in MKK3−/− FLS. The possibility that p38 regulates NF-κB has been examined in a few other systems with variable results. For instance, LPS-stimulated NF-κB activation in RAW264.7 cells and cytokine-stimulated FLS was not inhibited by SB203580 (26, 27). In contrast, p38 was required for NF-κB p65 transactivation in murine L929 cells and murine embryonic fibroblasts (28, 29). Our data strongly suggest that p38 or MKKs play a key role in NF-κB-mediated DNA binding and IL-6 production in synoviocytes.
The function of MKK3 was then evaluated in the passive K/BxN model of arthritis (30). MKK3 deficiency profoundly decreased both the incidence and severity of arthritis. In particular, the increase in synovial p38 phosphorylation observed in the WT mice was completely prevented in the MKK3−/− animals, as were MAPKAPK-2 phosphorylation and IL-6 and MMP-3 production. Early pathogenic events in the arthritis model are also modulated by MKK3. For instance, CXC ligand 1 expression in MKK3−/− joints in the early stages of arthritis is significantly decreased and could suppress accumulation of neutrophils to the synovium. Perhaps even more important, expression of IL-1β was also suppressed in the MKK3−/− mice. The critical role of defective IL-1β production in early disease was proven by administering exogenous cytokine to MKK3−/− mice, which restored arthritis to WT levels. Arthritis rapidly decreased after IL-1 treatment was discontinued, suggesting that cytokine replacement at the onset is not enough to establish chronic synovitis. Of interest, WT mice also had a small but persistent increase in arthritis severity.
In vivo studies confirm that MKK3 is not required for TLR4 responses. LPS administration to mice leads to a dramatic increase in IL-6 production, which was only slightly lower in the MKK3−/− animals. Mice pretreated with SB203580 had a significant decrease in cytokine levels after LPS injection, indicating that p38 still participates in the model. These results contrast with the arthritis studies, where cytokine production decreased to normal levels in the absence of MKK3. Hence, MKK3 deficiency dramatically decreases cytokine production and clinical arthritis while having a minimal impact on in vivo responses to TLR4 stimulation. The role of p38 in TLR4 responses may be similar in humans, where administration of a p38 inhibitor blocks IL-6 and TNF-α production after LPS challenge in normal volunteers (31). Although formal evaluation of host defense has not been performed, the fact that TLR4 signaling as well as IL-1-mediated events can remain intact with MKK3 deficiency compared with p38 blockade suggests an improved the safety profile of MKK3 inhibition compared with p38.
Our studies demonstrate that selective deficiency in MKK3 can suppress inflammation in an animal model of arthritis, in part through an IL-1β-dependent mechanism in early disease. More striking, the mice retain the ability to respond to TLR4 ligation in a p38-dependent fashion. The mechanism of TLR4 responses is probably related to preservation of p38 responses through other MKKs in combination with preserved NF-κB signaling. It is precisely this signaling diversity within the p38 pathway that provides an opportunity to block components of the p38 cascade that are implicated in pathogenic responses rather than host defense. These studies suggest that targeting upstream kinases such as MKK3 can offer a more selective approach to p38 inhibition than targeting p38 itself.
Materials and Methods
FLS.
Multiple FLS lines were derived from MKK3−/− mice (22) and matched WT littermates on the C57BL/6 background. FLS were grown in DMEM containing 10% FCS and used from passages three to four, during which time they are a homogeneous population of cells (<1% CD11b-positive, <1% phagocytic, and <1% FcγRII-positive) (32). All cell cultures were synchronized in 0.1% FCS in DMEM for 24 h before stimulation and maintained in low serum conditions throughout the experiment.
Antibodies and Reagents.
Anti-MKK3, anti-MKK6, anti-β-actin, and secondary antibodies (Santa Cruz Biotechnology); anti-phosho-p38 (Thr-180/Tyr-182), anti-p38, anti-phospho-MAPKAPK-2, anti-phospho-JNK1/2, anti-JNK1/2, anti-phospho-ERK1/2, and anti-ERK1/2 antibodies; GST-ATF-2 (Cell Signaling Technology, Beverly, MA); recombinant human TNF-α (rhTNF-α); recombinant mouse IL-1β (R & D Systems); and SB203580 (Calbiochem) were used.
Western Blot Analysis.
FLS were treated with medium, rhTNF-α (50 ng/ml), or LPS (1 μg/ml) for 15 min. Protein was extracted and fractionated on 10% SDS/PAGE, and then Western blotting was performed (14). The densitometry analysis was done by using nih image 1.61.
Immunoprecipitation and Kinase Assays.
FLS were treated with medium or rhTNF-α for 15 min, lysed in modified RIPA buffer (50 mM HEPES, pH 7.4/150 mM NaCl/1% Triton X-100/10% glycerol/2.5 mM MgCl2/1.0 mM EDTA, pH 8.0/20 mM β-glycerophosphate/10 mM NaF/1 mM Na2VO4/10 μg/ml aprotinin/1 μM pepstatin A/1 mM PMSF), and incubated with anti-p38 mAb, followed by protein G-Sepharose overnight. The immunoprecipitates were washed and incubated with medium containing 5 μCi (1 Ci = 37 GBq) of [γ-32P]ATP, 100 mM ATP, and 4 μg of GST-fusioned-ATF-2 at 37°C for 30 min. Reactions were fractionated on 12% SDS/PAGE and visualized by autoradiography (14).
Cell Transfection.
For virus infection, FLS were treated with purified adenovirus IKK2 DN overnight at a multiplicity of infection of 5,000 as described in ref. 25. Adenovirus encoding for β-gal (CMV-LacZ) was used as a control. For siRNA transfection, using a mouse embryonic fibroblast nucleofector kit (MEF2) with program T-20 (Amaxa, Gaithersburg, MD), FLS were transfected with 1 μg of p38α siRNA or control siRNA (Dharmacon) (14).
ELISA and mRNA Assay.
FLS were treated with medium, rhTNF-α (50 ng/ml), IL-1β (2 ng/ml), or LPS (1 μg/ml) for 24 h. Supernatants were harvested and assayed for IL-6 protein by ELISA (R & D Systems). Total RNA was harvested to assay IL-1 and IL-6 mRNA expression. Quantitative real-time PCR was performed by using the GeneAmp 5700 sequence detection system (Applied Biosystems) as described in ref. 33.
EMSA.
FLS were treated with medium, rhTNF-α (50 ng/ml), or LPS (1 μg/ml) for 60 min, and EMSA was performed by using the gel-shift assay system (Promega). Protein concentration-normalized nuclear extracts were mixed with the purified γ-32P-labeled oligonucleotides, resolved by 5% PAGE, and visualized by autoradiography.
Passive K/BxN Mice Serum Transfer Model of Arthritis.
Recipient MKK3−/− and matched WT mice were injected i.p. with 100 μl of pooled adult K/BxN mice serum on days 0 and 2. Arthritis was evaluated for each paw as described in ref. 34. For tissue protein assays, snap-frozen ankle joints were homogenized in lysis buffer and assayed by ELISA or immunoblotting. In some cases, RNA was isolated, and quantitative real-time PCR was performed. The other hind paws were fixed in 10% formalin and decalcified in EDTA. A semiquantitative scoring system was used to assess synovial inflammation, extraarticular inflammation, erosion, and proteoglycan loss (34).
Cytokine Levels in Endotoxic Shock.
MKK3−/− mice or matched WT mice (five animals per group) were injected i.p. with 400 μg of Escherichia coli 0111:B4 LPS (Sigma) (35). In some mice, SB203580 (50 mg/kg) in 0.03 M HCl–0.5% tragacanth (Sigma) was orally administered 30 min before LPS injection. Plasma was collected 3 h later and assayed for IL-6 by ELISA.
Statistical Analysis.
Data are expressed as mean ± SEM. Comparisons were performed by Student’s t test with Bonferroni’s correction where appropriate.
Acknowledgments
This work was supported by grants from the National Institutes of Health.
Abbreviations
- RA
rheumatoid arthritis
- FLS
fibroblast-like synoviocytes
- MAPK
mitogen-activated protein kinase
- MKK
MAPK kinase
- ATF
activating transcription factor
- MMP
matrix metalloproteinase
- TLR
Toll-like receptor
- ERK
extracellular signal-regulated kinase
- JNK
c-Jun N-terminal kinase
- DN
dominant negative
- siRNA
small interfering RNA
- IKK
IκB kinase
- APK
activator protein kinase
- rhTNF-α
recombinant human TNF-α.
Footnotes
Conflict of interest statement: No conflicts declared.
This paper was submitted directly (Track II) to the PNAS office.
References
- 1.Morel J., Berenbaum F. Joint Bone Spine. 2004;71:503–510. doi: 10.1016/j.jbspin.2004.03.004. [DOI] [PubMed] [Google Scholar]
- 2.Schett G., Tohidast-Akrad M., Smolen J. S., Schmid B. J., Steiner C. W., Bitzan P., Zenz P., Redlich K., Xu Q., Steiner G. Arthritis Rheum. 2000;43:2501–2512. doi: 10.1002/1529-0131(200011)43:11<2501::AID-ANR18>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 3.Mclay L. M., Halley F., Souness J. E., McKenna J., Benning V., Birrell M., Burton B., Belvisi M., Collis A., Constan A., et al. Bioorg. Med. Chem. 2001;9:537–554. doi: 10.1016/s0968-0896(00)00331-x. [DOI] [PubMed] [Google Scholar]
- 4.Pargellis C., Regan J. Curr. Opin. Investig. Drugs. 2003;4:566–571. [PubMed] [Google Scholar]
- 5.Saklatvala J. Curr. Opin. Pharmacol. 2004;4:372–377. doi: 10.1016/j.coph.2004.03.009. [DOI] [PubMed] [Google Scholar]
- 6.Kumar S., Boehm J., Lee J. C. Nat. Rev. Drug Discov. 2003;2:717–726. doi: 10.1038/nrd1177. [DOI] [PubMed] [Google Scholar]
- 7.Schnyder B., Meunier P. C., Car B. D. Biochem. J. 1998;331:489–495. doi: 10.1042/bj3310489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.van den Blink B., Juffermans N. P., ten Hove T., Schultz M. J., van Deventer S. J., van der Poll T., Peppelenbosch M. P. J. Immunol. 2001;166:582–587. doi: 10.4049/jimmunol.166.1.582. [DOI] [PubMed] [Google Scholar]
- 9.Raingeaud J., Whitmarsh A. J., Barrett T., Derijard B., Davis R. J. Mol. Cell. Biol. 1996;16:1247–1255. doi: 10.1128/mcb.16.3.1247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moriguchi T., Kuroyanagi N., Yamaguchi K., Gotoh Y., Irie K., Kano T., Shirakabe K., Muro Y., Shibuya H., Matsumoto K., et al. J. Biol. Chem. 1996;271:13675–13679. doi: 10.1074/jbc.271.23.13675. [DOI] [PubMed] [Google Scholar]
- 11.Brancho D., Tanaka N., Jaeschke A., Ventura J. J., Kelkar N., Tanaka Y., Kyuuma M., Takeshita T., Flavell R. A., Davis R. J. Genes Dev. 2003;17:1969–1978. doi: 10.1101/gad.1107303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Han J., Lee J. D., Jiang Y., Li Z., Feng L., Ulevitch R. J. J. Biol. Chem. 1996;271:2886–2891. doi: 10.1074/jbc.271.6.2886. [DOI] [PubMed] [Google Scholar]
- 13.Chabaud-Riou M., Firestein G. S. Am. J. Phathol. 2004;164:177–184. doi: 10.1016/S0002-9440(10)63108-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Inoue T., Hammaker D., Boyle D. L., Firestein G. S. J. Immunol. 2005;174:4301–4306. doi: 10.4049/jimmunol.174.7.4301. [DOI] [PubMed] [Google Scholar]
- 15.O’Neill L. A. Curr. Opin. Immunol. 2005;17:1–7. [Google Scholar]
- 16.Kyburz D., Corr M. Springer Semin. Immunopathol. 2003;25:79–90. doi: 10.1007/s00281-003-0131-5. [DOI] [PubMed] [Google Scholar]
- 17.Ohmura K., Johnsen A., Ortiz-Lopez A., Desany P., Roy M., Besse W., Rogus J., Bogue M., Puech A., Lathrop M., et al. Proc. Natl. Acad. Sci. USA. 2005;102:12489–12494. doi: 10.1073/pnas.0504325102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Firestein G. S. Nature. 2003;423:356–361. doi: 10.1038/nature01661. [DOI] [PubMed] [Google Scholar]
- 19.Neininger A., Kontoyiannis D., Kotlyarov A., Winzen R., Eckert R., Volk H. D., Holtmann H., Kollias G., Gaestel M. J. Biol. Chem. 2002;277:3065–3068. doi: 10.1074/jbc.C100685200. [DOI] [PubMed] [Google Scholar]
- 20.Hale K. K., Trollinger D., Rihanek M., Manthey C. L. J. Immunol. 1999;162:4246–4252. [PubMed] [Google Scholar]
- 21.Kuchen S., Seemayer C. A., Rethage J., von Knoch R., Kuenzler P., Michel B. A., Gay R. E., Gay S., Neidhart M. Autoimmunity. 2004;37:57–65. doi: 10.1080/08916930310001637977. [DOI] [PubMed] [Google Scholar]
- 22.Wysk M., Yang D. D., Lu H. T., Flavell R. A., Davis R. J. Proc. Natl. Acad. Sci. USA. 1999;96:3763–3768. doi: 10.1073/pnas.96.7.3763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Firestein G. S., Alvaro-Garcia J. M., Maki R. J. Immunol. 1990;144:3347–3353. [PubMed] [Google Scholar]
- 24.Miyazawa K., Mori A., Miyata H., Akahane M., Ajisawa Y., Okudaira H. J. Biol. Chem. 1998;273:24832–24838. doi: 10.1074/jbc.273.38.24832. [DOI] [PubMed] [Google Scholar]
- 25.Aupperle K. R., Bennett B. L., Han Z., Boyle D. L., Manning A. M., Firestein G. S. J. Immunol. 2001;166:2705–2711. doi: 10.4049/jimmunol.166.4.2705. [DOI] [PubMed] [Google Scholar]
- 26.Baldassare J. J., Bi Y., Bellone C. J. J. Immunol. 1999;162:5367–5373. [PubMed] [Google Scholar]
- 27.Suzuki M., Tetsuka T., Yoshida S., Watanabe N., Kobayashi M., Matsui N., Okamoto T. FEBS Lett. 2000;465:23–27. doi: 10.1016/s0014-5793(99)01717-2. [DOI] [PubMed] [Google Scholar]
- 28.Jijon H., Allard B., Jobin C. Cell. Signalling. 2004;16:1023–1032. doi: 10.1016/j.cellsig.2004.02.005. [DOI] [PubMed] [Google Scholar]
- 29.Vanden Berghe W., Plaisance S., Boone E., De Bosscher K., Schmitz M. L., Fiers W., Haegeman G. J. Biol. Chem. 1998;273:3285–3290. doi: 10.1074/jbc.273.6.3285. [DOI] [PubMed] [Google Scholar]
- 30.Ji H., Pettit A., Ohmura K., Ortiz-Lopez A., Duchatelle V., Degott C., Gravallese E., Mathis D., Benoist C. J. Exp. Med. 2002;196:77–85. doi: 10.1084/jem.20020439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Branger J., van den Blink B., Weijer S., Madwed J., Bos C. L., Gupta A., Yong C. L., Polmar S. H., Olszyna D. P., Hack C. E., et al. J. Immunol. 2002;168:4070–4077. doi: 10.4049/jimmunol.168.8.4070. [DOI] [PubMed] [Google Scholar]
- 32.Yamanishi Y., Boyle D. L., Pinkoski M. J., Mahboubi A., Lin T., Han Z., Zvaifler N. J., Green D. R., Firestein G. S. Am. J. Pathol. 2002;160:123–130. doi: 10.1016/S0002-9440(10)64356-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Boyle D. L., Rosengren S., Bugbee W., Kavanaugh A., Firestein G. S. Arthritis Res. Ther. 2003;5:R352–R360. doi: 10.1186/ar1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Simelyte E., Rosengren S., Boyle D. L., Corr M., Green D. R., Firestein G. S. Arthritis Rheum. 2005;52:1876–1884. doi: 10.1002/art.21099. [DOI] [PubMed] [Google Scholar]
- 35.Firestein G. S., Boyle D. L., Bullough D. A., Gruber H. E., Sajjadi F. G., Montag A., Sambol B., Mullane M. J. Immunol. 1994;152:5853–5859. [PubMed] [Google Scholar]