Abstract
We report that hbhA is differentially regulated during Mycobacterium tuberculosis infection. Upregulation was observed in epithelial cell infection but not in macrophage infection and in the lungs but not in the spleens of infected mice, and it was greater during the early steps of infection, when bacilli disseminate from the site of primary infection.
The heparin-binding hemagglutinin (HBHA) is a 28-kDa, methylated, surface-exposed protein that mediates the interaction of the tubercle bacilli with the host, acting as an adhesin for nonphagocytic cells (19, 22). Methylation of the C-terminal lysine residues affects both the biochemical and immunological properties of the protein (3, 14, 17, 23, 29). Several experimental findings have implicated HBHA in the process of extrapulmonary dissemination of Mycobacterium tuberculosis (22), a major step in the development of the active form of the disease (1). Recently, Menozzi et al. (18) have also shown that binding of HBHA to proteoglycans induces epithelial transcytosis, which may promote the systemic dissemination of M. tuberculosis. In light of these studies, it has been proposed that the development of an HBHA-based vaccine may represent an original and effective strategy to prevent the development of the disease, and the results obtained in the mouse model of tuberculosis (TB) have supported this hypothesis (20, 29). To further investigate the role of HBHA in the biology of M. tuberculosis, a comprehensive study, both in vitro and in vivo, was carried out to evaluate hbhA gene expression during the immunopathogenetic process of TB.
Expression of the hbhA, ftsZ, hsp70, and 16S rRNA genes was assessed using total RNA by real-time reverse transcriptase PCR (RT-PCR) with an i-Cycler iQ system (Bio-Rad Laboratories, Hercules, Calif.) (26). Cells and tissues homogenates were resuspended in RNAlater solution (QIAGEN, Hilden, Germany), and total RNA was extracted with an RNeasy Protect minikit (QIAGEN) by mechanical disruption, as previously described (11, 26). For each gene, a set of primer pairs and a TaqMan probe were designed with Beacon Designer 2 (version 2.06) software (Premier Biosoft International, Palo Alto, Calif.) and synthesized by MWG Biotech (Florence, Italy) (Table 1). Each reaction was run in quadruplicate, and amplification efficiencies for all genes were determined (21). Because the PCR protocols worked with equal efficiencies, relative mRNA expression levels of hbhA and hsp70 were normalized for input RNA against the level of ftsZ or 16S rRNA gene transcripts. The relative mRNA expression levels of the hbhA and hsp70 genes in each sample were calculated using the comparative cycle time method (16). Comparison between the categorical variables was performed by using the two-tailed Student t test. A P value of less than 0.05 was considered statistically significant.
TABLE 1.
Primers used in this study to amplify the gene and sequences of interest
| Gene | Annotationa | Forward primer | Reverse primer | Probeb |
|---|---|---|---|---|
| 16S rRNA gene | CAGGGCTTCACACATGCTACAAT | GTATTCACCGCAGCGTTGCT | Texas Red-TCTGCGATTACTAGCGACTCCGACTTCACG-BHQ2 | |
| ftsZ | Rv2150c | CGGTATCGCTGATGGATGCTTT | CGGACATGATGCCCTTGACG | Texas Red-CGCCGACGAGGTGCTGCTCAACG-BHQ2 |
| hsp70 | Rv0350 | GTTGAGGTCCGTGCCACTTC | ACTTGTCCACCAGCCAATCG | 6FAM-TGGTCCCAGTCGTCGCCGCCG-TAMRA |
| hbhA | Rv0475 | GCAGAGCTTCGAGGAAGTGTC | AACGCCTCCTGGGTCAACTC | 6FAM-CGCCTGGTCCACGTAGCCTTCGG-TAMRA |
Annotation as indicated in the TubercuList (http://genolist.pasteur.fr/TubercuList/).
6FAM, 6-carboxyfluorescein; TAMRA, 6-carboxy-N,N,N′,N′-tetramethylrhodamine; Texas Red, product from Molecular Probes; BHQ2, Black Hole Quencher 2.
To assess hbhA gene expression in axenic culture, M. tuberculosis strain Erdman (TMC 107) was grown in 7H9 liquid medium supplemented with albumin-dextrose-catalase enrichment (Microbiol, Cagliari, Italy) and Tween 80 (0.05%) at 37°C, and 2-ml aliquots were harvested at different time points to assess colony counts and to isolate total RNA. The 16S rRNA-normalized levels of ftsZ transcript remained constant during the in vitro growth phase, while the 16S rRNA-normalized amounts of hsp70 transcript increased at the stationary phase (Fig. 1B). An ≈100-fold increase in the expression of the hbhA gene relative to either the 16S rRNA (Fig. 1C) or ftsZ (Fig. 1D) gene was observed, demonstrating that the hbhA gene is strongly upregulated at the early stationary phase.
FIG. 1.
Expression of the hbhA gene by M. tuberculosis in axenic culture as determined by real-time RT-PCR. M. tuberculosis strain Erdman was grown in liquid medium with gentle shaking at 37°C. (A) Growth curve as determined by CFU counting at the indicated time points. (B) 16S rRNA-normalized levels of ftsZ and hsp70 transcripts at the different time points shown in panel A. (C and D) 16S rRNA (C)- and ftsZ (D)-normalized levels of hbhA determined during in vitro growth. Three samples per time point were analyzed, and RT-PCR was carried out four times on each sample. Error bars show standard deviations.
Expression of the hbhA gene in the intracellular environment was evaluated by infecting bone marrow-derived macrophages (BMMΦ) and A549 human pneumocytes with M. tuberculosis. BMMΦ were established in 24-well plates as previously described (4) and were infected with M. tuberculosis at a multiplicity of infection of 3:1. A549 type II human pneumocytes were cultured in Dulbecco modified Eagle medium containing 10% fetal bovine serum. A549 cells were cultured in 24-well plates and infected with M. tuberculosis (multiplicity of infection, ≈5:1) when they reached a confluence of 60 to 70%. Following infection, BMMΦ and A549 cells were incubated for 2 hours at 37°C and washed three times with antibiotic-free medium, and then medium containing gentamicin at 50 μg/ml was added to kill extracellular bacteria. At different time points, the cells were washed three times with phosphate-buffered saline (PBS) and resuspended in 0.5 ml of cell lysis solution (PBS containing 1% Triton X-100). Aliquots (0.4 ml) of the lysates were harvested by centrifugation and used to isolate total RNA, as described above.
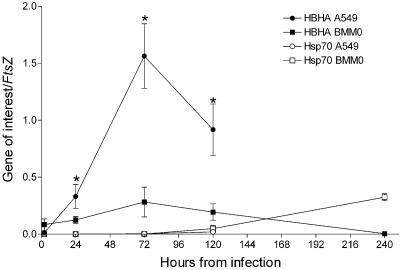
As shown in Fig. 2, on day 1 and, more significantly, on day 3 postinfection, an increase in hbhA mRNA levels was observed in M. tuberculosis infecting A549 cells. Conversely, the relative hbhA expression was lower and remained constant in M. tuberculosis infecting BMMΦ (Fig. 2). These results indicate that the hbhA gene is strongly upregulated in M. tuberculosis infecting type II human pneumocytes but not in M. tuberculosis infecting BMMΦ.
FIG. 2.
Expression of the hbhA and hsp70 genes in the intracellular environment. BMMΦ (squares) and A549 (circles) cells were infected with the virulent M. tuberculosis Erdman strain. Cells were harvested at different time points, and real-time RT-PCR was performed on the extracted RNA. Gene expression levels were normalized against the ftsZ gene transcripts. Three samples per time point were analyzed, and RT-PCR was carried out four times on each sample. Error bars show standard deviations. Comparisons were performed by using the two-tailed Student t test. Asterisks indicate statistically significant differences (P < 0.05).
In vivo gene expression was investigated using the mouse model of TB (2, 4). C57BL/6 female mice were obtained from Harlan (Italy) and were maintained under barrier conditions and fed commercial mouse chow and water ad libitum. All animal experiments were performed using protocols approved by the Catholic University Ethical Committee. Eight-week-old mice were aerogenically infected using a Glas-Col chamber with a low dose (≈2 × 102 CFU/mouse) of the M. tuberculosis strain (2). At different time points, five mice per group were sacrificed, the spleens and the right lung lobes were homogenized in PBS containing 0.05% Tween 80, and serial dilutions were plated on 7H11/oleic acid-albumin-dextrose-catalase agar plates to assess CFU per organ (4). One-milliliter aliquots of lung and spleen homogenates per animal were centrifuged, and the pellets were processed to isolate RNA. Right after tissue removal, the lung left lobes were perfused and fixed with neutral buffered paraformaldehyde, processed and stained with hematoxylin-eosin and Ziehl-Nielsen stain, and analyzed under light microscopy.
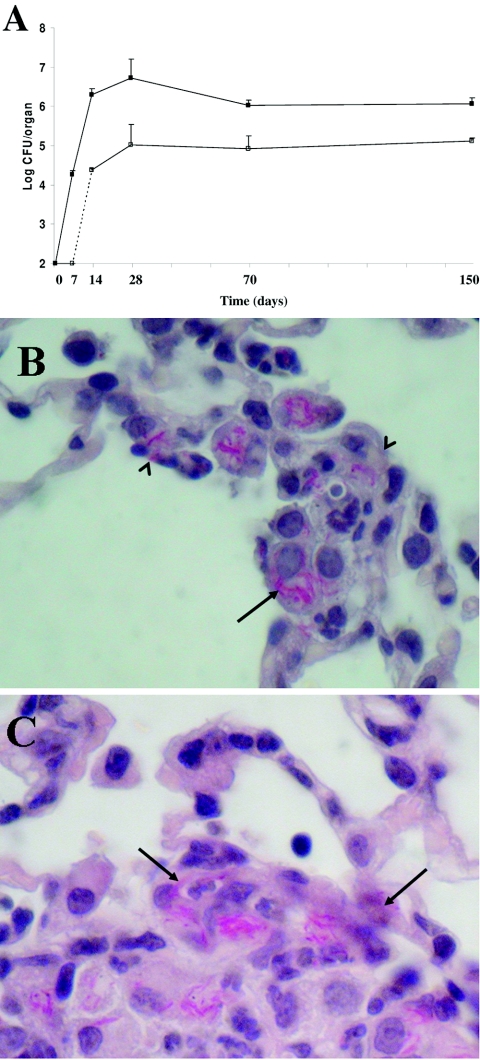
As shown in Fig. 3, during the first 2 weeks of infection M. tuberculosis multiplies logarithmically in lung tissue, reaching a peak of ≈6.8 log CFU/organ on day 28. Colonization of the spleen was observed starting by day 14 and reached the maximum on day 28 after infection, following a logarithmic growth curve similar to that seen in the lung tissue. Thereafter, M. tuberculosis persisted in the lung and spleen tissues for a long time (150 days).
FIG. 3.
Colonization of host tissues following aerogenic challenge with M. tuberculosis strain Erdman (≈100 CFU/mice). (A) Time course of colonization of lung (filled squares) and spleen (empty squares) tissues as assessed by CFU counts. (B and C) Histopathological analysis of lung tissues isolated from infected mice at 14 days postinfection. The left lobe of the lung was removed, fixed, and stained with hematoxylin-eosin and Ziehl-Nielsen stain. Representative slides are shown. Magnification, ×1,000. Arrows indicate AFBs; arrowheads indicate epithelial cells infected with AFBs.
Histopathological analysis of lung sections was found to be consistent with that in previous studies (24). Lung sections isolated from mice at day 14 after infection showed a cellular infiltrate typical of the category 1 lesions described by Rhoades et al. (24), with highly infected macrophages infiltrating the alveolar septa (Fig. 3B). Some of these macrophages were found in close contact with intact epithelial cells, which also appeared to be infected with acid-fast bacilli (AFBs) (Fig. 3B). A few more advanced type 2 lesions starting to organize in granulomas could be detected scattered in the tissue (24). Interestingly, most of the M. tuberculosis-infected cells were detected in the periphery of the lesions, in close contact with epithelial cells and lumen (Fig. 3C).
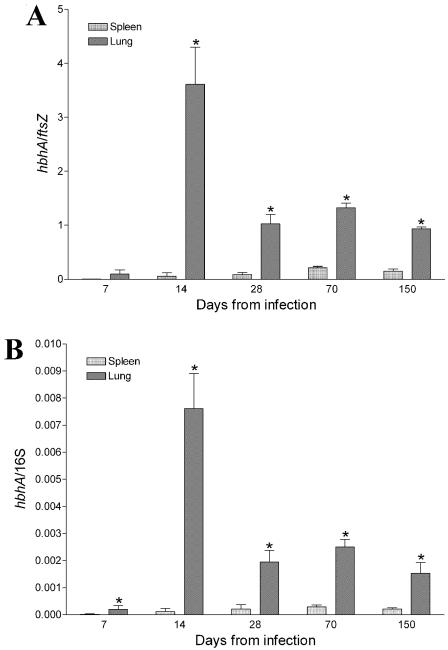
Figure 4A shows the ftsZ-normalized levels of hbhA in the lung and spleen tissue at the time points indicated in Fig. 3. hbhA gene expression was detected starting on day 7 in the lung, while expression in the spleen could not be demonstrated until day 14, although ftsZ and 16S RNA transcripts were detected (data not shown). On day 14 following infection, ftsZ-normalized levels of hbhA in lung tissue significantly increased (≈40-fold). On day 28, relative expression of the hbhA gene decreased, reaching an hbhA/ftsZ ratio of 1, which was maintained throughout the course of infection (day 70 and day 150). Expression of the hbhA gene in the spleen tissue remained low throughout the infection (hbhA/ftsZ ratio of ≈0.05 to 0.2), and no significant variability was observed at the different time points. Similar results were obtained when 16S rRNA was used as the housekeeping gene (Fig. 4B), indicating that ftsZ is a reliable housekeeping gene in vivo. A comparative analysis of the hbhA gene expression in M. tuberculosis infecting lung and spleen tissues indicates that overall expression of hbhA is 10 to 40 times higher in the lung than in the spleen.
FIG. 4.
Expression of the hbhA gene in the lung and spleen tissues of mice aerogenically challenged with the virulent M. tuberculosis Erdman strain as determined by real-time RT-PCR. ftsZ-normalized (A) and 16S rRNA-normalized (B) levels of hbhA were determined at the time points shown in Fig. 3. Five mice per time point were sacrificed, and RT-PCR was carried out four times on each sample. Error bars show standard deviations. Comparisons were performed by using the two-tailed Student t test. Asterisks indicate statistically significant differences (P < 0.05).
A major finding of this study is that the hbhA gene is specifically upregulated by M. tuberculosis in the lung but not in the spleen during the early steps of infection, when bacilli are actively multiplying in host tissues. Expression of the hbhA gene was calculated relative to 16S rRNA, whose expression remains constant in macrophages and host tissues (13, 28), and to the ftsZ gene. FtsZ is a protein involved in bacterial cell division, and its intracellular concentration remains constant regardless of doubling time or growth rate (25, 31). We reasoned that normalization of hbhA transcripts to those of the ftsZ gene would provide a further control to assess tissue-specific upregulation during active bacillus multiplication. Indeed, the results obtained in this study demonstrate that ftsZ is a reliable housekeeping gene.
Upregulation of the hbhA gene in the lung is tissue specific and cannot be simply associated with bacterial multiplication. The increase in hbhA expression is rather substantial in comparison to that observed for other M. tuberculosis genes known to play an important role in TB pathogenesis, for example, Esat-6, fbpABC, sodC, acr, icl1, mbtA, and others (5, 12, 27, 28, 30). hbhA upregulation appears to be quantitatively much more remarkable than what has been seen, for instance, for the fbpA and fbpB genes (27, 28).
It should be noted that upregulation of the hbhA gene occurs when bacilli are actively multiplying in the lung tissue and the cellular immune response has not yet contained the infected areas. Histopathological analysis of lung tissue carried out on day 14 indicated that AFBs reside primarily in early cellular infiltrates in the alveolar septa, infecting macrophages or epithelial cells. At this time of infection, M. tuberculosis can also be detected in the forming granulomas, mostly at the periphery of the lesions, where it is more likely to escape into the blood system and disseminate. Conversely, analysis of infected lung tissue at later time points indicates that AFBs infect macrophages residing primarily in the inner parts of the lesions, in areas tightly confined by wedges of lymphocytes.
Dissemination of the tubercle bacilli from the site of primary infection is a major step in TB pathogenesis (1). The current hypothesis is that inhalation of bacilli establishes primary lesions in the lungs that cannot be contained by either the innate or the slowly emerging acquired immune response. Multiplying bacteria escape from the primary lesion through a process of hematogenous dissemination that leads to the formation of secondary lesions that appear to be more favorable for the development of lesion reactivation (10, 15). Because HBHA has been clearly implicated in the process of dissemination of M. tuberculosis (19, 22), the results of this study suggest that M. tuberculosis promotes hbhA expression when the bacteria can easily escape from the forming lesions to the bloodstream. Because the hbhA gene is upregulated within A549 pneumocytes, it is possible that HBHA is important not only for adherence to and invasion of epithelial cells (22) but also for escape from these cells into the bloodstream.
The results of this study may also have more practical applications. Improved recombinant Mycobacterium bovis bacille Calmette-Guérin (BCG) strains expressing M. tuberculosis antigens are among the most promising vaccine candidates (7, 8, 9), and intranasal administration of a recombinant BCG vaccine has been shown to induce a strong immune response in the lung that resulted in greater protection than systemic administration (6). BCG strains expressing recombinant antigens under the control of the hbhA promoter should warrant a high expression level in the lung that may lead to improved vaccines for use in mucosal immunization strategies against M. tuberculosis or other infectious agents.
Acknowledgments
We thank M. J. Brennan for carefully reviewing the manuscript and for insight and advice.
This work was supported by a grant from the Catholic University of the Sacred Hearth (Linea D1), by a grant from the Italian MIUR (2004067822_004) (to S.Z.), and by the V Programma Nazionale Ricerca sull'AIDS (50F.13).
Editor: J. L. Flynn
REFERENCES
- 1.Balasubramanian, V., E. H. Wiegeshaus, B. T. Taylor, and D. W. Smith. 1994. Pathogenesis of tuberculosis: pathway to apical localization. Tuber. Lung Dis. 75:168-178. [DOI] [PubMed] [Google Scholar]
- 2.Collins, F. M. 1985. Protection to mice afforded by BCG vaccines against an aerogenic challenge by three mycobacteria of decreasing virulence. Tubercle 66:267-276. [DOI] [PubMed] [Google Scholar]
- 3.Delogu, G., and M. J. Brennan. 1999. Functional domains present in the mycobacterial hemagglutinin, HBHA. J. Bacteriol. 181:7464-7469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Delogu, G., A. Li, C. Repique, F. Collins, and S. L. Morris. 2002. DNA vaccine combinations expressing either tissue plasminogen activator signal sequence fusion proteins or ubiquitin-conjugated antigens induce sustained protective immunity in a mouse model of pulmonary tuberculosis. Infect. Immun. 70:292-302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gold, B., G. M. Rodriguez, S. A. E. Marras, M. Pentecost, and I. Smith. 2001. The M. tuberculosis IdeR is a dual functional regulator that controls transcription of genes involved in iron acquisition, iron storage and survival in macrophages. Mol. Microbiol. 42:851-865. [DOI] [PubMed] [Google Scholar]
- 6.Goonetilleke, N. P., H. McShane, C. M. Hannan, R. J. Anderson, R. H. Brookes, and A. V. S. Hill. 2003. Enhanced immunogenicity and protective efficacy against M. tuberculosis of Bacille Calmette-Guérin vaccine using mucosal administration and boosting with a recombinant modified vaccinia virus Ankara. J. Immunol. 171:1602-1609. [DOI] [PubMed] [Google Scholar]
- 7.Hess, J., D. Miko, A. Catic, V. Lehmensiek, D. G. Russell, and S. H. Kaufmann. 1998. M. bovis Bacille Calmette-Guerin strains secreting listeriolysin of Listeria monocytogenes. Proc. Natl. Acad. Sci. USA 95:5299-5304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Horwitz, M. A., G. Harth, B. J. Dillon, and S. Maslesa-Galic'. 2000. Recombinant bacillus Calmette-Guerin (BCG) vaccines expressing the M. tuberculosis 30-kDa major secretory protein induce greater protective immunity against tuberculosis than conventional BCG vaccines in a highly susceptible animal model. Proc. Natl. Acad. Sci. USA 97:13853-13858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kaufmann, S. H., and A. J. McMichael. 2005. Annulling a dangerous liaison: vaccination strategies against AIDS and tuberculosis. Nat. Med. 11:S33-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kraft, S. L., D. Dailey, M. Kovach, K. L. Stasiak, J. Bennett, C. T. McFarland, D. N. McMurray, A. A. Izzo, I. M. Orme, and R. J. Basaraba. 2004. Magnetic resonance imaging of pulmonary lesions in guinea pigs infected with Mycobacterium tuberculosis. Infect. Immun. 72:5963-5971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mahenthiralingam, E. 1998. Extraction of RNA from mycobacteria. Methods Mol. Biol. 101:75. [DOI] [PubMed] [Google Scholar]
- 12.Manganelli, R., E. Dubnau, S. Tyagi, F. R. Kramer, and I. Smith. 1999. Differential expression of 10 sigma factor genes in M. tuberculosis. Mol. Microbiol. 31:715-724. [DOI] [PubMed] [Google Scholar]
- 13.Manganelli, R., M. I. Voskuil, G. K. Schoolnik, and I. Smith. 2001. The M. tuberculosis ECF sigma factor sigmaE: role in global gene expression and survival in macrophages. Mol. Microbiol. 41:423-437. [DOI] [PubMed] [Google Scholar]
- 14.Masungi, C., S. Temmerman, J. P. Van Vooren, A. Drowart, K. Pethe, F. D. Menozzi, C. Locht, and F. Mascart. 2002. Differential T and B cell responses against M. tuberculosis heparin-binding hemagglutinin adhesin in infected healthy individuals and patients with tuberculosis. J. Infect. Dis. 185:513-520. [DOI] [PubMed] [Google Scholar]
- 15.McMurray, D. N. 2003. Hematogenous reseeding of the lung in low-dose, aerosol-infected guinea pigs: unique features of the host-pathogen interface in secondary tubercles. Tuberculosis (Edinburgh). 83:131-134. [DOI] [PubMed] [Google Scholar]
- 16.Meijerink, J., C. Mandingers, L. van de Locht, E. Tonnissen, F. Goodsaid, and J. Raemaekers. 2001. A novel method to compensate for different amplification effeciencies between patient DNA samples in quantitative real-time PCR. J. Mol. Diagn. 3:61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Menozzi, F. D., R. Bischoff, E. Fort, M. J. Brennan, and C. Locht. 1998. Molecular characterization of the mycobacterial heparin-binding hemagglutinin, a mycobacterial adhesin. Proc. Natl. Acad. Sci. USA 95:12625-12630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Menozzi, F. D., V. M. Reddy, D. Cayet, D. Raze, A. S. Debrie, M. P. Dehouck, R. Cecchelli, and C. Locht. 2005. Mycobacterium tuberculosis heparin-binding haemagglutinin adhesin (HBHA) triggers receptor-mediated transcytosis without altering the integrity of tight junctions. Microbes. Infect. 8:1-9. [DOI] [PubMed] [Google Scholar]
- 19.Menozzi, F. D., J. H. Rouse, M. Alavi, M. Laude-Sharp, J. Muller, R. Bischoff, M. J. Brennan, and C. Locht. 1996. Identification of a heparin-binding hemagglutinin present in mycobacteria. J. Exp. Med. 184:993-1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Parra, M., T. Pickett, G. Delogu, V. Dheenadhayalan, A. S. Debrie, C. Locht, and M. J. Brennan. 2004. The mycobacterial heparin-binding hemagglutinin is a protective antigen in the mouse aerosol challenge model of tuberculosis. Infect. Immun. 72:6799-6805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Peirson, S. N., J. N. Butler, and R. G. Foster. 2003. Experimental validation of novel and conventional approaches to quantitative real-time PCR data analysis. Nucleic Acids Res. 31:e73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pethe, K., S. Alonso, F. Biet, G. Delogu, M. J. Brennan, C. Locht, and F. D. Menozzi. 2001. The heparin-binding haemagglutinin of M. tuberculosis is required for extrapulmonary dissemination. Nature 412:190-194. [DOI] [PubMed] [Google Scholar]
- 23.Pethe, K., P. Bifani, H. Drobecq, C. Sergheraert, A.-S. Debrie, C. Locht, and F. D. Menozzi. 2002. Mycobacterial heparin-binding hemagglutinin and laminin-binding protein share antigenic methyllysines that confer resistance to proteolysis. Proc. Natl. Acad. Sci. USA 99:10759-10764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rhoades, E. R., A. A. Frank, and I. M. Orme. 1997. Progression of chronic pulmonary tuberculosis in mice aerogenically infected with virulent M. tuberculosis. Tuber. Lung Dis. 78:57-66. [DOI] [PubMed] [Google Scholar]
- 25.Rueda, S., M. Vicente, and J. Mingorance. 2003. Concentration and assembly of the division ring proteins FtsZ, FtsA, and ZipA during the Escherichia coli cell cycle. J. Bacteriol. 185:3344-3351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sanguinetti, M., B. Posteraro, B. Fiori, S. Ranno, R. Torelli, and G. Fadda. 2005. Mechanisms of azole resistance in clinical isolates of Candida glabrata collected during a hospital survey of antifungal resistance. Antimicrob. Agents Chemother. 49:668-679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shi, L., Y. Jung, S. Tyagi, M. L. Gennaro, and R. North. 2003. Expression of Th1-mediated immunity in mouse lungs induces a M. tuberculosis transcription pattern characteristic of nonreplicating persistence. Proc. Natl. Acad. Sci. USA 100:241-246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shi, L., R. North, and M. L. Gennaro. 2004. Effect of growth state on transcription levels of genes encoding major secreted antigens of Mycobacterium tuberculosis in the mouse lung. Infect. Immun. 72:2420-2424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Temmerman, S., K. Pethe, M. Parra, S. Alonso, C. Rouanet, T. Pickett, A. Drowart, A. S. Debrie, G. Delogu, F. D. Menozzi, C. Sergheraert, M. J. Brennan, F. Mascart, and C. Locht. 2004. Methylation-dependent T cell immunity to M. tuberculosis heparin-binding hemagglutinin. Nat. Med. 10:935-941. [DOI] [PubMed] [Google Scholar]
- 30.Timm, J., F. A. Post, L. G. Bekker, G. B. Walther, H. C. Wainwright, R. Manganelli, W. Chan, L. Tsenova, B. Gold, I. Smith, G. Kaplan, and J. D. McKinney. 2003. Differential expression of iron-, carbon-, and oxygen-responsive mycobacterial genes in the lungs of chronically infected mice and tuberculosis patients. Proc. Natl. Acad. Sci. USA 100:14321-14326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Weart, R. B., and P. A. Levin. 2003. Growth rate-dependent regulation of a medial FtsZ ring formation. J. Bacteriol. 185:2826-2834. [DOI] [PMC free article] [PubMed] [Google Scholar]