Abstract
In synthetic phenotype vascular smooth muscle cells (VSMC), activation of epidermal growth factor (EGF) receptor (EGFR) induces a sustained increase in intermediate conductance KCa (int-KCa; KCa3.1) channels that is essential for proliferation. However, a comparable mechanism has not been identified in native contractile phenotype VSMC, which express large conductance KCa (maxi-KCa; KCa1.1) channels, not int-KCa channels. Using patch clamp of freshly isolated contractile VSMC from rat basilar artery, we found that EGF (100 ng ml−1) caused hyperpolarization (7.9 ± 3.9 mV) due to activation of iberiotoxin-sensitive, maxi-KCa channels. The EGFR ligands EGF (100 ng ml−1), transforming growth factor α (0.4 ng ml−1) and heparin-binding EGF (100 ng ml−1) all caused a 20% increase in maxi-KCa channel current that was blocked by AG-1478 or by knock-down of EGFR expression using cisterna magna infusion of antisense oligodeoxynucleotide (AS-ODN). In controls, EGFR knock-down, and EGFR gain-of-expression (angiotensin II hypertension), the increase in maxi-KCa current correlated with the abundance of EGFR protein expressed. The EGFR-mediated increase in maxi-KCa channel activity was blocked by inhibiting cAMP-dependent protein kinase (cAK) using KT-5720 or Rp-cAMP, or by inhibiting adenylate cyclase type 5 (AC-5) using 2′,5′-dideoxyadenosine or knock-down of AC-5 expression by intracisternal AS-ODN. Direct infusion of EGF into cisterna magna caused up-regulation of proliferating cell nuclear antigen (PCNA) in VSMC that was prevented by coinfusion of iberiotoxin or of AG-1478. Our data, which are consistent with the hypothesis that hyperpolarization is critical for a proliferative response, are the first to implicate AC-5 and maxi-KCa channels in gene activation related to EGFR signalling in native contractile VSMC.
Hyperpolarization is essential for multifunctional growth signalling responses. In many types of cells, activation of K+ channels is required for G1 progression of the cell cycle, and proliferation is almost invariably inhibited by K+ channel blockers (Dubois & Rouzaire-Dubois, 1993; Wonderlin & Strobl, 1996). In vascular smooth muscle cells (VSMC) as well, K+ channel function is critical for growth factor signalling and growth factor-induced proliferation (Neylon, 2002).
Epidermal growth factor (EGF) receptor (EGFR) is a single transmembrane domain receptor tyrosine kinase that plays an important role in growth signalling. In a variety of cells, activation of EGFR induces a sustained increase in K+ channel activity that results in prolonged hyperpolarization (Pandiella et al. 1989; Lovisolo et al. 1992; Wang et al. 1997; Xu et al. 1999; Roderick et al. 2003). In the synthetic phenotype of VSMC, the phenotype that typifies cultured VSMC, EGFR induces hyperpolarization by direct tyrosine phosphorylation of intermediate conductance Ca2+-activated K+ (int-KCa) channels (Kohler et al. 2003). However, this mechanism cannot operate in contractile phenotype VSMC, the phenotype that typifies healthy VSMC in vivo, because contractile VSMC do not express int-KCa channels (Neylon et al. 1999). Contractile VSMC express predominantly large conductance Ca2+-activated K+ (maxi-KCa) channels which are not tyrosine phosphorylated by EGFR. Potential involvement of K+ channels in EGFR signalling in contractile VSMC has not been examined.
Proliferative responses have been studied extensively in synthetic phenotype VSMC, but not in the contractile phenotype. Primary cultured or early passage cultured cells are often represented as useful models for study of the contractile phenotype, but ultimately only VSMC in vivo or immediately after isolation meet the definitional criteria of contractility. Thus, studies of molecular and cellular mechanisms of proliferative responses that require hours or days to unfold present significant technical challenges if they are to address mechanisms in contractile phenotype VSMC. Notably, cerebral vessels such as the basilar artery are unique among arteries in the body, in that they contain a rete vasorum in the adventitia that is permeable to large molecules and that effectively places the extracellular space of VSMC in direct continuity with subarachnoid space (Zervas et al. 1982). The existence of a rete vasorum can be exploited to deliver substances directly to contractile phenotype VSMC in vivo by infusion into the cerebrospinal fluid of the cisterna magna.
In the present study, we made use of this feature of the basilar artery to study the proliferative response of native contractile VSMC following EGFR activation. First, we sought to determine if contractile VSMC respond to EGF stimulation by hyperpolarization, and if so, by what mechanism. Second, we sought to determine the effect of EGF stimulation on gene activation in vivo. Using freshly isolated basilar artery VSMC, we found that EGF and the related ligands transforming growth factor α (TGFα) and heparin binding-EGF (HB-EGF) act via EGFR to cause sustained cellular hyperpolarization attributable to activation of maxi-KCa but not int-KCa channels, and that activation of maxi-KCa channels by EGFR requires the intermediate molecules, AC-5 and cAK. Then, using cisterna magna infusions, we determined that key EGFR signalling events identified in freshly isolated cells are intimately involved in vivo in activation of proliferating cell nuclear antigen (PCNA), which is known to be critical for gene activation in the programme of VSMC proliferation (Lee et al. 1999; Paunesku et al. 2001). Our data, which are consistent with the hypothesis that hyperpolarization is critical for the proliferative response of VSMC following EGFR activation, are the first to implicate AC-5 and maxi-KCa channels in gene activation related to EGFR signalling in native contractile VSMC.
Methods
Animal models
Animal protocols adhered strictly to guidelines for the humane treatment of animals, and were approved by the Institutional Animal Care and Use Committee of the University of Maryland. Experiments were carried out using adult female Wistar rats (275–325 g). For survival surgery, animals were fasted overnight, anaesthetized (ketamine, 60 mg kg−1 plus xylazine, 7.5 mg kg−1, i.p.), and underwent surgical procedures using strictly aseptic techniques. For tissue harvest, animals were killed by intraperitoneal injection of an overdose of sodium pentobarbital (120 mg kg−1).
For knock-down of specific gene targets, rats were implanted with a mini-osmotic pump (Alzet 2001, Alza Corporation, Palo Alto, CA, USA; delivery rate, 1.0 μl h−1), with the body of the pump placed subcutaneously in the dorsal thorax, and the delivery catheter inserted 1–2 mm into the cisterna magna and secured in place with α-cyanoacrylate adhesive. Animals experiencing subarachnoid haemorrhage secondary to trauma at surgery, whether discovered at the time of surgery or at the time of kill, were discarded. For knock-down of EGFR, we used the following antisense oligodeoxynucleotide (AS-ODN): 5′-TGAGGGTCGCATCCC-3′, purified by HPLC. As a control, we used the corresponding sense oligodeoxynucleotide (SE-ODN): 5′-GGGATGCGACCCTCA-3′. For knock-down of AC-5, we used a 50 : 50 mixture of the following AS-ODN: 5′-GTCGTCTTCTTCCTCTTCCC-3′ and 5′-TCCTCCATCTCCTCCTTCTCT-3′, purified by HPLC. As a control, we used a 50 : 50 mixture of the corresponding SE-ODN: 5′-AGAGAAGGAGGAGATGGAGGA-3′ and 5′-GGGAAGAGGAAGAAGACGAC-3′. ODN solutions were made up as 60 μm solutions in aCSF, which contained (mm): NaCl, 119; KCl, 2.5; CaCl2, 2.5; MgCl2, 1.3; NaHCO3, 26; NaH2PO4, 1.0; d-glucose, 11. Pumps were left in place for 3–4 days, after which animals were killed for harvest of basilar arteries.
For direct delivery of ligands or other agents into cisterna magna, mini-osmotic pumps (Alzet 2001, delivery rate, 1.0 μl h−1) were implanted as above, but contained: (i) EGF, 10 μg ml−1; or (ii) EGF, 10 μg ml−1 plus AG-1478, 500 μg ml−1; or (iii) EGF, 10 μg ml−1 plus iberiotoxin, 4 μg ml−1, all in aCSF; or (iv) aCSF alone.
For gain-of-expression of EGFR, we used angiotensin-hypertensive rats (AHR), which were prepared as previously described (Gerzanich et al. 2003a, c). Briefly, under general anaesthesia, animals were implanted with mini-osmotic pumps (Alzet 2004; delivery rate, 0.25 μl h−1) placed subcutaneously in the dorsal thorax that delivered angiotensin II, 2.25 mg ml−1, in 0.9% NaCl (Sigma, St Louis MO, USA) for 4 weeks.
Patch clamp
Patch clamp experiments were carried out using VSMC from basilar arteries isolated enzymatically as described (Gerzanich et al. 2003a, b). Methods used for patch clamp recording of maxi-KCa channels in this lab have been described (Gerzanich et al. 2003a, b). All voltage clamp recordings were performed using a holding potential of 0 mV, and included on-line leak-subtraction (P/−4 protocol; pCLAMP), with leak currents measured during −15 or −20 mV pulses from −30 mV. For current clamp recordings, cells were discarded if they exhibited an unstable baseline membrane potential.
For standard whole cell recording, the pipette contained (mm): KCl, 145; MgCl2, 2; Hepes, 10; glucose, 10; Mg2ATP, 5; EGTA, 5; CaCl2, 1.8 (free [Ca2+], 100 nm); pH 7.2; and the bath contained (mm): NaCl, 140; KCl, 5; CaCl2, 0.1; MgCl2, 2; Hepes 10; glucose, 12.5; pH 7.4. For nystatin perforated patch recording, the pipette contained (mm): KCl, 25; K2SO4, 100; MgCl2, 8; Hepes, 10; and nystatin 130 μg ml−1; pH 7.2.
Drugs and reagents used included: epidermal growth factor (EGF), transforming growth factor α (TGFα), heparin-binding EGF (HB-EGF), iberiotoxin, 8-Br-cAMP and 8-Br-cGMP, which were obtained from Sigma; ATP-γS, AG-1478, AG-9, KT-5720, KT-5823, Rp-8Br-PET-cGMP and Rp-cAMP, which were obtained from Calbiochem (San Diego, CA, USA); and 2′,5′-dideoxyadenosine (2′,5′-dd-Ado), which was generously supplied by Dr R. A. Johnson (Johansson Biochemicals, Stony Brook, NY, USA).
Immunofluorescence
Animals were perfusion-fixed with 4% paraformaldehyde in PBS and brains were processed either for cryosectioning (10 μm, for AC-5 and caveolin-1) or for paraffin sectioning (4 μm for EGFR and PCNA). For caveolin-1 labelling, we performed antigen retrieval by microwaving sections at 800 W, 3 times for 2 min, with a 3-min interval between heatings, and followed by 30 min for cooling. We used primary antibodies directed against EGFR (1 : 200; Upstate, Lake Placid, NY, USA), AC-5 (1 : 200; FabGennix, Frisco, TX, USA), caveolin-1 (1 : 100; Clone 2297, BD Transduction Laboratories, San Jose, CA, USA) and PCNA (1 : 400; Santa Cruz Biotechnology, Santa Cruz, CA, USA). The secondary antibodies used were: CY3-conjugated goat antirabbit (1 : 400, Jackson ImmunoResearch Laboratories, West Grove, PA, USA) for EGFR and PCNA; Alexa 546-conjugated goat antirabbit (1 : 400, Molecular Probes/Invitrogen, Carlsbad, CA, USA) for AC-5; Alexa 488-conjugated goat antimouse (1 : 200, Molecular Probes) for caveolin-1. For all immunolabellings, omission of primary antibodies was used as a negative control, and labellings were carried out using tissues from three or more animals.
For quantitative immunofluorescence (Cress, 2000) for EGFR, tissue sections from all animals in all experimental group were immunolabelled as a single batch. Images were collected using a Nikon Eclipse E1000 microscope and a SenSys digital camera (Photometrics, Tucson, AZ, USA) with IPLab software (v. 3.06, Scanalytics, Fairfax, VA, USA) using uniform parameters of magnification and exposure. Single-plane wide-field images were deconvoluted using a point-spread function computed with microscope-specific optical parameters (Huygens Essential, SVI, Hilversum, the Netherlands), and the percentage area occupied by ‘bright particles’ in equal-sized regions-of-interest (%ROI) within VSMC layers was computed using IPLab software, as previously described (Gerzanich et al. 2003a).
Western Blots
For Western blots, basilar artery lyates were prepared as described (Gerzanich et al. 2003a). Blots were developed using antibodies directed against EGFR (UpState), AC-5 (FabGennix), phospho-EGFR (Upstate) and total actin (Santa Cruz Biotechnology).
Data analysis
For repeated measures of electrophysiological recordings, multiple cells from at least three animals were typically studied. Similarly, all immunohistochemical and Western blot analyses were carried out with tissues sampled from three or more animals. Statistical comparisons were evaluated using either ANOVA, with Tukey's means comparison, or Student's t test, as appropriate. Data are given as the mean ± s.e.m. unless otherwise noted.
Results
EGF induces hyperpolarization by activating maxi-KCa channel
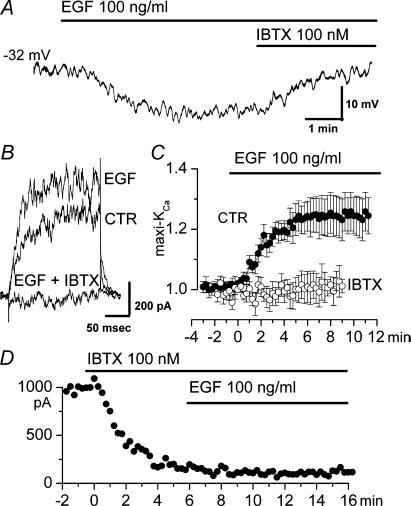
We first examined the effect of EGF on the membrane potential (Em) of freshly isolated VSMC from rat basilar artery. In a group of 43 cells with a stable resting potential, Em varied from −18 to −50 mV (mean ±s.d., −35.4 ± 9.6 mV), as previously observed (Holland et al. 1996). After monitoring cells for 5–10 min to assure stability of Em, addition of EGF (100 ng ml−1) to the bath caused a sustained hyperpolarization in 21/43 cells that ranged in magnitude from 4 to 15 mV (mean ±s.d., 7.9 ± 3.9 mV, 21 cells) (Fig. 1A). In 3/43 cells, an initial hyperpolarization was followed by depolarization, and in another 3/43, a small depolarization alone was observed. In 16/43 cells, EGF caused no change in baseline current. In cells with hyperpolarization, the response began ≈1 min after addition of EGF and reached a maximum at 3–5 min. The hyperpolarizing effect of EGF was not reversed by washout of ligand for 5 min or more (3 cells), but addition of iberiotoxin (100 nm) to the bath reversed the EGF-induced hyperpolarization and returned Em to its baseline value (5 cells) (Fig. 1A).
Figure 1. Epidermal growth factor (EGF) causes hyperpolarization by activating maxi-KCa channel in freshly isolated basilar artery smooth muscle cells.
A, current clamp recording showing hyperpolarization induced by EGF (100 ng ml−1) that was reversed by subsequent addition of iberiotoxin (100 nm). B, membrane current during test pulses to +60 mV (holding potential, 0 mV) before (CTR) and after addition of EGF (100 ng ml−1), and after addition of iberiotoxin (IBTX; 100 nm). C, normalized change (mean ± s.e.m.) in membrane current with addition of EGF (100 ng ml−1) in the absence of (•; 18 cells) and in the presence of (○; 8 cells) iberiotoxin (100 nm). Measurements of normalized currents were obtained from test pulses to +60 or +80 mV from a holding potential of 0 mV; conventional whole cell patch clamp technique. D, end-of-pulse current during test pulses to +60 mV (holding potential, 0 mV) before and after addition of iberiotoxin (IBTX; 100 nm) and after addition of EGF (100 ng ml−1).
Voltage-clamp experiments were used to identify the channel involved in the EGF-induced hyperpolarization. Because iberiotoxin had been found to reverse the EGF-induced hyperpolarization, we focused on maxi-KCa channels. We used a conventional whole cell configuration and recording conditions optimized for maxi-KCa channels, including a holding potential of 0 mV to inactivate voltage-dependent currents. As we and others previously reported (Holland et al. 1996; Gerzanich et al. 2003a), under these conditions, the cells exhibited macroscopic outward currents attributable to maxi-KCa but not int-KCa channels, as suggested by two lines of evidence. First, single channel recordings of inside-out patches showed channel openings with a single channel conductance of 150–160 pS, typical of maxi-KCa (Gerzanich et al. 2003a), but no openings attributable to int-KCa channels. Second, currents were sensitive to block by both iberiotoxin and charybdotoxin, but when first blocked using iberiotoxin, subsequent addition of charybdotoxin produced no further block. Since both toxins are potent blockers of maxi-KCa channels, but only charybdotoxin blocks both maxi-KCa and int-KCa channels (Neylon et al. 1999), this finding indicated that int-KCa channels did not contribute significantly to membrane currents.
When EGF (100 ng ml1) was added to the bath, an increase in current was observed in 18/25 cells tested (Fig. 1B and C, filled circles). The increase in current started 1–1.5 min after beginning perfusion with EGF, and reached a maximum at ∼6 min. The effect of EGF was not reversed by 5-min washout of ligand (2 cells). The EGF-induced increase in maxi-KCa current was not accompanied by any apparent change in kinetics or voltage dependence of the current (Fig. 1B). Also, the magnitude of the effect of EGF was the same at all voltages tested, i.e. the effect was not voltage dependent.
After a response to EGF had developed, subsequent addition of iberiotoxin (100 nm) to the bath caused a complete block of currents (3 cells) (Fig. 1B). When iberiotoxin (100 nm) was first added to the bath, subsequent addition of EGF had no effect on the outward current (8 cells) (Fig. 1D and C, open circles). These data demonstrated that the recording conditions we used favoured iberiotoxin-sensitive maxi-KCa channel current, and confirmed involvement of iberiotoxin-sensitive maxi-KCa channels in the response to EGF.
In our voltage clamp experiments, we studied effects of 5–500 ng ml−1 EGF. A clear concentration–response relationship was difficult to establish. This was due, in part, to cell-to-cell variability in the response to EGF, but also to an apparently steep concentration–response relationship. In general, concentrations < 10 ng ml−1 were ineffective, whereas concentrations > 50 ng ml−1 appeared to produce largely similar responses. Overall, when measured using test pulses to +60 or +80 mV (holding potential, 0 mV), 100 ng ml−1 EGF produced a mean increase in current of 21.6 ± 5.1% (18 cells) (Fig. 1C, filled circles). All subsequent experiments with EGF were carried out with 100 ng ml−1 of ligand.
Involvement of EGFR
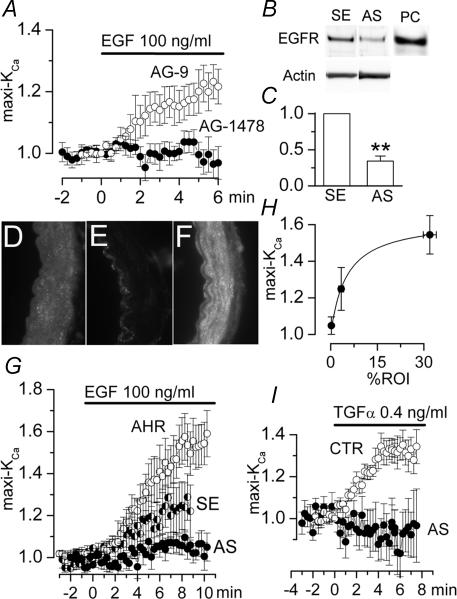
We used AG-1478, a selective blocker of EGFR (Levitzki & Gazit, 1995), to assess involvement of this receptor. When AG-1478 was included in the pipette solution, exposure of the cells to EGF no longer resulted in an increase in current (Fig. 2A, filled circles). By contrast, addition of the inactive tyrphostin AG-9 to the pipette solution did not prevent the EGF-induced increase in maxi-KCa current (Fig. 2A, open circles).
Figure 2. Link between EGFR activation and maxi-KCa channel activation.
A, normalized change (mean ± s.e.m.) in membrane current with addition of EGF (100 ng ml−1) in the presence of AG-9 (100 μm; ○; 4 cells) or AG-1478 (0.5 μm; •; 8 cells) in the pipette solution. B and C, Western blots (B) and bar graphs of densitometric analysis of Western blots (C) for EGFR in basilar artery lysates from controls (SE) and from EGFR knock-down (AS) rats; for EGFR knock-down, rats underwent constant infusion of antisence (AS) oligodeoxynucleotide directed against EGFR into the cisterna magna for 4 days (3 rats); for controls, rats underwent 4-day infusion into cisterna magna of corresponding sense (SE) oligodeoxynucleotide (3 rats); bar graphs show values normalized to controls, after first normalizing data from individual lanes to density of total actin; **P < 0.01; panel B also shows blots for total actin and a positive control (PC) for EGFR (30 μg total lysate of human epithelial carcinoma cells obtained from UpState Biotechnology, Lake Placid, NY, USA). D–F, immunofluorescence images of basilar artery sections from a control rat (D), from an EGFR knock-down rat (E) and from an EGFR gain-of-expression rat (F); the endothelial layer is always to the left; control and EGFR knock-down rats were prepared as above; EGFR gain-of-expression rats received constant infusion of angiotensin II subcutaneously for 28 days (angiotensin hypertensive rats; AHR); the secondary antibody was a CY3-conjugate, the filter for which shows the basal lamina as a dark band. G, normalized change (mean ± s.e.m.) in membrane current with addition of EGF (100 ng ml−1) in 7 cells from EGFR knock-down rats (AS), 7 cells from controls rats (SE), and 8 cells from EGFR gain-of-expression rats (AHR). H, plot of normalized change (mean ± s.e.m.) in membrane current 8 min after addition of EGF (100 ng ml−1) (7–8 cells/condition) (same data as in G) versus abundance (mean ± s.e.m.) of EGFR expressed (arteries from 3 to 4 rats/condition) (same data as in D–F); abundance of EGFR in smooth muscle layers is expressed as percentage ROI and was determined from analysis of immunofluorescence images, as described in Methods; the line represents a least-squares fit to a standard logistic equation. I, normalized change (mean ± s.e.m.) in membrane current with addition of TGFα (0.4 ng ml−1) in 9 cells from untreated control rats (CTR) and 7 cells from EGFR knock-down rats (AS). For A, G and I, measurements of normalized currents were obtained from test pulses to +60 or +80 mV from a holding potential of 0 mV; conventional whole cell patch clamp technique.
To further assess involvement of EGFR, we developed an EGFR knock-down model in which antisense oligodeoxynucleotide (AS-ODN) directed against EGFR was infused into the cisterna magna. Infusion of sense oligodeoxynucleotide (SE-ODN) was used as a control. Western blots (Fig. 2B and C) combined with immunofluorescence imaging (Fig. 2D and E) showed that basilar arteries from EGFR knock-down animals (AS-ODN) expressed significantly less EGFR compared to controls (SE-ODN). Notably, the reduction with AS-ODN appeared to be specific for VSMC layers, and was not evident in endothelium, consistent with the interpretation that the basal lamina had acted as a diffusion barrier for ODN placed in the subarachnoid space.
Patch clamp study of VSMC isolated from EGFR knock-down animals was carried out using the same conditions as above. Maxi-KCa currents showed no apparent changes in magnitude, kinetics, voltage dependence and block by pharmacological agents. However, in cells from EGFR knock-down animals, exposure to EGF resulted in little or no effect on maxi-KCa currents, whereas in control cells from SE-ODN animals, EGF caused the typical increase of ∼20% in maxi-KCa current (Fig. 2G, filled versus half-filled circles). The responses at 8 min for the two groups, SE versus AS, were significantly different (P < 0.05, by ANOVA).
Hypertension is known to up-regulate EGF signalling and EGFR expression in VSMC (Florian & Watts, 1999; Florian et al. 2001). We studied basilar arteries from angiotensin hypertensive rats (AHR) (Gerzanich et al. 2003a, c). Immunofluorescence imaging showed that basilar arteries from AHR expressed significantly more EGFR in VSMC layers compared to arteries from controls (Fig. 2FversusD), consistent with AHR being a useful model for EGFR gain-of-expression. Patch clamp study of VSMC isolated from AHR has previously been reported, but briefly, when studied under the same conditions as above, these cells show normal appearing maxi-KCa currents (Gerzanich et al. 2003a). In cells from AHR, exposure to EGF resulted in a large augmentation in maxi-KCa currents, with the magnitude of the response appreciably greater than controls (Fig. 2G, open circles). The responses at 8 min for the two groups, SE versus AHR, were significantly different (P < 0.05, by ANOVA).
We quantified the amount of EGFR expressed in VSMC layers of basilar arteries from each condition: control rats (SE-ODN), EGFR knock-down rats (AS-ODN), and EGFR gain-of-expression rats (AHR) (3–4 rats per condition). To permit analysis of VSMC without contamination by endothelium, we used a quantitative immunofluorescence technique (see Methods). A scatter plot of the relationship between EGFR expressed in VSMC layers versus the magnitude of the response to EGF (100 ng ml−1) in VSMC is shown for the three conditions (Fig. 2H). The data were fitted with a simple logistic equation.
Together, these data showing that the response to EGF was blocked by the specific EGFR inhibitor AG-1478 as well as by knock-down of EGFR expression, and that the magnitude of the response was directly correlated with the amount of EGFR expressed, provided strong evidence that the effect of EGF on maxi-KCa channels was mediated completely and exclusively by EGFR.
Other EGFR ligands
The most abundant endogenous ligand for EGFR in the brain is transforming growth factor α (TGFα) (Lazar & Blum, 1992). In voltage clamp experiments, we studied effects of 0.1–10 ng ml−1 of TGFα, with the optimal response obtained using 0.4 ng ml−1 of ligand. TGFα caused an increase in maxi-KCa channel activity, with a time course and magnitude similar to our previous observations with EGF (Fig. 2I, open circles). When measured using test pulses to +60 mV (HP, 0 mV), the mean increase in current with 0.4 ng ml−1 of TGFα was 31.6 ± 0.8% (9 cells).
We used basilar artery VSMC from the EGFR knock-down model to confirm involvement of this receptor in the actions of TGFα. In VSMC from the EGFR knock-down animals, exposure to TGFα resulted in no increase in maxi-KCa currents (Fig. 2I, filled circles), consistent with the effect of TGFα being mediated by EGFR.
Another important ligand for EGFR is heparin-binding EGF (HB-EGF), an endogenous membrane-bound ligand that is involved in EGFR transactivation by G-protein coupled receptors. Addition of HB-EGF (100 ng ml−1) caused an increase in maxi-KCa channel activity with a time course and magnitude similar to our observations with EGF and with TGFα (not shown). When measured using test pulses to +60 mV (HP, 0 mV), the mean increase in current with HB-EGF (100 ng ml−1) was 19.9 ± 1.3% (7 cells).
Cytoplasmic messengers
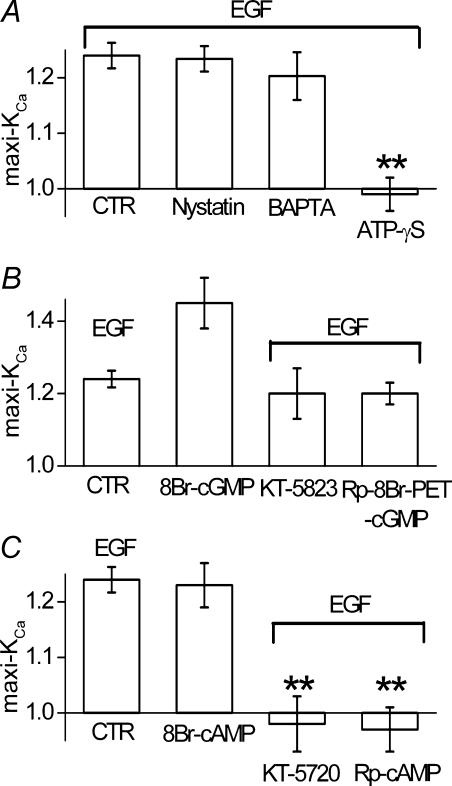
Our previous experiments were carried out using a conventional whole cell recording technique, which is associated with rapid depletion of small molecules from the cytoplasm. To check for possible involvement of cytoplasmic messengers that are potentially lost by whole cell dialysis, we studied a series of cells using a nystatin-perforated patch technique. In cells studied using a nystatin patch, EGF (100 ng ml−1) caused a mean increase in maxi-KCa current of 23.4 ± 2.3%, which was not significantly different (by ANOVA) from the response with the conventional whole cell method (Fig. 3A, nystatin versus CTR), suggesting that diffusible cytoplasmic molecules were unlikely to be critical for the response to EGF.
Figure 3. cAK mediates maxi-KCa channel activation by EGFR.
A, bar graph of normalized change in membrane current 8–10 min after addition of EGF (100 ng ml−1), measured using: (i) our ‘standard conditions’, including conventional whole cell technique plus 5 mm EGTA and 5 mm Mg2ATP in the pipette solution (CTR); (ii) a nystatin-perforated patch technique (Nystatin); (iii) our standard conditions except with 10 mm BAPTA instead of EGTA in the pipette (BAPTA); (iv) our standard conditions except with ATP-γS (5 mm) instead of Mg2ATP in the pipette (ATP-γS). B, bar graph of normalized change in membrane current measured using our standard conditions, after addition of EGF (100 ng ml−1) (CTR), after addition of 8-Br-cGMP (100 μm) (8-Br-cGMP), after addition of EGF (100 ng ml−1) in the presence of KT-5823 (1 μm in the bath) (KT-5823), after addition of EGF (100 ng ml−1) in the presence of Rp-8Br-PET-cGMP (1 μm in the pipette) (Rp-8Br-PET-cGMP). C, bar graph of normalized change in membrane current measured using our standard conditions, after addition of EGF (100 ng ml−1) (CTR), after addition of 8-Br-cAMP (100 μm) (8-Br-cAMP), after addition of EGF (100 ng ml−1) in the presence of KT-5720 (1 μm in the bath) (KT-5720), after addition of EGF (100 ng ml−1) in the presence of Rp-cAMP (0.5 μm in the pipette) (Rp-cAMP). **P < 0.01; all measurements of normalized currents were obtained from test pulses to +60 or +80 mV from a holding potential of 0 mV; bars for CTR are from the same data as in Fig. 1C; all bars for data other than CTR represent the mean ± s.e.m. for 5–9 cells.
Our previous whole cell experiments utilized EGTA to buffer intracellular Ca2+, but EGTA has a relatively slow on-rate of Ca2+ binding (Tsien, 1980), making it difficult to exclude potential involvement of a Ca2+ release mechanism in the effect of EGF (Naraghi & Neher, 1997). As a check on this possibility, we studied a series of cells in which EGTA (5 mm) was replaced with BAPTA (10 mm), which has much faster on-rate of Ca2+ binding (Tsien, 1980), maintaining [Ca2+]I at 100 nm. In cells studied with BAPTA, EGF (100 ng ml−1) caused a mean increase in maxi-KCa current of 20.3 ± 4.3%, which was not significantly different (by ANOVA) from the response with EGTA (Fig. 3A, BAPTA versus CTR), suggesting that a Ca2+-release mechanism was unlikely to be involved in the response to EGF.
We also examined whether different levels of extracellular Ca2+ would affect the response to EGF. No differences in response to EGF (100 ng ml−1) were observed by changing extracellular Ca2+ from our standard 100 μm to 0 mm (15 cells) and 2 mm (5 cells), suggesting that Ca2+ influx or extracellular Ca2+ binding were not critical in the response to EGF.
We also assessed for involvement phosphorylation. For this, we substituted non-hydrolysable ATP-γS for ATP in the pipette solution. With ATP-γS, maxi-KCa currents were very stable during prolonged recordings, but addition of EGF resulted in no significant change in current (Fig. 3A, ATP-γS versus CTR). This experiment indicated that one or more phosphorylation steps is important for EGFR activation of maxi-KCa channels.
Involvement of cAK but not cGK
To assess for potential involvement of cGK, we first confirmed that addition of the membrane-permeant activator of cGK, 8-Br-cGMP, would increase maxi-KCa current. Addition of 100 μm 8-Br-cGMP, a concentration that produces near-maximal activation of maxi-KCa channels (Gerzanich et al. 2003a), caused an increase in current of ∼40% (Fig. 3B, 8Br-cGMP). We next evaluated the response to EGF in the presence of the cGK inhibitor KT-5823. Upon addition to the bath, this compound itself suppressed maxi-KCa current by about 50%, but subsequent addition of EGF in the presence of KT-5823 still resulted in an increase in maxi-KCa current by 20 ± 7% (Fig. 3B, KT-5823). Similarly, a different inhibitor of cGK, Rp-8Br-PET-cGMP, added to pipette solution did not prevent the expected increase in maxi-KCa current with EGF (Fig. 3B, Rp-8Br-PET-cGMP). We interpreted these combined findings as indicating that cGK was unlikely to mediate the increase in maxi-KCa current induced by EGFR activation.
To assess for potential involvement of cAK, we first confirmed that addition of the membrane-permeant activator of cAK 8-Br-cAMP would increase maxi-KCa current. Addition of 100 μm 8-Br-cAMP caused an increase in current of 22.5 ± 4% (Fig. 3C, 8Br-cAMP). Higher concentrations of 8-Br-cAMP (500 μm) did not further increased maxi-KCa current (6 cells, not shown). The magnitude of effect observed with 8-Br-cAMP was not significantly different from that observed with EGF (by ANOVA). In cells exposed to 8-Br-cAMP, subsequent addition of EGF (100 ng ml−1) 5–7 min later resulted in no further increase in maxi-KCa current (6 cells; not shown). We next evaluated the response to EGF in the presence of the cAK inhibitors KT-5720 added to the bath solution, or Rp-cAMP added to pipette solution. Neither of these compounds appreciably affected baseline current, and both compounds completely prevented any increase in current expected with subsequent addition of EGF (Fig. 3C, KT-5720, Rp-cAMP). Together, these data provided strong evidence that cAK was involved in the increase in maxi-KCa current induced by EGFR activation.
Involvement of AC-5
Given that our data pointed to involvement of cAK in the EGF-induced activation of maxi-KCa channels, we sought to determine whether adenylate cyclase (AC) might be involved. A previous study using an expression system reported that AC type 5 (AC-5) is required for EGF-induced production of cAMP (Chen et al. 1995), and so our efforts focused on this isozyme.
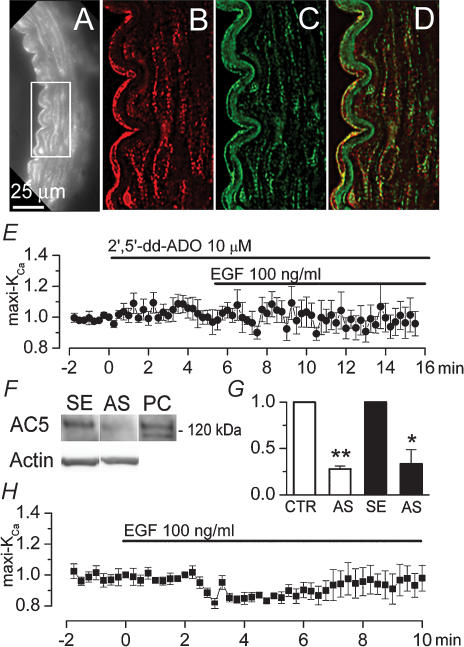
First, we sought to confirm that AC-5 is expressed in rat basilar artery VSMC. Immunolabelling experiments showed that AC-5 was abundantly expressed in both endothelial and VSMC layers (Fig. 4A and B). Labelling for AC-5 was punctate, and often appeared to be aligned with plasmalemmal membranes (Fig. 4B). Coimmunolabelling for caveolin-1 confirmed localization of AC-5 to the plasmalemmal membrane, and showed that AC-5 was often colocalized with caveolin-1 itself in both endothelium and VSMC (Fig. 4C and D) (Ostrom et al. 2002).
Figure 4. Adenylate cyclase 5 (AC-5) mediates maxi-KCa channel activation by EGFR.
A–D, fluorescence image of rat basilar artery immunolabelled for AC-5 (A and B) and coimmunolabelled for caveolin-1 (C); superimposed images of (B) and (C) demonstrate colocalization of AC-5 and caveolin-1 on plasmalemmal membranes of endothelial and smooth muscle cells (D); note autofluorescence of basal lamina at wavelengths for FITC filter (C), but not at wavelengths for CY3 filter (A and B). E, normalized change (mean ± s.e.m.) in membrane current measured using our standard conditions, after addition of EGF (100 ng ml−1) in the presence of 2′,5′-dd-ADO (10 μm) in the bath; 5 cells. F and G, Western blots (F) and bar graphs of densitometric analysis of Western blots (G) for AC-5 in basilar artery lysates from controls (CTR and SE) and from AC-5 knock-down (AS) rats; for AC 5-knock-down, rats underwent constant infusion of antisence (AS) oligodeoxynucleotide directed against AC-5 into the cisterna magna for 4 days (2 groups of 3 rats each); for controls, we used both normal, untreated rats (CTR; 3 rats), and rats with 4-day infusion into cisterna magna of corresponding sense (SE) oligodeoxynucleotide (3 rats); bar graphs show values normalized to corresponding controls, after first normalizing data from individual lanes to density of total actin; *P < 0.05; **P < 0.01; panel F also shows blots for total actin and a positive control (PC) for AC-5 (5 ng rat recombinant AC-5 obtained from FabGennix, Frisco TX). H, normalized change (mean ± s.e.m.) in membrane current in basilar artery VSMC from AC-5 knock-down rats, measured using our standard conditions, after addition of EGF (100 ng ml−1); 7 cells.
To provide an initial assessment for involvement of AC, we used 2′,5′-dideoxyadenosine (2′,5′-dd-Ado), a blocker with relative specificity for type 5 over types 2 and 3 (Iwatsubo et al. 2004). After 2′,5′-dd-Ado had been added to the bath, exposure of the cells to EGF resulted in no change in maxi-KCa current (Fig. 4E, filled circles).
To further assess involvement of AC-5, we developed an AC-5 knock-down model in which AS-ODN directed against AC-5 was infused into the cisterna magna. Western blots showed that basilar arteries from AC-5 knock-down animals exhibited significantly less AC-5 than arteries from controls (Fig. 4F and G). Patch clamp study of VSMC isolated from AC-5 knock-down animals was carried out using the same conditions as above. Maxi-KCa currents were normal in terms of magnitude, kinetics, voltage dependence and block by pharmacological agents. However, in cells from AC-5 knock-down animals, exposure to EGF resulted in no increase in maxi-KCa currents (Fig. 4H, filled squares).
PCNA up-regulation
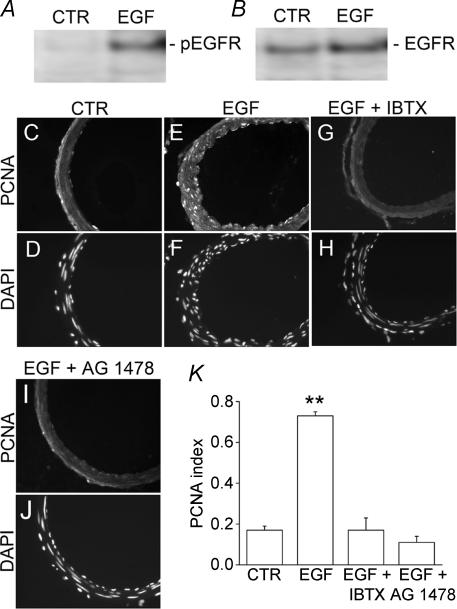
EGFR activation is expected to induce a proliferative response in VSMC, but this effect has only been demonstrated in synthetic phenotype VSMC, not in contractile phenotype VSMC. To assess the effect of EGFR activation on contractile VSMC, we applied EGF directly into cisterna magna, using mini-osmotic pumps to deliver a constant infusion for 1 day or for 3 days. Infusions of aCSF were used as controls. In these experiments, we confirmed that EGFR in basilar artery was being activated by performing Western blots for phospho-EGFR, a marker of EGFR activation. Arteries exposed to aCSF, both without and with EGF, exhibited similar levels of EGFR (Fig. 5B), but arteries exposed to EGF showed a clear increase in phosphorylation of the receptor, compared to controls (Fig. 5A), confirming that EGF infusion had resulted in EGFR activation.
Figure 5. Maxi-KCa channel is required for EGFR-mediated up-regulation of proliferating cell nuclear antigen (PCNA).
A and B, Western blots of basilar arteries exposed to aCSF without (CTR) or with EGF in vivo, blotted for phospho-EGFR (A) and reblotted for EGFR (B); aCSF without or with EGF (10 μg ml−1) was infused into cisterna magna at 1 μl h−1 for 24 h via mini-osmotic pump. C, E, G, I, fluorescence images of rat basilar artery sections immunolabelled for PCNA for a control rat (C), for a rat with cisterna magna infusion of EGF (E), for a rat with cisterna magna infusion of EGF plus iberiotoxin (IBTX) (G), for a rat with cisterna magna infusion of EGF plus AG-1478 (I); all infusions were for 3 days. D, F, H, J, fluorescence images of rat basilar artery corresponding to sections shown above, labelled with diamidino-2-phenylindole (DAPI) to show nuclei. K, bar graph of the PCNA index (number of nuclei with PCNA labelling divided by number of nuclei in the tissue section) for the four conditions, control, cisterna magna infusion of EGF, cisterna magna infusion of EGF plus iberiotoxin, cisterna magna infusion of EGF plus AG-1478; **P < 0.01; 3–4 arteries for each condition.
To assess for a proliferative response, we immunolabelled arteries for PCNA, up-regulation of which denotes a proliferative response in VSMC. Infusion of EGF for 1 day (not shown) or 3 days (Fig. 5E) resulted in a clear increase in nuclear labelling for PCNA, especially in VSMC layers, compared to controls (Fig. 5C). In addition, arteries exposed to EGF for 3 days appeared more corrugated, with a thicker arterial wall (Fig. 5CversusE). Both effects of EGF, i.e. PCNA up-regulation and apparent vasoconstriction, were completely prevented by coinfusion of iberiotoxin (Fig. 5G) or of AG-1478 (Fig. 5I). PCNA data from these and other similarly treated animals were quantified by computing a proliferation or PCNA index (number of nuclei with PCNA labelling divided by number of nuclei in the tissue section). Exposure to EGF resulted in a significant (by ANOVA, P < 0.01) increase in the PCNA index that was completely prevented by both iberiotoxin and by AG-1478 (Fig. 5K).
Discussion
The principal finding of the present study is that maxi-KCa channels are critically involved in growth-response signalling related to EGFR activation in native contractile VSMC in vivo. This finding reaffirms the widely recognized importance of K+ channel activation in growth factor signalling and cellular proliferation. A critical role for K+ channels and cellular hyperpolarization has been demonstrated in numerous studies on different cellular systems, with a surprising variety of channels and molecular mechanisms implicated. In VSMC alone, it appears that this critical step is carried out by two completely different mechanisms, depending upon the phenotype involved: in synthetic phenotype VSMC, EGFR tyrosine kinase phosphorylates int-KCa channels directly (Kohler et al. 2003), whereas in contractile phenotype VSMC, EGFR tyrosine kinase appears to act indirectly via AC-5 and cAK to cause phosphorylation of maxi-KCa channels. Since growth-response signalling in contractile VSMC has not been studied extensively, it remains to be determined whether activation of other growth-related genes or of other EGFR-induced signalling events also requires K+ channel activation. Regardless, our data indicate that maxi-KCa channels are both necessary and sufficient for EGFR-mediated activation of PCNA in vivo.
The signalling pathway that we identified in EGFR-mediated hyperpolarization in contractile VSMC, specifically the critical roles of AC-5 and of cAK, is similar to the pathway reported in heart. In cardiac cells, EGF causes activation of cAK, resulting in positive chronotropic and ionotropic effects (Nair et al. 1993). The mechanism involved includes EGFR-mediated tyrosine phosphorylation of GSα, resulting in activation of AC-5 and formation of cAMP (Nair et al. 1990; Poppleton et al. 1996). Although we did not explicitly study EGFR-mediated tyrosine phosphorylation of GSα in contractile VSMC, it seems likely that this would be the mechanism by which AC-5 becomes activated.
EGF does not increase cAMP accumulation in all tissues. EGF increases AC activity and elevates cAMP concentration only in cells expressing AC-5, not in cells overexpressing types 1, 2 and 6 isozymes (Chen et al. 1995). Of the 10 different mammalian isoforms of AC known, seven (AC-2–7 and AC-9) are expressed in smooth muscle cells, with types 3, 5 and 6 being particularly prominent (Webb et al. 2001; Ostrom et al. 2002). In the experiments reported here, we used immunochemistry, Western blots as well as knock-down experiments to confirm that contractile VSMC from rat basilar artery express AC-5, and that this isozyme is critically involved in growth-response signalling with EGFR. Our experiments are the first to specifically identify a distinct physiological function for AC-5 in VSMC.
Our results showing that EGF causes activation of AC-5, cAK and maxi-KCa channels may appear to be at odds with reports that EGF also acts as a potent vasoconstrictor (Florian & Watts, 1999). Whereas cAK and maxi-KCa channel activation are generally associated with vasodilatory responses, EGF causes modest but sustained contraction of rabbit (deBlois et al. 1992) and rat (Berk et al. 1985; Florian & Watts, 1999) aorta, and potentiates myogenic tone of mouse mesenteric arterioles (Lucchesi et al. 2004), with vasoconstrictive effects being significantly reduced by the EGFR inhibitor, AG-1478 (Lucchesi et al. 2004). Vasoconstriction is typically associated with an increase in intracellular Ca2+, a known consequence of EGF stimulation (Macara, 1986; Magni et al. 1991). EGF-induced Ca2+ influx may not be due to voltage-dependent mechanisms, but instead, to the voltage-independent non-selective cation channels, transient receptor potential (TRP) channels (Bezzerides et al. 2004). Notably, the recording protocols we used, specifically leak subtraction, would have negated any current due to a non-selective cation channel. In so far as EGFR signalling involves activation of both maxi-KCa channels and non-selective cation channels, it appears to constitute an example of ‘dissociation’ between vascular tone and membrane potential. Although we did not study Ca2+ influx or vasoconstriction specifically, our histological data showed a greater degree of corrugation and wall thickening in arteries exposed to cisterna magna infusion of EGF in vivo, consistent with a constrictive effect (Ono et al. 1997). However, additional study would be required to fully characterize constrictive effects of EGFR on basilar artery, as well as potential involvement of TRP channels.
Our results showing a critical role for AC-5 and for cAK in the proliferative response to EGFR activation may also seem paradoxical, given the extensive body of literature indicating that activation of cAK may be antiproliferative and cause G1 phase arrest of VSMC (Koyama et al. 2001; Thorin-Trescases et al. 2001). A plausible explanation for this apparent discrepancy would be that the effects that we observed were mediated by an AC-5/cAK system that is compartmentalized to the membrane and thereby affects only local phosphorylation of maxi-KCa channels, without broader involvement of cytoplasmic cAK. Support for this hypothesis comes from our experiments showing that effects of EGF were the same whether cells were studied using a nystatin-perforated patch technique to preserve intracellular contents, or with a whole cell technique in which cytoplasmic constituents are lost. Also, our immunolabelling experiments indicated that AC-5 was concentrated in plasmalemmal membranes, where it colocalized with caveolin-1, in accord with reports that AC-5 is a transmembrane protein localized to caveolin-rich membrane fractions (Ostrom et al. 2002). However, additional experiments, e.g. Western blots to show that VASP is not serine/threonine phosphorylated following EGFR activation, and patch clamp experiments to demonstrate that all of the molecular machinery involved can be localized to isolated inside-out patches, would be helpful to advance this hypothesis.
Studies on cultured cells indicate that contractile phenotype VSMC express low numbers of high affinity EGFR, but upon modulation from the contractile to the synthetic phenotype, the expression of EGFR increases 10-fold (Saltis et al. 1995). We also observed a 10-fold increase in EGFR expression in native basilar artery VSMC from AHR compared to controls, even though VSMC from AHR had not transitioned into a synthetic phenotype, but remained in a contractile phenotype, as suggested by continued expression of maxi-KCa channels. Our data from controls, EGFR knock-down and EGFR gain-of-expression (AHR) indicated that the ‘transfer function’ between EGFR activation and maxi-KCa channel activation varied non-linearly through the observed range of EGFR expression (Fig. 2H). The system is biased so that in the normal contractile phenotype, a relatively strong input signal is required to generate a given response, whereas when sensitized by chronic administration of angiotensin II, a weaker input signal is sufficient to generate the same response. If EGFR activation itself promotes conversion from a contractile to a synthetic phenotype, this bias would appear to provide a strong positive feedback favouring conversion to a synthetic phenotype. It has been suggested that expression of int-KCa channels may promote excessive neointimal VSMC proliferation (Kohler et al. 2003). However, our data would indicate that the specific K+ channel involved may be less important than the number of EGFR expressed.
Our experiments also confirmed that EGF applied in situ induces a proliferative response in contractile VSMC, as shown by PCNA up-regulation. Although not surprising, documentation of this has heretofore not been available. In general, claims of effects of ligands on contractile phenotype VSMC, based on effects in culture (Li et al. 1999), may be subject to question. The fact that cerebral vessels are bathed in cerebrospinal fluid in the subarachnoid space, coupled with the presence of a rete vasorum that allows substances in the cerebrospinal fluid to readily access VSMC (Zervas et al. 1982), provides a unique opportunity to expose contractile VSMC to a variety of agents in situ. For our experiments, we used direct infusions of ligand into cisterna magna to ensure effects on native contractile phenotype VSMC. Similarly, we used direct infusions of ODN into cisterna magna to selectively knock-down expression of molecular targets in VSMC, specifically EGFR and AC-5. Our experience with these techniques indicates that a diffusion barrier for ODN exists only at the level of the basal lamina, thereby allowing selective knock-down of selected molecular targets in VSMC of the basilar artery, with complete sparing of endothelium.
In summary, here we report that EGF and associated ligands, TGFα and HB-EGF, activated EGFR, resulting in activation of AC-5, cAK and maxi-KCa channels in native contractile VSMC from basilar artery. Also, we found that this signalling sequence was crucial for in vivo EGFR-mediated expression of PCNA, which itself is critical for gene activation in the programme of VSMC proliferation (Lee et al. 1999; Paunesku et al. 2001). Identification of the critical role of AC-5 suggests that therapeutic targeting of this molecule may be useful in preventing proliferative vasculopathies such as atherosclerosis and restenosis.
Acknowledgments
We thank Yohannes Adall for expert technical assistance. This work was supported by grants from the National Heart Lung and Blood Institute (HL51932) (J.M.S.), the American Heart Association (V.G.) and the National Research and Education Foundation of the American Association of Neurological Surgeons (R.DH.).
References
- Berk BC, Brock TA, Webb RC, Taubman MB, Atkinson WJ, Gimbrone MA, Jr, Alexander RW. Epidermal growth factor, a vascular smooth muscle mitogen, induces rat aortic contraction. J Clin Invest. 1985;75:1083–1086. doi: 10.1172/JCI111772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bezzerides VJ, Ramsey IS, Kotecha S, Greka A, Clapham DE. Rapid vesicular translocation and insertion of TRP channels. Nat Cell Biol. 2004;6:709–720. doi: 10.1038/ncb1150. [DOI] [PubMed] [Google Scholar]
- deBlois D, Drapeau G, Petitclerc E, Marceau F. Synergism between the contractile effect of epidermal growth factor and that of des-Arg9-bradykinin or of alpha-thrombin in rabbit aortic rings. Br J Pharmacol. 1992;105:959–967. doi: 10.1111/j.1476-5381.1992.tb09085.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z, Nield HS, Sun H, Barbier A, Patel TB. Expression of type V adenylyl cyclase is required for epidermal growth factor-mediated stimulation of cAMP accumulation. J Biol Chem. 1995;270:27525–27530. doi: 10.1074/jbc.270.46.27525. [DOI] [PubMed] [Google Scholar]
- Cress AE. Quantitation of phosphotyrosine signals in human prostate cell adhesion sites. Biotechniques. 2000;29:776–781. doi: 10.2144/00294st03. [DOI] [PubMed] [Google Scholar]
- Dubois JM, Rouzaire-Dubois B. Role of potassium channels in mitogenesis. Prog Biophys Mol Biol. 1993;59:1–21. doi: 10.1016/0079-6107(93)90005-5. [DOI] [PubMed] [Google Scholar]
- Florian JA, Dorrance A, Webb RC, Watts SW. Mineralocorticoids upregulate arterial contraction to epidermal growth factor. Am J Physiol Regul Integr Comp Physiol. 2001;281:R878–R886. doi: 10.1152/ajpregu.2001.281.3.R878. [DOI] [PubMed] [Google Scholar]
- Florian JA, Watts SW. Epidermal growth factor: a potent vasoconstrictor in experimental hypertension. Am J Physiol. 1999;276:H976–H983. doi: 10.1152/ajpheart.1999.276.3.H976. [DOI] [PubMed] [Google Scholar]
- Gerzanich V, Ivanov A, Ivanova S, Yang JB, Zhou H, Dong Y, Simard JM. Alternative splicing of cGMP-dependent protein kinase I in angiotensin-hypertension: novel mechanism for nitrate tolerance in vascular smooth muscle. Circ Res. 2003a;93:805–812. doi: 10.1161/01.RES.0000097872.69043.A0. [DOI] [PubMed] [Google Scholar]
- Gerzanich V, Ivanova S, van der Heijden MS, Zhou H, Simard JM. Trans-cellular proliferating cell nuclear antigen gene activation in cerebral vascular smooth muscle by endothelial oxidative injury in vivo. Arterioscler Thromb Vasc Biol. 2003b;23:2048–2054. doi: 10.1161/01.ATV.0000094962.07514.BC. [DOI] [PubMed] [Google Scholar]
- Gerzanich V, Ivanova S, Zhou H, Simard JM. Mislocalization of eNOS and upregulation of cerebral vascular Ca2+ channel activity in angiotensin-hypertension. Hypertension. 2003c;41:1124–1130. doi: 10.1161/01.HYP.0000066288.20169.21. [DOI] [PubMed] [Google Scholar]
- Holland M, Langton PD, Standen NB, Boyle JP. Effects of the BKCa channel activator, NS1619, on rat cerebral artery smooth muscle. Br J Pharmacol. 1996;117:119–129. doi: 10.1111/j.1476-5381.1996.tb15163.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwatsubo K, Minamisawa S, Tsunematsu T, Nakagome M, Toya Y, Tomlinson JE, Umemura S, Scarborough RM, Levy DE, Ishikawa Y. Direct inhibition of type 5 adenylyl cyclase prevents myocardial apoptosis without functional deterioration. J Biol Chem. 2004;279:40938–40945. doi: 10.1074/jbc.M314238200. [DOI] [PubMed] [Google Scholar]
- Kohler R, Wulff H, Eichler I, Kneifel M, Neumann D, Knorr A, Grgic I, Kampfe D, Si H, Wibawa J, Real R, Borner K, Brakemeier S, Orzechowski HD, Reusch HP, Paul M, Chandy KG, Hoyer J. Blockade of the intermediate-conductance calcium-activated potassium channel as a new therapeutic strategy for restenosis. Circulation. 2003;108:1119–1125. doi: 10.1161/01.CIR.0000086464.04719.DD. [DOI] [PubMed] [Google Scholar]
- Koyama H, Bornfeldt KE, Fukumoto S, Nishizawa Y. Molecular pathways of cyclic nucleotide-induced inhibition of arterial smooth muscle cell proliferation. J Cell Physiol. 2001;186:1–10. doi: 10.1002/1097-4652(200101)186:1<1::AID-JCP1012>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- Lazar LM, Blum M. Regional distribution and developmental expression of epidermal growth factor and transforming growth factor-alpha mRNA in mouse brain by a quantitative nuclease protection assay. J Neurosci. 1992;12:1688–1697. doi: 10.1523/JNEUROSCI.12-05-01688.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee M, Simon AD, Stein CA, Rabbani LE. Antisense strategies to inhibit restenosis. Antisense Nucl Acid Drug Dev. 1999;9:487–492. doi: 10.1089/oli.1.1999.9.487. [DOI] [PubMed] [Google Scholar]
- Levitzki A, Gazit A. Tyrosine kinase inhibition: an approach to drug development. Science. 1995;267:1782–1788. doi: 10.1126/science.7892601. [DOI] [PubMed] [Google Scholar]
- Li S, Sims S, Jiao Y, Chow LH, Pickering JG. Evidence from a novel human cell clone that adult vascular smooth muscle cells can convert reversibly between noncontractile and contractile phenotypes. Circ Res. 1999;85:338–348. doi: 10.1161/01.res.85.4.338. [DOI] [PubMed] [Google Scholar]
- Lovisolo D, Bonelli G, Baccino FM, Peres A, Alonzo F, Munaron L. Two currents activated by epidermal growth factor in EGFR-T17 fibroblasts. Biochim Biophys Acta. 1992;1104:73–82. doi: 10.1016/0005-2736(92)90133-7. [DOI] [PubMed] [Google Scholar]
- Lucchesi PA, Sabri A, Belmadani S, Matrougui K. Involvement of metalloproteinases 2/9 in epidermal growth factor receptor transactivation in pressure-induced myogenic tone in mouse mesenteric resistance arteries. Circulation. 2004;110:3587–3593. doi: 10.1161/01.CIR.0000148780.36121.47. [DOI] [PubMed] [Google Scholar]
- Macara IG. Activation of 45Ca2+ influx and 22Na+/H+ exchange by epidermal growth factor and vanadate in A431 cells is independent of phosphatidylinositol turnover and is inhibited by phorbol ester and diacylglycerol. J Biol Chem. 1986;261:9321–9327. [PubMed] [Google Scholar]
- Magni M, Meldolesi J, Pandiella A. Ionic events induced by epidermal growth factor. Evidence that hyperpolarization and stimulated cation influx play a role in the stimulation of cell growth. J Biol Chem. 1991;266:6329–6335. [PubMed] [Google Scholar]
- Nair BG, Parikh B, Milligan G, Patel TB. Gs alpha mediates epidermal growth factor-elicited stimulation of rat cardiac adenylate cyclase. J Biol Chem. 1990;265:21317–21322. [PubMed] [Google Scholar]
- Nair BG, Rashed HM, Patel TB. Epidermal growth factor produces inotropic and chronotropic effects in rat hearts by increasing cyclic AMP accumulation. Growth Factors. 1993;8:41–48. doi: 10.3109/08977199309029133. [DOI] [PubMed] [Google Scholar]
- Naraghi M, Neher E. Linearized buffered Ca2+ diffusion in microdomains and its implications for calculation of [Ca2+] at the mouth of a calcium channel. J Neurosci. 1997;17:6961–6973. doi: 10.1523/JNEUROSCI.17-18-06961.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neylon CB. Potassium channels and vascular proliferation. Vascul Pharmacol. 2002;38:35–41. doi: 10.1016/s1537-1891(02)00124-6. [DOI] [PubMed] [Google Scholar]
- Neylon CB, Lang RJ, Fu Y, Bobik A, Reinhart PH. Molecular cloning and characterization of the intermediate-conductance Ca2+-activated K+ channel in vascular smooth muscle: relationship between K(Ca) channel diversity and smooth muscle cell function. Circ Res. 1999;85:e33–e43. doi: 10.1161/01.res.85.9.e33. [DOI] [PubMed] [Google Scholar]
- Ono S, Date I, Nakajima M, Onoda K, Ogihara K, Shiota T, Asari S, Ninomiya Y, Yabuno N, Ohmoto T. Three-dimensional analysis of vasospastic major cerebral arteries in rats with the corrosion cast technique. Stroke. 1997;28:1631–1637. doi: 10.1161/01.str.28.8.1631. [DOI] [PubMed] [Google Scholar]
- Ostrom RS, Liu X, Head BP, Gregorian C, Seasholtz TM, Insel PA. Localization of adenylyl cyclase isoforms and G protein-coupled receptors in vascular smooth muscle cells: expression in caveolin-rich and noncaveolin domains. Mol Pharmacol. 2002;62:983–992. doi: 10.1124/mol.62.5.983. [DOI] [PubMed] [Google Scholar]
- Pandiella A, Magni M, Lovisolo D, Meldolesi J. The effect of epidermal growth factor on membrane potential. Rapid hyperpolarization followed by persistent fluctuations. J Biol Chem. 1989;264:12914–12921. [PubMed] [Google Scholar]
- Paunesku T, Mittal S, Protic M, Oryhon J, Korolev SV, Joachimiak A, Woloschak GE. Proliferating cell nuclear antigen (PCNA): ringmaster of the genome. Int J Radiat Biol. 2001;77:1007–1021. doi: 10.1080/09553000110069335. [DOI] [PubMed] [Google Scholar]
- Poppleton H, Sun H, Fulgham D, Bertics P, Patel TB. Activation of Gsalpha by the epidermal growth factor receptor involves phosphorylation. J Biol Chem. 1996;271:6947–6951. doi: 10.1074/jbc.271.12.6947. [DOI] [PubMed] [Google Scholar]
- Roderick C, Reinach PS, Wang L, Lu L. Modulation of rabbit corneal epithelial cell proliferation by growth factor-regulated K+ channel activity. J Membr Biol. 2003;196:41–50. doi: 10.1007/s00232-003-0623-1. [DOI] [PubMed] [Google Scholar]
- Saltis J, Thomas AC, Agrotis A, Campbell JH, Campbell GR, Bobik A. Expression of growth factor receptors in arterial smooth muscle cells. Dependency on cell phenotype and serum factors. Atherosclerosis. 1995;118:77–87. doi: 10.1016/0021-9150(95)05595-n. [DOI] [PubMed] [Google Scholar]
- Thorin-Trescases N, Orlov SN, Taurin S, Dulin NO, Allen BG, deBlois D, Tremblay J, Pshezhetsky AV, Hamet P. Antiproliferative effect of brief exposure to cholera toxin in vascular smooth muscle cells: role of cAMP and protein kinase A. Can J Physiol Pharmacol. 2001;79:471–480. [PubMed] [Google Scholar]
- Tsien RY. New calcium indicators and buffers with high selectivity against magnesium and protons: design, synthesis, and properties of prototype structures. Biochemistry. 1980;19:2396–2404. doi: 10.1021/bi00552a018. [DOI] [PubMed] [Google Scholar]
- Wang L, Xu B, White RE, Lu L. Growth factor-mediated K+ channel activity associated with human myeloblastic ML-1 cell proliferation. Am J Physiol. 1997;273:C1657–C1665. doi: 10.1152/ajpcell.1997.273.5.C1657. [DOI] [PubMed] [Google Scholar]
- Webb JG, Yates PW, Yang Q, Mukhin YV, Lanier SM. Adenylyl cyclase isoforms and signal integration in models of vascular smooth muscle cells. Am J Physiol Heart Circ Physiol. 2001;281:H1545–H1552. doi: 10.1152/ajpheart.2001.281.4.H1545. [DOI] [PubMed] [Google Scholar]
- Wonderlin WF, Strobl JS. Potassium channels, proliferation and G1 progression. J Membr Biol. 1996;154:91–107. doi: 10.1007/s002329900135. [DOI] [PubMed] [Google Scholar]
- Xu D, Wang L, Dai W, Lu L. A requirement for K+-channel activity in growth factor-mediated extracellular signal-regulated kinase activation in human myeloblastic leukemia ML-1 cells. Blood. 1999;94:139–145. [PubMed] [Google Scholar]
- Zervas NT, Liszczak TM, Mayberg MR, Black PM. Cerebrospinal fluid may nourish cerebral vessels through pathways in the adventitia that may be analogous to systemic vasa vasorum. J Neurosurg. 1982;56:475–481. doi: 10.3171/jns.1982.56.4.0475. [DOI] [PubMed] [Google Scholar]