Abstract
Nine healthy endurance-trained males were recruited to examine the effect of a dual dopamine/noradrenaline reuptake inhibitor on performance, thermoregulation and the hormonal responses to exercise. Subjects performed four trials, ingesting either a placebo (pla) or 2 × 300 mg bupropion (bup), prior to exercise in temperate (18°C) or warm (30°C) conditions. Trials consisted of 60 min cycle exercise at 55% Wmax immediately followed by a time trial (TT). TT performance in the heat was significantly improved by bupropion (pla: 39.8 ± 3.9 min, bup: 36.4 ± 5.7 min; P = 0.046), but no difference between treatments was apparent in temperate conditions (pla: 30.6 ± 2.2 min, bup: 30.6 ± 1.9 min; P = 0.954). While TT power output was consistently lower in the heat when compared to temperate conditions, this decrement was attenuated by bupropion. At the end of the TT in the heat, both core temperature (pla 39.7 ± 0.3°C, bup 40.0 ± 0.3°C; P = 0.017) and HR (pla 178 ± 7 beats min−1, bup 183 ± 12 beats min−1; P = 0.039), were higher in the bupropion trial than in the placebo. Circulating pituitary and adrenal hormone concentrations increased throughout exercise in all trials. Circulating serum prolactin was elevated above temperate levels during exercise in a warm environment (P < 0.001). These data indicate that performance in warm conditions is enhanced by acute administration of a dual dopamine/noradrenaline reuptake inhibitor. No such effect was apparent under temperate conditions. It appears that bupropion enabled subjects to maintain a greater TT power output in the heat with the same perception of effort and thermal stress reported during the placebo trial, despite the attainment of a higher core temperature.
Capacity to perform prolonged exercise is clearly impaired in high ambient temperatures (Galloway & Maughan, 1997; Parkin et al. 1999; Tatterson et al. 2000). While fatigue during prolonged exercise in temperate conditions is typically associated with the depletion of muscle glycogen, accumulation of metabolites, inadequate oxygen delivery and the development of hypohydration, factors contributing to fatigue when exercising in a warm environment are not clear at present. While exercise capacity is thought to be primarily limited by thermoregulatory and fluid balance factors (Hargreaves & Febbraio, 1998), it has been suggested that the central nervous system (CNS) may become important in the development of fatigue when body temperature is significantly elevated (Bruck & Olschewski, 1987; Nielsen, 1992). During prolonged exercise in the heat, exhaustion appears to coincide with the attainment of an internal body temperature of around 40.0°C (Nielsen et al. 1993; Gonzalez-Alonso et al. 1999). Hyperthermia has been proposed to accelerate the development of central fatigue during exercise, resulting in a reduction in maximal muscle activation (Nybo & Nielsen, 2001a), altered EEG brain activity (Nielsen et al. 2001) and increased perceived exertion (Nybo & Nielsen, 2001b), but the neurobiological mechanisms for these responses are not clear. Recently it has been suggested that exercise performance in the heat is governed through an anticipatory response that reduces skeletal muscle recruitment, thus limiting the rate of heat production (Marino, 2004; Marino et al. 2004; Tucker et al. 2004). A feed-forward regulation of power output may insure that the rate of body heat accumulation is restricted to prevent the development of heat illness.
Dopamine (DA) and noradrenaline (NA) have been implicated in arousal, motivation, reinforcement and reward, the control of motor behaviour and mechanisms of addiction (Nestler et al. 2001). The depletion of central catecholamines has been linked to CNS fatigue by a number of research groups (Owasoyo et al. 1992; Chaouloff, 1989; Davis, 2000). A series of animal studies conducted by Bailey and coworkers (Davis & Bailey, 1997) demonstrated that brain serotonin (5-HT) and DA activity were elevated during exercise, but at the point of exhaustion a marked fall in tissue DA content was apparent. This observation resulted in the suggestion that the ratio of 5-HT to DA activity may be important for the development of central fatigue, with a high ratio of 5-HT to DA associated with feelings of tiredness and reduced motivation (Davis & Bailey, 1997). Additionally, intracranial stimulation of the ventral tegmental area, an origin of the dopaminergic projections within the CNS, motivated rats to run without the need for aversive electric shocks (Burgess et al. 1991).
While there is good evidence that amphetamine, a potent DA releaser, reuptake inhibitor, and monoamine oxidase inhibitor, can enhance capacity to perform exercise (Borg et al. 1972; Gerald, 1978; Chandler & Blair, 1980), little evidence from other strategies to manipulate catecholaminergic neurotransmission supports a performance benefit (Meeusen et al. 1997; Struder et al. 1998; Chinevere et al. 2002; Piacentini et al. 2004). In particular, Piacentini et al. (2004) recently examined the effects of bupropion, a dual DA/NA reuptake inhibitor, on performance and hormonal responses to exercise in temperate conditions. Although bupropion failed to alter exercise performance when compared to a placebo treatment, a marked difference in the hormonal response to exercise was reported. As pituitary hormone release is largely governed through changes in hypothalamic neurotransmitter release (Checkley, 1980), this suggests that the treatment altered neurotransmission, but that this change was not sufficient to influence the development of fatigue under these conditions.
As the reduction in the capacity to perform prolonged exercise in a warm environment is thought to be mediated, in part, by a reduction in drive and motivation to continue exercise resulting from the development of whole-body hyperthermia (Bruck & Olschewski, 1987; Nielsen, 1992), the maintenance of central catecholaminergic neurotransmission may attenuate the loss of performance apparent when exercise is undertaken under conditions of heat stress. Additionally, dopamine has also been implicated in the control of body temperature (Hasegawa et al. 2000) and tolerance to exercise in the heat (Bridge et al. 2003). The aim of the present study was to investigate the influence of an acute dose of bupropion, a dual dopaminergic and noradrenergic reuptake inhibitor, on performance, thermoregulation and the hormonal responses to exercise in temperate and warm environmental conditions.
Methods
Subjects
Nine healthy males (age 22.7 ± 4.3 years; height 1.83 ± 0.07 m; mass 75.2 ± 6.7 kg; maximal workload, Wmax 372 ± 36 W; peak oxygen uptake, 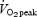 5.1 ± 0.5 l min−1) participated in this investigation. All subjects were well-trained cyclists or triathletes, but were not accustomed to exercise in a warm environment at the time of the study. Prior to the start of the study all volunteers received written information regarding the nature and purpose of the experimental protocol. Following an opportunity to ask any questions, a written statement of consent was signed. The protocol employed was approved by the Research Council of the Vrije Universiteit Brussel, Belgium.
5.1 ± 0.5 l min−1) participated in this investigation. All subjects were well-trained cyclists or triathletes, but were not accustomed to exercise in a warm environment at the time of the study. Prior to the start of the study all volunteers received written information regarding the nature and purpose of the experimental protocol. Following an opportunity to ask any questions, a written statement of consent was signed. The protocol employed was approved by the Research Council of the Vrije Universiteit Brussel, Belgium.
Experimental protocol
All subjects completed a preliminary maximal exercise test, a familiarization trial and four experimental trials. The preliminary trial consisted of continuous incremental cycle exercise to volitional exhaustion and was used to determine peak oxygen uptake 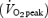 and the power output required to elicit 55% and 75% of maximal workload (Wmax). A familiarization trial was undertaken to ensure the subjects were accustomed to the procedures employed during the investigation and to minimize any potential learning or anxiety effects. This trial was performed in temperate environmental conditions with the placebo treatment administered in a single-blind manner, and was identical to the experimental trials in all respects. Experimental trials were undertaken in either temperate (18°C) or warm (30°C) conditions, with relative humidity maintained between 50 and 60% in both conditions. The experimental trials were separated by at least 7 days to minimize the development of heat acclimation and to ensure drug washout. Subjects were instructed to record dietary intake and physical activity during the two days before the first trial, and to replicate this in the two days prior to the subsequent experimental trials. No exercise or alcohol consumption was permitted in the 24 h before each trial.
and the power output required to elicit 55% and 75% of maximal workload (Wmax). A familiarization trial was undertaken to ensure the subjects were accustomed to the procedures employed during the investigation and to minimize any potential learning or anxiety effects. This trial was performed in temperate environmental conditions with the placebo treatment administered in a single-blind manner, and was identical to the experimental trials in all respects. Experimental trials were undertaken in either temperate (18°C) or warm (30°C) conditions, with relative humidity maintained between 50 and 60% in both conditions. The experimental trials were separated by at least 7 days to minimize the development of heat acclimation and to ensure drug washout. Subjects were instructed to record dietary intake and physical activity during the two days before the first trial, and to replicate this in the two days prior to the subsequent experimental trials. No exercise or alcohol consumption was permitted in the 24 h before each trial.
Subjects entered the laboratory in the morning approximately 90 min after consuming a standardized breakfast that included 500 ml of plain water. Nude post-void body mass was measured and an indwelling venous cannula was introduced into a superficial forearm vein to enable repeated blood sampling at rest and during exercise. Subjects inserted a rectal thermister (Gram Corporation LT-8 A, Saitama, Japan) 10 cm beyond the anal sphincter for the measurement of core temperature. Surface skin temperature probes (Gram Corporation LT-8 A, Saitama, Japan) were attached to four sites (chest, upper arm, thigh and calf) to enable the determination of weighted mean skin temperature (Ramanathan, 1964) and a heart rate telemetry band (Polar Accurex plus, Kempele, Finland) was positioned. Subjects were dressed in only cycling shorts, socks and shoes for all trials.
Subjects then entered a climatic chamber maintained at the appropriate environmental conditions and rested in a seated position for 15 min. During this period temperatures and heart rate were recorded at 5 min intervals and a resting venous blood sample was drawn immediately before the start of exercise. The exercise protocol consisted of 60 min constant load exercise at a workload corresponding to 55% Wmax, followed by a time trial (TT) to measure performance. There was a 1–2 min delay between the end of the constant load exercise and the beginning of the TT, to programme the ergometer (Lode Excalibur Sport, Groningen, Holland). The TT required the subjects to complete a predetermined amount of work equal to 30 min at 75% Wmax as quickly as possible (Jeukendrup et al. 1996). Subjects began the TT at a workload corresponding to 75% Wmax, but were free to increase or decrease their power output as desired from the outset. During the TT a computer program displayed a bar indicating the percentage of total work completed to give the subject an indication of their progress. Throughout the protocol no feedback was provided regarding time lapsed, power output, pedal cadence or heart rate. During exercise subjects had ad libitum access to plain water.
Core and skin temperatures and heart rate were recorded at 5 min intervals during exercise. Ratings of perceived exertion (Borg, 1982) and thermal stress (assessed using a 21-point scale ranging from unbearable cold to unbearable heat; adapted from Parsons, 2003) were assessed every 15 min during the initial 60 min and at 10 min intervals during the TT. Venous blood samples were drawn after 30 and 60 min of constant load exercise and at the end of the TT. Following the completion of the TT, subjects returned to a seated position where recovery was monitored for 15 min and a further blood sample was obtained. The probes and cannula were then removed and nude body mass was then re-measured to allow the estimation of sweat losses.
Drugs
Subjects ingested 600 mg bupropion (bup: ZybanTM., GSK, Middlesex, UK) or a placebo (pla: lactose) spread over two doses: one ingested at night immediately before sleeping, with the other taken upon waking on the morning of the trial (2 × 300 mg). This is equivalent to the maximum daily therapeutic dose administered during the treatment of depression or to assist in the cessation of cigarette smoking (Holm & Spencer, 2000), and has been demonstrated to alter the hormonal response to prolonged exercise (Piacentini et al. 2004). The treatment was randomized and administered in double-blind crossover manner. Drug and placebo capsules were prepared by an independent pharmacy to appear indistinguishable with regard to dimensions, weight and colour.
Blood collection and analysis
Venous blood samples were drawn directly into precooled vacutainer tubes (BD Vacutainer, Plymouth, UK). Samples of 10 ml were collected into a plain tubes and left to clot for 1 h at room temperature before centrifugation. The resulting serum was stored at −20°C for the determination of prolactin (Roche Diagnostics, Mannheim, Germany), cortisol (Diasorin, Stillwater, USA) and growth hormone (Pharmacia and Upjohn Diagnostics, Uppsala, Sweden). Samples for plasma adrenocorticotropic hormone (ACTH) and beta-endorphin (Nichols Institute Diagnostics, CA, USA) were collected into 4.5 ml tubes containing K3EDTA. An additional 7.5 ml was added to lithium heparin. A 0.5 ml aliquot of whole blood was extracted and used for the determination of haemoglobin and haematocrit; these were used to estimate percentage changes in plasma volume relative to the pre-exercise sample (Dill & Costill, 1974). Sodium metabisulphate (5 mg) was then added to the remaining whole blood prior to centrifugation. Plasma samples were stored frozen at −20°C ready for the determination of catecholamines by HPLC with electrical detection (Bio-Rad, Nazareth, Belgium).
Statistical analysis
Data are presented as means ± standard deviation (s.d.), unless otherwise stated. To evaluate differences in TT performance, two-factor (temperature × drug) repeated measures ANOVA was employed. Data collected over time were analysed using three-factor (temperature × drug × time) ANOVA with repeated measures. Pairwise differences were identified using Tukey's post hoc test as appropriate. Statistical significance was accepted at P < 0.05. To improve the clarity of figures, differences present between environmental conditions are described in the text.
Results
All subjects completed all trials. Some minor side-effects were reported (e.g. mild insomnia, headaches), but these were not consistent with bupropion and did not appear to influence performance in the exercise bout. Throughout the text the trials are referred to as: placebo trial at 18°C (pla18), bupropion trial at 18°C (bup18), placebo trial at 30°C (pla30) and the bupropion trial at 30°C (bup30).
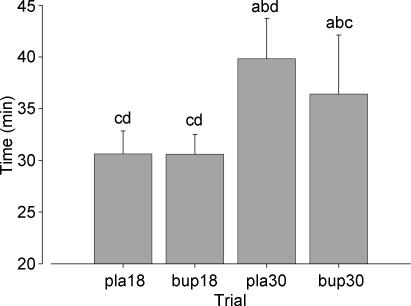
Exercise performance was significantly influenced by the ambient temperature, with subjects taking longer to complete the target amount of work in the warm than in the temperate trials (P < 0.001; Fig. 1). No difference in TT performance was apparent between treatments in the temperate condition, with times of 30.6 ± 2.2 min and 30.6 ± 1.9 min recorded in the placebo and bupropion trials, respectively (P = 0.954). When exercise was performed in a warm environment, subjects completed the TT 9% faster in the bupropion trial (36.4 ± 5.7 min) than in the placebo (39.8 ± 3.9 min; P = 0.046).
Figure 1. Time trial performance in the four experimental trials.
a, b, c, d denote a significant difference from corresponding values in pla18, bup18, pla30 and bup30 trials, respectively (P < 0.05). Values are mean ± SD.
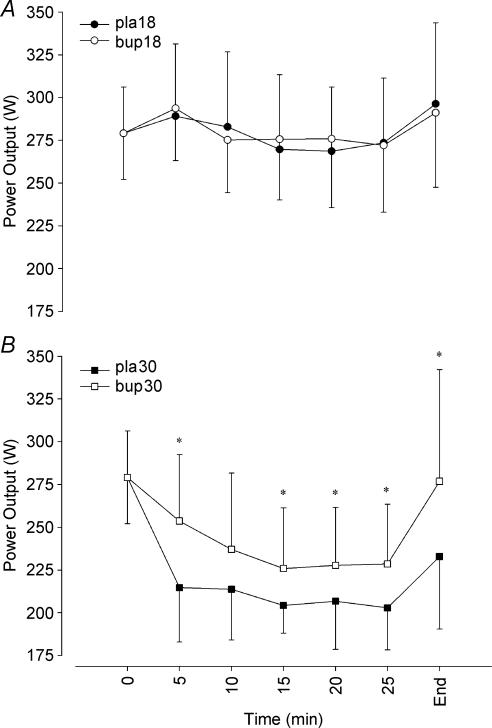
As the TT required the completion of a predetermined amount of work, the time taken to complete the protocol was directly related to the power output maintained throughout this period. TT power output was significantly higher during the temperate trials (P = 0.001; Fig. 2), with a similar workload maintained throughout the duration of the trial. Mean power output in temperate conditions was 275 ± 36 W and 279 ± 35 W in the placebo and bupropion trials, respectively (P = 0.531). Mean power output was greater throughout the bupropion trial in the warm (234 ± 35 W) than during the placebo trial in the same environmental conditions (211 ± 23 W). From the start of the TT in the heat, power output was reduced below the predetermined starting value, but the reduction was not as pronounced in the bupropion trial.
Figure 2. Time trial power output in temperate (A) and warm (B) conditions.
*Significant difference between the placebo trial and the corresponding time point on the bupropion trial (P < 0.05). Values are mean ± SD.
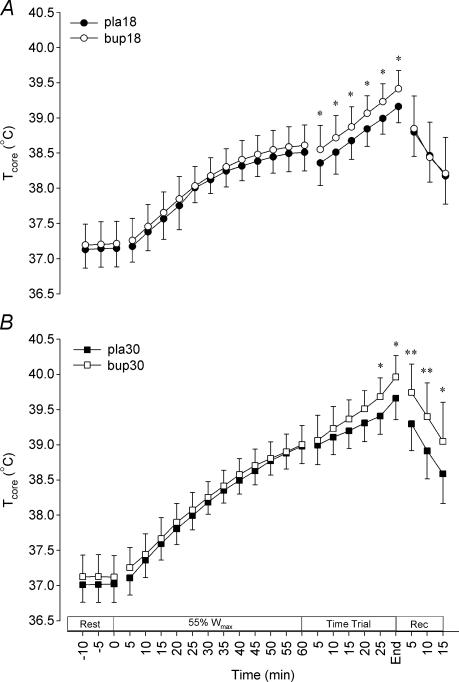
Core temperature (Tcore) at rest was not different between trials (P = 0.107; Fig. 3). Exercise produced a progressive increase in core temperature in all trials (P < 0.001). Ambient temperature influenced Tcore (P = 0.003), with values significantly elevated in the warm trials above those recorded in temperate conditions after 50 min of constant load exercise, with this difference maintained throughout the TT and recovery. There was no apparent effect of bupropion on Tcore during exercise at 55% Wmax, but core temperature was significantly higher on bupropion during the TT in both temperate and warm conditions. At the end of the performance test core temperature was elevated in bupropion trials in both temperate (pla18, 39.2 ± 0.2°C; bup18, 39.4 ± 0.3°C; P = 0.010) and warm (pla30, 39.7 ± 0.3°C; bup30, 40.0 ± 0.3°C; P = 0.017) environmental conditions. Weighted mean skin temperature increased rapidly from the onset of exercise, reaching a plateau after 20 min exercise in all trials. Skin temperature was higher throughout both trials at 30°C than during exercise in 18°C (P < 0.001), but was not influenced by bupropion (P = 0.386).
Figure 3. Core temperature responses to the experimental protocol in temperate (A) and warm (B) conditions.
*P < 0.05 and **P < 0.01, significant differences between the placebo trial and the corresponding time point on the bupropion trial. Values are mean ± SD.
Heart rate was higher in the warm when compared to values recorded during the corresponding trial in temperate conditions (P < 0.001). During the initial 60 min of exercise, heart rate was not affected by the drug treatment, but values were higher throughout the performance test with bupropion in the warm (bup30) compared to the same period in the placebo trial (pla30; P < 0.05). The change in body mass experienced during exercise, corrected for fluid intake, was significantly greater during trials in the warm (pla30: 3.03 ± 0.56 kg, bup30: 3.02 ± 0.54 kg) when compared to the temperate conditions (pla18: 2.10 ± 0.32 kg, bup18: 2.02 ± 0.43 kg; P < 0.001).
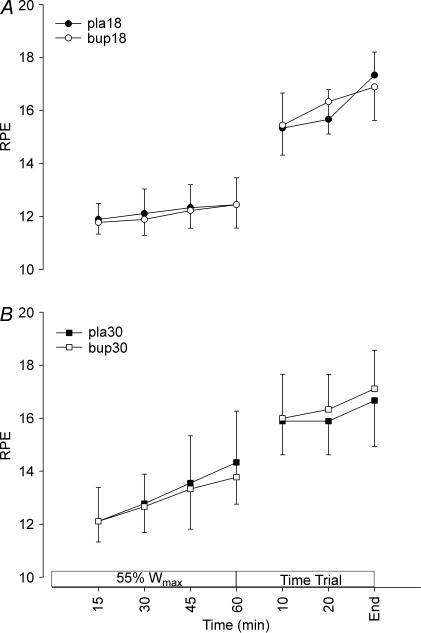
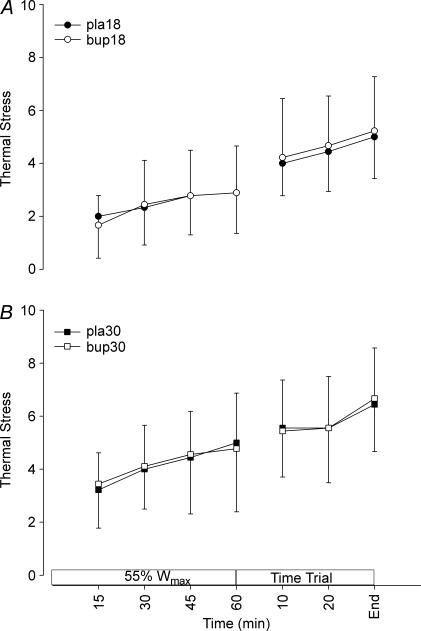
Ambient temperature did not influence subjects' rating of perceived exertion (RPE) during exercise, although there was a tendency for perceived exertion to be higher towards the end of the fixed-intensity exercise in 30°C (P = 0.072; Fig. 4). Bupropion had no effect on perceived exertion (P = 1.000). The subjects' rating of perceived thermal stress is shown in Fig. 5. Compared to the temperate trials, perceived thermal stress was significantly higher when exercise was performed in warm conditions (P < 0.001), but during the TT neither RPE nor perceived thermal stress was different between environmental conditions. No difference in the subjects' thermal stress was apparent between drug treatments in either temperate or warm conditions (P = 0.785).
Figure 4. Rating of perceived exertion during exercise in temperate (A) and warm (B) conditions.
Values are mean ± SD.
Figure 5. Perceived thermal stress during exercise in temperate (A) and warm (B) conditions.
Values are mean ± SD.
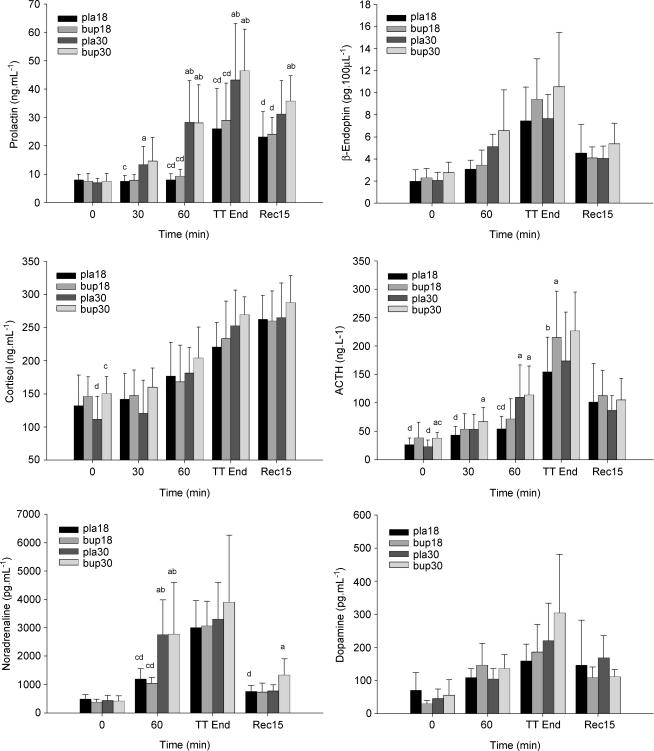
Circulating concentrations of pituitary and adrenal hormones are presented in Fig. 6. Ambient temperature altered the prolactin response to the experimental protocol (P < 0.001), with higher circulating concentrations observed at the end of the constant-load exercise and upon completion of the target amount of work in the heat. Serum prolactin was not influenced by bupropion at rest or during exercise (P = 0.173). Plasma ACTH concentrations at rest were increased by bupropion (P = 0.038). Beta-endorphin levels progressively increased throughout exercise under all conditions (P < 0.001), with bupropion producing a greater change (P = 0.049). ACTH increased during exercise in all trials (P = 0.005), with levels significantly higher during exercise in the heat at the end of the initial 60 min. At the end of the performance test plasma ACTH was higher in the bup18 trial (215 ± 81 ng l−1) than in the pla18 trial (155 ± 61 ng l−1; P = 0.028). Circulating serum cortisol increased progressively throughout exercise in all trials (P < 0.001). Ambient temperature did not appear to influence circulating cortisol concentrations (P = 0.191). While serum growth hormone was elevated 7–20-fold by the exercise protocol (P < 0.001), the environmental conditions (P = 0.131) and drug treatment (P = 0.223) did not influence the growth hormone response. Both plasma noradrenaline and dopamine concentrations increased throughout exercise (P < 0.001), with a return to near baseline levels following the 15-min recovery period.
Figure 6. Circulating concentrations of pituitary and adrenal hormones at rest and during exercise.
a, b, c, d denote a significant difference from corresponding values in pla18, bup18, pla30 and bup30 trials, respectively (P < 0.05). Values are mean ± SD.
Compared to resting values, both blood (P < 0.001) and plasma (P < 0.001) volumes were significantly reduced during exercise and after a 15-min recovery period. There was a difference in blood and plasma (P = 0.005) volume changes between environmental conditions (P = 0.003), with post hoc analysis identifying differences at the end of the constant-load exercise and following the recovery. Bupropion did not influence blood (P = 0.132), plasma (P = 0.080) or red cell (P = 0.897) responses to the experimental protocol.
Discussion
The present study investigated the effect of acute administration of a dual dopamine/noradrenaline reuptake inhibitor on performance, thermoregulation and hormonal responses to exercise in temperate and warm environments. While a number of studies have attempted to enhance exercise performance through the manipulation of central catecholaminergic neurotransmission in temperate conditions (Meeusen et al. 1997; Struder et al. 1998; Chinevere et al. 2002; Piacentini et al. 2004), this is the first study to evaluate the effect of a dual DA/NA reuptake inhibitor in warm ambient temperatures. Evidence suggests that exercise-induced hyperthermia may reduce motivation and drive to continue exercise, potentially contributing to the reduced capacity to perform prolonged exercise when ambient temperatures are elevated.
The results of the present study demonstrate that performance of a relatively short, preloaded TT was improved by bupropion when exercise was performed in warm ambient conditions, but no ergogenic benefit was apparent in a temperate environment. The 9% increase in performance observed in the warm was accompanied by higher TT heart rate and the attainment of a significantly higher core temperature at the completion of the TT compared to values measured during the placebo trial under the same environmental conditions. Interestingly, bupropion appeared to enable the maintenance of an elevated power output and the attainment of higher core body temperatures, with the same perception of effort and thermal discomfort reported during the placebo trial.
While there is good evidence that amphetamine, a potent DA agonist, can enhance exercise capacity in both rodents and humans (Borg et al. 1972; Gerald, 1978; Chandler & Blair, 1980), the failure to observe a performance effect at 18°C in the present study supports the findings of recent investigations of nutritional and pharmacological manipulation of catecholaminergic neurotransmission (Meeusen et al. 1997; Struder et al. 1998; Chinevere et al. 2002; Piacentini et al. 2004). Recently the influence of bupropion on exercise performance and the hormonal response to a 90 min TT has been investigated (Piacentini et al. 2004). While there were slight differences in the exercise protocol, the drug administration protocol in the present study was identical to that employed by Piacentini et al. (2004). The authors also reported no effect of bupropion on exercise performance in temperate environmental conditions (18°C).
Bupropion is a relatively weak, but selective, DA and NA reuptake inhibitor, with potency twice as great for DA as for NA, while showing little affinity for the 5-HT system (Holm & Spencer, 2000). Peak plasma concentrations are reached 3 h after ingestion, with the concentration of hydroxybupropion and threohydrobupropion, bupropion's major metabolites, reaching peak concentrations 6 h post-ingestion. While animal studies suggest that bupropion acts primarily through its effect on DA reuptake (Nomikos et al. 1989), the hormonal responses observed by Piacentini et al. (2004) led the authors to suggest that the central action of bupropion in humans was mediated largely through the noradrenergic system. This suggestion is supported by clinical studies (Cooper et al. 1994), and the observation that drugs acting exclusively on DA appear not to produce an appreciable antidepressant effect (Nestler et al. 2001). The discrepancy between human and animal data appears to result from differences in drug metabolism, with the metabolite hydroxybupropion not produced in rodents. This metabolite acts primarily on the NA transporter (Piacentini et al. 2003).
Core temperature was not influenced by the drug treatment during the initial 60 min period of fixed-intensity exercise, but marked differences were evident during the TT under both temperate and warm conditions. It appears that the elevated core temperature towards the end of the TT in the bup30 trial can be explained by an increased metabolic heat production, occurring as a result of the maintenance of a higher power output throughout the TT. It is interesting to note that only two (of nine) subjects attained a temperature of 40.0°C or greater during the pla30 trial, whereas seven breached this value when bupropion was administered (bup30 trial). It is not completely clear why a difference in core temperature was apparent between drug and placebo trials under temperate conditions, as power output was similar in these trials. Bupropion has been reported to possess thermogenic properties (Liu et al. 2002), possibility relating to its amphetamine-like structure (Nestler et al. 2001), and catecholaminergic neurotransmission may influence thermoregulation during exercise (Hasegawa et al. 2000). This may account for the different core temperature response during the TT in temperate conditions, but does not explain why no change in core temperature was measured during the fixed-intensity exercise and in the warm trials.
There is some limited evidence that higher dopaminergic activity increases tolerance to exercise in the heat (Bridge et al. 2003) and that the maintenance of a low central ratio of serotonergic to catecholaminergic activity is associated with feelings of arousal and motivation, consequently favouring increased performance (Davis & Bailey, 1997). It is possible to suggest that bupropion administration in the heat acted on central DA and NA neurotransmission to maintain motivation and arousal, enabling the subjects to continue to sustain a high power output despite approaching, and in some cases breaching, the ‘critical’ core temperature proposed by Nielsen et al. (1993). Attainment of a core temperature of 40.0°C has been suggested to result in a loss of drive to continue exercise (Nielsen, 1992), and has been associated with increased perception of effort (Nybo & Nielsen, 2001b), altered brain activity (Nielsen et al. 2001) and reduced voluntary activation during sustained contractions (Nybo & Nielsen, 2001a), all of which have been suggested to contribute to the development of fatigue and may serve as a protective mechanism limiting further heat production (Nielsen, 1992). This could indicate that subjects taking bupropion before exercise in a warm environment were capable of pushing into a potential ‘danger zone’ close to critical core temperature without, or with significantly dampened, negative feedback from the CNS. As evidence for a role of 5-HT during exercise in the heat is limited (Cheuvront et al. 2004; Strachan et al. 2004; Watson et al. 2004), these data suggest that catecholaminergic neurotransmission may act as an important neurobiological mediator of fatigue under conditions of heat stress.
Recent work has led to the observation that exercise performance in a warm environment may be in some way governed by a feed-forward mechanism that limits motor recruitment, consequently attenuating heat production (Marino, 2004; Marino et al. 2004; Tucker et al. 2004). Marino (2004) proposed that this ensures that the rate of heat accumulation by the body is restricted to allow the bout of exercise to be completed before the development of heat illness. The fall in power output observed from the outset of the time trial in the heat when compared to the temperate condition appears to lend support for this hypothesis, but the difference in core and skin temperatures at the end of the constant-load exercise may also be important factors in this response. An underlying physiological mechanism for the anticipatory regulation of power output has yet to be identified, but it seems likely that this is primarily a learned response developed through past experiences to enable the completion of a task before catastrophe (Ulmer, 1996; Marino, 2004). It is possible that acute manipulation of catecholaminergic neurotransmission with bupropion altered this feed-forward response in some way, allowing individuals to maintain a greater rate of heat production than was possible under placebo conditions. This model is relatively new, and future studies will help determine the importance of anticipatory regulation during prolonged exercise in the heat.
While Piacentini et al. (2004) failed to report a performance effect following bupropion, a marked difference in the hormonal response to exercise was apparent, suggesting that the drug administration produced an effect on central neurotransmission. Increased dopaminergic neurotransmission is associated with an inhibition of pituitary hormone release, with the exception of growth hormone (Checkley, 1980). Regulation of hormone release by NA is complicated by the type of adrenoceptor subtype activated, with conflicting responses produced by alpha and beta receptors. As stated earlier, it appears that bupropion acts primarily through changes to NA neurotransmission, making it difficult to predict expected changes to hormone release. Acute bupropion supplementation has been previously reported to increase the ACTH and cortisol response to exercise, while not influencing prolactin and growth hormone concentrations (Piacentini et al. 2004).
Changes in circulating prolactin concentrations have been widely employed as a peripheral marker of serotonergic activity (Van de Kar, 1997; Struder & Weicker, 2001), but its reliability has been questioned (Meeusen et al. 2001). Interpreting changes in plasma prolactin as an index of 5-HT activity may be misleading as prolactin release from the pituitary gland is governed through an complex interaction between a number of neurotransmitters rather than by serotonin alone (Meeusen et al. 2001). In the present study, serum prolactin concentrations were significantly elevated during exercise in the heat, but bupropion ingestion did not alter this response. A relationship between core temperature and circulating prolactin has been widely reported (Melin et al. 1988; Radomski et al. 1998; Pitsiladis et al. 2002), and it has been proposed that serotonergic activity may be upregulated during combined exercise and heat stress (Pitsiladis et al. 2002). As changes in core temperature and hypothalamic–pituitary–adrenal axis (HPA)-hormone secretion are primarily regulated by the hypothalamus, it appears that brain temperature during exercise may be causally related to the release of hormones from the pituitary gland (Radomski et al. 1998).
In contrast to previous reports (Brenner et al. 1997), serum cortisol concentrations were not influenced by the ambient conditions. Brenner and colleagues reported a marked elevation in circulating cortisol concentrations when moderate-intensity exercise was performed in warm (40°C) compared to temperate (23°C) conditions. A failure to observe a difference in cortisol between environmental conditions in the present investigation may be due to the attainment of relatively high core temperatures (>39°C) in the temperate trial, with clamping of body temperature previously demonstrated to abolish exercise-induced cortisol secretion (Cross et al. 1996). As previously reported (Piacentini et al. 2004), bupropion did not significantly influence the serum cortisol or growth hormone response, whereas plasma ACTH and beta-endorphin concentrations were higher at the end of the TT in temperate ambient conditions. In the present study there was a tendency for ACTH and beta-endorphin levels to be elevated in the heat with bupropion at this time point, but this difference failed to reach significance. The measurement of systemic beta-endorphin concentrations as a peripheral index of central opiate activity, however, has been questioned (Nybo & Secher, 2004). An acute dose of bupropion did not appear to alter plasma noradrenaline and dopamine concentrations under temperate or warm conditions, suggesting that the agent may have been acting directly on the CNS.
Two important findings arise from this study: (1) subjects completed a preloaded time trial 9% faster when bupropion was taken before exercise in a warm environment compared to a placebo treatment. This ergogenic effect was not apparent at 18°C; (2) 7 (of 9) subjects in the heat attained core temperatures equal to, or greater than, 40.0°C in the bupropion trial, compared to only two during the placebo trial. It is possible that this drug may dampen or override inhibitory signals arising from the CNS to cease exercise due to hyperthermia, and enable an individual to continue to maintain a high power output. This response appeared to occur, with the same perception of effort and thermal discomfort reported during the placebo trial, and may potentially increase the risk of developing heat illness. The present findings should be noted as the World Anti-Doping Agency (WADA) and International Olympic Committee (IOC), removed this drug from the list of prohibited substances in January 2003.
Acknowledgments
This project was partially supported by research funding from the Vrije Universiteit Brussel (OZR 387, 607, 990) and the Japanese Society for the Promotion of Sciences. We acknowledge the assistance of Dr Chris Dewaele and Luc Van Melckebeke at Biorad for the catecholamine analysis and Dr Ilse Smolders for the preparation of the treatments. The authors also wish to express their gratitude to Professor Ron Maughan and Dr Susan Shirreffs for help with the preparation of this manuscript.
References
- Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc. 1982;14:377–381. [PubMed] [Google Scholar]
- Borg G, Edstrom CG, Linderholm H, Marklund G. Changes in physical performance induced by amphetamine and amobarbital. Psychopharmacologia. 1972;26:10–18. doi: 10.1007/BF00421914. [DOI] [PubMed] [Google Scholar]
- Brenner IK, Zamecnik J, Shek PN, Shephard RJ. The impact of heat exposure and repeated exercise on circulating stress hormones. Eur J Appl Physiol Occup Physiol. 1997;76:445–454. doi: 10.1007/s004210050274. [DOI] [PubMed] [Google Scholar]
- Bridge MW, Weller AS, Rayson M, Jones DA. Responses to exercise in the heat related to measures of hypothalamic serotonergic and dopaminergic function. Eur J Appl Physiol. 2003;89:451–459. doi: 10.1007/s00421-003-0800-z. [DOI] [PubMed] [Google Scholar]
- Bruck K, Olschewski H. Body temperature related factors diminishing the drive to exercise. Can J Physiol Pharmacol. 1987;65:1274–1280. doi: 10.1139/y87-203. [DOI] [PubMed] [Google Scholar]
- Burgess ML, Davis JM, Borg TK, Buggy J. Intracranial self-stimulation motivates treadmill running in rats. J Appl Physiol. 1991;71:1593–1597. doi: 10.1152/jappl.1991.71.4.1593. [DOI] [PubMed] [Google Scholar]
- Chandler JV, Blair SN. The effect of amphetamines on selected physiological components related to athletic success. Med Sci Sports Exerc. 1980;12:65–69. [PubMed] [Google Scholar]
- Chaouloff F. Physical exercise and brain monoamines: a review. Acta Physiol Scand. 1989;137:1–13. doi: 10.1111/j.1748-1716.1989.tb08715.x. [DOI] [PubMed] [Google Scholar]
- Checkley SA. Neuroendocrine tests of monoamine function in man: a review of basic theory and its application to the study of depressive illness. Psychol Med. 1980;10:35–53. doi: 10.1017/s0033291700039593. [DOI] [PubMed] [Google Scholar]
- Cheuvront SN, Carter R, Kolka MA, 3rd, Lieberman HR, Kellogg MD, Sawka MN. Branched-chain amino acid supplementation and human performance when hypohydrated in the heat. J Appl Physiol. 2004;97:1275–1282. doi: 10.1152/japplphysiol.00357.2004. [DOI] [PubMed] [Google Scholar]
- Chinevere TD, Sawyer RD, Creer AR, Conlee RK, Parcell AC. Effects of 1-tyrosine and carbohydrate ingestion on endurance exercise performance. J Appl Physiol. 2002;93:1590–1597. doi: 10.1152/japplphysiol.00625.2001. [DOI] [PubMed] [Google Scholar]
- Cooper BR, Wang CM, Cox RF, Norton R, Shea V, Ferris RM. Evidence that the acute behavioral and electrophysiological effects of bupropion (Wellbutrin) are mediated by a noradrenergic mechanism. Neuropsychopharmacology. 1994;11:133–141. doi: 10.1038/npp.1994.43. [DOI] [PubMed] [Google Scholar]
- Cross MC, Radomski MW, VanHelder WP, Rhind SG, Shephard RJ. Endurance exercise with and without a thermal clamp: effects on leukocytes and leukocyte subsets. J Appl Physiol. 1996;81:822–829. doi: 10.1152/jappl.1996.81.2.822. [DOI] [PubMed] [Google Scholar]
- Davis JM. Nutrition, neurotransmitters and central nervous system fatigue. In: Maughan RJ, editor. Nutrition in Sport. Oxford: Blackwell Science; 2000. pp. 171–183. [Google Scholar]
- Davis JM, Bailey SP. Possible mechanisms of central nervous system fatigue during exercise. Med Sci Sports Exerc. 1997;29:45–57. doi: 10.1097/00005768-199701000-00008. [DOI] [PubMed] [Google Scholar]
- Dill DB, Costill DL. Calculation of percentage changes in volumes of blood, plasma and red cells in dehydration. J Appl Physiol. 1974;37:247–248. doi: 10.1152/jappl.1974.37.2.247. [DOI] [PubMed] [Google Scholar]
- Galloway SD, Maughan RJ. Effects of ambient temperature on the capacity to perform prolonged cycle exercise in man. Med Sci Sports Exerc. 1997;29:1240–1249. doi: 10.1097/00005768-199709000-00018. [DOI] [PubMed] [Google Scholar]
- Gerald MC. Effects of (+)-amphetamine on the treadmill endurance performance of rats. Neuropharmacology. 1978;17:703–704. doi: 10.1016/0028-3908(78)90083-7. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Alonso J, Teller C, Andersen SL, Jensen FB, Hyldig T, Nielsen B. Influence of body temperature on the development of fatigue during prolonged exercise in the heat. J Appl Physiol. 1999;86:1032–1039. doi: 10.1152/jappl.1999.86.3.1032. [DOI] [PubMed] [Google Scholar]
- Hargreaves M, Febbraio M. Limits to exercise performance in the heat. Int J Sports Med. 1998;19(Suppl. 2):S115–S116. doi: 10.1055/s-2007-971973. [DOI] [PubMed] [Google Scholar]
- Hasegawa H, Yazawa T, Yasumatsu M, Otokawa M, Aihara Y. Alteration in dopamine metabolism in the thermoregulatory center of exercising rats. Neurosci Lett. 2000;289:161–164. doi: 10.1016/s0304-3940(00)01276-3. [DOI] [PubMed] [Google Scholar]
- Holm KJ, Spencer CM. Bupropion: a review of its use in the management of smoking cessation. Drugs. 2000;59:1007–1024. doi: 10.2165/00003495-200059040-00019. [DOI] [PubMed] [Google Scholar]
- Jeukendrup A, Saris WH, Brouns F, Kester AD. A new validated endurance performance test. Med Sci Sports Exerc. 1996;28:266–270. doi: 10.1097/00005768-199602000-00017. [DOI] [PubMed] [Google Scholar]
- Liu YL, Connoley IP, Harrison J, Heal DJ, Stock MJ. Comparison of the thermogenic and hypophagic effects of sibutramine's metabolite 2 and other monoamine reuptake inhibitors. Eur J Pharmacol. 2002;452:49–56. doi: 10.1016/s0014-2999(02)02226-4. [DOI] [PubMed] [Google Scholar]
- Marino FE. Anticipatory regulation and avoidance of catastrophe during exercise-induced hyperthermia. Comp Biochem Physiol B Biochem Mol Biol. 2004;139:561–569. doi: 10.1016/j.cbpc.2004.09.010. [DOI] [PubMed] [Google Scholar]
- Marino FE, Lambert MI, Noakes TD. Superior performance of African runners in warm humid but not in cool environmental conditions. J Appl Physiol. 2004;96:124–130. doi: 10.1152/japplphysiol.00582.2003. [DOI] [PubMed] [Google Scholar]
- Meeusen R, Piacentini MF, De Meirleir K. Brain microdialysis in exercise research. Sports Med. 2001;31:965–983. doi: 10.2165/00007256-200131140-00002. [DOI] [PubMed] [Google Scholar]
- Meeusen R, Piacentini MF, Van Den Eynde S, Magnus L, De Meirleir K. Exercise performance is not influenced by a 5-HT reuptake inhibitor. Int J Sports Med. 2001;22:329–336. doi: 10.1055/s-2001-15648. [DOI] [PubMed] [Google Scholar]
- Meeusen R, Roeykens J, Magnus L, Keizer H, De Meirleir K. Endurance performance in humans: the effect of a dopamine precursor or a specific serotonin (5-HT2A/2C) antagonist. Int J Sports Med. 1997;18:571–577. doi: 10.1055/s-2007-972683. [DOI] [PubMed] [Google Scholar]
- Melin B, Cure M, Pequignot JM, Bittel J. Body temperature and plasma prolactin and norepinephrine relationships during exercise in a warm environment: effect of dehydration. Eur J Appl Physiol Occup Physiol. 1988;58:146–151. doi: 10.1007/BF00636618. [DOI] [PubMed] [Google Scholar]
- Nestler EJ, Hyman SE, Malenka RC. Molecular Neuropharmacology: a Foundation for Clinical Neuroscience. New York: McGraw-Hill; 2001. [Google Scholar]
- Nielsen B. Heat stress causes fatigue! Exercise performance during acute and repeated exposures to hot, dry environments. In: Marconnet P, Komi PV, Saltin B, Sejersted OM, editors. Muscle Fatigue Mechanisms in Exercise and Training. Basel: Karger; 1992. pp. 207–217. [Google Scholar]
- Nielsen B, Hales JR, Strange S, Christensen NJ, Warberg J, Saltin B. Human circulatory and thermoregulatory adaptations with heat acclimation and exercise in a hot, dry environment. J Physiol. 1993;460:467–485. doi: 10.1113/jphysiol.1993.sp019482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen B, Hyldig T, Bidstrup F, Gonzalez-Alonso J, Christoffersen GR. Brain activity and fatigue during prolonged exercise in the heat. Pflugers Arch. 2001;442:41–48. doi: 10.1007/s004240100515. [DOI] [PubMed] [Google Scholar]
- Nomikos GG, Damsma G, Wenkstern D, Fibiger HC. Acute effects of bupropion on extracellular dopamine concentrations in rat striatum and nucleus accumbens studied by in vivo microdialysis. Neuropsychopharmacology. 1989;2:273–279. doi: 10.1016/0893-133x(89)90031-6. [DOI] [PubMed] [Google Scholar]
- Nybo L, Nielsen B. Hyperthermia and central fatigue during prolonged exercise in humans. J Appl Physiol. 2001a;91:1055–1060. doi: 10.1152/jappl.2001.91.3.1055. [DOI] [PubMed] [Google Scholar]
- Nybo L, Nielsen B. Perceived exertion is associated with an altered brain activity during exercise with progressive hyperthermia. J Appl Physiol. 2001b;91:2017–2023. doi: 10.1152/jappl.2001.91.5.2017. [DOI] [PubMed] [Google Scholar]
- Nybo L, Secher NH. Cerebral perturbations provoked by prolonged exercise. Prog Neurobiol. 2004;72:223–261. doi: 10.1016/j.pneurobio.2004.03.005. [DOI] [PubMed] [Google Scholar]
- Owasoyo JO, Neri DF, Lamberth JG. Tyrosine and its potential use as a countermeasure to performance decrement in military sustained operations. Aviat Space Environ Med. 1992;63:364–369. [PubMed] [Google Scholar]
- Parkin JM, Carey MF, Zhao S, Febbraio MA. Effect of ambient temperature on human skeletal muscle metabolism during fatiguing submaximal exercise. J Appl Physiol. 1999;86:902–908. doi: 10.1152/jappl.1999.86.3.902. [DOI] [PubMed] [Google Scholar]
- Parsons K. Human Thermal Environments. London: Taylor & Francis; 2003. [Google Scholar]
- Piacentini MF, Clinckers R, Meeusen R, Sarre S, Ebinger G, Michotte Y. Effect of bupropion on hippocampal neurotransmitters and on peripheral hormonal concentrations in the rat. J Appl Physiol. 2003;95:652–656. doi: 10.1152/japplphysiol.01058.2002. [DOI] [PubMed] [Google Scholar]
- Piacentini MF, Meeusen R, Buyse L, De Schutter G, De Meirleir K. Hormonal responses during prolonged exercise are influenced by a selective DA/NA reuptake inhibitor. Br J Sports Med. 2004;38:129–133. doi: 10.1136/bjsm.2002.000760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitsiladis YP, Strachan AT, Davidson I, Maughan RJ. Hyperprolactinaemia during prolonged exercise in the heat: evidence for a centrally mediated component of fatigue in trained cyclists. Exp Physiol. 2002;87:215–226. doi: 10.1113/eph8702342. [DOI] [PubMed] [Google Scholar]
- Radomski MW, Cross M, Buguet A. Exercise-induced hyperthermia and hormonal responses to exercise. Can J Physiol Pharmacol. 1998;76:547–552. doi: 10.1139/cjpp-76-5-547. [DOI] [PubMed] [Google Scholar]
- Ramanathan LM. A new weighting system for mean surface temperature of the human body. J Appl Physiol. 1964;19:531–532. doi: 10.1152/jappl.1964.19.3.531. [DOI] [PubMed] [Google Scholar]
- Strachan A, Leiper J, Maughan R. The failure of acute paroxetine administration to influence human exercise capacity, RPE or hormone responses during prolonged exercise in a warm environment. Exp Physiol. 2004;89:657–664. doi: 10.1113/expphysiol.2004.027839. [DOI] [PubMed] [Google Scholar]
- Struder HK, Hollmann W, Platen P, Donike M, Gotzmann A, Weber K. Influence of paroxetine, branched-chain amino acids and tyrosine on neuroendocrine system responses and fatigue in humans. Horm Metab Res. 1998;30:188–194. doi: 10.1055/s-2007-978864. [DOI] [PubMed] [Google Scholar]
- Struder HK, Weicker H. Physiology and pathophysiology of the serotonergic system and its implications on mental and physical performance. Part II. Int J Sports Med. 2001;22:482–497. doi: 10.1055/s-2001-17606. [DOI] [PubMed] [Google Scholar]
- Tatterson AJ, Hahn AG, Martin DT, Febbraio MA. Effects of heat stress on physiological responses and exercise performance in elite cyclists. J Sci Med Sport. 2000;3:186–193. doi: 10.1016/s1440-2440(00)80080-8. [DOI] [PubMed] [Google Scholar]
- Tucker R, Rauch L, Harley YX, Noakes TD. Impaired exercise performance in the heat is associated with an anticipatory reduction in skeletal muscle recruitment. Pflugers Arch. 2004;448:422–430. doi: 10.1007/s00424-004-1267-4. [DOI] [PubMed] [Google Scholar]
- Ulmer HV. Concept of an extracellular regulation of muscular metabolic rate during heavy exercise in humans by psychophysiological feedback. Experientia. 1996;52:416–420. doi: 10.1007/BF01919309. [DOI] [PubMed] [Google Scholar]
- Van de Kar LD. 5-HT receptors involved in the regulation of hormone secretion. In: Baumgarten HG, Gothert M, editors. Serotonergic Neurons and 5-HT Receptors in the CNS. New York: Springer; 1997. pp. 557–562. [Google Scholar]
- Watson P, Shirreffs SM, Maughan RJ. The effect of acute branched-chain amino acid supplementation on prolonged exercise capacity in a warm environment. Eur J Appl Physiol. 2004;93:306–314. doi: 10.1007/s00421-004-1206-2. [DOI] [PubMed] [Google Scholar]