Abstract
Neuronal nicotinic acetylcholine receptors (nAChRs) are widely expressed in the brain where they are involved in a variety of physiological processes, including cognition and development. The nAChRs are ligand-gated cationic channels, and different subtypes are known to be differentially permeable to Ca2+; the α7-containing nAChRs are generally considered to be the most permeable. Ca2+ can activate and regulate a variety of signal transduction cascades, and the influx of Ca2+ through these receptors may have implications for synaptic plasticity. To determine the Ca2+ permeability of the nAChRs in rat hippocampal interneurones in the slice, which contain diverse subtypes of α7- and non-α7-containing nAChRs, we combined patch-clamp electrophysiology recordings with conventional fura-2 fluorescence imaging techniques. We estimated the relative Ca2+ permeability of the channels by determining the ratio of the increase in [Ca2+]i level (Δ[Ca2+]i) in the soma to the integrated transmembrane current (charge, Q) induced by the activation of the nAChRs, and compared this ratio to the highly Ca2+ permeable NMDA subtype of glutamate receptor channel. In all cells tested, the Δ[Ca2+]i/Q ratio was significantly larger (i.e. more than twice as big) for responses activated by NMDA than for α7-containing nAChRs in interneurones; the activation of the non-α7 nAChRs did not produce any significant increase in [Ca2+]i. Interestingly, the Ca2+ permeability of native α7 nAChRs in PC12 cells was significantly larger than in hippocampal interneurones, and not significantly different from NMDA receptors. Therefore, the α7-containing nAChRs in rat hippocampal interneurones are significantly less permeable to Ca2+ than not only NMDA receptors but also α7 nAChRs in PC12 cells.
Multiple subtypes of neuronal nicotinic acetylcholine receptor channels (nAChRs) are expressed in the brain, the functions of which have been linked to specific cognitive processes in humans and animal models, and development (Jones et al. 1999; Levin, 2002). Furthermore, dysfunctions in these receptors may be involved in various neurodegenerative diseases, such as Alzheimer's disease, epilepsy, and schizophrenia (Jones et al. 1999; Pettit et al. 2001; Liu et al. 2001). Functionally, nAChRs are ligand-gated cationic channels with the capacity to elicit local changes in cytoplasmic calcium ([Ca2+]i) levels, and different subtypes of neuronal nAChRs are known to be differentially permeable to Ca2+ (Bertrand et al. 1993; Séguéla et al. 1993; Fucile, 2004). Although much evidence indicates that nAChRs can directly regulate synaptic plasticity in the hippocampus (Hunter et al. 1994; Fujii & Sumikawa, 2001; Ji et al. 2001; McGehee, 2002), the cellular mechanisms involved are not fully understood, but likely to involve Ca2+. Calcium ions can activate and regulate a variety of signal transduction cascades, and play a key role in the short- and long-term regulation of nAChRs (Quick & Lester, 2002; Dajas-Bailador & Wonnacott, 2004; Fucile, 2004).
The influx of Ca2+ through nAChRs has implications not only for synaptic plasticity, but also for the release of neurotransmitter for those presynaptic terminals that express Ca2+-permeable nAChRs (Dajas-Bailador & Wonnacott, 2004; Fucile, 2004). For example in the hippocampus, Gray et al. (1996) found that activation of α7-containing nAChRs increased intraterminal Ca2+ levels and facilitated glutamatergic release; however, others failed to observe this effect (Vogt & Regehr, 2001). In heterologous expression systems, homomeric α7 nAChRs were shown to have a high permeability to Ca2+ (Bertrand et al. 1993; Séguéla et al. 1993), as do native α7-containing nAChRs in cultured rat hippocampal neurones (Castro & Albuquerque, 1995). There is a wide range of estimates for the Ca2+ permeability of both native and heterologously expressed nAChRs, but α7-containing receptors are thought to have the highest permeability to Ca2+ (Fucile, 2004).
Because of the importance of nAChR function in the hippocampus in relation to cognition and synaptic plasticity, and the fact that diverse subtypes of α7- and non-α7-containing nAChRs are preferentially expressed in hippocampal interneurones (Jones et al. 1999), it is critical to understand the Ca2+ permeability of nAChRs in these neurones in a more physiological setting such as the slice. To do this, we combined patch-clamp electrophysiology recordings with conventional fura-2 fluorescence imaging techniques to measure the changes in [Ca2+]i in the soma due to the activation of the various nAChRs. Recently we reported that the increase in [Ca2+]i levels due to the activation of α7-containing nAChRs in these interneurones in the slice was modest (Khiroug et al. 2003). Here we wanted to compare and contrast the Ca2+ permeability of these nAChRs (i.e. both α7- and non-α7-containing) with the permeability of the NMDA subtype of the ionotropic glutamate receptor channel, which is known to be highly Ca2+ permeable; in fact the Ca2+ permeability of α7 receptors expressed in Xenopus oocytes was greater than that for NMDA receptors expressed in the same cells (Séguéla et al. 1993). We also compared the Ca2+ permeability of native α7-containing nAChRs in cultured rat pheochromocytoma 12 (PC12) cells. Whereas we found that the relative Ca2+ permeability of NMDA receptors was similar to that of the α7 receptors in PC12 cells, the Ca2+ permeability of native α7-containing nAChRs in rat hippocampal interneurones was approximately half that of both of these receptors (i.e. native α7 receptors in PC12 cells and NMDA receptors in rat hippocampal interneurones). We suggest that the lower than expected Ca2+ permeability for hippocampal α7 nAChRs may indicate a differential subunit makeup, or regulation, of these receptors.
Methods
Slice preparation
All experiments were carried out in accordance with guidelines approved by the NIEHS Animal Care and Use Committee, which includes minimizing the number of animals used and their suffering. Standard techniques were used to prepare 350 mm thick acute hippocampal slices from 14- to 19-day-old rats (Fayuk & Yakel, 2004). Briefly, rats were anaesthetized with halothane (Sigma) and decapitated. Brains were quickly removed and placed into an ice-cold oxygenated, artificial cerebrospinal fluid (ACSF) containing (mm): 119 NaCl, 2.5 KCl, 1.3 MgCl2, 2.5 CaCl2, 1 NaH2PO4, 26.2 NaHCO3, and 11 glucose. Upon dissection, brain chunks were glued to the stage of a vibrating microtome (VT1000S; Leica, Germany) for slicing while immersed in the cooled oxygenated ACSF. Slices were then used for recordings within about 6 h, and after at least 1 h of recovery period.
Culturing of PC12 cells
Rat adrenal medulla pheochromocytoma cells (PC12) were obtained from American Type Culture Collection (ATCC, Rockville, MD, USA), and were grown in medium consisting of Kaighn's modified Ham's F12 medium (Gibco) supplemented with 15% heat-inactivated horse serum, 2.5% fetal bovine serum, 2 mm glutamine, 100 units ml−1 penicillin G and 100 μg ml−1 streptomycin. Cells were maintained at 37°C in a humidified incubator with 5% CO2, and plated on poly d-lysine-coated coverslips. After 2 or 3 days in culture, nerve growth factor (NGF; 50 ng ml−1) was added to differentiate the cells, and cells were used for experiments at least 3 days after NGF was added.
Electrophysiology
Whole-cell patch-clamp recordings were performed on either CA1 interneurones (stratum radiatum or stratum oriens) or pyramidal neurones in slices, or on cultured PC12 cells. Patch pipettes (Garner 7052 or 8250 glass, with resistances of 3–4 MΩ) were filled with a solution that contained (mm): 120 caesium gluconate, 2 NaCl, 4 Na2ATP, 0.4 Na2GTP, 4 MgCl2 and 20 Hepes (pH 7.3). Slices were superfused at room temperature (18–22°C) with ACSF. Synaptic activity was blocked with TTX (1 μm) added to the ACSF. In some experiments, atropine (1 mm) was also added to block putative muscarinic AChR-mediated responses. Cells were clamped using an Axopatch 200B amplifier (Axon Instruments, Union City, CA, USA) at a holding potential of −70 mV. Currents were recorded and analysed using pCLAMP software (Axon Instruments); recordings were analysed only if the holding current was less than 100 pA. Responses were induced by pressure application (Picospritzer II; General Valve Co., Fairfield, NJ, USA) of either ACh, choline, or NMDA delivered via a theta glass pipette (Harvard Apparatus, o.d. = 2 mm) pulled to a tip diameter of ∼3 μm (P-97 puller, Sutter Instruments, Novato, CA, USA) placed 20–30 μm from the cell body; in this way, two agonists could be rapidly applied to the same cell. Brief (20–200 ms) duration pulses at 10–20 p.s.i. were typically used to activate α7-containing nAChRs, whereas longer (5–10 s) pulses (< 10 p.s.i.) were typically used to activate non-α7 receptors. Drugs studied were diluted at final concentrations in ACSF and were delivered to the cell through a gravity-fed multichannel perfusion system ending with a non-metallic syringe needle (250 μm i.d., WPI) placed just above the slice surface to ensure the homogeneous coverage of the area surrounding the cell being studied.
Imaging of [Ca2+]i
Changes in intracellular Ca2+ levels ([Ca2+]i) in the soma in response to pressure application of either ACh, choline, or NMDA were assessed with conventional fura-2 fluorescence imaging techniques using an Eclipse E600FN upright microscope (Nikon, Japan) with a 40× fluorescence water-immersion objective, a Lambda DG-4 light source (Sutter Instrument Company, Novato, CA, USA) equipped with a stabilized xenon arc lamp (175 W) and four-channel filter interchange system, and a Cascade cooled (−40°C) 16-bit digital camera (Roper Scientific, USA). The AQM Advance imaging software (Kinetic Imaging Ltd, UK) was utilized to acquire and store images for off-line analysis, and to synchronize with membrane current recordings. Fura-2 pentapotassium salt (200–400 μm; Molecular Probes) was dialysed into the cell via the patch pipette, its fluorescence was alternately excited with 340 and 380 nm light, and pairs of images were recorded using a 510 nm emission filter at 5 Hz with 50 ms exposure time for each wavelength used. In each cell tested, background fluorescence was monitored until it is stabilized (usually by 5 min) prior to recordings. Four by four image binning was used to increase the signal-to-noise ratio and time resolution. A region of interest was selected and the 340/380 ratio of fluorescence intensities corrected for background signal were calculated using the imaging software. The ratio values were then converted to [Ca2+]i levels according to the calibration curve obtained with the in vitro Ca2+ calibration procedure using the Ca2+ calibration kit (Molecular Probes). This procedure consisted of measurements of the 340/380 ratio in solutions containing 11 different Ca2+ concentrations from 0 to 39 μm; the ratio value for 0 μm was 0.44, and for 39 μm was 8.4. We also made a calibration according to the method described by Grynkiewicz et al. (1985). In this case, [Ca2+]i levels were calculated using the following equation:
where R is the 340/380 ratio measured in experiments, and Rmin and Rmax are the 340/380 ratio values measured from cells filled with either a Ca2+-free intracellular solution (i.e. 10 mm EGTA and no added Ca2+), or where fura-2 was saturated with Ca2+ (1 mm of added Ca2+), respectively. All the types of cells studied here (i.e. interneurones, pyramidal cells, and PC12 cells) were calibrated with this method, and we found no significant difference in Rmin and Rmax values between cells (assuming that the Ca2+ signals in experiments were equally proportional to recorded 340/380 ratio changes); the Rmin and Rmax values for interneurones, pyramidal cells, and PC12 cells were, respectively (n = 3 cells for each), 0.37 ± 0.1 and 6.0 ± 0.3, 0.34 ± 0.1 and 7.1 ± 0.2, and 0.30 ± 0.1 and 6.1 ± 0.1. The difference between these two calibration methods is that the Rmin and Rmax values measured intracellularly (Grynkiewicz et al. 1985) were slightly lower than the 340/380 ratio measured in vitro using the Ca2+ calibration kit. However, according to our calculations this slight discrepancy may affect background Ca2+ measurements by no more than 20 nm, and will not have any significant effect on measured Ca2+ changes. Therefore the values reported here for [Ca2+]i levels will be based on the calibrations using the in vitro calibration kit. Statistical analyses were performed using Origin software (OriginLab Corp., Northampton, MA, USA). Averaged data were presented as means ± s.e.m., and statistical significance was tested using ANOVA.
Calculation of relative Ca2+ permeability
We estimated the relative Ca2+ permeability of the channels by the ratio of the increase in [Ca2+]i level (Δ[Ca2+]i) to the integrated membrane current (charge, Q) induced by the activation of these channels in the same cell; this ratio was referred to as the Permeability Index and will be in units of moles per litre per colouomb (m C−1). The time for current integration and [Ca2+]i amplitude measurement was 1 s after the beginning of agonist application because in most cases, this was the time the maximal Ca2+ response to choline occurred. Although NMDA responses typically had not peaked by 1 s, to maintain consistency we still used this time for measurement.
Results
[Ca2+]i level increase induced by the activation of α7 (but not non-α7) nAChRs in rat hippocampal interneurones in the slice
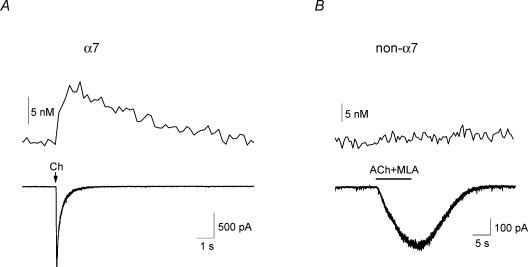
In acute slices of rat hippocampus, brief (20–200 ms) pressure applications of a maximal dose of the α7 nAChR-selective agonist, choline (10 mm; Papke et al. 1996), to voltage-clamped CA1 stratum radiatum or stratum oriens interneurones elicited rapidly activating α7-containing nAChR-mediated responses that decayed quickly (Fig. 1A); previously we have shown that these responses are blocked by the α7-selective antagonist methyllycaconitine (MLA; Fayuk & Yakel, 2004). To measure changes in cytoplasmic Ca2+ levels ([Ca2+]i) in the soma in these voltage-clamped cells, we used the conventional fura-2 fluorescent imaging technique and a digital camera (see Methods). In addition to inducing an inward current response, choline also induced a significant increase in [Ca2+]i level (Fig. 1A). The basal [Ca2+]i level was 44 ± 5 nm, and choline significantly increased this by 7 ± 1 nm (n = 41 cells).
Figure 1. Increased [Ca2+]i level due to activation of α7, but not non-α7, nAChRs.
A, brief (20–200 ms) duration pressure applications (arrows) of choline (‘Ch’; 10 mm) selectively activated α7-containing nAChRs in a neurone, inducing a rapidly activating and decaying inward current response (bottom trace), and a slower activating and decaying increase in fluorescence due to an increasing [Ca2+]i level (top trace). B, in a different neurone, the activation of non-α7 nAChRs by longer (10 s) pressure applications of ACh (2 mm), along with the α7-selective antagonist MLA (10 nm), induced a more slowly activating inward current response (bottom trace) without any significant change in [Ca2+]i level (top trace).
We also examined whether the activation of non-α7 nAChRs, which have previously been shown to be less permeable to Ca2+ than α7 nAChRs (Fucile, 2004), altered [Ca2+]i levels. To selectively activate non-α7 nAChRs, longer (5 s) pressure pulses of ACh (2 mm) were typically used, along with MLA (10 nm) to block α7 receptors (Fayuk & Yakel, 2004). Unlike with the activation of α7 nAChRs, the activation of the non-α7 nAChRs did not significantly change the [Ca2+]i level (Fig. 1B).
[Ca2+]i changes induced by the activation of α7 nAChRs are smaller than during depolarization
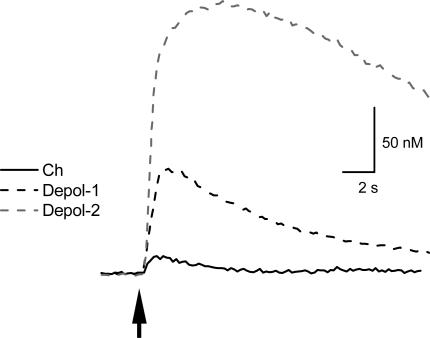
The increase in [Ca2+]i level induced by choline was compared to [Ca2+]i signals induced by depolarization to activate voltage-gated Ca2+ channels (VGCCs). When the membrane potential was depolarized to +10 mV for either 100 ms or 1 s, the increase in [Ca2+]i level was greater than that due to the activation of α7-containing nAChRs (Fig. 2); the average increase in [Ca2+]i level was 75 ± 9 nm (n = 8 cells) and 208 ± 28 nm (n = 3 cells) for the 100 ms and 1 s depolarizations, respectively. This suggests that under our current recording conditions, we are able to detect large changes in the [Ca2+]i level, and that the modest changes induced by the activation of α7-containing nAChRs were not due to technical limitations.
Figure 2. [Ca2+]i changes due to activation of α7 nAChRs are smaller than during depolarization.
The increase in [Ca2+]i level due to a brief application of choline (10 mm; arrow; continuous line) is smaller than the increase in [Ca2+]i level due to depolarization to +10 mV for a duration of either 100 ms (‘Depol-1’; black dashed line) or 1 s (‘Depol-2’; grey dashed line).
Comparison of [Ca2+]i signals induced by the activation of α7 nAChR and NMDA glutamate receptor subtypes
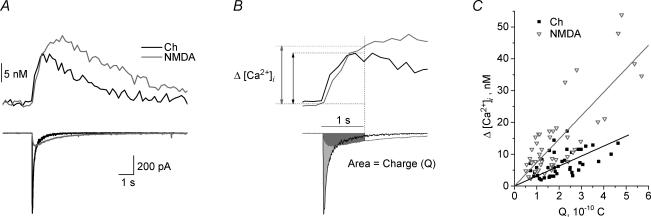
We compared the [Ca2+]i signal induced by the activation of the α7 nAChR with those from the activation of the NMDA receptor subtype of ionotropic glutamate receptor channel, the latter of which is known to be highly Ca2+ permeable. For the interneurone shown in Fig. 3A, the amplitude of the current response induced by NMDA (1 mm) was much smaller than that induced by choline (10 mm); however, NMDA induced a larger [Ca2+]i signal. To quantify and compare the relative Ca2+ permeability, we plotted the peak increase in [Ca2+]i level (Δ[Ca2+]i) versus the integrated membrane current (i.e. charge, Q) (see Methods for details); the slope of this line (i.e. the Δ[Ca2+]i/Q ratio) was termed the Permeability Index (PI; Fig. 3). For both NMDA and choline, there was a near-linear relationship between Q and Δ[Ca2+]i (Fig. 3C). The PI was significantly larger for responses activated by NMDA (78 ± 8 m C−1; n = 25 cells) than for choline (36 ± 3 m C−1; n = 41 cells; P < 0.001). Therefore on this basis, we estimate that the Ca2+ permeability of the α7-containing nAChRs in rat hippocampal interneurones is about 46% of that of NMDA receptors in the same cells.
Figure 3. Comparison of [Ca2+]i signals due to activation of α7 and NMDA receptors.
A, the application of choline (10 mm; black traces) and NMDA (1 mm; grey traces) in the same neurone induced inward current responses (bottom traces) with different peak amplitudes and kinetics, as well as increases in [Ca2+]i (top traces). Even though the amplitude of the NMDA current response was much smaller, the resulting increase in [Ca2+]i was larger than for choline. B, the total amount of Ca2+ influx (as indicated by the increase in [Ca2+]i level; top) is proportional to the integrated membrane current (i.e. charge, Q), the latter of which is estimated as the area under the current trace (bottom). The time for current integration and [Ca2+]i amplitude measurement was 1 s after the beginning of agonist application (horizontal bar). C, the plot of the peak increase in [Ca2+]i level versus the corresponding integrated current (Q) for choline (filled black squares) and NMDA (open grey triangles) responses yielded a near-linear relationship (linear regressions yielded R and P values of 0.52 and < 0.003 for choline, and 0.80 and < 0.0001 for NMDA). We have termed the slope of these lines the Permeability Index (PI).
[Ca2+]i signals induced by α7 nAChR activation are not due to Ca2+ intracellular store depletion, nor the activation of VGCCs
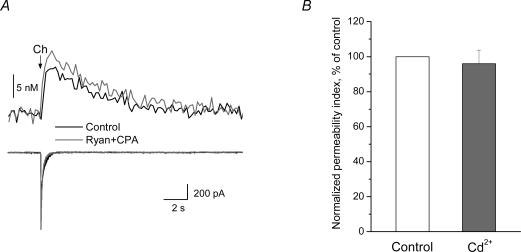
We tested whether the [Ca2+]i signals induced by activation of the α7 nAChRs were due to the influx of Ca2+ directly through these receptors, or via other potential sources of Ca2+. To test whether intracellular Ca2+ store depletion was involved, slices were pretreated with ryanodine and cyclopiazonic acid (CPA). Ryanodine binds to ryanodine receptors (RyRs), blocking the calcium-induced calcium release (CICR) from internal stores (McPherson et al. 1991). Internal stores may also release Ca2+ via activation of IP3 receptors (Berridge, 1993). CPA, a selective blocker of sarcoplasmic–endoplasmic reticulum Ca2+-ATPase (SERCA pumps; Seidler et al. 1989), was used to deplete the smooth endoplasmic reticulum-derived Ca2+ stores. After inducing a choline response, ryanodine and CPA (both at 20 μm) were applied for 10 min; this treatment slowly increased [Ca2+]i levels, which generally returned to baseline levels. After the addition of ryanodine and CPA, neither the peak amplitude of the [Ca2+]i signal, nor the amplitude of the choline-induced response, was significantly changed in all cells tested (n = 3 cells; tested both in control and with ryanodine and CPA; Fig. 4A). In addition, ryanodine and CPA had no significant effect on NMDA receptor-induced [Ca2+]i signals (n = 4 cells; data not shown). These data indicate that CICR does not contribute to the [Ca2+]i signals observed during either choline- or NMDA-induced responses.
Figure 4. [Ca2+]i signals due to α7 nAChR activation are not due to Ca2+ intracellular store depletion, nor the activation of VGCCs.
A, after inducing a choline response, slices were treated for 10 min with ryanodine and CPA (both at 20 μm) to prevent intracellular store depletion during α7 nAChR activation. Neither the peak amplitude of the [Ca2+]i signal (top traces) nor the current response (bottom traces) was significantly changed by pretreatment with ryanodine and CPA. B, the application of cadmium (Cd2+; 200 μm) had no significant effect on the PI (3 cells; tested both in control and with Cd2+).
We also tested whether Ca2+ influx through VGCCs might have contributed to the choline- or NMDA-induced [Ca2+]i signals. To do this, VGCCs were blocked with cadmium (Cd2+; 200 μm); this did not significantly affect the Ca2+ permeability index for responses to either choline (n = 3 cells; Fig. 4B) or NMDA (n = 6 cells; data not shown).
Ca2+ permeability of native α7 nAChRs in PC12 cells and NMDA receptors in pyramidal neurones
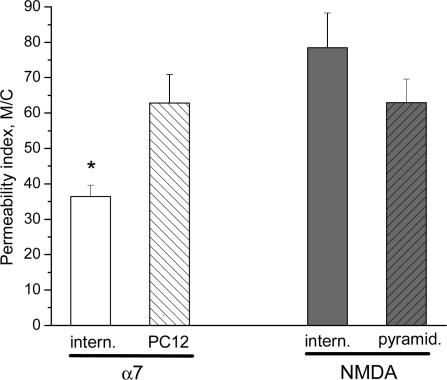
We explored the relative Ca2+ permeability of native α7 nAChRs and NMDA receptors in other cell types to determine if the lower permeability of the α7 nAChRs (as compared to NMDA receptors) was specific to interneurones. In cultured pheochromocytoma 12 (PC12) cells, the activation of α7 nAChRs with choline induced [Ca2+]i signals that were significantly larger (the PI was 63 ± 8 m C−1; n = 10 cells; P < 0.002) than the [Ca2+]i signals induced by choline in rat hippocampal interneurones (Fig. 5). Furthermore, the PI for the α7 nAChRs in PC12 cells was not significantly different from that for NMDA receptors in interneurones (Fig. 5). These data suggest that the Ca2+ permeability of the α7-containing nAChRs in rat hippocampal interneurones is significantly less than that for α7 nAChRs in PC12 cells.
Figure 5. Ca2+ permeability of native α7 nAChRs in PC12 cells and NMDA receptors in pyramidal neurones.
The PI values for the choline-induced activation of α7 nAChRs (left open bars) in interneurones (‘intern.’) was significantly smaller than in PC12 cells. For NMDA responses (right bars), there was no significant differences in the PI between interneurones and pyramidal (‘pyramid.’) cells.
We also compared the Ca2+ permeability of the NMDA receptors in CA1 pyramidal neurones to that in interneurones. The PI of NMDA responses in these pyramidal neurones (63 ± 7 m C−1; n = 5 cells) was not significantly different from either NMDA receptors in interneurones, or the α7 nAChRs in PC12 cells (Fig. 5). Therefore, it appears as if the α7-containing nAChRs in rat hippocampal interneurones are significantly less permeable to Ca2+ than either NMDA receptors (in both interneurones and pyramidal cells) or α7 nAChRs in PC12 cells.
Discussion
In the hippocampus, nAChRs are linked to synaptic plasticity and cognitive function, which may be due in part to their ability to elicit local changes in cytoplasmic Ca2+([Ca2+]i) levels. Different subtypes of nAChRs are known to be differentially permeable to Ca2+, with the α7-containing nAChRs generally considered to be the most highly Ca2+ permeable (Fucile, 2004). Rat hippocampal CA1 interneurones are known to express both α7- and non-α7-containing nAChRs (Jones et al. 1999), and an understanding of the Ca2+ permeability of these receptors in the slice will help to elucidate any involvement of Ca2+-dependent signal transduction cascades in nAChR-mediated synaptic plasticity. In addition, these functional data are helpful in the understanding of the molecular makeup of these receptors in these neurones.
Combining patch-clamp electrophysiological recordings with conventional fura-2 imaging techniques to measure changes in [Ca2+]i in the soma of voltage-clamped neurones, we have estimated the relative Ca2+ permeability of α7-containing nAChRs in rat hippocampal CA1 interneurones and compared this with the NMDA subtype of ionotropic glutamate receptor channels. We compared the ratio of the increase in somal [Ca2+]i level (Δ[Ca2+]i) to the integrated transmembrane current (charge, Q) induced by the activation of these channels; we refer to this ratio as the Permeability Index (PI). This method should be more sensitive to estimating the actual amount of Ca2+ entering cells than by using the classical Goldmann-Hodgkin-Katz (GHK) constant field equations, which rely on measuring shifts in reversal potential due to changes in the concentrations of permeant ions (Fucile, 2004). In addition, the strong rectification for both α7 and non-α7 nAChR-mediated single-channel currents in excised outside-out patches from these interneurones, in which no outward current flow through these channels was observed at positive holding potentials (Shao & Yakel, 2000), prevents the accurate determination of the reversal potential. By combining fluorescence imaging with patch-clamp recordings, we found that the relative Ca2+ permeability of the α7-containing receptors in interneurones was about half that of the NMDA receptors. Furthermore, we did not detect any significant Ca2+ permeability of the non-α7 nAChRs, although it is possible that these receptors do have significant Ca2+ permeability (although significantly lower than α7 receptors; Fucile, 2004), which was lower than the detection limit of our recording system. Previously we did observe non-α7 nAChR-mediated single-channel currents from patches excised from the soma, albeit at a low density (i.e. much lower than α7 receptors; Shao & Yakel, 2000).
We considered whether the Ca2+ permeability of native α7 receptors in other cell types might be similar to that in rat hippocampal interneurones, or whether the Ca2+ permeability of the α7 receptors in interneurones might be lower than in other cell types. This is critical information since Ca2+ regulates a variety of signal transduction cascades and plays a key role in the short- and long-term regulation of nAChRs (Berg & Conroy, 2002; Quick & Lester, 2002). In PC12 cells, the Ca2+ permeability of native α7 nAChRs, based on its PI, was significantly larger than in hippocampal interneurones, and interestingly not significantly different from that for NMDA receptors in hippocampal interneurones. Furthermore, we compared the PI of NMDA in pyramidal cells to that in interneurones, and found that there was no significant difference. These data suggest that α7-containing nAChRs in rat hippocampal interneurones are significantly less permeable to Ca2+ than either NMDA receptors (in both interneurones and pyramidal cells) or α7 nAChRs in PC12 cells. Furthermore, the relative Ca2+ permeability of α7 receptors in PC12 cells is similar to NMDA receptors, an indication that it may not be the type of cells that can explain the lower Ca2+ permeability of α7 nAChRs in the interneurones. However, currently we cannot rule out the possibility that α7-containing receptors selectively in interneurones are closely linked with elements that bind Ca2+, and therefore hinder its access to fura-2.
Measuring fluorescence signals in the soma to estimate the relative Ca2+ influx for membrane-bound ion channels underestimates the true increases in [Ca2+]i near the mouth of the channel due to diffusion, buffering and pumps, among other factors (Berridge et al. 2003; Dajas-Bailador & Wonnacott, 2004). Therefore the influx of Ca2+ through the α7 receptors, which we have shown significantly increases somal [Ca2+]i, might be enough to regulate these channels either directly, or through Ca2+-dependent signal transduction cascades. We previously have shown that the rate of recovery from desensitization of these α7 receptors is regulated by [Ca2+]i (Khiroug et al. 2003). However, whether or not it is the Ca2+ influx directly through the channel or through another Ca2+-dependent mechanism is at present unknown. Besides regulating ion channel function and neuronal excitability, the influx of Ca2+ through the α7 receptors may also regulate other processes such as neurotransmitter release, gene expression, cell proliferation, survival and death, and development (Role & Berg, 1996; Dajas-Bailador & Wonnacott, 2004).
Although we have shown here that the increase in [Ca2+]i due to the activation of the α7 nAChRs was not due to calcium-induced calcium release (CICR) from internal stores, nor the activation of voltage-gated Ca2+ channels (VGCCs), both of these have previously been shown to participate in the nAChR-mediated increases in [Ca2+]i in native systems. For example in unclamped cells, the activation of nAChRs will depolarize the neurones and induce the activation of VGCCs, the activation of which will dramatically increase [Ca2+]i and augment the nAChR-mediated increase in [Ca2+]i (Dajas-Bailador & Wonnacott, 2004). However local [Ca2+]i increases through α7 nAChRs, rather than through VGCCs, may be more useful in regulating signal transduction cascades. For example in chick ciliary ganglion neurones, the activation of α7-containing nAChRs induces Ca2+ influx, which activates the CaMK and MAPK pathways, leading to activation of the transcription factor CREB and gene expression (Chang & Berg, 2001). This effect is blocked by VGCCs, the activation of which initially leads to CREB activation; however, it also stimulates the phosphatases calcineurin and PP1, which ultimately blocks CREB activation and gene expression. The nAChR-mediated Ca2+ influx has also previously been shown to induce CICR (Dajas-Bailador & Wonnacott, 2004). For example, glutamatergic release onto hippocampal CA3 pyramidal neurones is facilitated presynaptically by CICR induced by presynaptic nAChRs (Sharma & Vijayaraghavan, 2003). In mouse substantia nigra pars compacta neurones in the slice, the block of intracellular Ca2+ stores by dantrolene prevents the increase in [Ca2+]i due to the activation of α7-containing nAChRs (Tsuneki et al. 2000). Lastly, both CICR and VGCCs appear to contribute to the nAChR-mediated Ca2+ signals in SH-SY5Y cells (Dajas-Bailador et al. 2002).
Although the physiological significance of the Ca2+ influx and modulation of the α7-containing nAChRs in hippocampal interneurones remain to be determined, the lower relative Ca2+ permeability of these receptors than those in PC12 cells, or from a variety of heterologous expression systems (Fucile, 2004), may be a further indication of a difference in the properties, and perhaps molecular makeup, of these receptors. For example, for recombinant homomeric receptors expressed in Xenopus oocytes, the Ca2+ permeability of α7 receptors was greater than that for NMDA receptors (Séguéla et al. 1993). Interestingly Castro & Albuquerque (1995), who estimated and compared the relative Ca2+ permeability of native α7 nAChRs and NMDA receptors in cultured hippocampal neurones, estimated that the Ca2+ permeability of the α7 receptors was about 59% that of the NMDA receptors, a value comparable to our estimate of 46% reported here. Perhaps this lower Ca2+ permeability of the hippocampal α7 receptors may be due to differential modulation (Fucile, 2004). However, recently a new α7 subunit isoform was discovered, α7–2, which is expressed in both central and peripheral neurones (Severance et al. 2004). When expressed in Xenopus oocytes, this α7–2 subunit forms functional channels with slower desensitization kinetics, reversible block by α-bungarotoxin, and higher affinity for ACh than the original α7–1 subunit (Severance et al. 2004). However, it was not determined if the Ca2+ permeability of these receptors was altered.
Another possibility to explain functional diversity among α7 receptors, both native and expressed, is coassembly with other nAChR subunits. Although it had been previously thought that mammalian α7 subunits mostly form homomeric receptors (McGehee & Role, 1995; Chen & Patrick, 1997; Drisdel & Green, 2000), multiple functional subtypes of α7-containing nAChRs, with properties not identical to those expected of homomeric α7 receptors, have been reported in rat autonomic ganglia (Cuevas & Berg, 1998; Cuevas et al. 2000) and hippocampal interneurones (Shao & Yakel, 2000; Sudweeks & Yakel, 2000), suggesting the possibility of heteromeric α7-containing nAChRs. In particular, the native α7-containing receptors in the hippocampal interneurones desensitize more slowly, and have a smaller single-channel conductance in comparison to the properties of recombinant heterologously expressed receptors (Shao & Yakel, 2000; Sudweeks & Yakel, 2000). We previously found that the α7 and β2 subunits were coexpressed in individual rat hippocampal interneurones and were both correlated with fast-activating responses (Sudweeks & Yakel, 2000), and that the α7 and β2 subunits coassembled to form a functional heteromeric nAChR in heterologous expression systems (i.e. Xenopus oocytes and TSA120 cells; Khiroug et al. 2002). In the chick, the properties of native α7 nAChRs often do not match those of heterologously expressed homomeric α7 nAChRs, and α7-containing heteromeric chick nAChRs can be formed in heterologous expression systems (Anand et al. 1993; Yu & Role, 1998; Girod et al. 1999; Palma et al. 1999). PC12 cells express multiple diverse subtypes of α7-containing nAChRs, and single-cell RT-PCR analysis revealed that these cells express (in addition to the α7 subunit) α3, α4, α5 and β4 subunits (Virginio et al. 2002). Furthermore, the nAChR-mediated single-channel currents from outside-out patches in PC12 cells (Sands & Barish, 1992) do not correspond to the α7-containing nAChR single-channel currents in rat hippocampal neurones in slices (Shao & Yakel, 2000). Therefore, coassembly with other nAChR subunits, as well as other α7 subunit isoforms (including potential ones yet to be discovered), may help to explain the apparent lower Ca2+ permeability of α7 receptors in rat hippocampal interneurones.
Using the local photolysis of caged carbachol to activate nAChRs, we previously showed that the α7-containing nAChRs on these rat hippocampal interneurones were located primarily at the soma and proximal dendrites (i.e. < 70 μm from the soma), which was different from that of the ionotropic glutamate receptors (Khiroug et al. 2003). The glutamate uncaging-induced currents, including both NMDA and non-NMDA components, are expressed on the soma and along the entire dendritic field in these interneurones (Pettit & Augustine, 2000; Khiroug et al. 2003). Therefore, since the Ca2+ signal induced by the activation of α7-containing nAChRs will be through receptors located primarily on the soma, with little to no contribution from receptors on the distal dendrites, the lower relative Ca2+ permeability of α7 receptors in rat hippocampal interneurones is not likely to be due to the differential distribution of these receptors.
In conclusion, we have found that the relative Ca2+ permeability of α7-containing nAChRs in rat hippocampal interneurones is approximately half that of the NMDA subtype of glutamate receptors in these neurones and native α7-containing nAChRs in PC12 cells. Although the reasons for this unexpected lower Ca2+ permeability are at present unknown, this may indicate a differential subunit makeup (e.g. the functional expression and coassembly with other α7 subunit isoforms or non-α7 subunits), or regulation, of these receptors.
Acknowledgments
We would like to thank C. Erxleben and S. Dudek for advice in preparing the manuscript.
References
- Anand R, Peng X, Lindstrom J. Homomeric and native α7 acetylcholine receptors exhibit remarkably similar but non-identical pharmacological properties, suggesting that the native receptor is a heteromeric protein complex. FEBS Lett. 1993;327:241–246. doi: 10.1016/0014-5793(93)80177-v. [DOI] [PubMed] [Google Scholar]
- Berg DK, Conroy WG. Nicotinic α7 receptors: synaptic options and downstream signaling in neurons. J Neurobiol. 2002;53:512–523. doi: 10.1002/neu.10116. [DOI] [PubMed] [Google Scholar]
- Berridge MJ. Inositol trisphosphate and calcium signalling. Nature. 1993;361:315–325. doi: 10.1038/361315a0. [DOI] [PubMed] [Google Scholar]
- Berridge MJ, Bootman MD, Roderick HL. Calcium signalling: dynamics, homeostasis and remodeling. Nat Rev Mol Cell Biol. 2003;4:517–529. doi: 10.1038/nrm1155. [DOI] [PubMed] [Google Scholar]
- Bertrand D, Galzi JL, Bertrand S, Changeux J-P. Mutations at two distinct sites within the channel domain M2 alter calcium permeability of neuronal α7 nicotinic receptor. Proc Natl Acad Sci U S A. 1993;90:6971–6975. doi: 10.1073/pnas.90.15.6971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castro NG, Albuquerque EX. α-Bungarotoxin-sensitive hippocampal nicotinic receptor channel has a high calcium permeability. Biophys J. 1995;68:516–524. doi: 10.1016/S0006-3495(95)80213-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang KT, Berg DK. Voltage-gated channels block nicotinic regulation of CREB phosphorylation and gene expression in neurons. Neuron. 2001;32:855–865. doi: 10.1016/s0896-6273(01)00516-5. [DOI] [PubMed] [Google Scholar]
- Chen D, Patrick JW. The α-bungarotoxin-binding nicotinic acetylcholine receptor from rat brain contains only the α7 subunit. J Biol Chem. 1997;272:24024–24029. doi: 10.1074/jbc.272.38.24024. [DOI] [PubMed] [Google Scholar]
- Cuevas J, Berg DK. Mammalian nicotinic receptors with α7 subunits that slowly desensitize and rapidly recover from α-bungarotoxin blockade. J Neurosci. 1998;18:10335–10344. doi: 10.1523/JNEUROSCI.18-24-10335.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuevas J, Roth AL, Berg DK. Two distinct classes of functional α7-containing nicotinic receptor on rat superior cervical ganglion neurons. J Physiol. 2000;525:735–746. doi: 10.1111/j.1469-7793.2000.t01-1-00735.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dajas-Bailador FA, Mogg AJ, Wonnacott S. Intracellular Ca2+ signals evoked by stimulation of nicotinic acetylcholine receptors in SH-SY5Y cells: contribution of voltage-operated Ca2+ channels and Ca2+ stores. J Neurochem. 2002;81:606–614. doi: 10.1046/j.1471-4159.2002.00846.x. [DOI] [PubMed] [Google Scholar]
- Dajas-Bailador F, Wonnacott S. Nicotinic acetylcholine receptors and the regulation of neuronal signaling. Trends Pharmacol Sci. 2004;25:317–324. doi: 10.1016/j.tips.2004.04.006. [DOI] [PubMed] [Google Scholar]
- Drisdel RC, Green WN. Neuronal α-bungarotoxin receptors are α7 subunit homomers. J Neurosci. 2000;20:133–139. doi: 10.1523/JNEUROSCI.20-01-00133.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fayuk D, Yakel JL. Regulation of nicotinic acetylcholine receptor channel function by acetylcholinesterase inhibitors in rat hippocampal CA1 interneurons. Mol Pharmacol. 2004;66:658–666. doi: 10.1124/mol.104.000042. [DOI] [PubMed] [Google Scholar]
- Fucile S. Ca2+ permeability of nicotinic acetylcholine receptors. Cell Calcium. 2004;35:1–8. doi: 10.1016/j.ceca.2003.08.006. [DOI] [PubMed] [Google Scholar]
- Fujii S, Sumikawa K. Nicotine accelerates reversal of long-term potentiation and enhances long-term depression in the rat hippocampal CA1 region. Brain Res. 2001;894:340–346. doi: 10.1016/s0006-8993(01)02058-3. [DOI] [PubMed] [Google Scholar]
- Girod R, Crabtree G, Ernstrom G, Ramirez-Latorre J, McGehee D, Turner J, Role L. Heteromeric complexes of α5 and/or α7 subunits. Effects of calcium and potential role in nicotine-induced presynaptic facilitation. Ann NY Acad Sci. 1999;868:578–590. doi: 10.1111/j.1749-6632.1999.tb11331.x. [DOI] [PubMed] [Google Scholar]
- Gray R, Rajan AS, Radcliffe KA, Yakehiro M, Dani JA. Hippocampal synaptic transmission enhanced by low concentrations of nicotine. Nature. 1996;383:713–716. doi: 10.1038/383713a0. [DOI] [PubMed] [Google Scholar]
- Grynkiewicz G, Poenie M, Tsien RY. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985;260:3440–3450. [PubMed] [Google Scholar]
- Hunter BE, de Fiebre CM, Papke RL, Kem WR, Meyer EM. A novel nicotinic agonist facilitates induction of long-term potentiation in the rat hippocampus. Neurosci Lett. 1994;168:130–134. doi: 10.1016/0304-3940(94)90433-2. [DOI] [PubMed] [Google Scholar]
- Ji D, Lape R, Dani JA. Timing and location of nicotinic activity enhances or depresses hippocampal synaptic plasticity. Neuron. 2001;31:131–141. doi: 10.1016/s0896-6273(01)00332-4. [DOI] [PubMed] [Google Scholar]
- Jones S, Sudweeks S, Yakel JL. Nicotinic receptors in the brain: correlating physiology with function. Trends Neurosci. 1999;22:555–561. doi: 10.1016/s0166-2236(99)01471-x. [DOI] [PubMed] [Google Scholar]
- Khiroug L, Giniatullin R, Klein RC, Fayuk D, Yakel JL. Functional mapping and Ca2+ regulation of nicotinic acetylcholine receptor channels in rat hippocampal CA1 neurons. J Neurosci. 2003;23:9024–9031. doi: 10.1523/JNEUROSCI.23-27-09024.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khiroug SS, Harkness PC, Lamb PW, Sudweeks SN, Khiroug L, Millar NS, Yakel JL. Rat nicotinic ACh receptor α7 and β2 subunits co-assemble to form functional heteromeric nicotinic receptor channels. J Physiol. 2002;540:425–434. doi: 10.1113/jphysiol.2001.013847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin ED. Nicotinic receptor subtypes and cognitive function. J Neurobiol. 2002;53:633–640. doi: 10.1002/neu.10151. [DOI] [PubMed] [Google Scholar]
- Liu Q, Kawai H, Berg DK. β-Amyloid peptide blocks the response of α7-containing nicotinic receptors on hippocampal neurons. Proc Natl Acad Sci U S A. 2001;98:4734–4739. doi: 10.1073/pnas.081553598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGehee DS. Nicotinic receptors and hippocampal synaptic plasticity … it's all in the timing. Trends Neurosci. 2002;25:171–172. doi: 10.1016/s0166-2236(00)02127-5. [DOI] [PubMed] [Google Scholar]
- McGehee DS, Role LW. Physiological diversity of nicotinic acetylcholine receptors expressed by vertebrate neurons. Annu Rev Physiol. 1995;57:521–546. doi: 10.1146/annurev.ph.57.030195.002513. [DOI] [PubMed] [Google Scholar]
- McPherson PS, Kim YK, Valdivia H, Knudson CM, Takekura H, Franzini-Armstrong C, Coronado R, Campbell KP. The brain ryanodine receptor: a caffeine-sensitive calcium release channel. Neuron. 1991;7:17–25. doi: 10.1016/0896-6273(91)90070-g. [DOI] [PubMed] [Google Scholar]
- Palma E, Maggi L, Barabino B, Eusebi F, Ballivet M. Nicotinic acetylcholine receptors assembled from the α7 and β3 subunits. J Biol Chem. 1999;274:18335–18340. doi: 10.1074/jbc.274.26.18335. [DOI] [PubMed] [Google Scholar]
- Papke RL, Bencherif M, Lippiello P. An evaluation of neuronal nicotinic acetylcholine receptor activation by quaternary nitrogen compounds indicates that choline is selective for the α7 subtype. Neurosci Lett. 1996;213:201–204. doi: 10.1016/0304-3940(96)12889-5. [DOI] [PubMed] [Google Scholar]
- Pettit DL, Augustine GJ. Distribution of functional glutamate and GABA receptors on hippocampal pyramidal cells and interneurons. J Neurophysiol. 2000;84:28–38. doi: 10.1152/jn.2000.84.1.28. [DOI] [PubMed] [Google Scholar]
- Pettit DL, Shao Z, Yakel JL. β-Amyloid 1-42 peptide directly modulates nicotinic receptors in the rat hippocampal slice. J Neurosci. 2001;21:RC120. doi: 10.1523/JNEUROSCI.21-01-j0003.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quick MW, Lester RA. Desensitization of neuronal nicotinic receptors. J Neurobiol. 2002;53:457–478. doi: 10.1002/neu.10109. [DOI] [PubMed] [Google Scholar]
- Yu CR, Role LW. Functional contribution of the α7 subunit to multiple subtypes of nicotinic receptors in embryonic chick sympathetic neurones. J Physiol. 1998;509:651–665. doi: 10.1111/j.1469-7793.1998.651bm.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Role LW, Berg DK. Nicotinic receptors in the development and modulation of CNS synapses. Neuron. 1996;16:1077–1085. doi: 10.1016/s0896-6273(00)80134-8. [DOI] [PubMed] [Google Scholar]
- Sands SB, Barish ME. Neuronal nicotinic acetylcholine receptor currents in phaeochromocytoma (PC12) cells: dual mechanisms of rectification. J Physiol. 1992;447:467–487. doi: 10.1113/jphysiol.1992.sp019012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Séguéla P, Wadiche J, Dineley-Miller K, Dani JA, Patrick JW. Molecular cloning, functional properties, and distribution of rat brain α7: a nicotinic cation channel highly permeable to calcium. J Neurosci. 1993;13:596–604. doi: 10.1523/JNEUROSCI.13-02-00596.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidler NW, Jona I, Vegh M, Martonosi A. Cyclopiazonic acid is a specific inhibitor of the Ca2+-ATPase of sarcoplasmic reticulum. J Biol Chem. 1989;264:17816–17823. [PubMed] [Google Scholar]
- Severance EG, Zhang H, Cruz Y, Pakhlevaniants S, Hadley SH, Amin J, Wecker L, Reed C, Cuevas J. The α7 nicotinic acetylcholine receptor subunit exists in two isoforms that contribute to functional ligand-gated ion channels. Mol Pharmacol. 2004;66:420–429. doi: 10.1124/mol.104.000059. [DOI] [PubMed] [Google Scholar]
- Shao Z, Yakel JL. Single channel properties of neuronal nicotinic ACh receptors in stratum radiatum interneurons of rat hippocampal slices. J Physiol. 2000;527:507–513. doi: 10.1111/j.1469-7793.2000.00507.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma G, Vijayaraghavan S. Modulation of presynaptic store calcium induces release of glutamate and postsynaptic firing. Neuron. 2003;38:929–939. doi: 10.1016/s0896-6273(03)00322-2. [DOI] [PubMed] [Google Scholar]
- Sudweeks SN, Yakel JL. Functional and molecular characterization of neuronal nicotinic ACh receptors in rat CA1 hippocampal neurons. J Physiol. 2000;527:515–528. doi: 10.1111/j.1469-7793.2000.00515.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuneki H, Klink R, Léna C, Korn H, Changeux J-P. Calcium mobilization elicited by two types of nicotinic acetylcholine receptors in mouse substantia nigra pars compacta. Eur J Neurosci. 2000;12:2475–2485. doi: 10.1046/j.1460-9568.2000.00138.x. [DOI] [PubMed] [Google Scholar]
- Virginio C, Giacometti A, Aldegheri L, Rimland JM, Terstappen GC. Pharmacological properties of rat α7 nicotinic receptors expressed in native and recombinant cell systems. Eur J Pharmacol. 2002;445:153–161. doi: 10.1016/s0014-2999(02)01750-8. [DOI] [PubMed] [Google Scholar]
- Vogt KE, Regehr WG. Cholinergic modulation of excitatory synaptic transmission in the CA3 area of the hippocampus. J Neurosci. 2001;21:75–83. doi: 10.1523/JNEUROSCI.21-01-00075.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu CR, Role LW. Functional contribution of the alpha7 scubunit to multiple subtypes of nicotinic receptors in embryonic chick Sympathetic neurones. J Physiol. 1998;509:651–665. doi: 10.1111/j.1469-7793.1998.651bm.x. [DOI] [PMC free article] [PubMed] [Google Scholar]