Abstract
Loss of γ-aminobutyric acid (GABA)-mediated inhibition in the spinal cord is thought to mediate allodynia and spontaneous pain after nerve injury. Despite extensive investigation of GABA itself, relatively little is known about how nerve injury alters the receptors at which GABA acts. This study examined levels of GABAB receptor protein in the spinal cord dorsal horn, and in the L4 and L5 dorsal root ganglia (DRG) one to 18 weeks L5 spinal nerve ligation. Mechanical allodynia was maximal by 1 week and persisted at blunted levels for at least 18 weeks after injury. Spontaneous pain behaviors were evident for 6 weeks. Western blotting of dorsal horn detected two isoforms of the GABAB(1) subunit and a single GABAB(2) subunit. High levels of GABAB(1a) and low levels of GABAB(1b) protein were present in the DRG. However, GABAB(2) protein was not detected in the DRG, consistent with the proposed existence of an atypical receptor comprised of GABAB(1) homodimers. The levels of GABAB(1a), GABAB(1b), and GABAB(2) protein in the ipsilateral dorsal horn were unchanged at any time after injury. Immunohistochemical staining also did not detect a change in GABAB(1) or GABAB(2) subunits in dorsal horn segments having a robust loss of isolectin B4 staining. The levels of GABAB(1a) protein were also unchanged in the L4 or L5 DRG at any time after spinal nerve ligation. Levels of GABAB(2) remained undetectable. Finally, baclofen-stimulated binding of GTPγS in dorsal horn did not differ between sham and ligated rats. Collectively, these results argue that a loss of GABAB receptor-mediated inhibition, particularly of central terminals of primary afferents, is unlikely to mediate the development or maintenance of allodynia or spontaneous pain behaviors after spinal nerve injury.
Keywords: Neuropathic, Pain, Allodynia
List of Abbreviations: ANOVA: analysis of variance, DRG: dorsal root ganglia, GABA: γ-aminobutyric acid, L: lumbar
Neuropathic pain, characterized by mechanical allodynia, thermal hyperalgesia, and spontaneous pain behaviors, is a frequent consequence of peripheral nerve injury. The exact etiology of neuropathic pain remains elusive. However, changes in the expression or function of ion channels, neurotransmitter receptors, or transporters in the dorsal horn are each implicated in the development or maintenance of neuropathic pain (Luo et al., 2001, Luo et al., 2002, Sung et al., 2003, Yang et al., 2004). Likely mechanisms include changes in the connectivity, phenotype, and excitability of primary afferent neurons (Woolf et al., 1992, Noguchi et al., 1995, Malcangio et al., 2000, Fukuoka et al., 2001, Moore et al., 2002, Wu et al., 2002, Woolf, 2004). Transsynaptic changes in dorsal horn neurons may also contribute to an enhanced excitability (Coull et al., 2003).
One receptor-based mechanism that may contribute to nociceptive behaviors after nerve injury is a loss of inhibitory receptors on dorsal horn neurons or at the synapse between primary afferent neurons and dorsal horn neurons. γ-aminobutyric acid (GABA) is an important inhibitory neurotransmitter in the dorsal horn. It is particularly well situated to modulate the transmission of sensory information in the spinal cord through its actions at metabotropic GABAB receptors (reviewed by Hammond, 1997, Bowery et al., 2002, Bettler et al., 2004). GABAB receptors are present on large and small diameter primary afferent neurons, as well as on the somata of dorsal horn neurons (Price et al., 1984, Price et al., 1987, Calver et al., 2000, Yang et al., 2001, Yang et al., 2002, Castro et al., 2004). Agonist stimulation of GABAB receptors in the spinal cord decreases neurotransmitter release from primary afferent fibers (Kangrga et al., 1991, Marvizon et al., 1999, Ataka et al., 2000) and hyperpolarizes or inhibits the nociceptive responses of dorsal horn neurons (Kangrga et al., 1991, Yang et al., 2001, Sokal and Chapman, 2003). Indeed, antagonism of spinal GABAB receptors by intrathecal injection of CGP35348 produces hyperalgesia and facilitates dorsal horn neuronal responses to noxious stimuli (Hao et al., 1994, Sokal and Chapman, 2001). Furthermore, GABAB(1) and GABAB(2) knockout mice exhibit hyperalgesia (Schuler et al., 2001, Gassmann et al., 2004). It is therefore possible that a loss of GABAB receptors could contribute to the development or maintenance of the allodynia, hyperalgesia and spontaneous pain behaviors that occur after peripheral nerve injury.
The GABAB receptor is an obligate heterodimeric receptor composed of a single GABAB(1) subunit and a single GABAB(2) subunit (Kaupmann et al., 1997, Kaupmann et al., 1998, Benke et al., 1999, Bowery et al., 2002, Bettler et al., 2004). The GABAB(1) subunit is responsible for ligand binding (Kaupmann et al., 1997), while the GABAB(2) subunit is responsible for intracellular signaling via G-proteins (Kaupmann et al., 1998, Bowery et al., 2002, Bettler et al., 2004). Loss of the GABAB(1) or the GABAB(2) subunit results in a loss of ligand binding, GTPγS coupling, and a decreased expression of the complementary subunit (Schuler et al., 2001, Gassmann et al., 2004). This study was undertaken to determine whether peripheral nerve injury alters the levels of either GABAB receptor subunit in the dorsal root ganglia (DRG) or the dorsal horn of the spinal cord. It also examined whether a temporal relationship exists between changes in the expression of these receptor subunits and the development or maintenance of neuropathic pain behaviors. Western blotting and immunohistochemistry were used to assess changes in expression of the GABAB(1) and GABAB(2) subunits in the dorsal horn and the L4 and L5 DRG of rats 1 week to 3 months after sham surgery or tight ligation of the L5 spinal nerve. Baclofen-induced stimulation of GTPγS binding was used to assess changes in receptor function in the dorsal horn 3 weeks after sham surgery or spinal nerve ligation.
EXPERIMENTAL PROCEDURES
Animals
Male Charles River Sprague-Dawley rats (Sasco stock; Charles River, Portage, IN) were housed under a reversed 12 h light cycle with lights off at 9:00 am. Rats were housed in pairs with water and soy-free food (Harlan Teklad 1026; Madison, WI) provided ad libitum. All animals were maintained for at least 7 days on the new diet and light conditions before start of the study. All procedures were conducted in accordance with the Guide for Care and Use of Laboratory Animals published by the National Institutes of Health and the ethical guidelines of the International Association for the Study of Pain. These experiments were approved by The University of Iowa Animal Care and Use Committee.
Surgical Procedure and Behavioral Testing
The L5 spinal nerve was ligated as described by Kim and Chung (Kim and Chung, 1992, Choi et al., 1994). Briefly, rats were anesthetized with halothane and the ipsilateral L6 transverse process was removed using aseptic surgical technique. The L5 spinal nerve was then ligated with 6-0 silk. Cohorts of rats underwent a sham surgery wherein the L5 spinal nerve was exposed but not ligated. Muscle and fascia were closed in layers using vicryl sutures, and the skin was closed with wound clips. Animals were randomly allocated to one of six different survival time points for Western blotting experiments (N=12/group/condition) or a two week survival time for immunohistochemical experiments (N=3/condition). Thus, rats were tested on only one occasion in this study. Rats that exhibited foot drop or other gross neurological deficits were excluded from the study.
On the scheduled day of testing, animals were acclimated to the testing room for 30 min before being removed from their cages. The treatment status of the rats was unknown to the tester. Spontaneous pain behaviors, mechanical allodynia and thermal hyperalgesia were assessed successively. At least 20 min elapsed between the different tests. To assess spontaneous pain behaviors, rats were individually placed on a 30°C copper plate and allowed to acclimate for 5 min. The number and duration of ipsilateral hindpaw lifts was then recorded for 10 min. Lifts associated with repositioning or locomotion were not counted. To assess mechanical allodynia, rats were placed on an elevated mesh surface and allowed to acclimate for 15 min. Monofilaments of increasing force (Smith & Nephew, Germantown, WI) were sequentially applied to the webbing between the 3rd and 4th digits of the hindpaw. Paw withdrawal threshold was determined using the Up-and-Down method of Dixon as described by Chaplan et al. (1994). The maximum filament used was 28.8 g. Rats that did not respond to this filament were assigned this value. To assess thermal hyperalgesia, rats were placed on a 25°C glass surface and allowed to acclimate for 15 min. A radiant thermal stimulus was then applied to the plantar surface of the hindpaw and time to paw withdrawal reflex was recorded electronically. A 20 sec cutoff time was imposed to prevent thermal injury. Rats that did not respond by 20 sec were assigned this value.
Western Blotting
Twenty-four hours after behavioral testing was conducted, the rats were deeply anesthetized with pentobarbital (75 mg/kg i.p.), and the L4–L5 section of the spinal cord, the ipsilateral L4 DRG, and the ipsilateral L5 DRG were removed. The spinal cord was sectioned into quadrants to isolate the ipsilateral and contralateral dorsal horns. All tissues were individually frozen on dry ice and stored at −70°C. Rats were selected on the basis that their mechanical withdrawal threshold corresponded to or closely approximated the median values for a particular time point.
Spinal cord sections were homogenized in an excess of ice-cold homogenization buffer (5 mM Tris pH 7.4; 0.32 M sucrose) with protease inhibitors (Roche Complete Mini protease cocktail; Indianpolis, IN) using a polytron (Tissue-Tearor). The solution was centrifuged at 1,000 g for 15 min at 4°C, and the supernatant was then centrifuged at 15,000 g for 30 min at 4°C. The resulting pellet (P2) was resuspended in homogenization buffer with protease inhibitors and the protein concentration was determined using a Lowry Assay (Bio-Rad; Hercules, CA) Thirty micrograms of protein was then added to an equal volume of sample buffer (125 mM Tris-HCl pH 6.8; 20% glycerol; 4% SDS; 5% β-mercaptoethanol; 0.02% bromophenol blue). The samples were not heated.
Two DRG were pooled for each lane and homogenized in ice-cold detergent homogenization buffer (50 mM Tris pH 8; 150 mM NaCl; 1% NP-40; 0.5% sodium deoxycholate; 0.1% sodium dodecylsulfate) with protease inhibitors (Roche Complete Mini protease cocktail) using motorized plastic Eppendorf tube pestles in a volume of 35 μl. The solution sat on ice for 30 min and then was centrifuged at 5,000 g for 15 min at 4°C. The resulting supernatant was then added to an equal volume of sample buffer. The samples were not heated.
Samples were separated on an 8% SDS-PAGE gel and transferred to PVDF membrane. The membrane was then blocked for 2 h in 3% bovine serum albumin in Tris buffered saline pH 7.4. The primary antibodies were then diluted in blocking solution and placed on the membrane overnight at 4°C. Rabbit polyclonal GABAB(1) antiserum (Ab25; generated against a glutathione-S-transferase fusion protein containing carboxyterminal residues R857-K960 of rat GABAB(1) protein) was diluted 1:3,000. The GABAB(2) antibody was purchased from Chemicon (AB5394; Temecula, CA) and diluted 1:5,000. The membranes were then incubated with appropriate horseradish peroxidase-linked secondary antibodies (Jackson ImmunoResearch, West Grove, PA) diluted 1:50,000 in 5% milk for 2 h at room temperature. Following thorough washing, the membranes were developed with a Pierce SuperSignal West Femto Kit according to manufacturer’s instructions. Images were acquired and analyzed using a Kodak Image Station. Multiple exposure times were acquired to ensure signals were not saturated. Expression levels for GABAB(1a), GABAB(1b), and GABAB(2) were normalized to values obtained for an aliquot of the same tissue from naïve rats, which were loaded on each gel as a control. The transferrin receptor (TFR12-M; Alpha Diagnostics, San Antonio, TX) was used as a loading control for the dorsal horn because actin was not detected in P2 membranes prepared with the Tissue-Tearor. The transferrin receptor was selected because it is a membrane bound receptor with a similar molecular mass as GABAB receptor subunits and, because of its involvement in iron homeostasis, is unlikely to be altered by spinal nerve ligation. Actin (MAB1501; Chemicon) was used as a loading control for the DRG.
For Western blotting, the specificity of the antisera was established by the absence of the 125 and 105 kDa bands when brain or spinal cord tissues from GABAB(1) knock-out mice were probed with the GABAB(1) antibody (Fig. 1A), and by the absence of the 115 kDa band when brain or spinal cord tissues from GABAB(2) knock-out mice were probed with the GABAB(2) antibody (Fig. 1B). The specificity of the GABAB(2) subunit antibody was additionally established in the rat by the absence of bands in dorsal horn tissue when the antibody was preabsorbed for 2 h at room temperature with a 10-fold excess of the cognate peptide (TASPRHRHVPPSFRVMVSGL) (Fig. 1C).
Fig. 1.
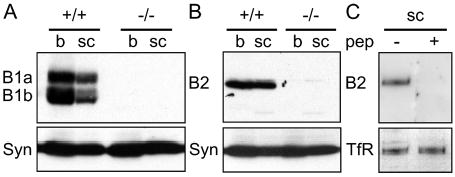
Specificity of antibodies for the GABAB(1) and GABAB(2) subunits was confirmed by western blotting membrane homogenates prepared from the brain (b) and spinal cord (sc) of GABAB(1) and GABAB(2) knock-out mice or wild-type mice. (A) Antibody to the GABAB(1) subunit detected two bands of 125 and 105 kDa in both the brain and spinal cord of wild-type (+/+) mice. These bands were absent in tissue from GABAB(1) knock-out mice (−/−). (B) Antibody to the GABAB(2) subunit detected a single band of 115 kDa in both brain and spinal cord of wild-type (+/+) mice. This band was absent in tissue from GABAB(2) (−/ −) knock-out mice. (C) The antibody to GABAB(2) also detected a band of 115 kDa molecular weight in homogenates of rat spinal cord. Preabsorption of the antibody with a 10-fold excess of cognate peptide eliminated this band. Uniform loading of protein was confirmed by probing membranes with antibodies to either syntaxin (syn) or the transferrin receptor (TfR).
Immunohistochemistry
Rats were deeply anesthetized with sodium pentobarbital (75 mg/kg i.p.) and transcardially perfused with 100 ml of 0.05 M phosphate buffered saline (PBS) pH 7.4 followed by 500 ml of 4% paraformaldehyde in 0.1 M phosphate buffer pH 7.4. The spinal cords were removed and cryoprotected in 0.1 M phosphate buffer (PB) containing 30% sucrose. The spinal cord was sectioned into sets of three serial sections each of 50-μm thickness beginning at L3 and ending at the L6. The first section in each set was processed for GABAB(1) , the third section for isolectin B4 (from Griffonia simplicifolia) binding, and the final section for GABAB(2). The sections stained for GABAB receptor subunits were incubated in 0.1 M PBS containing 10% normal goat serum containing 0.3% Triton X-100 and 0.1 M glycine for 2 h. The sections stained for IB4 were incubated in 100% methanol for 20 min followed by 1% sodium borohydride for 30 min. These sections were then incubated in 6% normal goat serum containing 0.3% Triton X-100. Respective sections were then incubated for 40 h at 4°C in rabbit anti-GABAB(1) (Ab25; 1:2,000), IB4-Biotin (1:8,000; Vector, Burlingame, CA) or guinea pig anti-GABAB(2) (1:2,000; Chemicon, Temecula, CA). The diluent for the antibodies was 0.1 M PBS with the respective blocking and permeabilizing agents. The sections were then washed with 0.1 M PBS and incubated for 1 h at room temperature in Cy3 goat anti-rabbit antiserum (1:200; Jackson ImmunoResearch, West Grove, PA), Cy3 conjugated streptavidin (1:200; Jackson ImmunoResearch), or Cy3 goat anti-guinea pig antiserum (1:200; Jackson ImmunoResearch), respectively. Sections were then washed in 0.1 M PBS, mounted on slides and dried overnight at room temperature. The slides were then cleared in xylenes and coverslipped with DPX. The immunohistochemical specificity of the GABAB(1) antisera was established by the absence of staining after omission of either the primary or secondary antibody and by a loss of staining in GABAB(1) knock-out mice. Briefly, BALB/c wildtype or GABAB(1) knockout mice were euthanized and the vertebral columns were removed and immersion fixed in 4% paraformaldehyde for 6 h. After cryoprotection in 30% sucrose, spinal cords were removed, sectioned on a cryostat and processed as described above. Figure 2 illustrates that the intensity of neuropil labeling in GABAB(1) knockout mice was greatly reduced in the dorsal horn (Fig. 2A, B), and there was a marked loss of neuronal staining in both the deep dorsal horn and in the ventral horn (Fig.2 C,D) compared to wildtype mice. The distribution and intensity of staining in the knockout mouse was not different from that obtained when the primary antibody was omitted, and likely reflects nonspecific staining of the secondary antibody at the concentration used. The specificity of the GABAB(2) antibody was established by the loss of staining in rat spinal cord when the primary or secondary antibody was omitted or when the primary antibody was preabsorbed for 2 h at room temperature with a 10-fold excess of cognate peptide (Fig. 2 E,F).
Fig. 2.
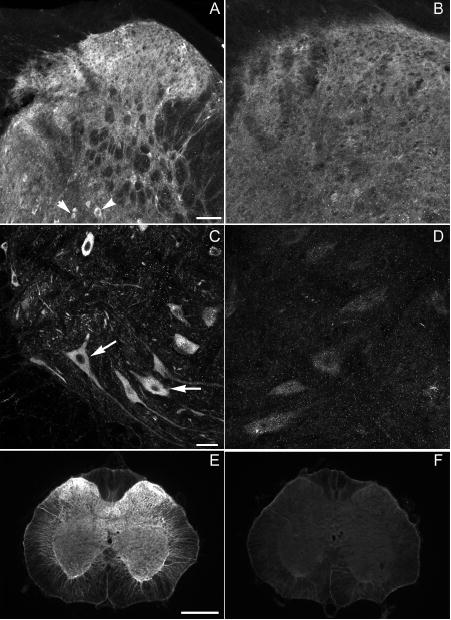
Specificity of antibsera to GABAB(1) and GABAB(2). Distribution of GABAB(1) immunoreactivity in (A,C) a wildtype BALB/c mouse and (B,D) a GABAB(1) knockout mouse. Note the dense labeling of neuropil throughout the dorsal horn (A), in addition to labeling of neurons in the deep dorsal horn (A, arrowheads) and ventral horn (C, arrow) of the wildtype mouse. Labeling in the dorsal (B) and ventral (D) horns of the knockout mouse is greatly decreased and comparable to that obtained when the primary antibody was omitted. (E) Distribution of GABAB(2) immunoreactivity in the neuropil of the spinal cord of the rat . Note the absence of somal labeling. Preabsorption of the antibody eliminated staining (F). Scale bars are 50 μm for A and B, 20 μm for C and D, and 500 μm for E, F.
Images of immunostaining in rat spinal cord were obtained using a SpotRT camera, Metamorph software, and a Nikon E800 epifluorescence microscope equipped with a narrow bandwidth Cy3 filter (Chroma Technology, Rockingham, VT). For each label, the same exposure times, gain and binning parameters were used to obtain images from sham-surgery and ligated rats. Images were exported to Photoshop for assembly into a figure; no changes in contrast, levels or brightness were made. Images of immunostaining in mouse spinal cord were obtained using a Zeiss LSM 510 with identical detector gain, amplifier offset, and amplifier gain conditions between conditions. These images were exported to Photoshop and converted to black and white. Changes in levels were equally applied to all images.
To quantify the intensity of labeling, sets of three serial sections were obtained at fixed intervals from spinal cord segments L3 through S1 from three sham-operated and three ligated rats. The tissue was concurrently processed for GABAB(1)- and GABAB(2)-like immunoreactivity, as well as IB4 staining. For quantitation, three sections in the L5 spinal cord with near complete loss of IB4 staining were selected from each of three ligated rats, along with the adjacent sections that were processed for GABAB(1) and GABAB(2)-like immunoreactivity. Three sets of sections in the L5 spinal cord were selected in each of three sham-operated rats. For each section, six imaging frames (99 μm wide x 113 μm high) were aligned across the medial-lateral extent of the superficial dorsal horn. These spanned the entirety of laminae I and 2. Five imaging frames were aligned immediately ventral to the first set of frames, corresponding to lamina 3. Lamina 2 and 3 were demarcated by IB4 staining. Another three imaging frames were situated in the dorsal column to determine background labeling. The average pixel intensity in each frame was measured using Metamorph software (Molecular Devices, Sunnyvale, CA). Values for the three imaging frames in the dorsal column were averaged to determine background labeling and subtracted from values determined for frames in the dorsal horn. The average pixel intensity of each of the six frames spanning laminae I and 2 were then averaged to yield a single value for that section for each rat. The same was done for the five frames in lamina 3. The intensity of staining for GABAB(1) and GABAB(2) , as well as IB4 did not differ across the medial-lateral aspect of either laminae 1 and 2 or lamina 3 (P>0.6 for all labels and regions). Therefore, the values for the six frames in laminae 1 and 2 and the five frames in lamina 3 were averaged to yield a single value for that region of that section. Average pixel intensity determined in the three sections were then calculated to yield a single value for laminae 1 and 2 and for lamina 3 for each rat. The mean and S.E.M. pixel intensity for each region in each treatment group was calculated from these values.
GTPγS Binding
Three weeks after either sham surgery or SNL, rats were euthanized by CO2 inhalation. The L4 and L5 segment of the spinal cord was rapidly removed to a ice-cold surface, divided into ipsilateral and contralateral halves, and then further divided into dorsal and ventral quadrants. The ipsilateral dorsal quadrant from each rat was homogenized in ice-cold membrane buffer (50 mM Tris pH 7.4, 5 mM MgCl2, 1 mM EGTA) using Potter-Elvehjem homogenization tubes and pestles. The homogenate was spun at 39,000 g at 4°C for 15 min, the supernatant was discarded and the pellet was resuspended in assay buffer (50 mM Tris pH 7.4, 5 mM MgCl2, 1 mM EGTA, 100 mM NaCl) and spun once again at 39,000 g at 4°C for 15 min. The resultant pellet was then resuspended in ice-cold assay buffer, an aliquot was taken for determination of protein content, and the membranes stored at −70°C until use.
Membrane homogenates (30–60 μg/tube) were incubated at 25°C for 2 hr in 1 ml of assay buffer with 0.1 μM – 1 mM (+)baclofen hydrochloride (Sigma, St. Louis, MO catalog number G013), 30 μM GDP and 50 pM [35S]GTPγS (specific activity, 1000 Ci/mmole; Amersham, Piscataway, NJ). The incubation mixture also contained adenosine deaminase (13.7 mU/ml) to eliminate adenosine-mediated stimulation of GTPγS binding. The reaction was terminated by filtration using a Brandel cell harvester and Whatman GF/B glass filters presoaked in 50 mM Tris-HCl (pH 7.4) and 5 mM MgCl2 at 4°C for 2 h. Filters were then washed 3 times with 5 ml of ice-cold Tris-HCl buffer (pH 7.4), transferred to scintillation counting vials containing Scintisafe Econo1 scintillation fluid, and equilibrated for 12 h. The radioactivity in the samples was determined with a liquid scintillation analyzer. Non-specific binding was measured in the presence of 10 μM unlabeled GTPγS. Tissue samples were analyzed in duplicate. Binding isotherms were fit with Graphpad Prism. Data were expressed as percent stimulation over control [(specific binding in presence of baclofen- specific binding in absence of baclofen)/specific binding in absence of baclofen) x 100)]. Data are expressed as the mean and S.E.M. of determinations in three ligated and three sham rats.
Statistical Analysis
Mechanical thresholds were analyzed with the non-parametric Mann-Whitney Rank Sum test to compare values between treatment groups at each time point and by Kruskal-Wallis for comparisons across all time points within a treatment group. Dunn’s test was used for post-hoc comparisons. Spontaneous hindpaw lifts in sham-operated and ligated rats were compared using the Mann-Whitney Rank Sum Test at each time point. As spontaneous pain behaviors could only increase and not decrease, a one-tailed test was applied in this one instance. Western blot band intensities were normally distributed and were analyzed using two-way ANOVA. Student’s t-tests were used to compare Emax and EC50 values in sham and ligated rats in the GTPγS binding assays, and also to compare the intensity of immunofluorescence in the dorsal horn of sham and ligated rats. For all statistical tests, a P<0.05 was significant.
RESULTS
L5 Spinal Nerve Ligation Causes Mechanical Allodynia and Spontaneous Pain Behaviors
Tight ligation of the L5 spinal nerve produced a rapid and robust decrease in mechanical withdrawal thresholds of the ipsilateral hindpaw within 1 week of ligation that persisted for the duration of the study (Fig. 3A). The reduction in threshold was maximal for post-operative weeks 1–3 with an attenuation occurring at post-operative week six that persisted through week 18 (P<0.05). Withdrawal thresholds for the ipsilateral hindpaw in sham-operated rats or the contralateral hindpaw in ligated rats were unchanged at all time points (P>0.4 and P>0.3, respectively).
Fig. 3.
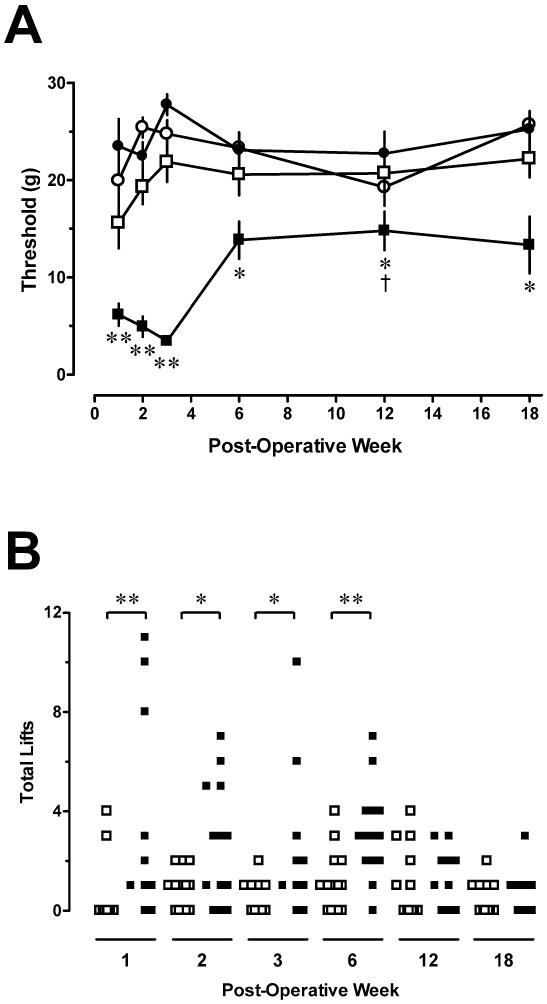
L5 spinal nerve ligation produces time-dependent mechanical allodynia and increase in spontaneous pain behaviors. Panel A illustrates withdrawal threshold to stimulation of the hindpaw with von Frey filaments. Open squares: Ipsilateral hindpaw of sham-operated rats; Solid squares: Ipsilateral hindpaw of ligated rats; Solid circles: contralateral hindpaw of ligated rats; Open circles: Contralateral hindpaw of sham rats. Ordinate: Withdrawal threshold (g). Abscissa: Time after surgery (weeks). Symbols represent mean ± S.E. of determinations in 10–13 rats. *P< 0.05, **P< 0.01 compared to sham-operated rats at the same time point; two-tailed Mann-Whitney test. †P< 0.05 compared to week 1 within the ligated treatment group; Kruskal-Wallis ANOVA. Panel B is a scatter plot of spontaneous pain behaviors exhibited by each animal. Ordinate: Total number of ipsilateral hindpaw lifts during 10 min on a 30ºC copper plate. Abscissa: Time after surgery (weeks). Open squares: Ipsilateral hindpaw of sham-operated rats; Solid squares: Ipsilateral hindpaw of ligated rats. * P<0.05, ** P<0.025, one-tailed Mann-Whitney test.
Rats that underwent L5 spinal nerve ligation also exhibited spontaneous pain behaviors (Fig. 3B). The total number of lifts, including sustained lifts and transient flinches of the ipsilateral hindpaw, was significantly greater in ligated rats than in sham rats during post-operative weeks 1 through 6 (P<0.05). Spontaneous pain behaviors subsided considerably by post-operative weeks 12–18 where ligated and sham operated rats did not differ in number of hindpaw lifts. No significant correlation was found between the magnitude of spontaneous pain behaviors and the degree of mechanical allodynia for the ligated paw at any time point.
Significant thermal hyperalgesia was not observed in rats with an L5 spinal nerve ligation. Paw withdrawal latencies in ligated rats did not differ from those in sham-operated rats in either hindpaw at any time point (P>0.14; data not shown). For example, withdrawal latency of the ipsilateral hindpaw in ligated rats was 9.8 ± 0.4 s two weeks after injury vs. 10.7 ± 0.5 s in sham-operated rats.
L5 Spinal Nerve Ligation Does Not Alter GABAB Receptor Subunit Levels in the Spinal Cord Dorsal Horn: Western Blotting
The levels of GABAB(1a), GABAB(1b), or GABAB(2) protein in rats that underwent L5 spinal nerve ligation were unchanged relative to sham animals at all time points (Fig. 4 and 5 A–C). In addition, the levels of the three receptor subunits did not correlate with either spontaneous pain behaviors or decreases in mechanical withdrawal thresholds. Although levels of GABAB(1a) and GABAB(1b) protein appeared to decrease in both treatment groups between 6 to 18 weeks, this trend was not statistically significant. In addition, the ratio of GABAB(1a) to GABAB(1b) was not significantly different as a function of treatment at any time point (Fig. 5D). Although the GABAB(1) antibody used in this study recognizes an identical sequence in the C-terminus of both GABAB(1) isoforms, the affinities of the antibodies to each isoform are not known to be equal. Therefore, a GABAB(1a) to GABAB(1b) ratio of 1 does not necessarily indicate an equimolar ratio of the two subunits. Additionally, the ratio of GABAB(1a) or GABAB(1b) to GABAB(2) was not significantly different as a function of treatment at any time point. Finally, levels of the GABAB(1a), GABAB(1b), and GABAB(2) subunits in the ipsilateral dorsal horn of ligated rats were not statistically different from those in the contralateral dorsal horn (data not shown). Power analysis demonstrated sufficient sensitivity to detect a 40% change in subunit expression levels with an N of 4–6/group. This analysis is congruent with a pilot study that demonstrated that Western blotting could readily detect a change of ~40% in levels of GABAB subunit protein applied as serial dilutions of dorsal horn tissue ranging from 10 to 40 μg of protein (data not shown). Finally, pilot studies showed that Western blotting methods detected a decrease in levels of GABAB(1a), GABAB(1b), and GABAB(2) subunit protein to 38% (P<0.05), 39% (P<0.01) and 62% (P<0.01) of values in sham-operated rats after rhizotomy of the L3, L4 and L5 dorsal roots.
Fig. 4.
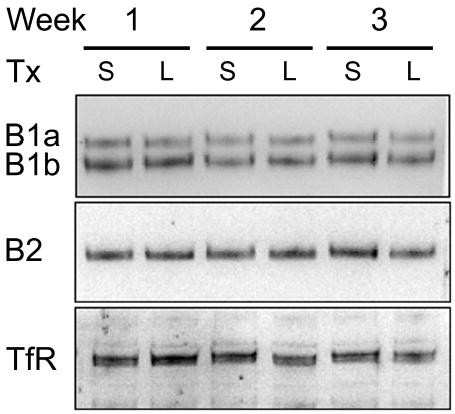
Representative Western blots of GABAB(1a) and GABAB(1b) and GABAB(2) protein in membrane homogenates of ipsilateral dorsal horn of individual rats that underwent either L5 spinal nerve ligation (L) or sham surgery (S) 1, 2, or 3 weeks earlier. Thirty μg of protein from the P2 pellet was loaded in each lane. Membranes were stripped and reprobed for the transferrin receptor (TfR) to establish uniform loading.
Fig. 5.
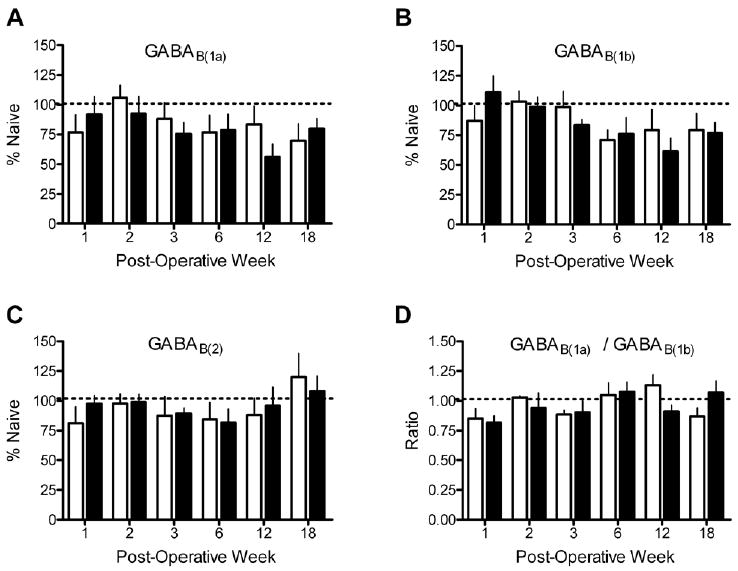
Spinal nerve ligation (filled bars) did not produce a significant change in levels of (A) GABAB(1a), (B) GABAB(1b) or (C) GABAB(2) protein in the ipsilateral L4–L5 dorsal horn compared to sham-operated rats (open bars) at times ranging from 1 to 18 weeks after surgery. Spinal nerve ligation also did not change the ratio of GABAB(1a) to GABAB(1b) (D). Band intensities for each isoforms were normalized to a sample from a naïve rat that was run on the gel. N = 4–6 for each treatment condition and time point.
L5 Spinal Nerve Ligation Does Not Alter GABAB Receptor Subunit Levels in the L4 or L5 DRG: Western Blotting
GABAB(1a) was the predominant isoform of the GABAB(1) subunit in DRG (Fig. 6A,B). Levels of this subunit were not significantly changed as a function of treatment or time after surgery in either the L4 (Fig. 6C) or the L5 (Fig. 6D) DRG. Levels of GABAB(1b) and GABAB(2) were not sufficient for quantitation even when as many as six DRG were pooled.
Fig. 6.
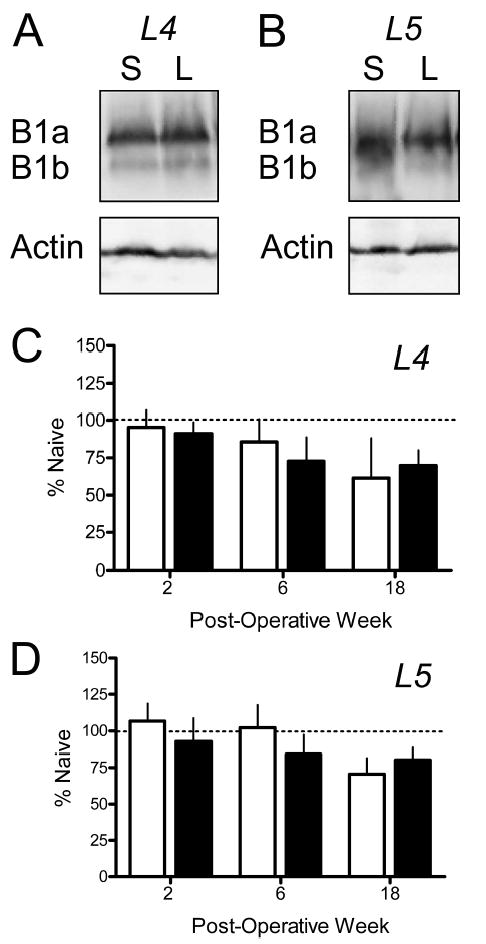
Lack of effect of spinal nerve ligation on levels of GABAB receptor subunits in the L4 and L5 DRG. Panels A and B respectively illustrate representative Western blots of whole cell lysates from L4 and L5 DRG obtained two weeks after ligation of the L5 spinal nerve (L) or sham surgery (S). Each lane contains lysates from two DRG. Note that the predominant isoform is GABAB(1a). Neither the GABAB(1b) nor the GABAB(2) subunits were present in quantifiable amounts. Panels C and D respectively illustrate mean data obtained for the L4 and L5 DRG of rats 2, 6 or 18 weeks after surgery. Abscissa: Time after surgery in weeks. Ordinate: Levels of subunit protein normalized to levels in DRG of naïve rats that were run on each blot. Open bars, Sham; Filled bars, Ligated. N = 4 for treatment condition and time point.
L5 Spinal Nerve Ligation Does Not Alter GABAB Receptor Subunit Levels in the Spinal Cord Dorsal Horn: Immunohistochemistry
As expected, high levels of immunoreactivity for both the GABAB(1) and GABAB(2) subunits were present in the dorsal horn of the spinal cord (Fig. 7). Isolectin B staining was confined to lamina II, consistent with its use as a marker for the terminals of C-fiber afferents (Ambalavanar and Morris, 1993). Two weeks after spinal nerve ligation, when mechanical allodynia was maximal, immunoreactivity for GABAB(1) (Fig. 7A) or GABAB(2) (Fig. 7C) subunits was unchanged as compared to sham-surgery rats (Fig. 7D and G, respectively) even at levels of the spinal cord where a near complete loss of the IB4 staining (Fig. 7B vs 7E) was evident. Table 1 quantifies the intensity of GABAB(1) and GABAB(2) immunoreactivity in regions of the lumbar dorsal cord where IB4 staining was significantly decreased.
Fig. 7.
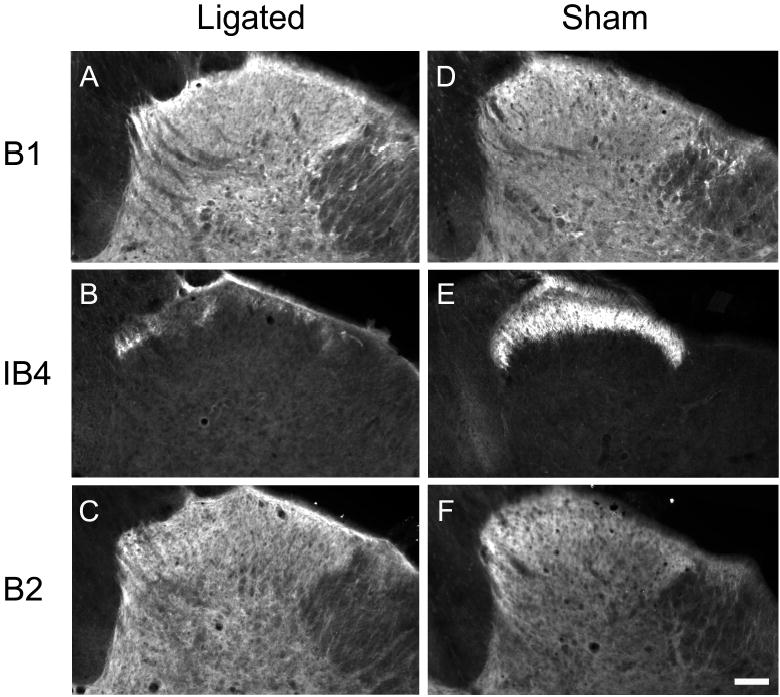
Spinal nerve ligation does not alter GABAB(1) or GABAB(2) subunit staining in the ipsilateral dorsal horn of the L5 spinal cord. Serial sections from both ligated and sham operated rats were processed for GABAB(1) (A,D), Isolectin B4 (B, E), and GABAB(2) (C,F). Isolectin B4 (IB4) staining was greatly reduced in ligated but not sham-operated tissue. Panel B indicates that the serial sections from this ligated spinal cord were taken from the region of greatest Ioss of IB4 staining.
Table 1.
Average pixel intensities for GABAB(1)- or GABAB(2)-immunoreactivity and IB4-staining in the ipsilateral dorsal horn 3 weeks after sham-surgery or spinal nerve ligation
| GABAB(1) | GABAB(2) | IB4 | ||||
|---|---|---|---|---|---|---|
| Ligated | Sham | Ligated | Sham | Ligated | Sham | |
| Laminae 1–2 | 97.7 ± 11.1 | 105.6 ± 15.3 | 94.0 ± 6.8 | 113.6 ± 23.0 | 14.8 ± 3.6** | 66.6 ± 5.1 |
| Lamina 3 | 66.9 ± 8.4 | 75.9 ± 12.9 | 77.3 ± 16.2 | 80.0 ± 23.4 | 1.5 ± 1.4 | 3.5 ± 1.3 |
Data are mean ± S.E.M of determinations in three sections obtained at the level of greatest IB4 loss in each of three ligated rats, and of three sections obtained at the corresponding level in three sham-operated rats.
P<0.01, t-test.
L5 Spinal Nerve Ligation Does Not Alter Baclofen-Stimulated GTPγS Binding
As previously reported (Sands et al., 2004), the maximal stimulation of GTPγS binding produced by 1 mM baclofen was on the order of 25% in the adult rat. No difference was observed in either the efficacy or potency of baclofen in ligated rats compared to sham-operated rats. The Emax for baclofen-induced stimulation of GTPγS binding in sham rats was 25.1 ± 4.4% vs. 21.4 ± 1.7% in ligated rats (P > 0.4, Students t-test). The EC50 values were 16.8 ± 6.5 μM in sham rats and 7.3 ± 3.8 μM in ligated rats (P > 0.2, Students t-test). In view of the lack of effect at a time when mechanical allodynia was maximal, additional time points were not examined.
DISCUSSION
At the outset of this study, published studies suggested that GABAB receptors in the dorsal horn would either be unchanged or decreased after peripheral nerve injury. Sciatic axotomy was reported to decrease GABAB mRNA levels in the spinal cord dorsal horn by 60% (Yang et al., 2004) and to decrease the number of GABAB receptors in lamina II by 30% (Castro-Lopes et al., 1995). However, levels of mRNA for the GABAB(1) subunit were reported to be unchanged in the DRG after sciatic axotomy (Xiao et al., 2002), and neither the Bmax nor the Kd of GABAB receptors in lamina II were altered after chronic constriction of the sciatic nerve (Smith et al., 1994). However, each of these studies has limitations. Levels of GABAB receptor mRNA may or may not be predictive of changes in protein (Princivalle et al., 2003, Sands et al., 2003). Furthermore, it is now understood that the functional GABAB receptor is an obligate heterodimer. Receptor binding methods would be unable to differentiate a decrease in receptor affinity that arises from a conformational change in the GABAB(1) subunit from a decrease in affinity that arises from an absence of the GABAB(2) subunit, whose presence is required for high affinity binding of GABA to the GABAB(1) subunit (Kaupmann et al., 1998). Receptor binding also would not allow differentiation of a decrease in receptor number that results from a loss of the GABAB(2) subunit, which is required for insertion of GABAB(1) in the plasma membrane (Bouvier, 2001), from a decrease in receptor number that results from a selective loss of the GABAB(1) subunit itself. The question of whether levels of the GABAB receptor in the dorsal horn are altered after peripheral nerve injury therefore merited further investigation using Western blotting methods with antibodies that recognize each subunit of this receptor, as well as with immunohistochemical methods that could provide better anatomical resolution of possible changes and GTPγS binding to assess possible changes in receptor function.
The first principal finding of this study was that the levels of GABAB(1a), GABAB(1b), and GABAB(2) subunit protein, as detected by Western blotting or immunohistochemical methods, were unchanged in the dorsal quadrant of the spinal cord at any time after L5 spinal nerve ligation as compared to sham-operated rats or naïve rats. Both the GABAB(1) and GABAB(2) antibodies used in this study identified bands at the correct molecular weight, and these bands were absent in spinal cord tissue obtained from mice lacking the respective receptor subunit. The GABAB(2) antibody could also be preabsorbed with its cognate peptide. It is possible that highly localized changes within the dorsal horn would not be detected when blotting the L4 and L5 spinal segments as a unit. However, loss of IB4 staining was evident to varying degrees throughout both the L4 and L5 segments of the spinal cord after L5 spinal nerve ligation (data not shown). Furthermore, quantitation of immunohistochemical staining revealed no change in GABAB(1) and GABAB(2) staining intensity in the superficial dorsal horn, even in sections of the L5 spinal cord where the loss of IB4 staining was marked. Collectively, these data suggest that the lack of change in GABAB subunit protein was not due to a failure to identify a highly localized effect or from the use of inappropriate antibodies.
The second principal finding of this study was that the levels of GABAB(1a) and GABAB(1b) subunit protein were unchanged in the L4 or L5 DRG of rats at any time after L5 spinal nerve ligation. The peripheral processes of L5 DRG neurons undergo Wallerian degeneration after L5 spinal nerve ligation (Ringkamp and Meyer, 2005). Neurons in the adjacent L4 DRG are not unaffected after this presumably restricted injury because their processes intermingle with the degenerating axons of L5 afferents in the periphery. Several groups have documented changes in the expression of growth factors, channels, neurotransmitters and receptors in neurons of the L4 DRG, as well as the L5 DRG after L5 spinal nerve ligation (Fukuoka et al., 1998, Fukuoka et al., 2001, Fukuoka et al., 2002). These changes may underlie the occurrence of spontaneous, paroxysmal discharges recorded in small and large diameter L4 afferent neurons after injury to the L5 spinal nerve (Wu et al., 2002, Ma et al., 2003). The lack of change in GABAB(1a) or GABAB(1b) levels in the L4 and L5 DRG suggests that a loss of GABAB receptors on the central terminals of primary afferent neurons is unlikely to contribute to the development or maintenance of tactile allodynia or spontaneous pain after tight ligation of the L5 spinal nerve. Xiao et al. (2002) also observed no change in mRNA for the GABAB(1) in the DRG following axotomy of the sciatic nerve.
Several investigators have reported that peripheral nerve injury results in a decrement in evoked inhibitory post synaptic currents in dorsal horn neurons (Moore et al., 2002) or that GABA can exert a frank excitatory effect on a subpopulation of lamina I neurons (Coull et al., 2003). Each observation is consistent with a loss of GABA-mediated inhibition in the dorsal horn. However, these studies were restricted to analysis of fast inhibitory synaptic currents mediated by GABAA receptors. Whether there is a concomitant decrement in the slow inhibitory currents mediated by the GABAB receptor in dorsal horn neurons or in GABAB-receptor mediated inhibition of neurotransmitter release from primary afferents has yet to be determined. However, the present data suggest that this is unlikely. Indeed, neither the affinity nor the efficacy of baclofen to stimulate GTPγS binding was decreased after spinal nerve ligation compared to sham-operated rats, suggesting that receptor function is unaltered. In this context, it is notable that intrathecal administration of baclofen, the archetype GABAB receptor agonist, reverses mechanical allodynia and thermal hyperalgesia induced by L5 and L6 spinal nerve ligation or partial nerve ligation (Patel et al., 2001, Malan et al., 2002) and inhibits the responses of dorsal horn neurons to Aβ, Aδ, and C fiber stimulation (Sokal and Chapman, 2003). These findings are consistent with the continued presence of functional GABAB receptors in the spinal cord dorsal horn. While the collective data indicate that neither GABAB receptor number nor function is altered after peripheral nerve injury, the possibility that injury alters the phosphorylation state of this receptor, its trafficking or the availability of accessory proteins should not be excluded.
Several interesting observations were made in the course of this study. As previously reported, (Poorkhalkali et al., 2000, Towers et al., 2000), GABAB(1a) was the primary and only quantifiable isoform of the GABAB(1) subunit found in the DRG. Although GABAB(2) mRNA is present in the DRG (Towers et al., 2000), this subunit was practically undetectable in the DRG even when protein from as many as six DRG was loaded on a gel (but see Charles et al., 2001). Both GABAB(1) and GABAB(2) subunits are necessary to form functional GABAB receptors having the conventional pharmacology. The disparate localization of GABAB(1a) and GABAB(2) subunits in the DRG suggests that functional GABAB receptors are not present on the soma of primary afferent neurons in situ, although their presence on cultured DRG neurons has been amply demonstrated (Dolphin and Scott, 1986). The finding that GABAB(2) was not detectable in the DRG, yet levels of this subunit were decreased in the dorsal horn after dorsal rhizotomy suggests that the GABAB(2) subunit is largely absent in the cell body, but present in the central terminals of primary afferents. Filippov et al. (2000) similarly concluded that soma of sympathetic neurons in the superior cervical ganglion express only the GABAB(1) subunit and that detectable levels of GABAB(2) are absent. Mice that are null mutant for the GABAB(2) subunit exhibit residual numbers of functional GABAB receptors possibly comprised of homodimers of GABAB(1) subunits. Interestingly, agonist application to hippocampal neurons in these mice causes closure of a K+ channel through a G-protein coupled mechanism and results in the occurrence of an inward, rather than outward current (Gassmann et al., 2004). Thus, in situ, the soma of DRG neurons may represent an easily accessible native population of this atypical GABAB receptor. The disparate localization of these two subunits in primary afferent neurons also raises some interesting issues and opportunities for future studies of GABAB receptor trafficking and assembly.
Acknowledgments
This work supported by the National Institute on Drug Abuse (1RO1DA16433 to D.L.H. and 1F30DA017418 to M.P.E) and the Swiss Science Foundation (3100-067100.01 to B.B.). We thank Elijah Frakes for fruitful discussions and Mary Bradford for technical assistance.
Footnotes
Section Editor: Yoland Smith
References
- Ambalavanar R, Morris R. An ultrastructural study of the binding of an alpha-D-galactose specific lectin from Griffonia simplicifolia to trigeminal ganglion neurons and the trigeminal nucleus caudalis in the rat. Neuroscience. 1993;52:699–709. doi: 10.1016/0306-4522(93)90418-f. [DOI] [PubMed] [Google Scholar]
- Ataka T, Kumamoto E, Shimoji K, Yoshimura M. Baclofen inhibits more effectively C-afferent than Adelta-afferent glutamatergic transmission in substantia gelatinosa neurons of adult rat spinal cord slices. Pain. 2000;86:273–282. doi: 10.1016/S0304-3959(00)00255-4. [DOI] [PubMed] [Google Scholar]
- Benke D, Honer M, Michel C, Bettler B, Mohler H. γ-aminobutyric acid type B receptor splice variant proteins GBR1a and GBR1b are both associated with GBR2 in situ and display differential regional and subcellular distribution. J Biol Chem. 1999;274:27323–27330. doi: 10.1074/jbc.274.38.27323. [DOI] [PubMed] [Google Scholar]
- Bettler B, Kaupmann K, Mosbacher J, Gassmann M. Molecular structure and physiological functions of GABAB receptors. Physiol Rev. 2004;84:835–867. doi: 10.1152/physrev.00036.2003. [DOI] [PubMed] [Google Scholar]
- Bouvier M. Oligomerization of G-protein-coupled transmitter receptors. Nat Rev Neurosci. 2001;2:274–286. doi: 10.1038/35067575. [DOI] [PubMed] [Google Scholar]
- Bowery NG, Bettler B, Froestl W, Gallagher JP, Marshall F, Raiteri M, Bonner TI, Enna SJ. International Union of Pharmacology. XXXIII. Mammalian γ-aminobutyric acidB receptors: structure and function. Pharmacol Rev. 2002;54:247–264. doi: 10.1124/pr.54.2.247. [DOI] [PubMed] [Google Scholar]
- Calver AR, Medhurst AD, Robbins MJ, Charles KJ, Evans ML, Harrison DC, Stammers M, Hughes SA, Hervieu G, Couve A, Moss SJ, Middlemiss DN, Pangalos MN. The expression of GABAB1 and GABAB2 receptor subunits in the CNS differs from that in peripheral tissues. Neuroscience. 2000;100:155–170. doi: 10.1016/s0306-4522(00)00262-1. [DOI] [PubMed] [Google Scholar]
- Castro-Lopes JM, Malcangio M, Pan BH, Bowery NG. Complex changes of GABAA and GABAB receptor binding in the spinal cord dorsal horn following peripheral inflammation or neurectomy. Brain Res. 1995;679:289–297. doi: 10.1016/0006-8993(95)00262-o. [DOI] [PubMed] [Google Scholar]
- Castro AR, Pinto M, Lima D, Tavares I. Nociceptive spinal neurons expressing NK1 and GABAB receptors are located in lamina I. Brain Res. 2004;1003:77–85. doi: 10.1016/j.brainres.2003.12.027. [DOI] [PubMed] [Google Scholar]
- Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods. 1994;53:55–63. doi: 10.1016/0165-0270(94)90144-9. [DOI] [PubMed] [Google Scholar]
- Charles KJ, Evans ML, Robbins MJ, Calver AR, Leslie RA, Pangalos MN. Comparative immunohistochemical localisation of GABAB1a, GABAB1b and GABAB2 subunits in rat brain, spinal cord and dorsal root ganglion. Neuroscience. 2001;106:447–467. doi: 10.1016/s0306-4522(01)00296-2. [DOI] [PubMed] [Google Scholar]
- Choi Y, Yoon YW, Na HS, Kim SH, Chung JM. Behavioral signs of ongoing pain and cold allodynia in a rat model of neuropathic pain. Pain. 1994;59:369–376. doi: 10.1016/0304-3959(94)90023-X. [DOI] [PubMed] [Google Scholar]
- Coull JA, Boudreau D, Bachand K, Prescott SA, Nault F, Sik A, De Koninck P, De Koninck Y. Trans-synaptic shift in anion gradient in spinal lamina I neurons as a mechanism of neuropathic pain. Nature. 2003;424:938–942. doi: 10.1038/nature01868. [DOI] [PubMed] [Google Scholar]
- Dolphin AC, Scott RH. Inhibition of calcium currents in cultured rat dorsal root ganglion neurones by (-)-baclofen. Br J Pharmacol. 1986;88:213–220. doi: 10.1111/j.1476-5381.1986.tb09489.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Filippov AK, Couve A, Pangalos MN, Walsh FS, Brown DA, Moss SJ. Heteromeric assembly of GABABR1 and GABABR2 receptor subunits inhibits Ca2+ current in sympathetic neurons. J Neurosci. 2000;20:2867–2874. doi: 10.1523/JNEUROSCI.20-08-02867.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuoka T, Kondo E, Dai Y, Hashimoto N, Noguchi K. Brain-derived neurotrophic factor increases in the uninjured dorsal root ganglion neurons in selective spinal nerve ligation model. J Neurosci. 2001;21:4891–4900. doi: 10.1523/JNEUROSCI.21-13-04891.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuoka T, Tokunaga A, Kondo E, Miki K, Tachibana T, Noguchi K. Change in mRNAs for neuropeptides and the GABA(A) receptor in dorsal root ganglion neurons in a rat experimental neuropathic pain model. Pain. 1998;78:13–26. doi: 10.1016/S0304-3959(98)00111-0. [DOI] [PubMed] [Google Scholar]
- Fukuoka T, Tokunaga A, Tachibana T, Dai Y, Yamanaka H, Noguchi K. VR1, but not P2X(3), increases in the spared L4 DRG in rats with L5 spinal nerve ligation. Pain. 2002;99:111–120. doi: 10.1016/s0304-3959(02)00067-2. [DOI] [PubMed] [Google Scholar]
- Gassmann M, Shaban H, Vigot R, Sansig G, Haller C, Barbieri S, Humeau Y, Schuler V, Muller M, Kinzel B, Klebs K, Schmutz M, Froestl W, Heid J, Kelly PH, Gentry C, Jaton AL, Van der Putten H, Mombereau C, Lecourtier L, Mosbacher J, Cryan JF, Fritschy JM, Luthi A, Kaupmann K, Bettler B. Redistribution of GABAB(1) protein and atypical GABAB responses in GABAB(2)-deficient mice. J Neurosci. 2004;24:6086–6097. doi: 10.1523/JNEUROSCI.5635-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammond, DL, 1997. Inhibitory neurotransmitters and nociception: Role of GABA and glycine. Berlin, Springer–Verlag.
- Hao JX, Xu XJ, Wiesenfeld-Hallin Z. Intrathecal gamma-aminobutyric acidB (GABAB) receptor antagonist CGP 35348 induces hypersensitivity to mechanical stimuli in the rat. Neurosci Lett. 1994;182:299–302. doi: 10.1016/0304-3940(94)90821-4. [DOI] [PubMed] [Google Scholar]
- Kangrga I, Jiang MC, Randic M. Actions of (-)-baclofen on rat dorsal horn neurons. Brain Res. 1991;562:265–275. doi: 10.1016/0006-8993(91)90630-e. [DOI] [PubMed] [Google Scholar]
- Kaupmann K, Huggel K, Heid J, Flor PJ, Bischoff S, Mickel SJ, McMaster G, Angst C, Bittiger H, Froestl W, Bettler B. Expression cloning of GABAB receptors uncovers similarity to metabotropic glutamate receptors. Nature. 1997;386:239–246. doi: 10.1038/386239a0. [DOI] [PubMed] [Google Scholar]
- Kaupmann K, Malitschek B, Schuler V, Heid J, Froestl W, Beck P, Mosbacher J, Bischoff S, Kulik A, Shigemoto R, Karschin A, Bettler B. GABA(B)-receptor subtypes assemble into functional heteromeric complexes. Nature. 1998;396:683–687. doi: 10.1038/25360. [DOI] [PubMed] [Google Scholar]
- Kim SH, Chung JM. An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain. 1992;50:355–363. doi: 10.1016/0304-3959(92)90041-9. [DOI] [PubMed] [Google Scholar]
- Luo ZD, Calcutt NA, Higuera ES, Valder CR, Song YH, Svensson CI, Myers RR. Injury type-specific calcium channel α2δ-1 subunit up-regulation in rat neuropathic pain models correlates with antiallodynic effects of gabapentin. J Pharmacol Exp Ther. 2002;303:1199–1205. doi: 10.1124/jpet.102.041574. [DOI] [PubMed] [Google Scholar]
- Luo ZD, Chaplan SR, Higuera ES, Sorkin LS, Stauderman KA, Williams ME, Yaksh TL. Upregulation of dorsal root ganglion α2δ calcium channel subunit and its correlation with allodynia in spinal nerve-injured rats. J Neurosci. 2001;21:1868–1875. doi: 10.1523/JNEUROSCI.21-06-01868.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma C, Shu Y, Zheng Z, Chen Y, Yao H, Greenquist KW, White FA, LaMotte RH. Similar electrophysiological changes in axotomized and neighboring intact dorsal root ganglion neurons. J Neurophysiol. 2003;89:1588–1602. doi: 10.1152/jn.00855.2002. [DOI] [PubMed] [Google Scholar]
- Malan TP, Mata HP, Porreca F. Spinal GABAA and GABAB receptor pharmacology in a rat model of neuropathic pain. Anesthesiology. 2002;96:1161–1167. doi: 10.1097/00000542-200205000-00020. [DOI] [PubMed] [Google Scholar]
- Malcangio M, Ramer MS, Jones MG, McMahon SB. Abnormal substance P release from the spinal cord following injury to primary sensory neurons. Eur J Neurosci. 2000;12:397–399. doi: 10.1046/j.1460-9568.2000.00946.x. [DOI] [PubMed] [Google Scholar]
- Marvizon JC, Grady EF, Stefani E, Bunnett NW, Mayer EA. Substance P release in the dorsal horn assessed by receptor internalization: NMDA receptors counteract a tonic inhibition by GABA(B) receptors. Eur J Neurosci. 1999;11:417–426. doi: 10.1046/j.1460-9568.1999.00445.x. [DOI] [PubMed] [Google Scholar]
- Moore KA, Kohno T, Karchewski LA, Scholz J, Baba H, Woolf CJ. Partial peripheral nerve injury promotes a selective loss of GABAergic inhibition in the superficial dorsal horn of the spinal cord. J Neurosci. 2002;22:6724–6731. doi: 10.1523/JNEUROSCI.22-15-06724.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noguchi K, Kawai Y, Fukuoka T, Senba E, Miki K. Substance P induced by peripheral nerve injury in primary afferent sensory neurons and its effect on dorsal column nucleus neurons. J Neurosci. 1995;15:7633–7643. doi: 10.1523/JNEUROSCI.15-11-07633.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel S, Naeem S, Kesingland A, Froestl W, Capogna M, Urban L, Fox A. The effects of GABAB agonists and gabapentin on mechanical hyperalgesia in models of neuropathic and inflammatory pain in the rat. Pain. 2001;90:217–226. doi: 10.1016/S0304-3959(00)00404-8. [DOI] [PubMed] [Google Scholar]
- Poorkhalkali N, Juneblad K, Jonsson AC, Lindberg M, Karlsson O, Wallbrandt P, Ekstrand J, Lehmann A. Immunocytochemical distribution of the GABAB receptor splice variants GABAB R1a and R1b in the rat CNS and dorsal root ganglia. Anat Embryol (Berl) 2000;201:1–13. doi: 10.1007/pl00008224. [DOI] [PubMed] [Google Scholar]
- Price GW, Kelly JS, Bowery NG. The location of GABAB receptor binding sites in mammalian spinal cord. Synapse. 1987;1:530–538. doi: 10.1002/syn.890010605. [DOI] [PubMed] [Google Scholar]
- Price GW, Wilkin GP, Turnbull MJ, Bowery NG. Are baclofen-sensitive GABAB receptors present on primary afferent terminals of the spinal cord? Nature. 1984;307:71–74. doi: 10.1038/307071a0. [DOI] [PubMed] [Google Scholar]
- Princivalle AP, Richards DA, Duncan JS, Spreafico R, Bowery NG. Modification of GABAB1 and GABAB2 receptor subunits in the somatosensory cerebral cortex and thalamus of rats with absence seizures (GAERS) Epilepsy Res. 2003;55:39–51. doi: 10.1016/s0920-1211(03)00090-1. [DOI] [PubMed] [Google Scholar]
- Ringkamp M, Meyer RA. Injured versus uninjured afferents: Who is to blame for neuropathic pain? Anesthesiology. 2005;103:221–223. doi: 10.1097/00000542-200508000-00002. [DOI] [PubMed] [Google Scholar]
- Sands SA, McCarson KE, Enna SJ. Relationship between the antinociceptive response to desipramine and changes in GABAB receptor function and subunit expression in the dorsal horn of the rat spinal cord. Biochem Pharmacol. 2004;67:743–749. doi: 10.1016/j.bcp.2003.10.008. [DOI] [PubMed] [Google Scholar]
- Sands SA, Purisai MG, Chronwall BM, Enna SJ. Ontogeny of GABAB receptor subunit expression and function in the rat spinal cord. Brain Res. 2003;972:197–206. doi: 10.1016/s0006-8993(03)02534-4. [DOI] [PubMed] [Google Scholar]
- Schuler V, Luscher C, Blanchet C, Klix N, Sansig G, Klebs K, Schmutz M, Heid J, Gentry C, Urban L, Fox A, Spooren W, Jaton AL, Vigouret J, Pozza M, Kelly PH, Mosbacher J, Froestl W, Kaslin E, Korn R, Bischoff S, Kaupmann K, van der Putten H, Bettler B. Epilepsy, hyperalgesia, impaired memory, and loss of pre- and postsynaptic GABA(B) responses in mice lacking GABAB(1) Neuron. 2001;31:47–58. doi: 10.1016/s0896-6273(01)00345-2. [DOI] [PubMed] [Google Scholar]
- Smith GD, Harrison SM, Birch PJ, Elliott PJ, Malcangio M, Bowery NG. Increased sensitivity to the antinociceptive activity of (±)-baclofen in an animal model of chronic neuropathic, but not chronic inflammatory hyperalgesia. Neuropharmacology. 1994;33:1103–1108. doi: 10.1016/0028-3908(94)90149-x. [DOI] [PubMed] [Google Scholar]
- Sokal DM, Chapman V. Spinal GABAB-receptor antagonism increases nociceptive transmission in vivo. Neuroreport. 2001;12:3247–3250. doi: 10.1097/00001756-200110290-00021. [DOI] [PubMed] [Google Scholar]
- Sokal DM, Chapman V. Inhibitory effects of spinal baclofen on spinal dorsal horn neurones in inflamed and neuropathic rats in vivo. Brain Res. 2003;987:67–75. doi: 10.1016/s0006-8993(03)03255-4. [DOI] [PubMed] [Google Scholar]
- Sung B, Lim G, Mao J. Altered expression and uptake activity of spinal glutamate transporters after nerve injury contribute to the pathogenesis of neuropathic pain in rats. J Neurosci. 2003;23:2899–2910. doi: 10.1523/JNEUROSCI.23-07-02899.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towers S, Princivalle A, Billinton A, Edmunds M, Bettler B, Urban L, Castro-Lopes J, Bowery NG. GABAB receptor protein and mRNA distribution in rat spinal cord and dorsal root ganglia. Eur J Neurosci. 2000;12:3201–3210. doi: 10.1046/j.1460-9568.2000.00237.x. [DOI] [PubMed] [Google Scholar]
- Woolf CJ. Dissecting out mechanisms responsible for peripheral neuropathic pain: implications for diagnosis and therapy. Life Sci. 2004;74:2605–2610. doi: 10.1016/j.lfs.2004.01.003. [DOI] [PubMed] [Google Scholar]
- Woolf CJ, Shortland P, Coggeshall RE. Peripheral nerve injury triggers central sprouting of myelinated afferents. Nature. 1992;355:75–78. doi: 10.1038/355075a0. [DOI] [PubMed] [Google Scholar]
- Wu G, Ringkamp M, Murinson BB, Pogatzki EM, Hartke TV, Weerahandi HM, Campbell JN, Griffin JW, Meyer RA. Degeneration of myelinated efferent fibers induces spontaneous activity in uninjured C-fiber afferents. J Neurosci. 2002;22:7746–7753. doi: 10.1523/JNEUROSCI.22-17-07746.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao HS, Huang QH, Zhang FX, Bao L, Lu YJ, Guo C, Yang L, Huang WJ, Fu G, Xu SH, Cheng XP, Yan Q, Zhu ZD, Zhang X, Chen Z, Han ZG, Zhang X. Identification of gene expression profile of dorsal root ganglion in the rat peripheral axotomy model of neuropathic pain. Proc Natl Acad Sci U S A. 2002;99:8360–8365. doi: 10.1073/pnas.122231899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang K, Ma WL, Feng YP, Dong YX, Li YQ. Origins of GABAB receptor-like immunoreactive terminals in the rat spinal dorsal horn. Brain Res Bull. 2002;58:499–507. doi: 10.1016/s0361-9230(02)00824-9. [DOI] [PubMed] [Google Scholar]
- Yang K, Wang D, Li YQ. Distribution and depression of the GABAB receptor in the spinal dorsal horn of adult rat. Brain Res Bull. 2001;55:479–485. doi: 10.1016/s0361-9230(01)00546-9. [DOI] [PubMed] [Google Scholar]
- Yang L, Zhang FX, Huang F, Lu YJ, Li GD, Bao L, Xiao HS, Zhang X. Peripheral nerve injury induces trans-synaptic modification of channels, receptors and signal pathways in rat dorsal spinal cord. Eur J Neurosci. 2004;19:871–883. doi: 10.1111/j.0953-816x.2004.03121.x. [DOI] [PubMed] [Google Scholar]


