Abstract
Since the inception of cryosurgery in the 1850s, landmark advances in chemistry, physics, materials science, and biology have culminated in the sophisticated cryosurgical devices currently in use. Effective cryosurgical tissue injury depends on four criteria: 1) excellent monitoring of the process; 2) fast cooling to a lethal temperature; 3) slow thawing; and 4) repetition of the freeze-thaw cycle. Meeting these criteria depends on understanding the imaging technology used to visualize the iceball, the type of cryogen used, the size of the probe, and probe arrangement. Third-generation cryosurgical equipment offers advantages over previous designs. These machines rely on argon for freezing but also use helium to warm probes and accelerate the treatment process, and they offer additional safety by being able to rapidly arrest iceball formation. Metallurgic advances have led to the development of thinner probes, which have been easily adapted to perineal templates similar to those used for prostate brachytherapy.
Key words: Prostate cancer, Cryotherapy, Ultrasound, Joule-Thomson effect, Freeze-thaw cycle
Cancer cryosurgery began in England in the 1850s, when James Arnott1 used local applications of salt solutions containing crushed ice to treat advanced cancers in accessible sites, such as the breast and the uterine cervix. Since then, landmark advances in chemistry, physics, materials science, and biology have culminated in the sophisticated cryosurgical devices currently in use. In this article, we will review the remarkable evolution of cryotherapy applications in human cancer, with a specific emphasis on prostate cancer, and discuss the fundamentals of cryobiology necessary for the understanding and practice of modern cryotherapy.
The Evolution and Development of Cryotherapy
James Arnott’s salt solutions reached temperatures of −18°C to −24°C and were adequate to freeze tumors, with resultant diminution in size, reduction in drainage, and amelioration of pain. Arnott wrote that “congelation arresting the accompanying inflammation, and destroying the vitality of the cancer cell, is not only calculated to prolong life for a great period, but may, not improbably, in the early stage of the disease, exert a curative action.”1
A quarter-century later, the technology necessary to compress and liquify atmospheric gases was developed, making the achievement of extremely low temperatures possible. Oxygen was first liquefied in small quantities, until the English scientist James Dewar developed a vacuum flask to store liquefied gases in 1892. The term “cryogen” came into use, and by the end of the 19th century, both liquid air and solid carbon dioxide were available for therapeutic use on localized malignancies.
The use of freezing techniques to treat deep-seated malignancies did not evolve rapidly after the discovery of liquified gases, although their use in a variety of dermatologic conditions became common. A notable exception was the work of Temple Fay,2 a neurosurgeon in Philadelphia, between 1936 and 1940. Fay used local and general refrigeration techniques to treat large inoperable cancers of the uterine cervix and breast with irrigations of refrigerated cold solutions and applications of ice packs. He also used implanted metal capsules connected to an external cold irrigation system in brain tumors. As Arnott had described nearly 90 years earlier, the result was reduction in tumor size and amelioration of pain symptoms.
After World War II, liquid nitrogen (−196°C) became commercially available. In 1950, Allington3 introduced this cryogen into clinical practice by the technique of using cotton swabs dipped in liquid nitrogen to treat a variety of nonneoplastic and neoplastic skin diseases. Liquid nitrogen applied with a cotton swab soon became a common practice for the treatment of verrucae, keratoses, and diverse nonneoplastic lesions. However, its use for skin tumors was not common because the swab technique produced only superficial freezing.
To address the problem of insufficient depth of tissue freezing, physicians began using solid copper cylinder discs, cooled by immersion in liquid nitrogen before application to the skin.4 The copper cylinders had much superior thermal capacity and heat exchange characteristics compared with the cotton applicators, and with exertion of pressure on the lesion to minimize blood flow, they could be used to limit the “heat sink” effect. These approaches allowed the doubling of the depth of tissue destruction from 1 to 2 mm to 4 to 5 mm. Nonetheless, freezing large volumes of tumor tissue still was not feasible.
The development of modern cryosurgery was based on experiments in the late 1940s and early 1950s, in which focal tissue destruction was produced by freezing and studied at the pathophysiologic level. Hass and Taylor5 described a technique producing sharply defined and uniformly necrotic lesions 2 to 3 cm in diameter in the brain, heart, liver, and kidneys, using an apparatus cooled by pressurized carbon dioxide. Importantly to modern cryotherapy, they observed that cryotherapy was associated with a lack of suppuration or sequestration and had a slow, uncomplicated healing process.
The introduction of an automated cryosurgical apparatus cooled by liquid nitrogen by Cooper and Lee6 in 1961 was the next stage in the evolution of cryosurgery. When applied to the tumor, the probe made the continuous and rapid extraction of heat possible, resulting in freezing in situ. Cooper, a neurosurgeon in New York City, designed the apparatus to produce a cryogenic lesion in the brain for the treatment of Parkinson’s disease.7 Importantly, Cooper modified his original 3-mm probes by adding a heating element to facilitate release from the tissue after freezing.
Prostate cryosurgery followed in the mid 1960s, when Gonder and colleagues8,9 developed a modified apparatus and probes suitable for the transurethral freezing of prostatic tissue. These investigators also carried out animal experiments leading to the clinical use of cryosurgery for both benign prostatic hypertrophy and prostatic cancer. These reports stimulated significant interest among the members of the urologic community, and extensive experience with prostatic cryosurgery followed,10–13 including a perineal approach to the gland by Flocks and coworkers in 1972.14 It is important to note that this perineal approach included an open exposure of the prostate.
Although liquid nitrogen had become the most common cryogen for use in the treatment of intra-abdominal tumors, a new approach to cryogenic therapy was emerging. This fundamental advance involved the application of the Joule-Thomson effect and, as will be described below, was vital to the evolution of the latest generation of prostate cryotherapy units. In his initial experiments, Torre15 used an experimental argon gas system (argon gas under a pressure of 1000 to 2500 pounds per square inch), which could deliver temperatures as cold as −185°C via the Joule-Thomson effect. The Joule-Thomson effect was named in honor of James Joule and William Thomson (Thomson is better known as Lord Kelvin), who discovered the effect in the 1850s by performing a set of careful experiments along with a detailed theoretical analysis. When a gas is expanded through a pinhole valve, its temperature might decrease. Controlling this effect was the cornerstone upon which many commonplace appliances used today were developed, such as the air conditioner, the refrigerator, and modern medical cryotherapy devices.
The fundamental reasons for this effect are as follows. The atoms or molecules that make up a gas are not like miniature snooker balls but in fact interact with each other. There are two primary types of interactions. In some gases, such as argon, the gas atoms stick to each other, whereas in others, such as helium, they repel each other. Hence, there are two major classes of so-called intermolecular interactions: attractive (sticking marmalade) and repulsive (pushing rubber) interactions. Seen from the outside of the gas container, the attractive interactions lead to a more stable gas. One would have to put in energy to stop the attractive interactions, because the gas itself has a lower internal energy. Conversely, to stop the repulsive interactions, one would have to take energy away from the molecules, because the gas itself has a high internal energy.
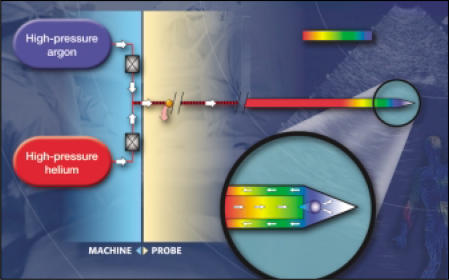
Under normal circumstances, the attractive interactions are by far the most dominant. Hence, a gas packed into a small volume at high pressure will be subject to many attractive interactions, or, in other words, have a relatively low internal energy. When it is released into a big volume at a lower pressure (as experienced at the tip of the cryoneedle for example) the particles will be subject to less interaction, or have a higher internal energy. This transition from a low internal energy state to a high internal energy state consumes energy, draining it from the environment. This is perceived as a drop in temperature. Importantly, a few gases, such as helium, show the opposite behavior, heating up under expansion. The repulsive forces in helium and hydrogen outweigh the sticking forces, resulting in heating instead of cooling, as described for the other gases. In cryotherapy, the application of this principle with gases with different Joule-Thomson behaviors offers the opportunity to clinically freeze or thaw tissues (Figure 1).
Figure 1.
Diagrammatic representation of the application of the Joule-Thomson effect for both freezing and thawing target tissue in cryotherapy with the SeedNet TM Cryotherapy System, (Oncura, Inc., Plymouth Meeting, PA). Figure courtesy of Oncura, Inc.
The 1970s was an era of reappraisal for cryotherapy, and some uses of cryosurgery fell into disfavor. Among these was the use of cryosurgery for prostatic disease, both benign and malignant, which became disused because of the prolonged catheter drainage required after operation, the high incidence of complications, such as prostatic rectal fistulas, and the emergence of transurethral resection for benign disease. Nevertheless, interest in cryotherapy for prostatic cancer persisted because of the potential benefits, possibly including a favorable immunologic response, as suggested by remission of metastatic cancer after tumor therapy.16,17 Sporadic reports on the use of cryosurgery for prostatic cancer were published from the few medical centers that could demonstrate a favorable survival rate compared with other therapy.18,19
In a way very reminiscent of the resurgence of brachytherapy, interest in prostate cryosurgery was renewed after the development of transrectal ultrasound in the 1990s. The technique, initially described by Onik and colleagues,20,21 had the potential to limit adjacent organ damage and thus minimize the major complications of prostatic rectal fistula and urinary incontinence. In addition, ultrasound-guided placement of the cryoprobe into the prostate allowed percutaneous perineal placement. With knowledge of the geometry of the ensuing iceball, exact placement could be carried out, resulting in complete freezing of the organ.
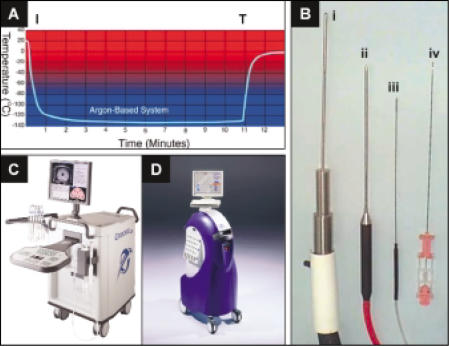
In the 1990s, improvements in cryosurgical apparatus and accessories led to greater precision of therapy and mitigation of complications. The first-generation machines included vacuum-insulated probes of smaller diameter, cooled by liquid nitrogen.22 These probes could be used percutaneously in the treatment of prostate cancer. In addition, it was determined that urethral warming was beneficial in reducing morbidity from this procedure, and its use was widely adopted. Subsequent generations of machines, in which compressed argon gas and the Joule-Thomson effect were used, resolved the problem of rapid arrest of the cooling process that had previously been a factor leading to adjacent organ damage and subsequent complications. The use of compressed argon gas offered many advantages over liquid nitrogen: 1) temperatures are equivalent to those of liquid nitrogen (Figure 2A); 2) the freezing process can start and stop almost instantaneously (Figure 2A); 3) argon is easily stored without evaporation and is ready for immediate use; 4) cryotherapy control and monitoring units and associated tubing for argon are much smaller than first-generation units (Figure 2B); and 5) cryotherapy machines are much smaller than liquid nitrogen units, thus allowing for easy transport from room to room or facility to facility (Figure 2C, D).
Figure 2.
(A) Graphic representation of iceball temperature as a function of time after initiation (I) and termination (T) of therapy with CRYOcare™ (Endocare, Inc., Irvine, CA), an argon-based Joule-Thomson effect cryoprobe. Figure courtesy of Endocare. (B) Cryoprobe designs: liquid nitrogen 5-mm probe (i); Argon Joule-Thomson effect 3-mm probe (ii); Argon Joule-Thomson effect 1.5-mm (17G) probe (iii); and prostate biopsy needle (iv). (C) CRYO-care Cryotherapy System; (D) SeedNet Cryotherapy System.
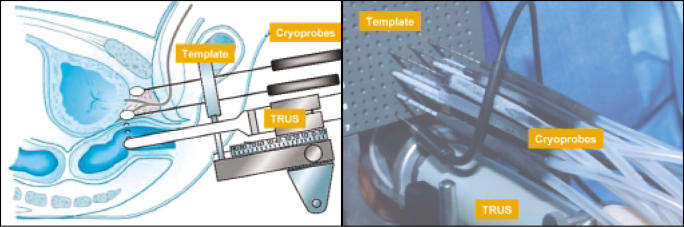
Currently available equipment offered several potential advantages compared with previous designs. Although these machines relied on the Joule-Thomson effect of argon gas for their freezing, they also used helium gas and its reverse Joule-Thomson effect to warm the probes and therefore accelerate the treatment process (Figure 2C, D). They also offered additional safety by being able to rapidly arrest iceball formation (Figure 2A). In addition, metallurgic advances led to the development of 17-gauge needles (Figure 3), which were easily adapted to perineal templates similar to those used for prostate brachytherapy (Figure 3). Since the resurgence of prostate brachytherapy, many urologists have become familiar with this technique, and the similarities between brachytherapy and third-generation cryotherapy instrumentation might facilitate the adoption of this technique in appropriately selected patients.
Figure 3.
Cryotherapy with 17-gauge cryoprobes is similar to that of brachytherapy (radioactive seed implantation). An insertion template similar to that used in brachytherapy is placed against the perineum. With transrectal ultrasound and template guidance, 17-gauge cryoprobes (CryoNeedles™, Oncura, Inc., Plymouth Meeting, PA) are inserted through the perineum into the prostate. Argon gas circulating through these needles (Figure 1) generates a very low temperature, causing the formation of ice. With transrectal ultrasound monitoring, iceballs fuse (Figure 7) to create a frozen region that conforms to the size and shape of the prostate. TRUS, transrectal ultrasound.
Cryobiology
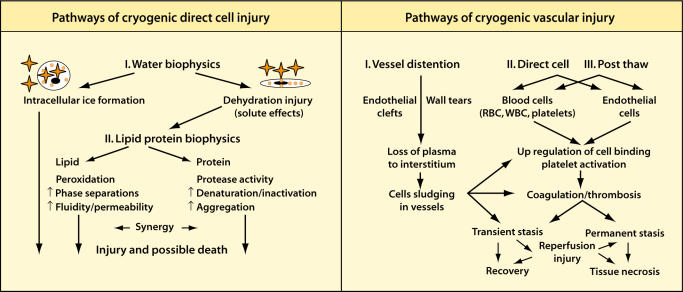
The science of cryobiology began with the study of cryopreservation and experimental and clinical work on patients with frostbite.23,24 Subsequently, Cooper25 concluded that temperatures of −20°C held for 1 minute would cause tissue necrosis. During the 1960s and 1970s, the importance of rapid freezing, slow thawing, and repetition of the freezethaw cycle was recognized in the study of cryosurgical injury.26,27 The destructive effects of cryosurgery can be grouped into two major mechanisms: cellular injury and vascular injury (Figure 4). Cellular injury is mainly immediate, and vascular injury is largely delayed. Cellular injury results from the deleterious effect of the cooling and warming cycles. Vascular injury, slower to take effect, results from the progressive failure of the microcirculation, ultimate vascular stasis, and subsequent necrosis. Although the relative importance of these two mechanisms is debated, the pragmatic approach for the clinician is to attempt to maximize the effects of both cellular and vascular damage to destroy a predetermined volume of tissue with the highest degree of certainty. These two mechanisms of injury are discussed below, with particular emphasis on the biology relevant to the practicing clinician.
Figure 4.
Diagrammatic representation of the pathways of freezing-based direct cell injury and those of vascular injury. RBC, red blood cells; WBC, white blood cells. Reproduced with permission from Hoffmann NE, Bischof JC. The cryobiology of cryosurgical injury. Urology. 2002;60(2 suppl 1):40–49.
Direct Cell Injury
The damaging effects of low temperature on cells begin gradually as temperature drops. Cell metabolism and structure are altered along with their constituent proteins and lipids. In fact, if continued for a sufficiently long time, cell death might result, even without exposure to freezing temperatures.22,28
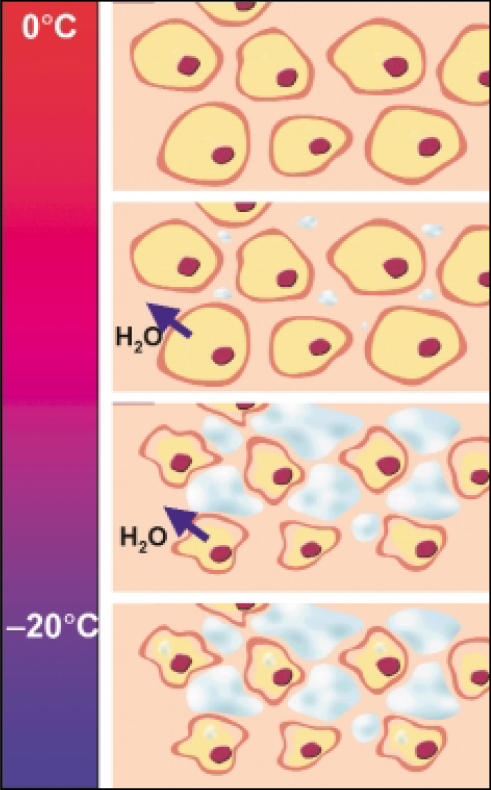
As the temperature falls to less than 0°C, water crystallizes. This results in more significant damage than with mere prolonged cooling. Crystal formation first occurs in the extracellular spaces, which withdraws water from the system and creates a hyperosmotic extracellular environment. This in turn draws water from the cells (Figure 5). Effective “cellular dehydration” occurs predominantly between 0°C and −20°C. Given enough time in this dehydrated state, the increased intracellular electrolyte concentration is often sufficient to destroy the cells.
Figure 5.
Direct cell injury from freezing. As the temperature falls to below 0°C, water crystallizes, which results in more damage than from mere prolonged cooling. Crystal formation first occurs in the extracellular spaces, which withdraws water from the system and creates a hyperosmotic extracellular environment. This in turn draws water from the cells. Effective “cellular dehydration” occurs predominantly between 0°C and −20°C. Given enough time in this dehydrated state, the increased intracellular electrolyte concentration is often sufficient to destroy the cells. Many cells might contain ice crystals by −15°C, but for intracellular ice formation to be certain, the temperature needs to drop to below −40°C.
However, in some cases the deleterious effects of cell dehydration and solute concentration, which is sometimes called solution-effect injury, might not be lethal to cells. In these situations, the formation of intracellular ice, which begins at temperatures below −20°C, is almost always lethal.24,29–32 Many cells might contain ice crystals by −15°C, but to be certain of intracellular ice formation the temperature needs to drop to less than −40°C.22 From the clinical perspective, it is important to note that intracellular ice crystal formation is more efficient at rapid cooling rates; slower rates of cooling will result in cellular dehydration as described above. Rapid cooling does not allow time for water to leave the cells and therefore keeps their solute freezing point higher. In this situation, the solution-effect injury is secondary to the results of intracellular ice crystal formation. It is important to note that regardless of whether the ice is extracellular or intracellular, water is removed from the biologic system, thus resulting in cellular desiccation.
During thawing, ice crystals fuse to form larger crystals, a process called recrystallization, which occurs at temperatures warmer than −40°C. In tissues with closely packed cells, these large crystals are disruptive to cell membranes and cause additional cell damage. As the ice melts, the extracellular environment becomes hypotonic. This occurs because extracellular ice formation occurs first, resulting in intracellular water being drawn out to compensate for the effective dehydration of the extracellular space. Therefore, upon thawing of the ice, more free water will exist outside the cells. This will enter the damaged cells, and the subsequent increase in cell volume might lead to cell membrane rupture.
Vascular Injury
Loss of circulation resulting in cellular anoxia and hypoxia is considered the main mechanism of injury in cryosurgery. Several in vivo studies have shown that upon thawing of frozen tissue, circulatory stasis develops rapidly. A sequence of events in the circulation during cryosurgical treatment results in vascular stasis. Most experimental data have been obtained by the use of transilluminated tissues, such as the mesentery and the cheek pouches of hamsters,33,34 to demonstrate that vascular changes can be seen at temperatures as warm as −20°C.35–37 During the initial freeze cycle, the tissue responds with vasoconstriction, with a resultant decrease in blood flow that eventually ceases when freezing is complete. During thawing, the circulation returns with a compensatory vasodilatation. However, the endothelial damage from cryotherapy results in increased permeability of the capillary walls, edema, platelet aggregation, and microthrombus formation. Progressive circulatory stagnation results over the ensuing hours. Many small blood vessels become completely thrombosed 3 to 4 hours after thawing. Larger arterioles might remain open for up to 24 hours.35 Together, these effects culminate in tissue necrosis, except at the periphery of the previously frozen volume of tissue.
Parameters of the Freeze-Thaw Cycle That Affect Tissue Destruction During Cryotherapy
Extensive investigation over the past 40 years suggests that the degree of cryosurgical tissue injury is a function of four different parameters: 1) temperature produced in the tissue; 2) cooling rate; 3) warming or thaw rate; and 4) repeated freezing. These parameters are under a certain degree of control by the treating physician and can be optimized by the engineering of the cryosurgical devices.
Effective tumor cryosurgery requires achieving a temperature lethal for all cells throughout the tumor. Although tissue temperature is easy to measure, a degree of uncertainty still exists regarding appropriate tissue temperature goals, owing to different cellular and tissue sensitivities to extremely low temperature. For example, most of the available data on tissue effects in cryosurgery are based on work with normal tissues, such as liver. In contrast, cancer cells seem to be more sensitive to freezing injury than normal cells. However, there are documented differences in the sensitivity of malignant cells to low temperatures, with some being resistant to injury at temperatures as low as −40°C.38–40 Given the data from many in vivo experiments, there is reason to consider the range of −40°C to −50°C as being critical for cell death in vivo after a single freeze-thaw cycle. This is primarily because of the certainty of intracellular ice formation occurring at temperatures below −40°C. For example, posttreatment viability of rat prostate tumor tissue was maximal at freeze rates greater than 10°C/min (see below) to final temperatures below −40°C. In these experiments, the endpoint temperature of less than −40°C was the major factor determining tissue destruction.41,42
The optimal duration for tissue to be held in the frozen state is a matter of some debate. The primary reason for this uncertainty is perhaps that the numerous reported experiments have examined many different temperatures. Taking into consideration much of the evidence, it would seem that duration of the freeze is unimportant if the tissue is held at temperatures below −50°C, whereas holding frozen tissue at temperatures above −40°C for longer times will increase tissue destruction.
In most cases, rapid tissue cooling has been shown to be more destructive than slow cooling.29,43 For this reason, the “rule of thumb” in cryosurgery is to obtain as high a cooling rate as possible with the available technology. However, a review of the published data shows that a wide range of cooling rates are associated with the formation of lethal intracellular ice crystals. These have varied between 50°C/min41 in prostate tumors to 22°C/min44 in experiments with liver slices. From a practical point of view, in cryosurgery rapid freezing (> 50°C/min) only occurs close to the cryosurgical probe. At approximately 1 cm from the probe, the cooling rate might be as low as 10°C/min, depending on the temperature of the probe. Cooling rates of 10°C/min are not likely to form a significant volume of intracellular ice.45 Experimental work in this area, however, supports the view that the cooling rate is not the primary factor determining cell survival after cryotherapy.43
Slow thawing is an important factor in tissue destruction after cryotherapy. The longer the thaw duration, the greater the damage to the cells because of increased solute effects and maximal growth of ice crystals.32,46,47 Current data suggest that slow thawing is a more important mechanism of cell death than rapid cooling.48–50 In addition, complete thawing, which takes full advantage of recrystallization, is the most destructive.
Initial reports on clinical cryosurgery for cancer emphasized repeating the freezing to optimize results.26,27,51 Repetition of the treatment cycle is associated with more extensive and more certain tissue destruction, because cells are subjected to additional deleterious physicochemical changes after they are already weakened by damage sustained in the first cycle. In addition, the intracellular ice crystals are larger in the second freezing cycle,52 potentially causing more damage. With each successive cycle, tissue cooling is faster, the volume of frozen tissue is enlarged, and the area of certain destruction is moved closer to the periphery of the frozen volume. Repetition of the cycle increases the resultant necrosis. Necrosis might be expected to involve approximately 80% of the previously frozen volume.53 Because ultrasound, the key monitoring tool, can only identify the periphery of the frozen volume, a close association between the zone of certain destruction and the periphery of the frozen volume is a distinct advantage in therapy.
The fundamental reason for the association of repeat cycles and an increased volume of certain destruction is that the enhanced lethal effect is best seen at the warmer freezing temperatures, in the −20°C to −30°C range, which exists near the border of the frozen tissue.54–57 In the relatively few scenarios in which the whole tumor and rim of normal tissue can be cooled to temperatures below −50°C, the temperature is sufficiently low to destroy tissue in a single freeze-thaw cycle, so there is no benefit associated with repetition of the cycle.58,59
Treatment Monitoring, Cryogen Type, Probe Size, and Probe Arrangement
As discussed above, the basic features of cryosurgical tissue destruction include: 1) excellent monitoring of the process; 2) fast cooling to a lethal temperature; 3) slow thawing; and 4) repetition of the freeze-thaw cycle. Meeting these basic criteria depends on understanding the imaging technology used to visualize the iceball, the type of cryogen used, the size of the probe, and probe arrangement.
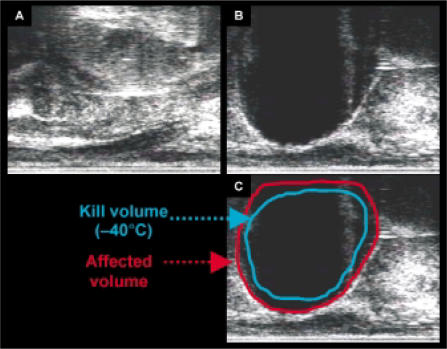
The use of intraoperative ultrasound as a monitoring technique made the current development of visceral cryosurgery possible. However, ultrasound has limitations that must be understood for this instrument to be used appropriately.20,21,60,61 For example, the ultrasound image does not provide information on the temperature isotherms in the frozen area. Therefore, when ultrasound is used to monitor cryotherapy, the tissue destroyed will not correspond to the ultrasound image but instead will be 4 mm to 10 mm away from the edge of the hypoechoic image62 (Figure 6 and Figure 7). This concept and its relationship to probe arrangement and size will be discussed in more detail below. Computed tomography and magnetic resonance imaging can also be used to image frozen tissue.63–68
Figure 6.
Transrectal ultrasound images during prostate cryotherapy with the SeedNet Cryotherapy System. (A) Images before initiation of argon flow. (B) Images after the prostate is engulfed by the iceball. (C) Diagrammatic approximation representing the probable tissue volumes at different temperatures. The “kill volume” −40°C isotherm is only a cartoon approximation. See text and Figure 7 for further details.
Figure 7.
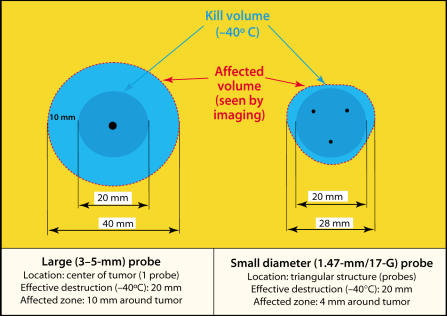
Temperature profiles generated in a gelatin phantom with 3-mm and 1.5-mm (17G) cryoprobes with single and multiple probe arrangements. Figure adapted from data and diagrams provided by Oncura, Inc. (www.oncura.com).
Summary
The choice of cryogen is clearly important. This has been demonstrated throughout the historic development of cryotherapy, with an increasing efficiency of tumor destruction as cryogens were developed that allowed lower and lower tissue temperatures. The coldest cryogen currently used is liquid nitrogen (−196°C). Despite its excellent thermal characteristics, the use of liquid nitrogen is associated with several disadvantages. First, it requires relatively large probes, with diameters greater than 3.5 mm. Second, storage is difficult and is associated with evaporation, resulting in material loss. Third, control of the freezing process, including starting and stopping, is slower than with newer technologies. Last, equipment is large, which affects mobility and easy storage. Owing to the limitations of liquid nitrogen, newer cryosurgical equipment uses argon in a Joule-Thomson-type apparatus. This cryogen freezes quickly and achieves temperatures nearly as low as those achieved with liquid nitrogen.69,70 The probe size can be reduced, with potential benefits, such as reduction in nonlethal damage within an iceball, as discussed in more detail below (Figure 7), and less risk of bleeding when probes are inserted in vascular organs, such as the kidney.
In cryosurgery, the probe is always used as cold as possible, because a large thermal gradient is needed to freeze a volume of tissue as quickly as possible to achieve a lethal temperature some distance from the heat sink. The only exception to this is seen with the use of multiple probes, which might be used at different temperatures to sculpt the frozen volume to a certain shape. This also offers advantages in nonspherical tumors or organs such as the prostate. For these reasons, the performance of cryosurgical probes is important to the production of the freezing injury.
A probe’s freezing capacity depends on its temperature, its surface area of heat transfer, and its thermal conductivity. Tissue conductivity is rarely an important factor because most tissues have high water content, and their thermal conductivity is similar to that of water. Importantly for prostatic cryotherapy, fatty tissues that contain little water have a lower thermal conductivity and therefore are more difficult to freeze, slowing the propagation of the iceball.
Technological advances in probe design and manufacturing have allowed the adoption of argon as the primary cryogen for most urologic procedures. A progressive decrease in probe diameter has been concurrent with this development (Figure 2B). Initial liquid nitrogen probes were 5 mm in diameter. Currently, the thinnest third-generation probes are only 17-gauge, comparable to a prostate biopsy needle. These advances allow great flexibility in covering organs or tumors of various shapes. In addition, there is evidence to suggest that when a given volume of tumor is subjected to lethal temperatures by three smaller probes, compared with one larger probe (Figure 7), the affected volume is greater with the multiple probe array. Because ultrasound can only detect the nonlethal edge of the iceball, minimizing the ratio between the “affected volume” and the “treated volume” is advantageous.
Main Points.
Successful cryosurgical tissue destruction depends on four criteria: excellent monitoring of the process; fast cooling to a lethal temperature; slow thawing; and repetition of the freeze-thaw cycle.
Data from many in vivo experiments indicate the range of −40°C to −50°C as being critical for cell death in vivo after a single freeze-thaw cycle.
The “rule of thumb” in cryosurgery is to obtain as high a cooling rate as possible with the available technology.
The longer the thaw duration, the greater the damage to the cells; current data suggest that slow thawing is a more important mechanism of cell death than rapid cooling.
Repetition of the treatment cycle is associated with more extensive and more certain tissue destruction, because cells are subjected to additional deleterious physicochemical changes after they are already weakened by damage sustained in the first cycle.
Technological advances in probe design and manufacturing have allowed the adoption of argon as the primary cryogen for most urologic procedures, and a progressive decrease in probe diameter has been concurrent with this development. Currently, the thinnest probes are only 17-gauge, comparable to a prostate biopsy needle, and thus allow great flexibility in covering organs or tumors of various shapes.
Compressed argon gas for cryotherapy offers many advantages over liquid nitrogen: temperatures are equivalent to those of liquid nitrogen; the freezing process can be started and stopped almost instantaneously; argon is easily stored without evaporation and is ready for immediate use; and cryotherapy control and monitoring units and associated tubing for argon are much smaller and thus are easily transported from room to room or facility to facility.
References
- 1.Arnott J. Practical illustrations of the remedial efficacy of a very low or anesthetic temperature. I. In cancer. Lancet. 1850;2:257–259. [PMC free article] [PubMed] [Google Scholar]
- 2.Fay T. Early experiences with local and generalized refrigeration of the human brain. J Neurosurg. 1959;16:239–259. doi: 10.3171/jns.1959.16.3.0239. [DOI] [PubMed] [Google Scholar]
- 3.Allington HV. Liquid nitrogen in the treatment of skin diseases. Calif Med. 1950;72:153–155. [PMC free article] [PubMed] [Google Scholar]
- 4.Zacarian SA, Adham MI. Cryotherapy of cutaneous malignancy. Cryobiology. 1966;2:212–218. doi: 10.1016/s0011-2240(66)80170-0. [DOI] [PubMed] [Google Scholar]
- 5.Hass GM, Taylor CB. A quantitative hypothermal method for the production of local injury of tissue. Arch Pathol. 1948;45:563–580. [Google Scholar]
- 6.Cooper IS, Lee A. Cryostatic congelation: a system for producing a limited controlled region of cooling or freezing of biologic tissues. J Nerv Ment Dis. 1961;133:259–263. [PubMed] [Google Scholar]
- 7.Cooper I. Cryogenic surgery of the basal ganglia. JAMA. 1962;181:600–605. doi: 10.1001/jama.1962.03050330030006. [DOI] [PubMed] [Google Scholar]
- 8.Gonder MJ, Soanes WA, Smith V. Experimental prostate cryosurgery. Invest Urol. 1964;1:610–619. [PubMed] [Google Scholar]
- 9.Gonder MJ, Soanes WA, Shulman S. Cryosurgical treatment of the prostate. Invest Urol. 1966;3:372–378. [PubMed] [Google Scholar]
- 10.Ortved WE, O’Kelly FM, Todd IA, et al. Cryosurgical prostatectomy: a report of 100 cases. Br J Urol. 1967;39:577–583. doi: 10.1111/j.1464-410x.1967.tb11797.x. [DOI] [PubMed] [Google Scholar]
- 11.Jordan WP, Jr, Walter D, Miller GH, Jr, Drylie D. Cryotherapy of benign and neoplastic tumors of the prostate. Surg Gynecol Obstet. 1967;125:1265–1268. [PubMed] [Google Scholar]
- 12.Dowd JB, Flint LD, Zinman LN, Tripathi VN. Experiences with cryosurgery of the prostate in the poor-risk patient. Surg Clin North Am. 1968;48:627–632. doi: 10.1016/s0039-6109(16)38545-0. [DOI] [PubMed] [Google Scholar]
- 13.Rouvalis P. Cryosurgery of the prostate under local anesthesia. J Urol. 1969;102:244–245. doi: 10.1016/s0022-5347(17)62122-0. [DOI] [PubMed] [Google Scholar]
- 14.Flocks RH, Nelson CM, Boatman DL. Perineal cryosurgery for prostatic carcinoma. J Urol. 1972;108:933–935. doi: 10.1016/s0022-5347(17)60910-8. [DOI] [PubMed] [Google Scholar]
- 15.Torre D. Alternate cryogens for cryosurgery. J Dermatol Surg. 1975;1:56–58. doi: 10.1111/j.1524-4725.1975.tb00073.x. [DOI] [PubMed] [Google Scholar]
- 16.Soanes WA, Ablin RJ, Gonder MJ. Remission of metastatic lesions following cryosurgery in prostatic cancer: immunologic considerations. J Urol. 1970;104:154–159. doi: 10.1016/s0022-5347(17)61690-2. [DOI] [PubMed] [Google Scholar]
- 17.Gursel E, Roberts M, Veenema R. Regression of prostate cancer following sequential cryotherapy to the prostate. J Urol. 1972;108:928–932. doi: 10.1016/s0022-5347(17)60909-1. [DOI] [PubMed] [Google Scholar]
- 18.Bonney WW, Fallon B, Gerber WL, et al. Cryosurgery in prostatic cancer: survival. Urology. 1982;19:37–42. doi: 10.1016/0090-4295(82)90042-5. [DOI] [PubMed] [Google Scholar]
- 19.Loening S, Lubaroff D. Cryosurgery and immunotherapy for prostatic cancer. Urol Clin North Am. 1984;11:327–336. [PubMed] [Google Scholar]
- 20.Onik G, Gilbert J, Hoddick W, et al. Sonographic monitoring of hepatic cryosurgery in an experimental animal model. AJR Am J Roentgenol. 1985;144:1043–1047. doi: 10.2214/ajr.144.5.1043. [DOI] [PubMed] [Google Scholar]
- 21.Onik G, Cobb C, Cohen J, et al. US characteristics of frozen prostate. Radiology. 1988;168:629–631. doi: 10.1148/radiology.168.3.3043544. [DOI] [PubMed] [Google Scholar]
- 22.Baust J, Chang Z. Underlying mechanisms of damage and new concepts in cryosurgical instrumentation. In: Baust J, Chang Z, editors. Cryosurgery: Mechanism and Applications. Paris, France: Institut international de froid; 1995. pp. 21–36. [Google Scholar]
- 23.Kreyberg L. Development of acute tissue damage due to cold. Physiol Rev. 1949;29:156–167. doi: 10.1152/physrev.1949.29.2.156. [DOI] [PubMed] [Google Scholar]
- 24.Mazur P. Kinetics of water loss from cells at subzero temperatures and the likelihood of intracellular freezing. J Gen Physiol. 1963;47:347–369. doi: 10.1085/jgp.47.2.347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cooper IS. Cryobiology as viewed by the surgeon. Cryobiology. 1964;1:44–54. doi: 10.1016/0011-2240(64)90019-7. [DOI] [PubMed] [Google Scholar]
- 26.Cahan W. Cryosurgery of malignant and benign tumors. Fed Proc. 1965;24:S241–S248. [PubMed] [Google Scholar]
- 27.Cooper IS. Cryogenic surgery for cancer. Fed Proc. 1965;24:S237–S240. [PubMed] [Google Scholar]
- 28.Taylor MJ. Physico-chemical principles in low temperature biology. In: Grout BWW, Morris GJ, editors. The Effects of Low Temperatures on Biological Systems. London, England: Edward Arnold; 1987. pp. 3–71. [Google Scholar]
- 29.Mazur P. Physical-chemical factors underlying cell injury in cryosurgical freezing. In: Rand R, Rinfret A, von Leden H, editors. Cryosurgery. Springfield, IL: Charles C. Thomas; 1968. pp. 32–51. [Google Scholar]
- 30.Mazur P. The role of intracellular freezing in the death of cells cooled at supraoptimal rates. Cryobiology. 1977;14:251–272. doi: 10.1016/0011-2240(77)90175-4. [DOI] [PubMed] [Google Scholar]
- 31.Mazur P. Fundamental cryobiology and the preservation of organs by freezing. In: Karow A, Pegg D, editors. Organ Preservation for Transplantation. New York, NY/Basel, Switzerland: Dekker; 1981. pp. 143–175. [Google Scholar]
- 32.Mazur P. Freezing of living cells: mechanisms and implications. Am J Physiol. 1984;143:C125–C142. doi: 10.1152/ajpcell.1984.247.3.C125. [DOI] [PubMed] [Google Scholar]
- 33.Mundth ED. Studies on the pathogenesis of cold injury: microcirculatory changes in tissue injured by freezing. Proc Symp Arctic Biol Med. 1964;4:51–72. [Google Scholar]
- 34.Quintanella R, Krusen F, Essex H. Studies on frostbite with special reference to treatment and the effect on minute blood vessels. Am J Physiol. 1947;149:149–161. doi: 10.1152/ajplegacy.1947.149.1.149. [DOI] [PubMed] [Google Scholar]
- 35.Bellman S, Ray JA. Vascular reactions after experimental cold injury. Angiology. 1956;7:339–367. doi: 10.1177/000331975600700404. [DOI] [PubMed] [Google Scholar]
- 36.Bowers WD, Hubbard RW, Daum RC, et al. Ultrastructural studies of muscle cells and vascular endothelium immediately after freeze-thaw injury. Cryobiology. 1973;10:9–21. doi: 10.1016/0011-2240(73)90003-5. [DOI] [PubMed] [Google Scholar]
- 37.Giampapa V, Oh C, Aufses A. The vascular effect of cold injury. Cryobiology. 1981;18:49–54. doi: 10.1016/0011-2240(81)90005-5. [DOI] [PubMed] [Google Scholar]
- 38.Heard BE. Nuclear crystals in slowly frozen tissues at very low temperatures: comparison of normal and ascites tumour cells. Br J Surg. 1955;42:659–663. doi: 10.1002/bjs.18004217617. [DOI] [PubMed] [Google Scholar]
- 39.Tatsutani K, Rubinsky B, Onik G, Dahiya R. The effect of thermal variables on frozen human prostatic adenocarcinoma cells. Urology. 1996;48:441–447. doi: 10.1016/S0090-4295(96)00199-9. [DOI] [PubMed] [Google Scholar]
- 40.Yamada S, Tsubouchi S. Rapid cell death and cell population recovery in mouse skin epidermis after freezing. Cryobiology. 1976;13:317– 327. doi: 10.1016/0011-2240(76)90113-9. [DOI] [PubMed] [Google Scholar]
- 41.Bischof JC, Smith D, Pazhayannur PV, et al. Cryosurgery of Dunning AT-1 rat prostate tumor: thermal, biophysical, and viability response at the cellular and tissue level. Cryobiology. 1997;34:42–69. doi: 10.1006/cryo.1996.1978. [DOI] [PubMed] [Google Scholar]
- 42.Roberts KP, Smith DJ, Ozturk H, et al. Biochemical alterations and tissue viability in AT-1 prostate tumor tissue after in vitro cryodestruction. Cryo Letters. 1997;18:241–250. [Google Scholar]
- 43.Farrant J, Walter CA. The cryobiological basis for cryosurgery. J Dermatol Surg Oncol. 1977;3:403–407. doi: 10.1111/j.1524-4725.1977.tb00319.x. [DOI] [PubMed] [Google Scholar]
- 44.Bischof JC, Cristov K, Rubinsky B. A morphological study of the cooling rate response of normal and neoplastic human liver tissue: cryosurgical implications. Cryobiology. 1993;30:482–492. doi: 10.1006/cryo.1993.1049. [DOI] [PubMed] [Google Scholar]
- 45.McGrath JJ, Caravalho EG, Huggins CE. An experimental comparison of intracellular ice formation and freeze-thaw survival of HeLa S-3 cells. Cryobiology. 1975;12:540–550. doi: 10.1016/0011-2240(75)90048-6. [DOI] [PubMed] [Google Scholar]
- 46.Meryman HT. Review of biological freezing. In: Meryman HT, editor. Cryobiology. New York: Academic Press; 1996. pp. 1–114. [Google Scholar]
- 47.Whittaker D. Electron microscopy of the ice crystals formed during cryosurgery: relationship to duration of freeze. Cryobiology. 1978;15:603– 607. doi: 10.1016/0011-2240(78)90084-6. [DOI] [PubMed] [Google Scholar]
- 48.Gage AA, Guest K, Montes M, et al. Effect of varying freezing and thawing rates in experimental cryosurgery. Cryobiology. 1985;22:175–182. doi: 10.1016/0011-2240(85)90172-5. [DOI] [PubMed] [Google Scholar]
- 49.Neel HB, Ketcham AS, Hammond WG. Cryonecrosis of normal and tumor-bearing rat liver potentiated by occlusion. Cancer. 1971;28:1211–1218. doi: 10.1002/1097-0142(1971)28:5<1211::aid-cncr2820280519>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- 50.Whittaker DK. Mechanisms of tissue destruction following cryosurgery. Ann R Coll Surg Engl. 1984;66:313–318. [PMC free article] [PubMed] [Google Scholar]
- 51.Gage AA, Koepf S, Wehrle D, Emmings F. Cryotherapy for cancer of the lip and oral cavity. Cancer. 1965;18:1646–1651. doi: 10.1002/1097-0142(196512)18:12<1646::aid-cncr2820181221>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 52.Whittaker DK. Repeat freeze cycles in cryosurgery of oral tissues. Br Dent J. 1975;139:459–465. doi: 10.1038/sj.bdj.4803667. [DOI] [PubMed] [Google Scholar]
- 53.Dilley AV, Dy DY, Walters A, et al. Laboratory and animal model evaluation of the Cryotech LCS 2000 in hepatic cryosurgery. Cryobiology. 1993;30:74–85. doi: 10.1006/cryo.1993.1007. [DOI] [PubMed] [Google Scholar]
- 54.Myers RS, Hammond WG, Ketcham AS. Cryosurgery of experimental tumors. J Cryosurg. 1969;2:225–228. [Google Scholar]
- 55.Neel HB, Ketcham AS, Hammond WG. Requisites for successful cryogenic surgery of cancer. Arch Surg. 1971;102:45–48. doi: 10.1001/archsurg.1971.01350010047012. [DOI] [PubMed] [Google Scholar]
- 56.Rand RW, Rand RP, Eggerding F, et al. Cryolumpectomy for carcinoma of the breast. Surg Gynecol Obstet. 1987;165:392–396. [PubMed] [Google Scholar]
- 57.Ravikumar TS, Steele G, Kane R, King V. Experimental and clinical observations on hepatic cryosurgery for colorectal metastases. Cancer Res. 1991;51:6323–6327. [PubMed] [Google Scholar]
- 58.Gage AA. Experimental cryogenic injury of the palate: observations pertinent to the cryosurgical destruction of tumors. Cryobiology. 1978;15:415–425. doi: 10.1016/0011-2240(78)90060-3. [DOI] [PubMed] [Google Scholar]
- 59.Natiella JR, Gage AA, Armitage JE, Greene GW. Tissue response to cryosurgery of oral cavity in rhesus monkeys. Arch Pathol. 1974;98:183–188. [PubMed] [Google Scholar]
- 60.Lam CM, Shimi SM, Cuschieri A. Ultrasonic characterization of hepatic cryolesions. Arch Surg. 1995;130:1068–1072. doi: 10.1001/archsurg.1995.01430100046010. [DOI] [PubMed] [Google Scholar]
- 61.Littrup P, Monti J, Mody A, et al. Prostatic cryotherapy: ultrasonic and pathologic correlation in the canine model. Urology. 1994;44:175–184. doi: 10.1016/s0090-4295(94)80123-1. [DOI] [PubMed] [Google Scholar]
- 62.Grampsas SA, Miller GJ, Crawford ED. Salvage radical prostatectomy after failed transperineal cryotherapy: histologic findings from prostate whole mount specimens correlated with intraoperative transrectal ultrasound images. Urology. 1995;45:936–941. doi: 10.1016/s0090-4295(99)80111-3. [DOI] [PubMed] [Google Scholar]
- 63.Gilbert JC, Rubinsky B, Roos MS, et al. MRI monitored cryosurgery in the rabbit brain. Magn Reson Imaging. 1993;11:1155–1164. doi: 10.1016/0730-725x(93)90243-7. [DOI] [PubMed] [Google Scholar]
- 64.Hong JS, Wong S, Pease G, Rubinsky B. MR imaging assisted temperature calculations during cryosurgery. J Magn Reson Imaging. 1994;12:1021–1031. doi: 10.1016/0730-725x(94)91233-m. [DOI] [PubMed] [Google Scholar]
- 65.Liang XG, Ge XS, Zhang YP, Wang GJ. A convenient method of measuring the thermal conductivity of biological tissue. Phys Med Biol. 1991;36:1599–1605. doi: 10.1088/0031-9155/36/12/005. [DOI] [PubMed] [Google Scholar]
- 66.Matsumoto R, Selig AM, Coluca MS, Jolesz GA. MR monitoring during cryotherapy in the liver: predictability of histologic outcome. J Magn Reson Imaging. 1993;3:770–776. doi: 10.1002/jmri.1880030513. [DOI] [PubMed] [Google Scholar]
- 67.Pease GR, Wong STS, Roos MS, Rubinsky B. MR image-guided control of cryosurgery. J Magn Reson Imaging. 1995;5:753–760. doi: 10.1002/jmri.1880050623. [DOI] [PubMed] [Google Scholar]
- 68.Rivoire ML, Voiglio EJ, Kaemmerlen P, et al. Hepatic cryosurgery precision: evaluation of ultrasonography, thermometry, and impedancemetry in a pig model. J Surg Oncol. 1996;61:242–248. doi: 10.1002/(SICI)1096-9098(199604)61:4<242::AID-JSO2>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 69.Hewitt PM, Zhao J, Akhter J, Morris DL. A comparative laboratory study of liquid nitrogen and argon gas cryosurgery systems. Cryobiology. 1997;35:303–308. doi: 10.1006/cryo.1997.2039. [DOI] [PubMed] [Google Scholar]
- 70.Homasson JP, Thiery JP, Angebault M, et al. The operative efficacy of cryosurgical nitrous oxide-driven cryoprobe. Cryobiology. 1994;31:290–304. doi: 10.1006/cryo.1994.1035. [DOI] [PubMed] [Google Scholar]