Abstract
Primary care clinicians are often responsible for the treatment of lower urinary tract symptoms (LUTS) and benign prostatic hyperplasia (BPH). The combination of the compelling epidemiologic presence of BPH, compromised quality of life due to LUTS, and the availability of highly effective oral therapeutic agents, offers an opportunity for substantial clinical impact in the primary care setting. Evolving management pathways include utilization of both symptom-modifying treatment and disease-modifying treatment. Alpha blockers are excellent to provide symptomatic treatment, but do not alter long-term disease progression. Alpha reductase inhibitors provide therapy by reducing the need for surgical intervention and the incidence of acute urinary retention. The combination of alpha blockers and alpha reductase inhibitors will be best for some patients, typically those with large prostate glands in whom disease progression is most likely. Primary care clinicians will want to become more familiar with recent clinical trails that are shaping emerging therapeutic practice.
Key words: Primary care clinicians, Lower urinary tract symptoms, Benign prostatic hyperplasia, 5α-reductase inhibitors, Dihydrotestosterone
Although benign prostatic hyperplasia (BPH) can be asymptomatic, it is often associated with problematic symptoms. The label “LUTS” (lower urinary tract symptoms) has been applied to any one or more of the symptoms of nocturia, urgency, frequency, a sensation of not completely emptying the bladder, stop-start urination, straining to urinate, a need to urinate soon after voiding, and weak urinary stream.1 These symptoms are often associated with BPH, though they may also be associated with various unrelated syndromes such as heart failure, urinary tract infections, and diabetes (see Table 1). Laboratory data should be obtained to ensure that another secondary cause is not masquerading as BPH-LUTS. In this paper, BPH-LUTS refers specifically to the symptom complex of LUTS associated with BPH.
Table 1.
LUTS and Differential Diagnosis
| SYMPTOM | |||||||
|---|---|---|---|---|---|---|---|
| Incomplete | Weak | Stop/ | Sexual | ||||
| Disorder | Frequency | Emptying | Stream | Nocturia | Urgency | Start | Dysfunction |
| BPH | + | + | + | + | + | + | + |
| UTI | + | + | + | ||||
| Prostatitis | + | + | + | + | + | ||
| Prostate CA | + | + | + | + | + | + | |
| Diabetes mellitus | + | + | + | ||||
| Diabetes insipidus | + | + | |||||
| Heart failure | + | ||||||
| Psychogenic water | + | + | |||||
| Bladder CA | + | + | + | ||||
LUTS, lower urinary tract symptoms; BPH, benign prostatic hyperplasia; UTI, urinary tract infection; CA, cancer.
The tools for diagnosis, as well as the varying pharmacotherapies to manage BPH, are readily available to primary care clinicians (PCCs). Treating BPH pharmacologically with 5α-reductase inhibitors (5ARIs) and α-blockers provides high levels of clinical success and patient satisfaction. These pharmacotherapies for BPH-LUTS are generally safe, and rarely have adverse effects that result in drug discontinuation. Alpha blockers may be categorized as symptom modifying as they relieve LUTS without eliciting a direct effect on the hyperplastic process. 5ARIs are both symptom and disease modifying because over the long term they both improve symptoms and cause BPH regression; the disease modifying impact is reflected in a reduction in need for invasive surgical interventions, and a reduced incidence of acute urinary retention (AUR). Because BPH is not a disorder that negatively affects mortality, treatment goals should include control of symptoms along with the prevention of AUR, need for invasive treatment, and hospitalization.
Epidemiology
BPH, which usually develops after age 40, is the most common benign neoplasm of aging men.2,3 Approximately 25% of men will be treated for BPH by age 80,4 despite the fact that twice this proportion will have suffered clinically significant symptoms by this age. It is estimated that more than one third of men ages 50 years or older will undergo surgery, medical intervention, or both for BPH during their lifetime.3 The majority of men over age 60 demonstrate histologic changes associated with BPH, although only about two thirds have referable symptoms, and only about one half are affected enough to perceive an impaired quality of life.4 Even in young men (ages 30 to 40 years), autopsy data (N = 1075) have demonstrated the presence of histologic BPH in as many as 8%.5 If trends prevail, about 20% of symptomatic patients will eventually require surgical intervention.6
Pathophysiology
In this communication, BPH refers to a clinically enlarged gland. Microscopic BPH refers to the histologic changes of stromal hyperplasia. Microscopic hyperplasia is found in up to 10% of men by age 40 and in almost 90% of men by age 80, but is inconsistently associated with symptoms.7 For purposes of clinical discussion, a gland volume > 30 cc may be considered enlarged.
Normal prostate tissue is composed of stroma (smooth muscle plus connective tissue) and epithelium (the glands), which are typically found in a 2:1 ratio. In BPH, not only are the number of cells increased (thus, the term hyperplasia), but there is tissue distortion in which the normal 2:1 stroma:epithelium ratio is altered to 5:1.8 Stromal smooth muscle proliferation appears to be responsible for the “dynamic component” of the symptomatology of BPH.9 In other words, although macroscopic gland growth produces an element of mechanical obstruction to the urethra, smooth muscle contraction at the bladder neck (the dynamic component) appears to also play a major role. Hyperplastic prostatic smooth muscle contracts in response to α-adrenergic stimuli, resulting in compromise of the urethral lumen, inhibiting urinary flow and bladder emptying. The pathologic combination of an excessive preponderance of muscle mass leads to the BPH-LUTS complex. Both prostatic urethral smooth muscle and bladder neck smooth muscle are innervated by α-adrenergic fibers. Alpha-adrenergic agonists may worsen symptoms of urinary outlet obstruction, but α-adrenergic antagonists usually improve symptoms.10
It remains unclear why the majority of men develop some degree of BPH during their lifetime, but it has been shown that the presence of the prostatic androgen dihydrotestosterone (DHT) is fundamental to its development.11,12 A clear example of this relationship is that men who undergo castration prior to age 40 do not develop BPH.9
Diagnosis and Differential Diagnosis
The most widely employed and validated scoring system for quantifying and monitoring of BPH-LUTS is the American Urological Association symptom index (AUA-SI). Based on 7 questions (scored 1–5 for increasing severity), the index quantifies the most consistently identified LUTS referable to BPH: nocturia, frequency, urgency, hesitancy, incomplete emptying, straining, and stop-start urination. In addition to the utility of the AUA-SI for monitoring symptom progression in clinical trials, clinicians may find this symptom score a useful way to quantitate and monitor LUTS.
The initial obstacle to managing BPH-LUTS is identification of the symptom complex. A survey of primary care patients listed prostate disorders as fourth among the top 10 most embarrassing disorders to discuss (Table 2).5 Numerous factors inhibit full disclosure of LUTS by patients, including:
Table 2.
Top 10 Clinical Entities Patients Find Most Embarrassing to Discuss With Their Family Physician
|
Data from Preboth MA et al.5
Some patients think that LUTS are just a normal part of growing older.
Symptom progression is insidious, allowing substantial periods of benign neglect.
Stories of problems caused by surgical treatments that were more frequently employed in the recent past may preclude initiating discussion, for fear of having to undergo such a procedure.
Historically, less well tolerated medical treatments (eg, first generation alpha blockers) may have led the patient to conclude that “medical treatment” was too problematic to be worth the bother.
To some men, the prostate is a “virility gland”; in these men, acknowledging prostate-related symptomatology may be tantamount to signaling impaired virility.
In the author’s experience, men between the ages of 15 and 50 follow the “used car” model of healthcare: they don’t come in unless parts are falling off in the street! Because men do not have the same ongoing link with the healthcare system as women (PAP testing, mammography, prescribed contraception), they may neglect their health, or enter the healthcare system only upon the insistence of their significant other.
The medical community has made patients well aware of the prototypic symptoms and signs of myocardial infarction, stroke, diabetes, breast cancer, and colon cancer. These “important diseases” occupy a major priority in ambulatory setting discussions, with a much lower priority given to disorders such as LUTS.
Clinical Evaluation
Most men have had symptoms for many years before seeking help from the PCC. When a patient does present with LUTS, secondary causes need to be ruled out. Other than BPH the 3 most common etiologies for nocturia and frequency are urinary tract infection (UTI), heart failure, and diabetes.
Laboratory tests. A urinalysis should be obtained for every patient who presents with LUTS. Initially, a dipstick urinalysis should be sufficient. Absence of nitrite, leukocyte esterase, and white blood cells essentially rules out UTI. Borderline urinalysis results should be repeated and/or followed with a urine culture. Although the benchmark for cultural positivity is 100,000 bacteria per milliliter, some clinicians consider lower colony counts (25,000 to 50,000 bacteria/mL) indicative of UTI, particularly when a single plausible organism is recovered.
Glucosuria or proteinuria may cause urinary frequency or nocturia as well. When either of these factors is present in sufficient degree to induce LUTS, urine dipstick results are positive. Prompt treatment of newly diagnosed diabetes can resolve or dramatically reduce the LUTS arising from glucosuria. Glucosuria insufficient to produce a positive dipstick response on urinalysis is not sufficient to induce LUTS. Proteinuria to a degree that results in nocturia or frequency essentially always indicates significant underlying renal disease, which will require prompt elucidation.
Occasionally, heart failure manifests solely as nocturia or (rarely) increased urinary frequency, although the symptom constellation of exertional dyspnea, peripheral edema, shortness of breath, fatigue, and cough are more typical manifestations. If any question about heart failure as a contributor or confounder to LUTS exists, simple physical examination may not be sufficient to ensure the absence of ventricular dysfunction. Brain natriuretic peptide testing, an inexpensive and widely available blood test, is capable of determining the presence or absence of heart failure with accuracy that closely rivals echocardiography.
Of course, diuretic medications are an obvious inducer of urinary frequency, but occasionally patients will have low awareness that their “anti-hypertensive pill” is indeed a diuretic.
Physical examination. Physical examination should include a prostate examination. Primary care clinicians do not generally estimate prostate size in grams, as is the custom by urologists. Normal prostate glands are approximately 20 grams or less, and significant enlargement is considered 35 grams or greater. Primary care clinicians usually do not have the availability of prostatic ultrasound directly after rectal examination to confirm their diagnostic impression of gland size. Hence, simply indicating whether palpation suggests moderate or severe gland enlargement is sufficient. It is useful to estimate gland size, because the larger the gland, the more likely disease progression will occur, and the greater responsivity to treatment with α-reductase inhibitors.6 Although the value of prostate-specific antigen (PSA) testing as a screening tool remains controversial in asymptomatic men, since prostate cancer may present as LUTS, it is appropriate to obtain a PSA measurement for these symptomatic men. Recent data indicate that PSA levels correlate sufficiently well with overall gland size to predict responsivity to 5ARIs; large glands (PSA level > 1.7 ng/mL) are more likely than smaller glands to regress with α-reductase inhibitors. Similarly, increased gland size predicts men most likely to enjoy the benefits of reduced hospitalization, reduced need for surgical procedures, and incidence of AUR through treatment with α-reductase inhibitors.6
The accuracy of digital rectal examination for the determination of prostate volume is imperfect. PSA levels increase with increasing prostate size and may function as a surrogate to estimate prostate volume, as well as to serve as a predictor for risk of urinary retention and the need for surgical intervention.13 However, one should not assume that an abnormally high PSA level correlates with increased benign gland size until prostate cancer has been ruled out. The current standard of care suggests that men with a PSA level > 4 ng/mL be considered for urologic referral. In persons with a positive family history of prostate cancer, and in African American men, an even lower PSA threshold (eg, > 2.5 ng/mL) may be warranted.
When testing PSA, some clinicians have been concerned that a rectal examination might spuriously alter the subsequent PSA measurement. At least 2 clinical trials have shown that although there is a statistically measurable change in PSA after rectal examination, it is insufficient to make a meaningful change in PSA levels (mean post-examination PSA change = 0.26).
When PSA is elevated into the “intermediate” category (PSA = 4–10 ng/mL) PSA density, PSA velocity, and free PSA concentration may help the clinician distinguish benign from malignant sources of the elevated PSA level, but, ultimately, prostate biopsy must be considered for most men with increased PSA concentrations. In men over age 50 with a total PSA concentration between 4 and 10 ng/mL, a lower ratio of free PSA to total PSA is associated with a higher risk of prostate cancer; free PSA levels greater than 25% support a benign process.14
Impact of BPH on Sexual Function
BPH is associated with sexual dysfunction, independent of age. The Multinational Survey of the Aging Male15 demonstrated that severity of LUTS in men with BPH correlates with sexual dysfunction. Most men with clinical BPH report that LUTS affect both sleep pattern and sexual activity.
Management
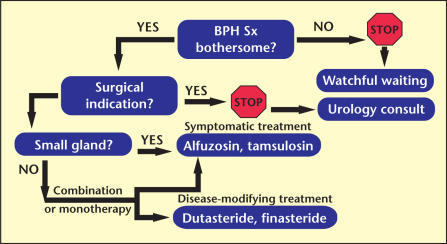
Not all patients with BPH-LUTS require treatment (see Figure 1). For some, reassurance that symptoms are not caused by cancer, combined with an explanation of the underlying disorder and its natural history, will suffice. When BPH-LUTS symptoms have been unmasked by pharmacotherapies for other disorders, a modulation of pharmacotherapy may be sufficient to provide adequate relief. For instance, when an alpha agonist has been part of a recent regimen for an upper respiratory infection, and hesitancy of urination occurs, simply omitting the alpha agonist will generally resolve the problematic symptoms.
Figure 1.
Benign prostatic hyperplasia (BPH) management algorithm. Sx, symptoms.
Regardless of the AUA symptom score, clinicians have been discouraged from offering intervention to a patient who does not express “bother” occasioned by symptoms. The AUA assembled a group of urological experts to develop guidelines for the management of BPH-LUTS. It has been suggested in the most recent iteration of the AUA guidelines (2003) that clinicians might consider 5ARI treatment in men with symptomatic BPH without bother. Specifically, 2 options could be considered: One option is that the 5ARIs finasteride and dutasteride are appropriate and effective treatments for patients with LUTS associated with demonstrable prostatic enlargement. And the second option is that patients with asymptomatic prostatic enlargement may be offered a 5ARI to prevent progression of the disease.
Assuming that the patient has expressed a significant amount of bother from BPH, the patient may be appropriately offered either symptomatic relief with an α-blocker, disease-modifying therapy with a 5ARI, or both, depending on the baseline prostate size. In unusual circumstances, invasive treatments may be considered as an initial option. It is generally recommended that all patients be offered medical therapy, unless absolutely contraindicated, before opting for invasive management.
α-Blockers
Treatment with α-blockers typically increases a peak flow rate up to 3 mL/sec and decreases AUA-SI by approximately 6 points.16 Data from a meta-analysis demonstrate that α-blocker treatment of BPH results in a mean 51% improvement in symptom scores in 59% to 86% of patients.1 By comparison, surgical interventions such as transurethral resection of the prostate may resolve symptoms in 90% of patients, with 70% to 85% improvement in AUA symptom scores.17,18 Despite the greater efficacy of surgical interventions, postsurgical complications such as impotence and incontinence are the reasons most men prefer to select nonsurgical options as their initial therapy.
The responsiveness of prostate adenoma smooth muscle to α-adrenergic stimulation is as much as 40-fold greater than that of normal bladder smooth muscle. Fortunately, the α1-adrenergic smooth muscle receptors are more densely populated in the prostate and bladder neck than in the bladder itself; otherwise α-blockers might induce decompensation of bladder wall contractility.19
Although α-blocker treatment will not ultimately alter the benign growth of the prostate, it is often satisfactory for symptom control over long time periods. Most men will achieve a clinically significant improvement in AUA symptom scores following initiation of α-blocker therapy. Most patients will elect to begin treatment with an α blocker. Of the currently available once daily non-selective α blockers, terazosin and doxazosin are equally appropriate initial choices. Because many men with BPH-LUTS also are receiving phosphodiesterase type-5 inhibitors for management of erectile dysfunction, care must be exercised to avoid drug interactions.
Both doxazosin and terazosin require titration over about 4 to 6 weeks. Dosing, and especially initial dosing, should be administered at bedtime, due to the uncommon but troublesome first-dose syncope reported with these agents. Indeed, if there is a hiatus in treatment greater than 72 hours, it is recommended that dosing resume at the initial dose to avoid a recurrence of first-dose syncope.
Occasionally, a patient will respond to one α-blocker but not another. In addition to ameliorating urinary symptoms, treatment with α-blockers may also improve sexual function, with the exception of ejaculation disorders that are predominantly associated with subtype-selective α-blockers, eg, tamsulosin.
Tamsulosin is a third-generation uroselective member of the pharmacotherapeutic class known as selective α-1-A adrenergic antagonists. Lack of affinity for α-receptors in the vascular wall smooth muscle affords tamsulosin freedom from first-dose syncope and minimal clinically relevant hypotensive effect. Because the initial therapeutic dose is the full therapeutic dose, no titration is necessary, which is especially helpful in persons with comorbid disorders (not related to BPH) that may contribute to LUTS.
All α-blockers can potentially induce retrograde ejaculation due to their capacity to relax the internal sphincter and bladder neck. Keeping in mind that ejaculation is a genital disorder, and orgasm a central nervous system phenomenon, it should be clear that altered ejaculation, such as retrograde ejaculation, does not alter the sensation of orgasm. Nonetheless, some patients (and/or their partners) may be dismayed by retrograde ejaculation. Although it is reported in almost 20% of patients receiving high dose tamsulosin (0.8 mg/d), this dose is taken by fewer than 5% of patients in the primary care setting and retrograde ejaculation results in discontinuation in less than 1% of patients. Alpha-blocker treatment does not appear to induce tachyphylaxis and may be continued indefinitely. Hence patients, once stable, may be considered for annual follow-up.
Disease- and Symptom-Modifying Therapy: 5ARIs
By preventing the conversion of testosterone to DHT, 5ARIs dramatically reduce the primary trophic stimulus to prostate growth. Within 6 to 12 months of consistent 5ARI therapy, prostate size is decreased by 15% to 25%. Indeed, it is recommended that the nadir PSA levels obtained after 6 months of continuous 5ARI treatment be doubled for future calculation of prostate cancer screening. Currently there are 2 identified isoforms of the enzyme for converting testosterone into DHT. Finasteride is highly effective at blocking 5ARI type 2; dutasteride, on the other hand, blocks both type 1 and type 2 isoforms of the 5-α-reductase enzyme, resulting in lower levels of intraprostatic DHT compared with finasteride. 5ARIs have been shown to be the most effective in men with prostate glands of at least 45 grams. Although alpha blockers effectively control symptoms, they do not ultimately change the course of the disease and thus combining them with 5ARIs becomes a logical approach.
A large Veterans Affairs Cooperative Study enrolled 1229 men in a comparison trial of finasteride and terazosin. Results indicated that symptom relief and improvement in urinary flow rates over 1 year were better with terazosin, that finasteride was no more effective than placebo in improving symptom scores, and that the combination of finasteride and terazosin was no more effective than terazosin alone. This trial has been widely misinterpreted. Because we know that alpha reductase inhibitors preferentially impact men with large prostate glands, and this trial had a substantial population of minimally enlarged glands, it should not come as a surprise that alpha blockers would emerge as most satisfactory in such a population, and especially since the trial was of such short duration (maximum 1 year treatment).
In contrast, a recent placebo-controlled trial studied more than 3000 men with BPH taking finasteride 5 g/day. Over 4 years, finasteride treatment resulted in a 55% reduction in surgery for BPH and a 57% reduction in episodes of AUR.20 Men who completed the study showed improvements in AUA symptom score and urinary flow rates. The patients enrolled in this study had moderate to severe symptoms and tended to have very large prostates (approximately 55 cm3), and therefore would be expected to be at fairly high risk for AUR and the need for surgical intervention. It would also be anticipated that they would be better responders to 5ARIs than men with smaller prostates. The most common adverse effects of finasteride noted in controlled clinical trials have been sexual dysfunction, reduced ejaculatory volume, loss of libido, and breast enlargement or tenderness.
Results from 3 large, multi-center, randomized, 2-year phase IIIa studies of dutasteride demonstrated that it not only reduced prostate volume and symptoms (similar to what has been seen with finasteride), but also circulating DHT by more than 90%, and cut in half the risk of AUR and the need for surgical intervention in men with enlarged prostates.21 A pooled analysis of two of the 2-year phase IIIa trials and a 2-year open-label trial extension with dutasteride indicated a sustained and durable benefit among men with symptomatic BPH at 4 years. Symptoms and flow rate improved, the incidence of AUR was low, and the need for BPH-related surgery was also low, suggesting that the “benefit of dutasteride in arresting BPH disease progression” continues for up to 48 months. No increase in adverse events was evident at 48 months versus 24 months, indicating that dutasteride was efficacious and well tolerated as long-term therapy for men with symptomatic BPH.22
The only data we have to support prevention of BPH come from an observational study of a highly selected population: a single report, derived from the very large (N = 24,488) Health Professionals Follow-up Study, suggested physical activity is inversely related to the occurrence of symptoms of BPH and the need for surgical intervention.23 These data indicate simply walking a total of 2 to 3 hours per week was associated with a 25% reduction in overall prostate-related symptoms.
Conclusion
Although BPH is usually treated effectively with conventional pharmacotherapy in the primary care setting, if uncertainty surrounds the patient’s progress, the PCC should consider a referral to a urologist for specialized testing.24 As treatment options for men with BPH and LUTS evolve, the primary care clinician is in a position to offer choices to the patient. The patient can now choose symptom alleviation with an α-blocker, or disease modification, or both. As with most therapies, the choice is dependent on the patient’s physical and financial condition, the adverse event profile of the pharmacotherapy being considered, and the advice of the PCC regarding the potential long-term outlook. The administration of combination therapy is gaining increasing acceptance as an initial treatment for men with bothersome symptoms and with a large prostate gland. Because of their capacity to provide disease modification, primary care clinicians should become more familiar with the use and efficacy of 5-α-reductase inhibitors.
Main Points.
The tools for diagnosis, as well as varying pharmacotherapies to manage benign prostatic hyperplasia (BPH), are readily available to primary care clinicians and typically provide high levels of clinical success and patient satisfaction. The pharmacotherapies for BPH and lower urinary tract symptoms (LUTS) can be symptom modifying, disease modifying, or both, and are generally safe, having rare adverse effects that end with drug discontinuation.
The most widely employed and validated scoring system for diagnosis and monitoring of BPH-LUTS is the American Urological Association (AUA) symptom score. Based on 7 questions (scored 1–5 by severity), the AUA symptom score quantifies the most consistently identified LUTS referable to BPH.
When a patient presents with LUTS, it is important to rule out possible secondary causes such as urinary tract infection, heart failure, and diabetes. Physicians should perform a urinalysis, a prostate examination, and test the patient’s prostate-specific antigen level.
A patient reporting bothersome symptoms from BPH may be offered symptomatic relief with an α-blocker, disease-modifying therapy with a 5α-reductase inhibitor, or both.
References
- 1.Barry MJ, Fowler FJ, Jr, O’Leary MP, et al. The American Urological Association symptom index for benign prostatic hyperplasia. J Urol. 1992;148:1549–1557. doi: 10.1016/s0022-5347(17)36966-5. [DOI] [PubMed] [Google Scholar]
- 2.Zorn B, Bauer JJ, Moul JW. Benign prostatic hyperplasia: new concepts for the millennium. Am J Manag Care. 1999;5:911–922. [Google Scholar]
- 3.Berry SJ, Coffey DS, Walsh C, Ewing LL. The development of human benign prostatic hyperplasia with age. J Urol. 1984;132:474–479. doi: 10.1016/s0022-5347(17)49698-4. [DOI] [PubMed] [Google Scholar]
- 4.Roberts RG. Benign prostatic hyperplasia: assessing severity, helping patients choose among management options. Consultant. 1994;34:1077–1085. [Google Scholar]
- 5.Preboth MA, Richard R, Wright S. Quantum sufficit. Am Fam Physician. 1999;59:17–21. [Google Scholar]
- 6.Roehrborn CG, Boyle P, Bergner D, et al. Serum prostate-specific antigen and prostate volume predict long-term changes in symptoms and flow rate: results of a four-year, randomized trial comparing finasteride versus placebo. Urology. 1999;54:662–669. doi: 10.1016/s0090-4295(99)00232-0. [DOI] [PubMed] [Google Scholar]
- 7.Chow RD. Benign prostatic hyperplasia. Patient evaluation and relief of obstructive symptoms. 2001;56:33–38. [PubMed] [Google Scholar]
- 8.Labreque J, Randolph R, Davis R. Benign prostatic hyperplasia: an overview for pharmacists. Am J Manag Care. 1999;5:661–669. [Google Scholar]
- 9.Kuritzky L. Benign prostatic hyperplasia. Compr Ther. 1998;24:130–136. [PubMed] [Google Scholar]
- 10.Lepor H. The emerging role of alpha antagonists in the therapy of benign prostatic hyperplasia. J Androl. 1991;12:389–394. [PubMed] [Google Scholar]
- 11.Bartsch G, Rittmaster RS, Klocker H. Dihydrotestosterone and the concept of 5α-reductase inhibition in human benign prostatic hyperplasia. Eur Urol. 2000;37:367–380. doi: 10.1159/000020181. [DOI] [PubMed] [Google Scholar]
- 12.Walsh PC, Madden JD, Harrod MJ, et al. Familial incomplete male pseudohermaphroditism, type 2. Decreased dihydrotestosterone formation in pseudovaginal perineoscrotal hypospadias. N Engl J Med. 1974;291:944–949. doi: 10.1056/NEJM197410312911806. [DOI] [PubMed] [Google Scholar]
- 13.Roehrborn CG, Boyle P, Gould AL, Waldstreicher J. Serum prostate-specific antigen as a predictor of prostate volume in men with benign prostatic hyperplasia. Urology. 1999;53:581–589. doi: 10.1016/s0090-4295(98)00655-4. [DOI] [PubMed] [Google Scholar]
- 14.Catalona WJ, Partin AW, Slawin KM, et al. Use of the percentage of free prostate-specific antigen to enhance differentiation of prostate cancer from benign prostatic disease. A prospective multicenter clinical trial. JAMA. 1998;279:1542–1547. doi: 10.1001/jama.279.19.1542. [DOI] [PubMed] [Google Scholar]
- 15.Rosen R, O’Leary M, Altwein J, et al. LUTS and male sexual dysfunction: the Multi-national Survey of the Aging Male (MSAM-7) 500161. Abstract presented at: American Urological Association Annual Meeting; May 26, 2002; Orlando, FL. [Abstract] [Google Scholar]
- 16.Lepor H, Williford WO, Barry MJ, et al. The efficacy of terazosin, finasteride, or both in benign prostatic hyperplasia. N Engl J Med. 1996;335:533–539. doi: 10.1056/NEJM199608223350801. [DOI] [PubMed] [Google Scholar]
- 17.Hicks RJ, Cook JB. Managing patients with benign prostatic hyperplasia. Am Fam Physician. 1995;52:135–142. [PubMed] [Google Scholar]
- 18.McConnell JD, Barry MJ, Bruskewitz RC, et al. Clinical Practice Guideline, Number 8. Rockville, MD: Agency for Health Care Policy and Research, Public Health Service, US Department of Health and Human Services; 1994. Benign prostatic hyperplasia: diagnosis and treatment. [PubMed] [Google Scholar]
- 19.Lepor H. Nonoperative management of benign prostatic hyperplasia. J Urol. 1989;141:1283–1289. doi: 10.1016/s0022-5347(17)41282-1. [DOI] [PubMed] [Google Scholar]
- 20.McConnell JD, Bruskewitz R, Walsh P, et al. The effect of finasteride on the risk of acute urinary retention and the need for surgical treatment among men with benign prostatic hyperplasia. N Engl J Med. 1998;338:557–563. doi: 10.1056/NEJM199802263380901. [DOI] [PubMed] [Google Scholar]
- 21.Roehrborn CG, Boyle P, Nickel JC, et al. Efficacy and safety of dual inhibitor of 5-alpha-reductase types 1 and 2 (dutasteride) in men with benign prostatic hyperplasia. Urology. 2002;60:434–441. doi: 10.1016/s0090-4295(02)01905-2. [DOI] [PubMed] [Google Scholar]
- 22.Roehrborn CG, Marks LS, Fenter T, et al. Efficacy and safety of dutasteride in the fouryear treatment of men with benign prostatic hyperplasia. Urology. 2004;63:709–715. doi: 10.1016/j.urology.2004.01.001. [DOI] [PubMed] [Google Scholar]
- 23.Platz EA, Kawachi I, Rimm EB, et al. Physical activity and benign prostatic hyperplasia. Arch Intern Med. 1998;158:2349–2356. doi: 10.1001/archinte.158.21.2349. [DOI] [PubMed] [Google Scholar]
- 24.Kuritzky L. A primary care physician’s perspective on benign prostate hyperplasia. Rev Urol. 2003;5(suppl 5):S42–S48. [PMC free article] [PubMed] [Google Scholar]