Abstract
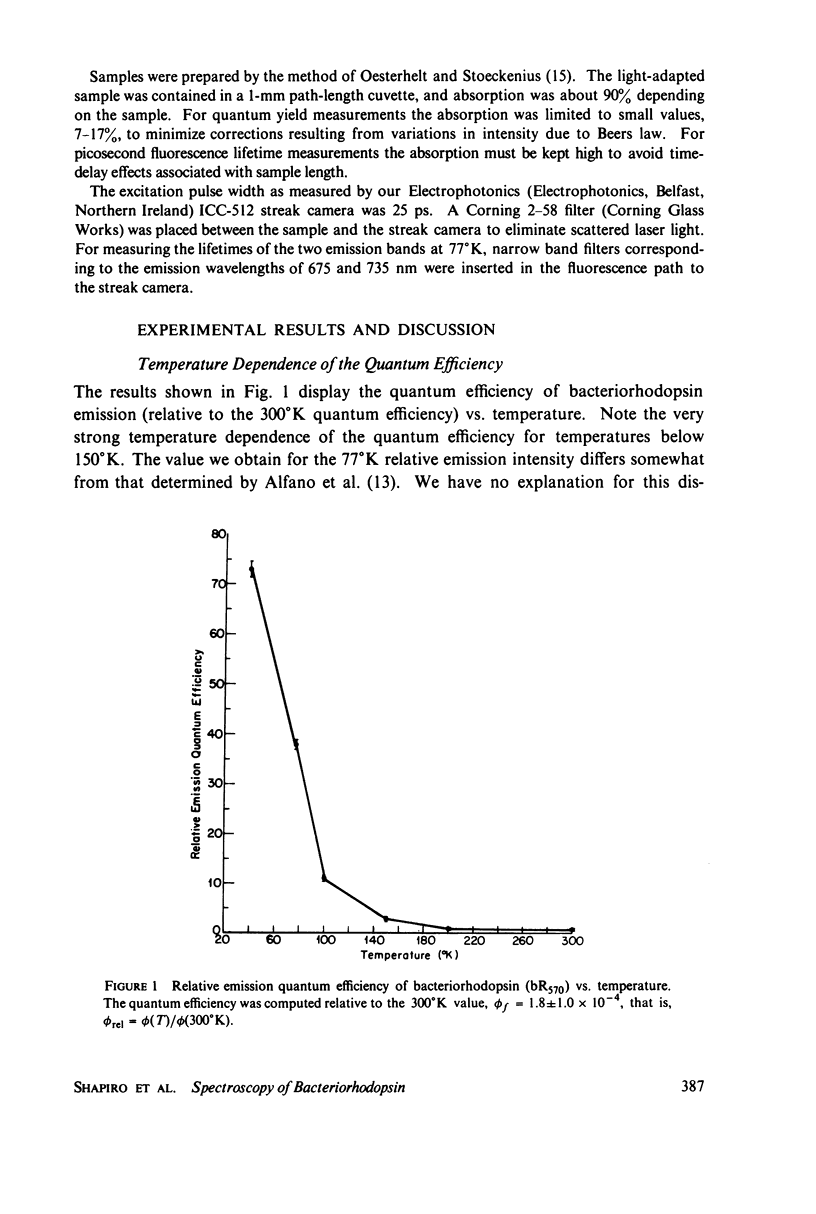
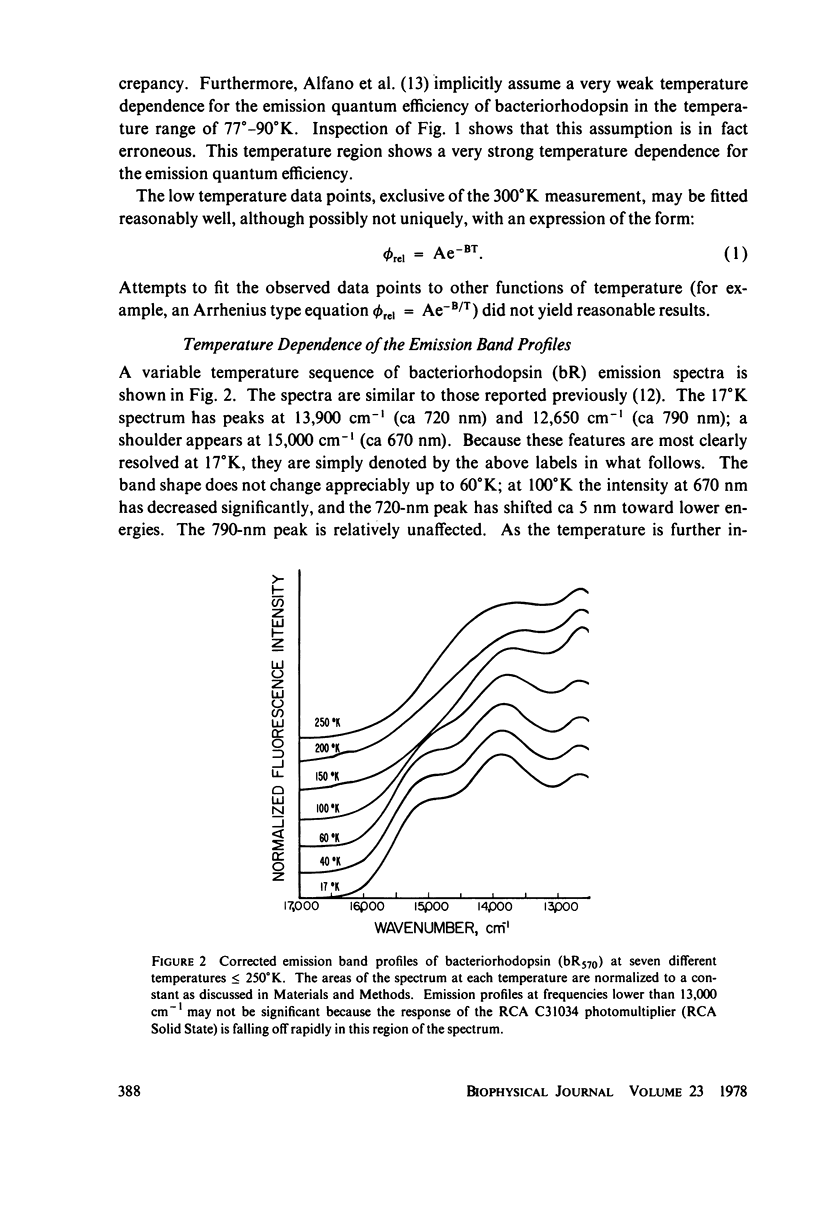
The bacteriorhodopsin emission lifetime at 77 degrees K has been obtained for different regions of the emission spectrum with single-pulse excitation. The data under all conditions yield a lifetime of 60 +/- 15 ps. Intensity effects on this lifetime have been ruled out by studying the relative emission amplitude as a function of the excitation pulse energy. We relate our lifetime to previously reported values at other temperatures by studying the relative emission quantum efficiency as a function of temperature. These variable temperature studies have indicated that an excited state with an emission maximum at 670 nm begins to contribute to the spectrum as the temperature is lowered. Within our experimental error the picosecond data seem to suggest that this new emission may arise from a minimum of the same electronic state responsible for the 77 degrees K emission at 720 nm. A correlation is noted between a 1.0-ps formation time observed in absorption by Ippen et al. (Ippen, E.P., C.V. Shank, A. Lewis, and M.A. Marcus. 1978. Subpicosecond spectroscopy of bacteriorhodopsin. Science [wash. D.C.]. 200:1279-1281 and a time extrapolated from relative quantum efficiency measurements and the 77 degrees K fluorescence lifetime that we report.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alfano R. R., Govindjee R., Becher B., Ebrey T. G. Picosecond kinetics of the fluorescence from the chromophore of the purple membrane protein of Halobacterium halobium. Biophys J. 1976 May;16(5):541–545. doi: 10.1016/S0006-3495(76)85709-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busch G. E., Applebury M. L., Lamola A. A., Rentzepis P. M. Formation and decay of prelumirhodopsin at room temperatures. Proc Natl Acad Sci U S A. 1972 Oct;69(10):2802–2806. doi: 10.1073/pnas.69.10.2802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campillo A. J., Hyer R. C., Monger T. G., Parson W. W., Shapiro S. L. Light collection and harvesting processes in bacterial photosynthesis investigated on a picosecond time scale. Proc Natl Acad Sci U S A. 1977 May;74(5):1997–2001. doi: 10.1073/pnas.74.5.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campillo A. J., Kollman V. H., Shapiro S. L. Intensity Dependence of the Fluorescence Lifetime of in vivo Chlorophyll Excited by a Picosecond Light Pulse. Science. 1976 Jul 16;193(4249):227–229. doi: 10.1126/science.193.4249.227. [DOI] [PubMed] [Google Scholar]
- Campillo A. J., Shapiro S. L., Kollman V. H., Winn K. R., Hyer R. C. Picosecond exciton annihilation in photosynthetic systems. Biophys J. 1976 Jan;16(1):93–97. doi: 10.1016/S0006-3495(76)85666-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geacintov N. E., Breton J., Swenberg C., Campillo A. J., Hyer R. C., Shapiro S. L. Picosecond and microsecond pulse laser studies of exciton quenching and exciton distribution in spinach chloroplasts at low temperatures. Biochim Biophys Acta. 1977 Aug 10;461(2):306–312. doi: 10.1016/0005-2728(77)90180-3. [DOI] [PubMed] [Google Scholar]
- Gillbro T., Kriebel A. N. Emission from secondary intermediates in the photocycle of bacteriorhodopsin at 77 degrees K. FEBS Lett. 1977 Jul 1;79(1):29–32. doi: 10.1016/0014-5793(77)80343-8. [DOI] [PubMed] [Google Scholar]
- Gillbro T., Kriebel A. N., Wild U. P. On the origin of the red emission of light adapted purple membrane of Halobacterium halobium. FEBS Lett. 1977;78(1):57–60. doi: 10.1016/0014-5793(77)80272-x. [DOI] [PubMed] [Google Scholar]
- Hirsch M. D., Marcus M. A., Lewis A., Mahr H., Frigo N. A method for measuring picosecond phenomena in photolabile species: the emission lifetime of bacteriorhodopsin. Biophys J. 1976 Dec;16(12):1399–1409. doi: 10.1016/S0006-3495(76)85783-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ippen E. P., Shank C. V., Lewis A., Marcus M. A. Subpicosecond spectroscopy of bacteriorhodopsin. Science. 1978 Jun 16;200(4347):1279–1281. doi: 10.1126/science.663607. [DOI] [PubMed] [Google Scholar]
- Kaufmann K. J., Rentzepis P. M., Stoeckenius W., Lewis A. Primary photochemical processes in bacteriorhodopsin. Biochem Biophys Res Commun. 1976 Feb 23;68(4):1109–1115. doi: 10.1016/0006-291x(76)90310-7. [DOI] [PubMed] [Google Scholar]
- Lewis A., Spoonhower J. P., Perreault G. J. Observation of light emission from a rhodopsin. Nature. 1976 Apr 22;260(5553):675–678. doi: 10.1038/260675a0. [DOI] [PubMed] [Google Scholar]
- Lewis A., Spoonhower J., Bogomolni R. A., Lozier R. H., Stoeckenius W. Tunable laser resonance raman spectroscopy of bacteriorhodopsin. Proc Natl Acad Sci U S A. 1974 Nov;71(11):4462–4466. doi: 10.1073/pnas.71.11.4462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lozier R. H., Bogomolni R. A., Stoeckenius W. Bacteriorhodopsin: a light-driven proton pump in Halobacterium Halobium. Biophys J. 1975 Sep;15(9):955–962. doi: 10.1016/S0006-3495(75)85875-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcus M. A., Lewis A. Kinetic resonance Raman spectroscopy: dynamics of deprotonation of the Schiff base of bacteriorhodopsin. Science. 1977 Mar 25;195(4284):1328–1330. doi: 10.1126/science.841330. [DOI] [PubMed] [Google Scholar]
- Oesterhelt D., Stoeckenius W. Functions of a new photoreceptor membrane. Proc Natl Acad Sci U S A. 1973 Oct;70(10):2853–2857. doi: 10.1073/pnas.70.10.2853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oesterhelt D., Stoeckenius W. Isolation of the cell membrane of Halobacterium halobium and its fractionation into red and purple membrane. Methods Enzymol. 1974;31:667–678. doi: 10.1016/0076-6879(74)31072-5. [DOI] [PubMed] [Google Scholar]
- Oesterhelt D., Stoeckenius W. Rhodopsin-like protein from the purple membrane of Halobacterium halobium. Nat New Biol. 1971 Sep 29;233(39):149–152. doi: 10.1038/newbio233149a0. [DOI] [PubMed] [Google Scholar]
- Porter G., Synowiec J. A., Tredwell C. J. Intensity effects on the fluorescence of in vivo chlorophyll. Biochim Biophys Acta. 1977 Mar 11;459(3):329–336. doi: 10.1016/0005-2728(77)90034-2. [DOI] [PubMed] [Google Scholar]
- Stoeckenius W., Lozier R. H. Light energy conversion in Halobacterium halobium. J Supramol Struct. 1974;2(5-6):769–774. doi: 10.1002/jss.400020519. [DOI] [PubMed] [Google Scholar]
- Swenberg C. E., Geacintov N. E., Pope M. Bimolecular quenching of excitons and fluorescence in the photosynthetic unit. Biophys J. 1976 Dec;16(12):1447–1452. doi: 10.1016/S0006-3495(76)85786-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zankel K. L., Reed D. W., Clayton R. K. Fluorescence and photochemical quenching in photosynthetic reaction centers. Proc Natl Acad Sci U S A. 1968 Dec;61(4):1243–1249. doi: 10.1073/pnas.61.4.1243. [DOI] [PMC free article] [PubMed] [Google Scholar]




