Abstract
The role of NK cells following solid organ transplantation remains unclear. We examined NK cells in acute allograft rejection using a high responder model (DA → Lewis) of rat orthotopic liver transplantation. Recipient-derived NK cells infiltrated liver allografts early after transplantation. Since chemokines are important in the trafficking of cells to areas of inflammation, we determined the intragraft expression of chemokines known to attract NK cells. CCL3 was significantly increased in allografts at 6 h post-transplant as compared to syngeneic grafts whereas CCL2 and CXCL10 were elevated in both syngeneic and allogeneic grafts. CXCL10 and CX3CL1 were significantly upregulated in allografts by day three post-transplant as compared to syngeneic grafts suggesting a role for these chemokines in the recruitment of effector cells to allografts. Graft-infiltrating NK cells were shown to be a major source of IFNγ and IFNγ levels in the serum were markedly increased, specifically in allograft recipients, by day three post-transplant. Accordingly, in the absence of NK cells the levels of IFNγ were significantly decreased. Furthermore, graft survival was significantly prolonged. These data suggest that IFNγ-producing NK cells are an important link between the innate and adaptive immune responses early after transplantation.
Keywords: Rodent, Transplantation, NK cells, Cytokines, Chemokines
Introduction
NK cells, large granular lymphocytes with the ability to lyse selected target cells without prior sensitization, play an important role in the host immune response against neoplastic cells and virally infected cells (1–3). NK cells have also been shown to inhibit the engraftment of allogeneic bone marrow (4–6) and demonstrate strong cytotoxicity against xenogeneic targets (7, 8). Rejection of solid organ transplants is thought to be mediated by allospecific T lymphocytes that recognize foreign MHC molecules on donor tissue (9, 10). However, we have previously demonstrated that depletion of host CD8+ T cells does not prevent rejection in fully MHC mismatched models of rat liver and small intestinal transplantation (11, 12), indicating that in the absence of CD8+ T cells, an alternate pathway can mediate graft rejection. Recent interest has focused on the role of NK cells in allograft rejection after solid organ transplantation. NK cell infiltration into allografts has been described in multiple experimental transplant models (13–17), and alloreactive NK cells have been shown to be activated in allogeneic cardiac transplantation (15).
Acute liver allograft rejection is characterized by a mixed portal tract infiltrate that containing mononuclear cells. The accumulation of activated lymphocytes into the allograft is essential to the pathogenesis of tissue injury. The mechanism by which activated lymphocytes are recruited to the graft from the circulation is poorly understood but probably involves local chemotactic factors that promote the migration, positioning, and retention of effector cells in the graft (18). Chemokines are low molecular weight proteins with chemotactic properties for leukocytes and are grouped into families based on their cysteine motif. They are expressed and secreted by a wide variety of cell types including lymphocytes (19) and endothelial components of rejecting allografts (18, 20, 21) in response to activation (22). Several studies have shown that CXCL10 (IFNγ-inducible protein-10), and CX3CL1 (fractalkine), are upregulated in rejecting murine cardiac allografts (23, 24). Both CXCL10 and CX3CL1 are induced by IFNγ (25, 26). The CC chemokines, CCL2 (monocyte chemoattractant protein-1) and CCL3 (macrophage inflammatory protein-1α), have also been detected in cardiac allografts (27, 28). NK cells, similar to T cells, are capable of migrating in response to the chemokines CXCL10, CX3CL1, CCL2, and CCL3 (29, 30). Recipient-derived NK cells can amplify the early chemokine expression in allogeneic skin grafts (14). In addition, NK cells migrate to sites of liver infection through an CCL3-dependent pathway and produce high levels of IFNγ in murine cytomegalovirus-infected livers (31, 32).
In the current study, we examine the role of NK cells in acute allograft rejection using a high responder model of rat orthotopic liver transplantation (OLTx). We demonstrate that increased expression of the chemokines CCL2, CCL3 and CXCL10 are detected in liver grafts as early as 6h post-transplant. This is accompanied by the accumulation of recipient-derived NK cells. Importantly, our data also indicate that graft-infiltrating NK cells are a major source of IFNγ and that liver allograft survival is prolonged in the absence of NK cells. These results suggest that recipient-derived NK cells recruited to the allograft early after transplantation produce IFNγ and facilitate the ensuing adaptive immune response that culminates in graft rejection.
Materials and Methods
Animals
Inbred male Dark Agouti (DA) rats (RT1a) and Lewis rats (RT1l), weighing 220–239 g, were purchased from Harlan (Indianapolis, IN). All animals were housed in accordance with institutional animal care and had access to water and standard laboratory chow ad libitum.
Orthotopic Liver Transplantation
Lewis rats were grafted with Lewis livers in the syngeneic group or with DA livers in the allogeneic group. Lewis recipients reject DA livers with a median survival time of 10 days (range 9–12 days) (33). Donor and recipient surgeries were carried out under anesthesia with isoflurane (Abbott Laboratories, North Chicago, IL). OLTx was performed with a modification of Kamada and Calne’s technique (34) without hepatic artery reconstruction. The liver was perfused with 15 ml of lactated Ringer’s solution at 4°C through the catheter placed in the abdominal aorta, and the excised graft was stored in lactated Ringer’s solution at 4°C. Cold ischemic time was ≤90 min. Upon completion of the recipient’s hepatectomy, the graft was transplanted orthotopically. The anhepatic phase was ≤16 min. No immunosuppression was given to the recipient rats in this study. In some experiments, NK cells were depleted in vivo by one intraperitoneal injection (50 μl) of rabbit anti-asialo-GM1 antibodies (Wako Chemicals, Richmond, VA) the day before transplantation. This protocol completely depletes NK cells for seven days after transplantation.
Specimens
The recipient rats were sacrificed at 6, 12, and 24 h and days 2, 3, 5, and 7, post-transplant. Liver tissue were snap frozen for mRNA analysis. When indicated, a portion of the liver graft was utilized for the isolation of liver infiltrating mononuclear cells (LIMC).
Isolation of LIMC
LIMC were prepared as previously described (11). After systemic heparinization followed by exsanguination, the graft was perfused in situ via the portal vein with 15 ml of Ca2+-free PBS to remove residual blood from the liver graft. The graft was subsequently removed and perfused through the portal vein with HBSS (GIBCO BRL, Grand Island, NY) containing 0.5 mg/ml collagenase (Sigma, St. Louis, MO), 0.02 mg/ml DNase I (Sigma), and 10% FCS. The liver tissue was cut into small pieces, resuspended in HBSS/collagenase/DNase I solution, and completely digested on a tilting device in a 37°C incubator for 30 min. The digested liver cell suspension was filtered through a nylon mesh (100 μm pore size) to remove debris, and LIMC were isolated by centrifugation over Ficoll (Ficoll-Paque Plus, Amersham Pharmacia Biotech, Piscataway, NJ). Isolated LIMC were washed and counted.
Flow Cytometric Analyses
Cells were incubated with specific mAbs; FITC-anti-αβTCR (R7.3, Serotec Inc, Raleigh NC), -anti-RT1Aa,b (C3, BD PharMingen, San Diego, CA), and PE-anti-NKR-P1 (10/78, Serotec) for 30 min on ice and then washed twice in FACS buffer (PBS, 1% FCS, 0.1% sodium azide). To distinguish between cells of donor or recipient origin, FITC-anti-RT1Aa,b mAb was used to detect MHC class I expressed on donor cells (RT1a) but not on recipient cells (RT1l). Flow cytometric analysis was performed on a FACScan flow cytometer using CellQuest software (Becton Dickinson, San Jose, CA). The corresponding isotype matched antibodies (Dako, Carpinteria, CA) were used for negative controls and to set appropriate quadrants.
RNase Protection Assay (RPA) and Densitometry Analysis
Total RNA was isolated from liver grafts using TRIzol (GIBCO BRL) (35). RNA integrity was confirmed by detection of the 28S and 18S RNA bands following electrophoresis in a 1.0% agarose gel. The concentration of RNA was measured by spectrophotometry (Beckman DU640B, Beckman Instruments, Inc., Fullerton, CA). RPA experiments (RiboQuant Multi-Probe RNase Protection Assay System, BD Pharmingen) were performed according to the manufacturer’s recommendations. Probes containing rat CCL2, CCL3, CXCL10, CX3CL1, rGAPDH, and r/mL32 (Torrey Pines Biolabs, La Jolla, CA) were used to detect chemokines. Antisense riboprobes were prepared by in vitro transcription with either T7 (CCL2, CXCL10, CX3CL1and r/mL32) (BD PharMingen) or SP6 (CCL3 and rGAPDH) (Invitrogen, Carlsbad, CA) RNA polymerase with the incorporation of [α32P]UTP (NEN Life Science Products, Boston, MA) at 37°C according to the manufacturer’s directions (BD PharMingen) using the PharMingen RiboQuant in vitro transcription kit. Total RNA (10.0 μg) was hybridized with a [α32P]UTP-labeled RNA probe (0.5–1.0 x 106 cpm each probe/sample) followed by RNase digestion. Protected bands were resolved on a 5.0% non-denaturing polyacrylamide gel and exposed to x-ray film. The undigested probe set was run also as a marker for each experiment. The intensity of the protected band was determined by densitometry (Image Analyzer IS 2000, Alpha Innotech, San Leandro, CA), and each value was normalized against its corresponding GAPDH band intensity.
IFNγ ELISA
The levels of IFNγ in rat serum were measured by ELISA using commercially available antibodies (BioSource International, Camarillo, CA). All serum samples were analyzed in triplicate. This assay was determined to have a sensitivity of 10 pg/ml using recombinant rat IFNγ as a standard (BioSource Int.).
Intracellular Cytokine Staining
LIMC were cultured at a density of 106 cells/ml in RPMI 1640 medium supplemented with 10% FCS, 50 μM 2-mercaptoethanol,10 mM Hepes, 100 U/ml penicillin, 100 μg/ml streptomycin (GIBCO BRL), and 1000 U/ml human recombinant IL-2 (Biological Resources Branch, NCI) for 24 h at 37°C. Brefeldin A (10 μg/ml; Sigma) was added during the last 6 h to inhibit cytokine secretion. Cells were washed, and surface staining using FITC-anti-αβTCR or -anti-NKR-P1 (10/78, Serotec) mAb was performed in FACS buffer plus brefeldin A for 30 min. Cells were then washed, fixed for 15 min using 250 μl of Cytofix (BD PharMingen), permeabilized for 15 min in permeabilization buffer (FACS buffer/0.5% saponin), and then incubated with PE-anti-rat IFNγ mAb (DB-1, BD PharMingen) or PE-mouse IgG1 (Dako) as an isotype control for 30 min at 4°C. After one wash with permeabilization buffer, cells were washed in FACS buffer without saponin. Stained cells were acquired by FACScan immediately and analyzed with CellQuest software.
Statistics
Data are expressed as mean values ± SD. Statistical analyses were performed by Student’s t test or Welch’s t test, where appropriate.
Results
Recipient-derived NK Cells Infiltrate Allogeneic Liver Grafts
In previous studies we determined that CD8+ T cell depletion does not prolong the survival of liver allografts in a high-responder OLTx (11). We did, however, note a marked infiltration of NKR-P1+ NK cells in these liver allografts. To expand upon this observation, NK cell infiltration was examined in a model where fully allogeneic donor DA (RT1a) livers were transplanted into Lewis (RT1l) recipients, and LIMC were isolated according to our previously published procedures (11). LIMC were labeled with mAbs against NKR-P1, αβTCR, and RT1Aa,b for analysis by two-color flow cytometry. Twenty-four hours after transplantation, 18.6 ± 3.0% (n=3) of the LIMC isolated from allografts were αβTCR+ T cells, while 56.4 ± 6.2% (n=3) were αβTCR− NKR-P1+ NK cells. A representative experiment is shown in Figure 1A.
Figure 1.
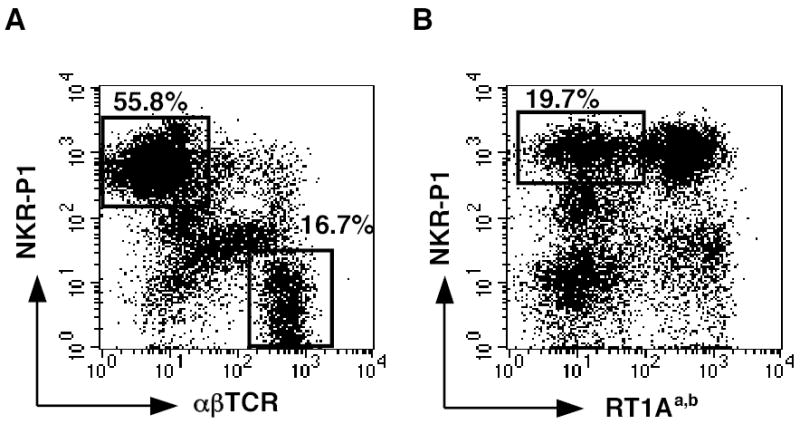
NK cells of recipient origin are detected in liver allografts. LIMC were isolated from an allograft, 24 h post-transplantation and analyzed by flow cytometry using mAbs against NKR-P1, αβTCR, and RT1Aa,b. Numbers indicate percentages of labeled cells in a mononuclear cell gate. A, LIMC were labeled with FITC-anti-α βTCR mAb and PE-anti-NKR-P1 mAb. 55.8% of the LIMC are αβTCR- NKR-P1+ NK cells, and 16.7% are αβTCR+ T cells. B, To determine if the NK cells are of donor or recipient origin, LIMC were labeled with PE-anti-NKR-P1 mAb and FITC-anti-RT1Aa,b mAb which specifically recognizes donor DA rat MHC class I. 19.7% of the LIMC are recipient-derived NK cells (RT1Aa,b− NKR-P1+ cells). Data are representative of three individual grafts.
To determine if the NK cells were of donor or recipient origin, LIMC were labeled with FITC-anti-RT1Aa,b mAb specific for donor MHC class I (RT1a) in combination with PE-anti-NKR-P1 mAb. At 24 h post-transplant, 22.9 ± 6.1% (n=3) of the LIMC were NK cells of recipient origin (RT1Aa,b,−). A representative experiment is shown in Figure 1B. We included only the NKR-P1high-positive subsets to eliminate NKT cell and monocyte populations, since hepatic NKT cells and activated monocytes exist mainly in NKR-P1dim-positive subsets in the rat (36, 37).
Recipient-derived NK Cells Infiltrate Liver Grafts in The First Few Hours Post-Transplant
As previously discussed, αβTCR− NKR-P1+ NK cells are a major component of the infiltrating cells in liver allografts. To evaluate the kinetics of NK cell infiltration after transplantation, LIMC isolated from allografts were analyzed for the proportion of NK cells at 8 time points post-transplant. The proportion of NK in LIMC cells peaked at 12-24 h post-transplant (56.4 ± 6.2%; n=3) and decreased to 35.6 ± 3.0% (n=3) by day 2 as the proportion of T cells increased in the allograft. NK cells of recipient origin (NKR-P1+ RT1Aa,b−), infiltrated the grafts very early post-transplant constituting over half of the NK cells at 6 h post-transplant (Fig. 2A). The proportion of recipient-derived NK cells in allografts peaked at 12 h post-transplant, decreased by day 3, then increased again at day 5 and comprised 29.5 ± 7.0% of the total number of infiltrating cells at day 7 post-transplant (Fig. 2B). Conversely, in syngeneic liver grafts the overall proportion of NK cells and T cells were 32.2± 2.1 % and 31.2 ± 4.0 % respectively, prior to transplant. NK cells peaked at 12 h post-transplant (NK cells, 49.7±3.3% and T cells 19.9±5.4%) and then decreased to pre-transplant levels by day 3 post-transplant (NK cells, 30.6±3.3% and T cells 32.4±3.4%) and remained constant (NK cells, 34.0±4.3% and T cells 30.4±4.4%). In syngeneic grafts it is not possible to separately quantitate the numbers of host and infiltrating NK and T cells thus these numbers reflect the total cell pool. These data suggest that there is a bimodal infiltration of NK cells into liver allografts, early as a result of non-specific surgical stress, including ischemia/reperfusion injury and later at the time that effector cells are infiltrating the allograft and mediating rejection.
Figure 2.
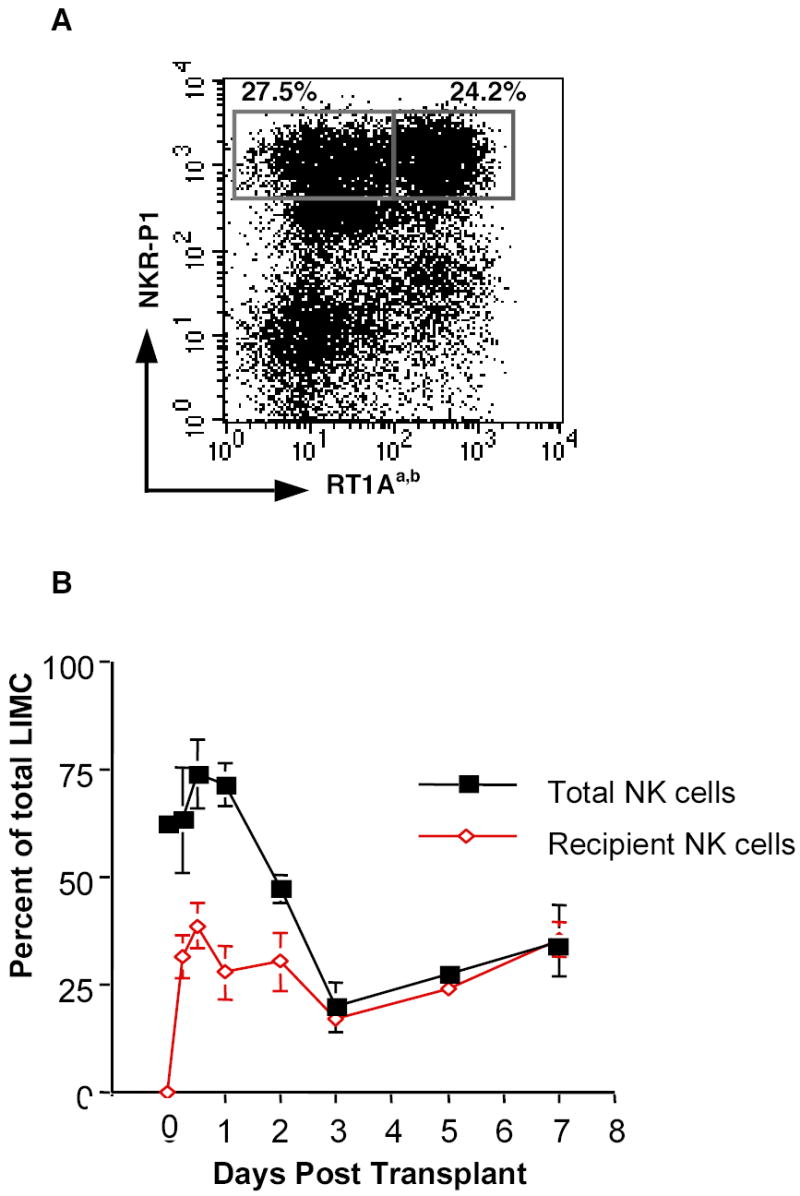
Recipient-derived NK cell infiltrate liver allografts by 6 h post-transplantation. A, Representative flow cytometric analysis of LIMC from an allograft at 6 h post-transplant. Over half of the NK cells are recipient-derived NK cells (RT1Aa,b− NKR-P1+ cells: 27.5% and RT1Aa,b+ NKR-P1+ cells: 24.2%). B, The kinetics of total (NKR-P1+, filled squares) and recipient-derived NK cells (RT1Aa,b- NKR-P1+, open diamonds) in allografts. LIMC isolated from allografts were analyzed for the proportion of recipient-derived NK cells among the total LIMC at serial time points post-transplant. Note that donor-derived NK cells are virtually gone by three days post-transplant. Data are presented as the mean percentage of three to four individual samples ± SD.
Increased Expression of Chemokines is Detected Early after Transplantation
Because NK cells were recruited to liver allografts rapidly following transplantation and because chemokines are important in the trafficking of lymphoid cells to areas of inflammation, we determined the expression of chemokines in liver grafts. Total RNA was isolated from both syngeneic and allogeneic grafts at 0, 6, 12, and 24 h and days 3 and 7 post-transplant and analyzed by RPA (Fig. 3). The chemokines CCL2 and CXCL10 were elevated as early as 6 h post-transplant in both syngeneic and allogeneic grafts, suggesting that these chemokines were increased in liver tissue due to non-specific surgical stress and ischemia/reperfusion injury. Expression of CCL3 was detected by 6 h post-transplant in both syngeneic and allogeneic grafts (Fig. 3A) but was significantly increased (P = 0.015) in the allografts as compared to the syngeneic grafts (Fig. 3B, bottom left panel). CXCL10 and CX3CL1 were significantly upregulated (P = 0.014 and 0.013, respectively) in the allografts at day 3 post-transplant as compared to the syngeneic grafts (Fig 3A and 3B, top right panel and bottom right panel), suggesting a role for these chemokines in promoting the recruitment of effector cells to allogeneic tissue.
Figure 3.
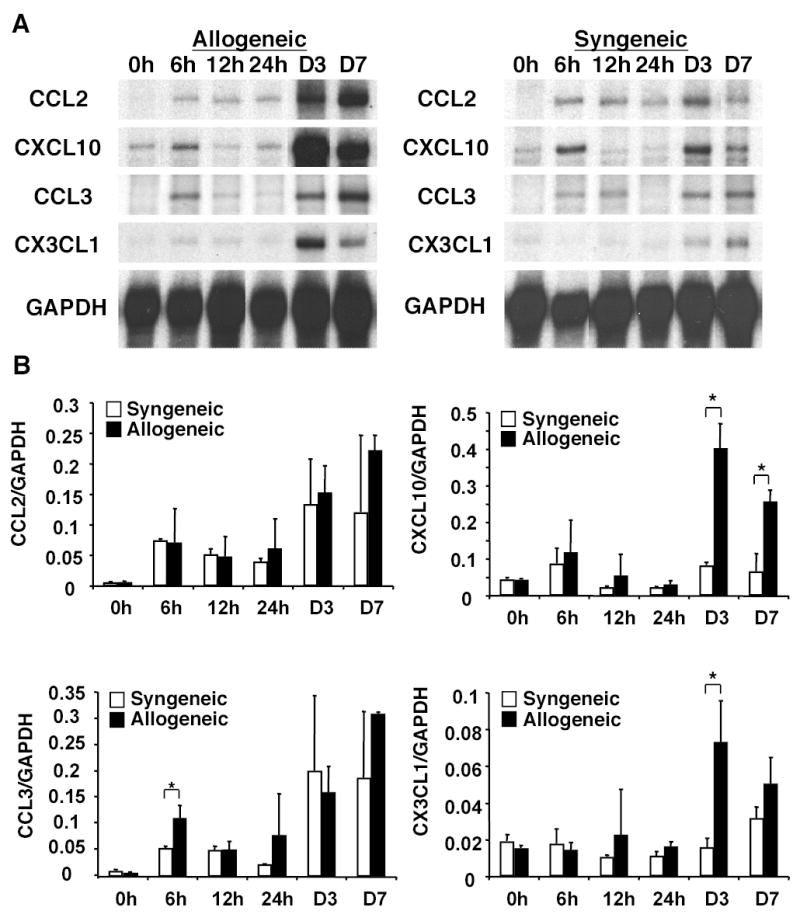
CCL2, CXCL10, CCL3 and CX3CL1 genes are expressed in liver grafts. Total RNA was isolated from syngeneic and allogeneic grafts at 0, 6, 12, and 24 h and days 3 and 7 post-transplant. Total RNA (10 μg) was hybridized with CCL2, CXCL10, CCL3, CX3CL1 and GAPDH riboprobes and analyzed by RPA. The day 0 sample is RNA isolated from nontransplanted Lewis livers for syngeneic grafts or DA livers for allogeneic grafts. A, Representative results from three individual grafts per group per time point are shown. B, The intensity of each chemokine band was measured and normalized to the GAPDH band intensity for each sample. Data are presented as the mean intensity of three individual samples ± SD. * P < 0.02 (syngeneic versus allogeneic grafts).
Depletion of NK Cells Prolongs Graft Survival
To define the role of NK cells after liver transplantation we treated a group of rat liver allograft recipients (n=7) with a single dose (50 μl) of rabbit anti-asialo-GM1 antibodies (AGM1) or control rabbit serum (500 μg), on the day prior to transplantation. Using this protocol NK cells are quickly (by 24 hours) depleted from the periphery, and begin to reappear in the blood seven days later (Fig 4B). Depletion of NK cells was confirmed by flow cytometry in all graft recipients treated with AGM1. We determined, by flow cytometry, that AGM1 does bind to rat NK cells, NKT cells and the majority of T cells, however, NK cells are AGM1bright as compared to T cells (data not shown). Interestingly, AGM1 treatment depletes virtually all of the NK cells and NKT cells, yet spares the majority of T cells (Fig. 4A, B). Allograft recipients depleted of NK cells had significantly prolonged survival (P=0.018) as compared to control allograft recipients (n=7) (Fig 4). These data suggest that NK cells have a role in the early events post-transplantation that contribute to liver allograft rejection.
Figure 4.
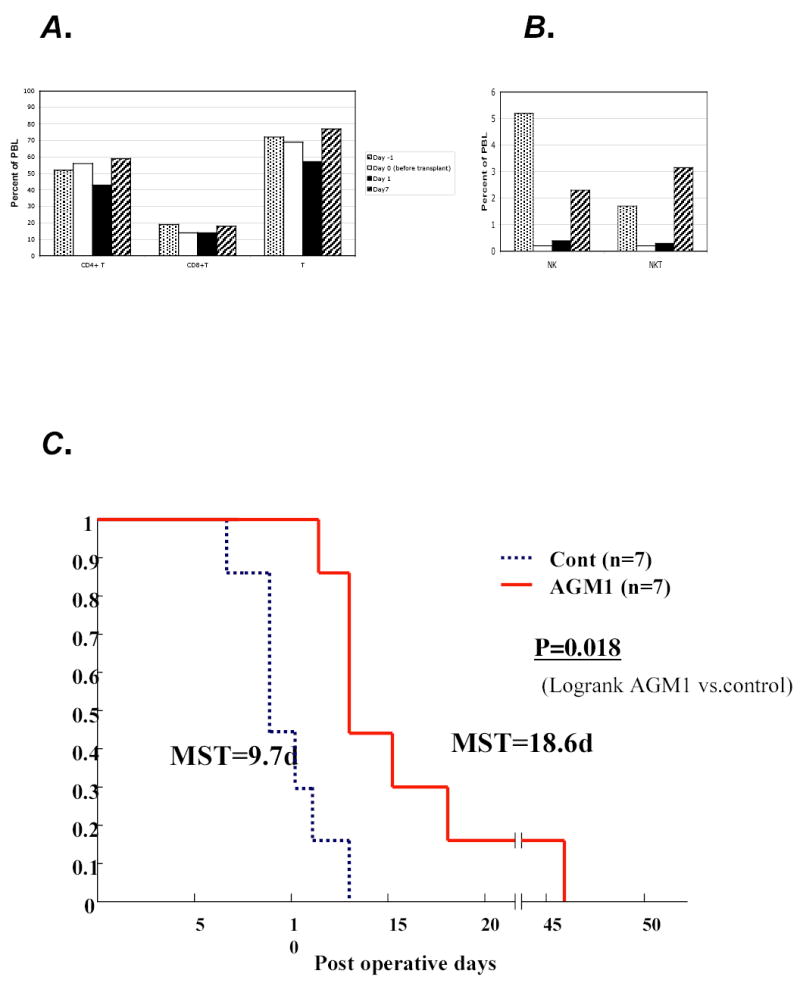
Depletion of NK cells prolongs allograft survival. AB, Lewis recipients of DA livers were treated with a single dose (50μl) of rabbit anti-asialo GM1 antibodies and bled prior to treatment (day –1) and after treatment and transplantation (day 0,1,7) for analysis of A. CD4T, CD8T and T cells B. NK and NKT cells. C. Lewis recipients of DA livers (n=7) have a MST of 9.7 days (dashed line) while depletion of NK cells by AGM1 treatment on the day before transplantation significantly (p=0.018) prolonged allograft survival to 18.6 days (solid lines) in another group of Lewis recipients of DA livers.
NK Cells are a Major Source of IFNγ in Liver Allografts
We and others have demonstrated that there is increased expression of inflammatory cytokines during rejection of liver allografts (33, 38, 39). To specifically analyze the IFNγ levels post-transplantation, serum was obtained from recipients of liver grafts (n=3–5) at 0, 6, 12, 24 hours and 2, 3, 5, and 7 days after transplant. IFNγ was generally below the level of detection in recipients of syngeneic grafts at all time points (Fig 5A). In contrast, the levels of IFNγ increased by day 1 post-transplantation in recipients of allogeneic liver grafts and peaked at 3 days post-transplantation.
Figure 5.

Serum levels of IFN-γ peak 3 days post-transplantation. Serum from recipients of liver grafts were analyzed for IFN-γ levels by ELISA. A, Rats that received syngeneic grafts (n=3) did not produce IFN-γ before or after transplantation. In contrast, recipients of allogeneic grafts (n=3-5) had robust levels of IFN-γ by day 3 post-transplantation. B, The levels of IFN-γ were markedly diminished in rats depleted of NK cells (AGM1-treated allogeneic, n=3) prior to transplantation as compared to rats that received allogeneic liver grafts (n=3). All ELISA measurements were performed in triplicate. Data are presented as the mean ± SD of all rats in the group. * P = 0.001 (AGM1-treated allogeneic versus allogeneic grafts).
To assess the role of NK cells in the production of IFNγ we depleted NK cells, in vivo, by injecting AGM1 into a group (n=3) of allograft recipients. The levels of IFNγ in the serum was significantly reduced (P=0.001) by >70%, three days after transplantation (the peak day for IFNγ production), in NK cell depleted graft recipients as compared to recipients of liver allografts treated with control rabbit serum (Fig 5B).
To further analyze the cellular production of IFNγ in the allografts, LIMC were isolated from allografts, post-transplant and IFNγ production was assessed in permeabilized cells and analyzed by flow cytometry. In a representative experiment, two-days post-transplant, 18% of NK cells and 9% of T cells within the allograft produced IFNγ (Fig. 6, top right and lower right panels, respectively), while 2% of NK cells and 0.4% of T cells in the nontransplanted Lewis rat liver were IFNγ positive (data not shown). Furthermore, the proportion of IFNγ+ NKR-P1+ cells was markedly greater (8.3%, middle top panel) than that of IFNγ+ αβTCR+ cells (3.4%, middle bottom panel) among the total LIMC. Similar results were observed at 12 hours post-transplant (absolute number=0.6 million IFNγ+ NK cells/g liver and 0.24 million IFNγ+ T cells/g liver), and 3 days post-transplant (0.55 million IFNγ+ NK cells/g liver and 0.2 million IFNγ+ T /g liver) as were observed on day two post-transplant (0.6 million IFNγ+ NK cells/g and 0.38 million IFNγ+ T cells/g). Specifically there are a greater number of NK cells expressing IFNγ than T cells. In contrast, on day 7 post-transplant there are more T cells than NK cells producing IFNγ (0.4 million IFNγ+ NK cells/g and 2.45 million IFNγ+ T cells/g). Donor NK cells are the source of 20–25% of the NK cell derived IFNγ on day 2post-transplant (data not shown). These data further indicate that NK cells are a major source of IFNγ in liver allografts early after transplantation but not at the time of rejection.
Figure 6.
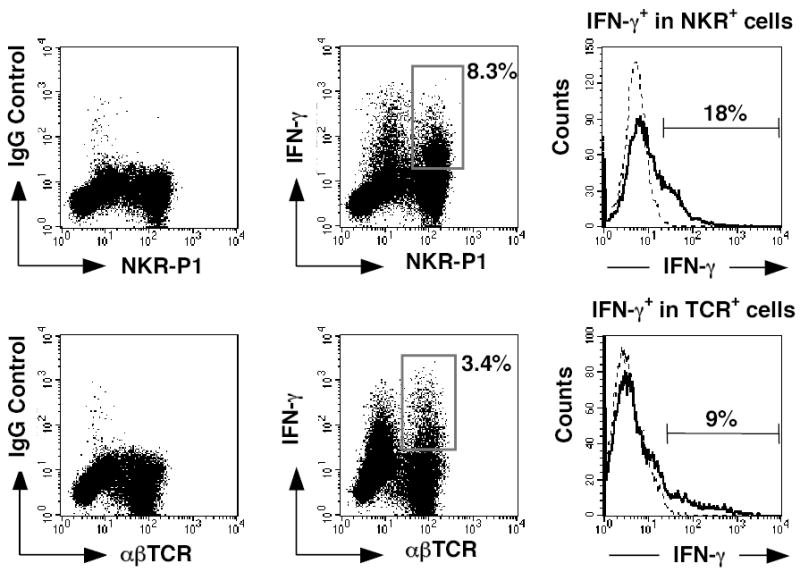
IFN-γ is expressed by infiltrating NK cells. LIMC were isolated from an allograft at day 2 post-transplant and cultured with IL-2 for 24 h. Cells were stained for cell surface antigens using FITC-anti-NKR-P1 or -anti-αβTCR mAb, fixed, permeabilized, and then stained with either PE-anti-IFN-γ mAb or PE-mouse IgG1 as an isotype control. Numbers in dot plots represent percentages of IFN-γ+ NKR-P1+ cells (upper middle panel) and IFN-γ+ αβTCR+ cells (lower middle panel) among the total LIMC. Profiles of IFN-γ staining are expressed as histograms after gating on the NKR-P1+ cell population (upper panel) or the αβTCR+ cell population (lower panel). Cells stained with an anti-IFN-γ mAb are shown with a solid black line; cells stained with an isotype control are shown with a dotted line. Numbers in histograms represent percentages of IFN-γ-producing cells among indicated cell populations. Representative results from three individual grafts are shown.
Discussion
We demonstrate that recipient-derived NK cells are recruited to liver allografts shortly after transplantation and that these NK cells produce IFNγ. NK cell infiltration into solid tissue allografts has been reported in cardiac, skin, and kidney experimental transplant models (13–16). NK cell infiltration is observed shortly after transplantation and often occurs before evidence of T cell infiltration (40). However, the role of early NK cells in solid tissue allografts remains unclear. Previous studies have suggested that NK cells are unable to mediate rejection of allogeneic skin or heart grafts (41, 42). In contrast, it has been reported that alloreactive NK effector cells are induced in rat cardiac allografts (15). Furthermore, inhibition of NK cells in the absence of CD28-B7 signaling prolonged graft survival of murine cardiac allografts (13). Our findings indicate that NK cells within liver allografts are an early source of IFNγ, and importantly that depletion of NK cells and the concomitant decrease in the level of IFNγ, is associated with prolonged graft survival.
The accumulation of activated lymphocytes into the allograft is essential to the pathogenesis of tissue injury. Chemokines are key factors in the trafficking of cells to sites of tissue inflammation, and several studies have documented intragraft chemokine expression during allograft rejection (14, 18–21, 23, 24, 27, 28). CCL2, CCL3, CXCL10 and CX3CL1 have been reported to attract NK cells as well as T cells and to activate NK function (29). In a murine cardiac transplant model, CCL2 gene expression was observed as early as 3 h and peaked at 6 h after transplantation in both syngeneic and allogeneic grafts (28). It is interesting to note that early expression of CCL2 and CXCL10 occurs prior to the peak of NK cell infiltration in the liver grafts. Indeed, a recent study indicates that the prolonged survival of CXCL10-deficient allografts was associated with an absence of NK cell infiltration into the allografts early after transplantation in murine cardiac transplant model (43). These results strongly suggest that non-specific surgical stress, including ischemia/reperfusion injury, elicit the early expression of these chemokines that can recruit recipient-derived NK cells to the liver graft shortly after transplantation.
Interestingly, we demonstrate that CCL3 is specifically upregulated in allografts as early as 6 h post-transplant. Similarly, CCL3 was induced in human liver allografts very early after transplantation (21). Furthermore, CCL3 has been shown to be critical for liver NK cell inflammation and IFNγ production in response to cytomegalovirus infection (31, 32). Thus, the early expression of CCL3 in liver allografts may lead to increased intragraft inflammation by attracting recipient-derived NK cells. The chemokines, CXCL10 and CX3CL1, are significantly upregulated in liver allografts at day 3 post-transplant as compared to the syngeneic grafts and are maintained at high levels through day 7 in agreement with the reported studies in experimental cardiac allograft models (23, 24). Expression of CXCL10 and CX3CL1 specifically in allografts may be important in directing the recruitment of effector cells, including antigen-activated T cells and NK cells, into the liver allografts during acute rejection. Interestingly, CXCR3, a receptor for CXCL10 has been shown to mediate the migration of NK cells to inflamed tissues (44). It is also interesting to note that in a murine skin allograft model the amplified expression of chemokines in allografts at day 3 post-transplant was mediated by recipient-derived NK cells (14).
We propose a model (Fig. 7) in which surgical stress and ischemia/reperfusion injury stimulate the liver allograft to induce the early expression of chemokines including CCL-2, CXCL10 and CCL3. These chemokines direct the recruitment of recipient-derived NK cells and T cells into the allograft early after transplantation. IFNγ produced by NK cells early after transplantation, further induces the production of chemokines and the recruitment of activated lymphocytes to the graft thereby augmenting effector function and graft damage.
Figure 7.

Model: Role of NK cells post-transplantation. Non-specific surgical stress and ischemia/reperfusion (I/R) injury stimulate the liver allograft to produce chemokines such as CCL2, CXCL10 and CCL3 by 6 h post-transplant. These chemokines attract recipient-derived NK cells to the liver allograft, and these graft infiltrating NK cells produce IFN-γ which can augment the later expression of chemokines including CXCL10 and CX3CL1 that promotes further infiltration of NK cells and T cells to the allograft. Finally, these cells produce cytokines including IFN-γ and mediate effector functions.
IFNγ is produced early post-transplantation with peak serum levels on the third day after transplant. Our data suggest that host-derived NK cells and, to a much lesser extent, donor NK cells are a source of much of the IFNγ produced in the early post-transplant period. Importantly, we demonstrate that serum levels of IFNγ are significantly diminished in the absence of NK cells and that graft survival is significantly prolonged. In our study AGM1 was used to deplete NK cells and we have determined in this model that AGM1 also depletes some T cells (mostly CD8+). In the murine model, AGM1 has been shown to deplete effector T cells (45, 46). However, we do not believe depletion of CD8+ T cells accounts for the beneficial effect of AGM1 in our model since we have not depleted the majority of CD8+T cells, we show that these cells do still express IFNγ and importantly, we have previously determined using this exact model, that CD8+ T cell depletion alone does not prolong graft survival (11). Confirmation of our findings using another antibody was not possible since the anti-NK 3.2.3 mAb resulted in only partial depletion of the NK cells in this model and additional reagents to deplete rat NK cells are not available. Taken together our results support the conclusion that NK cells are an important source of IFNγ in liver allografts. Recent studies indicate that NK cells can reach lymph nodes and participate in primary immune responses (47). NK cell entry into stimulated lymph nodes is mediated by CXCR3 rather than CCR7. NK cells recruited to lymph nodes have been shown to produce IFNγ which promotes T helper type 1 cell polarization (47). IFNγ is an important immunoregulatory cytokine after allogeneic transplantation, and studies have demonstrated a role for IFNγ in graft rejection (48–50). Interestingly, however, IFNγ has also been shown to be important in promoting tolerance induction (51). Clearly, the diverse effects of IFNγ on the immune response after allogeneic transplantation remain to be understood.
Our study suggests that NK cell-derived IFNγ is a pivotal link between the innate and adaptive immune responses after liver transplantation. Innate immunity and NK cells, in particular, may be important in the early events leading to allograft rejection.
Acknowledgments
We thank Drs. Ronald R. Nepomuceno, Anming Xiong, and Minoru Tanabe for technical advice.
Footnotes
This work was supported by NIH RO1AI44095 and an award from the Roche Organ Transplantation Research Foundation.
References
- 1.Koo GC, Peppard JR, Mark WH. Natural killer cells generated from bone marrow culture. J Immunol. 1984;132:2300–2304. [PubMed] [Google Scholar]
- 2.Trinchieri G. Biology of natural killer cells. Adv Immunol. 1989;47:187–376. doi: 10.1016/S0065-2776(08)60664-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Biron CA, Byron KS, Sullivan JL. Severe herpesvirus infections in an adolescent without natural killer cells. N Engl J Med. 1989;320:1731–1735. doi: 10.1056/NEJM198906293202605. [DOI] [PubMed] [Google Scholar]
- 4.Rolstad B, Benestad HB. The "natural resistance" to bone marrow allografts in normal and athymic nude rats. Rapid cytotoxic reactions both in vivo and in vitro. Eur J Immunol. 1984;14:793–799. doi: 10.1002/eji.1830140906. [DOI] [PubMed] [Google Scholar]
- 5.Murphy WJ, Kumar V, Bennett M. Acute rejection of murine bone marrow allografts by natural killer cells and T cells. Differences in kinetics and target antigens recognized. J Exp Med. 1987;166:1499–1509. doi: 10.1084/jem.166.5.1499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Murphy WJ, Kumar V, Bennett M. Rejection of bone marrow allografts by mice with severe combined immune deficiency (SCID). Evidence that natural killer cells can mediate the specificity of marrow graft rejection. J Exp Med. 1987;165:1212–1217. doi: 10.1084/jem.165.4.1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gourlay WA, Chambers WH, Monaco AP, Maki T. Importance of natural killer cells in the rejection of hamster skin xenografts. Transplantation. 1998;65:727–734. doi: 10.1097/00007890-199803150-00021. [DOI] [PubMed] [Google Scholar]
- 8.Lin Y, Vandeputte M, Waer M. Natural killer cell- and macrophage-mediated rejection of concordant xenografts in the absence of T and B cell responses. J Immunol. 1997;158:5658–5667. [PubMed] [Google Scholar]
- 9.Krensky AM, Weiss A, Crabtree G, Davis MM, Parham P. T-lymphocyte-antigen interactions in transplant rejection. N Engl J Med. 1990;322:510–517. doi: 10.1056/NEJM199002223220805. [DOI] [PubMed] [Google Scholar]
- 10.Sayegh MH, Turka LA. The role of T-cell costimulatory activation pathways in transplant rejection. N Engl J Med. 1998;338:1813–1821. doi: 10.1056/NEJM199806183382506. [DOI] [PubMed] [Google Scholar]
- 11.Ogura Y, Martinez OM, Villanueva JC, Tait JF, Strauss HW, Higgins JP, Tanaka K, Esquivel CO, Blankenberg FG, Krams SM. Apoptosis and allograft rejection in the absence of CD8+ T cells. Transplantation. 2001;71:1827–1834. doi: 10.1097/00007890-200106270-00020. [DOI] [PubMed] [Google Scholar]
- 12.Krams SM, Hayashi M, Fox CK, Villanueva JC, Whitmer KJ, Burns W, Esquivel CO, Martinez OM. CD8+ cells are not necessary for allograft rejection or the induction of apoptosis in an experimental model of small intestinal transplantation. J Immunol. 1998;160:3673–3680. [PubMed] [Google Scholar]
- 13.Maier S, Tertilt C, Chambron N, Gerauer K, Huser N, Heidecke CD, Pfeffer K. Inhibition of natural killer cells results in acceptance of cardiac allografts in CD28−/− mice. Nat Med. 2001;7:557–562. doi: 10.1038/87880. [DOI] [PubMed] [Google Scholar]
- 14.Kondo T, Morita K, Watarai Y, Auerbach MB, Taub DD, Novick AC, Toma H, Fairchild RL. Early increased chemokine expression and production in murine allogeneic skin grafts is mediated by natural killer cells. Transplantation. 2000;69:969–977. doi: 10.1097/00007890-200003150-00051. [DOI] [PubMed] [Google Scholar]
- 15.Petersson E, Ostraat O, Ekberg H, Hansson J, Simanaitis M, Brodin T, Dohlsten M, Hedlund G. Allogeneic heart transplantation activates alloreactive NK cells. Cell Immunol. 1997;175:25–32. doi: 10.1006/cimm.1996.1031. [DOI] [PubMed] [Google Scholar]
- 16.Fuggle SV. Immunophenotypic analysis of leukocyte infiltration in the renal transplant. Immunol Lett. 1991;29:143–146. doi: 10.1016/0165-2478(91)90216-w. [DOI] [PubMed] [Google Scholar]
- 17.Hsieh CL, Ogura Y, Obara H, Ali UA, Rodriguez GM, Nepomuceno RR, Martinez OM, Krams SM. Identification, cloning, and characterization of a novel rat natural killer receptor, RNKP30: a molecule expressed in liver allografts. Transplantation. 2004;77:121–128. doi: 10.1097/01.TP.0000110423.27977.6F. [DOI] [PubMed] [Google Scholar]
- 18.Goddard S, Williams A, Morland C, Qin S, Gladue R, Hubscher SG, Adams DH. Differential expression of chemokines and chemokine receptors shapes the inflammatory response in rejecting human liver transplants. Transplantation. 2001;72:1957–1967. doi: 10.1097/00007890-200112270-00016. [DOI] [PubMed] [Google Scholar]
- 19.Wang JD, Nonomura N, Takahara S, Li BS, Azuma H, Ichimaru N, Kokado Y, Matsumiya K, Miki T, Suzuki S, Okuyama A. Lymphotactin: a key regulator of lymphocyte trafficking during acute graft rejection. Immunology. 1998;95:56–61. doi: 10.1046/j.1365-2567.1998.00570.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Robertson H, Morley AR, Talbot D, Callanan K, Kirby JA. Renal allograft rejection: beta-chemokine involvement in the development of tubulitis. Transplantation. 2000;69:684–687. doi: 10.1097/00007890-200002270-00039. [DOI] [PubMed] [Google Scholar]
- 21.Adams DH, Hubscher S, Fear J, Johnston J, Shaw S, Afford S. Hepatic expression of macrophage inflammatory protein-1 alpha and macrophage inflammatory protein-1 beta after liver transplantation. Transplantation. 1996;61:817–825. doi: 10.1097/00007890-199603150-00024. [DOI] [PubMed] [Google Scholar]
- 22.Lukacs NW, Hogaboam C, Campbell E, Kunkel SL. Chemokines: function, regulation and alteration of inflammatory responses. Chem Immunol. 1999;72:102–120. doi: 10.1159/000058729. [DOI] [PubMed] [Google Scholar]
- 23.Fairchild RL, VanBuskirk AM, Kondo T, Wakely ME, Orosz CG. Expression of chemokine genes during rejection and long-term acceptance of cardiac allografts. Transplantation. 1997;63:1807–1812. doi: 10.1097/00007890-199706270-00018. [DOI] [PubMed] [Google Scholar]
- 24.Robinson LA, Nataraj C, Thomas DW, Howell DN, Griffiths R, Bautch V, Patel DD, Feng L, Coffman TM. A role for fractalkine and its receptor (CX3CR1) in cardiac allograft rejection. J Immunol. 2000;165:6067–6072. doi: 10.4049/jimmunol.165.11.6067. [DOI] [PubMed] [Google Scholar]
- 25.Fraticelli P, Sironi M, Bianchi G, D'Ambrosio D, Albanesi C, Stoppacciaro A, Chieppa M, Allavena P, Ruco L, Girolomoni G, Sinigaglia F, Vecchi A, Mantovani A. Fractalkine (CX3CL1) as an amplification circuit of polarized Th1 responses. J Clin Invest. 2001;107:1173–1181. doi: 10.1172/JCI11517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rollins BJ. Chemokines. Blood. 1997;90:909–928. [PubMed] [Google Scholar]
- 27.Miura M, Morita K, Kobayashi H, Hamilton TA, Burdick MD, Strieter RM, Fairchild RL. Monokine induced by IFN-gamma is a dominant factor directing T cells into murine cardiac allografts during acute rejection. J Immunol. 2001;167:3494–3504. doi: 10.4049/jimmunol.167.6.3494. [DOI] [PubMed] [Google Scholar]
- 28.Morita K, Miura M, Paolone DR, Engeman TM, Kapoor A, Remick DG, Fairchild RL. Early chemokine cascades in murine cardiac grafts regulate T cell recruitment and progression of acute allograft rejection. J Immunol. 2001;167:2979–2984. doi: 10.4049/jimmunol.167.5.2979. [DOI] [PubMed] [Google Scholar]
- 29.Robertson MJ. Role of chemokines in the biology of natural killer cells. J Leukoc Biol. 2002;71:173–183. [PubMed] [Google Scholar]
- 30.Maghazachi AA, al-Aoukaty A, Schall TJ. C-C chemokines induce the chemotaxis of NK and IL-2-activated NK cells. Role for G proteins. J Immunol. 1994;153:4969–4977. [PubMed] [Google Scholar]
- 31.Salazar-Mather TP, Orange JS, Biron CA. Early murine cytomegalovirus (MCMV) infection induces liver natural killer (NK) cell inflammation and protection through macrophage inflammatory protein 1alpha (MIP-1alpha)-dependent pathways. J Exp Med. 1998;187:1–14. doi: 10.1084/jem.187.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Salazar-Mather TP, Hamilton TA, Biron CA. A chemokine-to-cytokine-to-chemokine cascade critical in antiviral defense. J Clin Invest. 2000;105:985–993. doi: 10.1172/JCI9232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Egawa H, Martinez OM, Quinn MB, Villanueva JC, So S, Esquivel CO, Krams SM. Acute liver allograft rejection in the rat. An analysis of the immune response. Transplantation. 1995;59:97–102. doi: 10.1097/00007890-199501150-00017. [DOI] [PubMed] [Google Scholar]
- 34.Kamada N, Calne RY. Orthotopic liver transplantation in the rat. Technique using cuff for portal vein anastomosis and biliary drainage. Transplantation. 1979;28:47–50. [PubMed] [Google Scholar]
- 35.Hayashi M, Martinez OM, Krams SM, Burns W, Esquivel CO. Characterization of allograft rejection in an experimental model of small intestinal transplantation. J Gastrointest Surg. 1998;2:325–332. doi: 10.1016/s1091-255x(98)80071-1. [DOI] [PubMed] [Google Scholar]
- 36.Steiniger B, Stehling O, Scriba A, Grau V. Monocytes in the rat: phenotype and function during acute allograft rejection. Immunol Rev. 2001;184:38–44. doi: 10.1034/j.1600-065x.2001.1840104.x. [DOI] [PubMed] [Google Scholar]
- 37.Matsuura A, Kinebuchi M, Chen HZ, Katabami S, Shimizu T, Hashimoto Y, Kikuchi K, Sato N. NKT cells in the rat: organ-specific distribution of NK T cells expressing distinct V alpha 14 chains. J Immunol. 2000;164:3140–3148. doi: 10.4049/jimmunol.164.6.3140. [DOI] [PubMed] [Google Scholar]
- 38.Hoffmann MW, Wonigeit K, Steinhoff G, Herzbeck H, Flad HD, Pichlmayr R. Production of cytokines (TNF-alpha, IL-1-beta) and endothelial cell activation in human liver allograft rejection. Transplantation. 1993;55:329–335. doi: 10.1097/00007890-199302000-00019. [DOI] [PubMed] [Google Scholar]
- 39.Shirwan H, Cosenza CA, Wang HK, Wu GD, Makowka L, Cramer DV. Prevention of orthotopic liver allograft rejection in rats with a short-term brequinar sodium therapy. Analysis of intragraft cytokine gene expression. Transplantation. 1994;57:1072–1080. [PubMed] [Google Scholar]
- 40.Baldwin WM, 3rd, Larsen CP, Fairchild RL. Innate immune responses to transplants: a significant variable with cadaver donors. Immunity. 2001;14:369–376. doi: 10.1016/s1074-7613(01)00117-0. [DOI] [PubMed] [Google Scholar]
- 41.Heidecke CD, Araujo JL, Kupiec-Weglinski JW, Abbud-Filho M, Araneda D, Stadler J, Siewert J, Strom TB, Tilney NL. Lack of evidence for an active role for natural killer cells in acute rejection of organ allografts. Transplantation. 1985;40:441–444. [PubMed] [Google Scholar]
- 42.Markus PM, van den Brink M, Cai X, Harnaha J, Palomba L, Hiserodt JC, Cramer DV. Effect of selective depletion of natural killer cells on allograft rejection. Transplant Proc. 1991;23:178–179. [PubMed] [Google Scholar]
- 43.Hancock WW, Gao W, Csizmadia V, Faia KL, Shemmeri N, Luster AD. Donor-derived IP-10 initiates development of acute allograft rejection. J Exp Med. 2001;193:975–980. doi: 10.1084/jem.193.8.975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jiang D, Liang J, Hodge J, Lu B, Zhu Z, Yu S, Fan J, Gao Y, Yin Z, Homer R, Gerard C, Noble PW. Regulation of pulmonary fibrosis by chemokine receptor CXCR3. J Clin Invest. 2004;114:291–299. doi: 10.1172/JCI16861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Slifka MK, Pagarigan RR, Whitton JL. NK markers are expressed on a high percentage of virus-specific CD8+ and CD4+ T cells. J Immunol. 2000;164:2009–2015. doi: 10.4049/jimmunol.164.4.2009. [DOI] [PubMed] [Google Scholar]
- 46.Trambley J, Bingaman AW, Lin A, Elwood ET, Waitze SY, Ha J, Durham MM, Corbascio M, Cowan SR, Pearson TC, Larsen CP. Asialo GM1(+) CD8(+) T cells play a critical role in costimulation blockade-resistant allograft rejection. J Clin Invest. 1999;104:1715–1722. doi: 10.1172/JCI8082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Martin-Fontecha A, Thomsen LL, Brett S, Gerard C, Lipp M, Lanzavecchia A, Sallusto F. Induced recruitment of NK cells to lymph nodes provides IFN-gamma for T(H)1 priming. Nat Immunol. 2004;5:1260–1265. doi: 10.1038/ni1138. [DOI] [PubMed] [Google Scholar]
- 48.Yamaguchi Y, Matsumura F, Liang J, Akizuki E, Matsuda T, Okabe K, Ohshiro H, Ishihara K, Yamada S, Mori K, Ogawa M. Reduced interleukin-12, interleukin-18, and interferon-gamma production with prolonged rat hepatic allograft survival after donor-specific blood transfusion. Dig Dis Sci. 2000;45:2429–2435. doi: 10.1023/a:1005659529472. [DOI] [PubMed] [Google Scholar]
- 49.Stinn JL, Taylor MK, Becker G, Nagano H, Hasegawa S, Furakawa Y, Shimizu K, Libby P, Mitchell RN. Interferon-gamma-secreting T-cell populations in rejecting murine cardiac allografts: assessment by flow cytometry. Am J Pathol. 1998;153:1383–1392. doi: 10.1016/s0002-9440(10)65725-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hricik DE, Rodriguez V, Riley J, Bryan K, Tary-Lehmann M, Greenspan N, Dejelo C, Schulak JA, Heeger PS. Enzyme linked immunosorbent spot (ELISPOT) assay for interferon-gamma independently predicts renal function in kidney transplant recipients. Am J Transplant. 2003;3:878–884. doi: 10.1034/j.1600-6143.2003.00132.x. [DOI] [PubMed] [Google Scholar]
- 51.Konieczny BT, Dai Z, Elwood ET, Saleem S, Linsley PS, Baddoura FK, Larsen CP, Pearson TC, Lakkis FG. IFN-gamma is critical for long-term allograft survival induced by blocking the CD28 and CD40 ligand T cell costimulation pathways. J Immunol. 1998;160:2059–2064. [PubMed] [Google Scholar]


