Abstract
Storage compound accumulation during seed development prepares the next generation of plants for survival. Therefore, processes involved in the regulation and synthesis of storage compound accumulation during seed development bear relevance to germination and seedling establishment. The wrinkled1 (wri1) mutant of Arabidopsis (Arabidopsis thaliana) is impaired in seed oil accumulation. The WRI1 gene encodes an APETALA2/ethylene-responsive element-binding protein transcription factor involved in the control of metabolism, particularly glycolysis, in the developing seeds. Here we investigate the role of this regulatory factor in seed germination and seedling establishment by comparing the wri1-1 mutant, transgenic lines expressing the WRI1 wild-type cDNA in the wri1-1 mutant background, and the wild type. Plants altered in the expression of the WRI1 gene showed different germination responses to the growth factor abscisic acid (ABA), sugars, and fatty acids provided in the medium. Germination of the mutant was more sensitive to ABA, sugars, and osmolites, an effect that was alleviated by increased WRI1 expression in transgenic lines. The expression of ABA-responsive genes AtEM6 and ABA-insensitive 3 (ABI3) was increased in the wri1-1 mutant. Double-mutant analysis between abi3-3 and wri1-1 suggested that WRI1 and ABI3, a transcription factor mediating ABA responses in seeds, act in parallel pathways. Addition of 2-deoxyglucose inhibited seed germination, but did so less in lines overexpressing WRI1. Seedling establishment was decreased in the wri1-1 mutant but could be alleviated by sucrose. Apart from a possible signaling role in germination, sugars in the medium were required as building blocks and energy supply during wri1-1 seedling establishment.
Crucial to the life cycle of the plant is the effective utilization of the seed storage reserves during germination and seedling establishment. Seeds of Arabidopsis (Arabidopsis thaliana) contain a significant portion of the stored carbon and energy as triacylglycerols (TAGs) along with small amounts of Suc within the cotyledons of the mature embryo (Focks and Benning, 1998; Baud et al., 2002). Successful germination and seedling establishment rely upon the presence of sufficient seed storage reserves and the ability to utilize them. A number of Arabidopsis mutants have been isolated that are impaired in several aspects of the underlying processes. These include mutants that are affected in ATP/ADP transport into the plastid (Reiser et al., 2004), carbohydrate metabolism in the developing embryo (Focks and Benning, 1998; Periappuram et al., 2000), oil body formation (Lin et al., 1999, 2004), and TAG synthesis itself (Hobbs et al., 1999; Routaboul et al., 1999; Zou et al., 1999).
Seed germination in Arabidopsis initiates with the matrix-driven absorption of water, followed by cell expansion, splitting of the seed coat, and subsequent emergence of the radicle. Cell division is not a significant component of growth until after the radicle has emerged and root elongation begins (Bewley, 1997; Barroco et al., 2005). The mobilization and metabolism of seed TAG is an intricate but well-characterized process and has been reviewed recently by Penfield and colleagues (2005). Plants with reduced TAG mobilization have been identified, including those affected in lipid trafficking and β-oxidation (Hayashi et al., 1998; Germain et al., 2001; Zolman et al., 2001; Footitt et al., 2002; Lawand et al., 2002). The role of the glyoxylate cycle involved in the conversion of lipids into carbohydrates has also been examined through a series of mutants (Eastmond et al., 2000; Cornah et al., 2004; Hayashi et al., 2005), as has gluconeogenesis itself (Rylott et al., 2003; Penfield et al., 2004). Common phenotypes, many of which can be rescued by sugar supplementation or appropriate photosynthetic conditions, include a reduced germination rate, arrest of development after germination, inability to elongate the hypocotyl in the dark, and failure to elongate the root after opening and greening of the cotyledons. Based on the analysis of these mutants a picture has emerged suggesting that in Arabidopsis the energy for germination is derived from stored reserves other than lipids, and that seed oil becomes vital for continued growth and seedling establishment after the radicle has emerged. Thus seed germination and seedling establishment are metabolically distinct in Arabidopsis.
The signaling processes mediating the mobilization of storage compounds and germination involve growth factors and the sensing of sugars. High amounts of sugar in the medium will inhibit germination (Zhou et al., 1998; Dekkers et al., 2004) and Glc delays storage reserve breakdown (Lu and Hills, 2002; To et al., 2002). Trehalose in particular has been implicated as a sugar signal because ectopic expression of trehalose-6-P synthase causes seedlings to be abscisic acid (ABA) and Glc insensitive (Avonce et al., 2004; Avonce et al., 2005). The mechanism(s) by which Glc and other sugars are sensed and the signal propagated is still unclear. The use of mutants, such as glucose insensitive2 (gin2)/hexokinase 1 (hxk1), and sugar analogs that are metabolized to varying degrees, has shown that sugar signaling occurs through hexokinase-dependent and -independent pathways and likely through membrane receptors (Rolland et al., 2002; Arroyo et al., 2003; Moore et al., 2003; Gibson, 2005). There is also abundant evidence that sugar signaling/sensing is closely intertwined with ABA signaling. It has been shown that plants defective in ABA responses are also resistant to the inhibitory effects of high concentrations of sugar on growth. Several ABA biochemical or response mutants, aba1/gin1, aba2/gin5, and gin6/ABA-insensitive 4 (abi4), have been isolated during screens for ABA resistance and sugar resistance (Arenas-Huertero et al., 2000; Rook et al., 2001). In general, ABA acts in the embryo to regulate embryo maturation and induction of dormancy. Levels of ABA are maximal during seed maturation (Karssen et al., 1983), at the time of storage compound accumulation. This ABA is thought to be of maternal origin. With the ending of embryo maturation and the onset of dormancy ABA of embryonic origin comes into play. Additional evidence of a role for ABA in the regulation of seed storage compound accumulation was derived from in vitro embryo studies (Qi et al., 1998) and from the dynamics of protein and transcript accumulation for storage proteins and oleosin in an ABA response mutant (Nambara et al., 1992; Nambara et al., 1994; Crowe et al., 2000). Furthermore, the role of ABA in germination has been well established with a large collection of mutants (Finkelstein and Gibson, 2002; Finkelstein et al., 2002; Gibson, 2004). An example of a gene involved in the control of seed maturation and germination by ABA is ABI3. Severe alleles of the ABI mutant abi3 do not undergo complete maturation and desiccation. The abi3 mutant has also been shown to be defective in the accumulation of seed storage protein and oleosins (Finkelstein and Somerville, 1990; Giraudat et al., 1992; Nambara et al., 1992; Nambara et al., 1994; Raz et al., 2001; Nambara and Marion-Poll, 2003).
The wrinkled1 (wri1) mutant of Arabidopsis was originally isolated based on its low seed oil content and developing wri1 seeds showed a decreased activity of key glycolytic enzymes such as hexokinase and pyruvate kinase (Focks and Benning, 1998). WRI1 was subsequently shown to encode an APETALA2/ethylene-responsive element-binding protein (Cernac and Benning, 2004). The wri1-1 mutant allele has lost the first splice site leading to a premature stop codon prior to the APETALA2/ethylene-responsive element-binding protein domains. While it is most likely that no functional protein is produced in the mutant, wri1-1 mutant mRNA is still present. The ectopic expression of the WRI1 cDNA caused a sugar-inducible accumulation of seed oil in the transgenic seedlings. The general appearance of the transgenic seedlings suggested a resumption of embryonic development following germination. The overall conclusion was that the WRI1 gene product is involved in controlling seed development, in particular the phase of embryo maturation in which TAG accumulates, with sugar providing one important cue in the process (Cernac and Benning, 2004). Microarray comparison of the wri1-1 and wild-type developing seed transcript levels indicated a global down-regulation of transcripts encoding enzymes involved in carbohydrate metabolism in the mutant (Ruuska et al., 2002) consistent with a role for WRI1 in the regulation of sugar metabolism. Further support for the function of WRI1 was provided when WRI1 was recently identified in an activation-tagging screen targeting genes for which a strong expression resulted in increased transcription of a known sugar-inducible promoter (Masaki et al., 2005). Here we investigate whether WRI1 plays a role in ABA signaling and sugar sensing during germination and seedling establishment. Results suggest that in addition to regulating seed oil accumulation, WRI1 affects the regulation of sugar metabolism in the germinating seed and in the initial stage of establishment, but can be dispensed with once the seedling is established.
RESULTS
Decreased Germination of wri1-1 Seeds
In routine experiments it was noted that the wri1-1 mutant displayed impaired germination and establishment. Quantitative analysis showed that wri1-1 has a reduced germination frequency (defined as radicle emergence) that can be partially restored by offering fatty acids such as palmitic and oleic acid (data not shown) or the acylated detergent Tween-80 that serves as a fatty acid donor (Table I). The level of rescue by Tween-80 was consistent with that observed for an unrelated mutant deficient in storage lipid accumulation (Lin et al., 1999, 2004). Addition of gibberellic acid and/or sugars did not increase the germination frequency of wri1-1 within statistical limitations. We considered the possibility that the wri1-1 radicle could not emerge due to altered physical properties of the seed coat. However, scoring imbibed seeds with a needle to open the seed coat did not improve the rate of germination for wri1-1 (wild type, scored 95%, unscored 98%; wri1-1, scored 30%, unscored 35%; 50 seeds each). Therefore, germination but not dormancy is likely impaired in the mutant.
Table I.
The wri1-1 mutation causes a reduction in germination and establishment
Line 101 and line 106 express the WRI1 cDNA under the control of the 35S-CaMV promoter. Percentages of germinated seeds of all seeds plated, and establishment defined as percentage of plants of all seeds plated growing beyond radicle emergence are shown. The sd of the mean from three trials is given. Suc was added (+) at 50 mm, giberellic acid (GA) at 5 μm.
| Genotype | % Establishment Soil | % Germination − Suc | % Germination + Suc | % Germination Tween-80 | % Germination − Suc, + GA | % Germination + Suc, + GA |
|---|---|---|---|---|---|---|
| Wild type | 95 ± 3 | 98 ± 1 | 98 ± 1 | 99 ± 1 | 99 ± 1 | 98 ± 2 |
| Line 101 | 83 ± 6 | 90 ± 3 | 86 ± 9 | 90 ± 3 | 90 ± 3 | 91 ± 3 |
| Line 106 | 60 ± 5 | 87 ± 3/15 ± 3a | 89 ± 9/57 ± 10 | 79 ± 4/13 ± 3 | 83 ± 1/14 ± 2 | 86 ± 9/67 ± 5 |
| wri1-1 | 7 ± 4 | 53 ± 1 | 46 ± 3 | 67 ± 3 | 54 ± 4 | 38 ± 6 |
Percentage of yellow seedlings of all seeds plated plus or minus sd.
Establishment (defined as the formation of true leaves and root elongation) of plantlets in soil (Table I) or on agar-solidified medium lacking sugar was reduced 14-fold in the wri1-1 mutant. Expression of the wild-type WRI1 cDNA in the wri1-1 background restored germination and establishment to varying degrees depending on the strength of the transgene expression from the 35S-cucumber mosaic virus promoter. Previously described line 106 (Cernac and Benning, 2004) was a stronger expresser than line 101 (also in the wri1-1 background) isolated in the same study. Line 101 showed intermediate seed TAG accumulation between line 106 and the wri1-1 mutant. As observed for wild type, the seeds that did germinate for line 101 and line 106 established plantlets.
Occurrence of Abnormal Seedlings in WRI1 Overexpressors
A previously described phenotype, the formation of seed oil producing small yellow seedlings in the presence of sugars of strong WRI1-cDNA-expressing lines such as line 106 was studied more closely (Cernac and Benning, 2004). When line 106 was germinated in the presence of sugars (50 mm) with or without gibberellic acid (5 μm, a promoter of germination) we observed an increase in the formation of yellow seedlings as the concentration of sugar in the medium increased. The incidence of small yellow seedlings was not affected by the addition of gibberellic acid to the medium, when Suc was absent. Without Suc or with only Tween-80, the frequency of yellow seedlings was approximately 15%; with 50 mm Suc that number increased to 57%, and with Suc and gibberellic acid the number of storage compound accumulating seedlings increased to 67%. Tween-80 in addition to Suc in the plates had no additional effect on the number of yellow seedlings.
Altered ABA Response during Germination of wri1-1 Seeds
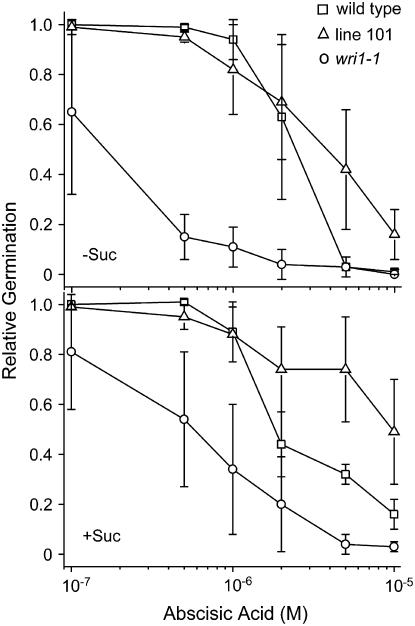
Germination is sensitive to the plant growth factor ABA. We exploited this fact to test a possible connection between ABA signaling in WRI1 function. Indeed, without Suc in the medium wri1-1 germination was approximately 100-fold more sensitive to ABA than that of the wild type (Fig. 1). In the absence of sugar the transgenic line 101 was approximately 2-fold more resistant to ABA compared to wild type and about 10-fold in the presence of sugar. As seen for wild type, the wri1-1 mutant was more resistant to ABA when sugar was present, namely 3- to 5-fold compared to germination in the absence of Suc. The alleviation of ABA inhibition of germination by sugars, including Suc, has been previously documented for wild type (Finkelstein and Lynch, 2000) and is taken as one piece of evidence for a link between sugar sensing and ABA signaling.
Figure 1.
Increased sensitivity of wri1-1 seed germination to ABA. Seed germination of wild type (squares), line 101 expressing the WRI1 cDNA under the control of the 35S-CaMV promoter in the wri1-1 background (triangles), and wri1-1 (circles) was analyzed. Plants were grown in the absence (top section) and in the presence (bottom section) of 50 mm Suc. Germination is reported as fraction relative to the zero control. Germination was scored by radicle emergence. Four independent sets of data were averaged per treatment and sd is indicated.
Increased Sensitivity of wri1-1 Germination to Osmotic Stress
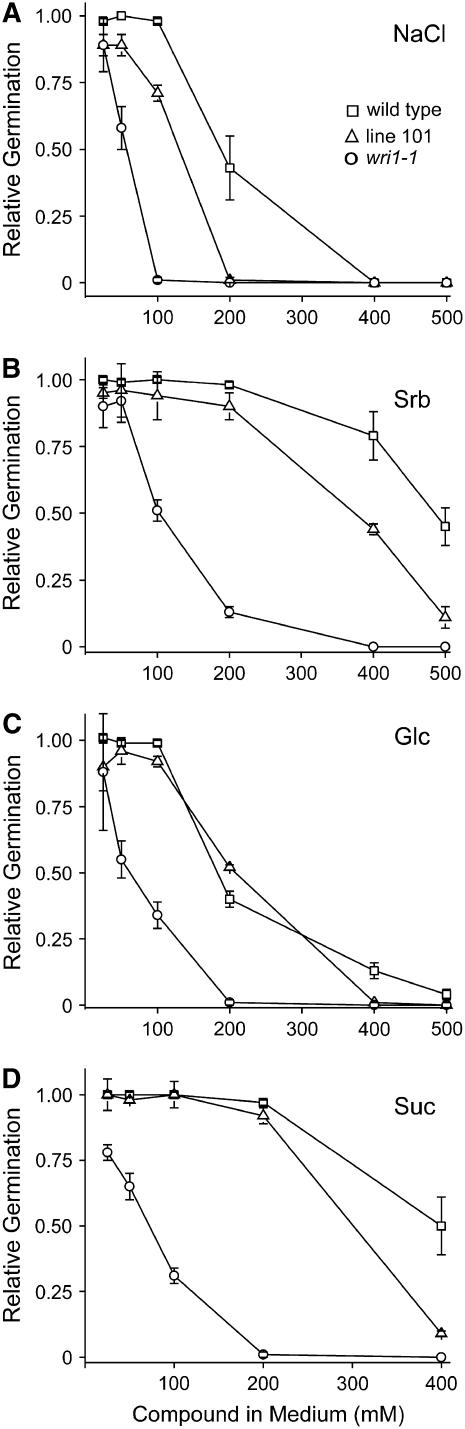
The role of ABA signaling in stress responses is well documented (Finkelstein et al., 2002). To explore this aspect we tested the germination of different lines under osmotic stress induced by osmolites such as sodium chloride and other small molecules. As shown in Figure 2, the wri1-1 mutant was more sensitive to the different compounds. It was noted that the amount of Glc and sodium chloride necessary to achieve 50% inhibition of wild-type germination was roughly 4 times that needed to inhibit wri1-1 germination, whereas the amount of sorbitol and Suc needed was 8 times greater for wild type than wri1-1. Of the various compounds tested, sodium chloride was the most effective at inhibiting wri1-1 germination while Suc and sorbitol were less effective. We observed for line 101 that the germination frequency was intermediate between wri1-1 and wild type, with one anomalous point for Glc. Taken together the data indicate a clear defect in wri1-1 with respect to the response of seed germination to osmotic stress.
Figure 2.
Impairment of wri1-1 germination in the presence of osmolites. Sodium chloride (NaCl; A), sorbitol (Srb; B), Glc (C), and Suc (D) were added to the medium at the concentrations indicated. The fraction of germination in the presence of osmolite is presented relative to germination without osmolite. Error bars represent sd of the mean of three independent assays. Plant lines and symbols are as described in Figure legend 1 and shown here in A.
The Expression of ABA-Responsive Genes Is Enhanced in wri1-1
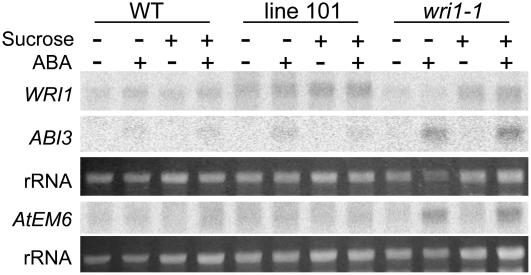
To test the role of WRI1 in the expression of known ABA-responsive genes, transcript levels were examined for the various lines using different concentrations of ABA and Suc in the medium. Of the 12 transcripts examined, which included cold responsive (COR), late embryogenesis, ABI genes, and genes encoding glycolytic enzymes as listed under “Materials and Methods,” two showed a difference between wild type and wri1-1. To directly draw conclusions with regard to germination, we would have preferred to use germinating seedlings for this experiment. However, for technical reasons (differential inhibition of the lines in response to ABA), the assay shown in Figure 3 was conducted by RNA-blot analysis of total RNA from seedlings that had been grown for 6 d on medium containing or lacking Suc, and then were transferred to medium containing or lacking Suc and/or ABA. The tissue from the response experiment was harvested after 3 d of induction by ABA. We found that ABA treatment resulted in a higher induction of ABI3 and AtEM6 in wri1-1 than in wild type or the transgenic lines. The response of the transgenic lines was similar to wild type.
Figure 3.
Expression of ABA-responsive genes in wri1-1. Transcript analysis was conducted following transfer of seedlings to medium containing 50 μm ABA, 50 mm Suc, or lacking these compounds as indicated by (−) or (+), respectively. Seedlings were grown on medium with or without Suc for 6 d and then transferred to the same medium containing or lacking ABA. RNA was extracted after three additional days. The RNA was blotted and the blots probed with the indicated gene. The rRNA bands from the ethidium bromide-stained gel are shown as a loading control.
Interaction between ABI3 and WRI1
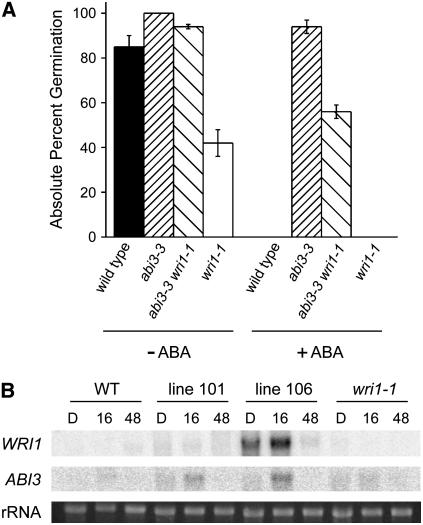
Based on our analysis and previously described effects of the abi3 mutation on storage compound accumulation as summarized in the introduction (Nambara et al., 1992), we hypothesized that WRI1 and ABI3 might act in the same signal transduction pathway. Towards testing this hypothesis, we generated a double mutant between abi3-3 (Nambara et al., 1992) and wri1-1 and examined the ABA response of germination for the double- and single-mutant lines using seeds dissected prior to desiccation (Fig. 4A). It should be noted that abi3-3 and wri1-1 are in the same genetic background. At 10 μm ABA wild type and wri1-1 were completely inhibited in germination (assayed after 7 d of incubation), while the single mutant abi3-3 and the double mutant abi3-3 wri1-1 were resistant to inhibition of germination by ABA but to different degrees (50% germination for the double mutant and 95% for abi3-3). We also noted that similar to severe alleles of abi3, the seeds of the double mutant were desiccation intolerant and like the immature, predesiccation seeds of abi3-3, the double mutant had close to complete germination. As observed for mature seeds, the wri1-1 mutant had a frequency of germination of approximately 50%. Thus the inability of wri1-1 to fully germinate can be alleviated by a reduction in activity of at least one ABA response protein, ABI3. The double mutant did not establish (judged by elongation of the root and formation of true leaves) as well as wild type or abi3-3, but it still did better than wri1-1 (20% ± 5% for the double mutant, 5% ± 2% for wri1-1). Oil content of dry seeds appeared to be lower for the double mutant than the wri1-1 mutant, but this conclusion is tentative because of the high sd for the wri1-1 sample (wild type, 5.9 ± 0.45 μg per seed; abi3-3, 3.7 ± 0.29 μg per seed; double mutant, 1.3 ± 0.1 μg per seed; wri1-1, 1.6 ± 0.67 μg per seed; n = 7 samples of 10 seeds each). Taken together, the results suggest an additive effect of the two mutations and imply that both transcription factors act in parallel pathways.
Figure 4.
Interaction of ABI3 and WRI1. A, Germination assays on early desiccation seeds of wild type (black bars), abi3-3 (densely hatched bars), abi3-3 wri1-1 (sparsely hatched bars), and wri1-1 (white bars) are shown. ABA was present (+) at 10 μm or absent (−). Absolute percent germination is reported as the average of the mean of three independent experiments with the error bars representing sd. B, mRNA abundance of WRI1 and ABI3 in dry seeds (D), 16 h (16), and 48 h (48) after imbibition. The RNA was blotted and the blots probed with the indicated gene. The rRNA bands from the ethidium bromide-stained gel are shown as a loading control.
When the abundance of the WRI1 and ABI3 mRNA was compared in dry seeds and during germination (Fig. 4B) ABI3 mRNA levels generally followed those of WRI1. The mRNA levels of ABI3 were particularly high in the WRI1 overexpressor line 106 and moderately increased in line 101 (Fig. 4B), negating the assumption that decreased ABA sensitivity of germination of the overexpressor lines (Fig. 1) is mediated by ABI3, as the opposite would be expected based on ABI3 abundance. More ABI3 should make germination more sensitive to ABA, not less. It should be pointed out though that the high mRNA levels do not necessarily imply high ABI3 protein levels.
Sugar Sensing Is Affected in the wri1-1 Mutant
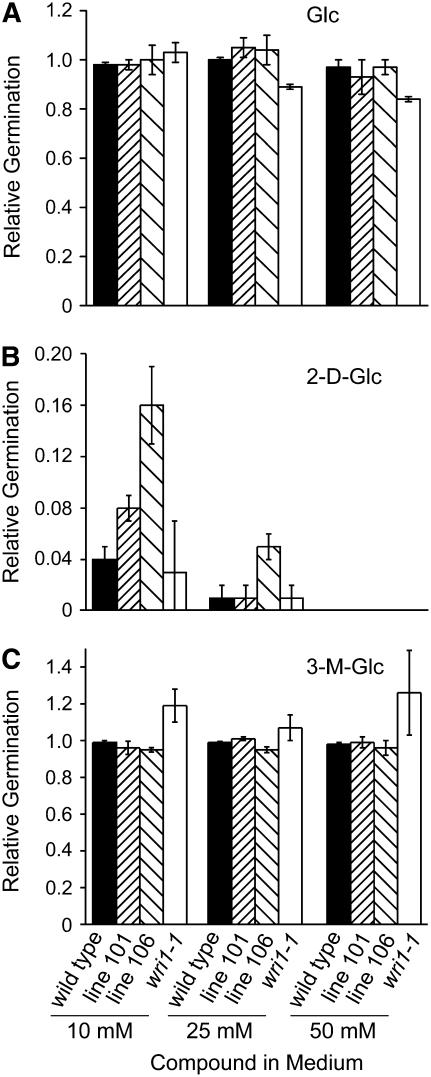
In the different germination assays described above we observed effects of sugars provided in the medium and the question arose whether sensing of sugars is affected in the wri1-1 mutant. To test this possibility and to distinguish sugar sensing effects from sugar metabolism effects, we examined the germination of the different lines in the presence of sugar analogs (Fig. 5). The Glc analogs 2-deoxyGlc and 3-O-methylGlc are not metabolized to any great extent by the glycolytic pathway (Rolland et al., 2002; Cortes et al., 2003). While 3-O-methylGlc is a poor substrate for phosphorylation by hexokinase, 2-deoxyGlc is phosphorylated but not further metabolized. Traditionally, 2-deoxyGlc and 3-O-methylGlc have been used to examine signaling effects versus metabolic effects of Glc, though it is known that the phosphorylation of these analogs could potentially interfere with metabolism by the sequestering of phosphorus. Relative to the zero control, wri1-1 and wild-type germination were both severely inhibited by 25 mm 2-deoxyGlc, while line 106 germination was more resistant (Fig. 5B). At 10 mm of 2-deoxyGlc the transgenic lines were clearly more resistant, with line 106 more so than line 101. Interestingly, germination on 3-O-methylGlc did not inhibit germination at the concentrations tested and for the wri1-1 mutant germination was slightly improved relative to the zero-addition control (Fig. 5C). This differential response to the different compounds might be related to the phosphorylation status of the three sugars with 2-deoxyGlc being the more phosphorylated and also active analog in this assay. Presumably the accumulation of the phosphorylated intermediate causes inhibition. The differences in response of the transgenic lines might be due to their altered sensitivity toward the phosphorylated hexose or their different rates of metabolization of the phosphorylated intermediate.
Figure 5.
Seed germination in the presence of sugar analogs. Germination is reported as fraction relative to the zero control. Glc (Glc; A), 2-deoxyglcuose (2-d-Glc; B), and 3-O-methylGlc (3-M-Glc; C) were added to the medium at concentrations as indicated. Black bars represent the wild type, densely hatched bars line 101, sparsely hatched bars line 106, which is similar to line 101 but a stronger WRI1 cDNA expresser, and white bars represent wri1-1. All assays were performed in triplicate with sd of the mean represented by the error bars.
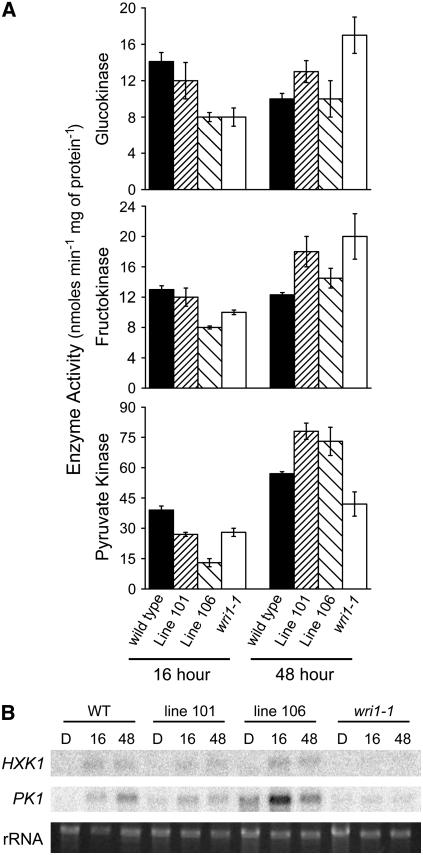
Glycolytic Activity Is Impaired in wri1-1 during Germination
To examine glycolytic activity during germination and at the transition to the seedling establishment phase, hexokinase and pyruvate kinase were assayed 16 h following the beginning of seed incubation and at 48 h, when the wild-type seedlings open the cotyledons and start greening. These are two key regulated enzymes of glycolysis in plants (Plaxton, 1996). Hexokinase can metabolize Fru or Glc and the two activities were assayed separately as glucokinase and fructokinase. To correlate the enzyme activity with the expression of the respective genes responsive to WRI1, mRNA abundance for HXK1 and for plastidic pyruvate kinase (PK1) were determined. Only one of the potential 14 pyruvate kinase genes was selected for this assay, which is expressed in the seed and is regulated by WRI1 (Cernac and Benning, 2004). The results are shown in Figure 6. At the 16 h time point, glycolytic activity was generally reduced in the wri1-1 mutant compared to wild type. Generally, the transgenic lines showed intermediate activity with the exception of the stronger expresser line 106 that had a decreased pyruvate kinase activity compared to wri1-1 at the 16 h time point. Possibly at this stage the plant is highly sensitive to the level of WRI1 protein such that higher levels would be inhibitory. At 48 h, when the seedlings transition into the seedling establishment phase, the activity trends were reversed, with the hexokinase being more active in the mutant compared to the wild type. This effect might have been due to a delay in the transient activity increase in the mutant following germination. The two transgenic lines had intermediate activity. Pyruvate kinase exhibited decreased activity in the mutant at both time points and increased activity over that observed for wild type in the two transgenic lines at 48 h. The mRNA levels were generally consistent with the activity levels but some deviations, as in case of the 16 h time point for the WRI1-overexpressing line, were obvious. To explain this observation one needs to take into consideration that the enzyme activity assays were done in crude extracts and reflect the combined activity of all isoforms present, while the mRNA analysis was specific to just one respective isoform. Taken together, these data are consistent with a shift in metabolism during the transition from germination to seedling establishment. Furthermore, the analysis of the transgenic lines and the wri1-1 mutant suggests that an orderly transition in metabolism is under the influence of WRI1.
Figure 6.
Glycolytic enzyme activities and mRNA abundance during germination and early seedling establishment. A, Enzyme activities for hexokinase represented as glucokinase or fructokinase activities and pyruvate kinase. Plants were grown on agar with half-strength Murashige and Skoog medium but lacking Suc. Tissues were harvested at times after incubation as indicated. The black bars represent wild type, densely hatched bars line 101, sparsely hatched bars line 106, and white bars the wri1-1 mutant. Error bars depict sd of the mean of enzyme activity obtained for three different protein preparations. B, mRNA abundance of HXK1 and PK1. The blot is identical to the blot shown in Figure 4B, but was stripped and reprobed with the gene-specific probes as indicated. The rRNA bands from the ethidium bromide-stained gel are shown as a loading control.
Endosperm Oil Storage and Hypocotyl Elongation Are Decreased in wri1-1
It was previously shown (Penfield et al., 2004) that TAG in the endosperm supplies a significant amount of energy for hypocotyl elongation in the dark and that gluconeogenesis is vital to this process. To discern the ability of the wri1-1 mutant to utilize either sugars stored in the mutant seed (in higher amounts in wri1-1 than in wild-type seeds [Focks and Benning, 1998]) or residual TAG and sugars derived from TAG by gluconeogenesis provided by the embryo or the endosperm, we examined the ability of the wri1-1 seedlings to elongate their hypocotyls in the dark in the absence and presence of sugar (Table II). Hypocotyl elongation of the wri1-1 mutant was reduced approximately 2.7-fold. Epidermal cell length was only 1.2-fold reduced for wri1-1 (wild type, 446 ± 58 μm; wri1-1, 367 ± 79 μm; 50 cells from approximately 10 different hypocotyls were measured each). Apparently, the reduction of hypocotyl length was caused by reduction in cell number as well as cell size. The defect in wri1-1 hypocotyl elongation was only slightly alleviated (within statistical limitations) on medium containing Suc but not on Tween-80 or fatty acid containing medium (data not shown). Apparently, while wri1-1 seedlings could utilize seed storage reserves for moderate hypocotyl elongation in the dark, they were impaired in their ability to utilize Suc in the medium. In this respect wri1-1 is clearly distinct from metabolic mutants more directly affected in TAG metabolism (Penfield et al., 2004). We also examined the transgenic lines in this hypocotyl elongation assay (Table II). The large sd for line 106 grown on media supplemented with 50 mm Suc was brought about, at least in part, by the appearance of the previously documented yellow seedling phenotype (Cernac and Benning, 2004). While some of the line 106 seedlings were capable of elongating the hypocotyl, others did not and resembled embryos.
Table II.
Endosperm lipid accumulation and hypocotyl elongation
Line 101 and line 106 express the WRI1 cDNA under the control of the 35S-CaMV promoter. F1 seed of reciprocal crosses between wild type and the homozygous wri1-1 mutant were analyzed as well. Fatty acid methyl esters (FAME) of total seeds and endosperm were analyzed. Three samples containing 15 seeds or endosperms each were analyzed and the sd of these three samples is shown. Hypocotyl length was measured on 50 mm Suc (+Suc) or in the absence of Suc (−Suc). The sd of the mean from three replicates is given. Hypocotyl measurements were averaged for 30 to 40 plants per treatment. n.d., Not determined.
| Genotype | Seed FAME | Endosperm FAME | Hypocotyl Length (mm) − Suc | Hypocotyl Length (mm) + Suc |
|---|---|---|---|---|
| μg/seed | μg/seed | |||
| Wild Type | 5.1 ± 0.8 | 0.72 ± 0.3 | 18.2 ± 2.1 | 19.4 ± 2.2 |
| Line 101 | 4.4 ± 0.2 | n.d. | 14.0 ± 0.3 | 17.6 ± 2.8 |
| Line 106 | 6.4 ± 1.1 | 1.1 ± 0.5 | 16.6 ± 2.1 | 7.1 ± 6.2 |
| wri1-1 | 1.4 ± 0.4 | 0.2 ± 0.1 | 6.7 ± 0.7 | 10.6 ± 4.8 |
| Wild type × wri1-1 | n.d. | 0.7 ± 0.1 | n.d. | n.d. |
| wri1-1 × wild type | n.d. | 0.7 ± 0.2 | n.d. | n.d. |
With the discovery by Penfield and colleagues (Penfield et al., 2004) that Arabidopsis endosperm stores approximately 10% of the seed oil and can, with some effort, be prepared for analysis, we wondered whether the residual TAG in the wri1-1 seed was solely based in the endosperm or was uniformly reduced across embryo and endosperm. These questions were answered through the use of test crosses, dissecting the endosperm, and analysis of total fatty acid methyl esters for wild type, wri1-1, transgenic line 106, and the F1 seed from reciprocal crosses between wri1-1 and the wild type. Our data indicated that the endosperm TAG is also reduced in a wri1-1 background and that the residual TAG in wri1-1 is present in both endosperm and embryo. As shown in Table II, there was a reduction in endosperm lipid in the wri1-1 background and the ratio of lipids in the endosperm over those of the whole seed remained approximately constant for all genotypes. The test crosses also showed that the endosperm defect in wri1-1 was gene dosage independent with regard to the triploid endosperm, suggesting that endosperm TAG storage is most likely governed by the homozygous wri1-1 embryo mutant genotype and dependent on substrates derived from the embryo. Taken together, our data from dark-grown seedlings and endosperm measurements provide further evidence that the wri1-1 carbon metabolism defect goes beyond the accumulation of TAG in the developing cotyledons and extends to the utilization of sugars in the developing seedling and deposition of storage reserves in the endosperm.
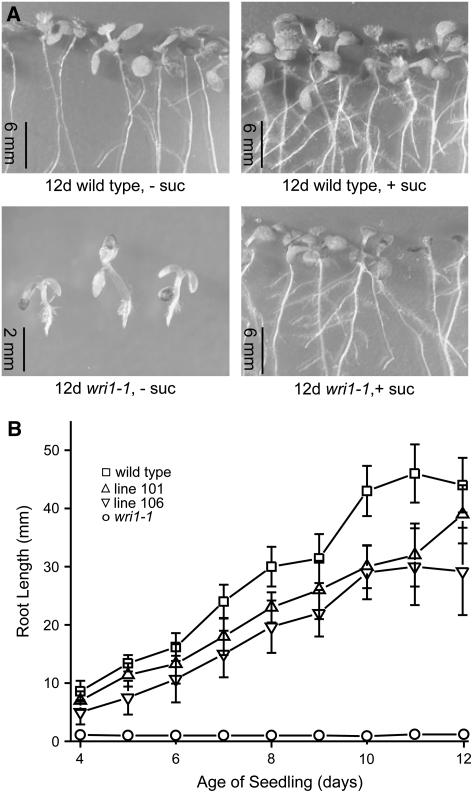
Seedling Establishment Is Impaired in the wri1-1 Mutant
To investigate the seedling establishment defect beyond that observed in soil (Table I), we examined root elongation in the absence and presence of Suc (50 mm). The poor establishment of wri1-1 in medium lacking sugar is qualitatively shown in Figure 7A and was quantified by measurements of root length from day 4 through day 12 post incubation (Fig. 7B). Without any sugar supplement, the wri1-1 seedlings failed to establish and even after 12 d they did not significantly elongate their root or form true leaves. This phenotype was similar to that observed for abi8, ntt2, and bou mutants. The abi8 mutant is defective in ABA signaling and allelic to eld1 (Cheng et al., 2000; Brocard-Gifford et al., 2004), bou is deficient in a mitochondrial acyl carnitine transporter (Lawand et al., 2002), and ntt2 is deficient in a plastidic ATP/ADP transporter (Reiser et al., 2004). While Suc did not enhance the germination frequency of wri1-1 (Table I) it did allow the seedlings to establish (Fig. 7A). As previously observed (Focks and Benning, 1998), wri1-1 plants grew similar to wild type on soil after they were raised on medium containing sugar. Expression of the WRI1 cDNA in the transgenic lines was able to rescue the wri1-1 root elongation defect almost to the level of wild type (Fig. 7B).
Figure 7.
Impaired seedling establishment in the wri1-1 mutant. A, Appearance of 12-d-old seedlings of wild type and wri1-1 grown in the presence (+) or absence (−) of 50 mm Suc. B, Root growth on medium lacking Suc. Symbols are as shown in the figure and as described in the Figure 1 legend with the addition of an inverted triangle for line 106. The error bars represent sd of the mean of at least 15 root length measurements.
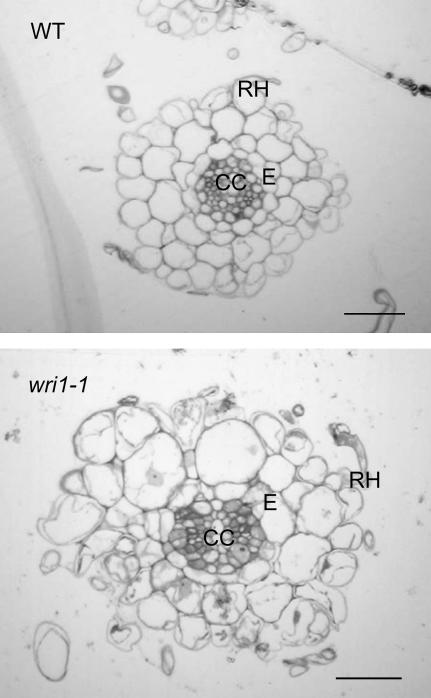
We examined the roots of wri1-1 plants grown on Suc and compared them to wild type using light microscopy (Fig. 8) and noticed that wri1-1 root cross sections were less organized, and that some of the cortex cells were enlarged. While Suc addition was capable of restoring root elongation, the histology of the wri1-1 root cross sections did not return to wild type in the presence of sugar. This altered histology might have been caused by the overall changes in carbohydrate metabolism in the wri1-1 mutant with possible effects on cell wall biosynthesis or changes in the cellular osmolite concentration affecting the water potential and cell expansion.
Figure 8.
Light micrographs of cross sections of wild-type and wri1-1 roots. The plants were grown in the presence of 50 mm Suc. The sections were derived from the differentiation zone of the roots. The size bar represents 200 μm. CC, Central cylinder; E, endodermis; RH, root hair.
To better understand the physiological effects described above, we examined the activity of hexokinase and pyruvate kinase and the expression of the respective genes in roots of 11-d-old developing seedlings of the different lines under investigation. Pyruvate kinase activity in roots grown on Suc was reduced. On the contrary, with Suc present the gluco- and fructokinase activities representing hexokinase were slightly increased. Between the three genotypes there were no gross differences (data not shown). Furthermore, there were minimal differences between wri1-1 and wild type in transcript accumulation of the genes encoding hexokinase and pyruvate kinase in 11-d-old seedlings (data not shown). There was less HXK1 transcript present in the wri1-1 mutant seedlings, but message levels for HXK1 were increased in the transgenic line and in the presence of sugars. Interestingly, transcript levels for the pyruvate kinase gene PK1 generally decreased in the presence of sugar contrary to the HXK1 transcript. Based on these results it is concluded that glycolysis is active in 11-d-old seedlings that have progressed through the establishment phase during which sugar supplementation could alleviate the deficiency in the wri1-1 mutant.
DISCUSSION
Previously studying the wri1 mutant and the recently identified WRI1 protein, our primary focus was on the role of this gene in embryo development, i.e. oil accumulation and associated metabolism (Focks and Benning, 1998; Ruuska et al., 2002; Cernac and Benning, 2004). However, as our continued wri1-1 mutant analysis described above showed, WRI1 exerts its effects beyond embryo development into seed germination and presumably indirectly into seedling establishment. These are two physiologically and developmentally distinct stages in the plant life cycle with different demands on metabolism. Analysis of germination in Arabidopsis is straight forward and, unlike seed development inside siliques, provides an experimental system readily amenable to the addition of growth factors and metabolically active compounds permitting the dissection of WRI1 function in new ways.
WRI1 Affects ABA Sensitivity and Sugar Sensing But Not through ABI3
The wri1-1 mutant was clearly more ABA sensitive with regard to germination and the expression of ABA-responsive genes, and was also more sensitive to osmotic stress. Because one of the ABA-responsive genes affected in the wri1-1 mutant was ABI3, we initially suspected that WRI1 might act through ABI3. However, the partial suppression of the germination phenotype of wri1-1 by abi3-3 and vice versa in the presence and absence of ABA (Fig. 4A) apparent in the double mutant suggests that the two factors act in parallel pathways, one negatively and one positively affecting germination. As WRI1 regulates the expression of genes involved in carbon metabolism (Ruuska et al., 2002; Cernac and Benning, 2004; Masaki et al., 2005) it might provide an indirect link between sugar and ABA signaling pathways evident in the enhanced ABA sensitivity of germination in the wri1-1 mutant. It has been known for some time that the addition of Suc to the medium alleviates the inhibition of germination by ABA (Finkelstein and Lynch, 2000; Finkelstein and Gibson, 2002). Based on our current findings, WRI1 might be a mechanistic component of this sugar response. Indeed, our analysis of germination of wri1-1 and the two WRI1-cDNA-expressing lines when grown on sugar analogs (Fig. 5) support a role for WRI1 in sugar signaling through the hexokinase pathway. The critical observation was the decreased sensitivity of germination of the WRI1-cDNA-expressing lines to low concentration of 2-deoxyGlc, which apparently can be phosphorylated by hexokinase, but cannot be efficiently further metabolized by glycolysis. Toxic effects of 2-deoxyGlc on germination have been interpreted as caused by its interference with sugar sensing through the hexokinase pathway (Pego et al., 1999). Apparently, increased activity of WRI1 in the transgenic lines counters the effect of the analog at low concentrations by increasing overall glycolytic activity (at least 48 h after imbibition [Fig. 6A] and more obvious at the transcript levels [Fig. 6B]), but at higher concentrations the system is overcome by the presumed accumulation of the phosphorylated 2-deoxyGlc intermediate. Accumulation of the phosphorylated analog might simply block the generation of energy through glycolysis needed for germination. Feeding sugar to seedlings was found to increase the WRI1 transcript amounts (Fig. 3; Cernac and Benning, 2004), an effect that was independently shown in leaf disc feeding experiments (Masaki et al., 2005) and suggests a stimulation of WRI1 activity in the presence of sugar. While WRI1 is directly involved in the control of expression of sugar-sensitive genes (Ruuska et al., 2002; Cernac and Benning, 2004; Masaki et al., 2005), it certainly does not represent the only means by which plants communicate changes in sugar levels because we found that Suc is still capable of alleviating the suppression of germination by ABA in a wri1-1 background, though to a lesser extent than in wild type.
The Role of WRI1 in Hypocotyl Elongation and Seedling Establishment
It has been documented that plants defective in gluconeogenesis or the conversion of lipids into precursors of the glyoxylate cycle show reduced hypocotyl elongation and seedling establishment (Eastmond et al., 2000; Rylott et al., 2003; Penfield et al., 2004). The wri1-1 seedlings failed to elongate their hypocotyl in the dark and supplementing the medium with sugar had only a slight effect on hypocotyl elongation. Furthermore, wri1-1 mutant plants showed a reduced germination that was only minimally rescued by Tween-80 and not at all by sugar or gibberellin (Table I). On the contrary, seedling establishment was rescued by Suc (Fig. 5), as was also shown for other mutants affected in seed oil catabolism (Pinfield-Wells et al., 2005). These findings clearly show that the roles played by WRI1 or the lack of it in the mutant are qualitatively different during germination and the later phase of seedling establishment with hypocotyl elongation in the dark being at the transition of these two processes. We have previously shown that WRI1 expression is primarily in the roots, flowers, developing embryos, and young seedlings (Cernac and Benning, 2004), findings that were confirmed by the global analysis of transcripts at different developmental stages of Arabidopsis (Schmid et al., 2005). These are tissues in which glycolysis and/or gluconeogenesis are expected to be highly active in the wild type, two processes that share a number of biochemical reactions and that are critical for germination and seedling establishment as they generate energy and/or building blocks. Our data suggest that in the wri1-1 mutant during elongation of the hypocotyls in the dark glycolysis is impeded, because neither Suc nor Tween-80 can rescue the wri1-1 defect in hypocotyl elongation. Indeed, glycolytic activity was found to be reduced in wri1-1 germinating seeds prior to entering the seedling establishment phase (Fig. 6). In addition, this conclusion is supported by a mutant specifically deficient in gluconeogenesis because it lacks the critical enzyme phosphoenolpyruvate carboxykinase (Penfield et al., 2004). While this mutant showed a similar hypocotyl elongation defect in the dark as wri1-1, its defect could be rescued by Suc. These findings on hypocotyl elongation in the wri1-1 mutant are consistent with our hypothesis and previous findings that WRI1 affects glycolytic activity in tissues in which it is active.
The Cause of Decreased Seedling Establishment in the wri1-1 Mutant
An impairment of sugar metabolism, i.e. glycolysis, in nonphotosynthetic tissues should have a detrimental effect on seedling establishment as the root is completely dependent upon the import and metabolism of sugar initially from the cotyledons breaking down lipids and converting them to sugars, and later from cotyledons and true leaves conducting photosynthesis. Our observations of the wri1-1 mutant mirror those seen with mutants deficient in the glyoxylate cycle (Eastmond et al., 2000; Cornah et al., 2004), gluconeogenesis (Rylott et al., 2003; Penfield et al., 2005), or transport of various intermediates derived from lipid breakdown (Hayashi et al., 1998; Footitt et al., 2002; Lawand et al., 2002). A striking difference in root elongation as compared to germination and hypocotyl elongation in the dark is that root elongation of wri1-1 can be rescued by sugar, suggesting that roots of the mutant are capable of utilizing and converting Suc for energy purposes and building block generation. Thus glycolysis must be functional in these tissues. This hypothesis was confirmed by measuring transcript amounts and enzyme activity for two enzymes critical in glycolysis, hexokinase and pyruvate kinase, in roots of 11-d-old plantlets. Subtle changes in enzyme activity observed between the treatments happened on top of a considerable background activity and it is likely that glycolytic activity was not limiting in 11-d-old seedlings. Therefore, WRI1 does not to seem to be directly involved in regulation of glycolysis once seedlings start growing during the process of establishment. However, what is presumably limiting during this phase of development in the mutant is the sparse amount of seed storage oil, which is reduced at least 80% in wri1-1 seeds (Focks and Benning, 1998). Therefore, offering Suc that can provide energy and building blocks at this stage completely restores seedling establishment. In this aspect, wri1-1 resembles a mutant lacking seed oil because of a defect in the gene encoding diacylglycerol acyltransferase, tag1 (Routaboul et al., 1999; Zou et al., 1999; Lu and Hills, 2002). Therefore, the role of WRI1 on seedling establishment is indirect, by way of its involvement in the up-regulation of carbohydrate metabolism in the developing seed thereby ensuring the presence of storage oil needed for seedling establishment.
MATERIALS AND METHODS
Plant Material and Seed Germination Assays
All Arabidopsis (Arabidopsis thaliana) plants were of the Columbia-2 ecotype. The wri1-1 mutant has been described by Focks and Benning (1998) and the abi3-3 mutant by Nambara et al. (1992). Transgenic lines 101 and 106 were independently isolated and were expressing the WRI1 cDNA under the control of the 35S-cauliflower mosaic virus (CaMV) promoter rescuing the homozygous wri1-1 mutation, with line 106 exhibiting higher WRI1 expression and stronger ectopic expression phenotypes (Cernac and Benning, 2004). Seeds used for germination assays were harvested from plants that were grown in parallel on the same shelf in the growth chamber using conditions as previously described (Cernac and Benning, 2004). The seeds were harvested as soon as the siliques had dried out (approximately 2.5 months after transfer to soil). Harvested seeds were after ripened for 6 weeks at room temperature prior to use. Seeds were surface sterilized and plated as described (Cernac and Benning, 2004). The medium used was half-strength Murashige and Skoog (Murashige and Skoog, 1962) pH 6.2 with additions as indicated. The ABA stock solution was prepared by dissolving mixed enantiomers of ABA from Sigma-Aldrich (A1049) in 100% ethanol. The ABA stock was less than 4 weeks old and was stored at −20°C. Tween-80 (Sigma-Aldrich P 1754) was added as described (Lin et al., 2004). Palmitic and oleic acids were purchased from Sigma-Aldrich (P-0500-10, O1008). Suc, Glc, sorbitol (Sigma-Aldrich S 8143), 2-deoxyGlc (Sigma-Aldrich D 8375), 3-O-methyGlc (Sigma-Aldrich M 4879-5G), and palatinose (Sigma-Aldrich P 2007-5G) used in germination assays were each prepared in 1 m stocks in water and filter sterilized. All compounds tested were added to autoclaved half-strength Murashige and Skoog medium after it had cooled to 44°C. Different lines were compared in sectors on the same plates. Between 120 and 200 seeds for each genotype were assayed per treatment. Four ABA assays were done with seed generated from three independently generated sets. Assays containing gibberellic acid (Sigma-Aldrich G 7645) were conducted in the same manner as those for ABA, with the same seed groups. The seeds were stratified for 3 d in the dark at 4°C prior to incubation as previously described (Cernac and Benning, 2004). For the analysis of immature seeds, 11-to-13-d-old siliques were harvested from wri1-1, wild type, abi3-3, and abi3-3 wri1-1. The seeds were dissected out and 120 to 200 sown onto the medium with water. Plates were incubated as above and germination was scored up to the 4th d after incubation.
Growth of Seedlings for Root Elongation and Enzyme Assays
Root elongation rate assays were carried out on half-strength Murashige and Skoog medium. Seeds were sown in a straight line, 15 to 20 for wild type and rescued lines and 30 to 50 for wri1-1. Seedling root length was measured every 24 h starting on the 4th d post incubation through the 12th d. For RNA and protein extraction seeds were sown as they were for root elongation except 15 × 150 mm plates were used. The seeds were stratified as above and the plates incubated vertically for 11 d. Entire wri1-1 seedlings were harvested for the 0 mm Suc treatment. Roots and shoots were separated by cutting along the line of plants at the base of the hypocotyl. Harvested material was wrapped in aluminum packets, weighed, and frozen in liquid nitrogen.
Endosperm Lipid Measurements
Seeds of plants grown as described for ABA assays were sown onto water-soaked 3MM Whatman paper and allowed to imbibe at 4°C for 3 h. The seed coats and attached endosperm of 15 seeds were prepared according to Penfield et al. (2004). Lipids were quantified by gas chromatography of fatty acid methyl esters (Cernac and Benning, 2004). A total of six measurements were done per genotype, representing two different groups of plants.
Preparation of Tissue for Light Microscopy
Roots from 8-d-old seedlings were harvested and fixed as previously described (Hoffmann-Benning et al., 1994). In short, roots were fixed in 4% glutaraldehyde in 0.1 m sodium phosphate buffer pH 7.2 on ice, followed by three washes and secondary fixation in 0.5% osmium tetroxide in the same buffer for 2 h at room temperature. The tissue was washed, dehydrated in a gradual series of ethanol, and embedded in Embed 812 (EM Sciences). Thin sections were cut in the area of the root differentiation zone and stained with 1% toluidine blue/1% boric acid. Pictures were taken with a Leica DMLB light microscope and recorded with a Canon Power Shot A95 digital camera.
Transcript Analysis
ABA induction assays were conducted according to Brocard-Gifford et al. (2004) with the following exceptions: Seeds were sterilized as above and sown onto nylon mesh placed on the surface of the agar plate. Water was used to spread the seed. Approximately 300 seeds were sown onto each plate, which were incubated vertically. After 6 d incubation, the filters containing the seedlings were transferred to plates containing or lacking ABA or Suc as indicated. RNA was extracted according to Ruuska and Ohlrogge (2001). Seedlings were harvested and frozen in liquid nitrogen. RNA gels, blotting, and probing were done as previously described (Cernac and Benning, 2004). The cDNAs used for probes were AY254038 (WRI1), At4g29130 (HXK1), At3g22960 (PK1), At5g52920 (PK2), At3g51810 (AtEM1), At2g40170 (AtEM6), At2g42540 (COR15a), At1g20440 (COR47), At5g52310 (COR78), At3g24650 (ABI3), At2g36270 (ABI5), and At2g40220 (ABI4).
Protein Extraction and Enzyme Assays
Approximately 100 mg of tissue was used for each protein extraction. Tissue was ground frozen using mortar and pestle and transferred to 1.5 mL tubes. Protein for enzyme assays was extracted as described previously except that bovine serum albumin was omitted from the extraction buffer (Focks and Benning, 1998). Protein extracts were used immediately for enzyme assays in a double-beam spectrophotometer (Uvicon 930, Kontron Instruments) equipped with a cell changer (model 900, Kontron). All reagents were from Sigma-Aldrich. Pyruvate kinase was assayed in 1 mL total volume in a reaction mix consisting of 50 mm MOPS pH 7.0, 5% PEG-8000, 50 mm KCl, 15 mm MgCl2, 1 mm dithiothreitol, 2 mm phosphoenolpyruvate, 1 mm ADP, 0.2 mm NADH, and 2 units of desalted lactate dehydrogenase. Pyruvate kinase was also assayed at pH 8.0 by using 50 mm HEPES-KOH pH 8.0 instead of MOPS. The same trends were observed using both conditions. Correction for phosphoenolpyruvate phosphatase activity was carried out by the omission of ADP from the reaction mix. Glucokinase and Fructokinase were assayed as previously described, but in 1 mL total volume (Wiese et al., 1999). All reactions were done at 30°C, were initiated by the addition of protein extracts, and were linear for at least 5 min. Total protein in each extract was determined using the DC protein assay kit from Bio-Rad.
Sequence data from this article can be found in the GenBank/EMBL data libraries under accession number AY254038.
Acknowledgments
We are grateful to Ruth Finkelstein and Mike Thomashow for providing us with cDNAs to probe RNA blots, and to Ruth Finkelstein for the abi3-3 mutant.
This work was supported in part by BASF-Plant Science.
The author responsible for distribution of materials integral to the findings presented in this article in accordance with the policy described in the Instructions for Authors (www.plantphysiol.org) is: Christoph Benning (benning@msu.edu).
Article, publication date, and citation information can be found at www.plantphysiol.org/cgi/doi/10.1104/pp.106.079574.
References
- Arenas-Huertero F, Arroyo A, Zhou L, Sheen J, Leon P (2000) Analysis of Arabidopsis glucose insensitive mutants, gin5 and gin6, reveals a central role of the plant hormone ABA in the regulation of plant vegetative development by sugar. Genes Dev 14: 2085–2096 [PMC free article] [PubMed] [Google Scholar]
- Arroyo A, Bossi F, Finkelstein RR, Leon P (2003) Three genes that affect sugar sensing (abscisic acid insensitive 4, abscisic acid insensitive 5, and constitutive triple response 1) are differentially regulated by glucose in Arabidopsis. Plant Physiol 133: 231–242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avonce N, Leyman B, Mascorro-Gallardo JO, Van Dijck P, Thevelein JM, Iturriaga G (2004) The Arabidopsis trehalose-6-P synthase AtTPS1 gene is a regulator of glucose, abscisic acid, and stress signaling. Plant Physiol 136: 3649–3659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avonce N, Leyman B, Thevelein J, Iturriaga G (2005) Trehalose metabolism and glucose sensing in plants. Biochem Soc Trans 33: 276–279 [DOI] [PubMed] [Google Scholar]
- Barroco RM, Van Poucke K, Bergervoet JH, De Veylder L, Groot SP, Inze D, Engler G (2005) The role of the cell cycle machinery in resumption of postembryonic development. Plant Physiol 137: 127–140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baud S, Boutin JP, Miquel M, Lepiniec L, Rochat C (2002) An integrated overview of seed development in Arabidopsis thaliana ecotype WS. Plant Physiol Biochem 40: 151–160 [Google Scholar]
- Bewley JD (1997) Seed germination and dormancy. Plant Cell 9: 1055–1066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocard-Gifford I, Lynch TJ, Garcia ME, Malhotra B, Finkelstein RR (2004) The Arabidopsis thaliana ABSCISIC ACID-INSENSITIVE8 encodes a novel protein mediating abscisic acid and sugar responses essential for growth. Plant Cell 16: 406–421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cernac A, Benning C (2004) WRINKLED1 encodes an AP2/EREB domain protein involved in the control of storage compound biosynthesis in Arabidopsis. Plant J 40: 575–585 [DOI] [PubMed] [Google Scholar]
- Cheng JC, Lertpiriyapong K, Wang S, Sung ZR (2000) The role of the Arabidopsis ELD1 gene in cell development and photomorphogenesis in darkness. Plant Physiol 123: 509–520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornah JE, Germain V, Ward JL, Beale MH, Smith SM (2004) Lipid utilization, gluconeogenesis, and seedling growth in Arabidopsis mutants lacking the glyoxylate cycle enzyme malate synthase. J Biol Chem 279: 42916–42923 [DOI] [PubMed] [Google Scholar]
- Cortes S, Gromova M, Evrard A, Roby C, Heyraud A, Rolin DB, Raymond P, Brouquisse RM (2003) In plants, 3-o-methylglucose is phosphorylated by hexokinase but not perceived as a sugar. Plant Physiol 131: 824–837 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crowe AJ, Abenes M, Plant A, Moloney MM (2000) The seed-specific transactivator, ABI3, induces oleosin gene expression. Plant Sci 151: 171–181 [DOI] [PubMed] [Google Scholar]
- Dekkers BJ, Schuurmans JA, Smeekens SC (2004) Glucose delays seed germination in Arabidopsis thaliana. Planta 218: 579–588 [DOI] [PubMed] [Google Scholar]
- Eastmond PJ, Germain V, Lange PR, Bryce JH, Smith SM, Graham IA (2000) Postgerminative growth and lipid catabolism in oilseeds lacking the glyoxylate cycle. Proc Natl Acad Sci USA 97: 5669–5674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkelstein RR, Gampala SS, Rock CD (2002) Abscisic acid signaling in seeds and seedlings. Plant Cell (Suppl) 14: S15–S45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkelstein RR, Gibson SI (2002) ABA and sugar interactions regulating development: cross-talk or voices in a crowd? Curr Opin Plant Biol 5: 26–32 [DOI] [PubMed] [Google Scholar]
- Finkelstein RR, Lynch TJ (2000) Abscisic acid inhibition of radicle emergence but not seedling growth is suppressed by sugars. Plant Physiol 122: 1179–1186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkelstein RR, Somerville CR (1990) 3 classes of abscisic-acid (Aba)-insensitive mutations of Arabidopsis define genes that control overlapping subsets of Aba responses. Plant Physiol 94: 1172–1179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Focks N, Benning C (1998) wrinkled1: a novel, low-seed-oil mutant of Arabidopsis with a deficiency in the seed-specific regulation of carbohydrate metabolism. Plant Physiol 118: 91–101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Footitt S, Slocombe SP, Larner V, Kurup S, Wu Y, Larson T, Graham I, Baker A, Holdsworth M (2002) Control of germination and lipid mobilization by COMATOSE, the Arabidopsis homologue of human ALDP. EMBO J 21: 2912–2922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Germain V, Rylott EL, Larson TR, Sherson SM, Bechtold N, Carde JP, Bryce JH, Graham IA, Smith SM (2001) Requirement for 3-ketoacyl-CoA thiolase-2 in peroxisome development, fatty acid beta-oxidation and breakdown of triacylglycerol in lipid bodies of Arabidopsis seedlings. Plant J 28: 1–12 [DOI] [PubMed] [Google Scholar]
- Gibson SI (2004) Sugar and phytohormone response pathways: navigating a signalling network. J Exp Bot 55: 253–264 [DOI] [PubMed] [Google Scholar]
- Gibson SI (2005) Control of plant development and gene expression by sugar signaling. Curr Opin Plant Biol 8: 93–102 [DOI] [PubMed] [Google Scholar]
- Giraudat J, Hauge BM, Valon C, Smalle J, Parcy F, Goodman HM (1992) Isolation of the Arabidopsis ABI3 gene by positional cloning. Plant Cell 4: 1251–1261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi M, Toriyama K, Kondo M, Nishimura M (1998) 2,4-Dichlorophenoxybutyric acid-resistant mutants of Arabidopsis have defects in glyoxysomal fatty acid beta-oxidation. Plant Cell 10: 183–195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi M, Yagi M, Nito K, Kamada T, Nishimura M (2005) Differential contribution of two peroxisomal protein receptors to the maintenance of peroxisomal functions in Arabidopsis. J Biol Chem 280: 14829–14835 [DOI] [PubMed]
- Hobbs DH, Lu C, Hills MJ (1999) Cloning of a cDNA encoding diacylglycerol acyltransferase from Arabidopsis thaliana and its functional expression. FEBS Lett 452: 145–149 [DOI] [PubMed] [Google Scholar]
- Hoffmann-Benning S, Klomparens KL, Kende H (1994) Characterization of growth-related osmiophilic particles in rice. Ann Bot (Lond) 74: 563–572 [Google Scholar]
- Karssen CM, Brinkhorst-van der Swan DLC, Breekland AE, Koorneef M (1983) Induction of dormancy during seed development by endogenous abscisic acid: studies on abscisic acid deficient genotypes of Arabidopsis thaliana (L.) Heynh. Planta 157: 158–165 [DOI] [PubMed] [Google Scholar]
- Lawand S, Dorne AJ, Long D, Coupland G, Mache R, Carol P (2002) Arabidopsis A BOUT DE SOUFFLE, which is homologous with mammalian carnitine acyl carrier, is required for postembryonic growth in the light. Plant Cell 14: 2161–2173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin Y, Cluette-Brown JE, Goodman HM (2004) The peroxisome deficient Arabidopsis mutant sse1 exhibits impaired fatty acid synthesis. Plant Physiol 135: 814–827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin Y, Sun L, Nguyen LV, Rachubinski RA, Goodman HM (1999) The Pex16p homolog SSE1 and storage organelle formation in Arabidopsis seeds. Science 284: 328–330 [DOI] [PubMed] [Google Scholar]
- Lu C, Hills MJ (2002) Arabidopsis mutants deficient in diacylglycerol acyltransferase display increased sensitivity to abscisic acid, sugars, and osmotic stress during germination and seedling development. Plant Physiol 129: 1352–1358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masaki T, Mitsui N, Tsukagoshi H, Nishii T, Morikami A, Nakamura K (2005) Activator of Spomin∷LUC1/WRINKLED1 of Arabidopsis thaliana transactivates sugar-inducible promoters. Plant Cell Physiol 46: 547–556 [DOI] [PubMed] [Google Scholar]
- Moore B, Zhou L, Rolland F, Hall Q, Cheng WH, Liu YX, Hwang I, Jones T, Sheen J (2003) Role of the Arabidopsis glucose sensor HXK1 in nutrient, light, and hormonal signaling. Science 300: 332–336 [DOI] [PubMed] [Google Scholar]
- Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15: 473–497 [Google Scholar]
- Nambara E, Keith K, McCourt P, Naito S (1994) Isolation of an internal deletion mutant of the Arabidopsis thaliana ABI3 gene. Plant Cell Physiol 35: 509–513 [PubMed] [Google Scholar]
- Nambara E, Marion-Poll A (2003) ABA action and interactions in seeds. Trends Plant Sci 8: 213–217 [DOI] [PubMed] [Google Scholar]
- Nambara E, Naito S, McCourt P (1992) A mutant of Arabidopsis which is defective in seed development and storage protein accumulation is a new abi3 allele. Plant J 2: 435–441 [Google Scholar]
- Pego JV, Weisbeek PJ, Smeekens SC (1999) Mannose inhibits Arabidopsis germination via a hexokinase-mediated step. Plant Physiol 119: 1017–1023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penfield S, Graham S, Graham IA (2005) Storage reserve mobilization in germinating oilseeds: Arabidopsis as a model system. Biochem Soc Trans 33: 380–383 [DOI] [PubMed] [Google Scholar]
- Penfield S, Rylott EL, Gilday AD, Graham S, Larson TR, Graham IA (2004) Reserve mobilization in the Arabidopsis endosperm fuels hypocotyl elongation in the dark, is independent of abscisic acid, and requires PHOSPHOENOLPYRUVATE CARBOXYKINASE1. Plant Cell 16: 2705–2718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Periappuram C, Steinhauer L, Barton DL, Taylor DC, Chatson B, Zou J (2000) The plastidic phosphoglucomutase from Arabidopsis: a reversible enzyme reaction with an important role in metabolic control. Plant Physiol 122: 1193–1199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinfield-Wells H, Rylott EL, Gilday AD, Graham S, Job K, Larson TR, Graham IA (2005) Sucrose rescues seedling establishment but not germination of Arabidopsis mutants disrupted in peroxisomal fatty acid catabolism. Plant J 43: 861–872 [DOI] [PubMed] [Google Scholar]
- Plaxton WC (1996) The organization and regulation of plant glycolysis. Annu Rev Plant Physiol Plant Mol Biol 47: 185–214 [DOI] [PubMed] [Google Scholar]
- Qi Q, Rose PA, Abrams GD, Taylor DC, Abrams SR, Cutler AJ (1998) (+)-Abscisic acid metabolism, 3-ketoacyl-coenzyme A synthase gene expression, and very-long-chain monounsaturated fatty acid biosynthesis in Brassica napus embryos. Plant Physiol 117: 979–987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raz V, Bergervoet JH, Koornneef M (2001) Sequential steps for developmental arrest in Arabidopsis seeds. Development 128: 243–252 [DOI] [PubMed] [Google Scholar]
- Reiser J, Linka N, Lemke L, Jeblick W, Neuhaus HE (2004) Molecular physiological analysis of the two plastidic ATP/ADP transporters from Arabidopsis. Plant Physiol 136: 3524–3536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolland F, Moore B, Sheen J (2002) Sugar sensing and signaling in plants. Plant Cell (Suppl) 14: S185–S205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rook F, Corke F, Card R, Munz G, Smith C, Bevan MW (2001) Impaired sucrose-induction mutants reveal the modulation of sugar-induced starch biosynthetic gene expression by abscisic acid signalling. Plant J 26: 421–433 [DOI] [PubMed] [Google Scholar]
- Routaboul JM, Benning C, Bechtold N, Caboche M, Lepiniec L (1999) The TAG1 locus of Arabidopsis encodes for a diacylglycerol acyltransferase. Plant Physiol Biochem 37: 831–840 [DOI] [PubMed] [Google Scholar]
- Ruuska SA, Girke T, Benning C, Ohlrogge JB (2002) Contrapuntal networks of gene expression during Arabidopsis seed filling. Plant Cell 14: 1191–1206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruuska SA, Ohlrogge JB (2001) Protocol for small-scale RNA isolation and transcriptional profiling of developing Arabidopsis seeds. Biotechniques 31: 752–758 [DOI] [PubMed] [Google Scholar]
- Rylott EL, Gilday AD, Graham IA (2003) The gluconeogenic enzyme phosphoenolpyruvate carboxykinase in Arabidopsis is essential for seedling establishment. Plant Physiol 131: 1834–1842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmid M, Davison TS, Henz SR, Pape UJ, Demar M, Vingron M, Schölkopf B, Weigel D, Lohmann J (2005) A gene expression map of Arabidopsis thaliana development. Nat Genet 37: 501–506 [DOI] [PubMed] [Google Scholar]
- To JP, Reiter WD, Gibson SI (2002) Mobilization of seed storage lipid by Arabidopsis seedlings is retarded in the presence of exogenous sugars. BMC Plant Biol 2: 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiese A, Groner F, Sonnewald U, Deppner H, Lerchl J, Hebbeker U, Flugge U, Weber A (1999) Spinach hexokinase I is located in the outer envelope membrane of plastids. FEBS Lett 461: 13–18 [DOI] [PubMed] [Google Scholar]
- Zhou L, Jang JC, Jones TL, Sheen J (1998) Glucose and ethylene signal transduction crosstalk revealed by an Arabidopsis glucose-insensitive mutant. Proc Natl Acad Sci USA 95: 10294–10299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zolman BK, Monroe-Augustus M, Thompson B, Hawes JW, Krukenberg KA, Matsuda SP, Bartel B (2001) chy1, an Arabidopsis mutant with impaired beta-oxidation, is defective in a peroxisomal beta-hydroxyisobutyryl-CoA hydrolase. J Biol Chem 276: 31037–31046 [DOI] [PubMed] [Google Scholar]
- Zou J, Wei Y, Jako C, Kumar A, Selvaraj G, Taylor DC (1999) The Arabidopsis thaliana TAG1 mutant has a mutation in a diacylglycerol acyltransferase gene. Plant J 19: 645–653 [DOI] [PubMed] [Google Scholar]