Abstract
Hepatitis B virus (HBV) X protein (HBx) is considered to play a role in the development of hepatocellular carcinoma (HCC) during HBV infection. HCC was shown to be more prevalent in men than in women. Estrogen, which exerts its biological function through estrogen receptor (ER), can inhibit HBV replication. ERΔ5, an ERα variant lacking exon 5, was found to be preferentially expressed in patients with HCC compared with patients with normal livers. Here, we report the biological role of ERΔ5 and a novel link between HBx and ERα signaling in hepatoma cells. ERΔ5 interacts with ERα in vitro and in vivo and functions as a dominant negative receptor. Both ERα and ERΔ5 associate with HBx. HBx decreases ERα-dependent transcriptional activity, and HBx and ERΔ5 have additive effect on suppression of ERα transactivation. The HBx deletion mutant that lacks the ERα-binding site abolishes the HBx repression of ERα. HBx, ERα and histone deacetylase 1 (HDAC1) form a ternary complex. Trichostatin A, a specific inhibitor of HDAC enzyme, can restore the transcriptional activity of ERα inhibited by HBx. Our data suggest that HBx and ERΔ5 may play a negative role in ERα signaling and that ERα agonists may be developed for HCC therapy.
INTRODUCTION
Prolonged infection with Hepatitis B virus (HBV) has been clearly recognized as a major etiological factor for hepatocellular carcinoma (HCC) (1). HBx, a virally encoded protein of 154 amino acids, has been shown to have multifunctional activities relevant to HBV-mediated oncogenesis (2). HBx is involved in neoplastic transformation in cultured cells and can induce liver cancer in transgenic mice. Although HBx does not bind to double-stranded DNA, it regulates transcription of a variety of cellular and viral genes by interacting with cellular proteins and/or components of signal transduction pathways. HBx has been shown to interact with transcriptional factors such as RPB5 of RNA polymerase (3), TATA-binding protein (4), basic region/leucine zipper (bZIP) proteins (5) and the tumor suppressor p53 (6). Besides, it can also associate with serine protease TL2 (7) and cellular DNA repair protein (8). The interaction of HBx with these proteins leads to activation of signal transduction pathways including the Ras/Raf/mitogen-activated protein kinase, protein kinase C, Jak1-STAT and nuclear factor κB pathways (9–12). However, the intracellular signaling pathways in which Hbx is involved are not fully elucidated.
Estrogen was shown to suppress HBV replication in male athymic mice transplanted with HBV-transfected HepG2 cells (13). The fact that HCC is more prevalent in men than in women suggests that estrogen may play an important role in the development of HCC (14–17). Estrogen exerts its function through its two nuclear receptors, estrogen receptor α and β (ERα and ERβ) (18–21). ERα and ERβ share structural similarity characterized by several functional domains. Two distinct activation function (AF) domains, AF-1 and AF-2, located at the N-terminus and the C-terminus, respectively, contribute to the transcriptional activity of the two receptors. The DNA-binding domain (DBD) of the two receptors is well conserved and centrally located. Activation of ERs is responsible for many biological processes, including cell growth, differentiation and apoptosis.
ERα has been well characterized in human liver (22). ERα is expressed in the liver of both healthy individuals and patients with HCC, with no differences in the pattern of expression (23,24). In contrast, the mutant form with the entire exon 5 deleted (ERΔ5) is preferentially expressed in patients with HCC compared with patients with normal livers (25). The presence of the liver ERΔ5 transcript in the tumor was the strongest negative predictor of survival in operable HCC (26–28). Its presence also correlates with a higher clinical aggressiveness of the tumor in comparison with tumors characterized by wild-type ERα (wt ERα) transcript. High rates of ERΔ5 expression have been shown to present in men at high-risk for HCC development. ERΔ5 encodes the hormone-independent AF-1 domain, as well as the DBD. Although ERΔ5 was demonstrated to be coexpressed with wt ERα in HCC, the role of ERΔ5 in ERα signaling remains to be investigated.
On the basis of in vivo and in vitro functional relevance of the estrogen/ERα axis and HBx in the development of HCC, we hypothesized that HBx may play a role in ERα signaling. Here, we show that ERΔ5 has a dominant negative activity in hepatoma cells when expressed together with wt ERα. HBx decreases ERα transcriptional activity, and HBx and ERΔ5 have additive effect on inhibition of ERα transactivation. We further present in vitro and in vivo evidence that both HBx and ERΔ5 interact with ERα. HBx inhibits ERα signaling possibly through recruitment of histone deacetylase 1 (HDAC1).
MATERIALS AND METHODS
Plasmids
The reporter constructs ERE-Luc (29), C3-LUC (30,31), pS2-LUC (32) and pS2ΔERE-LUC (33), and expression vector for ERα have been described previously. For the generation of FLAG-tagged full-length HBx, human HBx DNA was amplified by PCR using pHBV3091 as a template (34). The amplified HBx DNA was cloned into pcDNA3 vector harboring FLAG epitope sequence (pcDNA3-FLAG). The deletion mutant of HBx (Δ73-120) was constructed by inserting the recombinant PCR-generated fragment from the HBx DNA into the pcDNA3-FLAG vector. The expression vectors for the full-length ERα (1–595), ERα AF1 (1–185), ERα DBD (180–282), ERα AF2 (282-595), ERα AF2 (302–595) and ERΔ5 (370 amino acids with a novel five-amino acid residue COOH terminus) were made by introducing the corresponding cDNAs into pcDNA3 (Invitrogen). Enhanced green fluorescent protein (EGFP)-tagged HBx construct was generated by inserting HBx DNA into pEGFP-C1 (Clontech), and red fluorescent protein (RFP)-tagged ERα construct by inserting ERα cDNA into pDsRed-N1 (Clontech). A cDNA fragment encoding entire coding region of HDAC1 was obtained by RT–PCR using as a template total RNA from the human hepatoma cell line HepG2, and the cDNA fragment was inserted in frame into a pcDNA3 vector linked with HA tag at the amino terminus. Plasmids encoding GST-fusion proteins were prepared by amplification of each sequence by standard PCR methods, and the resulting fragments were cloned in frame into pGEX-KG (Amersham Pharmacia Biotech) using appropriate restriction sites. All of the constructs were confirmed by sequencing. Details of cloning are available upon request.
Transfection and luciferase assay
HepG2 and SMMC-7721 cells were routinely grown in DMEM (Invitrogen) supplemented with 10% fetal bovine serum (FBS). For transfection, cells were seeded in 12-well plates containing phenol red-free DMEM medium supplemented with 10% charcoal-stripped FBS (Hyclone). The cells were transfected using Lipofectamine 2000 (Invitrogen) with 0.2 µg of ERE-LUC, C3-LUC, pS2-LUC or pS2ΔERE-LUC reporter plasmid, 50 ng of ERα expression plasmid, 250 ng to 2 µg of the expression vector for HBx and 0.1 µg of β-galactosidase reporter as an internal control. The empty vector pcDNA3 was used to adjust the total amount of DNA. After treatment with 10 nM of 17β-estradiol (E2) and 100 nM 4-hydroxytamoxifen (4-OHT) for 24 h, or 100 nM trichostatin A (TSA) for 12 h, the cells were harvested, and luciferase and β-galactosidase activities were determined as described previously (35). All experiments were repeated at least five times.
GST pull-down assay
GST and GST-fusion proteins were expressed in E.coli DH5α, with the induction of protein expression performed at 20°C overnight (36). After large-scale preparation, purification of the recombinant proteins were performed according to the manufacturer's instruction (Pharmacia) using glutathione–Sepharose beads. The expression plasmid for the ERα, ERα deletion mutants, HBx or HDAC1 was used for in vitro transcription and translation in the TNT System (Promega). The 35S-labeled in vitro translated products were incubated with ∼10 µg of GST derivatives bound to glutathione–Sepharose beads in 500 µl binding buffer (50 mM Tris–HCl, pH 7.5, 150 mM NaCl, 1 mM EDTA, 0.3 mM DTT, 0.1% NP-40 and protease inhibitor tablets from Roche) at 4°C. The beads were precipitated, washed four times with binding buffer, eluted in SDS–PAGE sample buffer, and analyzed by SDS–PAGE. After electrophoresis, the gel was dried and exposed to X-ray films.
Coimmunoprecipitation
HepG2 cells were transfected with the indicated plasmids using Lipofectamine 2000 (Invitrogen), washed with phosphate-buffered saline (PBS), lysed in 0.5 ml lysis buffer (50 mM Tris, pH 8.0, 250 mM NaCl, 0.25% NP-40, 1 mM DTT and protease inhibitor tablets from Roche), and immunoprecipitated with anti-FLAG-agarose beads (Sigma) for 3 h at 4°C. The beads were centrifuged, washed four times with the lysis buffer, and eluted in 30 µl of SDS–PAGE sample buffer. The eluted proteins were separated by SDS–PAGE, followed by immunoblotting with anti-ERα (Santa Cruz Biotech), anti-HA (Sigma) or anti-FLAG (Sigma) according to the standard procedures.
For reimmunoprecipitation, the immune complexes precipitated with anti-FLAG were eluted under native condition by a competition with 3× FLAG peptide according to the manufacturer's instructions (Sigma). The eluate was precleared with 20 µl of 50% protein A agarose beads (Santa Cruz Biotech) for 30 min. Proteins were reprecipitated with anti-ERα or control serum (Santa Cruz Biotech) plus 20 µl of protein A agarose beads. Reprecipitates were washed four times with lysis buffer, eluted by boiling in SDS–PAGE sample buffer, and resolved by SDS–PAGE, followed by immunoblotting.
For detecting interaction of endogenous HBx with ERα, liver tissue from an HBV positive patient (General Hospital of PLA, Beijing) was lysed in 1.0 ml RIPA buffer (PBS, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS and protease inhibitor tablets from Roche), and immunoprecipitated with anti-ERα or control serum (Santa Cruz). After extensive washing with RIPA buffer, the immunoprecipitates were resolved by SDS–PAGE, followed by western blot analysis using anti-HBx (Chemicon).
Confocal microscopy
HepG2 cells were seeded in 6-well dishes with glass coverslips containing phenol red-free DMEM medium (Invitrogen) supplemented with 10% charcoal-stripped FBS (Hyclone). Cells were transiently transfected with the indicated plasmids using lipofectamine 2000. Six hours after transfection, cells were treated with 10 nM of 17β-estradiol (E2) for various times. Nuclear DNA was visualized with 4′,6′-diamidino-2-phenylindole (DAPI). The subcellular localization of EGFP-HBx and RFP-ERα was analyzed with a Radiance 2100 confocal microscope (Bio-Rad). Fluorescence was detected with appropriate filter sets (the green signal, excitation 488 nm, dichroic mirror 560 DCLPXR, emission HQ 515/30; the red signal, excitation 543 nm, dichroic mirror 650 DCLPXR, emission HQ590/70).
RESULTS
Repression of ERα transcriptional activity by ERΔ5
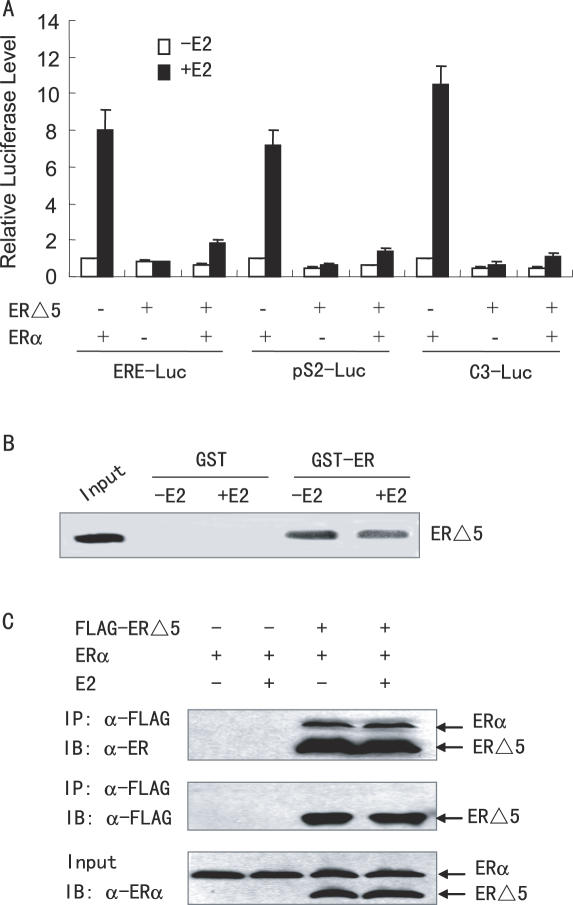
To examine the effects of wt ER and the ER variant ERΔ5 on E2-responsive gene transcription, ERα−negative human liver carcinoma HepG2 cells (37) were transiently transfected with wt ERα and/or ERΔ5, along with the synthetic estrogen-responsive reporter plasmid ERE-LUC, or the natural estrogen-responsive reporters pS2-Luc and Complement 3-Luc (C3-Luc). As shown in Figure 1, in the presence of E2, ERα stimulated the transcription of these reporter genes, whereas ERΔ5 had little effect. Importantly, when wt ER and ERΔ5 were co-transfected into the HepG2 cells in equal amounts, ERΔ5 was able to reduce the transcriptional activity of wt ER. These data suggest that ERΔ5 is able to interfere with the transcriptional activity of wt ER and to act as a dominant negative receptor.
Figure 1.
ERΔ5 represses ERα transcriptional activity through interaction with ERα. (A) ERΔ5 represses transcription of the ERE-Luc, pS2-Luc and C3-Luc reporters. HepG2 cells were co-transfected with 0.5 µg of the expression vector for ERΔ5, 0.5 µg of the expression plasmid for ERα and 0.2 µg of various luciferase reporter plasmids in the absence or presence of 10 nM of 17β-estradiol (E2). The luciferase activity obtained on transfection of the respective luciferase reporter and ERα without exogenous ERΔ5 in the absence of E2 was set as 1. (B) In vitro interaction of ERΔ5 with ERα. Glutathione–Sepharose beads bound with GST-ERα or with GST were incubated with 35S-labeled ERΔ5 in the absence or presence of 100 nM E2. After washing the beads, the bound proteins were eluted and subjected to SDS–PAGE and autoradiography. (C) In vivo interaction of ERΔ5 with ERα. ERα and FLAG-tagged ERΔ5 were co-transfected into HepG2 cells in the presence or absence of 10 nM E2. Cell lysates were immunoprecipitated (IP) by anti-FLAG M2 monoclonal antibody (Sigma), and the precipitates were then immunoblotted (IB) with anti-ERα polyclonal antibody (Santa Cruz Biotech).
Interaction of ERα with ERΔ5 in vitro and in vivo
The dominant negative property of the ER variant ERΔ5 could involve the formation of a heterodimer between ERΔ5 and wt ERα through protein–protein interactions. To test this possibility, GST pull-down experiments were performed in which in vitro translated 35S-methionine-labeled ERΔ5 was incubated with full-length GST-ERα or GST. As shown in Figure 1B, in both the absence and presence of E2, ERΔ5 bound to GST-ERα, but not to GST, suggesting that ERα physically interacts with ERΔ5 in vitro.
To determine whether ERΔ5 interacted with ERα in vivo, HepG2 cells were transfected with ERα and FLAG-tagged ERΔ5, and grown both in the absence and presence of 10 nM E2. The cells were then subjected to immunoprecipitation (IP) with FLAG antibody-conjugated agarose beads, followed by immunoblot (IB) with ERα antibody, which recognizes both ERα and ERΔ5 proteins. As shown in Figure 1C, ERα could be co-immunoprecipitated in a ligand-independent manner in the presence, but not in the absence, of FLAG- ERΔ5. These results suggest that ERΔ5 interacts with ERα in hepatoma cells.
Repression of ERα transcriptional activity by HBx
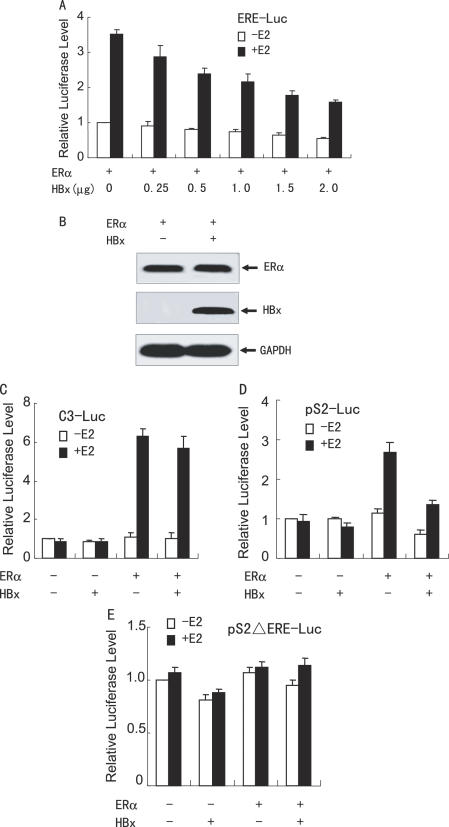
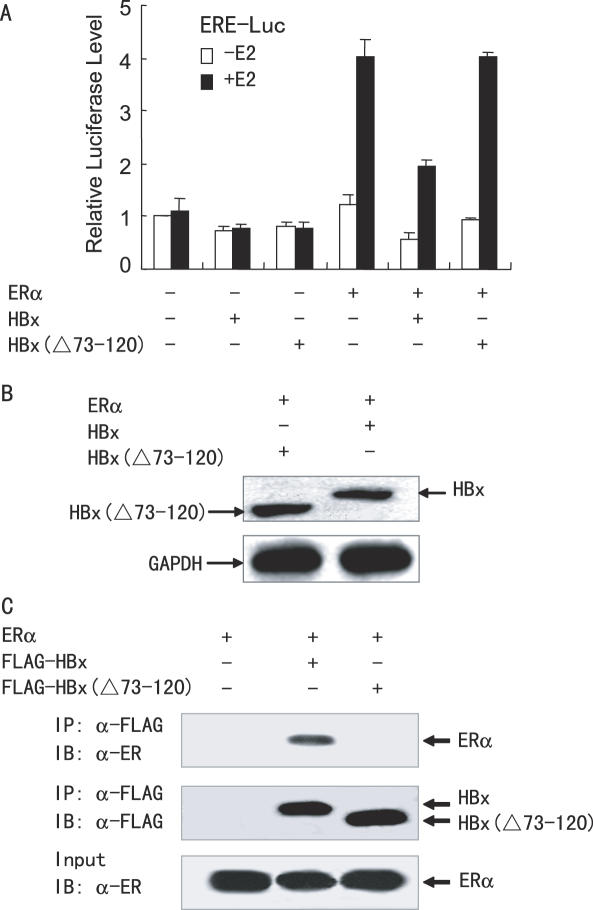
To gain insight into the functional role of HBx in HCC, the effect of HBx protein on ERα transactivation function was investigated. HepG2 cells were co-transfected with the synthetic estrogen response element (ERE)-containing reporter ERE-LUC, ERα, and increasing amounts of HBx. As shown in Figure 2A, as little as 250 ng of HBx was sufficient to exert a potent repression of ERα transactivation function and the extent of repression increased with increasing amount of HBx expression, suggesting that HBx decreased ERα transcriptional activity in a dose-dependent manner. Similar repression was observed in other liver cancer cells such as SMMC-7721 (data not shown). It should be noted that the decreased transcriptional activity was not a result of reduced ERα protein production (Figure 2B).
Figure 2.
HBx inhibits ERα-mediated transactivation function in hepatoma cells. (A) HepG2 cells were co-transfected with 0.2 µg of ERE-Luc, 50 ng of the expression plasmid for ERα and increasing amounts of the expression plasmid for FLAG-tagged HBx in the absence or presence of 10 nM E2. The luciferase activity obtained on transfection of ERE-Luc and ERα without exogenous HBx in the absence of E2 was set as 1. (B) Immunoblotting showing the ERα and HBx levels in HepG2 cells. Cells were transfected as in (A). Whole cell extracts were prepared from the cells transfected with 2.0 µg of the expression plasmid for HBx in the presence of 10 nM E2, and were detected with anti-ERα (Santa Cruz Biotech), anti-FLAG (Sigma) or anti-GAPDH (Biogenesis) antibody. (C–E) HepG2 cells were co-transfected with 50 ng of the expression plasmid for ERα, 1.0 µg of the expression plasmid for FLAG-tagged HBx, and 0.2 µg of C3-Luc (C), pS2-Luc (D) or pS2ΔERE-Luc (E), in the absence or presence of 10 nM E2. The luciferase activity obtained on transfection of the respective luciferase reporter without exogenous ERα and HBx in the absence of E2 was set as 1.
To test the effect of HBx on natural estrogen-responsive promoter activity, HepG2 cells were co-transfected with the natural ERE-containing reporter C3-Luc or pS2-Luc, together with expression vectors for ERα and HBx. As shown in Figure 2C and D, activation of the C3 promoter by ERα was not affected by HBx, whereas activation of the pS2 promoter by ERα was significantly repressed by HBx, indicating that the effect of HBx is promoter specific. Interestingly, mutations of the EREs at the pS2 promoter abolished both the E2-dependent gene activation and HBx-mediated repression (Figure 2E). A similar repressive effect of HBx on ERα-mediated transcription was also observed in SMMC-7721 cells (data not shown). As another control of the effects of HBx on transcription, HBx stimulated Smad-mediated gene transcription as reported previously (38), when HepG2 cells were co-transfected with the synthetic TGFβ-responsive transcriptional reporter p3TP-Lux (data not shown). Taken together, our data suggest that specific cis- and trans-acting elements are required for the HBx-mediated repression.
Additive repression of specific ERα responsive gene transcription by HBx and ERΔ5
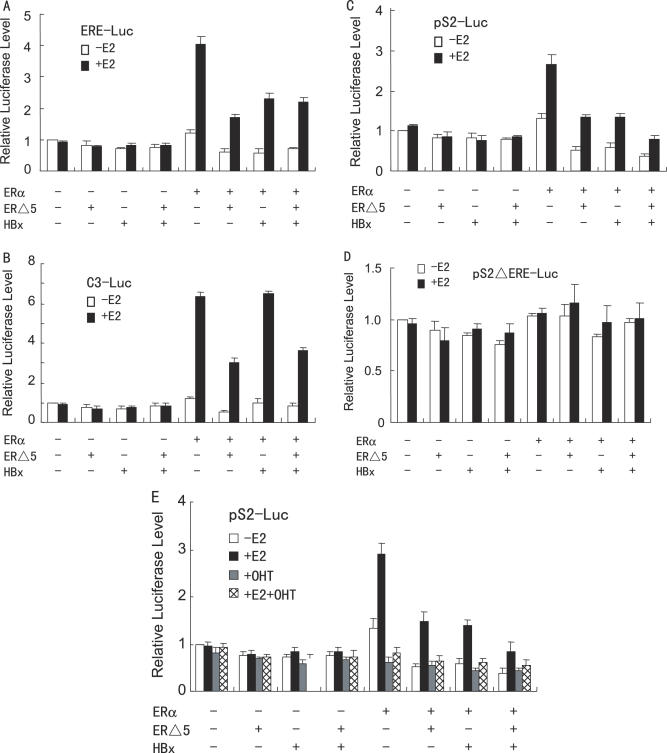
Since both HBx and ERΔ5 repressed ERα transcriptional activity, we determined if HBx and ERΔ5 had synergistic or additive effect on ERα transactivation. We co-transfected HepG2 cells with the ERE-Luc, C3-Luc, pS2-Luc or pS2ΔERE-Luc reporter construct, together with HBx or ERΔ5 or in combination. As expected, HBx alone inhibited the transcription of the ERE-Luc and pS2-LUC reporter genes but not the C3-Luc and pS2ΔERE-Luc reporter genes. ERΔ5 alone repressed the transcription of all of reporter genes except pS2ΔERE-Luc (Figure 3A–D). Cotransfection with HBx plus ERΔ5 expression vectors gave an additive effect in repressing the transcription of the pS2-LUC reporter gene but not the other reporter genes. These results indicate that the additive effect of HBx and ERΔ5 on ERα-responsive gene transcription is promoter specific.
Figure 3.
HBx and ERΔ5 have additive effect on repression of specific ERα responsive gene transcription. (A–D) HepG2 cells were co-transfected with 50 ng of the expression plasmid for ERα, 1.0 µg of the expression plasmid for FLAG-tagged HBx, 50 ng of the expression plasmid for ERΔ5, and 0.2 µg of ERE-Luc (A), C3-Luc (B), pS2-Luc (C) or pS2ΔERE-Luc (D), in the absence or presence of 10 nM E2. The luciferase activity obtained on transfection of the respective luciferase reporter without exogenous ERα, ERΔ5 and HBx in the absence of E2 was set as 1. (E) HepG2 cells were co-transfected with 0.2 µg of pS2-Luc, 50 ng of the expression plasmid for ERα, 1.0 µg of the expression plasmid for FLAG-tagged HBx and 50 ng of the expression vector for ERΔ5. Cells were then treated with control (0.1% ethanol) vehicle, 10 nM E2, 100 nM 4-hydroxytamoxifen (4-OHT) or 10 nM E2 plus 100 nM 4-OHT. The luciferase activity obtained on transfection of pS2-Luc without exogenous ERα, ERΔ5 and HBx in the absence of E2 was set as 1.
To examine the effect of antiestrogen on suppression of ERα transactivation by HBx and ERΔ5, HepG2 cells were co-transfected with the pS2-Luc reporter, ERα, and HBx or ERΔ5 or in combination, and subsequently treated with the antiestrogen 4-OHT (Figure 3E). 4-OHT alone did not have significant effect on HBx- or ERΔ5-mediated repression, whereas combination of 17β-estradiol (E2) and 4-OHT inhibited E2-induced ERα transactivation regardless of HBx and ERΔ5.
Interaction of HBx with ERα in vitro and in vivo
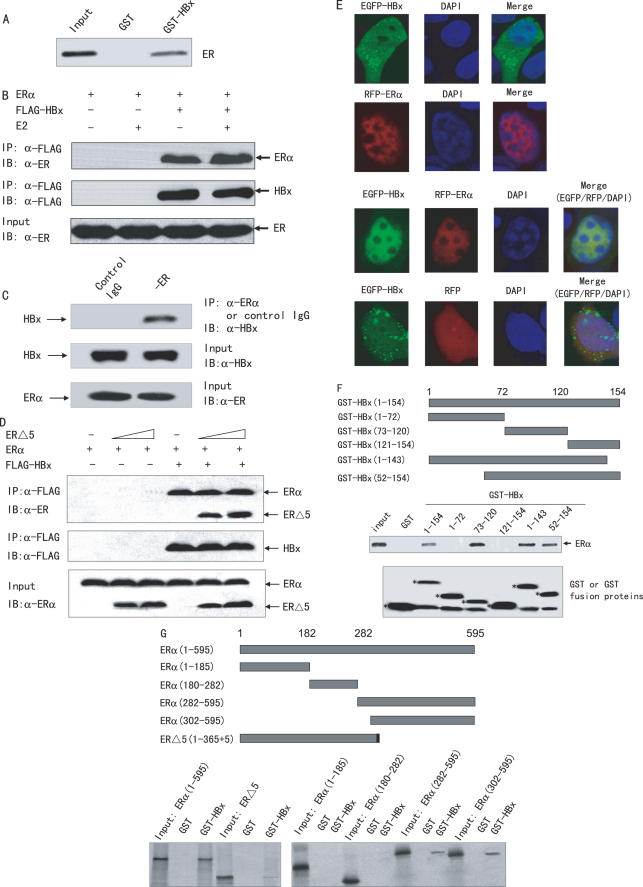
HBx has been shown to regulate viral and cellular gene transcription by interacting with transcription factors (3–6). Our observation that HBx could function as a co-repressor to repress ERα transactivation raised the possibility that HBx might physically interact with ERα. To test this possibility, GST pull-down experiments were performed using 35S-labeled full-length ERα and GST-tagged full-length HBx. As shown in Figure 4A, GST-HBx, but not GST, was able to pull down the 35S-labeled ERα, thus demonstrating an in vitro interaction between HBx and ERα.
Figure 4.
HBx interacts with ERα in vitro and in vivo. (A) Interaction of HBx with ERα in vitro. A GST pull-down assay was performed using 35S-labeled ERα, and GST or GST-HBx. The bound proteins were subjected to SDS–PAGE followed by autoradiography. (B) Interaction of HBx with ERα in vivo. ERα and FLAG-tagged HBx or empty vector were co-transfected into HepG2 cells. Cell lysates were immunoprecipitated (IP) by anti-FLAG M2 monoclonal antibody (Sigma), and the precipitates were then immunoblotted (IB) with anti-ERα polyclonal antibody (Santa Cruz Biotech). (C) Interaction of endogenous HBx with ERα in vivo. Liver tissue extracts from an HBV positive patient were immunoprecipitated with either anti-ERα polyclonal antibody or preimmune control serum (Santa Cruz Biotech). The precipitates were analyzed by immunoblot using anti-HBx (Chemicon). (D) Effect of ERΔ5 on the interaction between HBx and ERα. HepG2 cells were co-transfected with 2 µg ERα, 4 µg FLAG-tagged HBx and increasing amounts of ERΔ5 (2 and 4 µg). Cell lysates were immunoprecipitated by anti-FLAG monoclonal antibody, and the precipitates were detected with anti-ERα polyclonal antibody. (E) Co-localization of HBx and ERα in HepG2 cells. Cells were transfected with EGFP-tagged HBx and RFP-tagged ERα or empty vector (RFP) as indicated, and were treated with 10 nM E2 for 24 h. The images were captured by confocal immunofluorescence microscopy. HBx localization is shown with EGFP (green) and ERα is seen with RFP (red). The nuclei were stained with DAPI (blue). Co-localization of HBx with ERα is shown in merged images. (F) Mapping of the ERα interaction region in HBx. A GST pull-down assay was performed using 35S-labeled ERα and GST-HBx(1-72), GST-HBx(73-120), GST-HBx(121-154), GST-HBx(1-143), GST-HBx(52-154) and full-length GST-HBx(1–154) or GST. Schematic diagram of the HBx deletion constructs used is shown at the top, the binding of ERα to different regions of HBx is demonstrated in the middle, and SDS–PAGE analysis of the purified GST-fusion proteins is shown at the bottom. Asterisks indicate the positions of the expected purified GST or GST-fusion proteins. (G) Mapping of the HBx interaction region in ERα. A GST pull-down assay was performed using full-length GST-HBx(1–154) or GST, and 35S-labeled full-length ERα (1–595), ERα (1–185), ERα (180–282), ERα (282–595), ERα (302–595) or ERΔ5. Schematic diagram of the ERα deletion constructs used is shown at the top.
To test if HBx binds to ERα in mammalian cells, HepG2 cells were transfected with ERα and FLAG-tagged HBx, and harvested for coimmunoprecipitation experiments. Figure 4B demonstrates that ERα could be co-immunoprecipitated in a ligand-independent manner in the presence of FLAG-HBx but not FLAG-tagged empty vector. To ascertain the HBx–ERα interaction in a more physiological context, the endogenous ERα protein from liver tissue of an HBV positive patient was immunoprecipitated with an anti-ERα antibody. Subsequent immunoblotting with anti-HBx antibody indicated that the endogenous HBx was coprecipitated with ERα (Figure 4C). In the negative control experiment, normal rabbit serum or an irrelevant antibody, anti-FLAG antibody, did not immunoprecipitate HBx (Figure 4C and data not shown). Taken together, these data strongly suggest that HBx interacts with ERα in vivo.
Since HBx and ERΔ5 have additive effect on the ERα transactivation (Figure 3C), the effect of ERΔ5 on the HBx–ERα interaction was investigated. HepG2 cells were transfected with FLAG-tagged HBx, ERα, and increasing amounts of ERΔ5, and collected for coimmunoprecipitation assays. As shown in Figure 4D, Both ERα and ERΔ5 were coprecipitated with FLAG-tagged HBx, but not FLAG control vector. Consistent with the functional results (Figure 3C), ERΔ5 had little effect on the interaction of ERα with HBx (Figure 4D).
Co-localization of ERα with HBx
To confirm the protein–protein interaction between HBx and ERα in situ, constructs were made for EGFP-tagged HBx (EGFP-HBx) and RFP-tagged ERα. Based on their ability to regulate the ERE-Luc reporter activity, these fluorescent protein-tagged constructs were similar to those with or without the above mentioned FLAG tag (data not shown). HepG2 cells were then co-transfected with EGFP-HBx and RFP-ERα, and analyzed for co-localization of ERα with HBx. As expected, EGFP-HBx localizes in both the cytoplasm and the nucleus of HepG2 cells (39) (Figure 4E). RFP-ERα localizes essentially in the nucleus of HepG2 cells in both the presence and absence of estrogen (Figure 4E and data not shown). Co-localization studies indicated that EGFP-HBx colocalized with RFP-ERα, but not with the empty vector RFP, predominantly in the cell nucleus in a ligand-independent manner, suggesting that ERα may facilitate the nuclear localization of HBx (Figure 4E and data not shown).
Mapping of the ERα and HBx interaction regions
To define the interacting region(s) of HBx on ERα, GST-fusion proteins containing various regions of HBx were prepared and the ability of each of these to interact with 35S-methionine-labeled in vitro translated full-length ERα were determined by GST pull-down assay (Figure 4F). Deletion of only the first 51 or last 11 amino acids of HBx did not affect the ability to interact with ERα. The GST-HBx(73–120) containing part of the transactivation domain bound specifically to ERα, but the GST-HBx(1–72) and the GST-HBx(121–154) did not.
To map the domain of ERα responsible for interaction with HBx, a series of 35S-methionine-labeled in vitro translated ERα mutants were used in GST pull-down experiments (Figure 4G). The ERα(282–595) and the ERα(302–595) containing the AF2 domain were found to associate with HBx, whereas the ERα(1–185) containing the AF1 and the ERα(180–282) containing the DBD did not. ERΔ5, which has amino acid residues 1–365 of ERα also interacted with HBx, although with weak binding affinity.
Interaction of HBx with ERα is required for inhibition of ERα transactivation function
To test the possibility that the interaction of HBx with ERα is required for the repression of ERα transactivation function, the HBx mutant [HBx(Δ73–120)] in which the interaction region from amino acids 73 to 120 of HBx was deleted was constructed. HepG2 cells were co-transfected with the ERE-LUC reporter, ERα, and FLAG-tagged full-length HBx or HBx(Δ73–120). As shown in Figure 5A, the mutation lacking the ERα-binding site abrogated the repression of ERα transactivation function by HBx. Notably, FLAG-tagged HBx and HBx(Δ73–120) were expressed at comparable levels (Figure 5B). To determine if HBx(Δ73–120) did lose the ability to interact with ERα in HepG2 cells, coimmunoprecipitation experiments were performed. As expected, HBx(Δ73–120) did not interact with ERα (Figure 5C). Taken together, these findings suggest that interaction of HBx with ERα is required for repression of ERα transactivation function.
Figure 5.
The HBx deletion mutant abolishes HBx-induced repression of ERα transcriptional activity. (A) Luciferase reporter assays with the HBx deletion mutants. HepG2 cells were co-transfected with 0.2 µg of ERE-LUC, 50 ng of the expression plasmid for ERα and 1.0 µg of the expression vector for FLAG-tagged HBx or HBx(Δ73–120), in the presence or absence of 10 nM E2. (B) Western blotting showing expression of FLAG-tagged HBx and HBx(Δ73–120). Cells were transfected as in (A). Cell extracts were prepared from E2-treated cells, and equivalent amounts of each extract were detected with anti-FLAG or anti-GAPDH antibody. (C) The HBx deletion mutant abolishes the HBx–ERα interaction. HepG2 cells were co-transfected with the expression plasmid for ERα and the expression vector for FLAG-tagged HBx or HBx(Δ73–120). Cell lysates were immunoprecipitated by anti-FLAG, and the precipitates were probed with anti-ERα.
HBx, ERα and HDAC1 form a complex
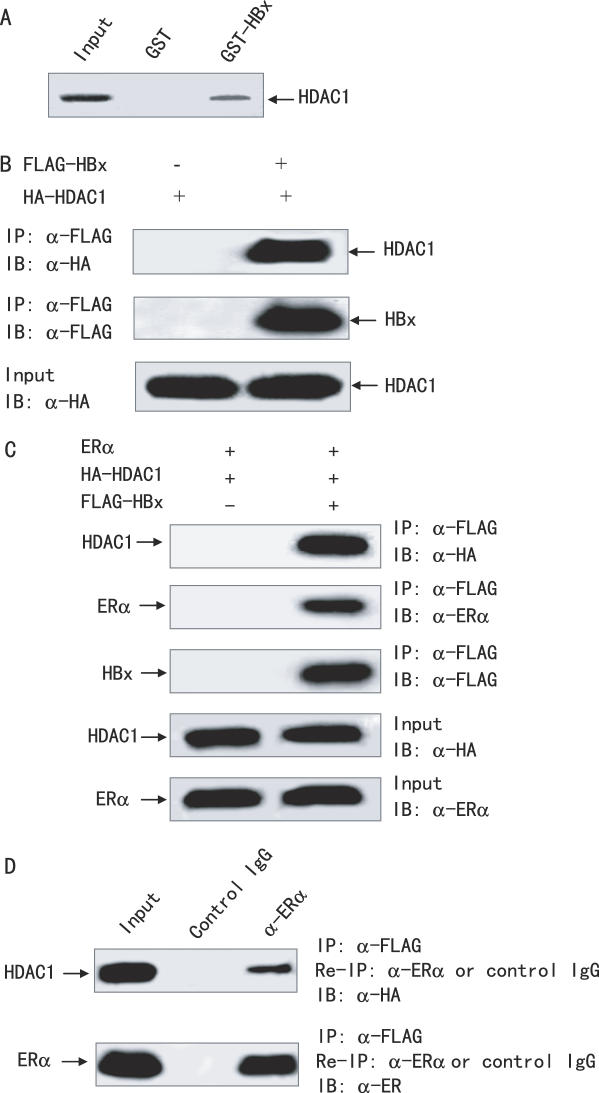
To investigate whether the observed repression of ERα-responsive gene transcription by HBx was associated with recruitment of HDAC complexes, we examined the interaction between HBx and HDAC1 by GST pull-down assay. As shown in Figure 6A, in vitro translated HDAC1 interacted with GST-HBx, but not with GST alone, indicating that HBx associated with HDAC1 in vitro.
Figure 6.
HBx forms a complex with ERα and HDAC1. (A) Association of HBx with HDAC1 in vitro. GST-HBx and GST were incubated with 35S-labeled ERα, and a GST pull-down assay was then performed. (B) Association of HBx with ERα in vivo. HepG2 cells were transiently transfected with HA-tagged HDAC1 and FLAG-tagged HBx or control vector. Immunoprecipitation (IP) was performed using anti-FLAG monoclonal antibody; immunoblotting (IB) was performed with the indicated antibodies. (C) HBx interacts with both HDAC1 and ERα in vivo. HepG2 cells were co-transfected with ERα, HA-tagged HDAC1, and FLAG-tagged HBx or control vector. The cell extracts were immunoprecipitated with anti-FLAG monoclonal antibody followed by immunoblotting with the indicated antibodies. (D) HBx, ERα and HDAC1 forms a ternary complex. HepG2 cells were transfected as in (C). The cell extracts were immunoprecipitated with anti-FLAG antibody. Immune complexes were eluted with FLAG peptide and re-immunoprecipitated (re-IP) using anti-ERα polyclonal antibody and normal rabbit serum as a negative control. The resulting precipitates were resolved by SDS–PAGE followed by immunoblotting with the indicated antibodies.
To determine whether the interaction of HBx with HDAC1 occurred in vivo, we performed coimmunoprecipitation and immunoblotting (Figure 6B). Transient expression of FLAG-tagged HBx, but not control FLAG vector, in HepG2 cells was accompanied by interaction with HA-tagged HDAC1. These results suggest that HBx interacts with HDAC1 in vivo.
To examine whether HBx, ERα and HDAC1 formed a complex, HepG2 cells were transfected with ERα and HA-tagged HDAC1 with or without FLAG-tagged HBx. The cells were then subjected to immunoprecipitation with FLAG antibody-conjugated agarose beads, followed by immunoblot with ERα and HA antibodies (Figure 6C). Both ERα and HDAC1 were coprecipitated with FLAG-tagged HBx, but not FLAG control vector. To further confirm the possibility of a ternary complex among HBx, ERα, and HDAC1, the immune complexes precipitated with FLAG antibody were eluted with a FLAG peptide, and subjected to a second immunoprecipitation with an anti-ERα antibody. The anti-ERα immunoprecipitates were then subjected to immunoblotting with anti-HA to detect HA-tagged HDAC1. HA-tagged HDAC1 was present after sequential immunoprecipitation (Figure 6D). In contrast, HA-tagged HDAC1 was absent after a second immunoprecipitation with control antibody. These data provide strong evidence that HBx, ERα and HDAC1 together can form a ternary complex in vivo.
HDAC inhibitor relieves repression of ERα transactivation function by HBx
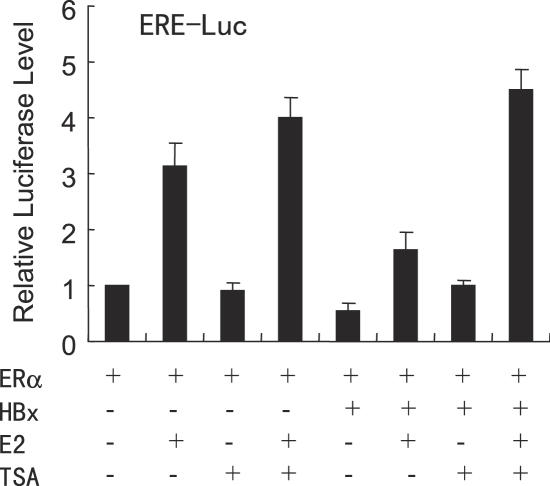
Our observation that HBx interacts with HDAC1 raises the possibility that the repression of ERα functions by HBx could be HDAC dependent. To this end, we examined the effect of TSA, a specific inhibitor of HDAC enzyme, on HBx-induced repression of ERE transcription in HepG2 cells (Figure 7). We found that HBx-mediated repression of ERα transcriptional activity could be effectively relieved by inhibiting HDAC activity. Interestingly, E2 and TSA have additive or synergistic effect in stimulating ERα transcriptional activity. These data suggest that HBx may recruit HDAC enzyme to repress ERE-mediated transcription.
Figure 7.
Treatment of hepatoma cells with the specific HDAC inhibitor TSA causes a drastic relieving of HBx-induced repression of ERα transactivation. HepG2 cells co-transfected with 0.2 µg of the ERE-Luc reporter, 50 ng of the expression vector for ERα, 1.0 µg of the expression plasmid for HBx. Cells were then treated with control (0.1% ethanol) vehicle, 10 nM E2 or 100 nM TSA as indicated. The luciferase activity obtained on transfection of ERE-Luc and ERα without exogenous HBx in the absence of E2 and TSA was set as 1.
DISCUSSION
In the present study, we demonstrated for the first time that both ERΔ5 and HBx can inhibit ERα transcriptional activity in human liver cancer cells. We found that the repression of ERα transactivation function by ERΔ5 and HBx is mediated by their physical interaction with ERα. The binding of HBx with ERα is important for HBx-induced repression of ERα transactivation function because the HBx deletion mutant that lacks ERα-binding site completely abolished the repression of ERα transcriptional activity by HBx. Furthermore, we have shown that ERΔ5 and HBx have additive but not synergistic effect on repression of ERα-responsive gene transcription, suggesting that ERΔ5, ERα and HBx may not form a complex.
Estrogen has been reported to promote the growth of certain human neoplasms, notably tumors of the breast, endometrium and pituitary (40,41). In sharp contrast, Estrogen was shown to suppress the replication of HBV that has been clearly recognized as a major etiological factor for HCC (13). Studies of chemical carcinogenesis also suggested that ERα may modulate HCC risk by inhibiting the malignant transformation of pre-neoplastic liver cells. The ERα variant ERΔ5 was shown to be preferentially expressed in patients with HCC compared with parents with normal livers and to be associated with poor clinical outcome (25–28). Therefore, it is important to determine whether the ERΔ5 is able to interfere with the transcriptional activity of wt ERα. When ERΔ5 was expressed alone in human ERα-negative hepatoma HepG2 cells, it had little effect on either basal or E2-mediated ERE-containing reporter activity. However, when ERΔ5 was coexpressed with wt ERα, the reporter activity was significantly decreased, similar to that in human breast cancer cells (42). Our data suggest that ERΔ5 functions as a dominant negative receptor in human liver cancer cells. Although both in breast and liver cancer cells, ERΔ5 displays dominant negative activity, ERΔ5 was found to act as a dominant positive receptor isoform and facilitate both basal and E2-stimulated ERE-mediated transcription of wt ERα when coexpressed in ERα-negative human osteosarcoma U2-OS and human endometrial cancer Ishikawa cells (43,44). These discordant results could be due to the different cell types used, suggesting that some factors required for ERα transcriptional activity may be tissue specific.
A number of studies have shown that HBx interacts with proteins involved in transcriptional regulation (3–5,45). Most of the studies identify HBx as a co-activator of transcription. For example, HBx enhances the transcription efficacy of the CREB transcription factor through interaction with CREB (46). HBx associates with hypoxia inducible factor-1α (HIF-1α), a major transcriptional factor that regulates expression of angiogenic factors such as vascular endothelial growth factor (VEGF), and enhances the transactivation function of HIF-1α (47,48). HBx was also shown to stimulate transcription by activating cellular signal transduction pathways. For instance, HBx is involved in activating Wnt/β-catenin signaling by stabilizing cytoplasmic β-catenin. HBx can stimulate activator protein 1 (AP-1) via two distinct pathways, the Ras-Raf-mitogen-activated protein kinase (MAPK) and the c-Jun amino-terminal kinase (JNK) cascades (49). On the other hand, HBx was found to act as a co-repressor of transcription. HBx interferes with p53 by direct binding and by sequestering p53 in the cytoplasm, resulting in the abrogation of p53-mediated transcriptional activity (6). HBx binds to DBD of peroxisome proliferator-activated receptor γ (PPARγ), a member of the steroid hormone receptor superfamily, and suppresses PPARγ-mediated transactivation (50). Our results showed that HBx represses ERα transcriptional activity through interaction with ERα and recruitment of HDAC1, which belongs to a class of enzymes involved in deacetylation of hyperacetylated histone tails, leading to compaction of chromatin and transcriptional repression (51). Importantly, the inhibitory effect of HBx on ERα transcriptional activity was antagonized by the HDAC inhibitor TSA. Interestingly, E2 and TSA are additive or synergistic in inducing ERα transcriptional activity. The fact that HBx represses ERα transcriptional activity in the presence or absence of E2 suggests that HBx regulates ERα transcriptional activity in a ligand-independent manner. Recently, the tumor suppressor BRCA1 has been shown to mediate ligand-independent transcriptional repression of ERα in a manner dependent on HDAC activity (52). Our observation that HBx, ERα and HDAC1 can form a complex and TSA can effectively reverse ligand-independent repression mediated by HBx suggests that one of the underlying mechanisms by which HBx mediates ligand-independent repression of ERα transcriptional activity may involve targeted recruitment by unliganded, promoter-bound ERα of a HBx-associated HDAC activity.
Tamoxifen is considered to be relatively more estrogenic than antiestrogenic in the urine, bone and liver tissues (37). Thus, tamoxifen has been used for the treatment of liver cancer. Indeed, initial studies with a relatively small population of patients with HCC show regression of liver tumor mass and improved survival in some of the tamoxifen-treated patients (53). However, more and more recent controlled trials with this drug were disappointing (54–56). Tamoxifen does not prolong survival in patients with HCC and has an increasingly negative impact with increasing dose. There is also no appreciable advantage to quality of life with tamoxifen. These studies showed conclusively that, although the mechanisms by which tamoxifen negatively impacts HCC are not known, tamoxifen does not benefit patients with HCC and is likely to be detrimental. Thus, the use of tamoxifen in patients with HCC is not recommended. Our study indicated that 4-OHT, a metabolite of the tamoxifen with a more potent estrogen agonist/antagonist activity than tamoxifen, acts as a pure estrogen antagonist in HepG2 cells, which is in agreement with the previous study showing that tamoxifen functions as a pure estrogen antagonist in HepG2 cells (37). This may at least in part explain why tamoxifen is ineffective in the treatment of HCC.
Recently, HDAC inhibitors have been used successfully to inhibit cancer cell growth in vitro and in vivo (57–59). TSA specifically inhibits classes I and II HDACs by binding directly to their catalytic site (3). Class I HDACs include HDAC1, HDAC2, HDAC3, HDAC8 and HDAC11, and Class II HDACs include HDACs 4–7 and HDACs 9–10. TSA regulates the expression of small subsets of growth-related genes and has potent antitumor activity in vitro and in vivo. In hepatoma cells, TSA induces a G2/M cell cycle arrest followed by apoptosis (60,61). Since E2 and TSA are additive or synergistic in inducing ERα transcriptional activity in hepatoma cells, it is important to develop more effective therapeutic agents for HCC that increase ERα transactivation function, with no obvious side effects.
Acknowledgments
We thank Xuemin Zhang for technical assistance. This work was supported by the Key Technologies R&D Program (2002BA711A02-5) and National Natural Science Foundation (30571655, 30530320 and 30428012). Funding to pay the Open Access publication charges for this article was provided by the Key Technologies R&D Program.
Conflict of interest statement. None declared.
REFERENCES
- 1.Lee W.M. Hepatitis B virus infection. N. Engl. J. Med. 1997;337:1733–1745. doi: 10.1056/NEJM199712113372406. [DOI] [PubMed] [Google Scholar]
- 2.Murakami S. Hepatitis B virus X protein: a multifunctional viral regulator. J. Gastroenterol. 2001;6:651–660. doi: 10.1007/s005350170027. [DOI] [PubMed] [Google Scholar]
- 3.Cheong J.H., Yi M., Lin Y., Murakami S. Human RPB5, a subunit shared by eukaryotic nuclear RNA polymerases, binds human hepatitis B virus X protein and may play a role in X transactivation. EMBO J. 1995;14:143–150. doi: 10.1002/j.1460-2075.1995.tb06984.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Qadri I., Maguire H.F., Siddiqui A. Hepatitis B virus transactivator protein X interacts with the TATA-binding protein. Proc. Natl Acad. Sci. USA. 1995;92:1003–1007. doi: 10.1073/pnas.92.4.1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Perini G., Oetjen E., Green M.R. The hepatitis B pX protein promotes dimerization and DNA binding of cellular basic region/leucine zipper proteins by targeting the conserved basic region. J. Biol. Chem. 1999;274:13970–13977. doi: 10.1074/jbc.274.20.13970. [DOI] [PubMed] [Google Scholar]
- 6.Wang X.W., Forrester K., Yeh H., Feitelson M.A., Gu J.R., Harris C.C. Hepatitis B virus X protein inhibits p53 sequence-specific DNA binding, transcriptional activity, and association with transcription factor ERCC3. Proc. Natl Acad. Sci. USA. 1994;91:2230–2234. doi: 10.1073/pnas.91.6.2230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Takada S., Kido H., Fukutomi A., Mori T., Koike K. Interaction of hepatitis B virus X protein with a serine protease, tryptase TL2 as an inhibitor. Oncogene. 1994;9:341–348. [PubMed] [Google Scholar]
- 8.Lee T.H., Elledge S.J., Butel J.S. Hepatitis B virus X protein interacts with a probable cellular DNA repair protein. J. Virol. 1995;69:1107–1114. doi: 10.1128/jvi.69.2.1107-1114.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brechot C., Jaffredo F., Lagorce D., Gerken G., Meyer zum Buschenfelde K., Papakonstontinou A., Hadziyannis S., Romeo R., Colombo M., Rodes J., et al. Impact of HBV, HCV and GBV-C/HGV on hepatocellular carcinomas in Europe: results of a European concerted action. J. Hepatol. 1998;29:173–183. doi: 10.1016/s0168-8278(98)80001-9. [DOI] [PubMed] [Google Scholar]
- 10.Kew M.C. Increasing evidence that hepatitis B virus X gene protein and p53 protein may interact in the pathogenesis of hepatocellular carcinoma. Hepatology. 1997;25:1037–1038. doi: 10.1002/hep.510250442. [DOI] [PubMed] [Google Scholar]
- 11.Murakami S. Hepatitis B virus X protein: structure, function and biology. Intervirology. 1999;42:81–99. doi: 10.1159/000024969. [DOI] [PubMed] [Google Scholar]
- 12.Yeh C.T. Hepatitis B virus X protein: searching for a role in hepatocarcinogenesis. J. Gastroenterol. Hepatol. 2000;15:339–341. doi: 10.1046/j.1440-1746.2000.02166.x. [DOI] [PubMed] [Google Scholar]
- 13.Almog Y., Klein A., Adler R., Laub O., Tur-Kaspa R. Estrogen suppresses hepatitis B virus expression in male athymic mice transplanted with HBV transfected HepG-2 cells. Antiviral Res. 1992;19:285–293. doi: 10.1016/0166-3542(92)90010-3. [DOI] [PubMed] [Google Scholar]
- 14.Chen C.J., Yu M.W., Liaw Y.F. Epidemiological characteristics and risk factors of hepatocellular carcinoma. J. Gastroenterol. Hepatol. 1997;12:S294–308. doi: 10.1111/j.1440-1746.1997.tb00513.x. [DOI] [PubMed] [Google Scholar]
- 15.Yu M.W., Chen C.J. Hepatitis B and C viruses in the development of hepatocellular carcinoma. Crit. Rev. Oncol. Hematol. 1994;17:71–91. doi: 10.1016/1040-8428(94)90020-5. [DOI] [PubMed] [Google Scholar]
- 16.Yu M.C., Tong M.J., Govindarajan S., Henderson B.E. Nonviral risk factors for hepatocellular carcinoma in a low-risk population, the non-Asians of Los Angeles County, California. J. Natl Cancer. Inst. 1991;83:1820–1826. doi: 10.1093/jnci/83.24.1820. [DOI] [PubMed] [Google Scholar]
- 17.De Maria N., Manno M., Villa E. Sex hormones and liver cancer. Mol. Cell. Endocrinol. 2002;193:59–63. doi: 10.1016/s0303-7207(02)00096-5. [DOI] [PubMed] [Google Scholar]
- 18.Kuiper G.G., Enmark E., Pelto-Huikko M., Nilsson S., Gustafsson J.A. Cloning of a novel receptor expressed in rat prostate and ovary. Proc. Natl Acad. Sci.USA. 1996;93:5925–5930. doi: 10.1073/pnas.93.12.5925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kuiper G.G., Carlsson B., Grandien K., Enmark E., Haggblad J., Nilsson S., Gustafsson J.A. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors α and β. Endocrinology. 1997;138:863–870. doi: 10.1210/endo.138.3.4979. [DOI] [PubMed] [Google Scholar]
- 20.Katzenellenbogen B.S., Katzenellenbogen J.A. Estrogen receptor transcription and transactivation: estrogen receptor α and estrogen receptor β: regulation by selective estrogen receptor modulators and importance in breast cancer. Breast Cancer Res. 2000;2:335–344. doi: 10.1186/bcr78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Klinge C.M. Estrogen receptor interaction with co-activators and co-repressors. Steroids. 2000;65:227–251. doi: 10.1016/s0039-128x(99)00107-5. [DOI] [PubMed] [Google Scholar]
- 22.Eisenfeld A.J., Aten R., Weinberger M., Haselbacher G., Halpern K., Krakoff L. Estrogen receptor in the mammalian liver. Science. 1976;191:862–865. doi: 10.1126/science.175442. [DOI] [PubMed] [Google Scholar]
- 23.Porter L.E., Elm M.S., Van Thiel D.H., Dugas M.C., Eagon P.K. Characterization and quantitation of human hepatic estrogen receptor. Gastroenterology. 1983;84:704–712. [PubMed] [Google Scholar]
- 24.Rossini G.P., Baldini G.M., Villa E., Manenti F. Characterization of estrogen receptor from human liver. Gastroenterology. 1989;96:1102–1109. doi: 10.1016/0016-5085(89)91629-6. [DOI] [PubMed] [Google Scholar]
- 25.Villa E., Camellini L., Dugani A., Zucchi F., Grottola A., Merighi A., Buttafoco P., Losi L., Manenti F. Variant estrogen receptor messenger RNA species detected in human primary hepatocellular carcinoma. Cancer Res. 1995;55:498–500. [PubMed] [Google Scholar]
- 26.Villa E., Dugani A., Fantoni E., Camellini L., Buttafoco P., Grottola A., Pompei G., De Santis M., Ferrari A., Manenti F. Type of estrogen receptor determines response to antiestrogen therapy. Cancer Res. 1996;56:3883–3885. [PubMed] [Google Scholar]
- 27.Villa E., Moles A., Ferretti I., Buttafoco P., Grottola A., Del Buono M., De Santis M., Manenti F. Natural history of inoperable hepatocellular carcinoma: estrogen receptors' status in the tumor is the strongest prognostic factor for survival. Hepatology. 2000;32:233–238. doi: 10.1053/jhep.2000.9603. [DOI] [PubMed] [Google Scholar]
- 28.Villa E., Colantoni A., Camma C., Grottola A., Buttafoco P., Gelmini R., Ferretti I., Manenti F. Estrogen receptor classification for hepatocellular carcinoma: comparison with clinical staging systems. J. Clin. Oncol. 2003;21:441–446. doi: 10.1200/JCO.2003.11.051. [DOI] [PubMed] [Google Scholar]
- 29.Ding L., Yan J., Zhu J., Zhong H., Lu Q., Wang Z., Huang C., Ye Q. Ligand-independent activation of estrogen receptor α by XBP-1. Nucleic Acids Res. 2003;31:5266–5274. doi: 10.1093/nar/gkg731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fan J.D., Wagner B.L., McDonnell D.P. Identification of the sequences within the human complement 3 promoter required for estrogen responsiveness provides insight into the mechanism of tamoxifen mixed agonist activity. Mol. Endocrinol. 1996;10:1605–1616. doi: 10.1210/mend.10.12.8961270. [DOI] [PubMed] [Google Scholar]
- 31.Aiyar S.E., Sun J.L., Blair A.L., Moskaluk C.A., Lu Y.Z., Ye Q.N., Yamaguchi Y., Mukherjee A., Ren D.M., Handa H., et al. Attenuation of estrogen receptor α-mediated transcription through estrogen-stimulated recruitment of a negative elongation factor. Genes Dev. 2004;18:2134–2146. doi: 10.1101/gad.1214104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Green S., Chambon P. A superfamily of potentially oncogenic hormone receptors. Nature. 1986;324:615–617. doi: 10.1038/324615a0. [DOI] [PubMed] [Google Scholar]
- 33.Tremblay G.B., Tremblay A., Copeland N.G., Gilbert D.J., Jenkins N.A., Labrie F., Giguere V. Cloning, chromosomal localization, and functional analysis of the murine estrogen receptor β. Mol. Endocrinol. 1997;11:353–365. doi: 10.1210/mend.11.3.9902. [DOI] [PubMed] [Google Scholar]
- 34.Protzer U., Nassal M., Chiang P.W., Kirschfink M., Schaller H. Interferon gene transfer by a hepatitis B virus vector efficiently suppresses wild-type virus infection. Proc. Natl Acad. Sci. USA. 1999;96:10818–10823. doi: 10.1073/pnas.96.19.10818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ye Q., Chung L.W., Cinar B., Li S., Zhau H.E. Identification and characterization of estrogen receptor variants in prostate cancer cell lines. J. Steroid Biochem. Mol. Biol. 2000;75:21–31. doi: 10.1016/s0960-0760(00)00118-7. [DOI] [PubMed] [Google Scholar]
- 36.Ye Q., Hu Y.F., Zhong H., Nye A.C., Belmont A.S., Li R. BRCA1-induced large-scale chromatin unfolding and allele-specific effects of cancer-predisposing mutations. J. Cell Biol. 2001;155:911–921. doi: 10.1083/jcb.200108049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Barkhem T., Andersson-Ross C., Hoglund M., Nilsson S. Characterization of the ‘estrogenicity’ of tamoxifen and raloxifene in HepG2 cells: regulation of gene expression from an ERE controlled reporter vector versus regulation of the endogenous SHBG and PS2 genes. J. Steroid Biochem. Mol. Biol. 1997;62:53–64. doi: 10.1016/s0960-0760(97)00017-4. [DOI] [PubMed] [Google Scholar]
- 38.Lee D.K., Park S.H., Yi Y., Choi S.G., Lee C., Parks W.T., Cho H., de Caestecker M.P., Shaul Y., Roberts A.B., et al. The hepatitis B virus encoded oncoprotein pX amplifies TGF-beta family signaling through direct interaction with Smad4: potential mechanism of hepatitis B virus-induced liver fibrosis. Genes Dev. 2001;15:455–466. doi: 10.1101/gad.856201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bontron S., Lin-Marq N., Strubin M. Hepatitis B virus X protein associated with UV-DDB1 induces cell death in the nucleus and is functionally antagonized by UV-DDB2. J. Biol. Chem. 2002;277:38847–38854. doi: 10.1074/jbc.M205722200. [DOI] [PubMed] [Google Scholar]
- 40.Curtis Hewitt S., Couse J.F., Korach K.S. Estrogen receptor transcription and transactivation: estrogen receptor knockout mice: what their phenotypes reveal about mechanisms of estrogen action. Breast Cancer Res. 2000;2:345–352. doi: 10.1186/bcr79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.McDonnell D.P., Norris J.D. Connections and regulation of the human estrogen receptor. Science. 2002;296:1642–1644. doi: 10.1126/science.1071884. [DOI] [PubMed] [Google Scholar]
- 42.Ohlsson H., Lykkesfeldt A.E., Madsen M.W., Briand P. The estrogen receptor variant lacking exon 5 has dominant negative activity in the human breast epithelial cell line HMT-3522S1. Cancer Res. 1998;58:4264–4268. [PubMed] [Google Scholar]
- 43.Chaidarun S.S., Alexander J.M. A tumor-specific truncated estrogen receptor splice variant enhances estrogen-stimulated gene expression. Mol. Endocrinol. 1998;12:1355–1366. doi: 10.1210/mend.12.9.0170. [DOI] [PubMed] [Google Scholar]
- 44.Bryant W., Snowhite A.E., Rice L.W., Shupnik M.A. The estrogen receptor (ER)α variant Δ5 exhibits dominant positive activity on ER-regulated promoters in endometrial carcinoma cells. Endocrinology. 2005;146:751–759. doi: 10.1210/en.2004-0825. [DOI] [PubMed] [Google Scholar]
- 45.Haviv I., Shamay M., Doitsh G., Shaul Y. Hepatitis B virus pX targets TFIIB in transcription coactivation. Mol. Cell. Biol. 1998;18:1562–1569. doi: 10.1128/mcb.18.3.1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Williams J.S., Andrisani O.M. The hepatitis B virus X protein targets the basic region-leucine zipper domain of CREB. Proc. Natl Acad. Sci. USA. 1995;92:3819–3823. doi: 10.1073/pnas.92.9.3819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yoo Y.G., Oh S.H., Park E.S., Cho H., Lee N., Park H., Kim D.K., Yu D.Y., Seong J.K., Lee M.O. Hepatitis B virus X protein enhances transcriptional activity of hypoxia-inducible factor-1alpha through activation of mitogen-activated protein kinase pathway. J. Biol. Chem. 2003;278:39076–39084. doi: 10.1074/jbc.M305101200. [DOI] [PubMed] [Google Scholar]
- 48.Moon E.J., Jeong C.H., Jeong J.W., Kim K.R., Yu D.Y., Murakami S., Kim C.W., Kim K.W. Hepatitis B virus X protein induces angiogenesis by stabilizing hypoxia-inducible factor-1α. FASEB J. 2004;18:382–384. doi: 10.1096/fj.03-0153fje. [DOI] [PubMed] [Google Scholar]
- 49.Cha M.Y., Kim C.M., Park Y.M., Ryu W.S. Hepatitis B virus X protein is essential for the activation of Wnt/β-catenin signaling in hepatoma cells. Hepatology. 2004;39:1683–1693. doi: 10.1002/hep.20245. [DOI] [PubMed] [Google Scholar]
- 50.Choi Y.H., Kim H.I., Seong J.K., Yu D.Y., Cho H., Lee M.O., Lee J.M., Ahn Y.H., Kim S.J., Park J.H. Hepatitis B virus X protein modulates peroxisome proliferator-activated receptor γ through protein-protein interaction. FEBS Lett. 2004;557:73–80. doi: 10.1016/s0014-5793(03)01449-2. [DOI] [PubMed] [Google Scholar]
- 51.Xue Y., Wong J., Moreno G.T., Young M.K., Cote J., Wang W. NURD, a novel complex with both ATP-dependent chromatin-remodeling and histone deacetylase activities. Mol. Cell. 1998;2:851–861. doi: 10.1016/s1097-2765(00)80299-3. [DOI] [PubMed] [Google Scholar]
- 52.Zheng Lei., Annab L.A., Afshari C.A., Lee W.H., Boyer T.G. BRCA1 mediates ligand-independent transcriptional repression of the estrogen receptor. Proc. Natl Acad. Sci. USA. 2001;98:9587–9592. doi: 10.1073/pnas.171174298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Martinez Cerezo F.J., Tomas A., Donoso L., Enriquez J., Guarner C., Balanzo J., Martinez Nogueras A., Vilardell F. Controlled trial of tamoxifen in patients with advanced hepatocellular carcinoma. J. Hepatol. 1994;20:702–706. doi: 10.1016/s0168-8278(05)80138-2. [DOI] [PubMed] [Google Scholar]
- 54.[No authors listed] Tamoxifen in treatment of hepatocellular carcinoma: a randomised controlled trial. CLIP Group (Cancer of the Liver Italian Programme). Lancet. 1998. pp. 17–20. [PubMed]
- 55.Perrone F., Gallo C., Daniele B., Gaeta G.B., Izzo F., Capuano G., Adinolfi L.E., Mazzanti R., Farinati F., Elba S., et al. Tamoxifen in the treatment of hepatocellular carcinoma: 5-year results of the CLIP-1 multicentre randomised controlled trial. Curr. Pharm. Des. 2002;8:1013–1019. doi: 10.2174/1381612024607063. [DOI] [PubMed] [Google Scholar]
- 56.Chow P.K., Tai B.C., Tan C.K., Machin D., Win K.M., Johnson P.J., Soo K.C. High-dose tamoxifen in the treatment of inoperable hepatocellular carcinoma: a multicenter randomized controlled trial. Hepatology. 2002;36:1221–1226. doi: 10.1053/jhep.2002.36824. [DOI] [PubMed] [Google Scholar]
- 57.Kramer O.H., Gottlicher M., Heinzel T. Histone deacetylase as a therapeutic target. Trends Endocrinol. Metab. 2001;12:294–300. doi: 10.1016/s1043-2760(01)00438-6. [DOI] [PubMed] [Google Scholar]
- 58.Jung M. Inhibitors of histone deacetylase as new anticancer agents. Curr. Med. Chem. 2001;8:1505–1511. doi: 10.2174/0929867013372058. [DOI] [PubMed] [Google Scholar]
- 59.Finnin M.S., Donigian J.R., Cohen A., Richon V.M., Rifkind R.A., Marks P.A., Breslow R., Pavletich N.P. Structures of a histone deacetylase homologue bound to the TSA and SAHA inhibitors. Nature. 1999;401:188–193. doi: 10.1038/43710. [DOI] [PubMed] [Google Scholar]
- 60.Herold C., Ganslmayer M., Ocker M., Hermann M., Geerts A., Hahn E.G., Schuppan D. The histone-deacetylase inhibitor Trichostatin A blocks proliferation and triggers apoptotic programs in hepatoma cells. J. Hepatol. 2002;36:233–240. doi: 10.1016/s0168-8278(01)00257-4. [DOI] [PubMed] [Google Scholar]
- 61.Yamashita Y., Shimada M., Harimoto N., Rikimaru T., Shirabe K., Tanaka S., Sugimachi K. Histone deacetylase inhibitor trichostatin A induces cell-cycle arrest/apoptosis and hepatocyte differentiation in human hepatoma cells. Int. J. Cancer. 2003;103:572–576. doi: 10.1002/ijc.10699. [DOI] [PubMed] [Google Scholar]