Abstract
Although we have witnessed advances in many aspects of cancer research and therapy in recent years, the ability to cure the majority of patients with advanced renal cell carcinoma (RCC) remains elusive. At the same time, it has become increasingly apparent that a better understanding of the genetic alterations and immune dysregulations in RCC will play a key role in finding a treatment. Therefore, clinical trials directed at specific genetic alterations and studies exploiting components of the immune system are being conducted. These studies provide new hope for an improved outlook for patients presenting with advanced RCC. The future prospects of RCC therapy will be, without doubt, built on the foundation of current investigative efforts in gene and immune therapy. This article reviews the current role of immunotherapy and gene therapy in the management of metastatic RCC. Finally, current clinical trials focusing on gene and immune therapies are listed.
Key words: Carcinoma, renal cell; Corrective gene therapy; Cytoreductive therapies; Immunomodulation; Immunotherapy, cancer vaccine
Conventionally, no curative treatment other than surgery exists for renal cell carcinoma (RCC), since RCC is resistant to radiation and chemotherapy. Patients with metastatic RCC have a poor prognosis, with an average survival of only 6 to 12 months from the time of diagnosis1 and with an objective response rate of only 6% with conventional chemotherapy.2 Therefore, the search for effective, novel therapy for patients with advanced RCC is ongoing. RCC has been shown to have immunogenic potential; thus, immunotherapy may be attractive for the treatment of RCC. The initial agents used to augment immune resistance to the tumor were recombinant interleukin-2 (rIL-2) and interferon-α (IFN-α). With the advent of recombinant DNA technology, the ability to produce large quantities of IL-2 has resulted in its widespread use. In a relatively short period, IL-2 has become an FDA-approved treatment for metastatic RCC.
Clinical Aspects of Molecular-Based Therapies
After a decade of experience, it became obvious that IL-2- and interferon-based therapies generate similar overall response rates (in the range of 15% to 20%3) but that, with IL-2 monotherapy, an increased frequency of complete responses and enhanced response duration were noted. It also became apparent that patient performance status is a strong predictor for response to IL-2 therapy and survival. When combination therapies and various dose regimens were compared, the response rate was 17% with high-dose rIL-2 alone, 11% with high-dose rIL-2/rIFN-α, 17% with outpatient subcutaneous rIL-2/rIFN-α, and 16% with outpatient rIL-2/rIFN-α plus 5-FU/rIFN-α.4 However, the high-dose rIL-2 regimen produced a 7% complete response rate, compared with 0%, 4%, and 4%, respectively, with each of the other regimens mentioned above. Median response duration was also much longer with high-dose intravenous rIL-2 alone (53 months), compared with 7 months, 12 months, and 9 months, respectively, with each of the other regimens. However, for rIL-2 therapy, subcutaneous low-dose administration generally resulted in much lower toxicity than high-dose intravenous administration.
Epidemiologic Aspects of Molecular-Based Therapies
The incidence of RCC continues to rise.5 Yet, regardless of the specific immunotherapy regimen used, patients included in immunotherapy clinical trials have had an improved prognosis, compared with those in clinical protocols in which no immunotherapy was given.6 This was reflected in the finding that survival was increased in patients participating in immunotherapy clinical trials, compared with those treated with conventional chemotherapy (for immunotherapy, median survival times for favorable-, intermediate-, and poor-risk patients were 26 months, 12 months, and 6 months, respectively). When these outcomes were compared with equivalent historical series in which regimens with chemotherapy alone were used, the difference in median survival between the most favorable groups of patients with metastases and the most grave ones did not exceed 7 months1; however, with immunotherapy, a 20-month difference in median survival is reported.
Classification and Definition of Terms
There has been a constant increase in molecular-based therapeutic strategies offered in various clinical trials as well as an explosion of different terms and nomenclature. We have formulated a simplified classification method to categorize these therapies (Table 1).
Table 1.
Classification of Molecular-Based Therapies for RCC
| Immunotherapy |
|
| Immune-based gene therapy (cancer vaccines) |
|
| Cytoreductive therapies |
|
| Corrective gene therapy |
|
IL, interleukin; GM-CSF, granulocyte-macrophage colony-stimulating factor; VHL, von Hippel-Lindau; TGF, transforming growth factor.
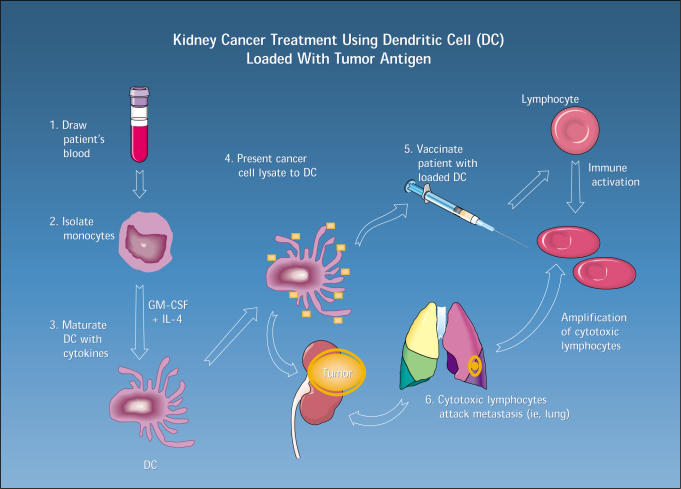
Immunotherapy. It has been hypothesized that tumor cells escape surveillance and destruction by the immune system through down-regulation of cell-surface antigens, such as the major histocompatibility complex7 and other mechanisms causing local inhibition of the immune-competent cells infiltrating the tumor. This led to the development of therapeutic strategies aimed at augmenting antitumor reactivity by either direct systemic administration of cytokines (such as IL-2, interferon, IL-4, granulocyte-macrophage colony-stimulating factor [GM-CSF], and IL-12) or by adoptive (passive) immunotherapy, in which immune lymphoid cells are exposed ex-vivo to cytokines and then rein-fused into the patient. A more recent approach is the use of dendritic cells (DC) that present tumor antigens to T cells alone, as a form of DC vaccine, or in combination with a cytokine, such as IL-2. A clinical trial using a DC vaccine pulsed with autologous tumor lysate is currently ongoing at the University of California, Los Angeles (Figure).
Figure 1.
The UCLA dendritic cell vaccine program. (GM-CSF, granulocyte-macrophage colony-stimulating factor; IL, interleukin.)
Immune-Based Gene Therapy (Cancer Vaccines). In this approach, tumor cells are transfected with cytokine genes or growth factor genes to induce expression of cell-surface proteins, such as HLA class I and class II, that then augment their immunogenicity, resulting in an enhanced immune response against the tumor. This approach carries significant advantage in that it is associated with lower toxicity than is systemic administration of cytokines. Alternatively, the immune activator gene can be delivered via a vector, such as a virus or a liposome (lipid form), or by direct injection of a naked DNA/gene.8
Cytoreductive Therapies. Using this strategy, a gene (suicide gene) is introduced into the tumor. In most cases, the gene encodes for an enzyme capable of converting an otherwise benign medication into a highly cytotoxic one. Administrating the benign medication will result in a high concentration of the cytotoxic agent in the tumor without causing significant systemic toxicity. Herpes-simplex thymidine kinase gene (HSV-tk) is the most commonly used system. HSV-tk phosphorylates ganciclovir to ganciclovir monophosphate, which is converted to ganciclovir triphosphate by cellular kinases. The resulting triphosphate acts as a false base, inhibiting DNA polymerase and DNA synthesis, leading to cell death. Other examples of cytoreductive strategies use virus systems that are capable of replicating and lysing only p53 deficient cells or diphtheria toxin gene, whose product induces necrosis and apoptosis.
Corrective Gene Therapy. Many different genetic alterations have been recognized in urologic tumors. Most of them are distinguished by either the overexpression of an oncogene or the inactivation of a tumor-suppressor gene, such as the p53 gene, by a mutation. The goal of corrective gene therapy is to correct a genetic alteration in the cancer cells and to restore normal growth control. Corrective gene therapy uses the same vectors as immunotherapy and cytoreductive therapy to introduce into the tumor cells a functioning wild-type suppressor gene.
A different approach is the use of antisense mRNA technique, in which small fragments of complementary DNA are bound to the RNA for oncogenes such as bcl-2 or immune inhibitory growth factor genes such as TGFß, thereby inactivating the oncogenic or growth factor gene expression by preventing translation.
RCC Tumorigenesis and Therapeutic Implications
Chromosome 3 Alterations and the VHL Gene. From studies of familial RCC in patients with the von Hippel-Lindau (VHL) syndrome, the tumorigenic pathways of RCC are becoming clearer, enabling investigators to pinpoint targets for future therapies. The loss of chromosome 3p in many sporadic and familial renal cell cancers has been noted.9 The VHL gene was identified at the 3p25.5 position (VHL gene) of chromosome 3.9,10 The wild-type VHL protein functions as a cell-cycle regulator, controlling cellular proliferation by restricting gene transcription, translation, or repair.11 Of all patients with sporadic RCC, 45% to 60% have a detectable mutation in one allele of the VHL gene12; loss in the other allele is seen in up to 98% of tumors.13,14 In fact, not only are allelic alterations of genes located on chromosome 3 associated with RCC, but also gross karyotypic changes of chromosome 3 have been observed, such as chromosomal 3p loss or monosomy.15,16 Moreover, even subtle molecular changes in the form of hypermethylation of the DNA in regulatory areas of the VHL gene are found in up to 20% of sporadic RCC17,18 and cause transcriptional arrest. Accumulating data suggest that other genes located on the 3p region of chromosome 3 may be defective, too, and also may play a role, either alone or in concert with VHL gene alterations, in the tumorigenesis of cases where a VHL gene alteration per se is not documented.19,20 Moreover, aberrations in chromosomes 5, 7, 14, and Y were also found to be associated with RCC. These loci may be able to act independently from the VHL locus, resulting in the development of RCC.21
Corrective Gene Therapy. The VHL gene serves as a potential target for corrective gene therapy. Initial studies have been performed to replace the defective tumor suppressor product in an attempt to reverse the cancer phenotype. Normal (wild-type) VHL gene was transfected into RCC cell lines lacking the normal expression of the gene. The wild-type VHL gene was attached to a constitutively activated cytomegalovirus promoter and put into a liposome vehicle.22 Transfection of the wild-type VHL gene resulted in growth suppression of an RCC cell line. This study showed that the suppression of cell growth was unique to RCC cell lines, which implied that the VHL protein is important in specially controlling the proliferation of kidney cells. Thus, gene replacement therapy using the wild-type VHL gene may have a role in treating patients with RCC, although the safety and efficacy of this treatment is yet to be defined.
P53 Mutation. Abnormal p53 protein has been implicated in a variety of tumor systems. The p53 protein manifests control on cellular proliferation by blocking the binding of DNA polymerase to the DNA strand23,24 in cells with uncorrectable genetic damage. This results in the arrest of cell proliferation at the G1 checkpoint of the cell cycle. If the p53 gene product is absent, cell proliferation will continue in the face of severe DNA damage, resulting in increased genetic instability and tumorigenesis. In RCC, p53 is associated with the aggressive sarcomatoid variant (79%).25 In vitro attempts to correct the p53 gene in RCC cell lines using liposome-p53 gene complexes have resulted in decreased growth of tumor cells in culture. Transfection of the p53 gene into a mouse-xenograft model resulted in a decrease in the number of metastatic lung lesions.26 The use of the p53 wild-type gene by intratumoral injection may prove effective in the future.27
Immune Therapy. Immune suppression has been shown to exist in many tumor systems. The theoretical basis for the immunologic therapies (namely, immunotherapy and cancer vaccine, Table 1) is that improved tumor killing by immune-competent cells may be achieved by maneuvers that augment the immune system. Recently, it was shown that the loss of the VHL gene is associated with increased expression of the immune inhibitory transforming growth factors TGF-α28 and TGF-ß1. This ties together the genetic and the immunologic alterations seen with RCC.
Tumor Vaccine. A novel strategy is to insert cytokine genes ex vivo directly into cancer cells, which are then reinfused into the patient as a vaccine in which the tumor cells act as antigen-presenting cells. Animal studies of this technique have demonstrated prevention of tumor growth, decreased metastatic spread, and prolonged immunologic memory, resulting in rejection of subsequent tumor challenges. The local production of high concentrations of cytokine may directly alter neoplastic properties associated with invasion and metastasis. This treatment strategy is the tumor vaccine addressed in Table 1. At UCLA, we have transfected the genes for IL-2 and IFN-α into human renal cancer cell lines. When these cells are implanted subcutaneously into nude mice, cytokine secretion from these cells prevents tumor growth locally and is more effective than systemic IL-2 and IFN-α administration. Another approach to cancer vaccine therapy is the direct adoptive transfer of ex vivo transfected tumor infiltrating lymphocytes (TILs) with cytokine genes. The rationale is that TILs will home back to tumor deposits with increased specificity and enhanced cytotoxicity induced by the high local cytokine concentration. IL-4, GM-CSF, HLA-137, and interferon-γ gene transfection are other immune system modulators that may have a role in future tumor vaccines for RCC,29,30 using different vectors and delivery systems (ie, a gene gun).31
Recent phase I trials using tumor vaccines have been tried in humans with metastatic RCC. Patients were given irradiated autologous tumor cells transfected in vitro with a retroviral vector carrying the GM-CSF gene. No significant toxicity was reported. One of 16 patients had a partial response.30 Additionally, studies using genetically modified dendritic cells, and studies using the injection of cytokine directly into the tumor, have been performed at UCLA by using the HLA-B7 and IL-2 gene carried in a liposomal vector. Besides these programs, there are at least three other tumor vaccine programs that have been initiated using either intratumoral HLA-B7 or IL-2 gene transfection to enhance the immunogenicity of the tumor. At this time, tumor vaccine-based gene therapy appears to be safe, but its efficacy in metastatic RCC has yet to be proved.
Adoptive Immunotherapy. Historically, adoptive immunotherapy was an attempt to induce cellular therapy by utilizing nonspecific lymphokine-activated killer (LAK) cells that were generated from the patient’s peripheral blood and incubated with IL-2, in vitro, before being reinfused into the patient. A number of clinical trials demonstrated an average response rate of 23.5%. A more specific application is the combination of TILs, which are cytotoxic T-lymphocytes, with immunologic memory to tumoral antigens. At UCLA, TILs are isolated from fresh nephrectomy specimens and are cultured with IL-2 in the presence of tumor extract. After in-vitro expansion with IL-2, TILs are reinfused back into the patient in the hope of directly or indirectly mediating tumor cell destruction without damaging normal cells. On a cellular basis, TILs are 50 to 100 times more potent than LAK cells in mediating tumor regression. Few clinical studies have been undertaken with TILs. The UCLA experience includes a response rate of 33% and an average response duration of 14 months, with a mean survival of 22 months.32
A tumor marker has been introduced recently for RCC. This new tumor antigen has been named G250. The function of this protein is unclear. High levels of G250 antigen can be detected in up to 90% of all kidney cancer cells, with normal renal parenchyma showing no detectable G250 antigen.33 This antigen has been used as a target for monoclonal antibody immunohistochemical staining for diagnostic purposes and has been used also in radionuclide scans to localize tumor sites.34,35 Because this antigen is found in a high proportion of RCCs, it has the potential to be a target for TILs. Recent studies demonstrate that immune activation could be enhanced by the administration of antibodies to G250 in a cytokine-stimulated human RCC xenograft model.36 Further studies are currently being undertaken to elucidate its role as a potential cancer vaccine. A list of clinical trials using molecular-based therapies is presented (Table 2).
Table 2.
Current Clinical Trials Using Molecular- Based Therapies for Advanced RCC
| Basic principle | Principal investigator | Site |
|---|---|---|
| HLA-B7 and IL-2 | Antonia | University of South Florida |
| Intratumoral injection | Belldegrun | UCLA |
| of IL-2 gene | ||
| (Leuvectin, | ||
| phase II) | ||
| Liposome HLA-B7/β2 | Chang | Multicenter |
| microglobulin | ||
| Autologous | Dillman | Multicenter |
| tumor cell | ||
| vaccine + | ||
| IFN/GM-CSF | ||
| (phase II) | ||
| TIL+ INF + IL2 | Belldegrun | UCLA |
| Anti-EGF receptor | Belldegrun | UCLA |
| antibody | ||
| HLA-B7 (Allovectin) | Figlin | UCLA |
| HLA-B7/β2 | Fox | Chiles Research Institute |
| microglobulin | ||
| IL-2 (allogeneic) | Gansbacher | MSKCC |
| Multiantigen loaded | Gitlitz | UCLA |
| dendritic cell vaccine | ||
| (adoptive | ||
| immunotherapy, | ||
| phase I) | ||
| IL-4 | Lotze | University of Pittsburgh |
| TNF-α | Rosenberg | NIH |
| IL-2 | Rosenberg | NIH |
| GM-CSF | Simons | Johns Hopkins |
Adapted with changes from National Cancer Institute Web site: http://cancernet.nci.nih.gov and from Rodrigez R et al. Urologic applications of gene therapy. Urology. 1999;54:401–406. IL, interleukin; TIL, tumor infiltrating lymphocyte; IFN, interferon; EGF, epidermal growth factor; TNF, tumor necrosis factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; UCLA, University of California, Los Angeles; MSKCC, Memorial Cancer Center.
Conclusion
In the past 20 years, there have been impressive advances in the application of immunotherapy to treating patients with RCC. At UCLA, we have seen a progressive increase in responses to treatment as therapy has evolved—from systemic IFN-α administration (16%), to combination IFN + IL-2 (25%), to the current method of bulk TILs (33%) and CD8-/TILs (40%). Patient characteristics that predict improved responsiveness to therapy have been identified, and treatment protocols that decrease toxicity have been developed. The most encouraging results have been the improved rates of complete clinical response, most of which are durable and long-lasting.
There is no doubt that current immunotherapeutic protocols produce changes in the natural history of this disease and cause significant and lasting remissions in select patients. Moreover, the role of surgery in the treatment of advanced RCC is no longer anecdotal. To utilize the advantages of TILs technology, nephrectomy is essential for the preparation of the TILs.
Main Points.
Patient performance status can predict response to interleukin-2 therapy and survival.
A potential target for corrective gene therapy is the von Hippel-Landau (VHL) gene.
Growth suppression of a renal cell carcinoma (RCC) line has been produced by transfection of a wild-type VHL gene.
Loss of the VHL gene is associated with increased expression of immune inhibitory transforming growth factors.
Efficacy of tumor vaccine-based gene therapy remains to be proved.
The tumor antigen G250 is a tumor marker for RCC.
References
- 1.Elson PJ, Witte RS, Trump DL. Prognostic factors for survival in patients with recurrent or metastatic renal cell carcinoma. Cancer Res. 1988;48:7310–7313. [PubMed] [Google Scholar]
- 2.Yagoda A, Abi-Rached B, Petrylak D. Chemotherapy for advanced renal-cell carcinoma: 1983–1993. Semin Oncol. 1995;22:42–60. [PubMed] [Google Scholar]
- 3.Bukowski RM. Natural history and therapy of metastatic renal cell carcinoma: the role of interleukin-2. Cancer. 1997;80:1198–1220. doi: 10.1002/(sici)1097-0142(19971001)80:7<1198::aid-cncr3>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 4.Dutcher JP, Atkins M, Fisher R, et al. Interleukin-2-based therapy for metastatic renal cell cancer: the Cytokine Working Group experience, 1989–1997. Cancer J Sci Am. 1997;3(suppl 1):S73–S78. [PubMed] [Google Scholar]
- 5.Chow WH, Devesa SS, Warren JL, Fraumeni JFJ. Rising incidence of renal cell cancer in the United States. JAMA. 1999;281:1628–1631. doi: 10.1001/jama.281.17.1628. [DOI] [PubMed] [Google Scholar]
- 6.Motzer RJ, Mazumdar M, Bacik J, et al. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol. 1999;17:2530–2540. doi: 10.1200/JCO.1999.17.8.2530. [DOI] [PubMed] [Google Scholar]
- 7.Garrido F, Ruiz-Cabello F. MHC expression on human tumors—its relevance for local tumor growth and metastasis. Semin Cancer Biol. 1991;2:3–10. [PubMed] [Google Scholar]
- 8.Saffran DC, Horton HM, Yankauckas MA, et al. Immunotherapy of established tumors in mice by intratumoral injection of interleukin-2 plasmid DNA: induction of CD8+ T-cell immunity. Cancer Gene Ther. 1998;5:321–330. [PubMed] [Google Scholar]
- 9.Glenn GM, Linehan WM, Hosoe S, et al. Screening for von Hippel-Lindau disease by DNA polymorphism analysis. JAMA. 1992;267:1226–1231. [PubMed] [Google Scholar]
- 10.Linehan WM, Lerman MI, Zbar B. Identification of the von Hippel-Lindau (VHL) gene: its role in renal cancer. JAMA. 1995;273:564–570. [PubMed] [Google Scholar]
- 11.Tory K, Brauch H, Linehan M, et al. Specific genetic change in tumors associated with von Hippel-Lindau disease. J Natl Cancer Inst. 1989;81:1097–1101. doi: 10.1093/jnci/81.14.1097. [DOI] [PubMed] [Google Scholar]
- 12.Shuin T, Kondo K, Torigoe S, et al. Frequent somatic mutations and loss of heterozygosity of the von Hippel-Lindau tumor suppressor gene in primary human renal cell carcinomas. Cancer Res. 1994;54:2852–2855. [PubMed] [Google Scholar]
- 13.Gnarra JR, Tory K, Weng Y, et al. Mutations of the VHL tumour suppressor gene in renal carcinoma. Nat Genet. 1994;7:85–90. doi: 10.1038/ng0594-85. [DOI] [PubMed] [Google Scholar]
- 14.Anglard P, Tory K, Brauch H, et al. Molecular analysis of genetic changes in the origin and development of renal cell carcinoma. Cancer Res. 1991;51:1071–1077. [PubMed] [Google Scholar]
- 15.Kovacs G, Emanuel A, Neumann HP, Kung HF. Cytogenetics of renal cell carcinomas associated with von Hippel-Lindau disease. Genes Chromosom Cancer. 1991;3:256–262. doi: 10.1002/gcc.2870030404. [DOI] [PubMed] [Google Scholar]
- 16.Foster K, Prowse A, van den Berg A, et al. Somatic mutations of the von Hippel-Lindau disease tumour suppressor gene in non-familial clear cell renal carcinoma. Hum Mol Genet. 1994;3:2169–2173. doi: 10.1093/hmg/3.12.2169. [DOI] [PubMed] [Google Scholar]
- 17.Glavac D, Ravnik-Glavac M, Ovcak Z, Masera A. Genetic changes in the origin and development of renal cell carcinoma (RCC) Pflugers Arch. 1996;431:R193–R194. doi: 10.1007/BF02346334. [DOI] [PubMed] [Google Scholar]
- 18.Herman JG, Latif F, Weng Y, et al. Silencing of the VHL tumor-suppressor gene by DNA methylation in renal carcinoma. Proc Natl Acad Sci U S A. 1994;91:9700–9704. doi: 10.1073/pnas.91.21.9700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Foster K, Crossey PA, Cairns P, et al. Molecular genetic investigation of sporadic renal cell carcinoma: analysis of allele loss on chromosomes 3p, 5q, 11p, 17 and 22. Br J Cancer. 1994;69:230–234. doi: 10.1038/bjc.1994.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ohta M, Inoue H, Cotticelli MG, et al. The FHIT gene, spanning the chromosome 3p14.2 fragile site and renal carcinoma-associated t(3;8) breakpoint, is abnormal in digestive tract cancers. Cell. 1996;84:587–597. doi: 10.1016/s0092-8674(00)81034-x. [DOI] [PubMed] [Google Scholar]
- 21.Iqbal MA, Akhtar M, Ali MA. Cytogenetic findings in renal cell carcinoma. Hum Pathol. 1996;27:949–954. doi: 10.1016/s0046-8177(96)90223-3. [DOI] [PubMed] [Google Scholar]
- 22.Chen F, Kishida T, Duh FM, et al. Suppression of growth of renal carcinoma cells by the von Hippel-Lindau tumor suppressor gene. Cancer Res. 1995;55:4804–4807. [PubMed] [Google Scholar]
- 23.Brewster SF, Gingell JC, Brown KW. Tumour suppressor genes in urinary tract oncology. Br J Urol. 1992;70:585–590. [PubMed] [Google Scholar]
- 24.Linehan WM. Molecular genetics of tumor suppressor genes in prostate carcinoma: the challenge and the promise ahead [editorial] J Urol. 1992;147:808–809. doi: 10.1016/s0022-5347(17)37391-3. [DOI] [PubMed] [Google Scholar]
- 25.Oda H, Nakatsuru Y, Ishikawa T. Mutations of the p53 gene and p53 protein overexpression are associated with sarcomatoid transformation in renal cell carcinomas. Cancer Res. 1995;55:658–662. [PubMed] [Google Scholar]
- 26.Moon WC, Kim YS, Moon CS. Experimental p53 gene therapy in renal cell carcinoma. J Urol. 1996;155:653A. [Google Scholar]
- 27.Mahato RI, Kawabata K, Nomura T, et al. Physicochemical and pharmacokinetic characteristics of plasmid DNA/cationic liposome complexes. J Pharm Sci. 1995;84:1267–1271. doi: 10.1002/jps.2600841102. [DOI] [PubMed] [Google Scholar]
- 28.Knebelmann B, Ananth S, Cohen HT, Sukhatme VP. Transforming growth factor alpha is a target for the von Hippel-Lindau tumor suppressor. Cancer Res. 1998;58:226–231. [PubMed] [Google Scholar]
- 29.Simons JW, Jaffee EM, Weber CE, et al. Bioactivity of autologous irradiated renal cell carcinoma vaccines generated by ex vivo granulocyte-macrophage colony-stimulating factor gene transfer. Cancer Res. 1997;57:1537–1546. [PMC free article] [PubMed] [Google Scholar]
- 30.Gitlitz BJ, Belldegrun A, Figlin RA. Immunotherapy and gene therapy. Semin Urol Oncol. 1996;14:237–243. [PubMed] [Google Scholar]
- 31.Seigne J, Turner J, Diaz J, et al. Feasibility study of gene gun mediated immunotherapy for renal cell carcinoma. J Urol. 1999;162:1259–1263. [PubMed] [Google Scholar]
- 32.Pierce WC, Belldegrun A, Figlin RA. Cellular therapy: scientific rationale and clinical results in the treatment of metastatic renal-cell carcinoma. 1995;22:74–80. [PubMed] [Google Scholar]
- 33.Bander NH, Divgi C, Finn R, et al. Renal cancer imaging with monoclonal antibody. J Urol. 1996;155:583A. [Google Scholar]
- 34.Oosterwijk E, Bander NH, Divgi CR, et al. Antibody localization in human renal cell carcinoma: a phase I study of monoclonal antibody G250. J Clin Oncol. 1993;11:738–750. doi: 10.1200/JCO.1993.11.4.738. [DOI] [PubMed] [Google Scholar]
- 35.Anton P, Tanke HJ, Allehoff EP, et al. Localized renal-cell carcinoma: detection of abnormal cells in peritumoral tissue: a cytophotometry and immunocytochemistry study. World J Urol. 1995;13:149–152. doi: 10.1007/BF00184869. [DOI] [PubMed] [Google Scholar]
- 36.van Dijk J, Uemura H, Beniers AJ, et al. Therapeutic effects of monoclonal antibody G250, interferons and tumor necrosis factor, in mice with renal-cell carcinoma xenografts. Int J Cancer. 1994;56:262–268. doi: 10.1002/ijc.2910560220. [DOI] [PubMed] [Google Scholar]