Abstract
Nearly all men will develop histological benign prostatic hyperplasia by the age of 80, but the degree of prostatic enlargement resulting from the hyperplasia is highly variable. Historically, it has often been assumed that the pathophysiology of lower urinary tract symptoms (LUTS) in men is the result of bladder outlet obstruction associated with prostatic enlargement. The observation that prostatic enlargement, bladder outlet obstruction, and LUTS are all age-dependent has been interpreted to indicate that these phenomena were causally related, but there is insufficient evidence for this. Undoubtedly, some men' prostatic enlargement causes obstruction and symptoms. Based upon the available data, however, this subset appears to be extremely small. Because of the many urological and nonurological conditions that cause LUTS and age-dependent changes in bladder and neurological function, it is unlikely that there exists a single dominant etiology for the aging male population. If this is the case, then the optimal management of LUTS will require different and possibly combination therapies.
Key words: Benign prostatic hyperplasia, Lower urinary tract symptoms, Bladder outlet obstruction, Urinary retention, Transurethral prostatic resection
The term benign prostatic hyperplasia (BPH) describes a proliferative process of the cellular elements of the prostate, an enlarged prostate, or the voiding dysfunction resulting from prostatic enlargement and bladder outlet obstruction. Histologically, BPH describes a proliferative process of both the stromal and epithelial elements of the prostate gland. BPH arises in the periurethral and transition zones of the prostate.1 Histological BPH represents an inescapable phenomenon for the aging male population. Approximately 90% of men will develop histologic evidence of BPH by 80 years of age.2 The relative proportion of stromal and epithelial hyperplasia is highly variable in men with clinical BPH (Table 1). In a group of 26 men with BPH, the individual cellular composition of the hyperplastic component of the prostate ranged in connective tissue from 16.1% to 56.1%, in smooth muscle from 20.2% to 59.3%, in epithelium from 4.3% to 24.8%, and in epithelial lumen by 5.3% to 21.9%.
Table 1.
Prostatic Cellular Composition in Men With Clinical BPH
| % Area Density | ||
|---|---|---|
| Tissue Elements | Mean | Range |
| Connective tissue | 38.6 | 16.1–56.1 |
| Smooth muscle | 38.8 | 20.2–59.3 |
| Epithelium | 11.9 | 4.3–24.8 |
| Lumen | 11.3 | 5.3–21.9 |
Although nearly all men develop histological BPH, the degree of prostatic enlargement resulting from hyperplasia is highly variable. The volume of the prostate is most accurately determined using imaging studies such as ultrasound, computerized tomography, and magnetic resonance imaging. Oesterling and colleagues3 measured the prostate volumes of 464 men between 40 and 80 years of age, selected at random from the population of Olmsted County, MN. The overwhelming majority of men had prostate volumes ranging between 20 g and 60 g. A statistically significant correlation existed between age and prostate volume (P = .0001; r2 = .185). Although prostate volume was age-dependent, there was substantial overlap between the 10-year categorical age groups.
The severity of lower urinary tract symptoms (LUTS) is best quantified using quantitative symptom indices. The most widely accepted instrument for quantifying symptom severity is the American Urological Association (AUA) symptom index.4 Results from population-based studies have shown that the prevalence of moderate-to-severe LUTS and reductions in Qmax both increase with patient age.5 Because the development of LUTS and prostatic enlargement are both age-dependent, the development of LUTS in the aging male population has often been attributed to the enlarging prostate or BPH. In fact, up until recently, the constellation of obstructive and irritative symptoms observed in aging men was termed “prostatism.” It is now widely recognized that the differential diagnosis of LUTS in the aging male population includes both urological and neurological conditions. Parkinson's disease, a cerebrovascular accident, diabetes mellitus, congestive heart failure, bladder cancer, prostate cancer, urinary tract infection, overactive bladder, urethral stricture, and bladder neck hypertrophy may all cause LUTS identical to BPH.6 Nevertheless, LUTS in the presence of some degree of prostatic enlargement have been sufficient to establish the clinical diagnosis of BPH.
Pathophysiology of BPH: Historical Perspective
The pathophysiology of bladder outlet obstruction in men with BPH has been attributed to both static and dynamic factors.7 The static obstruction is due to the bulk enlargement of the prostate encroaching upon the prostatic urethra and bladder outlet, whereas the dynamic obstruction is related to the tension of prostate smooth muscle. The medical therapies widely used today for treatment of BPH are targeted to diminishing bladder outlet obstruction in order to reduce prostate volume and relax prostate smooth muscle tension.6 Clinical data demonstrate that androgen suppression and α-blockade relieve and increase urinary flow rates in men with BPH; these data have been used to support the hypothesis that the pathophysiology of “prostatism” is due to bladder outlet obstruction.
Historically, it has often been assumed that the pathophysiology of LUTS in men is the result of bladder outlet obstruction associated with prostatic enlargement.8 The observation that prostatic enlargement, bladder outlet obstruction, and LUTS are all age-dependent was interpreted to indicate that these phenomena were causally related,9 but there is insufficient evidence for this. The relationships between prostate volume, bladder outlet obstruction, and LUTS are optimally defined by measuring these parameters in a group of men selected at random from the community. Girman and colleagues5 measured prostate volume using transrectal ultrasonography, peak flow rate, and the AUA symptom score in 464 men between the ages of 40 and 80 years, selected at random from the residents of Olmsted County, MN. The P and r2 values for the pairwise relationships between prostate volume and peak flow rate, prostate volume and symptom score, and peak flow rate and symptom score are shown in Table 2. These observations demonstrate that the size of the prostate is a very weak determinant of symptom severity and bladder outlet obstruction, and that bladder outlet obstruction is only a minor determinant of symptom severity.
Table 2.
Relationships Between Prostatism, Bladder Outlet Obstruction, and Prostate Enlargement
| Pairwise Comparisons | |||||||
|---|---|---|---|---|---|---|---|
| PV vs PFR | PV vs AUASS | PFR vs AUASS | |||||
| Reference | No. | P | r2 | P | r2 | P | r2 |
| Girman et al5 | 466 | < .001 | .034 | < .001 | 0.45 | < .001 | .123 |
| Barry et al10 | 198 | .06 | .020 | .22 | .008 | .27 | .005 |
PV, prostate volume; PFR, peak flow rate; AUASS, AUA symptom score.
Barry and coworkers10 reported the relationships between prostate volume, peak flow rate, and symptom scores in a cohort of men participating in a clinical trial examining different treatment options for BPH. The pairwise correlations between prostate volume and peak flow rate, prostate volume and symptom score, and peak flow rate and symptom score were not clinically or statistically significant (Table 2). The findings of both Girman5 and Barry10 strongly suggest that the widely held concept of the pathophysiology of LUTS in the aging male population—typically bladder outlet obstruction arising from enlarged prostate—is a gross oversimplification.
One explanation for the poor correlation between bladder outlet obstruction and symptom severity is that peak flow rate is not a reliable proxy for bladder outlet obstruction. Only a weak correlation exists between peak flow rate and synchronous pressure flow studies. Because pressure flow studies are invasive, these measurements have not been performed in a community population. Investigators have failed to show a clinically or statistically significant correlation between the severity of bladder outlet obstruction, based upon detrusor pressures at peak flow rate, and severity of LUTS.11
Studies comparing LUTS, bladder outlet obstruction, and prostate size among different races provide additional evidence that prostate size is not an important factor leading to the development of LUTS and lowered peak flow rate. The AUA symptom score, peak flow rate, and prostate volume were measured in community-based surveys of men in the United States (Olmsted County, MN) and Japan (Shimomaki-nura) (Table 3).12 The Japanese men had smaller prostates, higher peak flow rates, and more severe symptoms. The discordance between symptom scores and prostate volumes, and symptom scores and peak flow rates, in these two population studies, provides further evidence that the severity of LUTS is not explained by bladder outlet obstruction or prostate volume.
Table 3.
International Prostate Symptom Score, Peak Flow Rate, and Prostate Volume in Caucasian (American) and Asian (Japanese) Men
| PER (ML/sec) | |||
|---|---|---|---|
| Race | IPSS | (% ≥8) | PV (cm3) |
| Caucasian | 25.5 | 17.2 | 29.5 |
| Asian | 36.6 | 21.3 | 23.5 |
IPSS, international prostatic symptom score; PFR, peak flow rate; PV, prostate volume.
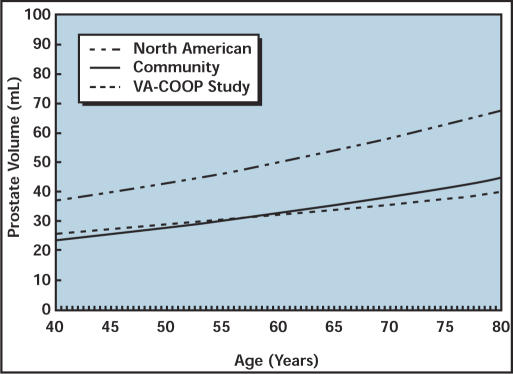
Oesterling and colleagues3 reported the distribution of prostate size in a cohort of randomly selected men living in Olmsted County, MN. Interestingly, the mean prostate volume and the distribution of prostate volumes were almost identical to subjects with both LUTS (AUA symptom score ≥ 8) and bladder outlet obstruction (peak urinary flow rates from 4 to 15 mL/sec) who were enrolled in the Veterans Affairs Cooperative Studies Benign Prostatic Hyperplasia Study (VA-COOP) (Figure 1).13 The fact that a random population of men has the same mean prostate volume and distribution of prostate volume as a group of age-matched men with clinical BPH, provides further evidence that prostate size is relatively unimportant in the development of LUTS and bladder outlet obstruction.
Figure 1.
Estimated median prostate volume as a function of age for the finasteride North American Study, VA Study, and Olmsted County Community Study. Reproduced from Boyle et al,13 with permission from the publisher, Elsever Science.
Additional evidence questioning the influence of prostatic enlargement on the pathophysiology of LUTS in the aging male comes from studies comparing the incidences of symptoms in men and women. Lepor and Machi14 administered the AUA symptom score to a group of 101 males and 99 females between the ages of 55 and 79 years. Subjects were attending a general health symposium with no emphasis on genitourinary diseases. Mean AUA symptom scores were equivalent in men (6.9) and women (7.7). The AUA score was subgrouped into obstructive and irritative scores. The obstructive symptom scores in men (2.7) and women (2.9) were not significantly different. Similarly, the irritative scores in the men (4.2) and women (4.8) were not significantly different. The development of LUTS characteristic of BPH is a non-gender- specific event associated with aging. It is conceivable that the pathophysiology of LUTS in men and women is different. Nevertheless, the observation that the prevalence of LUTS characteristic of clinical BPH is equivalent in men and women suggests that important nonprostatic mechanisms likely exist for the development of symptoms.
One of the life-threatening consequences of BPH is the development of urinary retention. Although prostate volume does not appear to be an important predictor of LUTS severity, there is increasing evidence that the risk of developing urinary retention is related to prostate size.15 Finasteride has been shown to reduce the risk of developing urinary retention but the effect of finasteride on reducing the risk of urinary retention is prostate-size—dependent.16 Reducing prostate volume with 5α-reductase inhibitors only represents a reasonable strategy for decreasing the risk of urinary retention in men with large prostates.
In summary, there is no clinically significant relationship between prostatic enlargement and LUTS in men with clinical BPH. There is only a weak relationship between LUTS and bladder outlet obstruction. Therefore, factors other than prostatic enlargement and bladder outlet obstruction must contribute to the development and severity of LUTS.
Identifying Prostatic Factors Contributing to the Pathophysiology of Clinical BPH
Despite the observation that both men and women develop LUTS, the unequivocal effectiveness of transurethral resection of the prostate (TURP) strongly suggests that the prostate and/or prostatic urethra must play an important role in the pathophysiology of clinical BPH. Schwartz and Lepor have also demonstrated that in men with clinically localized prostate cancer and LUTS, radical prostatectomy has the same beneficial effect on symptoms as does TURP.17 This provides further evidence that the prostate is an important factor in the pathophysiology of LUTS in men.
Caine and coworkers reported in 197618 that α-blockers are effective for the treatment of BPH. Over the last 20 years, over 20 randomized clinical trials have consistently demonstrated the safety and efficacy of various α1-blockers for the treatment of BPH.6 It is indisputable that α1 receptors are abundant in the human prostate,19 and these receptors mediate the tension of prostate smooth muscle.20 It has been assumed that the efficacy of α1-blockers is mediated via relaxation of prostate smooth muscle. The observation by Shapiro and colleagues,21 that 40% of the area density of BPH tissue is smooth muscle, provides further evidence that prostate smooth muscle is likely an important factor in the development of clinical BPH.
The most appropriate study design to elucidate the pathophysiology of clinical BPH would be a comparison of biochemical and histological properties in tissue specimens derived from age-matched men with and without clinical BPH. Ideally, the specimens would be derived from men with prostates of equivalent size. Several investigators have made comparisons between tissue specimens derived from the inner (transition zone) and outer (peripheral zone) regions of the prostate. In the enlarged prostate, the outer and inner zones correspond to the surgical capsule and the hyperplastic tissue, respectively. These comparisons do not provide insight into the pathophysiology of clinical BPH but instead compare regional differences in the prostate.
Between 1986 and 1989, our laboratory investigated the pathophysiology of clinical BPH utilizing inner gland tissue specimens derived from men undergoing TURP for clinical BPH (symptomatic BPH) and men undergoing cystoprostatectomy for bladder cancer (asymptomatic BPH). Preoperatively, symptom scores, peak flow rate, and prostate volumes were routinely measured. α1 Receptor density was observed to be equivalent in tissue specimens obtained from men with symptomatic and asymptomatic BPH (Table 4).20 The contractile response to α1 agonists was also similar between these groups (Table 4).19 These studies suggested that the development of clinical BPH was not due to upregulation of the α1 receptor or increased responsiveness of prostate smooth muscle to α1 agonists.
Table 4.
Comparison of Prostatic α1 Adrenoceptor Density, Phenylephrine Responsiveness, and Cellular Composition in Men With Symptomatic and Asymptomatic BPH
| α1AR Density | Phenylephrine | ||
|---|---|---|---|
| Tissue Source | (fmol/mg wet wt) | (gT/g wet wt) | S/E Ratio Emax |
| TURP | .17 | 11.4 | 5.5 |
| Cystoprostatectomy | .19 | 27 | 2.7 |
α1 AR, α1 adrenoceptor; Emax, maximal tension; S/E, stromal:epithelial; TURP, transurethral resection of prostate.
Although other investigators have reported that the α1 receptor is upregulated in men with BPH, these studies compared tissues from different regions of the prostate and not inner gland tissue from men with and without clinical BPH. In our studies, the stromal:epithelial ratio was greater in men with symptomatic BPH, suggesting that the cellular composition of the inner gland (transition zone) may represent an important factor contributing to the pathophysiology of clinical BPH (Table 4).22
Because the neurotransmitter for the α1 receptor is norepinephrine, another plausible mechanism contributing to the pathophysiology of clinical BPH is increased adrenergic innervation. Spitsbergen and coworkers23 have reported that the frequency of micturition in the spontaneous hypertensive rat (SHR) is greater than in controls, implying that the increased levels of norepinephrine may mediate voiding dysfunction. Lepor and colleagues24 reported an inverse relationship between the AUA symptom score and catecholamine level in consecutive men undergoing prostatic biopsy for an elevated prostate-specifc antigen (PSA) or abnormal digital rectal examination who had no evidence of prostate cancer. This observation strongly suggests that the pathophysiology of clinical BPH is not due to increased adrenergic innervation.
In summary, the studies from our laboratory identified the cellular composition of the prostate as the only parameter contributing to the pathophysiology of clinical BPH.
In order to further define the role of prostate smooth muscle in the pathophysiology of clinical BPH, 26 men with clinical BPH who were candidates for medical management completed the Boyarsky symptom score and underwent uroflowmetry and transrectal ultrasound-guided biopsy of the prostate before initiating therapy with the α1-blocker terazosin.25 The mean percent smooth muscle was quantified from the biopsy specimens. The pairwise relationships between baseline peak flow rate and percent smooth muscle, baseline total symptom score and percent smooth muscle, percent change in peak flow rate and percent smooth muscle, and percent change in the symptom score and percent smooth muscle are shown in Table 5. These pairwise relationships demonstrate a statistically and clinically significant relationship between the baseline peak flow rate and the percent smooth muscle, and no significant relationship between the baseline total symptom score and percent smooth muscle. These observations suggest that the amount of prostate smooth muscle contributes to bladder outlet obstruction and not to symptomatology.
Table 5.
The Role of Prostatic Smooth Muscle in the Pathophysiology of BPH
| Linear Correlation | |||
|---|---|---|---|
| Pairwise Comparisons | P | r | r2 |
| % SM vs baseline AUASS | .59 | .11 | .012 |
| % SM vs baseline PFR | .02 | -.44 | .194 |
| % SM vs ΔAUASS | .19 | -.27 | .073 |
| % SM vs Δ PFR | < .0001 | -.75 | .563 |
SM, smooth muscle; AUASS, AUA symptom score; PFR, peak flow rate.
These observations provide further evidence that LUTS and bladder outlet obstruction are not causally related. The relationship between the increase in peak flow rate and the percent smooth muscle was highly significant, suggesting that the improvement in bladder outlet obstruction secondary to terazosin is related to relaxation of prostatic smooth muscle. A very weak and statistically insignificant relationship was observed between the percent change in the total symptom score and the percent smooth muscle, suggesting that the symptom improvement associated with terazosin is most likely not mediated via relaxation of prostate smooth muscle. An important implication of these findings is that non-prostatic smooth muscle α1-mediated mechanisms may account for the symptom improvement elicited by α1-blockers in men with BPH.
In summary, prostate smooth muscle density contributes to the severity of bladder outlet obstruction and accounts for the α1-mediated reduction of bladder outlet obstruction in men with clinical BPH. Prostate smooth muscle density does not appear to be a major factor contributing to the severity of LUTS or to α1-mediated improvement in symptomatology in men with clinical BPH.
The Pathophysiology of Clinical BPH: Clinical Correlations
If the pathophysiology of LUTS is due to bladder outlet obstruction resulting from enlargement of the prostate, then the improvement in symptom scores in men undergoing treatment for BPH should be directly proportional to the increase in peak flow rates and the decrease in prostate volume. The overwhelming clinical evidence derived from both surgical and medical therapy databases provides evidence suggesting that decreases in symptom scores are not proportional to increases in peak flow rate or reduction of prostate volume.
Lepor and Rigaud26 reported the treatment outcome for 30 men with clinical BPH undergoing TURP. Overall, 87% of the patients experienced marked or moderate improvement in their symptomatology. In this prospective study, the relationship between changes in peak flow rate and changes in obstructive symptom score was not statistically significant (P = .49; r2 = .25). Similarly, changes in peak flow rate versus changes in irritative symptom score were not statistically significantly (P = .9; r2 = .085). Schaeffer has reported that symptom improvement following prostatectomy is equivalent in men with and without pressure flow evidence of bladder outlet obstruction.
An analysis of the VA-COOP study provides insights into the pathophysiology of clinical BPH.27 In this multicenter clinical trial, 1229 men with clinical BPH were randomized in equal proportions to receive placebo, terazosin, finasteride, or combination therapy. This study represented the first comparison of an α1-blocker and a 5α-reductase inhibitor and was the first to examine a combination of both drugs.
Over the entire 52 weeks of the randomized study, changes in peak flow rate and AUA symptom score were not significantly different between placebo and finasteride. The changes in peak flow rate and AUA symptom score between placebo and terazosin were highly statistically significant, whereas this relationship between terazosin and combination therapy was not statistically or clinically significant. The equivalent effectiveness of placebo versus finasteride and terazosin versus combination therapy is compelling evidence that finasteride has an extremely limited role in the medical management of BPH.
A subset analysis of the VA study demonstrated a small difference between the changes in peak flow rates and AUA symptom scores in men receiving finasteride versus placebo in those men with large prostates.28 In men with prostate volumes above 50 cm3, the treatment-related improvement in AUA symptom score attributed to terazosin was threefold greater than with finasteride.
The proposed mechanism of action for the efficacy of finasteride in men with BPH is reduction of prostate volume. Table 6 shows the P and r2 values for the pairwise relationship between changes in prostate volume and changes in peak flow rate, as well as the relationship between changes in prostate volume and changes in AUA symptom score for the 251 subjects randomized to the finasteride group in the VA study.28 The P values for both these pairwise relationships were not statistically significant. The mechanism for the minimal efficacy associated with finasteride is not related to reduction of prostate volume.
Table 6.
Pairwise Comparisons Between Changes in Prostate Volume and Changes in Primary Outcome Measures for Subjects Receiving Finasteride in the VA COOP Study
| Linear Correlation | ||
|---|---|---|
| Pairwise Comparisons | P | r2 |
| ΔPV vs Δ PFR | .097 | -.106 |
| Δ PV vs Δ AUASS | .068 | .116 |
PV, prostate volume; AUASS, AUA symptom score
If the mechanism for the improvement in symptomatology is related to alleviating bladder outlet obstruction, then a significant relationship should exist between changes in AUA symptom score and changes in the peak flow rate. A significant relationship was observed between changes in AUA symptom score and peak flow rate, only in the finasteride and combination groups (Table 7). These r2 values suggest that the relationship between changes in AUA symptom score and peak flow rate is weak for finasteride and that factors other than alleviating bladder outlet obstruction contribute to α1-mediated symptom improvement. There is no reasonable explanation as to why there exists a relationship between changes in AUA symptom scores and changes in peak flow rates for the combination and not the terazosin group.
Table 7.
Pairwise Comparisons in the VA COOP Study Between Changes in AUASS and PFR
| Pairwise Linear Correlation | |||
|---|---|---|---|
| Treatment Groups | Number | P | r2 |
| PLB | 264 | .684 | .025 |
| FIN | 251 | .004 | -.182 |
| TRZ | 274 | .124 | -.093 |
| CMB | 276 | .002 | -.225 |
AUASS, AUA symptom score; PFR, peak flow rate; PLB, placebo; FIN, finasteride; TRZ, terazosin; CMB, drug combination.
If the symptom improvement following terazosin administration is not related to the baseline peak flow rate, then excluding men with LUTS and no evidence of bladder outlet obstruction may not be justified. A subset analysis of the VA study examined the symptom improvement according to baseline peak flow rate quartile groups.28 Interestingly, improvements in the AUA symptom scores were equivalent in the lowest baseline peak flow rate quartile group (peak flow rate < 8.6 mL/sec) and the highest quartile baseline peak flow rate group (peak flow rate > 12.4 mL/sec), suggesting that α1-mediated symptom improvement may not be related to relaxing prostate smooth muscle. If this is the case, men with LUTS and no evidence of bladder outlet obstruction should also respond to α1-blockers.
Lepor and colleagues29 have recently reported that symptom improvements in age-matched men with prostate volumes of equivalent size were equivalent in men with normal and abnormal baseline peak flow rates. The assumption that men with LUTS must also have bladder outlet obstruction has likely limited the clinical utility of α-blockers in men with LUTS. Thus, men with lower urinary tract symptoms secondary to prostatitis, interstitial cystititis, radiation cystititis, and other clinical entities, may also respond to α1-blockers.
In summary, reduction of prostate volume at best accounts for only a small portion of finasteride's minimal effect on LUTS. Alleviation of bladder outlet obstruction does not account for LUTS improvement associated with α-blockers. The available clinical data suggest that symptom improvement elicited by α1-blockade and hormonal therapy may not be mediated primarily by relaxation of prostate smooth muscle and prostate volume reduction, respectively.
LUTS are the most prevalent but not the only clinical manifestation of BPH. The more severe manifestations of BPH include urinary retention, urinary tract infection, and renal insufficiency. There is increasing evidence that men with larger prostates are at greater risk of developing urinary retention.15 A 4-year, randomized, placebo-controlled study (PLESS trial) recently demonstrated that finasteride reduces the probability of developing urinary retention.16 Of the 3040 subjects randomized, only 1883 (62%) were evaluable at 4 years. The overall incidence of urinary retention in the finasteride and placebo groups was 3% and 7%, respectively. The difference between the rates of urinary retention in the finasteride and placebo group was manifested within 1 year of follow-up. Because the mean prostate volume in this study was 51 g, the conclusions are based on men with very large prostates and are not generalizable to men with the diagnosis of clinical BPH.
Whether men with enlarged prostates will take a medication that has minimal effects on symptoms in order to reduce the rate of urinary retention has yet to be determined. The cost-effectiveness of preventing urinary retention will play an important role in the ultimate acceptance of this new indication for finasteride. In the VA study, there was no difference in the rate of urinary retention between any of the four treatment groups. It is conceivable that α-blockers may also decrease the risk of urinary retention if examined in men with large prostates who are at risk for developing retention.
The Medical Therapy of Prostatic Symptoms (MTOPS) study compared the effects of doxazosin, finasteride, and the combination of these two agents versus placebo on acute urinary retention, urinary incontinence, renal insufficiency, recurrent urinary tract infection, and AUA symptom index scores in a large cohort of men with benign prostatic hyperplasia. The study enrolled 3047 patients greater than or equal to 50 years of age with AUA symptom scores ≥ 8 and followed them for a mean of 4.5 years. Risks of acute urinary retention were significantly reduced by combination therapy (81% risk reduction vs placebo, P < .001) and finasteride (68% risk reduction, P = .009). Although doxazosin alone also decreased risk of urinary retention by 34%, the difference was not statistically significant. Doxazosin, finasteride, and combination treatment all significantly lowered symptom scores versus placebo (64%, P < .001; 30%, P = .016; and 45%, P < .001, respectively). MTOPS analysis did not include stratification of subjects by prostate size, and thus it remains unknown whether finasteride and α-blocker monotherapies would have been even more effective in men with larger and smaller prostates, respectively.31
The incidence of acute urinary retention is lower in the 6-year, open-label extension studies of tamsulosin compared to the PLESS control group. This suggests that α-blockers may also play a beneficial role in preventing acute urinary retention in men with BPH.
Conclusion
What is the pathophysiology of clinical BPH? The development of microscopic BPH, bladder outlet obstruction, and LUTS is associated with aging. The overwhelming clinical evidence suggests that these three age-dependent parameters are not causally related. Undoubtedly there are some men whose prostatic enlargement causes obstruction and symptoms. Based upon the available data, however, this subset must be extremely small.
We know that TURP and radical prostatectomy result in highly significant improvements in LUTS. Obviously, the mechanisms of symptom improvement in these cases must be related to the prostate, bladder neck, or prostatic urethra, because only these tissues are resected or excised. Because men without bladder outlet obstruction respond equally well to prostatectomy, a plausible mechanism for the pathophysiology of BPH and the effectiveness of TURP may be a nonobstruction mechanism involving neurological pathways. Therefore drugs that influence this sensory afferent in the lower urinary tract, or the neural pathways that process this input, may represent an entirely new therapeutic strategy for men with LUTS.
We also know that α1-blockers represent an extremely effective pharmacological strategy for the treatment of BPH. There is increasing evidence that men with LUTS and no evidence of bladder outlet obstruction respond to α1-blockers. The mechanism for α1-mediated symptom improvement appears to be independent of bladder outlet obstruction. It is plausible that sensory innervation of the prostate may represent a target for α1 antagonists.
Because of the many urological and nonurological conditions that cause LUTS and the age-dependent changes in bladder and neurological function, it is unlikely that there exists a single dominant etiology for LUTS in the aging male population. If this is the case, then the optimal management of LUTS will require different and possibly combination therapies. The available data suggest that we should not direct our pharmacological strategies exclusively towards reducing prostate volume or diminishing bladder outlet obstruction. Only incremental advances in the medical management of BPH will be derived by developing subtype-selective α1 antagonists or more complete inhibitors of 5α-reductase. The quantum advances in the medical management of BPH will require a better understanding of the pathophysiology of LUTS.
Main Points.
Approximately 90% of men will develop histologic evidence of benign prostatic hyperplasia (BPH) by 80 years of age, and a statistically significant correlation also exists between age and prostate volume.
Medical therapies widely used today for treatment of BPH are targeted to diminish bladder outlet obstruction, thereby reducing prostate volume and relaxing prostate smooth muscle tension.
Clinical data demonstrate that androgen suppression and α-blockade relieve and increase urinary flow rates in men with BPH.
The observation that prostatic enlargement, bladder outlet obstruction, and lower urinary tract symptoms (LUTS) are all age-dependent has been interpreted to indicate that these phenomena are causally related, but there is insufficient evidence for this assumption.
The unequivocal effectiveness of transurethral resection of the prostate strongly suggests that the prostate and/or prostatic urethra must play an important role in the pathophysiology of clinical BPH.
Prostate smooth muscle density does not appear to be a major factor contributing to the severity of LUTS or to the α1-mediated improvement in symptomatology in men with clinical BPH.
Editor's Summary of Meeting Presentation
The finding that percent of smooth muscle content of the prostate correlates with flow rate improvement but not with symptom improvement, as shown by Dr. Lepor, is an interesting one, providing additional insight into one of the mechanisms of action of α-blocking agents, namely the relaxation of the smooth muscle via blockade of the effect of the adrenergic nerves on the α receptor. However, the fact that only flow rate but not symptom changes correlate with the smooth muscle density suggests that α-blockers may also act through other mechanisms.
Dr. Narayan suggested that in addition to the effects on the α receptors in the bladder neck and prostate, α-blockers may also have effects on the spinal cord and the central nervous system. Dr. Chapple agreed, stating that both spinal and pelvic postganglionic nerves may be involved in the overall effect of α-blocking agents on LUTS and BPH. Along this line of reasoning, the question of whether or not certain spinal cord injuries are known to mimic the typical lower urinary tract symptoms found in men with presumed BPH was raised. However, according to Dr. Nitti, most injuries of this nature have outcomes and sequelae that often are severe enough to override subtle changes that may occur regarding the typical LUTS symptomatology. Therapeutic options aiming to either anaesthetize nerves or destroy them, such as capsaicin or ethanol injection, may shed additional light on the role of the adrenergic nervous system, versus other sensory nerves in the bladder or prostate, in the pathophysiology of LUTS.
Age-matched men and women have similar levels of LUTS, and some of these symptoms have been attributed to the aging of the bladder. Along this line of thinking, it is interesting to note that, cystoscopically, the aging bladder in men and women takes on a different appearance, with trabeculation and diverticula formation less commonly seen in women, as Dr. Lowe stated. According to Dr. Nitti, however, thickened bladder walls and trabeculation are often encountered in women with voiding dysfunction. Although not necessarily conclusive, a study comparing morphometric aspects of detrusor muscle in men and women demonstrated remarkable similarities rather than the anticipated differences in terms of the ratio of connective tissue to smooth muscle (per Dr. Lepor). This finding is of particular interest, because secondary bladder changes are believed to be one the main, and thus far overlooked, causes of LUTS in aging men.
Dr. Lepor's presentation highlighted the evolution of our understanding of the pathophysiology of LUTS and BPH. Despite uncertainty regarding the relative contributions from various causes and etiologies, it is quite clear that the old terminology of prostatism and BPH, focusing exclusively on the prostate as the source of symptoms, is quite inaccurate. The modern term of LUTS is obviously a better choice when referring to this condition, because it does not refer to the prostate as the primary underlying cause.
References
- 1.McNeal JG. The prostate gland: morphology and pathobiology. Monogr Urol. 1983;4:3–33. [Google Scholar]
- 2.Berry SJ, Coffey DS, Walsh PC, et al. The development of human prostatic hyperplasia with age. J Urol. 1984;132:474–479. doi: 10.1016/s0022-5347(17)49698-4. [DOI] [PubMed] [Google Scholar]
- 3.Oesterling JE, Jacobsen SJ, Chute CG, et al. Serum prostate-specific antigen in a community-based population of health men: establishment of age-specific reference ranges. JAMA. 1993;270:860–864. [PubMed] [Google Scholar]
- 4.Barry MJ, O'Leary MP. The development and clinical utility of symptom scores. Urol Clin N Am. 1995;22:229–307. [PubMed] [Google Scholar]
- 5.Jacobsen SJ, Girman CJ, Lieber MM. Natural history of benign prostatic hyperplasia. Urology. 2001;58(suppl 1):5–16. doi: 10.1016/s0090-4295(01)01298-5. [DOI] [PubMed] [Google Scholar]
- 6.Lepor H, et al. Natural history, assessment of efficacy and nonsurgical treatment of BPH. In: Walsh PC, et al., editors. Campbell's Urology. 7th ed. Philadelphia, PA: WB Saunders Company; 1998. pp. 1453–1477. [Google Scholar]
- 7.Caine M. The present role of alpha-adrenergic blockers in the treatment of benign prostatic hypertrophy. J Urol. 1986;136:1–6. doi: 10.1016/s0022-5347(17)44709-4. [DOI] [PubMed] [Google Scholar]
- 8.Shapiro E, Lepor H. Pathophysiology of clinical BPH. Urol Clin N Am. 1995;22:285–290. [PubMed] [Google Scholar]
- 9.Isaacs JT, Coffey DS. Etiology and disease process of benign prostatic hyperplasia. Prostate Suppl. 1989;2:33–50. doi: 10.1002/pros.2990150506. [DOI] [PubMed] [Google Scholar]
- 10.Barry MJ, Cockett ATK, Holtgrewe HL, et al. Relationship of symptoms of prostatism to commonly used physiological and anatomical measures of the severity of benign prostatic hyperplasia. J Urol. 1993;150:351–358. doi: 10.1016/s0022-5347(17)35482-4. [DOI] [PubMed] [Google Scholar]
- 11.Nitti V, Kim Y, Combs AJ. Correlation of the AUA symptom index with urodynamics in patients with suspected benign prostatic hyperplasia. Neurourol Urodyn. 1994;13:521–529. doi: 10.1002/nau.1930130504. [DOI] [PubMed] [Google Scholar]
- 12.Tsukamoto T, Kumamoto Y, Masumori N, et al. Prevalence of prostatism in Japanese men in a population-based study with comparison to a similar American study. J Urol. 1995;154:391–395. doi: 10.1097/00005392-199508000-00018. [DOI] [PubMed] [Google Scholar]
- 13.Boyle P, Gould LA, Roehrborn CG. Prostate volume predicts outcome of treatment of benign prostatic hyperplasia with finasteride: meta-analysis of randomized clinical trials. Urology. 1996;48(3):398–404. doi: 10.1016/s0090-4295(96)00353-6. [DOI] [PubMed] [Google Scholar]
- 14.Lepor H, Machi GM. Comparison of AUA symptom index in unselected males and females between 55 and 79 years of age. Urology. 1993;42:36. doi: 10.1016/0090-4295(93)90332-5. [DOI] [PubMed] [Google Scholar]
- 15.Jacobsen SJ, Jacobson DJ, Girman CJ, et al. Natural history of prostatism: risk factors for acute urinary retention. J Urol. 1997;158:481–487. doi: 10.1016/s0022-5347(01)64508-7. [DOI] [PubMed] [Google Scholar]
- 16.McConnell JD, Bruskewitz R, Walsh PC, et al. The effect of finasteride on the risk of acute urinary retention and the need for surgical treatment among men with benign prostatic hyperplasia. N Engl J Med. 1998;338:557–563. doi: 10.1056/NEJM199802263380901. [DOI] [PubMed] [Google Scholar]
- 17.Schwartz EJ, Lepor H. Radical retropubic prostatectomy reduces symptom scores and improves quality of life in men with moderate and severe lower urinary tract symptoms. J Urol. 1999;161:1185–1188. [PubMed] [Google Scholar]
- 18.Caine M, Pfau A, Perlberg S. The use of alpha adrenergic blockers in benign prostatic obstruction. Br J Urol. 1976;48:255. doi: 10.1111/j.1464-410x.1976.tb03013.x. [DOI] [PubMed] [Google Scholar]
- 19.Gup DI, Shapiro E, Baumann M, Lepor H. Autonomic receptors in asymptomatic and symptomatic BPH. J Urol. 1990;143:179–185. doi: 10.1016/s0022-5347(17)39906-8. [DOI] [PubMed] [Google Scholar]
- 20.Gup DI, Shapiro E, Baumann M, et al. The contractile properties of human prostate adenomas and the development of infravesical obstruction. Prostate. 1989;15:105–114. doi: 10.1002/pros.2990150204. [DOI] [PubMed] [Google Scholar]
- 21.Shapiro E, Hartanto V, Lepor H. Anti-desmin vs. anti-actin for quantifying the area density of prostate smooth muscle. Prostate. 1992;20:259–267. doi: 10.1002/pros.2990200402. [DOI] [PubMed] [Google Scholar]
- 22.Shapiro E, Becich MJ, Lepor H. The relative proportion of stromal and epithelial hyperplasia as related to the development of clinical BPH. J Urol. 1992;147:1293–1297. doi: 10.1016/s0022-5347(17)37546-8. [DOI] [PubMed] [Google Scholar]
- 23.Spitsbergen JM, Clemon DB, McCarty R, et al. Neurally mediated hyperactive voiding in spontaneously hypertensive rats. Brain Res. 1998;790:151–159. doi: 10.1016/s0006-8993(98)00061-4. [DOI] [PubMed] [Google Scholar]
- 24.Lepor H, Shapiro E, Browsher R, Henry D. Tissue norepinephrine levels in prostate adenomas from men with symptomatic and asymptomatic BPH. J Urol. 1990;144:1263–1266. doi: 10.1016/s0022-5347(17)39716-1. [DOI] [PubMed] [Google Scholar]
- 25.Shapiro E, Hartanto V, Lepor H. The response to alpha blockade in benign prostatic hyperplasia is related to the percent area density of prostate smooth muscle. Prostate. 1992;21:297–307. doi: 10.1002/pros.2990210406. [DOI] [PubMed] [Google Scholar]
- 26.Lepor H, Rigaud G. The efficacy of transurethral resection of the prostate in men with moderate symptoms of prostatism. J Urol. 1990;143:533–537. doi: 10.1016/s0022-5347(17)40012-7. [DOI] [PubMed] [Google Scholar]
- 27.Lepor H, Williford WO, Barry MJ, et al. The efficacy of terazosin, finasteride, or both in benign prostatic hyperplasia. N Engl J Med. 1996;335:533–539. doi: 10.1056/NEJM199608223350801. [DOI] [PubMed] [Google Scholar]
- 28.Lepor H, Williford WU, Barry MJ, et al. The impact of medical therapy on bother due to symptoms, quality of life and global outcome and factors predicting response. Veterans Affairs Cooperative Studies Benign Prostatic Hyperplasia Study Group. J Urol. 1998;160:1358–1367. [PubMed] [Google Scholar]
- 29.Lepor H, Nieder A, Dixon CM, et al. The effectiveness of terazosin in men with normal and abnormal peak flow rates. Urology. 1997;49:476–480. doi: 10.1016/S0090-4295(96)00504-3. [DOI] [PubMed] [Google Scholar]