Abstract
We have evaluated the efficacy of voriconazole (VRC) in a systemic infection by Trichosporon asahii in immunosuppressed guinea pigs. VRC was more effective than amphotericin B in prolonging survival and reducing tissue burden. The best results were obtained with VRC at 10 mg/kg of body weight/day.
Trichosporonosis is an uncommon but frequently fatal invasive infection of immunocompromised patients caused by Trichosporon spp. (12, 18, 20, 21). Trichosporon asahii is the most common species involved (10, 11, 12, 13). Trichosporonosis is generally treated with amphotericin B (AMB) or fluconazole, commonly with an unsatisfactory outcome (1, 4, 6, 9, 12, 22). In a previous study we also demonstrated, using a murine model of disseminated infection by T. asahii, that the efficacy of both drugs was very limited but improved when each of them was combined with micafungin (16).
The aim of this study was to determine whether other therapeutic alternatives would also be effective in the treatment of trichosporonosis. Since voriconazole (VRC) has demonstrated in vitro activity (7, 9, 17, 19) even against multidrug-resistant strains of T. asahii (8), and favorable outcomes in several clinical cases have been reported (3, 9), we have evaluated the effectiveness of this drug in a guinea pig model. The pharmacokinetics of VRC in guinea pigs makes these animals appropriate for testing of this drug (15).
We have tested two isolates of T. asahii, IHEM 17910, from a blood culture of a patient with posthepatitis medullar aplasia, and IHEM 9325, from the floor of a swimming pool. On the day of infection, 24-h cultures on Sabouraud dextrose agar (SDA) were suspended in sterile saline and filtered through sterile gauze to remove clumps of cells or hyphae. The resulting suspensions, containing ≥95% conidial forms (arthroconidia and blastoconidia), were adjusted to the desired inoculum based on hemocytometer counts and by serial plating on SDA to confirm viability.
MICs of AMB and VRC against the two strains used in the study were determined by using a broth microdilution method for yeasts (14) and were defined as the lowest concentration resulting in 100% inhibition of growth for AMB and 50% inhibition of growth for VRC.
We used Hartley albino guinea pigs weighing 450 to 500g, and the Animal Welfare Committee at our university approved the conditions. Neutropenia was induced by intraperitoneal administration of cyclophosphamide at 100 mg/kg of body weight on days −3, −1, +1, +3, +8, and +13 (5). Animals were challenged intravenously with 6 × 106 CFU for strain IHEM 17910 and 1.2 × 107 CFU for IHEM 9325 in 0.2 ml. During a preliminary study to establish the lethal dose, we tested inocula of 6 × 106, 1.2 × 107, and 2 × 107 CFU, and we chose those inocula that produced an acute infection, with 100% of animals dying within 7 days of infection.
Groups of six animals were treated with VRC or AMB, both administered once a day. VRC was dissolved in polyethylene glycol and administered at 5 mg/kg/day (V5) or 10 mg/kg/day (V10) orally. AMB was administered at 1.5 mg/kg/day (A1.5) intraperitoneally. All the treatments began 24 h after infection and continued for 7 days. For tissue burden studies, we did a second experiment under the conditions described above, and the day after treatment finished, five animals for each strain and each treatment were sacrificed. Kidneys, spleens, and liver were removed aseptically, weighed, and homogenized in 2 ml of saline. Serial 10-fold dilutions were plated on SDA plates to determine numbers of CFU per gram, and plates were incubated at 35°C for 48 h.
Survival rates were evaluated by the Kaplan-Meier test with Graph Pad Prism software for Windows. CFU counts were analyzed by the Mann-Whitney U test using SPSS 11.5 for Windows.
In vitro results.
VRC showed very low MICs for both strains, IHEM 17910 and IHEM 9325 (0.06 and 0.03 μg/ml, respectively). The AMB MIC for both strains was 1 μg/ml.
In vivo results.
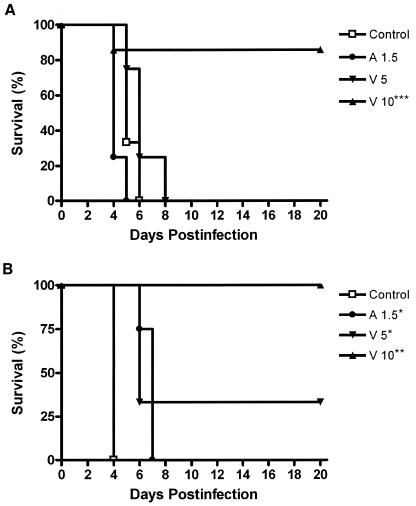
For strain IHEM 17910, A1.5 surprisingly decreased survival relative to that of the control group (P = 0.009) and V5 was not able to improve the survival of the control group (P = 0.2034). V10 prolonged survival relative to that of the control group (P = 0.0052), A1.5 (P = 0.0058), and V5 (P = 0.0069). Survival with V10 was 83.3%. For strain IHEM 9325, survival was improved over that of the control group by A1.5, V5, and V10 (P = 0.0047, P = 0.0082, and P = 0.0047, respectively). No significant mean survival time differences between V5 and V10 were observed for this strain (P = 0.0736). Only V10 prolonged survival relative to that with A1.5, attaining 100% survival (P = 0.0058) (Fig. 1).
FIG. 1.
Cumulative mortality of guinea pigs (n = 6) infected with T. asahii IHEM 17910 (A) or T. asahii IHEM 9325 (B) and given the indicated treatments. *P < 0.05 versus control; **, P < 0.05 versus control and A1.5; ***, P < 0.05 versus V5.
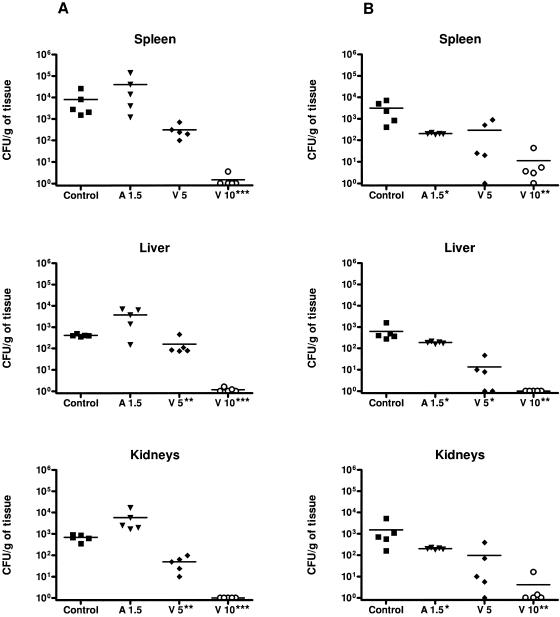
A1.5 did not reduce the fungal load in kidneys, spleens, and livers of animals infected with strain IHEM 17910. However, for strain IHEM 9325, this drug reduced the fungal loads in the three organs relative to those for the control group (P < 0.05). V5 for strain IHEM 17910 was able to reduce fungal loads significantly only in the kidneys and liver in comparison to the control group and A1.5. However, for strain IHEM 9325, V5 was able to reduce the fungal load in the liver only relative to that of the control group (P < 0.05), not versus A1.5. V10 significantly reduced tissue burdens for the two strains in comparison to controls and to the other two treatments (Fig. 2).
FIG. 2.
Effects of the antifungal treatments on colony counts of T. asahii strains IHEM 17910 (A) and IHEM 9325 (B) in spleens, livers, and kidneys of guinea pigs. *, P < 0.05 versus control; **, P < 0.05 versus control and A1.5; ***, P < 0.05 versus V5. Horizontal lines indicate mean values.
To our knowledge, this is the first report of disseminated trichosporonosis in a guinea pig model. The lack of efficacy of AMB observed here agrees with previous work using another animal model (16) and with clinical data (9). However, in some clinical cases, after failure of AMB and fluconazole therapy, the infection improved with VRC (3, 9). Our results confirmed the effectiveness of this drug (3, 9).
Pharmacodynamic parameters have proved to be useful in predicting antifungal efficacy (2). For azoles, values of the AUC (the 24-h area under the concentration-time curve)/MIC ratio over 25 are associated with a good outcome (2). In guinea pigs, the AUC for VRC at 10 mg/kg given once a day for 7 days is 29.0 (15). Consequently, the drug AUC/MIC ratio in our study was clearly superior to 25, even considering that the plasma protein binding of VRC in guinea pigs is 45% (15).
In summary, VRC has shown efficacy against systemic trichosporonosis and may represent an important advance in the therapy of this disease.
Acknowledgments
This work was supported by a grant from Fondo de Investigaciones Sanitarias from the Ministerio de Sanidad y Consumo of Spain (PI 050031).
REFERENCES
- 1.Anaissie, E. J., G. P. Bodey, H. Kantarjian, J. Ro, S. E. Vartivarian, R. Hopfer, J. Hoy, and K. Rolston. 1989. New spectrum of fungal infections in patients with cancer. Rev. Infect. Dis. 11:369-378. [DOI] [PubMed] [Google Scholar]
- 2.Andes, D., K. Marchillo, T. Stamstad, and R. Conklin. 2003. In vivo pharmacokinetics and pharmacodynamics of a new triazole, voriconazole, in a murine candidiasis model. Antimicrob. Agents Chemother. 47:3165-3169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Antachopoulos, C., E. Papakonstantinou, J. Dotis, E. Bibashi, M. Tamiolaki, D. Koliouskas, and E. Roilides. 2005. Fungemia due to Trichosporon asahii in a neutropenic child refractory to amphotericin B: clearance with voriconazole. J. Pedriatr. Hematol. Oncol. 27:283-285. [DOI] [PubMed] [Google Scholar]
- 4.Cawley, M. J., G. R. Braxton, L. R. Haith, K. J. Reilly, R. E. Guilday, and M. L. Patton. 2000. Trichosporon beigelii infection: experience in a regional burn center. Burns 26:483-486. [DOI] [PubMed] [Google Scholar]
- 5.Chandrasekar, P. H., J. Cutright, and E. Manavathu. 2000. Efficacy of voriconazole against invasive pulmonary aspergillosis in a guinea pig model. J. Antimicrob. Chemother. 45:673-676. [DOI] [PubMed] [Google Scholar]
- 6.Ebright, J. R., M. R. Fairfax, and J. A. Vazquez. 2001. Trichosporon asahii, a non-Candida yeast that caused fatal septic shock in a patient without cancer or neutropenia. Clin. Infect. Dis. 33:28-30. [DOI] [PubMed] [Google Scholar]
- 7.Espinel-Ingroff, A. 1998. In vitro activity of the new triazole voriconazole (UK-109,496) against opportunistic filamentous and dimorphic fungi and common and emerging yeast pathogens. J. Clin. Microbiol. 36:198- 202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Falk, R., D. G. Wolf, M. Shapiro, and I. Polacheck. 2003. Multidrug-resistant Trichosporon asahii isolates are susceptible to voriconazole. J. Clin. Microbiol. 41:911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fournier, S., W. Pavageau, M. Feuillhade, S. Deplus, A. M. Zagdanski, O. Verola, H. Dombret, and U. M. Molina. 2002. Use of voriconazole to successfully treat disseminated Trichosporon asahii infection in a patient with acute myeloid leukemia. Eur. J. Clin. Microbiol. Infect. Dis. 21:892-896. [DOI] [PubMed] [Google Scholar]
- 10.Groll, A. H., and T. J. Walsh. 2001. Uncommon opportunistic fungi: new nosocomial threats. Clin. Microbiol. Infect. 7:824. [DOI] [PubMed] [Google Scholar]
- 11.Guého, E., M. T. Smith, G. S. de Hoog, G. Billon-Grand, R. Christen, and W. H. Batenburg-van der Vegte. 1992. Contributions to a revision of the genus Trichosporon. Antonie Leeuwenhoek 62:289-316. [DOI] [PubMed] [Google Scholar]
- 12.Itoh, T., H. Hosokawa, U. Kohdera, N. Toyazaki, and Y. Asada. 1996. Disseminated infection with Trichosporon asahii. Mycoses 39:195-199. [DOI] [PubMed] [Google Scholar]
- 13.Moretti-Branchini, M. L., K. Fukushima, A. Z. Schreiber, K. Nishimura, P. M. Papaiordanou, P. Trabasso, R. Tanaka, and M. Miyaji. 2001. Trichosporon species infection in bone marrow transplanted patients. Diagn. Microbiol. Infect. Dis. 39:161-164. [DOI] [PubMed] [Google Scholar]
- 14.National Committee for Clinical Laboratory Standards. 2002. Reference method for broth dilution antifungal susceptibility testing of yeasts. Approved standard M27-A2, 2nd ed. National Committee for Clinical Laboratory Standards, Wayne, Pa.
- 15.Roffey, S. J., S. Cole, P. Comby, D. Gibson, S. G. Jezequel, A. N. R. Nedderman, D. A. Smith, D. K. Walker, and N. Wood. 2003. The disposition of voriconazole in mouse, rat, rabbit, guinea pig, dog and human. Drug Metab. Dispos. 31:731-741. [DOI] [PubMed] [Google Scholar]
- 16.Serena, C., F. J. Pastor, F. Gilgado, E. Mayayo, and J. Guarro. 2005. Efficacy of micafungin in combination with other drugs in a murine model of disseminated trichosporonosis. Antimicrob. Agents Chemother. 49:497-502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Serena, C., F. J. Pastor, M. Ortoneda, J. Capilla, N. Nolard, and J. Guarro. 2004. In vitro antifungal susceptibility of uncommon basidiomycetous yeasts. Antimicrob. Agents Chemother. 48:2724-2726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tashiro, T., H. Nagai, H. Nagaoka, Y. Goto, P. Kamberi, and M. Nasu. 1995. Trichosporon beigelii pneumonia in patients with hematologic malignancies. Chest 108:190-195. [DOI] [PubMed] [Google Scholar]
- 19.Uzun, O., S. Arikan, S. Kocagoz, B. Sancak, and S. Unal. 2000. Susceptibility testing of voriconazole, fluconazole, itraconazole and amphotericin B against yeast isolates in a Turkish university hospital and effect of time of reading. Diagn. Microbiol. Infect. Dis. 38:101-107. [DOI] [PubMed] [Google Scholar]
- 20.Vasta, S., M. Menozzi, R. Scime, A. Indovina, A. Speciale, G. Liberti, C. Spano, and I. Majolino. 1993. Central catheter infection by Trichosporon beigelii after autologous blood stem cell transplantation. A case report and review of the literature. Haematologica 78:64-67. [PubMed] [Google Scholar]
- 21.Walsh, T. J., K. R. Newman, M. Moody, R. C. Wharton, and J. C. Wade. 1986. Trichosporonosis in patients with neoplastic disease. Medicine (Baltimore) 65:268-279. [DOI] [PubMed] [Google Scholar]
- 22.Walsh, T. J., G. P. Melcher, M. G. Rinaldi, J. Lecciones, D. A. McGough, P. Kelly, J. Lee, D. Callender, M. Rubin, and P. A. Pizzo. 1990. Trichosporon beigelii, an emerging pathogen resistant to amphotericin B. J. Clin. Microbiol. 28:1616-1622. [DOI] [PMC free article] [PubMed] [Google Scholar]