Abstract
We report the construction and application of a novel insertion element for transposase-mediated mutagenesis in gram-negative bacteria. Besides Kmr as a selectable marker, the insertion element InsTetG−1 carries the anhydrotetracycline (atc)-regulated outward-directed PA promoter so that atc-dependent conditional gene knockouts or knockdowns are generated. The complex formed between the purified hyperactive transposase and InsTetG−1 was electroporated into Escherichia coli or Salmonella enterica serovar Typhimurium, and mutant pools were collected. We used E. coli strains with either TetR or the reverse variant revTetRr2, while only TetR was employed in Salmonella. Screening of the InsTetG−1 insertion mutant pools revealed 15 atc-regulatable auxotrophic mutants for E. coli and 4 atc-regulatable auxotrophic mutants for Salmonella. We have also screened one Salmonella mutant pool in murine macrophage-like J774-A.1 cells using ampicillin enrichment. Two mutants with the InsTetG−1 insertion in the gene pyrE or argA survived this procedure, indicating a reduced intracellular growth rate in J774-A.1 cells. The nature of the mutants and the modes of their regulation are discussed.
In recent years, the application of sophisticated tools has led to a rapidly growing wealth of genomic, transcriptomic, and proteomic information for bacteria. It has been assumed that many new targets for antiinfectives may be identified from these data. However, the function of about 40% of the genes (26) and their encoded proteins turns out to be unknown in most organisms. Therefore, there is now a need to develop new generally applicable methods to analyze gene functions. Responding to this need, several efficient mutagenesis protocols have been developed for a wide range of pathogenic and nonpathogenic bacteria. Many of them contain transposable elements to create insertion mutants because the site of mutation is tagged. The expression of transposase in the target cell is required for transposition but should be eliminated afterwards to ensure the stability of the mutants (16). Therefore, in vitro transposition systems, like the one developed previously by Goryshin et al. in which an insertion element is complexed with purified transposase, which is followed by electroporation of this complex into bacterial cells, are used for mutagenesis in vitro or in vivo (10, 17). While this system is very efficient for creating randomized knockouts of genes, it is incapable of revealing essential genes directly. Insertion elements carrying a regulatable promoter yielding conditional expression of downstream genes may circumvent this limitation. Several such constructs make use of the arabinose-inducible promoter pBAD in combination with transposition (32). This promoter responds to arabinose, which requires an uptake system to enter bacteria. Furthermore, it cannot be used in mammalian pathogenicity models because that inducer is metabolized by the infected animal. The Tn5tac system (8) responds to IPTG (isopropyl-β-d-thiogalactopyranoside) and is limited by the strong Ptac promoter, causing overexpression effects and a lack of tight repression. We have designed and constructed an insertion element for gram-negative bacteria using a tetracycline-dependent transcriptional control system to regulate genes located downstream from the insertion site. The inducers tetracycline, doxycycline, and anhydrotetracycline (atc) do not require an uptake system to penetrate most cells (33, 34) and function in eukaryotic cell lines (13) and transgenic mammals (38). Thus, the regulation of genes should also be possible in infection models. We describe here the construction and efficiency of an insertion element, based on the Tn5 in vitro system developed previously by Goryshin et al. (12), to obtain atc-responsive conditional gene knockouts in Escherichia coli and Salmonella enterica serovar Typhimurium.
MATERIALS AND METHODS
Bacterial strains, media, and growth conditions.
Bacterial strains used in this study are listed in Table 1. E. coli and Salmonella serovar Typhimurium strains were grown at 37°C in Luria-Bertani (LB) broth or M9 minimal medium containing 0.2% glucose as a carbon source. For antibiotic resistance marker selection, bacterial media were additionally supplemented with 25 μg/ml of chloramphenicol (Cm), 60 μg/ml or 100 μg/ml of kanamycin (Km), or 100 μg/ml of ampicillin (Ap) where needed.
TABLE 1.
Bacterial strains and plasmids used
| Strain or plasmid | Genotype | Reference or source |
|---|---|---|
| Strains | ||
| E. coli K-12 | ||
| DH5α | recA1 endA1 gyrA96 thi relA1 hsdR17 (rK− mK+) supE44 φ80dlacZΔ ΔlacU169 | 4 |
| MG1655 | rph-1 | 5 |
| ER2566 | fhuA2 lacZ::T7 gene 1 [lon] ompT gal sulA11 R(mcr-73::mini-Tn10-Tets)2 [dcm] R(zgb-210::Tn10-Tets) endA1Δ (mcrC-mrr) 114::IS10 | 7 |
| Salmonella enterica serovar | Wild type | ATCC 14028 |
| Typhimurium NCTC12023 | ||
| Plasmids | ||
| pCB302b | Apr, pBR322 derivative, divergent promoterless lacZ and galK | 29 |
| pDG972 | Kmr, pMTL23 derivative | 14 |
| pTYB2 | Cmr, pACYC derivative, C-terminal intein-chitin binding domain fusion | 7 |
| pUC18 | AprlacZα | 37 |
| pWH1411BD | Cmr, pACYC177 derivative, tetR(BD)51-208 | 31 |
| pWH1411r2 | Cmr, pACYC177 derivative, tetRr2(BD)E15A, L17G, L25V; XbaI/NcoI from pWH1925-TetRr2 | This study |
| pWH1865 | Apr/Kmr, pUC18 derivative, InsTetG−1 in XbaI/NcoI pWH1865 ΔKm | This study |
| pWH1865ΔKmr | Apr, pUC18 derivative, InsTetG−1 ΔaphAIII XbaI/NcoI | This study |
| pWH1866 | Apr/Kmr, pUC18 derivative, InsTetG−1 flanked by PvuII sites | This study |
| pWH1867 | Apr/Kmr, pCB302b derivative, InsTetG−1 upstream of a promoterless lacZ | This study |
| pWH1891 | Apr, pTYB2 derivative, Tn5 transposase E54K, M56A, L372P; C-terminal intein-chitin binding domain fusion | This study |
| pWH1925r2 | Apr, pUC19 derivative, tetR(BD)E15A, L17G, L25V | 30 |
General methods.
For measurements of β-galactosidase (β-gal) activity, E. coli cells were cotransformed with pWH1867 and pWH1411BD, encoding wild-type (wt) TetR, or pWH1411r2, encoding revTetRr2, respectively (Table 1). Three independent clones of each strain were taken from log-phase cultures at 37°C, and β-gal activities were determined as described previously (25, 30).
For the preparation of electrocompetent cells, 1 liter of LB broth containing 25 mg chloramphenicol was inoculated with E. coli MG1655/pWH1411BD or pWH1411r2 or Salmonella serovar Typhimurium 12023/pWH1411BD. After the optical density at 600 nm (OD600) had reached 0.4, the culture was chilled on ice for 30 min and then harvested by centrifugation at 4,000 × g for 10 min. The supernatant was discarded, and the pellet was washed four times in 10 ml of 10% ice-cold glycerol and finally resuspended in 1 ml of 10% ice-cold glycerol. This cell suspension was stored in 100-μl aliquots at −70°C.
Electroporation of InsTetG−1-transposase complexes into E. coli and Salmonella serovar Typhimurium was carried out in 2-mm-gap electroporation cuvettes with 40 μl of cell suspension and 1 μl of the reaction mixtures, as specified in Results, at 2,500 V (τ = 5 ms) using a Bio-Rad (Munich, Germany) Gene Pulser. For E. coli, the suspension was diluted to 2 ml with LB medium, transferred into 2.2-ml microcentrifuge tubes, and incubated for 60 min at 37°C before plating onto agar as specified in Results. In the case of Salmonella serovar Typhimurium, SOC medium (100 μl 1 M MgCl2, 100 μl 1 M MgSO4, 200 μl 20% glucose diluted in 10 ml SOB medium [10 g/liter tryptone, 2.5 g/liter yeast extract, 0.85 ml 5 M NaCl, 1 ml 1 M KCl]) was used for dilution after electroporation.
For ampicillin enrichment of the Salmonella serovar Typhimurium InsTetG−1 insertion mutant library, a final ampicillin concentration of 100, 200, or 500 μg/ml was used.
Chromosomal DNA was isolated from kanamycin-resistant clones of E. coli MG1655 containing either pWH1411BD or pWH1411BDr2 and Salmonella serovar Typhimurium 12023/pWH1411BD according to the QIAamp DNA Mini kit protocol (QIAGEN, Hilden, Germany). The DNA was digested with EcoRI, which was followed by 20 min heat inactivation at 70°C. A total of 1.5 μg of the DNA was sequenced using 6 μl Big Dye Terminator mix (Applied Biosystems, Weiterstadt, Germany) and 2 μl of the sequencing primer Tn-out_neu (5′-GGTCCTAATTTTTGTTGACACTC-3′) (20 pmol) and adjusted with deionized water to a total volume of 20 μl. The reaction products were ethanol precipitated, washed once with 70% ethanol, and resuspended in 10 μl formamide, and the sequence was determined using an ABI 310 genetic analyzer (PE Biosystems, Weiterstadt, Germany).
Construction of the insertion element InsTetG−1.
The oligonucleotides IE1-1 (5′-GGGAATTCTCTAGACTGTCTCTTATACACATCTTAAGTAGGTAAAAATATAATGACCCTCTTG-3′), IE1-2 (5′-TATATTTTTACCTACTTAAGATGTGTATAAGAGACAGTCTAGAGAATTCCC-3′), IE2-1 (5′-TACTGGGAGAACTATTGGGTTCTCCCGTAAAAAATGGTCCCAGGATTAAAAACAACTGTGAGA-3′), IE2-2 (5′-TTTTAATCCTGGGACCATTTTTTACGGGAGAACCCAATA-3′), IE3-1 (5′-TGTTGACACTCTATCATTGATAGAGTTATTTTACCTCTCCCTATCAGTGATAGAGAAAAAGAT-3′), IE3-2 (5′-ATCACTGATAGGGAGAGGTAAAATAACTCTATCAATGAT-3′), IE-4-1 (5′-TCTCTTTTTCTACACATATTCTCTGTCTTCGAATACATACTCACTACACC-3′), and IE4-2 (5′-GGTGTAGTGAGTATGTATTCGAAGACAGAGAATATGTG-3′) comprise the sequence of the insertion element InsTetG−1 with an NcoI restriction site for insertion of the kanamycin resistance gene. They were phosphorylated with T4 polynucleotide kinase (NEB, Frankfurt/Main, Germany). Equimolar amounts of complementary oligonucleotides were hybridized by heating to 96°C and slow cooling to ambient temperature. The hybridized DNA fragments were ligated for 16 h at 4°C, amplified by PCR using the outside primers IE1-1 and IE4-2, digested with BamHI and XbaI, and ligated with pUC18 restricted with the same nucleases. The resulting plasmid, pWH1865ΔKm, was digested with NcoI and ligated with the kanamycin resistance cassette obtained by PCR from plasmid pDG792 (14) with the oligonucleotides Km1-neu (5′-GAAGAGGATGCCATGGCAGATTGCCTTG-3′) and Km2 (5′-GTAGTTAAAGCTCCATGGACATCTAAATCTAGGTAC-3′) to obtain plasmid pWH1865. A PCR with the oligonucleotides PvuII-hind (5′-GCCAGTGCCAAGCAGCTGTCTCTTATAC-3′) and PvuII-xba (5′-CGGGGATCCTCTCAGCTGTCTCTTATACAC-3′) introduced PvuII sites at each end of InsTetG−1. The PCR fragment and pUC18 were digested with PvuII, and the vector was additionally dephosphorylated and ligated. The resulting plasmid, pWH1866, was cut with PvuII, and InsTetG−1 was purified from an agarose gel using the Nucleo Spin Extract kit (Macherey & Nagel, Düren, Germany). The DNA concentration was determined from the absorption at 260 nm. The reporter plasmid pWH1867 contains InsTetG−1 transcriptionally fused to a promoterless lacZ. InsTetG−1 was obtained from pWH1865 by XbaI restriction, blunt ending using T4-DNA polymerase (NEB, Frankfurt/Main, Germany), and then restriction with HindIII. pCB302b (29) was digested with AgeI and HindIII and ligated with the InsTetG−1 fragment to obtain pWH1867.
Cloning and purification of the hyperactive Tn5 transposase.
The hyperactive transposase mutant (12) was generated from transposon Tn5 by PCR mutagenesis and cloned into the overexpression vector pTYB2 to yield pWH1891. This plasmid contains a self-cleaving intein tag fused to a chitin binding domain originating from the IMPACT-CN kit (NEB, Frankfurt/Main, Germany) and a T7 promoter for overexpression. E. coli ER2566 transformed with pWH1891 was grown in 3 liters of LB medium at 23°C until an OD600 of 0.5 was reached, 0.5 mM of IPTG was then added, and the culture was incubated for another 5 h. The cells were harvested, washed once with 80 ml of TEGX (20 mM Tris-HCl, pH 7.5, 0.7 M NaCl, 1 mM EDTA, 10% [vol/vol] glycerol, 0.1% [vol/vol] Triton X-100), resuspended in 20 ml of TEGX containing protease inhibitors (Complete Mini EDTA Free; Roche, Penzberg, Germany), sonified, and centrifuged at 45,000 rpm (L7-55 ultracentrifuge, 60 TI rotor; Beckmann, Munich, Germany) for 1 h at 4°C. A chitin affinity column was loaded with the supernatant, washed with 10 column volumes of TEGX, flushed with 1.5 volumes of TEGX containing 50 mM dithiothreitol, incubated overnight, and eluted the next day with TEGX. Fractions containing transposase were collected, aliquoted, and stored at −20°C in storage buffer (TEGX with 50% glycerol).
Transposition reactions in vitro and in vivo.
In vitro insertion of InsTetG−1 into pUC19 was done according to the protocol provided by Epicenter (Madison, Wis.). One hundred nanograms of InsTetG−1 was mixed with 100 ng of pUC19 and different molar ratios of the hyperactive transposase in a total volume of 10 μl in reaction buffer (0.5 mM Tris-acetate, pH 7.5, 1.5 M potassium acetate, 100 mM magnesium acetate, 40 mM spermidine). After incubation at 37°C for 2 h, 1 μl of a 1% sodium dodecyl sulfate solution was added, and the incubation continued for 10 min at 70°C. One microliter of the reaction mixture was transformed into E. coli DH5α, and the transformants were plated onto LB agar with kanamycin (60 mg/liter).
Creation of the InsTetG−1-transposase complex (transposomes) in vitro was performed by mixing 110 fmol (E. coli) or 545 fmol (Salmonella serovar Typhimurium) InsTetG−1 with a fivefold molar excess of the hyperactive Tn5 transposase. Glycerol was then added to a final concentration of 25% in a total volume of 10 μl, the mixture was incubated at 37°C for 30 min, and 1 μl of the mixture was used for electroporation.
Ampicillin enrichment of intracellular InsTetG−1-derived Salmonella serovar Typhimurium mutants.
J774-A.1 cells were maintained in a 100-mm petri dish in a humidified atmosphere with 5% CO2 at 37°C in Dulbecco's modified Eagle's medium (DMEM) containing 5% fetal bovine serum and 2 mM l-glutamine. About 0.5 × 106 J774-A.1 cells between passages 6 and 30 were seeded into each well of a six-well plate and incubated for 24 h. A culture of the InsTetG−1 insertion library of Salmonella serovar Typhimurium was grown overnight at 37°C in LB medium and used to inoculate two cultures. One culture was grown in M9 medium to an OD600 of about 0.3, and the other culture was first grown in LB medium to an OD600 of about 0.3 and was then washed twice with M9 medium, resuspended in M9 medium, and incubated for 1.5 h at 37°C. The J774-A.1 cells were washed with 1 ml 1× phosphate-buffered saline (PBS) and infected with about 1 × 107 Salmonella serovar Typhimurium cells in 1 ml 1× PBS. After a 30-min incubation at 37°C, the macrophage cells were washed three times with 1× PBS followed by another incubation for 1 h with DMEM and 100 μg/ml gentamicin. The monolayers were then washed three times with 1× PBS and incubated for 20 h in DMEM with 500 μg/ml of ampicillin and 10 μg/ml of gentamicin. The macrophage cells were washed three times with 1× PBS, and the J774-A.1 cells were lysed with 1 ml of sterile distilled water. The lysate was spread onto LB plates containing kanamycin and incubated overnight at 37°C, and the Salmonella serovar Typhimurium mutants were tested for intracellular growth deficiencies by employing the same procedure described above for intracellular selection. The chromosomal locations of the InsTetG−1 insertions in the surviving mutants were determined as described above.
RESULTS AND DISCUSSION
Construction and features of the insertion element InsTetG−1.
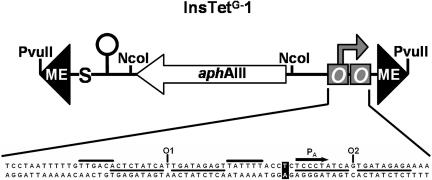
The features of the insertion element designed and constructed in this work are depicted in Fig. 1. It consists of 1,376 bp flanked by PvuII sites. The element is bordered by two 19-bp transposase recognition sequences (ME) necessary to form the transposase complex. The tetA promoter (PA) containing two tetO sequences is located next to ME on the right side of the figure and transcribes out of the insertion element. It was mutated at one position to eliminate PR promoter activity and prevent TetR-dependent transcription into the element (15). The middle portion of the insertion element consists of the kanamycin resistance cassette, which is needed for selection, flanked by NcoI sites. The left side of Fig. 1 shows the stop codons in all three reading frames and a transcriptional terminator to protect the element from incoming translation or transcription. InsTetG−1 was constructed by hybridizing eight oligonucleotides containing both MEs, the tetA promoter, and the translational and transcriptional stop sequences. Cloning of this DNA was followed by insertion of a kanamycin resistance cassette into the NcoI site, as detailed in Materials and Methods.
FIG. 1.
Architecture of the insertion element InsTetG−1. The entire element is shown schematically. It is 1,376 bp long and flanked by 19-bp recognition sites for hyperactive Tn5 transposase (ME). The modified tet PA promoter (right-directed gray arrow) on the one side containing two tet operators (gray boxes) can drive transcription of downstream genes. The kanamycin resistance gene (aphAIII) allows the selection of candidates. A bidirectional transcription terminator from Tn10 (stem-loop) upstream of the Kmr cassette blocks ingoing and outgoing transcription, and stop codons in all three forward reading frames (“S”) terminate possible translation from upstream. Restriction sites for NcoI at both ends of the kanamycin cassette facilitate the exchange of the selection marker, and restriction sites for PvuII at both ends of the insertion element enable blunt cutting for in vitro transposome formation. The sequence of the modified tetracycline-dependent control region of transposon Tn10 is displayed underneath. One base pair mutation (black boxed letters) shuts down the activity of both PR promoters without influencing the PA promoter. O1/O2 and the underlined sequences indicate the operator sequences, and the −35 and −10 regions of the PA promoter are indicated by black boxes. The arrow depicts the transcription start site of the promoter.
Testing of the regulatory properties of InsTetG−1 in E. coli and Salmonella serovar Typhimurium.
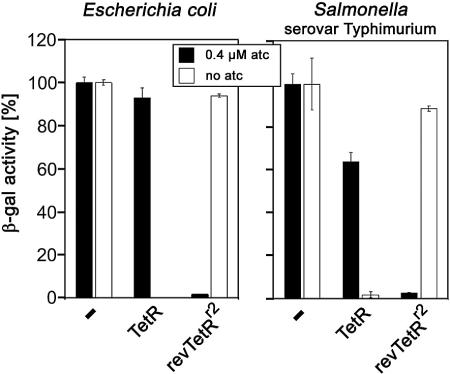
Plasmid pWH1867 contains the insertion element located upstream of lacZ, placing it under the control of the PA promoter. Two different variants of TetR were used for regulation. pWH1411BD carries wt TetR, which allows transcription only in the presence of atc, while pWH1411r2 encodes revTetRr2 (3) and shows expression of lacZ in the absence and repression in the presence of 0.4 μM atc, an amount that ensures full induction without growth inhibition, when cotransformed with pWH1867 in E. coli DH5α and Salmonella serovar Typhimurium 12023. Western blot analysis confirmed that the two TetR variants are expressed at the same levels in both organisms (data not shown). The results of β-gal expression for each strain are shown in Fig. 2 and reveal efficient regulation in all four strains. The induction factors are 2,080-fold for TetR and 60-fold for revTetRr2 in E. coli and 50-fold for TetR and 30-fold for revTetRr2 in Salmonella serovar Typhimurium. Regulation by TetR is not as tight in Salmonella serovar Typhimurium as it is in E. coli with this combination of plasmids, but revTetRr2 regulation is almost the same. These results establish the large regulation window of the atc-inducible promoter. Wild-type TetR efficiently shuts down PA in the absence of an inducer, as does revTetRr2 in the presence of atc, while with the latter regulator, a slightly increased basal expression level is observed. The PA promoter may be too strong in front of some genes; however, this can be circumvented by the use of lower atc concentrations or by less-efficient inducers (19, 23). We assume that the available options for the adjustment of induction levels will meet the needs of most genes. In an attempt to make use of these properties, a Tn10-based system was developed previously (28); however, this transposon has the disadvantage of nonrandom insertion, leading to mutational hotspots (1). Furthermore, a Tn10 variant in which the tetracycline resistance gene tetA was present has been employed (20-22). As a result, tetracycline-induced transcription must proceed from PA through the resistance gene before reaching the target gene downstream of the insertion site. Since tetA overexpression decreases bacterial fitness (9), this may influence the observed phenotype.
FIG. 2.
Test of the regulatory function of the insertion element InsTetG−1 cloned in front of a promoterless lacZ gene and cotransformed with TetR-expressing plasmids. The large regulatory window of InsTetG−1 was determined by measurements of β-gal activity. The percent β-gal activity can be seen on the y axis. The left panel shows the results for E. coli and the right panel shows results for Salmonella serovar Typhimurium with atc (black columns) and without atc (white columns). Measurements without a regulator (−) represent the 100% values, with 1,120 Miller units (MU) (with atc) and 1,200 MU (without atc) for E. coli and 2,350 MU (with atc) and 1,720 MU (without atc) for Salmonella serovar Typhimurium. TetR and revTetRr2 represent the two TetR variants expressed from pWH1411.
Optimizing the insertion reaction in vitro.
To determine the conditions that yield the maximal number of insertions with the transposase-InsTetG−1 complex, we performed an in vitro insertion experiment using pUC19 DNA. One hundred nanograms of purified insertion element InsTetG−1 was mixed with an equimolar amount or up to a 20-fold excess of purified transposase. The insertion reaction was done as detailed in Materials and Methods. The products were transformed into E. coli DH5α, and the transformants were selected for kanamycin resistance. A fivefold molar excess of the monomeric transposase over insertion element yielded the largest number of kanamycin-resistant candidates (data not shown) and was used for further experiments. Thirty of the candidates were randomly picked, their plasmids were purified, and the InsTetG−1 insertion sites were determined by sequencing with an outward primer. Twenty-four of these candidates exhibited different insertion loci (data not shown). Most of the sites were found within bla, lacZ, or the polylinker region, while no insertions occurred in ori, thus indicating random target site selection.
Creating pools of E. coli mutants with InsTetG−1.
To explore the insertion properties of InsTetG−1 in E. coli, pools of insertion mutants were created and subsequently screened for auxotrophy. For that purpose, 1 μl of the freshly prepared InsTetG−1-transposase complex (for details, see Material and Methods) was electroporated into freshly prepared electrocompetent cells, which were then spread onto kanamycin/chloramphenicol plates (60 mg/liter Km and 25 mg/liter Cm) and incubated overnight at 37°C. All colonies were washed from the agar plates using LB medium, the suspension was centrifuged and resuspended in 1 ml of LB medium containing 10% (vol/vol) dimethyl sulfoxide, and 100-μl aliquots were stored at −70°C as stocks. Following this procedure, one mutant pool with about 25,000 candidates was created from E. coli MG1655/pWH1411BD encoding wt TetR, and another mutant pool containing about 105,000 candidates was created from E. coli MG1655/pWH1411r2 encoding the reverse TetRr2.
Screening for auxotrophic E. coli mutants.
We developed a screening protocol by employing 96-well plates so that the growth of the candidates could be monitored by reading the OD595. Aliquots of the mutant pools were streaked onto LB plates containing kanamycin and chloramphenicol so that individual colonies could be picked from the plates. Ninety-six-well plates were supplemented with 200 μl of M9 minimal medium containing 0.2% glucose and kanamycin and chloramphenicol for selection, except for the well containing the wild-type strain, for which kanamycin was omitted. Each well was inoculated from one colony, the resulting suspension was mixed, and 100 μl was transferred into a well of a second plate containing 25 μl of the same medium supplemented with 2 μM atc. Subsequently, 25 μl of minimal medium was added to the wells in the first plate so that all wells contained the same volume of 125 μl of broth. The plates were then incubated at 37°C for 24 h on a stroking shaker in the dark to account for the light sensitivity of atc, and their OD595 was determined in a well plate reader. Using this procedure, we obtained four different phenotypes. Most of the strains were prototrophic, showing the same OD595 in both wells. The second-largest population displayed an unregulated auxotrophic phenotype (a mutant was scored auxotrophic when the OD595 was below 0.1) where mutants could not grow in either minimal medium. The third-largest number of mutants displayed a regulatable auxotrophic phenotype only growing in the absence (E. coli strain with TetRr2) or presence (E. coli strain with TetR) of atc. The fewest candidates had a reverse regulatable phenotype and were able to grow only in the absence of atc. All these candidates originated from the E. coli strain with TetR. All mutants with an auxotrophic phenotype were streaked onto LB plates and restreaked onto minimal medium plates with and without atc to verify their phenotypes. After 36 h of incubation in the dark, the mutants with a confirmed phenotype were grown in liquid LB medium to obtain stock cultures. The screening procedure described above was carried out twice for each of the mutant pools so that a total of 9,920 candidates were examined, leading to 396 candidates with one of the three auxotrophic phenotypes. However, only 106 of the candidates were confirmed on plates. This is a somewhat disappointing result, which may be related to the different growth conditions in the well plates compared to agar plates. Ninety-one of these mutants had a nonregulatable auxotrophic phenotype, and 15 mutants displayed atc-regulatable auxotrophy, as shown in Fig. 3.
FIG. 3.
M9 minimal medium agar plates containing 2% glucose as a carbon source with and without 0.4 μM atc (+atc and −atc, respectively) showing the growth phenotypes of the regulatable E. coli candidates. Letters A to M refer to the candidates described in Table 3.
Locations of the InsTetG−1 insertions in E. coli.
From the 106 mutants with a confirmed phenotype, chromosomal DNA was prepared and restricted with EcoRI, and the insertion sites were determined by sequencing with an InsTetG−1-specific primer. The resulting sequences were analyzed using the BLAST server at the EcoCyc database (http://ecocyc.org). The affected genes are shown in Table 2, where they are grouped according to their functions. The regulatable mutants are depicted in boldface type. Seventy-four different genes were mutated, with 32 of them showing more than one insertion site. Two to three identical mutants were found only in the genes ilvC, pgi, and b3557. The insertion sites of all mutants are randomly distributed in the E. coli chromosome (data not shown). The 9 bp of the short direct repeat flanking the insertion sites and the 1 bp immediately adjacent to it (sequences not shown) agree with the previously published consensus sequence (11), in which only the first of the 9 bp shows a clear preference for one nucleotide. For all other positions, no single nucleotide is present in more than 40% of all isolates. This result clearly demonstrates the random distribution of the insertions and the low redundancy obtained with this mutagenesis protocol.
TABLE 2.
Insertion mutants of E. coli and functions of the affected genesa
| Classification | Gene(s) | Pathway |
|---|---|---|
| Biosynthetic pathways | argC, D, E, G | Arginine biosynthesis |
| aroB | Chorismate biosynthesis | |
| bioA, B | Biotin biosynthesis | |
| carB | Pyrimidine and arginine biosynthesis | |
| cvpA | Colicin V biosynthesis | |
| cysD, G, J, Q | Sulfur metabolism | |
| entB, F | Enterobactin biosynthesis | |
| hflD | Membrane protein in operon with purB | |
| hisB | Histidine biosynthesis | |
| ilvA, C, D | Valine, isoleucine biosynthesis | |
| leuA, B, C | Leucine biosynthesis | |
| menD | Menaquinone biosynthesis | |
| metA, B, C, E | Methionine biosynthesis | |
| panB, C | Pantothenate biosynthesis | |
| pdxB | Pyridoxal 5′-phosphate biosynthesis | |
| proA | Proline biosynthesis | |
| purC, H | Purine nucleotide biosynthesis | |
| pyrD, F | Pyrimidine ribonucleotide biosynthesis | |
| rfaG | Lipopolysaccharide core biosynthesis | |
| serA, C | Serine biosynthesis | |
| thiC | Thiamine biosynthesis | |
| thrA | Homoserine biosynthesis | |
| trpA, B, C, E | Tryptophan biosynthesis | |
| tyrA | Phenylalanine/tyrosine biosynthesis | |
| Catabolism | fruK | 1-Phosphofructokinase |
| gltA | Citrate synthase | |
| mtlA | Subunit of EIImtlA | |
| pgi | Phosphoglucose isomerase | |
| ppc | Phosphoenolpyruvate carboxylase | |
| Regulation | argP | Transcriptional regulator |
| cspA | Transcriptional activator | |
| cysB | Transcriptional regulator | |
| ilvY | Transcriptional regulator | |
| Transporters | nirC | Nitrite transporter |
| cysP | Thiosulfate ABC transporter | |
| glcA | Glycolate transporter | |
| xylE | Xylose major facilitator superfamily transporter | |
| Essential genes | lhr | Member of ATP-dependent helicase superfamily II |
| mraZ | Member of dcw cluster (cell division) | |
| prmA | Methylation of 50S ribosomal subunit protein L11 | |
| rplM | L13 protein of 50S ribosomal subunit | |
| sfmH | Involved in fimbrial assembly | |
| truA | Pseudouridylate synthase I | |
| Unknown function | b3557 | IS5-related protein |
| ydeO | Putative AraC-type regulatory protein | |
| yfgL | Putative dehydrogenase | |
| yhdP | Putative transport protein | |
| yidL | Putative AraC-type regulatory protein | |
| yjhF | Member of Gnt transporter family | |
| ykgC | Putative oxidoreductase | |
| yrbK | Conserved hypothetical protein |
Genes are classified depending on their function; regulatable candidates are shown in boldface type.
Regulation of expression of the E. coli InsTetG−1 insertion mutants.
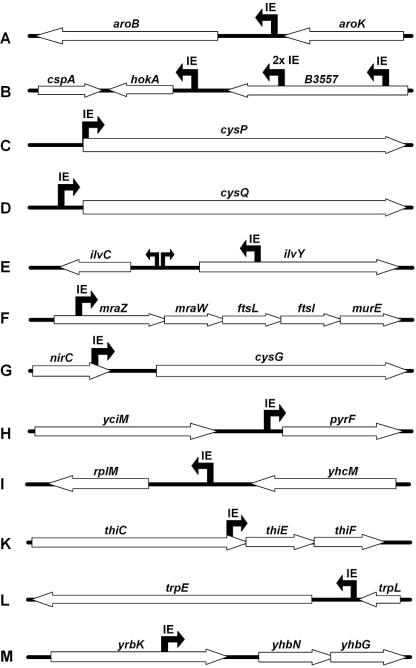
The 15 atc-regulatable mutants are summarized in Table 3 in alphabetical order of their affected genes. The two different phenotypes (regulated auxotrophy [regaux] and reverse regulated auxotrophy [revreg]) and the regulator type, TetR or revTetRr2, are depicted as well. They clearly demonstrate the advantages resulting from the variation of regulation for obtaining different types of mutants. Most notably, regulation by revTetRr2 opens the window for the direct detection of essential genes that reveal their auxotrophy only in the presence of atc. Until now, this was only possible by negative selection (18). The genes surrounding the insertion sites are depicted in Fig. 4.
TABLE 3.
All regulatable auxotrophic E. coli mutantsa
| Designation | Gene | Function | Phenotype | Regulator |
|---|---|---|---|---|
| A | aroB | 3-Dehydroquinate synthase | regaux | revTetRr2 |
| B1 | b3557 | IS5 protein | revreg | TetR |
| B2 | b3557 | IS5 protein | revreg | TetR |
| B3 | b3557 | IS5 protein | revreg | TetR |
| B4 | cspA | Intergenic region downstream of cspA ccw | revreg | TetR |
| C | cysP | Thiosulfate ABC transporter | regaux | TetR |
| D | cysQ | Involved in sulfite synthesis | regaux | TetR |
| E | ilvY | Transcriptional dual regulator | regaux | revTetRr2 |
| F | mraZ | Involved in cell wall synthesis and cell division | regaux | revTetRr2 |
| G | nirC | Nitrite FNT transporter | regaux | TetR |
| H | pyrF | Subunit of orotidine-5′-phosphate-decarboxylase | regaux | revTetRr2 |
| I | rplM | Upstream of rplM (product, 50S ribosomal subunit protein L13) | regaux | revTetRr2 |
| K | thiC | Thiamine biosynthesis | regaux | TetR |
| L | trpE/trpL | Subunit of anthranilate synthase | regaux | revTetRr2 |
| M | yrbK | Conserved hypothetical protein | regaux | revTetRr2 |
The involved genes are depicted with their functions. “regaux” indicates the regulatable auxotrophic phenotype, whereas “revreg” denotes the reverse regulatable phenotype. “revTetRr2” denotes the reverse TetRr2 variant present in the respective strains.
FIG. 4.
Schematic overview of all insertion sites of the regulatable E. coli candidates found in the screens. “IE” depicts the insertion site of the element. The black arrow represents the direction of the tetA promoter.
Eight mutants regulated by TetR were found. Four of them showed a normal phenotype, and four showed a reverse regulated phenotype in which the mutants cannot grow in the presence of atc in minimal medium. The reverse regulated mutants, designated B1 to B4, show insertion sites in the same orientation behind hokA (Fig. 4). This gene encodes a small toxic peptide and originates from plasmid R1, where the hok/sok locus mediates plasmid stabilization by the killing of plasmid-free cells. The hokA gene on the E. coli chromosome is not active due to an insertion of an IS150 element just upstream of the reading frame (27). We therefore assume that PA drives transcription of the toxin gene, causing cell death in the presence of atc. The observation that these mutants also die on rich medium with atc (data not shown) corroborates this interpretation. The other four TetR-regulated mutants, designated C, D, G, and K in Fig. 4 and Table 3, show a normal auxotrophic behavior due to mutations in biosynthetic pathways.
Seven revTetRr2-regulated mutants were found. Four of them, designated A, E, H, and L in Fig. 4 and in Table 2, contain insertions in genes encoding biosynthetic pathways. The E mutant shows an interesting genotype, with the promoter of the insertion element oriented towards ilvC (36). Thus, InsTetG−1 located in ilvY has replaced the regulator of ilvC, obviously making its expression dependent on the presence of atc. The other three mutants, designated F, I, and M, contain insertions in front of essential genes, as verified by their lack of growth on LB plates with atc. Mutant F contains InsTetG−1 in mraZ, the first gene in the dcw cluster. This operon is essential for cell wall synthesis and cell division (35). The I mutant contains the 50S ribosomal subunit protein L13 under atc control, and the M mutant contains InsTetG−1 at the 3′ end of yrbK, with PA directed towards yhbG and yhbN, encoding a putative ABC transporter for which an arabinose-regulated mutant with the same phenotype has been described previously (32). The yrbK knockout does not have a detectable phenotype on glucose minimal medium. Thus, InsTetG−1 seems to block transcription from an upstream promoter, while PA drives the transcription of the two ABC transporter-encoding genes yhbNG. This is an example of identifying unknown genes and enabling their direct analysis by regulated expression using InsTetG−1.
Application of InsTetG−1 to Salmonella serovar Typhimurium.
In order to analyze genes in Salmonella serovar Typhimurium, a well-understood pathogenic relative of E. coli, it was transformed with plasmid pWH1411BD or pWH1411r2. Mutant pools of Salmonella serovar Typhimurium were created with InsTetG−1 according the E. coli protocol described above, except that a fivefold-higher amount of InsTetG−1-transposase complexes had to be used for electroporation to obtain large pools of insertion mutants. One pool of about 156,000 candidates contained TetR-regulated mutants. The screening of 1,880 randomly chosen candidates on microtiter plates yielded 33 auxotrophic strains. Twenty-eight of those strains were confirmed on solid medium. Of these, 25 mutants exhibited constitutive auxotrophy, while 1 mutant displayed auxotrophy in the absence of atc and 2 mutants showed auxotrophy in the presence of atc. The screening of 1,600 randomly chosen candidates from another pool containing about 10,200 candidates yielded 15 auxotrophic strains. This rather low yield of auxotrophic mutants prompted the application of a more efficient screen. After an ampicillin enrichment of the pool containing 10,200 candidates, 700 candidates were screened. One hundred ninety-four of these were auxotrophs, and four showed auxotrophy in the absence of atc. Thirty-seven of these 198 mutants were sequenced, with the results displayed in Table 4.
TABLE 4.
Insertion mutants found in Salmonella serovar Typhimuriuma
| Gene | Function | Phenotype |
|---|---|---|
| argC | N-Acetyl-γ-glutamylphosphate reductase | aux |
| argE | Acetylornithine deacetylase | aux |
| argG | Argininosuccinate synthetase | aux |
| argI | Ornithine carbamoyltransferase 1 | aux |
| aroB | 3-Dehydroquinate synthase | aux |
| carB | Carbamoyl-phosphate synthase large subunit | aux |
| cysC | Adenylylsulfate kinase | aux |
| cysH | Phosphoadenosine phosphosulfate reductase | aux |
| cysI | Sulfite reductase α subunit | aux |
| cysJ | Sulfite reductase β subunit | aux |
| cysP | Thiosulfate transport protein | aux |
| cysW | Thiosulfate permease W protein | aux |
| glnA | Glutamine synthetase | aux |
| hisC | Histidinol-phosphate aminotransferase | aux |
| hisD | Histidinol dehydrogenase | aux |
| ilvA | Threonine dehydratase | aux |
| ilvC | Ketol acid reductoisomerase | aux |
| ilvD | Dihydroxy acid dehydratase | aux |
| ilvE | Branched-chain amino acid aminotransferase | aux |
| ilvY | Regulatory protein | aux |
| leuA | 2-Isopropylmalate synthase | aux |
| leuC | Isopropylmalate isomerase large subunit | aux |
| metA | Homoserine O-succinyltransferase | aux |
| metR | metE/metH regulator | regaux |
| nadB | l-Aspartate oxidase | aux |
| panC | Pantoate-β-alanine ligase | aux |
| ppc | Phosphoenolpyruvate carboxylase | aux |
| purC | Phosphoribosylaminoimidazole-succinocarboxamide synthase | aux |
| purD | Phosphoribosylglycinamide synthetase | aux |
| purF | Amidophosphoribosyltransferase | aux |
| purH | Bifunctional phosphoribosylamino- imidazolecarboxamide formyltransferase/ IMP cyclohydrolase | aux |
| pyrD | Dihydroorotate dehydrogenase | aux |
| pyrF | Orotidine 5′-phosphate decarboxylase | aux |
| recE | Exodeoxyribonuclease VIII | revreg |
| rfbI | CDP-6-deoxy-Δ-3,4-glucoseen reductase | aux |
| STM 4450 | Putative inner membrane protein | revreg |
| serA | d-3-Phosphoglycerate dehydrogenase | aux |
| serB | 3-Phosphoserine phosphatase | aux |
| serC | Phosphoserine aminotransferase | regaux |
| thrC | Threonine synthase | aux |
Regulatable candidates are shown in boldface type. The involved genes are depicted with their encoded functions. “aux” indicates a constitutive auxotrophic phenotype, and “regaux” denotes a regulatable auxotrophic phenotype, whereas “revreg” indicates the reverse regulatable phenotype. TetR was used as the regulator in the screen.
Ampicillin enrichment of intracellular growth-deficient Salmonella serovar Typhimurium mutants.
We next attempted to enrich growth-deficient mutants of Salmonella serovar Typhimurium with ampicillin in the murine macrophage-like cell line J774-A.1 using an approach similar to the ones previously described for Listeria monocytogenes (6) and Salmonella serovar Typhimurium (24). J774-A.1 cells were infected with the insertion mutant library (for details, see Materials and Methods). We found two different Salmonella serovar Typhimurium mutants that survived the ampicillin treatment in J774-A.1 cells. Mutant A has an InsTetG−1 insertion in pyrE, encoding orotate phosphoribosyltransferase, and mutant B has the insertion in argA, encoding N-acetylglutamate synthase. These two genes have not been specifically recognized as essential for intracellular growth previously, although pyrimidine auxotrophy leads to an intracellular growth deficiency (24).
Conclusion.
The constructed insertion element InsTetG−1 generates conditional knockout mutants in E. coli and Salmonella serovar Typhimurium that respond to the presence of atc. The use of revTetRr2 or TetR leads to the generation of mutants that can grow only in the presence or in the absence of atc. In principle, induction of transcription can lead to the induction of expression of a gene fused downstream of the PA promoter. However, it may also lead to repression of expression when the affected gene is silenced by antisense RNA. The yield of regulated mutants is expected to be much lower than that of obtaining unregulated auxotrophs, but the successful enrichment by counterselection, even in intracellularly growing Salmonella, should greatly facilitate the isolation of a sufficiently high number of regulated auxotrophs. Since the inducer is able to enter most bacterial cells without requiring an uptake protein, this insertion mutagenesis should be widely applicable, as has recently been demonstrated for Bacillus subtilis (2). Moreover, since atc and its analogs are not metabolized by higher organisms or bacteria, the regulation should also work fine in in vivo models of pathogenicity, like infected mice, which can be provided with atc in their drinking water or by infusion. Thus, this approach should be a useful tool for the functional analysis of known genes under various conditions or for elucidating the function of unknown genes.
Acknowledgments
We thank Chris Berens for fruitful discussions and critical reading of the manuscript.
This work was supported by the Deutsche Forschungsgemeinschaft through SFB473 and GRK805 and the Fonds der Chemischen Industrie.
REFERENCES
- 1.Bender, J., and N. Kleckner. 1992. Tn10 insertion specificity is strongly dependent upon sequences immediately adjacent to the target-site consensus sequence. Proc. Natl. Acad. Sci. USA 89:7996-8000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bertram, R., M. Köstner, J. Müller, J. Vazquez Ramos, and W. Hillen. 2005. Integrative elements for Bacillus subtilis yielding tetracycline-dependent growth phenotypes. Nucleic Acids Res. 33:e153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bertram, R., C. Kraft, S. Wisshak, J. Mueller, O. Scholz, and W. Hillen. 2004. Phenotypes of combined tet repressor mutants for effector and operator recognition and allostery. J. Mol. Microbiol. Biotechnol. 8:104-110. [DOI] [PubMed] [Google Scholar]
- 4.Bethesda Research Laboratories. 1986. BRL pUC host: E. coli DH5α competent cells. Bethesda Res. Lab. Focus 8:9. [Google Scholar]
- 5.Blattner, F. R., G. Plunkett III, C. A. Bloch, N. T. Perna, V. Burland, M. Riley, J. Collado-Vides, J. D. Glasner, C. K. Rode, G. F. Mayhew, J. Gregor, N. W. Davis, H. A. Kirkpatrick, M. A. Goeden, D. J. Rose, B. Mau, and Y. Shao. 1997. The complete genome sequence of Escherichia coli K-12. Science 277:1453-1474. [DOI] [PubMed] [Google Scholar]
- 6.Camilli, A., C. R. Paynton, and D. A. Portnoy. 1989. Intracellular methicillin selection of Listeria monocytogenes mutants unable to replicate in a macrophage cell line. Proc. Natl. Acad. Sci. USA 86:5522-5526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chong, S., F. B. Mersha, D. G. Comb, M. E. Scott, D. Landry, L. M. Vence, F. B. Perler, J. Benner, R. B. Kucera, C. A. Hirvonen, J. J. Pelletier, H. Paulus, and M. Q. Xu. 1997. Single-column purification of free recombinant proteins using a self-cleavable affinity tag derived from a protein splicing element. Gene 192:271-281. [DOI] [PubMed] [Google Scholar]
- 8.Chow, W. Y., and D. E. Berg. 1988. Tn5tac1, a derivative of transposon Tn5 that generates conditional mutations. Proc. Natl. Acad. Sci. USA 85:6468-6472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Eckert, B., and C. F. Beck. 1989. Overproduction of transposon Tn10-encoded tetracycline resistance protein results in cell death and loss of membrane potential. J. Bacteriol. 171:3557-3559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Goryshin, I. Y., J. Jendrisak, L. M. Hoffman, R. Meis, and W. S. Reznikoff. 2000. Insertional transposon mutagenesis by electroporation of released Tn5 transposition complexes. Nat. Biotechnol. 18:97-100. [DOI] [PubMed] [Google Scholar]
- 11.Goryshin, I. Y., J. A. Miller, Y. V. Kil, V. A. Lanzov, and W. S. Reznikoff. 1998. Tn5/IS50 target recognition. Proc. Natl. Acad. Sci. USA 95:10716-10721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Goryshin, I. Y., and W. S. Reznikoff. 1998. Tn5 in vitro transposition. J. Biol. Chem. 273:7367-7374. [DOI] [PubMed] [Google Scholar]
- 13.Gossen, M., and H. Bujard. 2002. Studying gene function in eukaryotes by conditional gene inactivation. Annu. Rev. Genet. 36:153-173. [DOI] [PubMed] [Google Scholar]
- 14.Guerout-Fleury, A. M., K. Shazand, N. Frandsen, and P. Stragier. 1995. Antibiotic-resistance cassettes for Bacillus subtilis. Gene 167:335-336. [DOI] [PubMed] [Google Scholar]
- 15.Gülland, U., and W. Hillen. 1992. The Tn10-encoded tetR mRNA has heterogeneous 5′ ends in vivo and in vitro. Gene 114:97-101. [DOI] [PubMed] [Google Scholar]
- 16.Hare, R. S., S. S. Walker, T. E. Dorman, J. R. Greene, L. M. Guzman, T. J. Kenney, M. C. Sulavik, K. Baradaran, C. Houseweart, H. Yu, Z. Foldes, A. Motzer, M. Walbridge, G. H. Shimer, Jr., and K. J. Shaw. 2001. Genetic footprinting in bacteria. J. Bacteriol. 183:1694-1706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hayes, F. 2003. Transposon-based strategies for microbial functional genomics and proteomics. Annu. Rev. Genet. 37:3-29. [DOI] [PubMed] [Google Scholar]
- 18.Hensel, M., J. E. Shea, C. Gleeson, M. D. Jones, E. Dalton, and D. W. Holden. 1995. Simultaneous identification of bacterial virulence genes by negative selection. Science 269:400-403. [DOI] [PubMed] [Google Scholar]
- 19.Henssler, E. M., R. Bertram, S. Wisshak, and W. Hillen. 2005. Tet repressor mutants with altered effector binding and allostery. FEBS J. 272:4487-4496. [DOI] [PubMed] [Google Scholar]
- 20.Hidalgo, A. A., A. N. Trombert, J. C. Castro-Alonso, C. A. Santiviago, B. R. Tesser, P. Youderian, and G. C. Mora. 2004. Insertions of mini-Tn10 transposon T-POP in Salmonella enterica sv. Typhi. Genetics 167:1069-1077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Karlinsey, J. E., S. Tanaka, V. Bettenworth, S. Yamaguchi, W. Boos, S. I. Aizawa, and K. T. Hughes. 2000. Completion of the hook-basal body complex of the Salmonella typhimurium flagellum is coupled to FlgM secretion and fliC transcription. Mol. Microbiol. 37:1220-1231. [DOI] [PubMed] [Google Scholar]
- 22.Kofoid, E., C. Rappleye, I. Stojiljkovic, and J. Roth. 1999. The 17-gene ethanolamine (eut) operon of Salmonella typhimurium encodes five homologues of carboxysome shell proteins. J. Bacteriol. 181:5317-5329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Krueger, C., K. Pfleiderer, W. Hillen, and C. Berens. 2004. Tetracycline derivatives: alternative effectors for Tet transregulators. BioTechniques 37:546, 548, 550. [DOI] [PubMed] [Google Scholar]
- 24.Leung, K. Y., and B. B. Finlay. 1991. Intracellular replication is essential for the virulence of Salmonella typhimurium. Proc. Natl. Acad. Sci. USA 88:11470-11474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Miller, J. H. 1992. A short course in bacterial genetics: laboratory manual, p. 72-74. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.
- 26.Moir, D. T., K. J. Shaw, R. S. Hare, and G. F. Vovis. 1999. Genomics and antimicrobial drug discovery. Antimicrob. Agents Chemother. 43:439-446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pedersen, K., and K. Gerdes. 1999. Multiple hok genes on the chromosome of Escherichia coli. Mol. Microbiol. 32:1090-1102. [DOI] [PubMed] [Google Scholar]
- 28.Rappleye, C. A., and J. R. Roth. 1997. A Tn10 derivative (T-POP) for isolation of insertions with conditional (tetracycline-dependent) phenotypes. J. Bacteriol. 179:5827-5834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schneider, K., and C. F. Beck. 1986. Promoter-probe vectors for the analysis of divergently arranged promoters. Gene 42:37-48. [DOI] [PubMed] [Google Scholar]
- 30.Scholz, O., E. M. Henssler, J. Bail, P. Schubert, J. Bogdanska-Urbaniak, S. Sopp, M. Reich, S. Wisshak, M. Köstner, R. Bertram, and W. Hillen. 2004. Activity reversal of Tet repressor caused by single amino acid exchanges. Mol. Microbiol. 53:777-789. [DOI] [PubMed] [Google Scholar]
- 31.Scholz, O., M. Köstner, M. Reich, S. Gastiger, and W. Hillen. 2003. Teaching TetR to recognize a new inducer. J. Mol. Biol. 329:217-227. [DOI] [PubMed] [Google Scholar]
- 32.Serina, S., F. Nozza, G. Nicastro, F. Faggioni, H. Mottl, G. Deho, and A. Polissi. 2004. Scanning the Escherichia coli chromosome by random transposon mutagenesis and multiple phenotypic screening. Res. Microbiol. 155:692-701. [DOI] [PubMed] [Google Scholar]
- 33.Sigler, A., P. Schubert, W. Hillen, and M. Niederweis. 2000. Permeation of tetracyclines through membranes of liposomes and Escherichia coli. Eur. J. Biochem. 267:527-534. [DOI] [PubMed] [Google Scholar]
- 34.Thanassi, D. G., G. S. Suh, and H. Nikaido. 1995. Role of outer membrane barrier in efflux-mediated tetracycline resistance of Escherichia coli. J. Bacteriol. 177:998-1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vicente, M., M. J. Gomez, and J. A. Ayala. 1998. Regulation of transcription of cell division genes in the Escherichia coli dcw cluster. Cell. Mol. Life Sci. 54:317-324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wek, R. C., and G. W. Hatfield. 1988. Transcriptional activation at adjacent operators in the divergent-overlapping ilvY and ilvC promoters of Escherichia coli. J. Mol. Biol. 203:643-663. [DOI] [PubMed] [Google Scholar]
- 37.Yanisch-Perron, C., J. Vieira, and J. Messing. 1985. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33:103-119. [DOI] [PubMed] [Google Scholar]
- 38.Zhu, Z., T. Zheng, C. G. Lee, R. J. Homer, and J. A. Elias. 2002. Tetracycline-controlled transcriptional regulation systems: advances and application in transgenic animal modeling. Semin. Cell Dev. Biol. 13:121-128. [DOI] [PubMed] [Google Scholar]