Abstract
Blastocystis is an enteric protozoan purportedly associated with numerous clinical cases of diarrhea, flatulence, vomiting, and other gastrointestinal symptoms. Despite new knowledge of Blastocystis cell biology, genetic diversity, and epidemiology, its pathogenic potential remains controversial. Numerous clinical and epidemiological studies either implicate or exonerate the parasite as a cause of intestinal disease. Therefore, the aim of this study was to investigate the pathogenic potential of Blastocystis by studying the interactions of Blastocystis ratti WR1, an isolate of zoonotic potential, with a nontransformed rat intestinal epithelial cell line, IEC-6. Here, we report that B. ratti WR1 induces apoptosis in IEC-6 cells in a contact-independent manner. Furthermore, we found that B. ratti WR1 rearranges F-actin distribution, decreases transepithelial resistance, and increases epithelial permeability in IEC-6 cell monolayers. In addition, we found that the effects of B. ratti on transepithelial electrical resistance and epithelial permeability were significantly abrogated by treatment with metronidazole, an antiprotozoal drug. Our results suggest for the first time that Blastocystis-induced apoptosis in host cells and altered epithelial barrier function might play an important role in the pathogenesis of Blastocystis infections and that metronidazole has therapeutic potential in alleviating symptoms associated with Blastocystis.
Blastocystis is an intestinal protozoan parasite found in humans and other mammals, birds, reptiles, and insects. Blastocystis is frequently reported in human fecal samples of symptomatic patients and healthy individuals (43, 44, 49, 51) with the prevalence of Blastocystis infections in developing countries ranging from 30 to 50% (2, 16, 30, 43). Since its description in the early 1900s, our knowledge of its cell biology, genetic diversity, and epidemiology has advanced significantly (1, 43, 44). There are many reports associating Blastocystis with gastrointestinal symptoms, including recurrent watery diarrhea, mucous diarrhea, abdominal cramps, vomiting, and bloating (13, 33, 38, 44). Accumulating phylogenetic data suggest that Blastocystis is a zoonotic organism, and it is believed that the transmission of animal infections to humans is common (34, 50). Various studies suggested that immunocompromised patients are more likely to suffer Blastocystis-related diarrheal diseases (6, 35). Despite its description about a century ago, the exact role of Blastocystis as a cause of human disease is controversial and inconclusive. Some clinical and epidemiological studies implicate the parasite as a potential pathogen, while others exonerate the organism as a cause of intestinal disease (23).
The primary site for Blastocystis colonization is the intestine, and it has been suggested that proteases produced by Blastocystis help in colonization and pathogenesis of this parasite (37, 42). In vitro studies have demonstrated that Blastocystis is capable of causing significant cytopathic effects on Chinese hamster ovary cells (48). Recently, a variety of intestinal pathogens were found to induce apoptosis in intestinal epithelial cells, and their association with pathogenicity was suggested (11, 14, 20, 22, 29, 46). It has been suggested that epithelial cell apoptosis caused by the protozoan Giardia lamblia can contribute to the intestinal permeability disorders (9).
Intestinal permeability was reported to be considerably increased in Blastocystis hominis-infected patients, and it was suggested that Blastocystis infections may damage the intestinal wall (12). Many gastrointestinal disorders, such as bacterial enteritis, celiac disease, and inflammatory bowel disease, are reported to be associated with a breakdown of epithelial barrier function (4, 9).
Intestinal barrier function is mainly regulated by tight junctions, which consist of a number of transmembrane proteins, such as claudins and occludin, and cytoplasmic peripheral membrane proteins, including zonula-occludens-1, -2, and -3 and cingulin (10). Many tight-junction proteins interact with F-actin, and these interconnections are considered to stabilize the tight junction. Intestinal pathogens, such as Giardia lamblia and Escherichia coli, were reported to rearrange F-actin distribution in intestinal epithelial cells and compromise barrier function (15, 45).
In this study, we aim to investigate the interactions of Blastocystis ratti WR1 using a nontransformed rat intestinal epithelial cell line model. We demonstrate for the first time that Blastocystis can induce apoptosis, rearrange F-actin distribution, decrease transepithelial electrical resistance (TER), and increase epithelial permeability. Additionally, our results show that the antiprotozoal drug metronidazole abrogates the effects of B. ratti WR1 on epithelial barrier function, and this suggests that it has therapeutic potential in Blastocystis infections.
MATERIALS AND METHODS
Culture of nontransformed rat intestinal cell line.
All experiments were performed with a nontransformed rat intestinal epithelial cell line, IEC-6 (ATCC). IEC-6 cell stock was maintained in T-75 flasks in a humidified 37°C incubator with 5% CO2, and passages 5 to 15 were used for all experiments. The growth medium consisted of Dulbecco's modified Eagle's medium (Sigma) with 10% heat-inactivated fetal bovine serum (HyClone), 1% sodium pyruvate (Gibco), and 0.1% bovine insulin (Sigma). The culture medium was replenished every 2 to 3 days. Cell viability was analyzed in a trypan blue assay, and cell cultures with >95% viability were used. Cells were trypsinized with 0.25% trypsin-EDTA (Gibco), and a seeding density of 2 × 105 cells/ml was used. Cells were grown to confluence on 12-well tissue culture plates (Costar) or on poly-l-lysine-treated 12-mm glass coverslips. For transepithelial resistance and monolayer permeability assays, cells were grown on 0.6-cm2 Millicell-HA filters using 24-well tissue culture plates (Costar).
Parasite culture and preparation of lysate.
Blastocystis ratti isolate WR1 was originally isolated from rats housed in the National University of Singapore's Animal Holding Unit and subsequently axenized (8). Parasites were cultured in prereduced Iscove's modified Dulbecco's medium supplemented with 10% inactivated horse serum (Gibco) and incubated anaerobically at 37°C in an ANAEROJAR (Oxoid, United Kingdom) as previously described (18). Five-day-old parasites at log phase were washed two times in ice-cold IEC-6 complete medium at 500 × g for 10 min at 4°C. The pellet was resuspended in IEC-6 cell complete medium, parasites were counted with a hemocytometer, and the concentration was adjusted to 1 × 107 parasites/ml. Parasites were examined microscopically for their viability in IEC-6 complete medium and found to be viable for >48 h in IEC-6 growth conditions (data not shown). Parasitic lysates were prepared by three freeze-thaw cycles in liquid nitrogen and a 37°C water bath.
Inoculation protocol and experimental planning.
For all experiments, a density of 1 × 107 parasites/ml was used. Either live parasites or parasitic lysate was added to 12-well plates (2 ml) or 24-well plates (1 ml) or to the apical side of confluent monolayers grown on 0.6-cm2 Millicell-HA filters (400 μl). For apoptosis experiments and phalloidin-fluorescein isothiocyanate (FITC) staining, 2- or 3-day-old confluent monolayers were used. For TER and epithelial permeability experiments, monolayers grown on Millicell-HA filters were used 5 or 6 days after they were seeded when they reached peak electrical resistance of ∼30 Ω/cm2 (data not shown). In caspase inhibition experiments, cell monolayers were pretreated for 2 h with either general caspase inhibitor Z-VAD-fmk (50 μM) or caspase-3 inhibitor Z-DEVD-fmk (120 μM) (where Z is benzyloxycarbonyl group, fmk is fluoromethyl ketone, VAD is Val-Ala-Asp, and DEVD is Asp-Glu-Val-Asp) prior to the addition of parasites or lysate. In some TER and permeability assays, the antiprotozoal drug metronidazole (10 μg/ml, Sigma) was added in medium prior to the addition of live parasites. Monolayers were washed twice with cold Hanks balanced salt solution (HBSS; Gibco) before the addition of parasite or lysate suspension. In order to test whether apoptosis of IEC-6 cells requires direct contact with parasites, cells were grown in 12-well plates and kept separate from parasite by porous filters (Millicell-HA filter) during coincubation.
Apoptosis assays.
Various assays, as described below, were employed to analyze apoptosis of IEC-6 cells. Staurosporin (Sigma) (0.25 μM) was used to induce apoptosis as a positive control.
(i) DNA binding dye.
IEC-6 cell monolayers grown on 12-mm glass coverslips were coincubated with B. ratti WR1 live parasites and parasitic lysate for 12 h. After incubation, monolayers were washed with phosphate-buffered saline (PBS) and fixed with 2% (wt/vol) paraformaldehyde in PBS (pH 7.4). The cells were then washed twice with PBS and stained with 0.5 μg/ml of DAPI (4′,6-diamidino-2-phenylindole; Sigma) for 5 min at room temperature in the dark. Then, cells were washed in PBS and mounted on a glass slide with fluorescence mounting medium (VECTASHIELD). Cells were observed with a fluorescence microscope (Olympus BX60; Olympus, Japan).
(ii) Assay for expression of phosphatidylserine on the cell surface.
Annexin V binding assay was performed as an early marker of apoptosis. IEC-6 cell monolayers were grown in 12-well culture plates and then coincubated with B. ratti WR1 live parasites and parasitic lysate for 5 h. Plasma membrane asymmetry was measured by using an annexin V-FITC apoptosis detection kit (BD Pharmingen) following the manufacturer's instructions. Propidium iodide (PI) was used to exclude dead cells from apoptotic cells. After the cells were stained, samples were analyzed by flow cytometry (Dako Cytomation Cyan LX) using a 488-nm excitation wavelength, a 515-nm band-pass filter for fluorescein detection, and a 600-nm filter for PI detection. A region was defined to represent the apoptotic cells showing annexin-positive and PI-negative staining (lower right quadrant).
(iii) TUNEL.
IEC-6 cells grown on poly-l-lysine-coated 12-mm glass coverslips were used for fluorescent microscopic analysis, and cells grown in six-well culture plates were used for flow cytometric analysis. Cells were coincubated with B. ratti WR1 live parasites and lysate for 12 h. Terminal deoxynucleotidyltransferase-mediated dUTP nick end labeling (TUNEL) was performed using the in situ cell death detection kit (Roche) following the manufacturer's instructions for the detection of in situ DNA fragmentation. The samples were viewed with a fluorescence microscope or analyzed by flow cytometry using a 525-nm band-pass filter to detect green fluorescence.
(iv) Evaluation of caspase-3 activity.
Caspase-3 activity was measured using ApoAlert caspase fluorescent assay kit (BD Pharmingen) after infection for 6, 12, and 24 h. Briefly, after coincubation, cells were washed twice with PBS and harvested using a cell scraper. Cells were then resuspended in 50 μl of chilled cell lysis buffer (component no. 6636KC; BD Pharmingen) and incubated on ice for 10 min. Cell lysates were centrifuged at maximum speed for 10 min to precipitate cellular debris. The supernatants were transferred to new microcentrifuge tubes, and then 50 μl of 10 mM dithiothreitol in 1 ml of 2× reaction buffer mix followed by 5 μl of 1 mM caspase-3 substrate (DEVD-7-amino-4-trifluoromethylcoumarin; 50 μM final concentration) was added to each sample and incubated for 1 h at 37°C in a water bath. Samples were transferred to a 96-well plate, and absorbance was measured in a fluorometer (SpectraMax) with a 400-nm excitation filter and 505-nm emission filter. Caspase-3 activity was expressed as relative fluorescence units.
Phalloidin-FITC staining of F-actin.
IEC-6 cells were grown on poly-l-lysine-coated glass coverslips, and confluent monolayers were coincubated with live parasites, parasitic lysate, or 5 μg/ml cholera toxin as positive control for 24 h. After incubation, cells were washed with PBS and fixed in 3.7% paraformaldehyde in PBS for 5 min. Cells were washed with PBS, dehydrated with acetone, permeabilized with 0.1% Triton X-100 in PBS, and washed again in PBS. Cells were stained with 50 μg/ml phalloidin-FITC conjugate solution in PBS for 40 min at room temperature and then washed five or six times with PBS to remove unbound phalloidin conjugate. Coverslips were mounted on glass slides with fluorescence mounting medium and viewed by confocal microscopy (Olympus Fluoview FV500).
Measurement of transepithelial resistance.
Transepithelial resistance was determined with a Millipore electrical resistance system (Millipore-ERS). IEC-6 cell monolayers were grown on 12-mm Millicell-HA filters, and TER was tested on alternate days until it peaked (∼30 Ω/cm2) by day 6. Parasites or lysate was added to the apical side of confluent monolayers grown on Millicell-HA filters and coincubated for 12, 24, and 48 h. After coincubation, the monolayers were washed two times with HBSS to ensure that the parasites would not affect TER measurements. Four hundred microliters of warm (37°C) complete medium was added to the apical compartment, and TER was measured with Millipore-ERS. To avoid fluctuations in temperature, TER was measured at 37°C with the aid of a heating plate. Epithelial resistance was expressed in Ω/cm2.
Determination of epithelial permeability by Lucifer yellow.
To confirm confluence of cell monolayers grown on Millicell-HA filters, transepithelial resistance was measured with Millipore-ERS. After reaching peak transepithelial resistance (∼30 Ω/cm2), the effect of Blastocystis on epithelial barrier function was assessed with Lucifer yellow (molecular weight of 482; Molecular Probes). Briefly, after 24 h of coincubation, apical and basolateral compartments were washed gently three times with HBSS. Three hundred microliters of Lucifer yellow (100 μg/ml in HBSS) was added to the apical compartment, and 500 μl of HBSS was added to the basolateral compartment. After 3 h of incubation at 37°C in a humidified incubator with 5% CO2, 250 μl of sample was taken from the basolateral compartment. Fluorescence absorbance was determined using wavelengths of 485 nm (excitation) and 535 nm (emission). Values were expressed as a percentage of Lucifer yellow passage across the cell monolayer.
Statistical analysis.
The mean values of experimental groups were compared by using the paired Student t test. Differences were considered significant if P was <0.05.
RESULTS
Blastocystis ratti WR1 live parasites and parasitic lysates induce IEC-6 cell apoptosis.
We employed a series of experiments to assess the effects of Blastocystis on IEC-6 cell apoptosis. These included (i) DAPI staining, which detects chromatin condensation and nuclear fragmentation; (ii) annexin V-FITC binding assay to detect externalization of phosphatidylserine during the early phase of apoptosis; (iii) TUNEL assay for the detection of internucleosomal cleavage of DNA during the late stages of apoptosis, and (iv) caspase-3 fluorescence assays that detect the presence of active caspase-3. Results of all assays consistently showed that coincubation of IEC-6 cells with B. ratti WR1 live parasites and parasitic lysates induces apoptosis of IEC-6 cells.
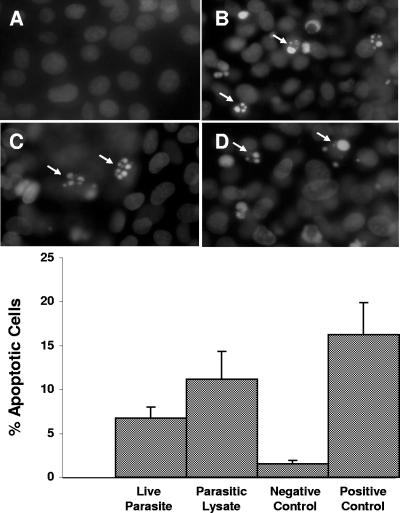
Apoptosis was originally defined on the basis of a distinctive pattern of morphological features seen in the nucleus as the cell dies, features observable by light or fluorescence microscopy (21). DAPI staining, which detects changes in the nucleus, provides the first unequivocal evidence of the onset of apoptosis. Nuclear morphological characteristics of infected IEC-6 cells showed typical apoptotic nuclear condensation and fragmentation when stained with DAPI (Fig. 1). IEC-6 cells cocultured with live parasites and parasitic lysates for 12 h showed shrunken, highly condensed, and fragmented nuclei in apoptotic cells (Fig. 1B and C). Control IEC-6 cells exhibited regularly shaped intact nuclei with normal chromatin distribution (Fig. 1A). Cells incubated with live parasites (6.7% ± 1.3%; P < 0.05) and parasitic lysate (11.2% ± 3.1%; P < 0.05) showed significant increases of four- and sevenfold in the percentage of apoptotic cells compared with the value for the control (1.5% ± 0.3%), respectively.
FIG. 1.
Fluorescence photomicrographs and histograms obtained from DAPI fluorescence assay for apoptosis of nontransformed rat intestinal epithelial IEC-6 cells. Cell monolayers were grown on glass coverslips and incubated for 24 h with either growth medium (A), B. ratti WR1 live parasites (B), parasitic lysate (C), and 0.25 μM staurosporin as a positive control (D). Cells coincubated with live parasites, parasitic lysate, and staurosporin show nuclear condensation and fragmentation (B, C, and D). From the histogram, a significant increase in the percentage of apoptotic cells in monolayers coincubated with live parasites and lysate can be noticed in comparison to the negative control. For each sample, 1,000 cells were counted under a magnification of ×40. Values are means ± standard deviations (error bars) (three monolayers in each group). The values were significantly different (P < 0.05) from the value for the negative control.
The flipping of phosphatidylserine molecules from the inner to outer leaflet of the plasma membrane is one of the key early indications of apoptosis. Annexin V, a 35- to 36-kDa molecule, specifically binds, in the presence of calcium ions, with high affinity to phosphatidylserine molecules (39). Using annexin V as a FITC conjugate in combination with propidium iodide, one can distinguish between viable, apoptotic, and necrotic cells. This combination appears to be a potent discriminator between necrotic and apoptotic cells (47).
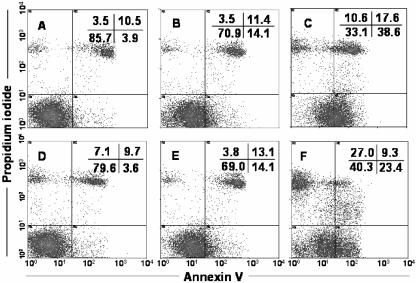
Apoptotic cells showed annexin-positive and PI-negative staining (lower right quadrants in Fig. 2A to F) populations in flow cytometry dot plots. IEC-6 cells cocultured with Blastocystis for 5 h showed a marked increase in the percentage of apoptotic cells coincubated with live parasites (14.1% [Fig. 2B]) and with parasitic lysate (38.6% [Fig. 2C]) relative to the control (3.9% [Fig. 2A]) in the lower right quadrants. The dot plots in Fig. 2B, C, E, and F compared to that in Fig. 2A show an overall shift in the population of cells from the lower left quadrant towards the lower right quadrant, which indicates the percentage of apoptotic cells with preservation of the integrity of the plasma membrane. Pretreatment with the general caspase inhibitor Z-VAD-fmk prior to coincubation with parasites significantly prevented the apoptosis of IEC-6 cells, bringing the percentage of annexin-positive and PI-negative cells close to that of the negative control (3.6% [Fig. 2D]). To investigate contact-independent cell death induction due to parasite secretory factors, we used Millicell-HA filters to separate live parasites from host cells. There was a significant increase in the number of apoptotic cells induced by live parasites in a contact-independent manner when these filters were used (14.1% [Fig. 2E]). This finding suggests that B. ratti WR1 secretes some parasitic factors that may induce apoptosis of IEC-6 cells without the need for direct contact. All the dot plots show consistent percentages of a baseline population of necrotic cells in the upper right quadrants, but cells incubated with parasitic lysate showed an increase in the necrotic cell population. Parasitic lysate consists of all secretory and nonsecretory cellular components of the parasite. This increase in necrotic population (annexin positive and PI positive) in cells incubated with parasitic lysate may be due to the effects of some intracellular nonsecretory Blastocystis products, such as proteases present only in parasitic lysates (Fig. 2C).
FIG. 2.
Annexin V-FITC staining of IEC-6 cells by flow cytometry. Representative dot plots of cells subjected to different treatments are shown. Untreated IEC-6 cells (A) are shown as a control. IEC-6 cells were incubated for 5 h with B. ratti WR1 live parasites (B), parasitic lysate (C), live parasites after pretreatment with caspase inhibitor Z-VAD-fmk (D), live parasites on Millicell-HA filter for contact-independent assay (E), and 0.25 μM staurosporin as a positive control (F). Panels B, C, E, and F show significant increases in annexin V-positive apoptotic cell population (lower right quadrants). A total of 2 × 104 cells from each sample were analyzed. The upper right quadrant represents apoptotic or necrotic cells positive for both annexin V and PI. The lower left quadrant represents healthy cells negative for annexin V and PI staining, and the upper left quadrant represents necrotic cells positive for only PI. Results are representative from two experiments, and the values were significantly different (P < 0.05) from the value for the negative control for all samples.
We employed TUNEL as a late marker for the detection of endonuclease activity and quantification of apoptosis in IEC-6 cells. Endonucleolysis is an important biochemical hallmark during apoptosis, and TUNEL is considered to be a sensitive method for detecting DNA fragmentation when used in conjunction with alternative techniques (19). TUNEL relies on the specific binding of a labeled polynucleotide molecule (dUTP) to the exposed 3′-OH ends of DNA, catalyzed by exogenous terminal deoxynucleotidyltransferase.
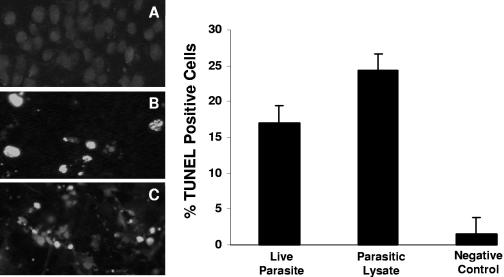
Fluorescence microscopy results showed maximal labeling of distinctly fragmented DNA (Fig. 3). The intensity of FITC staining in IEC-6 cells coincubated with live parasites (Fig. 3B) and parasitic lysate (Fig. 3C) samples showed marked increases in fluorescence compared to that of the negative control (Fig. 3A). Flow cytometric results (Fig. 3) revealed significantly higher percentages of TUNEL-labeled cells for cells incubated with live parasites (17% ± 2.3%) and parasitic lysate (24.3% ± 2.9%) compared to that of the negative control (2.4% ± 1.5%) (P < 0.05 for both in comparison to the control). Taken together, TUNEL and DAPI staining results clearly indicate that Blastocystis causes extensive DNA fragmentation in IEC-6 cells.
FIG. 3.
TUNEL for in situ DNA fragmentation of IEC-6 cells. Fluorescence micrographs of cells grown on glass coverslips and coincubated for 12 h with growth medium (A), B. ratti WR1 live parasites (B), and parasitic lysate (C). A significant number of TUNEL-positive cells (green fluorescence) can be seen in panels B and C. (Right) Histogram of TUNEL-positive cell population determined by flow cytometry shows a significant increase in TUNEL-positive IEC-6 cells after coincubation with live parasites and parasitic lysates. Values are means ± standard deviations (error bars) (two sets of cells grown on coverslips per group). The values were significantly different (P < 0.05) from the value for the negative control.
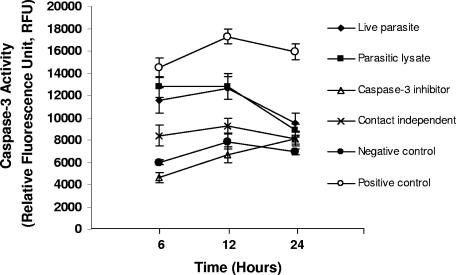
Caspases are the central components and executioners of apoptosis (40), and they control the dismantling and clearance of dying cells. Caspase-3 is an active cell death protease involved in the execution phase of apoptosis, during which cells undergo morphological changes, such as DNA fragmentation, chromatin condensation, and apoptotic body formation (36). Caspase-3 activity was found to be significantly increased in IEC-6 cells treated with B. ratti WR1 live parasites and parasitic lysates (Fig. 4). Caspase-3 activity peaked at 12 h of incubation with and without direct contact of live parasites, after which activity decreased for up to 24 h. This was also observed in cells incubated with parasitic lysates. Interestingly, caspase-3 activity was found to be high in IEC-6 cells coincubated with live parasites in a contact-independent manner, using Millicell-HA filters. This result was consistent with annexin V binding results (Fig. 2) and therefore provides supporting evidence that B. ratti WR1 can induce apoptosis of IEC-6 cells in a contact-independent manner. In addition, to provide direct evidence that apoptosis of IEC-6 cells by Blastocystis involved caspase-3, IEC-6 monolayers were pretreated with caspase-3-specific inhibitor, Z-DEVD-fmk, before coincubation with parasites. Caspase-3 activity was found to be completely blocked with the use of Z-DEVD-fmk (Fig. 4). Results suggest active involvement of caspase-3 in Blastocystis-induced apoptosis of IEC-6 cells.
FIG. 4.
Effect of B. ratti WR1 on caspase-3 activity of IEC-6 cells. Caspase-3 activity was measured at the indicated time points as described in Materials and Methods. A significant increase in caspase-3 activity was observed in cells treated with live parasites, parasitic lysates, and positive control (0.25 μM staurosporin) after 6 and 12 h. Caspase-3 activity was gradually reduced at 24 h. Pretreatment of cells with caspase-3 inhibitor Z-DEVD-fmk reduced caspase-3 activity significantly. Values are means ± standard deviations (error bars) (n = 3 per group). The values were significantly different (P < 0.05) from the value for the negative control.
Rearrangement of F-actin.
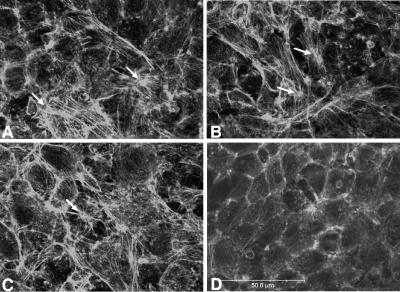
The cytoskeleton, in particular the actin microfilaments, is key in maintaining cell shape and regulating tight junction permeability. The organization of actin filaments is crucial for the assembly of intestinal tight junctions (27). Phalloidin, a fungal toxin, binds to F-actin with high affinity and stabilizes it. We used phalloidin-FITC conjugate to detect changes in F-actin distribution. When IEC-6 cell monolayers were coincubated with B. ratti WR1 live parasites, elongation of cortical actin filaments, actin disruption, and formation of stress fibers in the cytoplasmic zone were noticed (Fig. 5A). A more pronounced formation of stress fibers and actin condensation was noticed in the monolayers incubated with parasitic lysate and cholera toxin (positive control) (Fig. 5B and C). IEC-6 cell monolayers in the control showed normal actin distribution in the cortex zone of cells, with negligible stress fiber formation in the cytoplasmic area (Fig. 5D). IEC-6 cells incubated with live parasites and parasitic lysate for 24 h showed significantly higher percentages of cells showing stress fiber formation (34.3% and 36.4%) in comparison to that of the control (5.7%) (Table 1). The percentage of cells showing stress fiber formation after 48 h of coincubation was slightly higher than that after 24 h of coincubation (Table 1).
FIG. 5.
Effect of B. ratti WR1 exposure on actin cytoskeleton. IEC-6 cells were stained with fluorescein-phalloidin and analyzed by confocal microscopy. Prominent formation of stress fibers (white arrows) can be noticed in monolayers coincubated with B. ratti WR1 live parasites (A), parasitic lysate (B), and 5 μg/ml cholera toxin (C) as a positive control. Negative control (D) showing normal distribution of F-actin in the cortex zone of cells. Bar, 50 μm.
TABLE 1.
IEC-6 cells showing stress fiber formation in response to Blastocystis ratti WR1 infectiona
| Period of incubation (h) | % of IEC-6 cells showing stress fiber formation
|
|||
|---|---|---|---|---|
| Cells coincubated with WR1 live parasite | Cells coincubated with parasitic lysate | Negative control | Cells coincubated with 5 μg/ml cholera toxin (positive control) | |
| 24 | 34.3 | 36.4 | 5.7 | 42.8 |
| 48 | 37.2 | 45.7 | 7.8 | 60.7 |
A total of 500 cells were counted for each sample.
Decrease in transepithelial resistance.
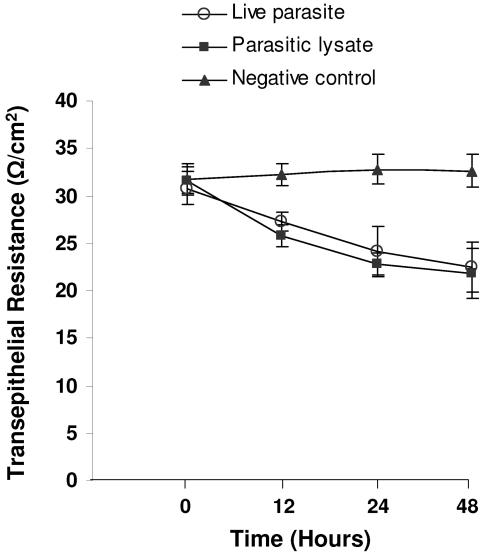
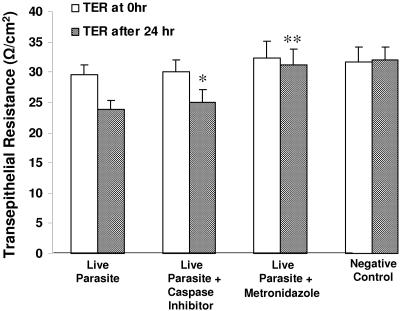
F-actin rearrangement has been associated with reduced TER in a number of studies (17, 45). Hence, we conducted experiments to investigate whether rearrangement of F-actin induced by Blastocystis is associated with changes in TER. IEC-6 cell monolayers achieved a peak average resistance of ∼30 Ω/cm2 after about 6 days after seeding. B. ratti WR1 caused a significant decrease in the transepithelial resistance of IEC-6 cell monolayers in a time-dependent manner (Fig. 6). The epithelial resistance of monolayers coincubated with B. ratti WR1 live parasites dropped from 32.2 ± 1.1 to 27.2 ± 1.1 Ω/cm2 by 12 h, from 32.8 ± 1.5 to 24.2 ± 2.6 Ω/cm2 by 24 h, and dropped further from 32.6 ± 1.7 to 22.4 ± 2.6 Ω/cm2 by 48 h (for all, P < 0.05 in comparison to the control value). IEC-6 cell monolayers characteristically achieve average peak resistance of ∼30 Ω/cm2 about 6 days after seeding. This is in contrast to those of other cell lines (e.g., Caco-2, T-84), which range from a few hundred to a few thousand Ω/cm2. Hence, in the case of 12 h of incubation of live parasites with IEC-6 cell monolayer, a TER drop of 5 Ω/cm2 from a peak of 32.2 Ω/cm2 translates to a significant (P < 0.05 versus negative control) 16% decrease. A similar significant decrease in TER was observed with parasitic lysate, which suggested that parasitic soluble and nonsoluble products are capable of inducing a decrease in TER. To show that the effect on transepithelial resistance was due to live parasites, we added an antiprotozoal drug, metronidazole (10 μg/ml), in the incubation medium as the drug has been reported to induce Blastocystis cell death (32). The addition of metronidazole to monolayers coincubated with live parasites significantly prevented a reduction in TER after 24 h, bringing the levels of TER close to control values (P < 0.05 in comparison to monolayers incubated with live parasites only). In contrast, a significant reduction in TER was noted in monolayers coincubated with live parasites only (Fig. 7).
FIG. 6.
Effect of B. ratti WR1 on the transepithelial resistance of IEC-6 cell monolayers. Confluent monolayers of IEC-6 cells were grown on Millicell-HA filters and coincubated for the indicated times with live parasites, parasitic lysate, or growth medium (negative control). Thereafter, transepithelial resistance was measured as described in Materials and Methods. Live-parasite- and lysate-treated monolayers showed a significant drop in TER after 12, 24, and 48 h. Values are means ± standard deviations (error bars) (three monolayers per group). The values were significantly different (P < 0.05) from the value for the negative control.
FIG. 7.
Effects of caspase inhibition and metronidazole on B. ratti WR1-induced decrease in transepithelial resistance of IEC-6 monolayers. Confluent monolayers of IEC-6 cells were grown on Millicell-HA filters and incubated for 24 h with live parasites, live parasites after pretreatment of cells with broad-spectrum caspase inhibitor Z-VAD-fmk, or live parasites along with the antiprotozoal drug metronidazole. Monolayers incubated with growth medium served as a negative control. Thereafter, transepithelial resistance was measured as described in Materials and Methods. Pretreatment of IEC-6 cells with caspase inhibitors does not considerably rescue these cells from Blastocystis-induced effect, but exposure of Blastocystis to metronidazole abolished the effect significantly. Values are means ± standard deviations (error bars) (three monolayers per group). Values that were significantly different from the values for live-parasite-treated samples are indicated (*, P = 0.07; **, P < 0.05).
To test whether Blastocystis-induced apoptosis of IEC-6 cells plays an important role in inducing a reduction in TER, monolayers were pretreated with general caspase inhibitor Z-VAD-fmk before coincubation with parasites. Results show that the inhibition of caspases in IEC-6 cells did not significantly prevent reduction of TER (P = 0.07 in comparison with monolayers incubated with parasites only [Fig. 7]).
Increase in epithelial permeability.
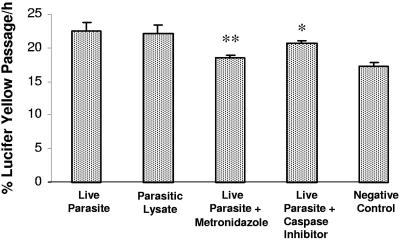
The effects of B. ratti WR1 live parasites and parasitic lysate on the permeability of IEC-6 cell monolayers were analyzed by measuring the apical to basolateral flux of Lucifer yellow. IEC-6 cells exposed to B. ratti WR1 live parasites or parasitic lysates disrupted the monolayer barrier function as evidenced by an increased flux of Lucifer yellow across the monolayer (Fig. 8). There were significant increases in the flux of Lucifer yellow in monolayers coincubated for 24 h with live parasites (22.4% ± 1.2%; P < 0.05) and with parasitic lysates (22.1% ± 1.2%; P < 0.05) in comparison to that of control monolayers (17.2% ± 0.6%). Pretreatment of IEC-6 cell monolayers with the general caspase inhibitor Z-VAD-fmk prior to coincubation with live parasites did not significantly decrease Lucifer yellow flux (20.6% ± 0.3%; P = 0.2 in comparison to that value for live-parasite-treated monolayers) across cell monolayers compared to that of the parasite-free control (17.2% ± 0.6%). This suggests that Blastocystis-induced apoptosis of intestinal epithelial cells might not play a major role in disrupting epithelial permeability, but there are other factors responsible for disruption of permeability.
FIG. 8.
Flux measurement with Lucifer yellow. Confluent monolayers of IEC-6 cells were grown on Millicell-HA filters and incubated for 24 h with live parasites, live parasites after pretreatment of cells with the broad-spectrum caspase inhibitor Z-VAD-fmk, or live parasites along with the antiprotozoal drug metronidazole. Monolayers incubated with only growth medium served as a negative control. Permeability was determined by measurement of Lucifer yellow fluxes across the monolayer as described in Materials and Methods. A significant increase in the epithelial permeability can be noticed after incubation with live parasites and parasitic lysate in comparison to the control monolayer. Pretreatment of IEC-6 cells with caspase inhibitors does not considerably rescue these cells from Blastocystis-induced effect on permeability, but exposure of Blastocystis to metronidazole abolished the effect significantly. Values are means ± standard deviations (error bars) (three monolayers per group). Values that were significantly different from the values for live-parasite-treated samples are indicated (*, P = 0.2; **, P < 0.05).
To prove that changes in the permeability of IEC-6 cell monolayer were due to the effect of the live parasites of Blastocystis, we added an antiprotozoal drug, metronidazole, during coincubation and found that it significantly abrogated the increased flux of Lucifer yellow across the IEC-6 cell monolayer (18.5% ± 0.4%; P < 0.05 in comparison to the value for live-parasite-treated monolayers).
DISCUSSION
The exact role of Blastocystis as a cause of human disease is, to date, controversial and inconclusive. Whether it is a true pathogen or a commensal or is capable of being a pathogen under specific circumstances is currently a matter of debate. Several studies have associated Blastocystis with a variety of gastrointestinal symptoms, but only a few in vitro studies have been carried out to elucidate the pathogenesis of this organism.
We demonstrated that B. ratti WR1 live parasites and parasitic lysates induce apoptosis of a nontransformed rat intestinal epithelial cell line, IEC-6. This apoptosis occurred in a contact-independent manner and suggests that Blastocystis may be secreting certain virulent factors that can initiate the apoptotic pathway in IEC-6 cells. In addition, B. ratti WR1 rearranges F-actin distribution, decreases transepithelial electrical resistance, and increases epithelial permeability across the cell monolayer. Furthermore, our results showed that the antiprotozoal drug metronidazole abolishes the effects of B. ratti WR1 on epithelial barrier function. Together, these findings suggest for the first time that Blastocystis-induced apoptosis of host intestinal cells and disruption of the epithelial barrier function may lead to the pathogenesis of Blastocystis-related gastrointestinal symptoms.
Recently, numerous protozoan parasites, such as G. lamblia, Cryptosporidium parvum, and Entamoeba histolytica, and bacterial pathogens, including Listeria monocytogenes, E. coli, and Clostridium difficile, have been found to induce apoptosis of intestinal epithelial cells (9, 11, 14, 20, 22, 29, 46). The ability of Blastocystis to kill host cells in vitro was observed previously (48), but the mechanism of cell death was not noted. In clinical cases of Blastocystis infections, evidence of intestinal epithelium damage was documented (12), but specific features of cell apoptosis were not reported. Results from our study show for the first time that protozoan B. ratti WR1 can cause apoptosis of intestinal epithelial cells and it can occur in a contact-independent manner. In our study, the percentages of cells showing nuclear fragmentation are comparable in significance with the results of other in vitro studies involving protozoan parasites, such as those with C. parvum, giving rise to 8% cell death in intestinal cell lines (29). In another study (9), 13% apoptosis was seen in the nontransformed cell line SCBN after exposure to G. lamblia lysate, similar to what was observed for Blastocystis lysate (12%). Our findings on contact-independent cell death suggest that Blastocystis has the potential to cause in vivo pathogenic effects without adhering to the gut mucosa. However, studies have shown that intestinal pathogens, such as E. histolytica, C. parvum, and enteropathogenic E. coli, usually establish contact with the host cells to cause apoptosis (11, 20, 29). In contrast, the ability of Blastocystis to adhere to the intestinal epithelium or invade intestinal mucosa has not been established. Most studies suggest Blastocystis to be a noninvasive protozoan (43, 44). Therefore, our current data suggesting that Blastocystis has the ability to cause apoptosis without attaching to the gut mucosa have significant implications for its pathogenicity. Many intestinal protozoan parasites are known to secrete parasitic factors, such as proteases that are responsible for their virulence (41). Blastocystis-secreted proteases can cleave human secretory immunoglobulin A and were suggested to be virulence factors (37). Pathogen invasion and induction of apoptosis are discrete processes, and there are pathogens that can invade but do not induce apoptosis (11). It appears that induction of apoptosis of host intestinal cells would not be advantageous to a noninvasive parasite like Blastocystis, as it would result in the loss of colonization sites for the parasite. This unintended induction of host cell apoptosis might be a host response against some parasitic factors like proteases which are necessary for the parasite's own life cycle. There is a need to further investigate and characterize Blastocystis secretory products which can subsequently shed light on its pathogenesis.
When exposed to certain toxins, the intestinal epithelium can be induced to undergo cell death via apoptosis or necrosis, which can in turn affect intestinal barrier function, resulting in decreased TER and increased permeability (5). In our experiments, we tested whether Blastocystis-induced apoptosis of IEC-6 cells has an effect on epithelial permeability by pretreating the cell monolayers with the general caspase inhibitor Z-VAD-fmk before coincubation with parasites. Our results show that the inhibition of caspases in IEC-6 cells did not prevent significant reduction of TER and did not significantly decrease Lucifer yellow flux across the cell monolayer. Therefore, in the case of Blastocystis infection, we suggest that unintended activation of host cell apoptosis in a contact-independent manner does not play a major role in increased epithelial permeability, but there are other factors responsible for increased permeability. In contrast, studies on G. lamblia have established that while Giardia is a noninvasive pathogen, it releases parasitic factors that may induce enterocyte apoptosis and increase epithelial permeability (9). The apparent discrepancy in our observation may be due to two factors. First, the contribution of apoptosis to increased permeability may be minor because of the low percentage of apoptotic cells, evidenced by DAPI staining, compared to cells with F-actin rearrangement. Second, Blastocystis may have induced nonapoptotic cell death, such as necrosis, autophagy, or paraptosis (7) that may have contributed to the impaired barrier function.
A variety of intestinal pathogens have been shown to cause changes in F-actin arrangement in intestinal epithelial cells (45), and it was reported that dynamic rearrangements of F-actin contribute to an increase in epithelial permeability (17) and affect regulation of epithelial salt and water transport (28), which may consequently lead to diarrhea. Blastocystis has been shown to cause cytopathic effects in Chinese hamster ovary cells (48), but its effects on intestinal epithelial cytoskeleton and barrier functions were not reported. The present study demonstrates for the first time that Blastocystis adversely affects the cytoskeletal proteins in intestinal epithelial cells, causing a rearrangement of F-actin distribution. IEC-6 cells exposed to live B. ratti WR1 and parasitic lysate showed enhanced formation of condensed actin filaments in cells. A significant number of IEC-6 cells exhibited the formation of stress fibers in the cytoplasmic zone of cells.
The findings from the present study that B. ratti WR1 causes apoptosis of intestinal epithelial cells and rearrangement of F-actin distribution prompted us to assess the effects of Blastocystis on transepithelial TER and permeability. An in vitro model involving confluent monolayers of IEC-6 cells grown on filters was used, and the findings demonstrated that on interactions with the apical surface of the epithelial cells, B. ratti WR1 causes a significant decrease in epithelial resistance and an increase in permeability across the cell monolayer. The intestinal mucosa forms a barrier that separates luminal contents from the interstitium, and the intestinal barrier can become compromised in parasitic infections, bacterial infections, and in many intestinal diseases, such as inflammatory bowel disease (10). Intestinal barrier function is mainly regulated by tight junctions, and the function of tight junctions can be measured as a decrease in the transepithelial electrical resistance and an increase in the paracellular flux of macromolecules (3). Studies have suggested that Blastocystis may interact with epithelial cells of the gastrointestinal tract to modulate their transport characteristics and increase intestinal permeability (12). In vitro findings from our study show for the first time that Blastocystis has the ability to disrupt the barrier function of the intestinal epithelium. Our results consistently showed an increase in the transepithelial flux of Lucifer yellow with a decrease in the TER in the IEC-6 monolayers exposed to live B. ratti WR1 parasites and parasitic lysates. The results of our experiments suggest that Blastocystis-induced F-actin rearrangement is associated with reduced TER and altered permeability. The cause of Blastocystis-associated diarrhea is so far unidentified, but an association between increases in epithelial permeability and diarrhea has been described for other diseases (25, 26). It is possible that Blastocystis-induced changes in permeability may also occur in vivo and lead to electrolyte imbalance and diarrhea.
We observed that monolayers coincubated with B. ratti WR1 live parasites in the presence of metronidazole did not show any change in barrier function and retained their normal TER and epithelial permeability. This finding suggests that the effect on monolayer barrier function was due to live parasites only and that dead parasites cannot induce such effects on epithelial TER and permeability. Metronidazole is known to induce programmed cell death in Blastocystis, and the integrity of the plasma membrane of the parasite remains preserved (32); therefore, there is no leakage of intracellular parasitic proteases and other products that can induce an effect on TER and permeability. However, we noticed a change in TER and permeability in monolayers coincubated with parasitic lysates, as the lysates contain all parasitic soluble or nonsoluble products that would have resulted in the observed effect. G. lamblia live parasites and parasitic lysates were shown to exhibit similar effects on intestinal epithelial cytoskeleton and TER (45). In our study; we also found that B. ratti WR1 live parasites and parasitic lysates have almost similar effects on F-actin distribution and epithelial barrier function. In contrast, coincubation of E. histolytica parasitic lysate with human colonic epithelial monolayer failed to lower the epithelial resistance (24).
The need to treat Blastocystis infections is controversial because of its uncertain pathogenicity. There is a lack of standardized treatment for Blastocystis infections, and studies have shown that metronidazole inhibits the growth of Blastocystis (31). In clinical cases where Blastocystis is implicated by gastrointestinal symptoms, metronidazole is the drug of choice (44). Our results show that metronidazole can avert the adverse effects of Blastocystis on intestinal epithelial barrier function and suggest that metronidazole is a therapeutic drug for Blastocystis infections.
In summary, the present study reports for the first time that Blastocystis induces apoptosis in enterocytes and that this may occur in a contact-independent manner. Our findings also demonstrate for the first time that Blastocystis can rearrange F-actin distribution and disrupt epithelial barrier function and that metronidazole can abrogate these effects. In addition, our findings show that IEC-6 monolayers grown on permeable filters provide a useful model for studying the interactions of Blastocystis with intestinal epithelial cells.
There is currently no animal model available to study Blastocystis infections (44). Therefore, future work should also focus on the development of animal models to extend in vitro studies of Blastocystis-host interactions.
Acknowledgments
This work was supported by generous grants from the National Medical Research Council (R-182-000-058-213) and the School of Medicine, National University of Singapore.
We thank Wong Siew Heng and Sylvie Alonso for helpful discussions. We are grateful to Toh Kok Tee, Ong Ling Yeow, and Lee Kong Heng of the National University Medical Institute for their help in flow cytometry analysis and confocal microscopy. We thank Julie Yeo, Tan Mui Hong, and Tan Li Li of the Defense Medical and Environmental Research Institute, DSO, for their contributions. We also thank Geok Choo Ng and Ramachandran for technical support.
Editor: W. A. Petri, Jr.
REFERENCES
- 1.Alexeieff, A. 1911. Sur la nature des formations dites kystes de Trichomonas intestinalis. C. R. Soc. Biol. 71:296-298. [Google Scholar]
- 2.Ashford, R. W., and E. A. Atkinson. 1992. Epidemiology of Blastocystis hominis infection in Papua New Guinea: age-prevalence and associations with other parasites. Ann. Trop. Med. Parasitol. 86:129-136. [DOI] [PubMed] [Google Scholar]
- 3.Berkes, J., V. K. Viswanathan, S. D. Savkovik, and G. Hecht. 2003. Intestinal epithelial responses to enteric pathogens: effects on the tight junction barrier, ion transport, and inflammation. Gut 52:439-451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bjarnason, I., A. MacPherson, and D. Hollander. 1995. Intestinal permeability: an overview. Gastroenterology 108:1566-1581. [DOI] [PubMed] [Google Scholar]
- 5.Bojarski, C., K. Bendfeldt, A. H. Gitter, J. Mankertz, M. Fromm, S. Wagner, E. O. Riecken, and J. D. Schulzke. 2000. Apoptosis and intestinal barrier function. Ann. N. Y. Acad. Sci. 915:270-274. [DOI] [PubMed] [Google Scholar]
- 6.Brites, C., M. G. Barberino, M. A. Bastos, S. M. Sampaio, and N. Silva. 1997. Blastocystis hominis as a potential cause of diarrhoea in AIDS patients: a report of six cases in Bahia. Braz. J. Infect. Dis. 1:91-94. [PubMed] [Google Scholar]
- 7.Broker, L. E., F. A. Kruyt, and G. Giaccone. 2005. Cell death independent of caspases: a review. Clin. Cancer Res. 11:3155-3162. [DOI] [PubMed] [Google Scholar]
- 8.Chen, X. Q., M. Singh, L. C. Ho, K. T. Moe, S. W. Tan, and E. H. Yap. 1997. A survey of Blastocystis sp. in rodents. Lab. Anim. Sci. 47:91-94. [PubMed] [Google Scholar]
- 9.Chin, A. C., D. A. Teoh, K. G. E. Scott, J. B. Meddings, W. K. Macnaughton, and A. G. Buret. 2002. Strain-dependent induction of enterocyte apoptosis by Giardia lamblia disrupts epithelial barrier function in a caspase-3-dependent manner. Infect. Immun. 70:3673-3680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Clayburgh, D. R., L. Shen, and J. R. Turner. 2004. A porous defense: the leaky epithelial barrier in intestinal disease. Lab. Investig. 84:282-291. [DOI] [PubMed] [Google Scholar]
- 11.Crane, J. K., S. Majumdar, and D. F. Pickhardt III. 1999. Host cell death due to enteropathogenic Escherichia coli has features of apoptosis. Infect. Immun. 67:2575-2584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dagci, H., S. Ustun, M. S. Taner, G. Ersoz, F. Karacasu, and S. Budak. 2002. Protozoon infections and intestinal permeability. Acta Trop. 81:1-5. [DOI] [PubMed] [Google Scholar]
- 13.Doyle, P. W., M. M. Helgason, R. G. Mathias, and E. M. Proctor. 1989. Epidemiology and pathogenicity of Blastocystis hominis. J. Clin. Microbiol. 28:116-121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fiorentini, C., A. Fabbri, L. Falzano, A. Fattorossi, P. Matarrese, R. Rivabene, and G. Donelli. 1998. Clostridium difficile toxin B induces apoptosis in intestinal cultured cells. Infect. Immun. 66:2660-2665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gerhard, R., G. Schmidt, F. Hofmann, and K. Aktories. 1998. Activation of Rho GTPases by Escherichia coli cytotoxic necrotizing factor 1 increases intestinal permeability in Caco-2 cells. Infect. Immun. 66:5125-5131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Guimaraes, S., and M. I. L. Sogayar. 1993. Blastocystis hominis: occurrence in children and staff members of municipal day-care centres from Botucatu, Sao Paulo State, Brazil. Mem. Inst. Oswaldo Cruz. 88:427-429. [DOI] [PubMed] [Google Scholar]
- 17.Hecht, G., C. Pothoulakis, J. T. LaMont, and J. L. Madara. 1988. Clostridium difficile toxin A perturbs cytoskeletal structure and tight junction permeability of cultured human intestinal epithelial monolayers. J. Clin. Investig. 82:1516-1524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ho, L. C., M. Singh, G. Suresh, G. C. Ng, and E. H. Yap. 1993. Axenic culture of Blastocystis hominis in Iscove's modified Dulbecco's medium. Parasitol. Res. 79:614-616. [DOI] [PubMed] [Google Scholar]
- 19.Huppertz, B., H. G. Frank, and P. Kaufmann. 1999. The apoptosis cascade—morphological and immunohistochemical methods for its visualization. Anat. Embryol. 200:1-18. [DOI] [PubMed] [Google Scholar]
- 20.Huston, C. D., E. R. Houpt, B. J. Mann, C. S. Hahn, and W. A. Petri, Jr. 2000. Caspase 3-dependent killing of host cells by the parasite Entamoeba histolytica. Cell. Microbiol. 2:617-625. [DOI] [PubMed] [Google Scholar]
- 21.Kerr, J. F., A. H. Wyllie, and A. R. Currie. 1972. Apoptosis: a basic biological phenomenon with wide ranging implications in tissue kinetics. Br. J. Cancer 26:239-267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim, J. M., L. Eckmann, T. C. Savidge, D. C. Lowe, T. Witthoft, and M. F. Kagnoff. 1998. Apoptosis of human intestinal epithelial cells after bacterial invasion. J. Clin. Investig. 102:1815-1823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lader, K., M. E. Hellard, M. I. Sinclair, C. K. Fairley, and R. Wolfe. 2005. No correlation between clinical symptoms and Blastocystis hominis in immunocompromised individuals. J. Gastroenterol. Hepatol. 20:1390-1394. [DOI] [PubMed] [Google Scholar]
- 24.Leroy, A., T. Lauwaet, G. De Bruyne, M. Cornelissen, and M. Mareel. 2000. Entamoeba histolytica disturbs the tight junction complex in human enteric T84 cell layers. FASEB J. 14:1139-1146. [DOI] [PubMed] [Google Scholar]
- 25.Madara, J. L. 1988. Tight junction dynamics: is paracellular transport regulated? Cell 53:497-498. [DOI] [PubMed] [Google Scholar]
- 26.Madara, J. L. 1990. Contributions of paracellular pathway to secretion, absorption, and barrier function in the epithelium of the small intestine, p. 125-138. In F. Lebenthal and M. Duffy (ed.), Textbook of secretory diarrhea. Raven Press, New York, N.Y.
- 27.Madara, J. L., D. Barenberg, and S. Carlson. 1986. Effects of cytochalasin D on occluding junctions of intestinal absorptive cells: further evidence that the cytoskeleton may influence paracellular permeability and junctional charge selectivity. J. Cell Biol. 102:2125-2136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Matthews, J. B., K. J. Tally, J. A. Smith, and C. S. Awtrey. 1994. F-actin differentially alters epithelial transport and barrier function. J. Surg. Res. 56:505-509. [DOI] [PubMed] [Google Scholar]
- 29.McCole, D. F., L. Eckmann, F. Laurent, and M. F. Kagnoff. 2000. Intestinal epithelial cell apoptosis following Cryptosporidium parvum infection. Infect. Immun. 68:1710-1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mercado, R., and B. Arias. 1991. Blastocystis hominis: frecuencia de infeccion en pacientes ambulatorios del sector norte de Santiago, Chile. Bol. Chil. Parasitol. 46:30-32. [PubMed] [Google Scholar]
- 31.Moghaddam, D. D., E. Ghadirian, and M. Azami. 2005. Blastocystis hominis and the evaluation of efficacy of metronidazole and trimethoprim/sulfamethoxazole. Parasitol. Res. 96:273-275. [DOI] [PubMed] [Google Scholar]
- 32.Nasirudeen, A. M., Y. E. Hian, M. Singh, and K. S. Tan. 2004. Metronidazole induces programmed cell death in the protozoan parasite Blastocystis hominis. Microbiology 150:33-43. [DOI] [PubMed] [Google Scholar]
- 33.Nimri, L., and R. Batchoun. 1994. Intestinal colonization of symptomatic and asymptomatic schoolchildren with Blastocystis hominis. J. Clin. Microbiol. 32:2865-2866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Noel, C., F. Dufernez, D. Gerbod, V. P. Edgcomb, P. D. Viscogliosi, L. C. Ho, M. Singh, R. Wintjens, M. L. Sogin, M. Capron, R. Pierce, L. Zenner, and E. Viscogliosi. 2005. Molecular phylogenies of Blastocystis isolates from different hosts: implications for genetic diversity, identification of species, and zoonosis. J. Clin. Microbiol. 43:348-355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ok, U. Z., M. Cirit, A. Uner, E. Ok, F. Akcicek, A. Basci, and M. A. Ozcel. 1997. Cryptosporidiosis and blastocystosis in renal transplant recipients. Nephron 75:171-174. [DOI] [PubMed] [Google Scholar]
- 36.Porter, A. G., and R. U. Janicke. 1999. Emerging role of caspase-3 in apoptosis. Cell Death Differ. 6:99-104. [DOI] [PubMed] [Google Scholar]
- 37.Puthia, M. K., A. Vaithilingam, J. Lu, and K. S. W. Tan. 2005. Degradation of human secretory immunoglobulin A by Blastocystis. Parasitol. Res. 97:386-389. [DOI] [PubMed] [Google Scholar]
- 38.Qadri, S. M. H., G. A. Al-Okaili, and F. Al-Dayel. 1989. Clinical significance of Blastocystis hominis. J. Clin. Microbiol. 27:2407-2409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Raynal, P., and H. B. Pollard. 1994. Annexins: the problem of assessing the biological role for a gene family of multifunctional calcium and phospholipid-binding proteins. Biochem. Biophys. Acta 1197:63-93. [DOI] [PubMed] [Google Scholar]
- 40.Riedl, S. J., and Y. Shi. 2004. Molecular mechanisms of caspase regulation during apoptosis. Nat. Rev. Mol. Cell Biol. 5:897-907. [DOI] [PubMed] [Google Scholar]
- 41.Rosenthal, P. J. 1999. Proteases of protozoan parasites. Adv. Parasitol. 43:105-159. [DOI] [PubMed] [Google Scholar]
- 42.Sio, S. W. S., M. K. Puthia, A. S. Y. Lee, J. Lu, and K. S. W. Tan. 4. March 2006, posting date. Protease activity of Blastocystis hominis. Parasitol. Res. [Online.] doi: 10.1007/s00436-006-0131-1. [Epub ahead of print.] [DOI] [PubMed]
- 43.Stenzel, D. J., and P. F. L. Boreham. 1996. Blastocystis hominis revisited. Clin. Microbiol. Rev. 9:563-584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tan, K. S. W. 2004. Blastocystis in humans and animals: new insights using modern methodologies. Vet. Parasitol. 126:121-144. [DOI] [PubMed] [Google Scholar]
- 45.Teoh, D. A., D. Kamieniecki, G. Pang, and A. G. Buret. 2000. Giardia lamblia rearranges F-actin and alpha-actinin in human colonic and duodenal monolayers and reduces transepithelial electrical resistance. J. Parasitol. 86:800-806. [DOI] [PubMed] [Google Scholar]
- 46.Valenti, P., R. Greco, G. Pitari, P. Rossi, M. Ajello, G. Melino, and G. Antonini. 1999. Apoptosis of Caco-2 intestinal cells invaded by Listeria monocytogenes: protective effect of lactoferrin. Exp. Cell Res. 250:197-202. [DOI] [PubMed] [Google Scholar]
- 47.Vermes, I., C. Haanen, H. Steffens-Nakken, and C. Reutelingsperger. 1995. A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. J. Immunol. Methods 184:39-51. [DOI] [PubMed] [Google Scholar]
- 48.Walderich, B., S. Bernauer, M. Renner, J. Knobloch, and G. D. Burchard. 1998. Cytopathic effects of Blastocystis hominis on Chinese hamster ovary (CHO) and adeno carcinoma HT29 cell cultures. Trop. Med. Int. Health 3:385-390. [DOI] [PubMed] [Google Scholar]
- 49.Walker, J. C., G. Bahr, and A. S. Ehl. 1985. Gastrointestinal parasites in Sydney. Med. J. Aust. 143:480. [DOI] [PubMed] [Google Scholar]
- 50.Yoshikawa, H., K. Morimoto, Z. Wu, M. Singh, and T. Hashimoto. 2004. Problems in speciation in the genus Blastocystis. Trends Parasitol. 20:251-255. [DOI] [PubMed] [Google Scholar]
- 51.Zierdt, C. H., W. S. Zierdt, and B. Nagy. 1995. Enzyme-linked immunosorbent assay for detection of serum antibody to Blastocystis hominis in symptomatic infections. J. Parasitol. 81:127-129. [PubMed] [Google Scholar]