Abstract
Agrobacterium tumefaciens delivers transferred DNA (T-DNA) into cells of plants and yeast. In plants, the T-DNA integrates at random positions into the genome by non-homologous recombination (NHR), whereas in yeast the T-DNA preferably integrates by homologous recombination (HR). Here we show that T-DNA integration by HR in yeast requires the recombination/repair proteins Rad51 and Rad52, but not Rad50, Mre11, Xrs2, Yku70 and Lig4. In the HR events a remarkable shift from insertion-type events to replacement events was observed in rad50, mre11 and xrs2 mutants. Residual integration in the rad51 mutant occurred predominantly by HR, whereas in the rad52 mutant integration occurred exclusively by NHR. Previously, we found that T-DNA integration by NHR is abolished in a yku70 mutant. Thus, Rad52 and Yku70 are the key regulators of T-DNA integration, channeling integration into either the HR or NHR pathway.
INTRODUCTION
Agrobacterium tumefaciens is in use as an efficient vector for the genetic modification of plants. This is based on the bacterium’s ability to introduce part of its Ti plasmid, the transferred DNA (T-DNA), as a single-stranded nucleoprotein complex into cells of plants (1,2). The incoming T-DNA integrates at random positions into the plant genome by a process of non-homologous recombination (NHR) (3). In principle the T-DNA can be targeted to a specific region of the genome by the incorporation of a segment from this genomic area in the T-DNA (4). However, gene targeting is still not routinely applicable in plants as even T-DNAs that share extensive homology with the plant genome integrate preferably by NHR (5).
Some time ago it was shown that Agrobacterium is also able to transfer its T-DNA to the yeast Saccharomyces cerevisiae (6). In yeast, T-DNA sharing homology with the genome preferably integrates by homologous recombination (HR) (6,7), but in the absence of DNA homology integration occurs by NHR as observed in plants (8,9). This indicated that the process of T-DNA integration is predominantly determined by the enzymatic machinery of the host. Using the yeast S.cerevisiae as a model we recently demonstrated that the YKU70, LIG4, RAD50, MRE11, XRS2 and SIR4 genes are required for T-DNA integration by NHR (9). These genes have been shown to be involved in the repair of DNA double-strand breaks by non-homologous end-joining (NHEJ). In addition to functioning in NHEJ, these genes also play a role in telomere length maintenance (YKU70, RAD50, MRE11 and XRS2), telomere-associated transcriptional silencing (YKU70 and SIR4), meiotic recombination and some types of HR (RAD50, MRE11 and XRS2) (10–13). However, genes with an exclusive role in HR, such as RAD51 and RAD52 (12,14), were not necessary for T-DNA integration by NHR (9). In the present study we used yeast as a model system to establish which genes are required for the integration of T-DNA by HR.
MATERIALS AND METHODS
Yeast strains
The yeast strains used in this study are listed in Table 1. The rad52yku70 double mutant was constructed by one step disruption after lithium acetate transformation (17) of the RAD52::TRP1 BamHI fragment of pSM21 (18) into the YPH250yku70 strain. Disruption of RAD52 was confirmed by PCR and Southern blot analysis.
Table 1. Yeast strains.
| Strain | Genotype | Reference |
|---|---|---|
| YPH250 | MATα, ura3-52, lys2-801, ade2-101, trp1-Δ1, his3-Δ200, leu2-Δ1 | 15 |
| YPH250rad51 | MATα, ura3-52, lys2-801, ade2-101, trp1-Δ1, his3-Δ200, leu2-Δ1, rad51::LEU2 | 9 |
| YPH250rad52 | MATα, ura3-52, lys2-801, ade2-101, trp1-Δ1, his3-Δ200, leu2-Δ1, rad52::LEU2 | 9 |
| YPH250yku70 | MATα, ura3-52, lys2-801, ade2-101, trp1-Δ1, his3-Δ200, leu2-Δ1, yku70::LEU2 | 9 |
| YPH250rad50 | MATα, ura3-52, lys2-801, ade2-101, trp1-Δ1, his3-Δ200, leu2-Δ1, rad50::hisG | 9 |
| YPH250lig4 | MATα, ura3-52, lys2-801, ade2-101, trp1-Δ1, his3-Δ200, leu2-Δ1, lig4::HIS3 | 9 |
| YPH250rad52yku70 | MATα, ura3-52, lys2-801, ade2-101, trp1-Δ1, his3-Δ200, leu2-Δ1, rad52::TRP1, yku70::LEU2 | This study |
| JKM115 | MATα, Δho, Δhml::ADE1, Δhmr::ADE1, ade1, leu2-3,112, lys5, trp1::hisG, ura3-52 | 16 |
| JKM129 | MATα, Δho, Δhml::ADE1, Δhmr::ADE1, ade1, leu2-3,112, lys5, trp1::hisG, ura3-52, xrs2::LEU2 | 16 |
| JKM138 | MATα, Δho, Δhml::ADE1, Δhmr::ADE1, ade1, leu2-3,112, lys5, trp1::hisG, ura3-52, mre11::hisG | 16 |
Construction of the binary vector pSDM8001
A 1476 bp BamHI–EcoRI fragment containing the KanMX marker from pFA6a was ligated into BamHI and EcoRI digested pIC20H to form pIC20HKanMX. The KanMX marker was inserted between the PDA1 flanks by replacement of a 2610 bp BglII fragment from pUC4E1α10 (19) with a 1465 BglII fragment from pIC20HKanMX. A 3721 bp XhoI–KpnI fragment from this construct, which carried the KanMX marker and the PDA1 flanks, was then inserted into the XhoI and KpnI sites of pSDM14 to create pSDM8001. Plasmid pSDM8001 was introduced into A.tumefaciens by electroporation (20).
Co-cultivations/T-DNA transfer experiments
Co-cultivations were performed as described (9,21) using A.tumefaciens LBA1119(pSDM8001) (22) and the yeast strains listed in Table 1.
Analysis of T-DNA integration at the PDA1 locus
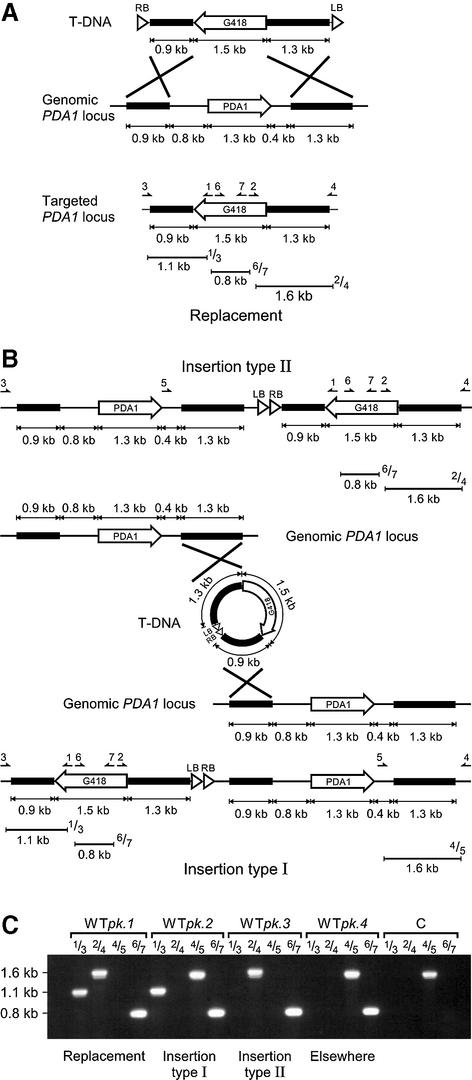
In order to detect whether the T-DNA from pSDM8001 had integrated by HR at the PDA1 locus, PCR analysis was done on G418-resistant yeast colonies obtained after co-cultivation as described (16). The T-DNA primers p1 (5′-TCGCAGGTCTGCAGCGAGGAGC-3′) and p2 (5′-TCGCCTCGACATCATCTGCCCAG-3′) were used in combination with primers p3 (5′-TGTGGTGGGCAACAAGAATGTC-3′), p4 (5′-TTTCTTGTCAAGGGATTATCGGG-3′) and p5 (5′-GCTGTAGTCTGTTATGCCAATCAGG-3′) that anneal to sequences of the PDA1 locus which are absent in the T-DNA (Fig. 1A and B). A replacement event was detected when p1 and p3 generated a 1.1 kb fragment and p2 and p4 a 1.6 kb fragment. When p1 and p3 generated a 1.1 kb fragment and p4 and p5 a 1.6 kb fragment an insertion-type I event was detected. An insertion-type II event was found when p4 and p2 generated a 1.6 kb fragment. The primers p6 (5′-AGACTCACGTTTCGAGGCC-3′) and p7 (5′-TCACCGAGGCAGTTCCATAG-3′) amplified a 0.8 kb fragment to detect the presence of a T-DNA. PCR products were analyzed in a 1% agarose gel (SphaeroQ) after electrophoresis.
Figure 1.
Targeted integration of the T-DNA from pSDM8001 at the PDA1 locus. (A–B) A schematic overview of the different T-DNA integration events. T-DNA integration by homologous recombination can lead to a replacement event or an insertion-type event. Insertion-type events were seen when T-DNA circles were formed by fusion of the T-DNA ends (6). Depending on the PDA1 flanking sequence that was used in the insertion reaction insertion-type I or II events were found. (C) Example of the PCR analysis which was performed to detect T-DNA integration by homologous recombination at the PDA1 locus. G418-resistant wild-type colonies, WTpk.1, WTpk.2, WTpk.3 and WTpk.4, were obtained after co-cultivation with Agrobacterium LBA1119(pSD8001). The untransformed wild-type (WT) was used as a control. PCR was performed as described in Materials and Methods. Four different PCRs were performed using primers p1 + p3, p2 + p4, p4 + p5 and p6 + p7. The position of the primers and the expected sizes of the amplified fragments are indicated in (A) and (B).
CHEF gel analysis was performed using a T-DNA probe as described previously (9).
RESULTS
A novel T-DNA vector to study T-DNA integration by homologous recombination in yeast
In order to be able to study T-DNA integration by HR in any yeast strain a novel T-DNA vector was constructed. The T-DNA of this vector (pSDM8001) carries the dominant KanMX selectable marker, which allows selection of transgenic yeast resistant to G418, surrounded by PDA1 flanking sequences (Fig. 1A). The pSDM8001 vector was introduced into A.tumefaciens strain LBA1119 and used in T-DNA transfer experiments with the S.cerevisiae strains YPH250 and JKM115. The T-DNA from pSDM8001 integrated at frequencies of 3 × 10–5 and 6 × 10–5 into the genomes of these strains, respectively (Table 2). A number of G418-resistant colonies obtained from these experiments were analyzed by PCR to investigate whether the T-DNA from pSDM8001 had integrated by HR at the PDA1 locus. Integration of the T-DNA by HR can lead to gene replacement (double crossover or gene conversion) or insertion of the entire T-DNA (single crossover). Insertion-type events can occur when the ends of the linear T-DNA fuse to form a circle (Fig. 1A and B) (6). In order to detect gene replacement and insertion-type events by PCR we combined primers that anneal to the T-DNA (p1 and p2) with primers that anneal to sequences of the PDA1 locus absent from the T-DNA (p3, p4 and p5) in different reactions (Fig. 1). We found in 27 out of 30 (90%) G418-resistant colonies (YPH250 and JKM115) that the T-DNA from pSDM8001 had indeed integrated at the PDA1 locus by HR (Table 3). Both gene replacement (39%) and insertion-type events (61%) were observed as described previously (6). The presence of a T-DNA was detected in the remaining three (14%) colonies, but it appeared not to be present at the PDA1 locus (Fig. 1C, WTpk.4). This indicated that the T-DNA had integrated elsewhere into the genome. For one of these strains we confirmed by CHEF gel analysis that the T-DNA was not present on chromosome V, which carries the PDA1 locus, but was located on chromosome IV or XII, which co-migrated in the gel (Fig. 2, WTpk.4).
Table 2. Frequencies of T-DNA integration by homologous recombination in recombination-defective yeast mutants.
| Strain | Genotype | Frequency of G418-resistant colonies ± SEM (× 10–6)a | Relative frequency of T-DNA integrationb |
|---|---|---|---|
| YPH250 | WT | 25 ± 7 | 100 |
| YPH250rad51 | rad51Δ | 2.2 ± 0.9c | 9 |
| YPH250rad52 | rad52Δ | 2.4 ± 0.7c | 10 |
| YPH250yku70 | yku70Δ | 33 ± 11 | 134 |
| YPH250rad50 | rad50Δ | 25 ± 5 | 102 |
| YPH250lig4 | lig4Δ | 27 ± 12 | 110 |
| YPH250rad52yku70 | rad52yku70Δ | 3.8 ± 0.4c | 15 |
| JKM115 | WT | 63 ± 18 | 100 |
| JKM129 | xrs2Δ | 77 ± 8 | 122 |
| JKM138 | mre11Δ | 82 ± 7 | 130 |
aAll yeast strains were co-cultivated with Agrobacterium LBA1119(pSDM8001). Averages of two or more independent experiments are shown. Frequencies are depicted as the number of G418-resistant colonies divided by the output number of yeast cells (cells/ml).
bThe relative frequency of T-DNA integration by HR = (frequency of G418-resistant colonies obtained in the mutant/frequency in the wild-type) × 100%.
cThe means of the frequency of G418-resistant colonies of the wild-type and the mutant were tested significantly different in a Student’s t-test (P < 0.05).
Table 3. Integration of homologous T-DNA in the genome of recombination-defective yeast mutants.
| Strain | G418R colonies analyzed | Replacement | Insertiona | Elsewhere | HR (%)b | NHR (%) |
|---|---|---|---|---|---|---|
| WT (YPH250) | 20 | 8 | 10 (7/3) | 2 | 90 (44/56) | 10 |
| rad51 | 17 | 11 | 3 (3/0) | 3 | 82 (79/21) | 18 |
| rad52 | 40 | 0 | 0 (0/0) | 40 | 0 (0/0) | 100 |
| rad50 | 30 | 26 | 0 (0/0) | 4 | 87 (100/0) | 13 |
| yku70 | 10 | 3 | 7 (0/7) | 0 | 100 (30/70) | 0 |
| lig4 | 10 | 5 | 5 (1/4) | 0 | 100 (50/50) | 0 |
| rad52yku70 | 40 | 0 | 0 (0/0) | 0 | 0 (0/0) | 0 |
| WT (JKM115) | 10 | 3 | 6 (0/6) | 1 | 90 (33/66) | 10 |
| xrs2 | 10 | 9 | 0 (0/0) | 1 | 90 (100/0) | 10 |
| mre11 | 10 | 8 | 2 (0/2) | 0 | 100 (80/20) | 0 |
aThe numbers in parentheses indicate the number of insertions by single crossover of type I versus type II.
bThe numbers in parentheses indicate the percentage of replacement versus insertion events after T-DNA integration by homologous recombination at the PDA1 locus.
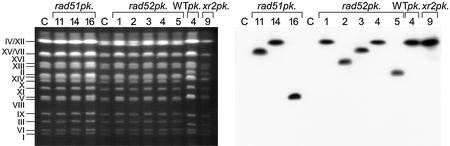
Figure 2.
Non-targeted integration of the T-DNA from pSDM8001. Non-targeted integration events were examined by CHEF gel analysis in wild-type (WT), rad51, rad52 and xrs2 strains. Intact chromosomes were isolated from G418-resistant colonies obtained after co-cultivation with Agrobacterium LBA1119(pSDM8001), separated in a CHEF gel (left panel) and blotted on a membrane. The membrane was hybridized with a labeled probe that anneals to the T-DNA and an autoradiograph was made (right panel).
We conclude that the T-DNA of pSDM8001 preferably integrates by HR at the PDA1 locus and thus that this vector is suitable to study T-DNA integration by HR in yeast.
Rad51 and Rad52 are required for the efficient integration of homologous T-DNA
Recently we demonstrated that the Yku70, Lig4, Rad50, Mre11 and Xrs2 proteins are required for T-DNA integration by NHR (9). In contrast, the Rad51 and Rad52 proteins, which are exclusively involved in HR (12), were found to be dispensable for the integration of T-DNA by NHR (9). In order to study the role of these DNA repair proteins in T-DNA integration by HR we performed T-DNA transfer experiments with the Agrobacterium strain LBA1119(pSDM8001) and a set of isogenic yeast strains carrying disruptions of RAD51, RAD52, YKU70, LIG4, MRE11, RAD50 and XRS2 and compared integration in these mutants with that in the wild-type. The results of these experiments are shown in Table 2. In the rad51 and rad52 mutants the frequency of integration was reduced dramatically and remained only 9 and 10% of that observed in the wild-type. However, in the yku70, lig4, rad50, mre11 and xrs2 mutants the frequency of integration did not differ significantly from the wild-type. Therefore, we conclude that RAD51 and RAD52 are required for T-DNA integration by HR, but that the other DNA repair genes are dispensable.
Mutations in DNA repair genes differently affect T-DNA integration by homologous recombination
Using PCR analysis we then investigated whether the T-DNA had integrated by HR at the PDA1 locus or by NHR at other loci in the various DNA repair mutants. In 14 out of 17 (82%) G418-resistant rad51 colonies the T-DNA had integrated at the PDA1 locus (Table 3). In the remaining three colonies the T-DNA had not integrated at the PDA1 locus on chromosome V as we detected insertions at chromosome XV or VII, IV or XII, VIII or V (outside the PDA1 locus), respectively (Fig. 2, rad51pk.11, rad51pk.14 and rad51pk.16). Thus, although the transformation efficiency was much reduced, integration still predominantly occurred by HR in the rad51 mutant.
In none of the G418-resistant rad52 colonies was the T-DNA present at the PDA1 locus (Table 3). CHEF gel analysis of five of the G418-resistant rad52 colonies demonstrated that the T-DNA had inserted elsewhere into the genome as we detected insertions at chromosome IV or XII, XVI, XV or VII, IV or XII and II, respectively (Fig. 2, rad52pk.1–rad52pk.5). Thus, whereas there is residual T-DNA integration by HR detectable in the absence of Rad51, integration by HR is completely abolished in the rad52 mutant. This demonstrates that RAD52 is essentiel for T-DNA integration by HR.
In 45 out of 50 (90%) G418-resistant colonies of the rad50, mre11 and xrs2 mutants, which lack a functional Rad50– Mre11–Xrs2 complex, integration had occurred by HR, which was comparable with the wild-type (Table 3). However, in contrast to the wild-type, predominantly gene replacement events were seen after integration by HR (44 versus 90%, respectively). In the remaining five (one xrs2 and four rad50) G418-resistant colonies (10%) the T-DNA had integrated elsewhere into the genome by NHR as PCR did not detect the presence of a T-DNA at the PDA1 locus and CHEF gel analysis of the xrs2 strain detected a T-DNA insertion at chromosome IV or XII (Fig. 2, xrs2pk.9). As the frequency of integration by HR is not altered in cells lacking a functional Rad50–Mre11–Xrs2 complex when compared to wild-type, the complex is not required for T-DNA integration by HR per se. However, as replacement events were prevalent amongst the G418-resistant rad50, mre11 and xrs2 colonies, it seems that the Rad50–Mre11–Xrs2 complex is important to bring about insertion-type events.
In the G418-resistant yku70 and lig4 colonies only T-DNA integration events at the PDA1 locus were detected (Table 3). The ratios of gene replacement and insertion-type events were comparable to those seen with the wild-type (Table 3). Thus, we conclude that mutations in YKU70 and LIG4 do not affect T-DNA integration by HR.
Rad52 and Yku70 are the key regulators of T-DNA integration
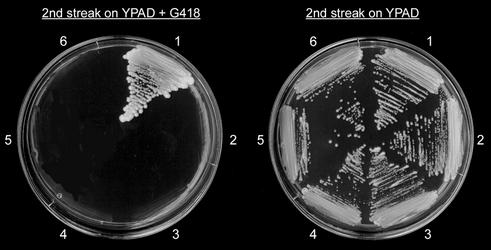
It has been proposed that Rad52 and Yku70 may be competing agents channeling a double-strand break for repair into either the HR or NHEJ pathway (23). Previously we demonstrated that Yku70 is essential for T-DNA integration by NHR (9). We have shown here that Rad52 is essential for T-DNA integration by HR. This suggests that Yku70 and Rad52 are also the key regulators of T-DNA integration, which channel integration into either the NHR or HR pathway. To investigate this further we examined whether disruption of RAD52 in the yku70 mutant strain would inactivate both pathways for T-DNA integration and performed T-DNA transfer experiments with LBA1119(pSDM8001) and a rad52yku70 mutant strain. The frequency of G418-resistant colonies obtained after co-cultivation with the rad52yku70 mutant was much reduced when compared to wild-type, but to our surprise a small number of G418-resistant rad52yku70 colonies were still found (Table 2). However, neither PCR nor CHEF gel analysis revealed the presence of a T-DNA in the genome of 40 G418-resistant rad52yku70 colonies analyzed (data not shown). Apparently, the T-DNA had not integrated into the genome. This demonstrated that T-DNA integration is blocked by the mutations in both RAD52 and YKU70. It has been reported that transformed non-homologous DNA can be ligated in vivo to linear fragments of mitochondrial DNA present in the nucleus to form DNA elements that are not stably maintained (24). To investigate whether the T-DNA was maintained as an unstable extrachromosomal element four G418-resistant rad52yku70 colonies were first grown for 25 generations on medium without G418 and subsequently on medium containing G418. None of the initially G418-resistant rad52yku70 strains were able to grow on the selective medium (Fig. 3), which indicated that the T-DNA was maintained as an unstable extrachromosomal element in the rad52yku70 mutant. Notably, we did not obtain any G418-resistant colonies from co-cultivation experiments with the rad52yku70 mutant when a non-homologous T-DNA that carried the KanMX selectable marker, but not the PDA1 flanking sequences, was transferred (data not shown). This suggests that the PDA1 sequences are involved in maintenance of the T-DNA as extrachromosomal elements when integration into the genome is blocked. We conclude that the Yku70 and Rad52 proteins are absolutely required for the genomic integration of T-DNA and channel integration into the NHR and HR pathway, respectively.
Figure 3.
Loss of the T-DNA in a rad52yku70 double mutant. G418-resistant rad52yku70 colonies, obtained after co-cultivation with Agrobacterium LBA1119(pSDM8001), were purified on YPAD medium containing G418. Single colonies were taken and grown for 25–30 generations on YPAD medium without G418 (first streak). Growth was then assayed on YPAD medium in the presence and absence of G418 (second streak). (1) WTpk.1 (positive control), (2) untransformed WT (negative control), (3) rad52yku70pk.1, (4) rad52yku70pk.2, (5) rad52yku70pk.3, (6) rad52yku70pk.4.
DISCUSSION
Agrobacterium delivers the T-DNA as a single-stranded nucleoprotein complex (T-complex) into cells of its eukaryotic hosts (1,2,6,25). The VirD2 relaxase protein is covalently attached to the 5′ end of the single-stranded T-DNA (26) and the VirE2 protein, which is delivered into the host cell separately from the T-DNA–VirD2 complex (27), binds to the single-stranded T-DNA cooperatively, so that it becomes completely coated by this single-stranded DNA-binding protein (28). This prevents knotting of the T-DNA and leads to the formation of a thin thread-like structure that can pass through the nuclear pore into the nucleus. The presence of VirE2 also prevents degradation of the T-DNA by nucleases that are present in the host cell (29). How the presence of the VirD2 and VirE2 proteins in the T-complex affects T-DNA integration is unknown at the moment. In this study we used the yeast S.cerevisiae as a model to determine the genetic requirements for the targeted integration of Agrobacterium T-DNA. To this end we compared the fate of a T-DNA that carries segments of DNA with homology to the yeast genome in wild-type yeast to that in isogenic strains carrying disruptions of a set of recombination/repair genes. We found that integration of the T-DNA by HR is dependent on Rad52 and also requires Rad51. The absence of the Yku70, Lig4, Rad50, Mre11 and Xrs2 proteins, which previously were found to be involved in T-DNA integration by NHR (9), did not affect the efficiency of integration by HR. For Yku70 and Lig4 this was expected as these proteins are specific for the process of NHEJ and do not play a role in HR (11,13). However, it is known that the Rad50–Mre11–Xrs2 complex is not only involved in NHEJ, but also in some types of HR (10,12,14).
We have chosen the PDA1 locus to study T-DNA integration by HR. PDA1 encodes the E1α subunit of the pyruvate dehydrogenase complex and is a non-essential gene in yeast (19). Previously, this locus was successfully used as the insertion site for T-DNA (6). When the T-DNA sequence was used in a BLAST search to compare it with the yeast genomic sequence in the database, it appeared that the 0.9 kb PDA1 flanking sequence of the T-DNA (Fig. 1A) also showed homology to a region of the BMH2 locus on chromosome IV (76% homology over a 371 bp region). Therefore, we cannot exclude that insertions in chromosome IV had occurred by HR instead of NHR (Fig. 2). However, we think this is unlikely as we have shown that the T-DNA integrates by NHR into several different chromosomes (Fig. 2).
As mentioned above, the structure of the T-DNA which enters the host cell differs significantly from that of naked plasmid DNA that was previously used in transformation experiments. Do these differences also affect the requirements for integration by HR? We found that Rad52 is essential for T-DNA integration by HR. Likewise, it was found that the integration of linear plasmid DNA is blocked in rad52 mutants (30,31). In contrast, the role of RAD52 in the integration of circular plasmid DNA is less clear. Two groups reported little or no effect on integration of single-stranded and double-stranded DNA molecules (31,32), whereas others observed a dramatic reduction in the efficiency of integration in rad52 strains (33,34). In the absence of Rad51, we found that residual T-DNA integration still occurred mainly by HR. Likewise, Schiestl et al. (30) reported that integration of a homologous DNA fragment is reduced (10-fold), but not abolished, in a rad51 mutant. To our knowledge, the effect of a RAD51 mutation on the integration of transformed homologous naked single-stranded DNA molecules has not been determined yet. A clear role for Rad51 in the integration of exogenous DNA has thus not been defined and we may only speculate about its function in DNA integration. Recent studies have shown that RAD51-independent mechanisms of HR exist. For instance, mechanisms such as break-induced replication (BIR), which are dependent on Rad52, can operate in the absence of Rad51 and may be used for T-DNA integration in the rad51 mutant (10).
The efficiency of T-DNA integration by HR was not affected by the absence of a functional Rad50–Mre11–Xrs2 complex. However, an interesting observation was that a shift to gene replacement events was seen in cells that lack this complex. It is not likely that this is due to impaired T-circle formation in the absence of a Rad50–Mre11–Xrs2 complex as the efficiency of circularization, measured by transfer of a T-DNA that carries the KanMX selectable marker and the yeast 2µ replicator sequence, in rad50, mre11 and xrs2 mutants was comparable with wild-type (data not shown). However, an insertion-type event results in a direct repeat. Such direct repeats may be much more unstable in rad50, mre11 and xrs2 mutants, leading to the loss of one of the repeats and the intervening sequences by HR (11,35). The result of such a recombination event would convert an insertion-type event into an apparent replacement event, which may form an explanation for the shift towards replacement events in the rad50, mre11 and xrs2 mutants.
We showed that in the absence of Rad52, integration by NHR becomes the sole pathway for integration. Previously we found that in the absence of Yku70, which is essential for T-DNA integration by NHR (9), HR becomes the sole pathway for integration. Indeed, we have found now that T-DNA integration becomes totally abolished when both RAD52 and YKU70 are inactivated. We conclude that Rad52 and Yku70 are the key regulators of T-DNA integration, channeling integration into the HR and NHR pathway, respectively. These data demonstrate that the pathway used for T-DNA integration, HR or NHR, can be influenced by controlling the activity of specific DNA repair genes. In plants T-DNA integrates efficiently by NHR, even when it shares extensive homology with the plant genome. As plant orthologs of the yeast YKU70, LIG4, RAD50 and MRE11 genes have been identified, it is now of interest to see whether these genes also play an important role in T-DNA integration in plants and whether inactivation of these genes could facilitate targeted integration by preventing non-targeted integration of the T-DNA.
Acknowledgments
ACKNOWLEDGEMENTS
The authors thank Prof. J. Haber (Rosenstiel Center and Biology Department, Brandeis University, Waltham, MA) for sending yeast strains, Dr P. Bundock, Dr D. Weijers and R. Benjamins for many useful discussions, Peter Hock for assistance with the figures and Dr S. de Pater for useful comments on the manuscript. Research in our laboratory was supported by the EU RECOMBIO Biotech program BIO-CT97-2028.
REFERENCES
- 1.Tzfira T., Rhee,Y., Chen,M.H., Kunik,T. and Citovsky,V. (2000) Nucleic acid transport in plant-microbe interactions: the molecules that walk through the walls. Annu. Rev. Microbiol., 54, 187–219. [DOI] [PubMed] [Google Scholar]
- 2.Zhu J., Oger,P.M., Schrammeijer,B., Hooykaas,P.J.J., Farrand,S. and Winans,S. (2000) The bases of crown gall tumorigenesis. J. Bacteriol., 182, 3885–3895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tinland B. and Hohn,B. (1995) Recombination between prokaryotic and eukaryotic DNA: integration of Agrobacterium tumefaciens T-DNA into the plant genome. In Setlow,J.K. (ed.), Genetic Engineering. Plenum Press, New York, NY, pp. 209–229. [PubMed]
- 4.Offringa R., De Groot,M.J.A., Haagsman,H.J., Does,M.P., Van den Elzen,P.J.M. and Hooykaas,P.J.J. (1990) Extrachromosomal homologous recombination and gene targeting in plant cells after Agrobacterium mediated transformation. EMBO J., 9, 3077–3084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vergunst A.C. and Hooykaas,P.J.J. (1999) Recombination in the plant genome and its application in biotechnology. Crit. Rev. Plant Sci., 18, 1–31. [Google Scholar]
- 6.Bundock P., Den Dulk-Ras,A., Beijersbergen,A. and Hooykaas,P.J.J. (1995) Transkingdom T-DNA transfer from Agrobacterium tumefaciens to Saccharomyces cerevisiae. EMBO J., 14, 3206–3214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Risseeuw E., Franke-van Dijk,M.E. and Hooykaas,P.J. (1996) Integration of an insertion-type transferred vector from Agrobacterium tumefaciens into the Saccharomyces cerevisiae genome by gap-repair. Mol. Cell. Biol., 16, 5924–5932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bundock P. and Hooykaas,P.J.J. (1996) Integration of Agrobacterium tumefaciens T-DNA in the Saccharomyces cerevisiae genome by illegitimate recombination. Proc. Natl Acad. Sci. USA, 93, 15272–15275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Van Attikum H., Bundock,B. and Hooykaas,P.J.J. (2001) Non-homologous end-joining proteins are required for Agrobacterium T-DNA integration. EMBO J., 20, 6550–6558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kraus E., Leung,W.-Y. and Haber,J.E. (2001) Break-induced replication: a review and an example in budding yeast. Proc. Natl Acad. Sci. USA, 98, 8255–8262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lewis L.K. and Resnick,M.A. (2000) Tying up loose ends: nonhomologous end-joining in Saccharomyces cerevisiae. Mutat. Res., 451, 71–89. [DOI] [PubMed] [Google Scholar]
- 12.Sung P., Trujillo,K.M. and Van Komen,S. (2000) Recombination factors of Saccharomyces cerevisiae. Mutat. Res., 451, 257–275. [DOI] [PubMed] [Google Scholar]
- 13.Critchlow S.E. and Jackson,S.P. (1998) DNA end-joining: from yeast to man. Trends Biochem. Sci., 23, 394–398. [DOI] [PubMed] [Google Scholar]
- 14.Pâques F. and Haber,J.E. (1999) Multiple pathways of recombination induced by double-strand breaks in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev., 63, 349–404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sikorski R.S. and Hieter,P. (1989) A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics, 122, 19–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Moore J.K. and Haber,J.E. (1996) Cell cycle and genetic requirements of two pathways of nonhomologous end-joining repair of double-strand breaks in Saccharomyces cerevisiae. Mol. Cell. Biol., 16, 2164–2173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gietz D., St Jean,A., Woods,R.A. and Schiestl,R.H. (1992) Improved method for high efficiency transformation of intact yeast cells. Nucleic Acids Res., 20, 1425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schild D., Calderon,I.L., Contopoulou,C.R. and Mortimer,R.K. (1983) Cloning of yeast recombination repair genes and evidence that several are nonessential genes. In Friedberg,E.C. and Bridges,B.A. (eds), Cellular responses to DNA damage. Alan R. Liss Inc., New York, NY, pp. 417–427.
- 19.Steensma H.Y., Holterman,L., Dekker,I., Van Sluis,C.A. and Wenzel,T.J. (1990) Molecular cloning of the gene for the E1 alpha subunit of the pyruvate dehydrogenase complex from Saccharomyces cerevisiae. Eur. J. Biochem., 191, 769–774. [DOI] [PubMed] [Google Scholar]
- 20.Den Dulk-Ras A. and Hooykaas,P.J.J. (1995) Electroporation of Agrobacterium tumefaciens. Methods Mol. Biol., 55, 63–72. [DOI] [PubMed] [Google Scholar]
- 21.Van Attikum H., Bundock,P., Den Dulk-Ras,A., Vergunst,A.C. and Hooykaas,P.J.J. (2002) Agrobacterium tumefaciens mediated transformation of Saccharomyces cerevisiae.TTO, 1, T02732. [Google Scholar]
- 22.Hood E.E., Gelvin,S.B., Melchers,L.S. and Hoekema,A. (1993) New Agrobacterium helper plasmids for gene transfer to plants. Transgenic Res., 2, 208–218. [Google Scholar]
- 23.Haber J.E. (1999) Gatekeepers of recombination. Nature, 398, 665–667. [DOI] [PubMed] [Google Scholar]
- 24.Schiestl R.H., Dominska,M. and Petes,T.D. (1993) Transformation of Saccharomyces cerevisiae with nonhomologous DNA: illegitimate integration of transforming DNA into yeast chromosomes and in vivo ligation to mitochondrial DNA sequences. Mol. Cell. Biol., 13, 2697–2705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.De Groot M.J.A., Bundock,P., Hooykaas,P.J.J. and Beijersbergen,A.G.M. (1998) Agrobacterium-mediated transformation of filamentous fungi. Nat. Biotechnol., 16, 839–842. [DOI] [PubMed] [Google Scholar]
- 26.Ward E.R. and Barnes,W.M. (1988) VirD2 protein of Agrobacterium tumefaciens very tightly linked to the 5′end of T-strand DNA. Science, 242, 927–930. [Google Scholar]
- 27.Vergunst A.C., Schrammmeijer,B., Den Dulk-Ras,A., De Vlaam,C.M., Regensburg-Tuink,T.J. and Hooykaas,P.J.J. (2000) VirB/D4-dependent protein translocation from Agrobacterium into plant cells. Science, 290, 979–982. [DOI] [PubMed] [Google Scholar]
- 28.Christie P.J., Ward,J.E., Winans,J.C. and Nester,E.W. (1988) The Agrobacterium tumefaciens virE2 gene product is a single-stranded DNA binding protein that associates with T-DNA. J. Bacteriol., 170, 2584–2591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rossi L., Hohn,B. and Tinland,B. (1996) Integration of complete transferred DNA units is dependent on the activity of virulence E2 protein of Agrobacterium tumefaciens. Proc. Natl Acad. Sci. USA, 93, 126–130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schiestl R.H., Zhu,J. and Petes,T.D. (1994) Effect of mutations in genes affecting homologous recombination on restriction enzyme-mediated and illegitimate recombination in Saccharomyces cerevisiae. Mol. Cell. Biol., 14, 4493–4500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Orr-Weaver T.L., Szostak,J.W. and Rothstein,R.J. (1981) Yeast transformation: a model system for the study of recombination. Proc. Natl Acad. Sci. USA, 78, 6354–6358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Simon J.R. and Moore,P.D. (1990) Transformation and recombination in rad mutants of Saccharomyces cerevisiae. Mol. Gen. Genet., 223, 241–248. [DOI] [PubMed] [Google Scholar]
- 33.Malone R.E., Montelone,B.A., Edwards,C., Carney,K. and Hoekstra,M.F. (1988) A reexamination of the role of the RAD52 gene in spontaneous mitotic recombination. Curr. Genet., 14, 211–223. [DOI] [PubMed] [Google Scholar]
- 34.Schiestl R.H. and Prakash,S. (1990) RAD10, an excision repair gene of Saccharomyces cerevisiae, is involved in the RAD1 pathway of mitotic recombination. Mol. Cell. Biol., 10, 2485–2491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gherbi H., Gallego,M.E., Jalut,N., Lucht,J.M., Hohn,B. and White,C.I. (2001) Homologous recombination in planta is stimulated in the absence of Rad50. EMBO Rep., 2, 287–291. [DOI] [PMC free article] [PubMed] [Google Scholar]