Abstract
OBJECTIVES
To identify potential obstacles to bone mineral density (BMD) testing, we performed a structured review of current osteoporosis screening guidelines, studies of BMD testing patterns, and interventions to increase BMD testing.
DESIGN
We searched medline and HealthSTAR from 1992 through 2002 using appropriate search terms. Two authors examined all retrieved articles, and relevant studies were reviewed with a structured data abstraction form.
MEASUREMENTS AND MAIN RESULTS
A total of 235 articles were identified, and 51 met criteria for review: 24 practice guidelines, 22 studies of screening patterns, and 5 interventions designed to increase BMD rates. Of the practice guidelines, almost one half (47%) lacked a formal description of how they were developed, and recommendations for populations to screen varied widely. Screening frequencies among at-risk patients were low, ranging from 1% to 47%. Only eight studies assessed factors associated with BMD testing. Female patient gender, glucocorticoid dose, and rheumatologist care were positively associated with BMD testing; female physicians, rheumatologists, and physicians caring for more postmenopausal patients were more likely to test patients. Five articles described interventions to increase BMD testing rates, but only two tested for statistical significance and no firm conclusions can be drawn.
CONCLUSIONS
This systematic review identified several possible contributors to suboptimal BMD testing rates. Osteoporosis screening guidelines lack uniformity in their development and content. While some patient and physician characteristics were found to be associated with BMD testing, few articles carefully assessed correlates of testing. Almost no interventions to improve BMD testing to screen for osteoporosis have been rigorously evaluated.
Keywords: osteoporosis, research methods, diagnosis, clinical reviews
Screening for osteoporosis, primarily with bone mineral density (BMD) testing, permits prediction of future fractures among white women.1–3 However, screening may not be adequately performed on at-risk patients. As the population ages and osteoporosis increases in prevalence, there is a growing need to develop practical public health initiatives to bring effective screening technologies and strategies to widespread use. This is particularly important in light of the substantial morbidity, mortality, and medical costs caused by osteoporosis.4 One of the research priorities described by the 2000 NIH Consensus Development Conference on Osteoporosis was the “need to study the most effective method of educating the public and health care professionals about the diagnosis and treatment of osteoporosis.”5 To realize this priority, the scope of the problem must be adequately assessed, obstacles to providing screening must be identified, and prior interventions to improve screening must be examined.
We framed three questions in an effort to better understand the apparent underutilization of bone mineral density testing and how the rate at which at-risk populations undergo screening could be optimized. First, do recommendations for the use of BMD testing as a screening test for osteoporosis present a unified and coherent message to clinicians? Second, are predictors for BMD testing well defined? Last, are there proven strategies for improving rates of BMD testing? We systematically reviewed three article types to address these questions. We first examined practice guidelines for osteoporosis screening. Next, we reviewed studies that described rates of BMD testing in different populations to explore what patient or physician factors have been identified as predictors of undergoing or not undergoing densitometry. Finally, we examined articles that described interventions designed to increase the use of such testing.
METHODS
Study Selection
We searched medline and HealthSTAR for English-language articles published between January 1992 and December 31, 2002 on the following MESH terms: osteoporosis + guideline, osteoporosis + practice pattern, osteoporosis + provider characteristics, osteoporosis + management, osteoporosis + screening, osteoporosis + x-ray densitometry, and screening + x-ray densitometry. References from relevant articles were reviewed, as were abstracts presented at national meetings; when possible, authors were contacted for full text references. Last, experts in the field were consulted to obtain additional references.
Article selection was performed in two stages. First, two authors (DHS and HC) examined all abstracts to determine their relevance to this review. We excluded meta-analyses, review articles, public policy statements about osteoporosis, articles about hormone replacement therapy (HRT) that did not focus on osteoporosis, articles without primary data, aides to decide whom to screen, and articles not about osteoporosis management. The remaining articles were reviewed completely by two authors (CAM and DC), using the same exclusion criteria. The articles were organized into three categories: clinical practice guidelines published in peer-reviewed journals or issued by national organizations, studies that reported the rate of and/or predictors of BMD testing, and interventions to increase BMD testing rates.
Data Abstraction
Each included article was reviewed using a structured data abstraction tool (available upon request). For all articles, we recorded the source of funding. For clinical guidelines we noted the method for creating the guidelines, including whether a formal consensus process was described, whether evidence for the guidelines was graded, and whether cost effectiveness was mentioned as a criterion for the guidelines. While our primary purpose was to focus on the process and methods by which guidelines are developed, we also recorded specific screening recommendations, including the target patient populations, osteoporosis risk factors, the screening modalities discussed, and the presence of a practice algorithm.
For articles describing patterns of bone densitometry, we abstracted demographic information and relevant clinical characteristics of the study population. We noted the frequency of BMD testing and the source of this information. Any data on patient, physician, or system characteristics predictive of screening were assessed. BMD rates that were adjusted for patient and/or physician characteristics were preferred when available. Articles examining the frequency of bone densitometry by different physician groups were considered separately. For interventions to improve bone densitometry, similar demographic information about the target population was recorded, as well as trial design, the details of the intervention, endpoints, and effect estimates.
Analysis
Both reviewers discussed all data, and achieved consensus by re-review of the papers if necessary. The articles were too heterogeneous in design and study populations to attempt a formal meta-analysis. Data for BMD testing frequencies were summarized as a weighted mean.
RESULTS
We identified a total of 235 articles on screening and treatment of osteoporosis.6 After reviewing abstracts, 155 articles were excluded: 62 only concerned hormone replacement therapy (HRT), 24 described decision aides, 19 did not discuss management of osteoporosis, 12 lacked data, and 38 were excluded for other reasons. The remaining 80 studies of osteoporosis screening included 36 guidelines, 35 studies of screening practices, and 9 studies of interventions. Of the 36 articles identified as potential guidelines, 12 were excluded from the analysis: 5 were public policy statements, 3 were review articles, and 4 lacked specific guidelines. Among the 24 guidelines were 7 pairs of revised recommendations from the same sponsor; for the purpose of this review only the most recent version was considered. Of the 35 articles on patterns of BMD testing, 10 were excluded from the review: 6 articles that discussed treatment patterns only and 4 articles that lacked data on BMD testing as the outcome of interest. We identified 9 intervention studies, and excluded 4: 3 articles had bone densitometry as the intervention rather than the outcome, and 1 was unrelated to osteoporosis.
Clinical Practice Guidelines
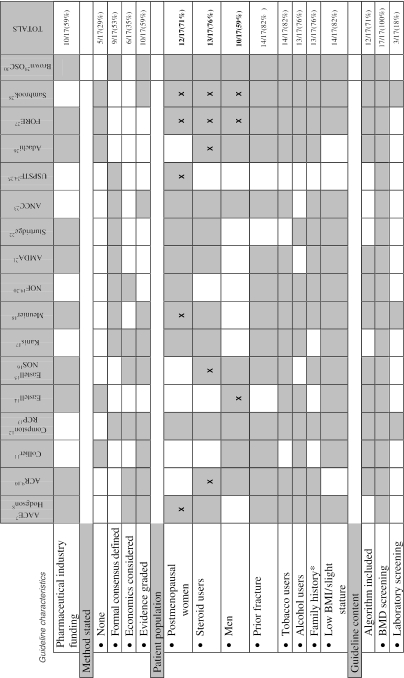
Results of our review of the literature on clinical practice guidelines for osteoporosis screening are described in the Appendix and summarized in Table 1.7–30 The processes used in the development of guidelines varied widely. Five of 17 (29%) did not describe how the guidelines were derived.11,14,26,27,28 While guidelines were often described as the product of a “consensus,” only 9 of 17 (53%) mentioned how that consensus was defined.12,17,18,20,21,23,25,28,29 Of the 17 guidelines, 10 (59%) commented on how evidence was graded or evaluated for guideline development,7,9,11,12,15,17,18,23,28,29 and 6 (35%) considered the cost of screening.7,9,12,15,17,20 Ten of 17 (59%) guidelines were developed with support from the pharmaceutical industry.7,9,14,15,18,21,22,26,27,29
Table 1.
Clinical Practice Guidelines for Osteoporosis
| Percent | |
|---|---|
| Guideline development (N = 17)* | |
| No methods stated | 29 |
| Consensus process defined | 53 |
| Economics considered | 35 |
| Evidence graded | 59 |
| Pharmaceutical funding | 59 |
| Algorithm included | 71 |
| Guideline content† | |
| BMD screening recommended in: | |
| Women (N = 11)‡ | |
| All women >65 years of age | 55 |
| All postmenopausal women with: | |
| Low BMI | 91 |
| Family history of osteoporosis | 91 |
| Oral glucocorticoid use | 82 |
| Alcohol/tobacco use | 55 |
| Osteopenia on radiograph | 55 |
| Women with previous fracture alone | 82 |
| Premenopausal women with additional risk factors | 0 |
| Men (N = 4)§ | |
| With one additional risk factor | 100 |
| Oral glucocorticoid users (N = 5)|| | |
| Any use | 40 |
| ≥5 mg daily | 40 |
| ≥7.5 mg daily | 20 |
| Recommended screening with modality other than BMD (N = 17) | 18 |
An analysis of development and content characteristics for published osteoporosis guidelines.
Among all guidelines that included specific recommendations for one or more specific target populations (women, men, or oral glucocorticoid users; some guidelines considered more than one population).
The guidelines also varied in content. Twelve of 17 (71%) guidelines included an algorithm either in graphic or text form for clinicians,9,11,12,14,15,18,19,21,26–29 and 3 of 17 (18%) guidelines suggested a role for screening modalities other than bone densitometry, such as serum chemistries or bone turnover markers.12,18,27 To compare the specific screening recommendations, we omitted 1 specialty guideline that focused only on patients with chronic liver disease11 and 1 guideline that did not specify osteoporosis risk factors.21 Among guidelines that considered postmenopausal women, 6 of 11 (55%) recommended universal screening for women over the age of 65.7,18,19,25,27,29 All guidelines endorsed screening postmenopausal women under the age of 65 who had an additional risk factor, though there was no agreement on which risk factors should be considered. The most commonly cited were low body mass index, family history of osteoporosis, alcohol and/or tobacco use, and a previous fracture. Only 4 of 17 (24%) guidelines discussed screening men with risk factors.12,14,27,29 Five guidelines recommended screening patients taking oral glucocorticoids.9,15,26–28 Of these, two endorsed screening patients regardless of dose,27,28 two recommended screening patients who take a daily prednisone dose of 5 mg or more,9,15 and another proposed a ≥7.5 mg dose threshold.26
Patterns of Bone Densitometry
Twenty-two papers provided data on rates of BMD testing.23–52 Tables 2 and 3 describe patterns of BMD screening for 2 of the most frequently studied subgroups, postfracture patients38–44 and oral glucocorticoid users.45–52 Studies of physicians’ characteristics were included.34–36
Table 2.
Osteoporosis Screening in Patients with Fractures
| Author | Study Period | Country | N | Data Source | Mean Age, y (Range) | Female, % | Postmenopausal, % | Type of Fracture(s) | Screened with BMD, % |
|---|---|---|---|---|---|---|---|---|---|
| Harrington38 | 1997 to 1999 | U.S. | 502 | MR review | 80 | 82 | N/A | Hip | 14 |
| Kiebzak39 | 1996 to 2000 | U.S. | 363 | Self-report | (53 to 99) | 70 | N/A | Hip | 0.9 |
| Riley40 | 1993 to 1999 | U.S. | 43 | MR review | 72 | 0 | 0 | Hip | 12 |
| Smith41 | N/A | Australia | 218 | Self-report | 70* | 78 | N/A | Hip Wrist | 32 |
| Freedman42 | 1994 to 1997 | U.S. | 1162 | Claims | (55 to 95+) | 100 | 100 | Wrist | 2.8 |
| Hajcsar43 | 1996 to 1997 | Canada | 108 | MR review | 64 | 89 | 87 | Hip Wrist Proximal humerus Vertebrae | 18 |
| Kamel44 | 1996 to 1998 | U.S. | 170 | MR review | 80 | 75 | 75 | Hip | 3 |
Weighted mean of 4 median ages.
BMD, bone mineral densitometry; MR, medical record; PM, postmenopausal; N/A, data not available.
Table 3.
Osteoporosis Screening in Glucocorticoid Users
| Author | Study Period | Country | N | BMD Data Source | Mean Age, y | Female, % | Postmenopausal, % | Screened with BMD, % |
|---|---|---|---|---|---|---|---|---|
| Gudbjornsson45 | 1995 to 1996 | Iceland | 191 | MR | 66 | 55 | 76 | 1† |
| Solomon*46 | 1999 | U.S. | 236 | MR | 60 | 80 | 67 | 23 |
| Ettinger47 | 1998 to 1999 | U.S. | 8,807 | CD | N/A | 60 | N/A | 7 |
| Mudano*48 | 1995 to 1998 | U.S. | 6,821 | CD | 52 | 62 | N/A | 9 |
| Smith49 | 1999 | Australia | 189 | MR | 75 | 38 | 100 | 47 |
| Yood*50 | 1997 to 1998 | U.S. | 224 | CD | 70 | 57 | N/A | 31 |
| Osiri*51 | 1993 to 1999 | U.S. | 365 | MR | 53 | 59 | N/A | 19 |
| Buckley*52 | 1997 to 1998 | U.S. | 147 | SR | 51 | 58 | 50 | 29 |
All data adjusted.
Limited BMD equipment in geographic area studied.
MR, medical records; CD, claims data; SR, self-report; PM, postmenopausal.
BMD testing rates ranged from 1% to 32% of postfracture patients and 1% to 47% of oral glucocorticoid users. One article that reported a 0% rate of screening in a postfracture population acknowledged that BMD testing was unavailable during the study period, so it was omitted from consideration.33 The weighted average screening rates were 8% in the postfracture population and 9% in patients using oral glucocorticoids. In the three studies that examined physician characteristics for performing BMD testing, the percentage of doctors ordering bone densitometry as a screening test for osteoporosis varied from 38% to 62%.34–36 Two of these studies used physician self-report as the primary data source.34,36
Fourteen studies examined potential predictors of bone densitometry and 8 presented data that were adjusted for covariates.34,35,41,46,48,50–52 Female gender39,41,46,48–50 and having care provided by a rheumatologist46,48,49,51,52 were found to predict BMD testing in at least 2 studies. Neither patient age nor presence of comorbidities was associated with BMD testing. Female physicians and doctors caring for larger numbers of postmenopausal women associated with higher rates of use of bone densitometry in 2 studies, while physician age and years since medical school graduation were not associated with rates of bone density testing.34–36 One article found higher rates of BMD testing in areas with more bone densitometers.37
Interventions
Interventions to increase the rate of BMD testing are presented in Table 4.53–57 Two were multifaceted interventions designed to address both osteoporosis screening and treatment.53,55 Two studies were randomized controlled trials,54,57 and 1 used a nonrandomized design.56 The interventions targeted both patients and physicians. One randomized trial examined the effect of varying the emphasis of pretesting counseling between positively and negatively focused consequences of BMD testing. Those who received negatively focused counseling (e.g., mentioning anxiety associated from a diagnosis of osteoporosis or potential side effects of osteoporosis treatment) were significantly less likely to proceed to BMD testing than a control group or those who were given positively focused counseling. The second randomized trial examined the effects of BMD report length on physician ordering of BMD testing. Those physicians receiving long clinical narrative reports were more likely to order BMD testing than those receiving shorter reports of a technical nature. While 3 of 5 interventions we found reported an increased rate of BMD tests ordered after the intervention was completed, only the 2 randomized trials demonstrated statistically significant differences.
Table 4.
Interventions to Improve Rates of Screening Bone Densitometry
| Author | N | Mean Age, y | Target Population | Intervention | Control | Data Source | Study Design | Relative BMD Rate Increase |
|---|---|---|---|---|---|---|---|---|
| Chevalley53 | 385 | 73 | Patients w/prior fracture | • RN risk assessment | Standard care | CD | Before-after* | N/A |
| • Need for BMD assessed/ordered by MD | ||||||||
| • Recommendations sent to PCP and orthopedic surgeon | ||||||||
| Wroe54 | 90 | 51 | Patients | • Positively and negatively focused statements about BMD† | “Neutral” discussion | BMDR | RCT | 91%‡ |
| Newman55 | N/A | N/A | Patients | • Algorithm dissemination | Standard care | CD | Before-after | 167%§ |
| • Pharmacist education | ||||||||
| • Patient education classes | ||||||||
| • Postcard reminders | ||||||||
| • Patient surveys | ||||||||
| • BMD machines to health centers | ||||||||
| Pazirandeh56 | 672 | PreM: 48 PM: 55 | Physicians | • CME on osteoporosis for one group of physicians | No CME | SR | Nonrandomized controlled trial | 0% |
| Stock57 | 57 | N/A | Physicians | • Short BMD reports | Long BMD reports | SR | RCT | 81%|| |
Designed as “before-and-after” trial for effect of intervention upon treatment, but only “after” data provided for BMD.
“Positive group” patients received information about the benefits of testing, while “negative group” patients learned about the possible negative effects of an osteoporosis diagnosis (anxiety, drug side effects).
The effect of positively focused versus negatively focused statements. Positively focused statements versus control: −13% (NS), negatively focused statements versus control: −54% (P < .02).
1999 BMD rates compared to 1996 BMD rates.
Long reports versus short reports (control).
N/A, information not available; RCT, randomized controlled trial; BMD, bone mineral densitometry; PCP, primary care physicians; PreM, premenopausal; PM, postmenopausal; CME, continuing medical education; CD, claims data; SR, self-report; BMDR, bone mineral densitometry appointment registry.
DISCUSSION
Our systematic review of three categories of articles from the osteoporosis screening literature found that osteoporosis screening guidelines lack uniformity in their development and content. In populations for which there was agreement in guidelines about the utility of performing BMD screening, such as older patients with fractures and oral glucocorticoid users, we found low screening rates across multiple studies. Several patient and physician characteristics were associated with BMD testing, yet few articles carefully assessed correlates of screening using adjusted analyses. Only a small number of interventions to improve osteoporosis screening have been undertaken, and none of the findings have been reproduced.
There is concern that bone densitometry is performed infrequently in at-risk populations, and our findings may provide insight into this underutilization. Differing methods for guideline development, from evidence selection and grading to the degree of pharmaceutical support, may affect their credibility. In addition, variable recommendations across different practice guidelines limit the effectiveness of such recommendations. For example, while many guidelines endorsed screening all postmenopausal women under 65 with an additional risk factor for osteoporosis, there was little agreement on what those risk factors were. This lack of consensus may lead physicians to conclude there is no convincing evidence for any screening strategy. Recommendations were discrepant for other groups, including older postmenopausal women, men, and oral glucocorticoid users. Such heterogeneity of practice guidelines may produce confusion among physicians and patients and impair decision making regarding osteoporosis. Confusion about appropriate populations on which to conduct BMD testing may extend even to high-risk groups for whom there is clinical agreement. The low rates of BMD screening reported in oral glucocorticoid users and postfracture populations may in part be due to conflicting practice guidelines.
Almost all of the studies reported low rates of BMD testing: less than one tenth of postfracture or glucocorticoid-using patients, most of whom were postmenopausal women, received BMD testing. While not entirely comparable, these average screening rates are far lower than those for other diseases with accepted screening strategies including prostate cancer and hypercholesterolemia.58–60 Few predictors of BMD testing beyond patient gender, physician specialty, physician gender, and dose of oral glucocorticoids have been examined or identified. The limited information related to correlates of bone densitometry use prevents understanding which patient subgroups are more or less likely to undergo recommended screening. Equally important is the possibility that unrecognized groups of low-risk patients may be receiving BMD unnecessarily.
Improving the identification of individuals with osteoporosis through BMD testing requires a better understanding of how and where to intervene. The lack of well-defined strategies for improving rates of BMD testing prevents physicians, administrators, and health policy advocates from addressing the needs of at-risk populations. The lack of well-designed controlled trials of interventions to improve rates of BMD testing hampers quality improvement in this area.
The findings of this review may be subject to several limitations. It is possible our search strategy failed to identify published articles that would have been relevant to our analysis. Hospitals, clinics, insurers, and other health care organizations produce their own practice guidelines that were not included for review. While we did collect information on the content of guidelines, our review was not a meta-analysis or a “meta-summary.” It was not intended as a comprehensive synthesis of the recommendations of all available practice guidelines. Various studies correlated BMD testing data from different sources, possibly confusing the interpretation of a weighted average of BMD testing rates. We did not collect data on treatment rates in this analysis. High rates of treatment could partially explain low rates of BMD testing if physicians opted to initiate therapy for high-risk patients without documentation of low bone density. While some evidence supports this approach,61,62 a review we conducted of osteoporosis treatment found low rates of pharmacological therapy in similar populations.6
Further research is necessary to better determine the factors that predict BMD testing, and how best to optimize the rate at which at-risk populations undergo screening. There is a need to clarify patterns of BMD use across less-studied populations: nonwhite postmenopausal women, premenopausal women with multiple risk factors, hospitalized patients, and men. Studies that report screening rates should include analyses that adjust for relevant patient and physician characteristics, and assess awareness of osteoporosis and its risk factors, accessibility of densitometry equipment, reimbursement of testing, and familiarity with reports and therapeutic options. A better understanding of factors that predict osteoporosis screening is critical for the development of effective interventions.
Acknowledgments
This work was supported by the Arthritis Foundation and NIH grants K23-AR48616, K24-AR02123, and P60-AR47782.
APPENDIX
FIGURE 1.
Practice Guidelines for Osteoporosis Screening
Shading indicates that component discussed in the guideline; X indicates primary patient group(s).
*Of fracture or osteoporosis.
AACE, American Association of Clinical Endocrinologists; ACR, American College of Rheumatology; RCP, Royal College of Physicians; NOS, National Osteoporosis Society; NOF, National Osteoporosis Foundation; AMDA, American Medical Directors Association; ANCC, Australian National Consensus Conference; USPSTF, United States Preventative Services Task Force; FORE, Foundation for Osteoporosis Research and Education; OSC, Osteoporosis Society of Canada; BMI, body mass index; BMD, bone mineral density.
REFERENCES
- 1.Marshall D, Joyhnell O, Weddel H. Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ. 1996;312:1254–9. doi: 10.1136/bmj.312.7041.1254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Woodhouse A, Black DM. BMD at various sites for the prediction of hip fracture: a meta-analysis. J Bone Miner Res. 2002;15:S145. [Google Scholar]
- 3.Cummings SR, Black DM, Nevitt MC, et al. Bone density at various sites for prediction of hip fractures. Lancet. 1993;341:72–5. doi: 10.1016/0140-6736(93)92555-8. [DOI] [PubMed] [Google Scholar]
- 4.Centers for Disease Control and Prevention. Healthy People 2010. Available at: http://www.health.gov/healthypeople/, volume1/02Arthritis.htm#_Toc49053811. Accessed April 30, 2004.
- 5.Consensus Development Panel. National Institutes of Health Consensus Development Conference Statement: Osteoporosis Prevention, Diagnosis, and Therapy. Rockville, Md: National Institutes of Health; 2000. p. 27. [Google Scholar]
- 6.Solomon DH, Morris CA, Cheng H, et al. A structured review of treatment guidelines, osteoporosis treatment rates, and quality improvement interventions. Under review. [DOI] [PubMed]
- 7.American Association of Clinical Endocrinologists. American Association of Clinical Endocrinologists 2001 medical guidelines for clinical practice for the prevention and management of postmenopausal osteoporosis. Endocr Pract. 2001;7:294–312. [PubMed] [Google Scholar]
- 8.Hodgson SF, Johnston CC, Avioli LV, et al. AACE clinical practice guidelines for the prevention and treatment of postmenopausal osteoporosis. Endocr Pract. 1996;2:157–71. [Google Scholar]
- 9.American College of Rheumatology Ad Hoc Committee on Glucocorticoid-induced Osteoporosis. Recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis: 2001 update. Arthritis Rheum. 2001;44:1496–503. doi: 10.1002/1529-0131(200107)44:7<1496::AID-ART271>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 10.American College of Rheumatology Task Force on Osteoporosis Guidelines. Recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Rheum. 1996;39:1791–801. doi: 10.1002/art.1780391104. [DOI] [PubMed] [Google Scholar]
- 11.Collier JD, Ninkovic M, Compston JE. Guidelines on the management of osteoporosis associated with chronic liver disease. Gut. 2002;50(suppl 1):1–9. doi: 10.1136/gut.50.suppl_1.i1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Compston J. Prevention and treatment of osteoporosis: clinical guidelines and new evidence. J R Coll Physicians Lond. 2000;34:518–21. [PMC free article] [PubMed] [Google Scholar]
- 13.Guideline Development Group of the Royal College of Physicians. Osteoporosis: Clinical Guidelines for Prevention and Treatment. London: Royal College of Physicians; 1999. [Google Scholar]
- 14.Eastell R, Boyle IT, Compston J, et al. Management of male osteoporosis: report of the UK Consensus Group. QJM. 1998;91:71–92. doi: 10.1093/qjmed/91.2.71. [DOI] [PubMed] [Google Scholar]
- 15.Eastell R, Reid DM, Compston J, et al. A UK Consensus Group on management of glucocorticoid-induced osteoporosis: an update. J Intern Med. 1998;244:271–92. doi: 10.1046/j.1365-2796.1998.00408.x. [DOI] [PubMed] [Google Scholar]
- 16.National Osteoporosis Society. Guidance on the Prevention and Management of Corticosteroid-induced Osteoporosis. Bath, UK: National Osteoporosis Society; 1998. [Google Scholar]
- 17.Kanis JA, Delmas P, Burckhardt P, et al. The European Foundation for Osteoporosis and Bone Disease, for guidelines for diagnosis and management of osteoporosis. Osteoporos Int. 1997;7:390–406. doi: 10.1007/BF01623782. [DOI] [PubMed] [Google Scholar]
- 18.Meunier PJ, Delmas PD, Eastell R, et al. Diagnosis and management of osteoporosis in postmenopausal women: clinical guidelines. International Committee for Osteoporosis Clinical Guidelines. Clin Ther. 1999;21:1025–44. doi: 10.1016/s0149-2918(99)80022-8. [DOI] [PubMed] [Google Scholar]
- 19.National Osteoporosis Foundation. Osteoporosis: Review of the Evidence for Prevention, Diagnosis, and Treatment and Cost-effectiveness Analysis. Washington, DC: The Foundation; 1998. [Google Scholar]
- 20.National Osteoporosis Foundation. Physician's Guide to the Prevention and Treatment of Osteoporosis. Available at: http://www.nof.org. Accessed July 18, 2002.
- 21.Osteoporosis Clinical Practice Guideline. Columbia, Md: American Medical Directors Association; 1998. and American Health Care Association, Washington, DC. [Google Scholar]
- 22.Sturtridge W, Lentle B, Hanley D. The use of bone density measurement in the diagnosis and management of osteoporosis. CMAJ. 1996;155:924–9. [PMC free article] [PubMed] [Google Scholar]
- 23.The prevention and management of osteoporosis: consensus statement. Australian National Consensus Conference 1996. Med J Aust. 1997;167(suppl):S1–S15. [PubMed] [Google Scholar]
- 24.U.S. Preventive Services Task Force. Guide to Clinical Preventive Services: Report of the USPreventive Services Task Force. Baltimore, Md: Williams & Wilkins; 1996. [Google Scholar]
- 25.U.S. Preventive Services Task Force. Screening for osteoporosis in postmenopausal women: recommendations and rationale. Ann Intern Med. 2002;137:526–8. doi: 10.7326/0003-4819-137-6-200209170-00014. [DOI] [PubMed] [Google Scholar]
- 26.Adachi JD, Olszynski WP, Hanley DA, et al. Management of corticosteroid-induced osteoporosis. Semin Arthritis Rheum. 2000;29:228–51. doi: 10.1016/s0049-0172(00)80011-6. [DOI] [PubMed] [Google Scholar]
- 27.Foundation for Osteoporosis Research and Education. Guidelines of Care on Osteoporosis for the Primary Care Physician. Oakland, Calif: Foundation for Osteoporosis Research and Education; 1998. [Google Scholar]
- 28.Sambrook PN, Seeman E, Phillips SR, et al. Preventing osteoporosis: outcomes of the Australian Fracture Prevention Summit. Med J Aust. 2002;176:S1–S16. doi: 10.5694/j.1326-5377.2002.tb04475.x. [DOI] [PubMed] [Google Scholar]
- 29.Brown JP, Josse RG. Clinical practice guidelines for the diagnosis and management of osteoporosis in Canada. CMAJ. 2002;167(10 suppl):S1–S34. [PMC free article] [PubMed] [Google Scholar]
- 30.Scientific Advisory Board, Osteoporosis Society of Canada. Clinical practice guidelines for the diagnosis and management of osteoporosis. CMAJ. 1996;155:1113–29. [PMC free article] [PubMed] [Google Scholar]
- 31.Sahota O, Worley A, Hosking DJ. An audit of current clinical practice in the management of osteoporosis in Nottingham. J Public Health Med. 2000;22:466–72. doi: 10.1093/pubmed/22.4.466. [DOI] [PubMed] [Google Scholar]
- 32.Gallagher TC, Geling O, Comite F. Missed opportunities for prevention of osteoporosis fracture. Arch Intern Med. 2002;162:450–6. doi: 10.1001/archinte.162.4.450. [DOI] [PubMed] [Google Scholar]
- 33.Colon-Emeric C, Yballe L, Sloane R, Pieper CF, Lyles KW. Expert physician recommendations and current practice patterns for evaluating and treating men with osteoporotic hip fracture. J Am Geriatr Soc. 2000;48:1261–3. doi: 10.1111/j.1532-5415.2000.tb02599.x. [DOI] [PubMed] [Google Scholar]
- 34.Ridout R, Hawker GA. Use of bone densitometry by Ontario family physicians. Osteoporos Int. 2000;11:393–9. doi: 10.1007/s001980070105. [DOI] [PubMed] [Google Scholar]
- 35.Solomon CG, Connelly MT, Collins K, Okamura K, Seely EW. Provider characteristics: impact on bone density utilization at a health maintenance organization. Menopause. 2000;7:391–4. doi: 10.1097/00042192-200011000-00004. [DOI] [PubMed] [Google Scholar]
- 36.Papa LJ, Weber BE. Physician characteristics associated with the use of bone densitometry. J Gen Intern Med. 1997;12:781–3. doi: 10.1046/j.1525-1497.1997.07165.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jaglal SB, McIsaac WJ, Hawker G, Jaakkimainene L, Cadarette SM, Chan BTB. Patterns of use of the bone mineral density test in Ontario, 1992–1998. CMAJ. 2000;163:1139–43. [PMC free article] [PubMed] [Google Scholar]
- 38.Harrington JT, Broy SB, DeRosa AM, et al. Hip fracture patients are not treated for osteoporosis: a call for action. Arthritis Care Res. 2002;47:651–4. doi: 10.1002/art.10787. [DOI] [PubMed] [Google Scholar]
- 39.Kiebzak GM, Beinart GA, Perser K, et al. Undertreatment of osteoporosis in men with hip fracture. Arch Intern Med. 2002;162:2217–22. doi: 10.1001/archinte.162.19.2217. [DOI] [PubMed] [Google Scholar]
- 40.Riley RL, Carnes ML, Gudmundsson A, Elliott ME. Outcomes and secondary prevention strategies for male hip fractures. Ann Pharmacother. 2002;36:17–23. doi: 10.1345/aph.1A094. [DOI] [PubMed] [Google Scholar]
- 41.Smith MD, Ross W, Ahern MJ. Missing a therapeutic window of opportunity: an audit of patients attending a tertiary teaching hospital with potentially osteoporotic hip and wrist fractures. J Rheumatol. 2001;28:2504–8. [PubMed] [Google Scholar]
- 42.Freedman KB, Kaplan FS, Bilker WB, Strom BL, Lowe RA. Treatment of osteoporosis: are physicians missing an opportunity? J Bone Joint Surg Am. 2000;82-A:1063–70. doi: 10.2106/00004623-200008000-00001. [DOI] [PubMed] [Google Scholar]
- 43.Hajcsar EE, Hawker G, Bogoch ER. Investigation and treatment of osteoporosis in patients with fragility fractures. CMAJ. 2000;163:819–22. [PMC free article] [PubMed] [Google Scholar]
- 44.Kamel HK, Hussain MS, Tariq S, Perry HM, III, Morley JE. Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture. Am J Med. 2000;109:326–8. doi: 10.1016/s0002-9343(00)00457-5. [DOI] [PubMed] [Google Scholar]
- 45.Gudbjornsson B, Juliusson UI, Gudjonsson FV. Prevalence of long term steroid treatment and the frequency of decision making to prevent steroid induced osteoporosis in daily clinical practice. Ann Rheum Dis. 2002;61:32–6. doi: 10.1136/ard.61.1.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Solomon DH, Katz JN, Jacobs JP, La Tourette AM, Coblyn J. Management of glucocorticoid-induced osteoporosis in patients with rheumatoid arthritis—rates and predictors of care in an academic rheumatology practice. Arthritis Rheum. 2002;46:3136–42. doi: 10.1002/art.10613. [DOI] [PubMed] [Google Scholar]
- 47.Ettinger B, Chidambaran P, Pressman A. Prevalence and determinants of oteoporosis drug prescription among patients with high exposure to glucocorticoid drugs. Am J Manag Care. 2001;7:597–605. [PubMed] [Google Scholar]
- 48.Mudano A, Allison J, Hill J, Rothermel T, Saag K. Variations in glucocorticoid-induced osteoporosis prevention in a managed care cohort. J Rheumatol. 2001;28:1298–305. [PubMed] [Google Scholar]
- 49.Smith MD, Cheah SP, Taylor K, Ahern MJ. Prevention of corticosteroid induced osteoporosis in inpatients recently discharged from a tertiary teaching hospital. J Rheumatol. 2001:566–70. [PubMed] [Google Scholar]
- 50.Yood RA, Harrold LR, Fish L, et al. Prevention of glucocorticoid-induced osteoporosis: experience in a managed care setting. Arch Intern Med. 2001;161:1322–7. doi: 10.1001/archinte.161.10.1322. [DOI] [PubMed] [Google Scholar]
- 51.Osiri M, Saag KG, Ford AM, Moreland LW. Practice pattern variation among internal medicine specialists in the prevention of glucocorticoid-induced osteoporosis. J Clin Rheumatol. 2000;6:117–22. doi: 10.1097/00124743-200006000-00001. [DOI] [PubMed] [Google Scholar]
- 52.Buckley LM, Marquez M, Feezor R, Ruffin DM, Benson LL. Prevention of corticosteroid induced osteoporosis: results of a patient survey. Arthritis Rheum. 1999;42:1736–9. doi: 10.1002/1529-0131(199908)42:8<1736::AID-ANR24>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 53.Chevalley T, Hoffmeyer T, Bonjour JP. An osteoporosis clinical pathway for the medical management of patients with low-trauma fracture. Osteoporosis Int. 2002;13:450–5. doi: 10.1007/s001980200053. [DOI] [PubMed] [Google Scholar]
- 54.Wroe A, Salkovskis PM, Rimes KA. The effect of nondirective questioning on women's decisions whether to undergo bone density screening: an experimental study. Health Psychol. 2000;19:181–91. doi: 10.1037//0278-6133.19.2.181. [DOI] [PubMed] [Google Scholar]
- 55.Newman ED, Starkey RH, Ayoub WT, et al. Osteoporosis disease management: best practices from the Penn State Geisinger Health System. J Clin Outcomes Manage. 2000;7:23–8. [Google Scholar]
- 56.Pazirandeh M. Does patient partnership in continuing medical education (CME) improve the outcome in osteoporosis management? J Contin Educ Health Prof. 2002;22:142–51. doi: 10.1002/chp.1340220303. [DOI] [PubMed] [Google Scholar]
- 57.Stock JL, Waud CE, Corderre JA, et al. Clinical reporting to primary care physicians leads to increased use and understanding of bone densitometry and affects the management of osteoporosis. Ann Intern Med. 1998;128:996–9. doi: 10.7326/0003-4819-128-12_part_1-199806150-00006. [DOI] [PubMed] [Google Scholar]
- 58.Etzioni R, Bery KM, Legler JM, Shaw P. Prostate-specific antigen testing in black and white men: an analysis of Medicare claims from 1991–1998. Urology. 2002;59:251–5. doi: 10.1016/s0090-4295(01)01516-3. [DOI] [PubMed] [Google Scholar]
- 59.Ruffin MT, Gorenflo DW, Woodman B. Predictors of screening for breast, cervical, colorectal, and prostatic cancer among community-based primary care practices. J Am Board Fam Pract. 2000;13:1–10. doi: 10.3122/jabfm.13.1.1. [DOI] [PubMed] [Google Scholar]
- 60.Anonymous. State-specific cholesterol screening trends—United States, 1991–1999. MMWR Morb Mortal Wkly Rep. 2000;49:750–5. [PubMed] [Google Scholar]
- 61.Moss K, Keen R. Usefulness of bone densitometry in postmenopausal women with clinically diagnosed vertebral fractures (letter) Ann Rheum Dis. 2002;61:667–8. doi: 10.1136/ard.61.7.667-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Nolla JM, Gomez-Vaquero C, Fiter C, et al. Usefulness of bone densitometry in postmenopausal women with clinically diagnosed vertebral fractures. Ann Rheum Dis. 2002;61:73–5. doi: 10.1136/ard.61.1.73. [DOI] [PMC free article] [PubMed] [Google Scholar]