Abstract
Vac8p is a vacuolar membrane protein that is required for efficient vacuole inheritance and fusion, cytosol-to-vacuole targeting, and sporulation. By analogy to other armadillo domain proteins, including β-catenin and importin α, we hypothesize that Vac8p docks various factors at the vacuole membrane. Two-hybrid and copurfication assays demonstrated that Vac8p does form complexes with multiple binding partners, including Apg13p, Vab2p, and Nvj1p. Here we describe the surprising role of Vac8p-Nvj1p complexes in the formation of nucleus–vacuole (NV) junctions. Nvj1p is an integral membrane protein of the nuclear envelope and interacts with Vac8p in the cytosol through its C-terminal 40–60 amino acids (aa). Nvj1p green fluorescent protein (GFP) concentrated in small patches or rafts at sites of close contact between the nucleus and one or more vacuoles. Previously, we showed that Vac8p-GFP concentrated in intervacuole rafts, where is it likely to facilitate vacuole-vacuole fusion, and in “orphan” rafts at the edges of vacuole clusters. Orphan rafts of Vac8p red-sifted GFP (YFP) colocalize at sites of NV junctions with Nvj1p blue-sifted GFP (CFP). GFP-tagged nuclear pore complexes (NPCs) were excluded from NV junctions. In vac8-Δ cells, Nvj1p-GFP generally failed to concentrate into rafts and, instead, encircled the nucleus. NV junctions were absent in both nvj1-Δ and vac8-Δ cells. Overexpression of Nvj1p caused the profound proliferation of NV junctions. We conclude that Vac8p and Nvj1p are necessary components of a novel interorganelle junction apparatus.
INTRODUCTION
The yeast vacuole, like the animal lysosome, is an acidified organelle filled with soluble protein hydrolases that degrade proteins, lipids, nucleic acids, and carbohydrates (Jones et al., 1997). Vacuole biogenesis occurs by several routes. Most vacuolar membrane proteins and soluble vacuolar hydrolases are initially transported into the endoplasmic reticulum (ER). The ER in Saccharomyces cerevisiae is limited to the nuclear envelope (NE) and ribbon-like peripheral cisternae (peripheral ER) (Preuss et al., 1991; Shaywitz et al., 1997). Vacuole proteins pass through the ER to the Golgi apparatus before being delivered to the vacuole by either of two known routes (reviewed in Conibear and Stevens, 1998). The carboxypeptidase Y (CPY) pathway directs vesicular cargo through a prevacuolar compartment, which is analogous to the endosome of animal cells (Vida et al., 1993). The second pathway, which delivers alkaline phosphatase (ALP) and a vacuolar T-snare (Vam3p), bypasses the prevacuolar compartment and requires the clathrin adaptor protein-related complex AP-3 (Conibear and Stevens, 1998). A third distinct route for the delivery of vacuolar proteins is the cytosol-to-vacuole targeting (Cvt) pathway, which delivers pre-aminopeptidase I (API) directly from the cytosol to the vacuole. API is sequestered in small double-membrane vesicles that ultimately fuse with the vacuole (Baba et al., 1997; Kim et al., 1997; Scott et al., 1996, 1997; Klionsky and Ohsumi, 1999).
A major function of the vacuole is the autophagocytic degradation and recycling of cell components. Autophagy of the cytoplasm serves to recycle cell components, and is significantly induced by starvation and various metabolic signals (Jones et al., 1997), during normal growth. Macroautophagy involves the formation of double-membrane autophagosomes around bulk cytoplasm and organelles (Takeshige et al., 1992; Baba et al., 1994). Fusion of mature autophagosomes with vacuoles releases single-membrane autophagic bodies into the vacuole lumen where they are degraded and their contents recycled (Takeshige et al., 1992). Macroautophagy and the Cvt pathway in S. cerevisiae are morphologically similar processes (Baba et al., 1997) that are mediated by an overlapping set of gene products (Scott et al., 1996). Thus, most Cvt and autophagy mutants are defective in both API targeting and macroautophagy.
VAC8 was originally identified in a screen for genes required for the mitotic transfer of vacuoles from mother to daughter cells (Wang et al., 1996). vac8-Δ cells are also defective in Cvt targeting and contain fragmented vacuoles (Wang et al., 1998; Pan and Goldfarb, 1998; Fleckenstein et al., 1998). Vacuole fragmentation is thought to indicate a defect in vacuole–vacuole fusion. Vac8p is anchored in the vacuole membrane by N-terminal myristate and palmitate moieties. Vac8p localizes over the entire surface of the vacuole (Wang et al., 1998) but concentrates in rafts between vacuoles (intervacuolar rafts) and at the edges of clustered vacuoles (orphan rafts) (Pan and Goldfarb, 1998; this study). The domain structure of Vac8p is similar to a small family of proteins that includes β-catenin and karyopherin/importin α. These proteins contain central Armadillo (Arm) domains composed of ≥ 10 tandem ∼42-aa Arm repeats bracketed by short N- and C-terminal domains. X-ray crystal structures revealed that Arm domains fold into right-handed superhelical structures with extended grooves that serve as surfaces for binding protein cargo (Huber et al., 1997; Conti et al., 1998). Arm repeats are related to HEAT motifs (Malik et al., 1997), which fold into polypeptides with similar structures and protein-binding characteristics (Groves et al., 1999; Vetter et al., 1999; Chook et al., 1999; Cingolani et al., 1999). In large part, α-catenin and importin α are adapters that mediate the docking of various cargo at their cognate membranes. The experiments in this article indicate that Vac8p plays an analogous role in docking proteins at the vacuole membrane. Surprisingly, we find that Vac8p interacts with Nvj1p to mediate the formation of NV junctions.
MATERIALS AND METHODS
Yeast Strains, Plasmids, and Growth Conditions
Yeast strains used in this study were YEF473a (MATa), YEF473α (MATα), YEF473 (MATa/MATα) trp1-Δ63 leu2-Δ1 ura3–52 his3-Δ200 lys2–801, and L40 (MATa) his-Δ200 trp1–901 leu2–3112 ade2 LYS2::(lexAop)4-HIS3 URA3::(lexAop)8-lacZ GAL4. pRK2 and pJN40 (obtained from D. Hinkle, University of Rochester), derived from YEplac181 and YEplac195 (Gietz and Sugino, 1988), were used to control expression using the CUP1 promoter (PCUP1) (Macreadie et al., 1989; Ward et al., 1994). pRK2 and pJN40 were constructed by inserting a BamHI/Klenow–EcoRI 443-base pair fragment encompassing the PCUP1 into a Nar/Klenow–EcoRI cut YEplac181. BamHI and EcoRI sites both were reformed. pNUP188-GFP was created by inserting polymerase chain reaction (PCR)-amplified NUP188 into the HindIII and ClaI sites of pGFP-C-fus (Neidenthal et al., 1996). Yeast genes were amplified by PCR from genomic DNA, GFP from pGFP-C-fus (Niedenthal et al., 1996), YFP/CFP from pEYFP/pECFP (Clontech, Palo Alto, CA), and glutathione-S-transferase (GST) was from pGEX-2TK (Pharmacia Biotech, Piscataway, NJ). Expand Hi-Fi DNA polymerase (Boehringer Mannheim, Indianapolis, IN) was used for all PCR amplifications. nvj1-Δ null cells were created by replacing NVJ1 with a kanamycin resistance marker (kanr) cassette, as described (Guldener et al., 1996; Wach, 1996). A similar strategy was employed to produce vac7-Δ cells. A chromosomal cNVJ1-GFP gene fusion was similarly constructed using a PCR-amplified NVJ1-YFP-kanr cassette. kanr replacements were verified by PCR with appropriate primers. Cells were cultured in standard YPD/YEPD and SCGlu media (Sherman, 1986). Nitrogen starvation medium (SD-N) contained 0.17% yeast nitrogen base without aa and without ammonium sulfate plus 2% glucose.
Diploid nvj1-Δ cells were obtained by mating haploid YEF473a nvj1-Δ + pNJ (URA3 marker) with YEF473α nvj1-Δ + pRK (LEU2 marker). Diploid cells were grown in YEPA (1% bacto-yeast extract, 2% peptone, and 2% potassium acetate) for at least five generations to a titer of ∼1 × 107 cells/ml, were washed once in sterile ddH2O, and were resuspended in SPM (1% potassium acetate, 0.02% raffinose, plus appropriate aa supplements at 25% of the level for complete medium). Asci began to develop within 24 h. Attempts to induce diploid vac8-Δ cells to sporulate included using liquid medium, as described above, and solid medium (1% potassium acetate, 0.1% bacto-yeast extract, 0.05% dextrose, appropriate aa supplements at 25% of the level for complete medium, and 2% bacto-agar) with or without prior incubation on presporulation medium (Sherman, 1986).
Glutathione Sepharose Affinity Chromatography
nvj1-Δ::kanr cells containing pRK-GST or pRK-GST-NVJ1 were used to purify GST or GST-Nvj1p using glutathione Sepharose 4B (Pharmacia). For 3 h, 0.1 mM CuSO4 was added to early log cells (OD600 = 0.3–0.7) in SCGlu in order to induce the expression of GST or GST-NVJ1. Total cell lysates were prepared from ∼ 1 g of cells by vortexing with glass beads in 2 ml PBS (137 mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 1.76 mM KH2PO4) containing protease inhibitors (Sigma, St. Louis, MO). Cells were vortexed again after NP-40 was added to a final concentration of 1%. The lysate was clarified by centrifugation at 5,000 × g and then at 100,000 × g for 30 min at 4°C. The supernatant was diluted 10 times with PBS before it was incubated with 120 μl of a 50% suspension of glutathione Sepharose 4B beads (Pharmacia Biotech) per gram of protein of starting cell pellet. After rocking overnight at 4°C, the beads were collected, washed three times with 10-bed volumes of PBS at 4°C, and eluted with 75 μl of standard protein gel sample loading buffer. Immunoblots were probed with rabbit anti-Vac8p serum at 1:50,000 dilution in TST buffer (20 mM Tris-HCl, pH 7.6, 150 mM NaCl, 0.05% Tween 20) containing 0.2% nonfat dry milk and developed using ALP-conjugated secondary antibodies (1:3000) (Bio-Rad, Richmond, CA).
Two-Hybrid Screen
An EcoRI–BamHI restriction fragment encoding VAC8 was inserted into the bait plasmid pBTM116 (Bartel and Fields, 1995) to produce pBTM116-VAC8. L40 cells (Vojtek et al., 1993) harboring pBTM116-VAC8 were transformed with a yeast cDNA library (87002; American Type Culture Collection, Manassas, VA) by the lithium acetate procedure (Schiestl and Gietz, 1989). Approximately 105 transformants were pooled and replated at 1:1000 on plates supplemented with 3 mM 3-amino-1,2,4-triazole (3-AT) (Sigma). Extracts from cells that grew on −His, + 3-AT media were assayed for β-galactosidase activity, as described, using L40 cells containing pBTM116-VAC8, and a plasmid encoding only the GAL4 activation domain was used as a negative control (Bartel and Fields, 1995). The regions of Nvj1p interacting with Vac8p were identified by PCR-amplifying portions of NVJ1 and cloning the PCR products into pGAD424 (Clontech). The resulting plasmids were transformed into L40 cells containing pBTM116-VAC8.
Cell Fractionation
Cells of 600 ml weight containing either pRK-Nvj1p-GFP or pRK-GFP-NVJ1 were grown to an OD600 of 0.45 and were supplemented for 2.75 h with 0.1 mM CuSO4. Cells were pelleted and resuspended in 2 ml PBS containing protease inhibitors (Boehringer Mannheim). An equal volume of glass beads was added, and the cells were broken by vortexing at 4°C. Suspensions were cleared by centrifugation at 500 × g for 15 min. Each supernatant was divided into four aliquots, adjusted to 1 ml with PBS, and centrifuged at 100,000 × g at 4°C for 30 m. S100 supernatants were collected leaving P100 pellets, which were resuspended in 1 ml PBS, 1% NP-40 in PBS, 1 M NaCl in PBS, or 0.1 mM Na2CO3 (pH 11.5). Suspensions then were centrifuged at 100,000 × g. The resulting supernatants were collected, and the pellets solubilized in 1 ml protein gel sample loading buffer. Equal amounts of extracts from ∼1.4 OD600 equivalents (∼ 1.5 ml of original culture) were used for immunoblotting, as described above. Anti-GFP antiserum (Clontech) was used at 1:20,000, and the Vac8p antibody was used at 1:50,000.
Analysis of API processing
Midlog-phase cells (2 × 107) were incubated in 4 ml SD-N (nitrogen starvation conditions) or 4 ml YEPD for 4 h at 30°C. Cells were collected, resuspended in 400 μl of 50 mM sodium phosphate, 20 mM MES, pH 7.0, 1% SDS, 3 M urea, 1 mM sodium azide, and 2 mM phenylmethanesulfonate, and lysed with 0.15-g glass beads by vortexing 3 × 1 m. After the addition of SDS sample buffer, lysates were spun at 14,000 × g for 10 min and extracts from ∼0.5-OD equivalents were analyzed by SDS-PAGE. API was detected by immunoblotting using a rabbit anti-API polyclonal antiserum (1/10,000 dilution, gift of D. Klionsky), followed by HRP-conjugated goat-antirabbit secondary antibody (Zymed, San Francisco, CA) and chemilumescent detection (ECL, Amersham, Arlington Heights, IL).
Anti-Vac8p Antibodies
A BamHI–EcoRI restriction fragment encoding Vac8p was inserted into the bacterial GST fusion expression vector pGEX-2TK (Pharmacia). The GST–Vac8p fusion protein was affinity purified from BLR Escherichia coli cells (Novagen, Madison, WI) using glutathione Sepharose 4B (Pharmacia). Full-length Vac8p was released from GST by thrombin (Sigma). Polyclonal antibodies were prepared by Pocono Rabbit Farm & Laboratory (Canadensis, PA).
Microscopy
A Nikon fluorescence microscope equipped with a SPlan 100 objective (NA 1.25), an Image1 (Universal Imaging, West Chester, PA) controlled CCD camera, and filter sets for YFP and CFP (sets 41028 and 31044; Chroma Technology, Brattleboro, VT) were used to capture CFP and YFP images. Confocal microscopy was performed on a Leica TCS NT microscope (Zeiss, Thornwood, NY) equipped with UV, Ar, Kr/Ar, and He/Ne lasers. Images were processed using MetaMorph (Universal Imaging) or Photoshop 5.0 (Adobe Systems, San Jose, CA). FM4–64 staining of vacuoles was performed as previously described (Pan and Goldfarb, 1998).
Cells were prepared for electron microscopy basically as described (Banta et al., 1988). Briefly, logarithmically growing cells were harvested by centrifugation and were fixed in 3% glutaraldehyde in buffer A (170 mM KH2PO4, 0.1 mM MgCl2, and 30 mM sodium citrate, at pH 5.8) for 2 h at 22°C. Cells were washed once in buffer A, resuspended in a minimal volume of buffer A containing 1 M sorbitol and zymolyase-100T (ICN Biomedicals, Costa Mesa, CA), and incubated for 2 h at 30°C. Spheroplasts were washed two times in buffer A before post fixation with osmium and embedding in Spurr's resin.
RESULTS
Nvj1p interacts with Vac8p
The yeast two-hybrid screen was used to identify putative Vac8p binding partners. Vac8p fused to the lexA DNA-binding domain was used to screen a yeast cDNA library fused to the GAL4 activation domain (Bartel and Fields, 1995; Vojtek and Hollenberg, 1995). From ∼105 two-hybrid clones, partial cDNA fragments encoding five different putative Vac8p binding proteins were isolated multiple times. The interactions of Vac8p with three of these proteins, Apg13p, Vab2p, and Nvj1p, were further characterized. APG13 was originally identified in a screen for genes required for viability in starvation medium (Tsukada and Ohsumi, 1993) and was subsequently shown to be required for autophagy (Funakoshi et al., 1997). VAB2 (Vac8 binding protein 2) is encoded by an uncharacterized ORF (YEL005c) with no similarity to known proteins. The third, encoded by the uncharacterized locus YHR195w, was named Nvj1p for its role in NV junctions (see below).
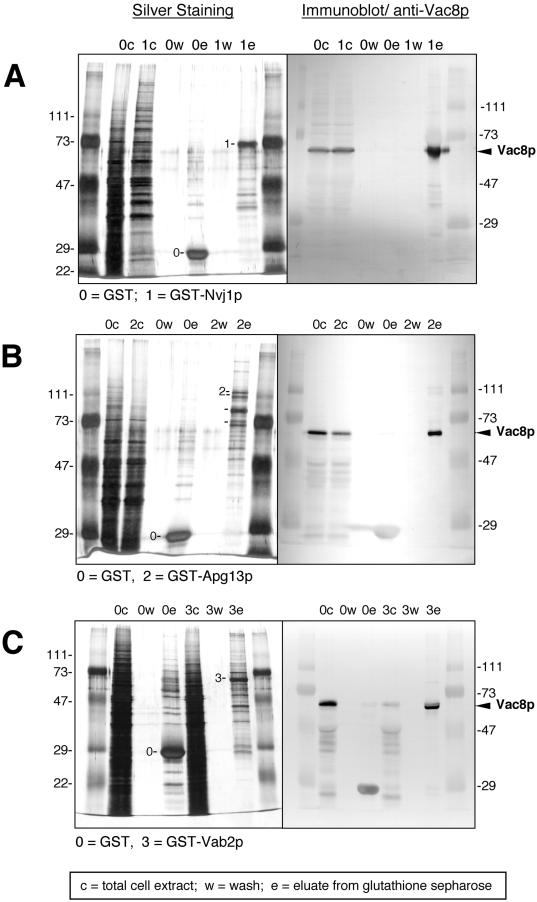
The two hybrid interactions were corroborated by the copurification of Vac8p with the three putative binding partners. When whole-cell extracts expressing GST fusions of the binding partners, or GST alone, were incubated with glutathione Sepharose beads, significant amounts of Vac8p coeluted with GST-Nvj1p (Figure 1A), GST-Apg13p (Figure 1B), and GST-Vab2p (Figure 1C). In each case, the GST-fusion proteins appeared as silver-stained bands (Figure 1, A to C, left panels). Vac8p was detected on immunoblots using anti-Vac8p antibodies (Figure 1, A to C, right panels). These interactions were specific since Vac8p was absent when extracts from cells expressing GST alone were treated in parallel (Figure 1, A to C). In addition, Nvj1p with a C-terminal GFP tag copurified by glutathione affinity chromatography with Vac8p-GST (our unpublished results). Therefore, both N-terminal (GST-Nvj1p) and C-terminal (Nvj1p-GFP) fusions retain the capacity to associate with Vac8p. Although we have presumed that the two-hybrid and copurification results indicate a direct interaction between Vac8p and the three putative binding partners, it remains a remote possibility that one or more of the interactions are indirect. In this study, we have focused on the interaction between Vac8p and Nvj1p. Possible implications regarding the interactions of Apg13p and Vab2p with Vac8p are presented in the Discussion section.
Figure 1.
Glutathione–Sepharose affinity copurification of Vac8p with three binding partners. Proteins from cell extracts (c), wash solutions (w), and eluates (e) were separated by SDS-PAGE and were analyzed by silver staining (left panels) and immunoblot using anti-Vac8p antibodies (right panels). (A) Copurification of Vac8p with GST-Nvj1p. (B) Copurification of Vac8p with GST-Apg13p. (C) Copurification of Vac8p with GST-Vab2p. Labeled bands correspond to GST (27 kDa), GST-Nvj1p (63.4 kDa), and Vac8p (63.2 kDa).
Nvj1p Fractionates as an Integral Membrane Protein
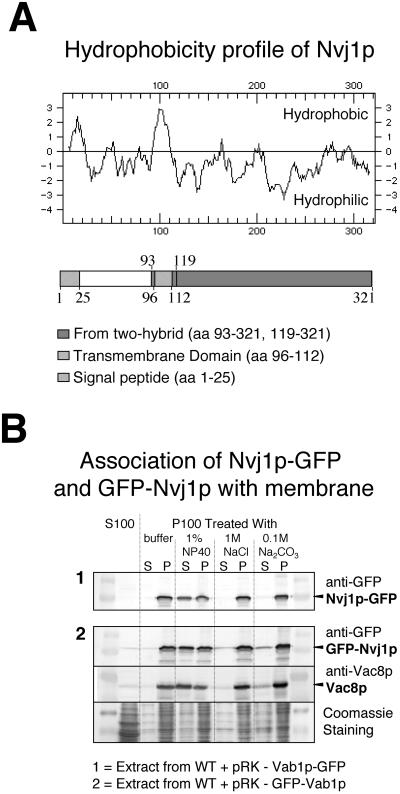
Nvj1p does not show significant sequence similarity to known proteins. A Kyte-Doolittle hydrophobicity plot and sequence analysis suggested that Nvj1p contains an N-terminal signal peptide (M1TRPPLVRGIFSLGLSVAVLKGVEK25,) and a single membrane-spanning domain (V96LILLLSFLLPIAWTVL112) (Figure 2A). The putative signal peptide is typical in that it contains a basic residue followed by a core of hydrophobic residues (Watson, 1984). Evidence that Nvj1p is indeed an integral membrane protein was obtained by biochemical fractionation. A reporter protein containing GFP fused to the C-terminus of Nvj1p (Nvj1p-GFP) was used for this purpose in order to facilitate its detection by anti-GFP antibodies. Results presented below validate the use of Nvj1p-GFP as a functional reporter. A whole-cell lysate from cells expressing Nvj1p-GFP was centrifuged to separate soluble and insoluble fractions. Immunoblot analysis of these fractions showed that Nvj1p-GFP fractionated with the particulate membrane fraction (Figure 2B). Membrane fraction-associated Nvj1p-GFP was resistant to extraction by high salt and high pH but was solubilized by nonionic detergent (NP-40) treatment (Figure 2B). Vac8p, which is anchored into the vacuole membrane by myristate and palmitate residues, also behaved as an integral membrane protein (Figure 2B). Nvj1p with an N-terminal GFP tag also fractionated as an integral membrane protein, even though its signal peptide is blocked.
Figure 2.
Nvj1p is an integral membrane protein. (A) Hydrophobicity plot and map of Nvj1p. The map shows the location of the putative N-terminal signal peptide (M1TRPPLVRGIFSLGLSVAVLKGVEK25), the significant hydrophobic region (V96LILLSFLLPIAWTVL112), and the portions of NVJ1 that were isolated in independent two-hybrid clones (aa 93–321 and 119–321). (B) Nvj1p fractionates as an integral membrane protein. Panel 1: A whole cell extract from cells expressing Nvj1p-GFP was separated into supernatant (S100) and membrane (P100) fraction by centrifugation. P100 was subsequently treated as described in Material and Methods (S, supernatant; P, pellet). The presence of Nvj1p in each fraction was assayed by immunoblot using anti-GFP antibodies. Panel 2: Extract from cells expressing GFP-Nvj1p and Vac8p were treated as described in panel 1.
C-Terminus of Nvj1p Is Sufficient and Necessary for Binding Vac8p
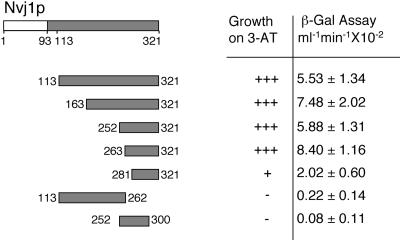
The combination of an N-terminal signal peptide and a single internal membrane-spanning domain predicts that Nvj1p is a type I integral membrane protein with its C-terminal domain exposed to the cytosol where it could interact with Vac8p. Consistent with this hypothesis, the four independently isolated NVJ1 two-hybrid clones all contained the C-terminal portion of NVJ1 but not the N-terminal domain upstream of the membrane-spanning sequence (Figure 2A). The putative Vac8p binding domain of Nvj1p was further delineated using a two-hybrid interaction assay. Based on relative growth rates on 3-AT plates and LacZ expression levels, the C-terminal 40 aa of Nvj1p (aa 281–321) were sufficient to mediate a minimal two-hybrid interaction with Vac8p; however, an additional 20 aa were necessary for a strong interaction (Figure 3). The deletion of the C-terminal 60 aa of Nvj1p abolished the two-hybrid interaction, demonstrating that these residues are also necessary for Vac8p binding. The two-hybrid assay results were corroborated by showing that a GFP reporter protein containing only the C-terminal 40 aa of Nvj1p (aa 281–321) partially localized to the vacuole membrane (our unpublished results).
Figure 3.
Two hybrid mapping of the C-terminal domain of Nvj1p that interacts with Vac8p.
NVJ1 Is Not Required for Growth or Vacuole Biogenesis
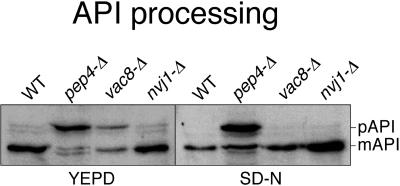
A complete disruption of NVJ1 was generated by replacing the entire ORF with a kanr cassette (Guldener et al., 1996; Wach, 1996). nvj1-Δ cells grew normally at 30 and 37°C. vac8-Δ cells exhibit a number of characteristic defects. Because Nvj1p associates with Vac8p, we asked whether nvj1-Δ cells also exhibited these phenotypes. vac8-Δ cells are defective in Cvt sorting but are normal for both CPY and ALP pathways (Wang et al., 1998). As shown in Figure 4, both vac8-Δ and pep4-Δ control cell extracts contained significant amounts of unprocessed API (∼50 and 90% respectively). In contrast, parental wt and nvj1-Δ cells contained, for the main part, fully processed API. PrA (Pep4p) is required for the proteolytic activation of PrB, which, together with PrA, activates many vacuolar hydrolases, including API after their targeting to the vacuole (Jones et al., 1997). Thus, pep4-Δ cells import but do not process preAPI. Following the induction of autophagy by shifting the cells into a nitrogen-limited medium (SD-N), only pep4-Δ cells contained unprocessed API (Figure 4). We note that using the vac8-Δ strain constructed by Wang et al., (1998), which is in a different genetic background, a virtually complete API processing defect in YEPD medium was observed, consistent with previous studies (Wang et al., 1998), and a moderate defect (∼50 unprocessed) in SD-N medium (our unpublished results). We attribute these variations in API processing to strain differences and conclude that Vac8p probably plays a role in the kinetic efficiency of both Cvt sorting and autophagy. A role for Vac8p in autophagy also is suggested by its interaction with Apg13p, and the finding that vac8-Δ cells exhibited a loss of viability in SD-N medium (our unpublished results). The key result here is that nvj1-Δ cells had no significant defect in the VAC8-dependent Cvt of AP1. nvj1-Δ cells were also normal for CPY and ALP targeting (our unpublished results).
Figure 4.
API processing is normal in nvj1-Δ cells. Midlog-phase cells were incubated for 4 h in rich (YEPD) or nitrogen starvation (SD-N) medium. Cell lysates were prepared and processed for immunoblotting with anti-API antibody. The positions of precursor and mature API are indicated.
vac8-Δ cells exhibit abnormally large numbers of small vacuoles and are defective in vacuole inheritance (Wang et al., 1998; Pan and Goldfarb, 1998) and sporulation (our unpublished results, see Material and Methods). nvj1-Δ cells were tested for these defects. The numbers of vacuoles per cell and the efficiency of vacuole inheritance (see below) and sporulation were normal in nvj1-Δ cells. Therefore, although there is strong evidence that Vac8p and Nvj1p form a complex in vivo, nvj1-Δ cells displayed none of the defects that have been described for vac8-Δ cells.
Evidence for a physiologically significant interaction between Vac8p and Nvj1p was obtained by studying the effects of overexpressing NVJ1 on vacuolar inheritance. The efficiency of vacuolar inheritance is normally expressed as the percentage of daughter cells that receive FM4–64-stained vacuoles from their mothers (Vida and Emr, 1995; Wang et al.,1998). The efficiency of vacuolar inheritance dropped from 96.9 ± 1.1% in wt cells to 6.2 ± 1.3% in vac8-Δ cells. In contrast, the deletion of NVJ1 did not affect the efficiency of vacuolar inheritance (95.9 ± 0.6%). This result indicates that the role of the Vac8p–Nvj1p interaction is unrelated to the role of Vac8p in vacuolar inheritance. Interestingly, however, the overexpression of NVJ1 in wt cells decreased the efficiency of vacuolar inheritance more than twofold to 40.8% ± 0.7. Overexpression of NVJ1-GFP had a similar effect (39.1 ± 3.6%). Thus NVJ1 overexpression has an inhibitory effect on vacuole inheritance. Because the deletion of NVJ1 did not affect vacuolar inheritance, the inhibitory effect of NVJ1 overexpression is likely to be indirect. Possibly, the pool of Vac8p that is normally employed in vacuolar inheritance is partially sequestered by excess Nvj1p. Thus, the overexpression of NVJ1 would indirectly effect vacuole inheritance by sequestering Vac8p. If true, then the coordinate overexpression of both VAC8 and NVJ1 should replete the Vac8p pool and suppress the transfer defect caused by the overexpression of NVJ1. The vacuolar inheritance defect of NVJ1 overexpressing cells was, in fact, partially suppressed from an efficiency of 40.8% to 63.7 ± 14% in wt cells by the coordinate overexpression of both genes.
Vac8p Recruits Nvj1p into NV Junctions
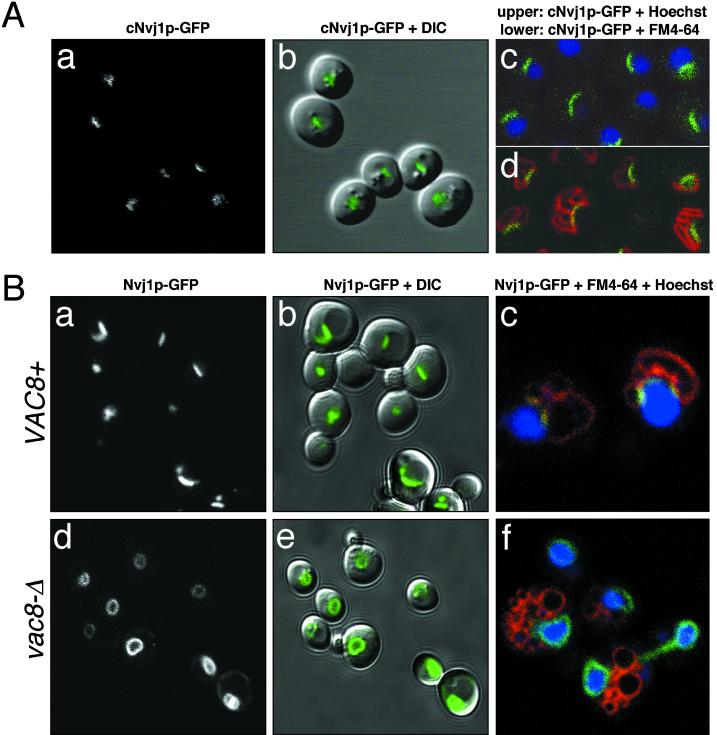
To investigate the localization of Nvj1p expressed from its own promoter, GFP was integrated into the chromosome in frame with the C-terminal codons of NVJ1 to create cNVJ1-GFP. When cells were grown to midlog, cNvj1p-GFP localized to short stripes or patches (Figure 5A, panels a and b). In stationary-phase cells, cNvj1p–GFP stripes had expanded in many cells into arcs that were sandwiched between FM4–64-stained vacuole clusters and Hoechst-stained nuclei (Figure 5A, panels c and d). The increase at higher cell densities in the length (and intensity) of cNvj1p-GFP stripes is consistent with gene array analysis, which showed that NVJ1 mRNA levels rose two- to threefold after the diauxic shift (DeRisi et al., 1997). When viewed en face, stripes appear as patches. We conclude that Nvj1p-GFP concentrates in rafts in the NE.
Figure 5.
Nvj1p-GFP localizes to a specialized region of the NE in a VAC8-dependent manner. (A) Localization of cNvj1p-GFP in wt cells grown to an OD600 of 2.8 in SCGlu (panels a and b), and in cells grown to an OD600 of 10 in YPD and costained for DNA (Hoechst) (panel c) and vacuoles (FM4–64) (panel d). (B) Nvj1p-GFP in wt (VAC8+) (panels a and b) and vac8-Δ cells (panels d and e) grown in SC medium to OD600 ∼0.1–0.3. Localization of Nvj1p-GFP in wt and vac8-Δ cells stained for DNA (Hoechst) and vacuoles (FM4–64) (panels c and f). Plasmid-borne Nvj1p-GFP was expressed uninduced from the CUP1 promoter.
The dependence of Nvj1p localization on VAC8 was determined using a plasmid copy of NVJ1-GFP expressed from the PCUP1. When cells containing this plasmid were grown in a medium containing small amounts of copper (∼0.25 μM Cu2+), PCUP1–NVJ1–GFP expression levels were held at a minimum and the localization of Nvj1p-GFP resembled that of cNvj1p-GFP (Figure 5B). In vac8-Δ cells, Nvj1p-GFP generally failed to concentrate in rafts (Figure 5B, panels d and e) and, instead, delocalized over the entire surface of the Hoechst-stained nuclei (Figure 5B, panel f). Especially at higher cell densities, some rafting of Nvj1p-GFP in vac8-Δ cells was observed. Therefore, binding to Vac8p appears to facilitate Nvj1p-GFP rafting, which still can occur at a low frequency in its absence. These results are consistent with the hypothesis that the pool of Nvj1p in the NE is recruited into rafts predominantly through direct interaction with Vac8p. The lower frequency occurrence of Vac8p-independent rafting suggests that Nvj1p may coassociate with itself. Alternatively, factor(s) beside Vac8p may facilitate rafting of Nvj1p.
Following the induction of PCUP1–NVJ1–GFP expression, elevated levels of Nvj1p-GFP concentrated in expanded rafts (see below) but at very high levels spilled out of its rafts and spread over the surface of the NE (see Figure 6C). Therefore, the amount of Nvj1p-GFP that can accumulate in rafts is determined by a limiting factor. The endogenous level of Nvj1p is normally maintained at levels sufficiently below this limit to allow low levels of ectopically expressed Nvj1p-GFP to concentrate in rafts. When expressed at levels that exceed this limit, excess Nvj1p-GFP is free to distribute randomly over the surface of the nucleus, presumably by diffusing in the plane of the NE. It is noteworthy that when expressed at fully induced levels, Nvj1p-GFP still did not spread into peripheral ER membranes. Thus, it is apparent that Nvj1p is selectively sorted to the NE and, subsequently, concentrated in rafts.
Figure 6.
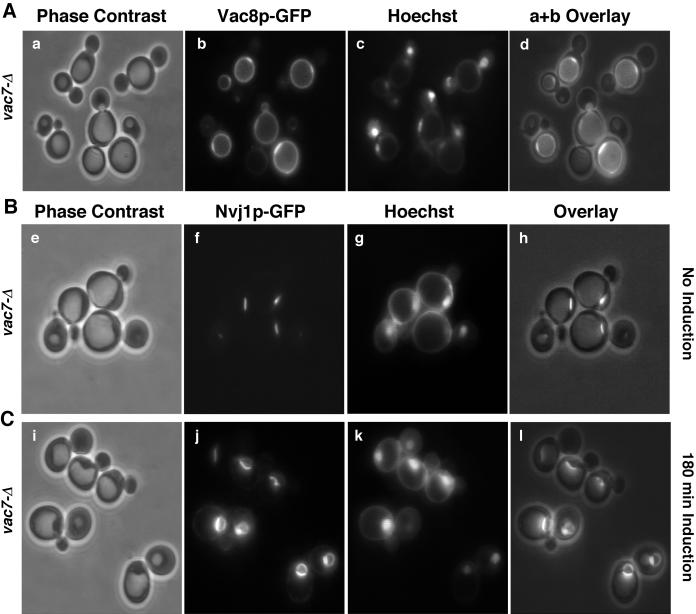
Localization of Vac8p-GFP and Nvj1p-GFP to single perinuclear rafts in vac7-Δ cells. Vac8p-GFP localization (A), Nvj1p-GFP localization before (B) and after (C) the induction of expression by 0.1 mM CuSO4. Phase contrast (panels a, e, and i), GFP (panels b, f, and j), Hoecht (panels c, g, and k), and overlay of phase contrast and Hoechst images (panels d, h, and l). Bar: 5 μm.
The localization of Vac8p in intervacuole rafts suggests that it may play a structural or regulatory role in homotypic vacuole docking and/or fusion. This notion is supported by the observation that vac8-Δ cells contain fragmented vacuoles (Pan and Goldfarb, 1998; Wang et al., 1998; Fleckenstein et al., 1998). The relationship between intervacuole and orphan rafts of Vac8p-GFP was studied in vac7-Δ cells, which contain a single enlarged vacuole (Bonangelino et al., 1997; Bryant et al., 1998; Gary et al., 1998). As shown in Figure 6A, Vac8p-GFP in vac7-Δ cells concentrated in single perinuclear rafts adjacent to Hoechst-stained nuclei. As in wt cells, Nvj1p-GFP localized to a single perinuclear patch in vac7-Δ cells when expressed at low levels (Figure 6B) and spread over the surface of the NE when overexpressed (Figure 6C). Therefore, orphan rafts of Vac8p-GFP occur independently from intervacuole rafts. In summary, intervacuole Vac8p-GFP rafts occur where vacuoles abut vacuoles, and orphan Vac8p-GFP rafts occur where vacuoles abut perinuclear rafts of Nvj1p-GFP.
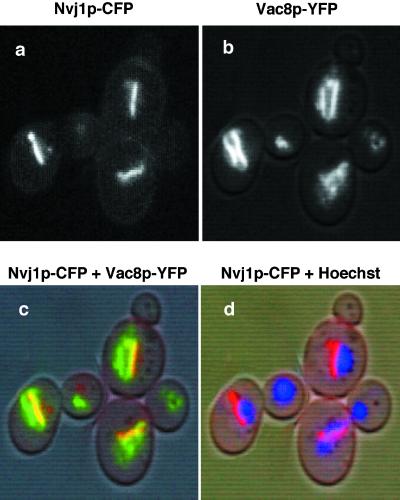
The colocalization of rafts of Vac8p and Nvj1p to NV contact sites was achieved by fusing the two proteins to different spectrally shifted GFP variants. Blue-shifted CFP (cyan) was fused to NVJ1 (Nvj1p-CFP) and red-shifted YFP (yellow) was fused to VAC8 (Vac8p-YFP). Vac8p-YFP decorated the entire vacuole surface and, as with Vac8p-GFP, accumulated in intervacuole and orphan rafts (Figure 7). Nvj1p-CFP localized to single rafts that colocalized exclusively with Vac8p-YFP orphan rafts at the edges of nuclei (Figure 7). In conclusion, orphan rafts of Vac8p-GFP colocalize with perinuclear rafts of Nvj1p-GFP.
Figure 7.
Partial colocalization of Vac8p and Nvj1p. Nvj1p-CFP (blue-shifted GFP) and Vac8p-YFP (red-shifted GFP) were localized in the same cells. Monochromic images of Nvj1p-CFP (a) and Vac8p-YFP (b), and color overlay images over a DIC mask of Nvj1p-CFP and Vac8p-YFP (c), and Nvj1p-CFP, Hoechst, and DIC (d). In panel c, yellow occurs where Nvj1p-CFP and Vac8p-YFP overlap.
Effect of NVJ1 Expression on NV Junction Morphology
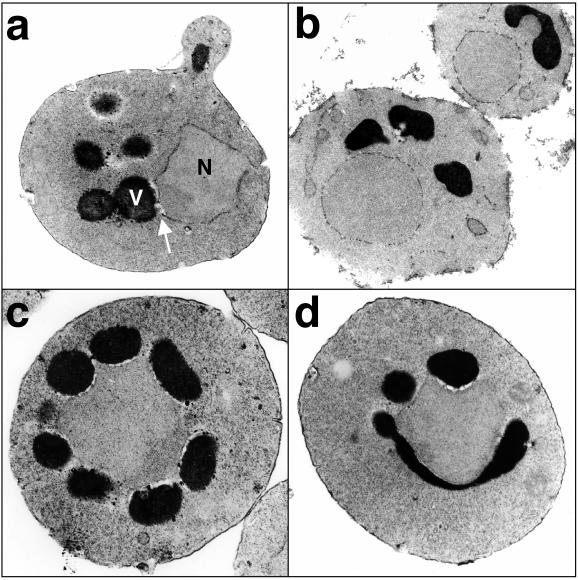
Using the electron microscope (TEM), we attempted to demonstrate a causal relationship between the expression of NVJ1 and the formation of physical junctions between the nucleus and vacuole (NV junctions). Thin sections of cells were scored for the presence of at least one area of contact between the nucleus and a vacuole. Only those thin sections exhibiting both vacuoles and a nucleus were scored. Sixty-two percent (71 of 115) of thin sections of wt cells exhibited NV contacts (Figure 8, panel a) and only 5% (6 of 113) of nvj1-Δ thin sections exhibited NV contacts (Figure 8, panel b). The rare NVJ1-independent NV contacts may represent instances where the two organelles were simply pressed together. NV contacts were also mostly absent from thin sections of vac8-Δ cells (our unpublished results, see below) indicating that both partners are required for the formation of NV junctions.
Figure 8.
NVJ1 expression levels control the occurrence and morphology of NV junctions. (A) wt cells + empty vector; (B) nvj1-Δ cells + empty vector; (C) wt cells + PCUP1-NVJ1; (D) wt cells + PCUP1--NVJ1-GFP. Overexpression from the CUP1 promoter was achieved by adding 0.1 mM CuSO4 to growth media for 3 h. Cells were subsequently fixed and prepared for TEM as described in Material and Methods.
The overexpression of NVJ1 in wt cells had a striking effect on the frequency and morphology of NV contacts. NVJ1 overexpression increased the frequency of NV contacts to 97% of thin sections (146 of 151). Both the numbers of separate contacts per cell and the extensiveness of individual junctions were increased (Figure 8, panel c). The overexpression of Nvj1p-GFP also increased the frequency of NV contacts to 95% (99 of 104) and had virtually the identical effects that elevated levels of native Nvj1p had on the morphology of NV junctions (Figure 8, panel d). These effects indicate that the total area of NV junctions in a cell is normally limited by NVJ1, and not VAC8, expression. Consistent with this conclusion, the overexpression of PGAL1-VAC8 in cells expressing cNvj1p-GFP altered neither the morphology of cNvj1p-GFP localization nor increased the area of NV junctions (our unpublished results). In addition, these results provide compelling evidence that Nvj1p-GFP is an active and valid reporter of Nvj1p function.
Nuclear Pore Complexes Are Excluded From NV Junctions
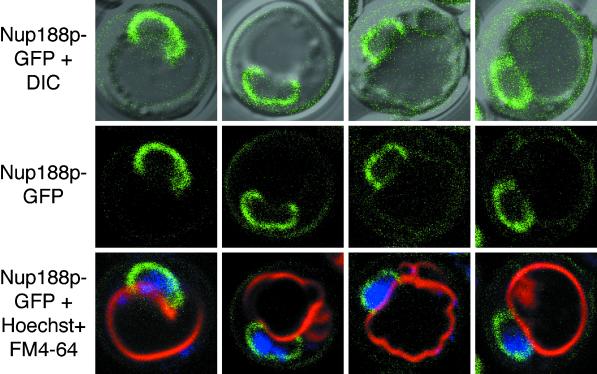
Severs et al. (1976) noted that pore complexes were absent from regions of the NE that were adjacent vacuole membranes. We sought to confirm this observation by studying the localization of Nup188p-GFP in nup188-Δ cells. Nup188p is an abundant nonessential nucleoporin that assembles with other nucleoporins in symmetrical structures located on both the cytoplasmic and nuclear faces of the pore complex (Nehrbass et al., 1996; Rout et al., 2000). As shown in Figure 9, Nup188p-GFP localized to the NE of nup188-Δ cells. Significantly, Nup188p-GFP staining was absent in contact sites between nuclei and vacuoles. These results suggest that NPCs are normally either actively or passively excluded from NV junctions.
Figure 9.
Nup188p-GFP-tagged NPCs are excluded from NV junctions. Pore complexes in nup188-Δ cells were labeled with Nup188p-GFP. Vacuoles were stained with FM4–64, and chromatin with Hoechst (Materials and Methods). Examples of four cells that exhibited large NV junctions are shown. Upper panels: Overlays of GFP-Nup188p and DIC images. Middle panels: GFP-Nup188p. Lower panels: Overlays of GFP-Nup188p, FM4–64, and Hoechst.
DISCUSSION
In this study, we define the role of Vac8p and Nvj1p in the formation of NV junctions. The discovery that the yeast nucleus and vacuole are physically linked by protein–protein interactions was foreshadowed by published observations. Foremost among these is the commonplace occurrence in published TEM images of extensive contact sites between the nuclei and vacuoles of vegetatively growing (Severs et al., 1976; Heath et al., 1995) and sporulating S. cerevisiae cells (Lynn and Magee, 1970). Second, Severs et al. (1976) noted that NPCs were absent from NV contact sites. This observation implies that some form of contact or communication, either direct or indirect, occurs between the two organelles. Finally, Koning et al. (1993) reported that the two organelles often move together in a manner that is consistent with their being attached.
Vac8p and Nvj1p Mediate NV Junctions
Four lines of evidence support the hypothesis that Vac8p and Nvj1p interact with one another to form NV junctions. First, two-hybrid and copurification assays demonstrate that Vac8p and Nvj1p form specific and stable complexes. Second, the specific topologies of Vac8p and Nvj1p within their respective membranes provide for their interaction at the interface between the nucleus and vacuole. Thus, Vac8p is posttranslationally anchored in the cytoplasmic face of the vacuole membrane by N-terminal myristate and palmitate residues, and Nvj1p is an integral membrane protein of the NE whose C-terminal domain extends into the cytosol. As predicted by this model, the cytosolic C-terminal 40–60 aa of Nvj1p are necessary and sufficient to interact with Vac8p. Third, Vac8p-GFP and Nvj1p-GFP accumulate in membrane rafts that coincide with contact sites between vacuoles and nuclei. The accumulation of Vac8p-GFP and Nvj1p-GFP in membrane rafts is dependent on the expression and proper localization of its binding partner. Thus, Nvj1p-GFP accumulated in single NE rafts in VAC8 cells but delocalized over the entire surface of the NE in vac8-Δ cells. Conversely, Vac8p-GFP orphan rafts were absent in nvj1-Δ cells (X. Pan, unpublished results). Because rafts of Nvj1p-GFP still occur at a low frequency in vac8-Δ cells, especially at high cell densities, Nvj1p may self-assemble or there may be factors in addition to Vac8p that facilitate Nvj1p rafting. Fourth, the appearance in electron micrographs of close contacts between nuclei and vacuoles was dependent on the expression of Vac8p and Nvj1p. Particularly convincing was the finding that elevated levels of Nvj1p and Nvj1p-GFP caused a marked increase in numbers and total surface area of NV junctions. This indicates that Vac8p can be recruited into NV junctions from around the vacuole when Nvj1p levels are naturally or ectopically increased. In cells expressing very high levels of Nvj1p, the available Vac8p pool can be sequestered into NV junctions so efficiently as to cause a defect in Vac8p-dependent vacuole inheritance.
The use of Nvj1p-GFP as a valid reporter is supported by functional criteria. First, Nvj1p-GFP selectively binds and copurifies with Vac8p. Second, the overexpression of Nvj1p and Nvj1p-GFP had identical effects on the efficiency of vacuolar inheritance. Third, Nvj1p-GFP successfully recruited Vac8p-GFP into NV junctions in nvj1-Δ cells (our unpublished results). Fourth, the ectopic overexpression of Nvj1p and Nvj1p-GFP had identical effects on NV junction morphology.
Previous structure–function studies on mitochondria-associated ER membranes provides some precedent for the kinds of processes that might occur at NV junctions. For example, specific steps in the biosynthesis of glycosylphosphatidylinositols, which serve to anchor proteins in membranes, occur at sites of contact between mitochondria and ER (reviewed in Vidugiriene et al., 1999). Mitochondria–ER contact sites also facilitate the transfer of lipids between ER and mitochondria in yeast (Achleitner et al., 1999) and may participate in the regulation of cytosolic Ca2+ (Vidugiriene et al., 1999). In yeast, the vacuole rather than the ER is the major repository of cellular Ca2+ (Jones et al., 1997; Strayle et al., 1999). Finally, the recycling of nuclear components by vacuolar hydrolases could be facilitated by a form of microautophagy within the context of NV junctions (Dunn, 1994; Sakai et al., 1998).
Vac8p Docks Multiple Factors at the Vacuole Membrane
In addition to defining the role of Vac8p in NV junction formation, these investigations suggest that Vac8p plays other roles in vacuole-associated processes. Three lines of evidence support this hypothesis. First, vac8-Δ cells display several phenotypes that are absent in nvj1-Δ cells, including defects in vacuole inheritance, Cvt, sporulation, and vacuole fragmentation. Second, the pattern of Vac8p-GFP localization on the surface of the vacuole suggests multiple functions. Whereas Nvj1p-GFP localizes exclusively to a single membrane patch on the surface of the NE, Vac8p localizes over the entire surface of the vacuole (Wang et al., 1998) and in at least two types of rafts, vacuole-vacuole and orphan (Pan and Goldfarb, 1998; this study). As shown here, orphan rafts correspond to NV junctions. The function of intervacuolar rafts is not known, however, they are likely to be related to the vacuole fragmentation phenotype of vac8-Δ cells. In fact, Wang et al. (1998) suggested that Vac8p has a role in vacuole fusion.
A third line of evidence is that Vac8p binds at least two other proteins, Vab2p and Apg13p, whose characteristics suggest that they function at the vacuole membrane. In addition to the interaction of Vab2p with Vac8p, Vab2p interacts by two-hybrid assay with the vacuolar ATPase subunit Vma8p (Roberts and Goldfarb, unpublished results). Thus, Vac8p may participate in the assembly or regulation of the vacuolar ATPase. Another Vac8p-binding protein, Apg13p, was originally identified as being required for efficient autophagy (Funakoshi et al., 1997). Because autophagy and Cvt are mediated by many of the same factors, the requirement for Vac8p in Cvt and autophagy may result from its interaction with Apg13p. Both vac8-Δ and apg13-Δ cells, but not nvj1-Δ cells, exhibit decreased viability in SD-N medium (our unpublished results). Thus, it is likely that Vac8p is required for efficient Cvt and autophagy. Neither VAB2 nor APG13 are required for NV junction formation (Pan and Goldfarb, unpublished observations). Together, these results support the hypothesis that Vac8p mediates independent vacuole-specific processes, including the formation of NV junctions through its interaction with different binding partners. This study provides support for the hypothesis that the primary function of Arm domain proteins such as Vac8p, importin α, and β-catenin is to dock binding partners at their cognate membranes.
ACKNOWLEDGMENTS
We thank Joanna Olmsted and Hiram Lyon for help with confocal microscopy, Karen Jenson of the University of Rochester Imaging Core for technical assistance with TEM, and Kathy Wilson for suggesting the name NVJ1. API antibodies were the generous gift of Dan Klionsky. This work was supported by National Institutes of Health grants GM40362 (D. S. G.) and GM55796 (S. K. L.) and by a grant from the March of Dimes (D. S. G.).
REFERENCES
- Achleitner G, Gaigg B, Krasser A, Kainersdorfer E, Kohlwein SD, Perktold A, Zellnig G, Daum G. Association between endoplasmic reticulum and mitochondria of yeast facilitates interorganelle transport of phospholipids through membrane contact. Eur J Biochem. 1999;264:545–553. doi: 10.1046/j.1432-1327.1999.00658.x. [DOI] [PubMed] [Google Scholar]
- Baba M, Takeshige K, Baba N, Osumi Y. Ultrastructural analysis of the autophagic process in yeast: detection of autophagosomes and their characterization. J Cell Biol. 1994;124:903–913. doi: 10.1083/jcb.124.6.903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baba M, Osumi M, Scott SV, Klionsky DJ, Ohsumi Y. Two distinct pathways for targeting proteins from the cytoplasm to the vacuole/lysosome. J Cell Biol. 1997;139:1687–1695. doi: 10.1083/jcb.139.7.1687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banta LM, Robinson JS, Klionsky DJ, Emr SD. Organelle assembly in yeast: characterization of yeast mutants defective in vacuolar biogenesis and protein sorting. J Cell Biol. 1988;107:1369–1383. doi: 10.1083/jcb.107.4.1369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartel PL, Fields S. Analyzing protein-protein interactions using two-hybrid system. Methods Enzymol. 1995;254:241–263. doi: 10.1016/0076-6879(95)54018-0. [DOI] [PubMed] [Google Scholar]
- Bonangelino CJ, Catlett NL, Weisman LS. Vac7p, a novel vacuolar protein, is required for normal vacuole inheritance and morphology. Mol Cell Biol. 1997;17:6847–6858. doi: 10.1128/mcb.17.12.6847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bryant NJ, Piper RC, Weisman LS, Stevens TH. Retrograde traffic out of the yeast vacuole to the TGN occurs via the prevacuolar/endosomal compartment. J Cell Biol. 1998;142:651–663. doi: 10.1083/jcb.142.3.651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chook YM, Blobel G. Structure of the nuclear transport complex karyopherin-β2-Ran GppNHp. Nature. 1999;399:230–237. doi: 10.1038/20375. [DOI] [PubMed] [Google Scholar]
- Chu S, DeRisi J, Eisen M, Mulholland J, Botstein D, Brown PO, Herskowitz I. The transcriptional program of sporulation in budding yeast [published erratum appears in Science 1998, 282, 1421] Science. 1998;282:699–705. doi: 10.1126/science.282.5389.699. [DOI] [PubMed] [Google Scholar]
- Cingolani G, Petosa C, Weis K, Muller CW. Structure of importin-β bound to the IBB domain of importin-α. Nature. 1999;399:221–229. doi: 10.1038/20367. [DOI] [PubMed] [Google Scholar]
- Conibear E, Stevens TH. Multiple sorting pathways between the late Golgi and the vacuole in yeast. Biochim Biophys Acta. 1998;1404:211–230. doi: 10.1016/s0167-4889(98)00058-5. [DOI] [PubMed] [Google Scholar]
- Conti E, Uy M, Leighton L, Blobel G, Kuriyan J. Crystallographic analysis of the recognition of a nuclear localization signal by the nuclear import factor karyopherin alpha. Cell. 1998;94:193–204. doi: 10.1016/s0092-8674(00)81419-1. [DOI] [PubMed] [Google Scholar]
- DeRisi JL, Iyer VR, Brown PO. Exploring the metabolic and genetic control of gene expression on a genomic scale. Science. 1997;278:680–686. doi: 10.1126/science.278.5338.680. [DOI] [PubMed] [Google Scholar]
- Dunn WA., Jr Autophagy and related mechanisms of lysosome-mediated protein degradation. Trends Cell Biol. 1994;4:139–143. doi: 10.1016/0962-8924(94)90069-8. [DOI] [PubMed] [Google Scholar]
- Fleckenstein D, Rohde M, Klionsky DJ, Rudiger M. Yel013p (Vac8p), an armadillo repeat protein related to plakoglobin and importin alpha is associated with the yeast vacuole membrane. J Cell Sci. 1998;111:3109–3118. doi: 10.1242/jcs.111.20.3109. [DOI] [PubMed] [Google Scholar]
- Funakoshi T, Matsuura A, Noda T, Osumi Y. Analyses of APG13 gene involved in autophagy in yeast, Saccharomyces cerevisiae. Gene. 1997;192:207–213. doi: 10.1016/s0378-1119(97)00031-0. [DOI] [PubMed] [Google Scholar]
- Gary JD, Wurmser AE, Bonangelino CJ, Weisman LS, Emr SD. Fab1p is essential for PtdIns(3)P 5-kinase activity and the maintenance of vacuolar size and membrane homeostasis. J Cell Biol. 1998;143:65–79. doi: 10.1083/jcb.143.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gietz RD, Sugino A. New yeast-Escherichia coli shuttle vectors constructed with in vitro mutagenized yeast genes lacking six-base pair restriction sites. Gene. 1988;74:527–534. doi: 10.1016/0378-1119(88)90185-0. [DOI] [PubMed] [Google Scholar]
- Groves MR, Hanlon N, Turowski P, Hemmings BA, Barford D. The structure of the protein phosphatase 2A PR65/A subunit reveals the conformation of its 15 tandemly repeated HEAT motifs. Cell. 1999;96:99–110. doi: 10.1016/s0092-8674(00)80963-0. [DOI] [PubMed] [Google Scholar]
- Guldener U, Heck S, Fielder T, Beinhauer J, Hegemann JH. A new efficient gene disruption cassette for repeated use in budding yeast. Nucleic Acids Res. 1996;24:2519–2524. doi: 10.1093/nar/24.13.2519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heath CV, Copeland CS, Amberg DC, Del Priori V, Snyder M, Cole CN. Nuclear pore complex clustering and nuclear accumulation of poly(A)+ R.N.A. associated with mutation of the Saccharomyces cerevisiae RAT2/NUP120 gene. J Cell Biol. 1995;131:1677–1697. doi: 10.1083/jcb.131.6.1677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huber AH, Nelson WJ, Weis WI. Three-dimensional structure of the armadillo repeat region of beta-catenin. Cell. 1997;90:871–882. doi: 10.1016/s0092-8674(00)80352-9. [DOI] [PubMed] [Google Scholar]
- Jones EW, Webb GC, Hiller MA. Biogenesis and function of the yeast vacuole. In: Pringle J R, Broach J R, Jones E W, editors. The Molecular and Cellular Biology of the Yeast Saccharomyces. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 1997. pp. 363–470. [Google Scholar]
- Kim J, Scott SV, Oda MN, Klionsky DJ. Transport of a large oligomeric protein by the cytoplasm to vacuole protein targeting pathway. J Cell Biol. 1997;137:609–618. doi: 10.1083/jcb.137.3.609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klionsky DJ, Ohsumi Y. Vacuolar import of proteins and organelles from the cytoplasm. Annu Rev Cell Dev Biol. 1999;15:1–32. doi: 10.1146/annurev.cellbio.15.1.1. [DOI] [PubMed] [Google Scholar]
- Koning AJ, Lum PY, Williams JM, Wright R. DiOC6 staining reveals organelle structure and dynamics in living yeast cells. Cell Motil Cytoskeleton. 1993;25:111–128. doi: 10.1002/cm.970250202. [DOI] [PubMed] [Google Scholar]
- Lynn RR, Magee PT. Development of the spore wall during ascospore formation in Saccharomyces cerevisiae. J Cell Biol. 1970;44:688–692. doi: 10.1083/jcb.44.3.688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macreadie IG, Jagadish MN, Azad AA, Vaughan PR. Versatile cassettes designed for the copper inducible expression of proteins in yeast. Plasmid. 1989;21:147–150. doi: 10.1016/0147-619x(89)90059-0. [DOI] [PubMed] [Google Scholar]
- Malik HS, Eickbush TE, Goldfarb DS. Evolutionary specialization of the nuclear targeting apparatus. Proc Natl Acad Sci USA. 1997;94:13738–13742. doi: 10.1073/pnas.94.25.13738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nehrbass U, Rout MP, Maguire S, Blobel G, Wozniak RW. The yeast nucleoporin Nup188p interacts genetically and physically with the core structures of the nuclear pore complex. J Cell Biol. 1996;133:1153–1162. doi: 10.1083/jcb.133.6.1153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niedenthal RK, Riles L, Johnston M, Hegemann JH. Green fluorescent protein as a marker for gene expression and subcellular localization in budding yeast. Yeast. 1996;12:773–786. doi: 10.1002/(SICI)1097-0061(19960630)12:8%3C773::AID-YEA972%3E3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- Pan X, Goldfarb DS. YEB3/VAC8 encodes a myristylated armadillo protein of the Saccharomyces cerevisiae vacuolar membrane that functions in vacuole fusion and inheritance. J Cell Sci. 1998;111:2137–2147. doi: 10.1242/jcs.111.15.2137. [DOI] [PubMed] [Google Scholar]
- Preuss D, Mulholland J, Kaiser CA, Orlean P, Albright C, Rose MD, Robbins PW, Botstein D. Structure of the yeast endoplasmic reticulum: localization of ER proteins using immunofluorescence and immunoelectron microscopy. Yeast. 1991;7:891–911. doi: 10.1002/yea.320070902. [DOI] [PubMed] [Google Scholar]
- Rout MP, Aitchison JD, Suprapto A, Hjertaas K, Zhao Y, Chait BT. The yeast nuclear pore complex: composition, architecture, and transport mechanism. J Cell Biol. 2000;148:635–652. doi: 10.1083/jcb.148.4.635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakai Y, Koller A, Rangell LK, Keller GA, Subramani S. Peroxisome degradation by microautophagy in Pichia pastoris: identification of specific steps and morphological intermediates. J Cell Biol. 1998;141:625–636. doi: 10.1083/jcb.141.3.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schiestl RH, Gietz RD. High efficiency transformation of intact yeast cells using single stranded nucleic acids as a carrier. Curr Genet. 1989;16:339–346. doi: 10.1007/BF00340712. [DOI] [PubMed] [Google Scholar]
- Scott SV, Hefner-Gravink A, Morano KA, Noda T, Ohsumi Y, Klionsky DJ. Cytoplasm-to-vacuole targeting and autophagy employ the same machinery to deliver proteins to the yeast vacuole. Proc Natl Acad Sci USA. 1996;93:12304–12308. doi: 10.1073/pnas.93.22.12304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Severs NJ, Jordan EG, Williamson DH. Nuclear pore absence from areas of close association between nucleus and vacuole in synchronous yeast cultures. J Ultrastruct Res. 1976;54:374–387. doi: 10.1016/s0022-5320(76)80023-8. [DOI] [PubMed] [Google Scholar]
- Shaywitz DA, Espenshade PJ, Gimeno RE, Kaiser CA. COPII subunit interactions in the assembly of the vesicle coat. J Biol Chem. 1997;272:25413–25416. doi: 10.1074/jbc.272.41.25413. [DOI] [PubMed] [Google Scholar]
- Sherman F. Laboratory Course Manual for Methods in Yeast Genetics. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory; 1986. [Google Scholar]
- Spellman PT, Sherlock G, Zhang MQ, Iyer VR, Anders K, Eisen MB, Brown PO, Botstein D, Futcher B. Comprehensive identification of cell cycle-regulated genes of the yeast Saccharomyces cerevisiae by microarray hybridization. Mol Biol Cell. 1998;9:3273–3297. doi: 10.1091/mbc.9.12.3273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strayle J, Pozzan T, Rudolph HK. Steady-state free Ca2+ in the yeast endoplasmic reticulum reaches only 10 μM and is mainly controlled by the secretory pathway pump Pmr1. EMBO J. 1999;18:4733–4743. doi: 10.1093/emboj/18.17.4733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeshige K, Baba M, Tsuboi S, Noda T, Osumi Y. Autophagy in yeast demonstrated with proteinase-deficient mutants and conditions for its induction. J Cell Biol. 1992;119:301–311. doi: 10.1083/jcb.119.2.301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsukada M, Ohsumi Y. Isolation and characterization of autophagy-defective mutants of Saccharomyces cerevisiae. FEBS Lett. 1993;333:169–174. doi: 10.1016/0014-5793(93)80398-e. [DOI] [PubMed] [Google Scholar]
- Vetter IR, Arndt A, Kutay U, Gorlich D, Wittinghofer A. Structural view of the ran-importin β interaction at 2.3 A resolution. Cell. 1999;97:635–646. doi: 10.1016/s0092-8674(00)80774-6. [DOI] [PubMed] [Google Scholar]
- Vida TA, Emr SD. A new vital stain for visualizing vacuolar membrane dynamics and endocytosis in yeast. J Cell Biol. 1995;128:779–792. doi: 10.1083/jcb.128.5.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vida TA, Huyer G, Emr SD. Yeast vacuolar proenzymes are sorted in the late Golgi complex and transported to the vacuole via a prevacuolar endosome-like compartment. J Cell Biol. 1993;121:1245–1256. doi: 10.1083/jcb.121.6.1245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vidugiriene J, Sharma DK, Smith TK, Baumann NA. Segregation of glycosylphosophatidylinositol biosynthetic reactions in a subcompartment of the endoplasmic reticulum. J Biol Chem. 1999;274:15203–15212. doi: 10.1074/jbc.274.21.15203. [DOI] [PubMed] [Google Scholar]
- Vojtek AB, Hollenberg SM. Ras-Raf interaction: two-hybrid analysis. Methods Enzymol. 1995;255:331–342. doi: 10.1016/s0076-6879(95)55036-4. [DOI] [PubMed] [Google Scholar]
- Vojtek AB, Hollenberg SM, Cooper JA. Mammalian Ras interacts directly with the serine/threonine kinase Raf. Cell. 1993;74:205–214. doi: 10.1016/0092-8674(93)90307-c. [DOI] [PubMed] [Google Scholar]
- Wach A. PCR-synthesis of marker cassettes with long flanking homology regions for gene disruptions in S. cerevisiae. Yeast. 1996;12:259–265. doi: 10.1002/(SICI)1097-0061(19960315)12:3%3C259::AID-YEA901%3E3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- Wang YX, Kauffman EJ, Weisman LS. Vac8p functions in vacuole fusion at a post-SNARE step and may account for the palmitoyl-CoA stimulation of fusion. Mol Biol Cell. 1998;9:322A. Suppl. [Google Scholar]
- Wang YX, Catlett NL, Weisman LS. Vac8p, a vacuolar protein with armadillo repeats, functions in both vacuole inheritance and protein targeting from the cytoplasm to vacuole. J Cell Biol. 1998;140:1063–1074. doi: 10.1083/jcb.140.5.1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang YX, Zhao H, Harding TM, Gomes de Mesquita DS, Woldringh CL, Klionsky DJ, Munn AL, Weisman LS. Multiple classes of yeast mutants are defective in vacuole partitioning yet target vacuole proteins correctly. Mol Biol Cell. 1996;7:1375–1389. doi: 10.1091/mbc.7.9.1375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward AC, Castelli LA, Macreadie IG, Azad AA. Vectors for Cu(2+)-inducible production of glutathione S-transferase fusion proteins for single-step purification from yeast. Yeast. 1994;10:441–449. doi: 10.1002/yea.320100403. [DOI] [PubMed] [Google Scholar]
- Watson ME. Compilation of published signal sequences. Nucleic Acids Res. 1984;12:5145–5164. doi: 10.1093/nar/12.13.5145. [DOI] [PMC free article] [PubMed] [Google Scholar]