Abstract
OBJECTIVE
Factors associated with impaired functioning in patients with lower extremity peripheral arterial disease (PAD) are not fully understood. The purpose of this study was to determine the relationship between depressive symptoms and objective measures of lower extremity functioning in persons with PAD.
DESIGN
Cross-sectional.
PATIENTS/PARTICIPANTS
Four hundred twenty-three men and women with PAD identified from 3 Chicago area medical centers.
MEASUREMENTS AND MAIN RESULTS
PAD was defined as ankle brachial index (ABI) <0.90. The Geriatric Depression Scale short form (GDS-S) (0–15 scale, 15 = worst) was completed by all participants. A clinically significant number of depressive symptoms was defined as a GDS-S score ≥6. Six-minute walk distance and usual-and fast-pace walking velocity were determined for all participants. A GDS-S score ≥6 was present in 21.7% of participants with PAD. Adjusting for age, increasing numbers of depressive symptoms were associated with an increasing prevalence of leg pain on exertion and rest (P = .004). Adjusting for age, sex, race, ABI, number of comorbidities, current smoking, and antidepressant medications, increasing numbers of depressive symptoms were associated with shorter 6-minute walk distance (P < .001), slower usual-pace walking velocity (P = .005), and slower fast-pace walking velocity (P = .005). These relationships were attenuated slightly after additional adjustment for presence versus absence of leg pain on exertion and rest and severity of exertional leg symptoms.
CONCLUSIONS
Among men and women with PAD, the prevalence of a clinically significant number of depressive symptoms is high. Greater numbers of depressive symptoms are associated with greater impairment in lower extremity functioning. Further study is needed to determine whether identifying and treating depressive symptoms in PAD is associated with improved lower extremity functioning.
Keywords: depression, peripheral vascular disease, physical functioning
Studies of community-dwelling older men and women show that depressive symptoms are risk factors for disability and that disability is associated with an increased risk of depressive symptoms.1–4 Disability may induce depressive symptoms because of disability-associated loss of social functioning and loss of independence in the community. Depressive symptoms may induce subsequent functional decline by way of several psychologically and/or biologically mediated pathways.4 For example, depressive symptoms may reduce initiative to seek necessary medical assistance for specific medical problems. Depressive symptoms also may inhibit healthy behaviors integral to improving functioning and mobility. Finally, by inducing immunosuppression and/or by increasing sympathetic tone, depressive symptoms may promote susceptibility to infections and other diseases that may in turn further impair functioning.5–7
Patients with lower extremity peripheral arterial disease (PAD) have reduced lower extremity functioning as compared to individuals without PAD,8–10 and greater severity of PAD as measured by the ankle brachial index (ABI) is associated with greater impairment of lower extremity functioning.9,10 Patients with ABI <0.50, consistent with severe PAD, are nearly 12 times more likely to stop during a 6-minute walk test compared to individuals with a normal ABI.9 Because of their high burden of functional impairment, patients with PAD may have an increased prevalence of depressive symptoms. However, the prevalence of depressive symptoms in patients with PAD has not been well studied.
We documented the relation between depressive symptoms and lower extremity functioning in persons with PAD. Because persons with PAD already have impaired functioning as compared to persons without PAD, it is not clear whether depressive symptoms in patients with PAD are associated with greater impairment in lower extremity functioning. We hypothesized that patients with a greater number of depressive symptoms would have more impairment in lower extremity functioning than patients with fewer depressive symptoms. If depressive symptoms are related to functional impairment, then treating depressive symptoms in patients with PAD may improve lower extremity functioning.
METHODS
Participant Identification
The protocol was Institutional Review Board–approved by Northwestern University's Feinberg School of Medicine and Catholic Health Partners Hospitals. All participants gave informed consent. Participants were part of the Walking and Leg Circulation Study (WALCS), a prospective observational study of men and women age 55 and older with and without PAD.9 All WALCS participants with PAD were potentially eligible for the present study.
Peripheral arterial disease participants were identified consecutively from among patients diagnosed with PAD in 3 Chicago-area noninvasive vascular laboratories. Individuals were contacted and invited to return to the medical center for a study visit between October 1998 and January 2000. A small proportion of participants with PAD were identified from among consecutively identified men and women in a large general internal medicine practice at Northwestern who were found to have a low ABI consistent with PAD during their study visit.
Exclusion Criteria
On the basis of a previous study, PAD was defined as ABI <0.90,10–13 and absence of PAD was defined as ABI ≥0.90 and ≤1.50.8–10,13 ABI <0.90 is 95% sensitive and 99% specific for angiographically diagnosed PAD.14 Individuals with ABI >1.50 were excluded because this indicates poorly compressible leg arteries and inability to gauge the degree of arterial ischemia accurately.12
Individuals with PAD diagnosed in the noninvasive vascular laboratory were excluded if their study visit ABI indicated absence of PAD. This occasionally occurred when PAD participants were revascularized between vascular laboratory testing and their study visit. It also occurred in individuals with borderline ABI values of ∼0.90, due to measurement variation. The total number of individuals excluded because of their ABI was 136. In addition, patients with dementia were excluded because of poor reliability during interviewing. Dementia was defined as a Mini-Mental Status Examination Score <18.15 Nursing home residents, wheelchair-bound patients, and patients with foot or leg amputations were excluded because of their severely and uniquely impaired functioning. Non–English-speaking patients were excluded because investigators were not fluent in non-English languages. Patients with recent major surgery were excluded.
Ankle Brachial Index Measurement
The ABI was measured using established methods.8–10,12,13,16,17 A hand-held Doppler probe (Nicolet Vascular Pocket Dop II, Golden, Colo) was used to measure systolic pressures in the right brachial artery, right dorsalis pedis and posterior tibial arteries, left dorsalis pedis and posterior tibial arteries, and left brachial artery. Pressures were measured twice, in the order listed here and then in reverse order. The ABI was calculated in each leg by dividing the average of the 4 pressures in each leg by the average of the 4 brachial pressures.9,17 Average brachial pressures in the arm with highest pressure were used when one brachial pressure was higher than the opposite brachial pressure in both measurement sets, and the 2 brachial pressures differed by 10 or more mm Hg in at least 1 measurement set, because in such cases, subclavian stenosis was possible.9,16 Lowest leg ABI was used in analyses.
Leg Symptom Groups
Leg symptoms were characterized into 1 of 5 mutually exclusive groups, using the San Diego claudication questionnaire, based on a previous study.8 Four groups had exertional leg symptoms: 1) intermittent claudication (exertional calf pain that does not begin at rest, causes the participant to stop walking, and resolves within 10 minutes of rest); 2) leg pain on exertion and rest (exertional leg pain that sometimes begins at rest); 3) atypical exertional leg pain/carry on (exertional leg symptoms that do not begin at rest and do not stop the individual from walking); 4) atypical exertional leg pain/stop (exertional leg symptoms that do not begin at rest, stop the individual from walking, and do not involve the calves or resolve within 10 minutes of rest). The fifth group had no exertional leg pain.
Comorbidities
Algorithms developed for the Women's Health and Aging Study and the Cardiovascular Health Study were used to adjudicate comorbidities.15 These algorithms combine data from patient report, physical examination, medical record review, medications, laboratory values, and a primary care physician questionnaire. Criteria developed by the American College of Rheumatology were used to diagnose knee and hip osteoarthritis.18,19 Comorbidities assessed were angina, diabetes mellitus, myocardial infarction, stroke, heart failure, pulmonary disease, knee and hip arthritis, spinal stenosis, disk disease, and hip fracture. These comorbidities have been shown to influence lower extremity functioning.20–23
Functional Measures
Six-minute Walk
In older men and women, the 6-minute walk test is a better measure of community walking ability than is treadmill performance.24–27 After instructions, and following a standardized protocol,28 participants walk up and down a 100-foot hallway for 6 minutes, covering as much distance as possible during 6 minutes.
Four-meter Walking Velocity
Walking velocity was measured with a 4-meter walk performed at “usual” and “fastest” pace. Each walk was performed twice. The faster walk in each pair was used in analyses.29,30
Assessment of Depressive Symptoms
We used the Geriatric Depression Scale-Short Form (GDS-S) to measure depressive symptoms.31 The GDS-S is a 15-item questionnaire assessing the number of depressive symptoms. The GDS-S score ranges from 0 to 15, where 0 indicates no depressive symptoms and 15 indicates that all depressive symptoms measured by GDS-S are present. In men and women age 60 and older, a GDS-S score ≥6 has a sensitivity of 92% and a specificity of 81% for clinical depression, as defined by a Structured Clinical Interview according to the Diagnostic and Statistical Manual of Mental Disorders, Third Edition, Revised (DSM-IIIR).31 Therefore, we considered a GDS-S score ≥6 to represent a clinically significant number of depressive symptoms. In this article, the GDS-S score and number of depressive symptoms are used interchangeably.
Other Assessments
Cigarette Smoking
Current cigarette smoking was assessed via patient report.
Antidepressant Use
Participants listed their medications at their study visit. The principal investigator (MMM) reviewed all medications and classified each according to whether it was an antidepressant medication.
Walking Impairment Questionnaire
Using the Walking Impairment Questionnaire (WIQ), participants recorded the degree to which specific symptoms (including joint symptoms and chest pain) impaired walking during the past week on a Likert scale ranging from 0 to 4.32 We calculated a WIQ leg symptom score in which responses to questions regarding the degree to which specific leg symptoms (pain in calf or buttock, pain in thighs, and leg weakness) impaired walking were summed to achieve a score ranging from 0 to 12 (12 = worst).
Statistical Analyses
Participants with PAD were categorized according to the number of their depressive symptoms, based on the GDS-S score as follows: 0 to 1, 2 to 3, 4 to 5, and ≥6. This classification system was selected a priori and allowed us to relate the number of depressive symptoms in a continuous fashion to outcome variables of interest.
Characteristics of the PAD population were expressed as means (SD) for continuous variables and proportions for binary variables. For continuous variables, the test for trend was based on linear regression analyses using the actual GDS-S scores as an independent variable. The Cochran-Armitage test was used to check for linear trend in binomial proportions across the GDS-S categories. Mean WIQ-measured walking impairment scores were compared between the GDS-S categories using general linear models, and the test for trend was based on linear regression analyses.
Mean lower extremity functioning was compared between the GDS-S categories using general linear models, adjusting for known and potential confounders (age, sex, race, ABI, number of comorbidities, current smoking, use of antidepressant medications, exertional leg pain, and WIQ-measured leg symptoms), and the test for trend was based on multiple linear regression analyses using the actual GDS-S scores as an independent variable. Proportions for the binary lower extremity functioning variables were estimated using adjusted general linear models, and the test of significance was based on logistic regression models. Analyses were repeated among participants with and without WIQ-measured walking impairment due to chest discomfort, and then repeated among participants with and without WIQ-measured walking impairment due to leg symptoms. All analyses were performed using SAS statistical software, version 8.2 (SAS Institute Inc., Cary, NC). All P values were 2-tailed, and P < .05 was considered to indicate statistical significance.
RESULTS
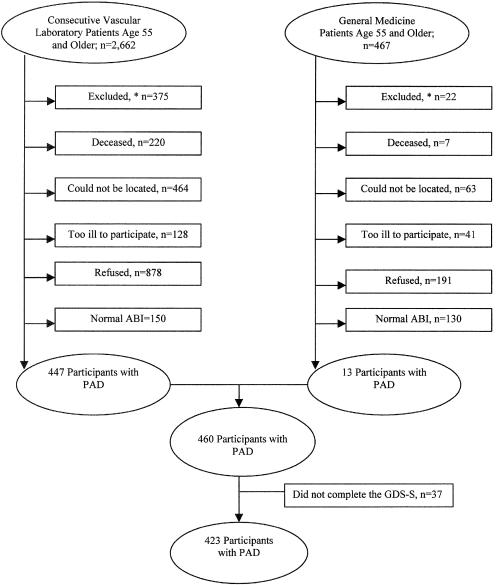
Figure 1 shows reasons for nonparticipation among individuals identified for the study. Of 460 enrolled men and women with PAD, 37 (8%) did not complete the GDS-S, leaving 423 participants (Fig. 1).
FIGURE 1.
Reasons for nonparticipation among individuals identified for the study. *See Methods section for details on excluded persons. PAD, peripheral arterial disease.
Overall, 21.7% of participants with PAD had a GDS-S score of ≥6. Table 1 shows characteristics of participants according to their GDS-S score. Increasing numbers of depressive symptoms were associated with a higher prevalence of antidepressant use and a greater average number of comorbidities. The prevalence of leg pain on exertion and rest was higher in participants with greater GDS-S scores.
Table 1.
Characteristics of Men and Women Age 55 and Older with Peripheral Arterial Disease According to Number of Depressive Symptoms (N = 423)*
| GDS-S Score | |||||
|---|---|---|---|---|---|
| 0–1 (n = 154) | 2–3 (n = 120) | 4–5 (n = 57) | 6+ (n = 92) | P Value | |
| Age, y (SD) | 72.1 (8.0) | 72.8 (8.6) | 72.3 (8.5) | 70.0 (8.3) | .014 |
| African-American race, % | 14.3 | 13.3 | 22.8 | 16.5 | .369 |
| Male, % | 64.3 | 55.0 | 57.9 | 58.7 | .398 |
| Mean ankle brachial index (SD) | 0.665 (0.13) | 0.645 (0.15) | 0.635 (0.16) | 0.637 (0.16) | .068 |
| Antidepressant use, % | 3.2 | 8.3 | 19.3 | 19.6 | <.001 |
| Leg pain with exertion and rest, % | 13.6 | 16.7 | 21.0 | 28.3 | .004 |
| Mean body mass index (SD) | 26.6 (5.4) | 26.6 (5.3) | 27.0 (4.2) | 26.5 (7.5) | .738 |
| Currently smoke, % | 18.2 | 15.8 | 21.0 | 31.5 | .013 |
| Mean total number of comorbidities (SD) | 2.0 (1.5) | 2.4 (1.6) | 2.7 (1.7) | 3.0 (1.7) | <.001 |
| Mean number of cardiovascular diseases (SD) | 0.86 (1.0) | 1.05 (1.0) | 1.24 (1.1) | 1.25 (1.2) | <.001 |
| Mean number of arthritis (SD) | 0.47 (0.72) | 0.52 (0.76) | 0.56 (0.80) | 0.79 (0.99) | <.001 |
| Mean number of other comorbidities (SD) | 0.66 (0.72) | 0.82 (0.76) | 0.93 (0.82) | 0.94 (0.77) | .007 |
P values derived from tests for trend across GDS-S categories (0–1, 2–3, 4–5, and 6+). The GDS-S score is measured on a 0–15 scale, where higher score represents greater depression. Comorbidities included: myocardial infarction, heart failure, angina, stroke, knee arthritis, hip arthritis, hip fracture, spinal stenosis, disk disease, diabetes, cancer, and pulmonary disease. Antidepressant use and currently smoke were defined as dichotomized variables, where yes = 1 and no = 0. Race was categorized as African American or non-African American.
GDS-S, Geriatric Depression Scale-Short Form.
Table 2 compares associations between GDS-S scores and the degree to which participants with PAD report that specific symptoms impair their leg functioning. Higher GDS-S scores were associated with greater walking impairment from lower extremity symptoms, chest discomfort, shortness of breath, and heart palpitations.
Table 2.
Reported Causes of Walking Impairment Among Men and Women Age 55 and Older with Peripheral Arterial Disease According to Depressive Symptom Severity (N = 423)*
| GDS-S Score | |||||
|---|---|---|---|---|---|
| 0–1 (n = 154) | 2–3 (n = 120) | 4–5 (n = 57) | 6+ (n = 92) | P Value | |
| Pain, aching or cramps in calves/buttocks | 1.66 (1.1) | 2.01 (1.2) | 1.98 (1.0) | 2.24 (1.2) | <.001 |
| Pain or aching in thighs | 0.80 (1.1) | 1.13 (1.2) | 1.07 (1.0) | 1.55 (1.3) | <.001 |
| Pain, stiffness, aching in joints (knee/ankle/hip) | 1.24 (1.1) | 1.59 (1.2) | 1.54 (1.1) | 1.77 (1.3) | <.001 |
| Weakness in one or both legs | 1.20 (1.1) | 1.57 (1.3) | 1.44 (1.1) | 2.15 (1.3) | <.001 |
| Pain or discomfort in chest | 0.31 (0.67) | 0.34 (0.73) | 0.56 (0.93) | 0.74 (0.96) | <.001 |
| Shortness of breath | 0.69 (0.86) | 1.05 (1.1) | 0.95 (1.0) | 1.26 (1.1) | <.001 |
| Heart palpitations | 0.26 (0.60) | 0.32 (0.65) | 0.33 (0.66) | 0.62 (0.84) | <.001 |
Responses to the Walking Impairment Questionnaire were scored on a 0–4 scale, where 4 represented greatest limitation and 0 represented least limitation. Values are expressed as means (SD). P values derived from tests for trend across GDS-S categories (0–1, 2–3, 4–5, and 6+). Data on pain, aching, or cramps in calves/buttocks were missing for 1 patient, and data on pain or aching in thighs were missing for 1 patient.
GDS-S, Geriatric Depression Scale-Short Form.
Table 3 shows associations between GDS-S scores and lower extremity functional measures. Adjusting for age, sex, race, ABI, number of comorbidities, current smoking status, and use of antidepressant drugs, higher GDS-S scores were associated with significantly shorter distance achieved during the 6-minute walk and with slower usual-paced and fast-paced walking velocities. Additional adjustment for presence versus absence of leg pain on exertion and rest as well as WIQ-measured severity of exertional leg pain symptoms further attenuated the relationships between the GDS-S scores and lower extremity functioning (Table 3). However, significant differences in distance achieved in the 6-minute walk, usual-pace walking velocity, and fast-pace walking velocity remained across categories of GDS-S scores. We observed similar associations between GDS-S scores and lower extremity functioning when Table 3 analyses were repeated within subsets of participants with and without WIQ-measured walking impairment due to chest discomfort. Results were also similar within subsets of participants with and without WIQ-measured walking impairment due to leg pain (data not shown).
Table 3.
Adjusted Functional Outcomes in Men and Women Age 55 and Older with Peripheral Arterial Disease According to Depressive Symptom Severity (N = 423)*
| GDS-S Score | |||||
|---|---|---|---|---|---|
| 0–1 (n = 154) | 2–3 (n = 120) | 4–5 (n = 57) | 6+ (n = 92) | P Value | |
| Adjusted for age, sex, race, ABI, number of comorbidities, current smoking status, and antidepressant use | |||||
| 6-Min walk, ft | 1,193 | 1,150 | 1,119 | 1,015 | <.001 |
| Stopped during 6-min walk, % | 25.4 | 28.2 | 26.3 | 34.2 | .048 |
| 4-Meter normal pace walking velocity, m/s | 0.90 | 0.88 | 0.86 | 0.84 | .005 |
| 4-Meter rapid pace walking velocity, m/s | 1.22 | 1.20 | 1.19 | 1.12 | .005 |
| Adjusted for age, sex, race, ABI, number of comorbidities, current smoking status, antidepressant use, and presence versus absence of leg pain on exertion and rest | |||||
| 6-Min walk, ft | 1,190 | 1,146 | 1,119 | 1,027 | <.001 |
| Stopped during 6-min walk, % | 25.6 | 28.5 | 26.3 | 33.4 | .077 |
| 4-Meter normal pace walking velocity, m/s | 0.90 | 0.88 | 0.86 | 0.84 | .016 |
| 4-Meter rapid pace walking velocity, m/s | 1.22 | 1.19 | 1.18 | 1.13 | .014 |
| Adjusted for age, sex, race, ABI, number of comorbidities, current smoking status, antidepressant use, and WIQ leg symptom score | |||||
| 6-Min walk, ft | 1,178 | 1,153 | 1,112 | 1,043 | <.001 |
| Stopped during 6-min walk, % | 27.4 | 27.9 | 27.4 | 30.4 | .453 |
| 4-Meter normal pace walking velocity, m/s | 0.90 | 0.88 | 0.85 | 0.84 | .030 |
| 4-Meter rapid pace walking velocity, m/s | 1.22 | 1.20 | 1.18 | 1.13 | .018 |
Higher GDS-S scores represent greater numbers of depressive symptoms. Presence of leg pain on exertion and rest was determined by the San Diego Claudication Questionnaire. Data on 6-minute walk and stopped during 6-minute walk were missing for 7 patients, and data on 4-meter normal pace walking velocity were missing for 3 patients.
GDS-S, Geriatric Depression Scale-Short Form; WIQ, Walking Impairment Questionnaire.
DISCUSSION
We found that PAD participants with higher GDS-S scores achieved shorter distances in the 6-minute walk and had slower usual- and fast-paced walking velocities as compared to PAD participants with lower GDS-S scores. These relationships between greater numbers of depressive symptoms and poorer lower extremity functioning were independent of comorbid diseases, ABI, and other known and potential confounders. Our findings also suggest that differences in the quality of leg pain symptoms and/or greater severity of leg pain symptoms in PAD participants with higher GDS-S scores may contribute to some of the functional impairment associated with greater depressive symptoms.
On the basis of our findings, clinicians should evaluate patients with PAD for depression, because symptoms of depression are both common and associated with significant functional impairment in patients with PAD. Although treatment of depressive symptoms may improve functional impairment in patients with PAD, to our knowledge this hypothesis has not been tested previously in clinical trials of patients with PAD.
In a separate study of 167 patients (93 with PAD), we showed previously that the prevalence of GDS-S ≥6 was 24% in the patients with PAD versus 12% in patients without PAD.33 In this previous smaller cohort of patients with PAD, a shorter 6-minute walk distance was associated with an increased prevalence of GDS-S ≥6.33 However, linear relations between GDS-S scores and 6-minute walk performance or 4-meter walking velocity were not examined. In addition, associations between GDS-S scores and leg symptoms, chest discomfort, and other symptoms were not examined. Although men and women with PAD already have impaired lower extremity functioning compared to persons without PAD, our data reported here show that greater depressive symptoms are associated with further functional impairment beyond that already related to PAD.
PAD participants with higher GDS-S scores reported greater walking impairment from chest discomfort, shortness of breath, and heart palpitations compared to PAD participants with lower GDS-S scores. Although patients with depressive disorders typically have an increased frequency of these physical symptoms, this relationship has not been evaluated previously among individuals with PAD to our knowledge. Because patients with PAD have a high prevalence of comorbid disease and exertional leg symptoms, the relationship between depressive symptoms and physical symptoms associated with walking impairment might have differed in patients with PAD as compared to patients without PAD. Conceivably, the higher prevalence of comorbid disease among participants with PAD and higher GDS-S scores may have contributed to the observed associations between higher GDS-S scores and greater walking impairment from physical symptoms. However, we observed similar linear relations between higher GDS-S scores and poorer functioning among patients with and without walking impairment due to chest discomfort and leg symptoms, respectively. These latter findings suggest that the relations between GDS-S scores and functioning are independent of symptomatic comorbid disease.
Our study has several limitations. In patients with PAD, shorter distances achieved in the 6-minute walk are highly correlated with lower levels of physical activity.34 Slower walking speed has been associated with an increased risk of nursing home placement, mortality, loss of mobility, and loss of the ability to perform Activities of Daily Living in older community-dwelling men and women populations.29,30 However, the clinical significance of observed differences in lower extremity performance between our participants with the fewest versus the greatest number of depressive symptoms has not been assessed longitudinally in patients with PAD. We cannot exclude the possibility that poorer effort among PAD participants with greater numbers of depressive symptoms contributed to slower walking velocity and shorter distance achieved in the 6-minute walk as compared to PAD participants with fewer depressive symptoms. However, our findings are consistent with those of a previous study in non-PAD populations demonstrating that greater depressive symptoms are associated with functional impairment and with greater decline in functioning over time.4,35,36 Finally, although presence versus absence of comorbid diseases was rigorously adjudicated using previously validated methods, we are not aware of validated methods for assessing severity of comorbid disease. It is conceivable that variation in severity of comorbid disease according to the number of depressive symptoms may contribute to observed differences in lower extremity functioning by the number of depressive symptoms.
In this cross-sectional study, we could not determine whether greater depressive symptoms preceded functional impairment or vice versa. Previous studies of community-dwelling populations suggest that disability promotes depression and also that depression is associated with functional decline.1–4 Lower extremity arterial ischemia-associated functional impairment and pain may induce depressive symptoms in patients with PAD. In addition, PAD patients with depressive symptoms may be less likely to practice healthy behaviors such as exercise and smoking cessation, and these unhealthy behaviors may lead to greater disability in PAD. Further study is needed to define the mechanism of association between increasing depressive symptoms and greater impairment in functioning among persons with PAD. Further study also is needed to determine whether treatment of depressive symptoms is associated with improvement in lower extremity functioning.
Acknowledgments
Supported by grant #R01-HL58099 from the National Heart, Lung, and Blood Institute and by grant #RR-00048 from the National Center for Research Resources, National Institutes of Health. Dr. McDermott was a Robert Wood Johnson Generalist Physician Faculty Scholar during the time these data were collected. Dr. McDermott is recipient of an Established Investigator Award from the American Heart Association.
REFERENCES
- 1.Broadhead WE, Blazer DG, George LK, Tee C. Depression, disability days, and days lost from work in a prospective epidemiologic survey. JAMA. 1990;264:2524–8. [PubMed] [Google Scholar]
- 2.Gallo JJ, Rabins PV, Lyketsos CG, Tien AY, Anthony JC. Depression without sadness: Functional outcomes of nondysphoric depression in later life. J Am Geriatr Soc. 1997;45:570–8. doi: 10.1111/j.1532-5415.1997.tb03089.x. [DOI] [PubMed] [Google Scholar]
- 3.Bruce ML, Seeman TE, Merrill SS, Blazer DG. The impact of depressive symptomatology on physical disability: MacArthur Studies of Successful Aging. Am J Public Health. 1994;84:1796–9. doi: 10.2105/ajph.84.11.1796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pennix BWJH, Guralnik JM, Lerrucci L, Simonsick EM, Deeg DJH, Wallace RB. Depressive symptoms and physical decline in community-dwelling older persons. JAMA. 1998;279:1720–6. doi: 10.1001/jama.279.21.1720. [DOI] [PubMed] [Google Scholar]
- 5.Kronfel Z, House J. Depression, cortisol and immune function. Lancet. 1984;1:1026–7. doi: 10.1016/s0140-6736(84)92379-1. [DOI] [PubMed] [Google Scholar]
- 6.Miller AH, Spencer RL, McEwen BS, Stein M. Depression, adrenal steroids, and the immune system. Ann Med. 1993;25:481–7. doi: 10.3109/07853899309147316. [DOI] [PubMed] [Google Scholar]
- 7.Stein M, Miller AH, Trestman RL. Depression, the immune system, and health and illness. Arch Gen Psychiatry. 1991;48:171–7. doi: 10.1001/archpsyc.1991.01810260079012. [DOI] [PubMed] [Google Scholar]
- 8.McDermott MM, Greenland P, Liu K, et al. Leg symptoms in peripheral arterial disease: associated clinical characteristics and functional impairment. JAMA. 2001;286:1599–606. doi: 10.1001/jama.286.13.1599. [DOI] [PubMed] [Google Scholar]
- 9.McDermott MM, Greenland P, Liu K, et al. The ankle brachial index is associated with leg function and physical activity: the Walking and Leg Circulation Study. Ann Intern Med. 2002;136:873–83. doi: 10.7326/0003-4819-136-12-200206180-00008. [DOI] [PubMed] [Google Scholar]
- 10.McDermott MM, Fried L, Simonsick E, Ling S, Guralnik JM. Asymptomatic peripheral arterial disease is independently associated with impaired lower extremity functioning: the women's health and aging study. Circulation. 2000;101:1007–12. doi: 10.1161/01.cir.101.9.1007. [DOI] [PubMed] [Google Scholar]
- 11.Olin JW. The clinical evaluation and office based detection of peripheral arterial disease. In: Hirsch AT, Olin FW, editors. An Office-based Approach to the Diagnosis and Treatment of Peripheral Arterial Disease, I: The Epidemiology and Practical Detection of Peripheral Arterial Disease. Am J Med Continuing Education Series. Belle Mead, NJ: Exerpta Medica; 1998. pp. 10–7. [Google Scholar]
- 12.Weitz JI, Byrne J, Clagett P, et al. Diagnosis and treatment of chronic arterial insufficiency of the lower extremities: a critical review. Circulation. 1996;94:3026–49. doi: 10.1161/01.cir.94.11.3026. [DOI] [PubMed] [Google Scholar]
- 13.Newman AB, Siscovick DS, Manolio TA, Plak J, Fried LP, Borhani NO. Ankle-arm index as a marker of atherosclerosis in the Cardiovascular Health Study. Circulation. 1993;88:837–45. doi: 10.1161/01.cir.88.3.837. [DOI] [PubMed] [Google Scholar]
- 14.Bernstein EF, Fronek A. Current status of non-invasive tests in the diagnosis of peripheral arterial disease. Surg Clin North Am. 1982;62:473–87. doi: 10.1016/s0039-6109(16)42739-8. [DOI] [PubMed] [Google Scholar]
- 15.Guralnik JM, Fried LP, Simonsick EM, et al. The Women's Health and Aging Study: Health and Social Characteristics of Older Women with Disability. Bethesda, Md: National Institute on Aging; 1995. NIH publication No. 95–4009, Appendix E. [Google Scholar]
- 16.Hiatt WR, Hoag S, Hamman RF. Effect of diagnostic criteria on the prevalence of peripheral arterial disease. The San Luis Valley Diabetes Study. Circulation. 1995;91:1472–9. doi: 10.1161/01.cir.91.5.1472. [DOI] [PubMed] [Google Scholar]
- 17.McDermott MM, Criqui MH, Liu K, et al. Lower ankle/brachial index, as calculated by averaging the dorsalis pedis and posterior tibial arterial pressures, and association with leg functioning in peripheral arterial disease. J Vasc Surg. 2000;32:1164–71. doi: 10.1067/mva.2000.108640. [DOI] [PubMed] [Google Scholar]
- 18.Altman R, Alarcon G, Appelrouth D, et al. The American College of Rheumatology criteria for the classification and reporting of osteoarthritis of the hip. Arthritis Rheum. 1991;34:505–14. doi: 10.1002/art.1780340502. [DOI] [PubMed] [Google Scholar]
- 19.Altman R, Asch E, Bloch D, et al. Development of criteria for the classification and reporting of osteoarthritis. Classification of osteoarthritis of the knee. Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum. 1986;29:1039–49. doi: 10.1002/art.1780290816. [DOI] [PubMed] [Google Scholar]
- 20.Ettinger WH, Fried LP, Harris T, Shemanski L, Schulz R, Robbins J. Self-reported causes of physical disability in older people: the Cardiovascular Health Study. J Am Geriatr Soc. 1994;42:1035–44. doi: 10.1111/j.1532-5415.1994.tb06206.x. [DOI] [PubMed] [Google Scholar]
- 21.Boult C, Kane RL, Louis TA, Boult L, McCaffrey D. Chronic conditions that lead to functional limitation in the elderly. J Gerontol. 1994;49:M28–36. doi: 10.1093/geronj/49.1.m28. [DOI] [PubMed] [Google Scholar]
- 22.Fried LP, Ettinger WH, Lind B, Newman AB, Gardin J. Physical disability in older adults: a physiological approach. Cardiovascular Health Study Research Group. J Clin Epidemiol. 1994;47:747–60. doi: 10.1016/0895-4356(94)90172-4. [DOI] [PubMed] [Google Scholar]
- 23.Fried LP, Bandeen-Roche K, Kasper JD, Guralnik JM. Association of comorbidity with disability in older women: the Women's Health and Aging Study. J Clin Epidemiol. 1999;52:27–37. doi: 10.1016/s0895-4356(98)00124-3. [DOI] [PubMed] [Google Scholar]
- 24.Swerts PMJ, Mostert R, Wouters EFM. Comparison of corridor and treadmill walking in patients with severe chronic obstructive pulmonary disease. Phys Ther. 1990;70:439–42. doi: 10.1093/ptj/70.7.439. [DOI] [PubMed] [Google Scholar]
- 25.Simonsick EM, Gardner AW, Poehlman ET. Assessment of physical function and exercise tolerance in older adults: reproducibility and comparability of five measures. Aging. 2000;12:274–80. doi: 10.1007/BF03339847. [DOI] [PubMed] [Google Scholar]
- 26.Greig C, Butler F, Skelton D, Mahmud S, Young A. Treadmill walking in old age may not reproduce the real life situation. J Am Geriatr Soc. 1993;41:15–8. doi: 10.1111/j.1532-5415.1993.tb05941.x. [DOI] [PubMed] [Google Scholar]
- 27.Peeters P, Mets T. The six-minute walk as an appropriate exercise test in elderly patients with chronic heart failure. J Gerontol A Biol Sci Med Sci. 1996;51:M147–51. doi: 10.1093/gerona/51a.4.m147. [DOI] [PubMed] [Google Scholar]
- 28.Guyatt GH, Sullivan MJ, Thompson PJ, et al. The six-minute walk: a new measure of exercise capacity in patients with chronic heart failure. Can Med Assoc J. 1985;132:919–23. [PMC free article] [PubMed] [Google Scholar]
- 29.Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49:M85–94. doi: 10.1093/geronj/49.2.m85. [DOI] [PubMed] [Google Scholar]
- 30.Guralnik JM, Ferrucci L, Simonsick E, Salive ME, Wallace RB. Lower extremity function in persons over 70 years as a predictor of subsequent disability. N Engl J Med. 1995;332:556–61. doi: 10.1056/NEJM199503023320902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lyness JM, Noel TK, Cox C, King DA, Conwell Y, Caine ED. Screening for depression in elderly primary care patients. Arch Intern Med. 1997;157:449–54. [PubMed] [Google Scholar]
- 32.Regensteiner JG, Steiner JF, Panzer RJ, Hiatt WR. Evaluation of walking impairment by questionnaire in patients with peripheral arterial disease. J Vasc Med Biol. 1990;2:142–52. [Google Scholar]
- 33.Arseven A, Guralnik JM, O'Brien E, Liu K, McDermott MM. Peripheral arterial disease and depressed mood in older men and women. Vasc Med. 2001;6:229–34. doi: 10.1177/1358836X0100600405. [DOI] [PubMed] [Google Scholar]
- 34.Gardner AW, Womack CJ, Sieminski DJ, Montgomery PS, Killewich LA, Fonong T. Relationship between free-living daily physical activity and ambulatory measures in older claudicants. Angiology. 1998;49:327–37. doi: 10.1177/000331979804900501. [DOI] [PubMed] [Google Scholar]
- 35.Lyness JM, King DA, Cox C, Yoediono Z, Caine ED. The importance of subsyndromal depression in older primary care patients: prevalence and associated functional disability. J Am Geriatr Soc. 1999;47:647–52. doi: 10.1111/j.1532-5415.1999.tb01584.x. [DOI] [PubMed] [Google Scholar]
- 36.Penninx BWJH, Deeg DJH, van Eijk JThM, Beekman ATF, Guralnik JM. Changes in depression and physical decline in older adults: a longitudinal perspective. J Affect Disord. 2000;61:1–12. doi: 10.1016/s0165-0327(00)00152-x. [DOI] [PubMed] [Google Scholar]