Abstract
AIDS-related human cytomegalovirus (HCMV) retinitis continues to be a chronic ophthalmologic problem among human immunodeficiency virus type 1 (HIV-1)-infected patients who do not respond to highly active antiretroviral therapy. Although HCMV retinitis occurs during HIV-1-induced immunosuppression, the precise effector mechanism(s) that fails during the immunopathogenesis of AIDS to allow onset and progression of HCMV retinal disease remains unclear. We therefore performed a series of experiments to explore the relative roles of distinct pathways of lymphocyte-mediated cytotoxicity in either resistance or susceptibility to experimental murine cytomegalovirus (MCMV) retinitis in mice. Whereas mutant C57BL/6 mice deficient in the Fas/FasL cytotoxic pathway (gld mice) were identical to normal C57BL/6 mice and exhibited absolute resistance to retinal necrosis following subretinal MCMV inoculation, knockout C57BL/6 mice deficient in the perforin cytotoxic pathway (PKO mice) were susceptible to MCMV retinitis. Susceptibility of PKO mice to MCMV retinitis correlated with increased ocular MCMV titers when compared with ocular MCMV titers of gld and normal mice. Since mice with retrovirus-induced immunodeficiency syndrome (MAIDS) exhibited a frequency and severity of MCMV retinitis that were equivalent to those observed in PKO mice, we hypothesized that susceptibility to MCMV retinitis during MAIDS correlates with a decrease in the perforin cytotoxic pathway. To test this hypothesis, we developed a quantitative competitive reverse transcription-PCR assay to measure mouse perforin mRNA levels in the splenic T lymphocytes and MCMV-inoculated eyes of normal mice or mice with MAIDS. Perforin mRNA levels in splenic T lymphocytes were significantly decreased during MAIDS, by ∼100-fold, from perforin mRNA levels in normal mice. Moreover, MCMV-inoculated eyes destined to develop retinitis during MAIDS also showed a significant decrease in perforin mRNA levels from the perforin mRNA levels of MCMV-inoculated eyes of normal mice destined to be resistant to retinitis. As expected, perforin mRNA could not be detected in unmanipulated and uninfected eyes of normal mice. These results provide the first evidence that the perforin cytotoxic pathway is more important than the Fas/FasL cytotoxic pathway in providing resistance to experimental MCMV retinitis and that loss of the perforin cytotoxic pathway predisposes to MCMV retinitis.
Prior to the era of highly active antiretroviral therapy to manage human immunodeficiency virus type 1 (HIV-1) infection, human cytomegalovirus (HCMV) retinitis was the leading cause of vision loss and blindness among patients with AIDS (8, 20). HCMV retinitis continues to be a chronic sight-threatening ophthalmologic problem among HIV-1-infected patients who do not respond to highly active antiretroviral therapy (22, 40). Although AIDS-related HCMV retinal disease occurs during HIV-1-induced immunosuppression, the precise effector mechanism(s) that fails during the immunopathogenesis of AIDS to allow onset and progression of HCMV retinitis remains unclear. Clinical studies attempting to correlate immune function to onset and severity of HCMV retinitis during AIDS have focused on quantification of absolute numbers of effector cell populations such as CD4+ T cells (30) and/or CD8+ T cells (32). These studies indicate that HCMV retinitis does not occur until late in the course of the disease when peripheral blood CD4+-T-cell counts have declined to less than 50 cells/ml3 (20). Studies with experimental models of murine CMV (MCMV) retinitis have provided additional evidence that underscores the importance of CD8+ T cells (2) and natural killer (NK) cells (3, 21) in protection against onset of MCMV retinal disease.
Cytotoxic CD8+ T cells and NK cells kill virus-infected cells by several possible pathways that have been investigated extensively during the past decade. Two distinct pathways of lymphocyte-mediated cytotoxicity have emerged that fall under the broad categories of perforin-mediated cytotoxicity and receptor-mediated cytotoxicity (27, 34, 35, 48). Although these pathways use distinct mediators of cytolysis and differ sharply with respect to calcium dependence, they both exhibit rapid kinetics (<4 h) and involve induction of a series of intracellular events that leads to apoptosis and cell death.
Perforin-mediated cytotoxicity is calcium dependent and involves the directional release of the contents of cytoplasmic granules from the cytotoxic lymphocyte to the target cell. These granules contain a number of proteins that include a pore-forming protein, perforin, and a family of serine proteases, granzymes (23, 42). Perforin monomers initiate pore formation in the plasma membrane of the target cell that eventually would lead to osmotic damage. Prior to appreciable perforin-mediated cell lysis, however, an endogenous cell death cascade is triggered via activation by granzymes of key target cell caspases, most notably granzyme B. Perforin and granzyme B must therefore work in concert to trigger apoptotic death of the target cell.
In comparison, receptor-mediated cytotoxicity is calcium independent and involves the engagement of a receptor molecule on the target cell (Fas or tumor necrosis factor receptor) with its ligand found on the cytotoxic lymphocyte (FasL or tumor necrosis factor) (16, 38, 44, 48). Receptor-ligand signaling leads to target cell death through activation of an endogenous apoptotic pathway involving cytoplasmic caspases (37). A second independent apoptotic pathway activated by Fas alone has also been described (6).
The relative roles of the perforin-mediated and receptor-mediated cytotoxic pathway in providing resistance to HCMV retinitis in immunologically normal persons with systemic HCMV infection have not yet been investigated. Conversely, the specific pathway(s) of cytolysis that fails during the immunopathogenesis of HIV-1 infection to allow onset of HCMV retinitis during AIDS has not yet been defined. We therefore initiated a series of experiments to explore the relative contributions of the perforin and Fas/FasL cytotoxic pathways in either resistance or susceptibility to experimental MCMV retinal disease in mice. Results demonstrated that the perforin cytotoxic pathway is more important than the Fas/FasL cytotoxic pathway in providing resistance to MCMV retinitis in immunocompetent mice. Moreover, additional results support the conclusion that mice with retrovirus-induced immunodeficiency syndrome (MAIDS) are highly susceptible to MCMV retinitis due to loss of the perforin pathway. Taken together, these findings provide the first direct evidence to suggest that the perforin-mediated pathway of cytotoxicity plays a prominent and defining role in determining resistance or HIV-1-induced susceptibility to HCMV retinitis in humans. These findings also have important implications with respect to the management and diagnosis of AIDS-related HCMV retinitis.
MATERIALS AND METHODS
Mice.
C57BL/6 knockout mice with a null mutation in the perforin gene (perforin−/− [PKO] mice) (26, 33, 51) (Transgenic Mouse Facility, University of Miami School of Medicine, Miami, Fla.) and C57BL/6 mutant mice deficient in FasL (gld mice) (38, 39) (Taconic Farms, Germantown, N.Y.) were used in experiments to establish the relative roles of the perforin and Fas/FasL cytotoxic pathways, respectively, in resistance or susceptibility to experimental MCMV retinitis. C57BL/6 euthymic mice (Taconic Farms) served as normal, healthy, control mice in all retinitis experiments and were used also for induction of MAIDS. All mice were allowed unrestricted access to food and water and were maintained in alternating 12-h light-dark cycles. All animal procedures were performed in accordance with the Association for Research in Vision and Ophthalmology Resolution on the Use of Animals in Research.
Viruses.
Stocks of MCMV were prepared in mouse salivary glands of BALB/c mice (Taconic Farms) as described previously (10). Mice were infected intraperitoneally with 102 to 103 PFU of the Smith strain of MCMV contained within a 0.2-ml volume. Fourteen days later, the salivary glands were removed aseptically, homogenized (10% [wt/vol]) in Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal bovine serum, and clarified by centrifugation, and 0.5-ml aliquots of the supernatants were stored in liquid N2. Virus stocks were titered on monolayers of mouse embryo fibroblasts grown in DMEM. A fresh aliquot of MCMV stock was thawed and used for a single experiment.
Stocks of the murine retrovirus mixture (LP-BM5 MuLV) were prepared by using SC1/MuLV LP-BM5 cells obtained from the AIDS Research and Reference Reagent Program, Division of AIDS, National Institute of Allergy and Infectious Diseases, National Institutes of Health. Approximately 106 SC1/MuLV LP-BM5 cells were mixed with approximately 106 uninfected SC1 cells and maintained for 6 days in DMEM. The cells were then scraped into the medium, pelleted by centrifugation, resuspended in phosphate-buffered saline, and frozen at −70°C. Prior to use, the suspension was thawed and clarifed by centrifugation to remove cellular debris.
Induction of MAIDS.
MAIDS was induced in normal C57BL/6 mice by intraperitoneal injection of 1.0 ml of LP-BM5 MuLV preparation (18). The inoculum contained approximately 5 × 103 to 5 × 104 infectious murine retroviruses. Mice with MAIDS of 8 to 10 weeks' duration were used in all experiments. These animals displayed clinical signs and symptoms (chronic generalized lymphadenopathy and splenomegaly) consistent with MAIDS (10).
Experimental mouse model of MCMV retinitis.
AIDS-related HCMV retinitis is thought to originate from virus that invades the retina from the blood during systemic infection (20). Intravenous or intraperitoneal injection of immunosuppressed mice with MCMV to produce systemic MCMV infection fails to infect the neurosensory retina and cause retinal disease, although virus infects the choroid, retinal pigment epithelium, and ciliary of the eye (9, 15). Consequently, several laboratories (1, 10, 15, 29) routinely induce experimental MCMV retinitis in mice by supraciliary (subretinal) MCMV injection, a procedure that induces reproducible retinal disease in mice with histopathologic features similar to those found in AIDS-related HCMV retinitis (1, 10). Any initial mechanical disruption of the blood-ocular barrier due to subretinal injection of virus in a 2-μl volume is focal, minimal, and temporary (R. D. Dix and S. W. Cousins, unpublished data). In comparison, extensive and progressive disruption of the blood-ocular barrier occurs during the evolution of necrotizing MCMV retinitis in eyes of mice susceptible to MCMV retinitis. All mice in the present study were anesthesized and injected subretinally with approximately 104 PFU of MCMV as described previously (10).
Histopathologic analysis of eyes and evaluation of retinitis.
At 8 to 10 days after subretinal MCMV injection, eyes were carefully removed from euthanatized animals, fixed in 10% buffered formalin, paraffin embedded, sectioned, stained with hematoxylin and eosin, and examined by light microscopy. Since necrotizing retinitis can remain regionally localized within the retina, at least six histopathologic sections representing different regions of the retina were examined. The histopathologic sections from MCMV-infected eyes were scored by using a modification of two types of scoring systems described previously (10) to quantify the frequency and severity of MCMV retinitis in mice. Firstly, the sections were scored for the presence (i.e., score of 2 or more on at least two sections) or absence of necrotizing retinitis to determine the frequency of retinitis within each animal group. Secondly, each section was evaluated semiquantitatively for posterior segment inflammation. All six sections of each eye were scored, and the scores for all sections were summed (with a maximum score of 24). The medium score and the percentage of the maximum score were calculated for each group. The frequency data were analyzed for significance by the chi-square test. The severity data for posterior segment inflammation were analyzed for significance by the Whitney U test because the scoring system provided categorical rather than continuous measurements.
Recovery of infectious MCMV.
Whole MCMV-infected eyes collected from mice at 8 days after subretinal MCMV inoculation and whole livers collected from mice at 5 days after intraperitoneal MCMV inoculation were frozen at −70°C. Whole livers were weighed prior to freezing. At time of quantitative plaque assay, eyes or livers were thawed, homogenized individually in 1.0 ml of cold DMEM, and clarified by centrifugation. Tenfold dilutions of the resulting supernatants were titered in duplicate onto monolayers of mouse embryo fibroblasts contained within six-well plates, allowed to adsorb for 1 h at 37°C, overlaid with methylcellulose containing DMEM, and incubated for 5 or 6 days at 37°C in a humidified CO2 atmosphere. Individual plaques were counted with an inverted light microscope. Results for whole eyes and whole livers are reported as number of PFU/ml/eye and number of PFU/ml/0.1 g of fresh weight, respectively.
Measurement of mouse perforin mRNA.
A competitive quantitative reverse transcription (RT)-PCR assay was developed to measure mouse perforin mRNA levels in individual spleens and individual eyes (posterior segment) collected from normal mice or mice with MAIDS at 3 days after uniocular subretinal MCMV inoculation, a time during initial rounds of intraocular virus replication but before the development of clinical retinal disease. Following extraction of total RNA (∼20 μg per eye), RT and quantitative competitive RT-PCR for perforin mRNA content were performed with primers specific for mouse perforin (GenBank accession no. M23182): 5′-CACAGTAGAGTGTCCGATGTA-3′ (upstream primer), 5′-CTTGGTTCCCGAAGAGCAGAT-3′ (downstream primer), and 5′-CTTGGTTCCCGAAGAGCAGATAAAGAGGTGGCGATTTTGTGCT-3′ (composite primer). Conditions for PCR were 94°C for 30 s, 53.4°C for 30 s, and 72°C for 2 min for a total of 40 cycles. The pattern of cDNA products (442 bp for authentic perforin and 380 bp for the competitive template) was visualized on ethidium-bromide-stained agarose gels. The sensitivity of the competitive RT-PCR assay was ∼1 fg of perforin mRNA per 500 ng of total RNA.
RESULTS
Susceptibility of PKO and gld mice to MCMV retinitis.
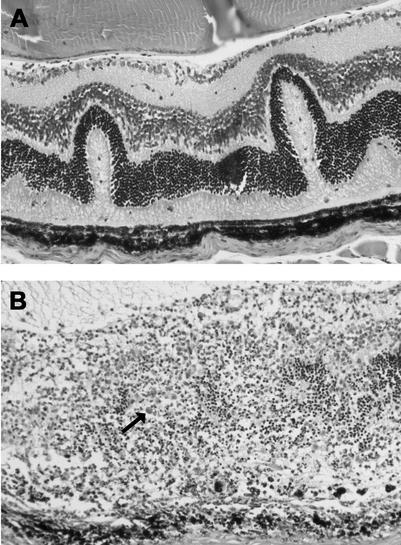
Clearance of lymphocytic choriomeningitis virus fails to occur in PKO mice that have a null mutation in the perforin gene (51). We therefore hypothesized that PKO mice would be susceptible to experimental MCMV retinitis. This hypothesis was tested by determining the frequency and severity of MCMV retinal necrosis in PKO mice at 10 days following uniocular subretinal MCMV inoculation and by then comparing these results with those obtained with mice deficient in FasL (gld mice), mice immunosuppressed due to MAIDS, or normal immunocompetent mice. Results are shown in Table 1. None (0%) of the MCMV-infected eyes of gld mice (zero of eight) or normal mice (zero of eight) developed retinal necrosis, although retinal folding was apparent in some histopathologic sections as noted previously in virus-infected eyes of normal mice (10, 12) (Fig. 1A). In sharp contrast, 92% of MCMV-infected eyes of PKO mice (12 of 13) and 88% of MCMV-infected eyes of mice with MAIDS (seven of eight) developed moderate-to-severe MCMV retinitis characterized by full-thickness retinal necrosis and the appearance of cytomegalic cells (cytomegalocytes) (Fig. 1B). Thus, gld mice deficient in Fas/FasL-mediated cytotoxicity were identical to normal immunocompetent mice and exhibited absolute resistance to experimental MCMV retinitis, whereas PKO mice deficient in perforin-mediated cytotoxicity exhibited a high susceptibility to MCMV retinitis at a level that was equivalent to that observed in mice with MAIDS.
TABLE 1.
Frequency and severity of necrotizing MCMV retinitis at 8 to 10 days postinfectiona
| Mouse type | Frequency (%) of necrotizing retinitis (no. with retinitis/total) | Median posterior segment score, range, and % maximum |
|---|---|---|
| Normal | 0 (0/8) | 1.25, 0-2.0, 5 |
| gld | 0 (0/8) | 1.0, 0-1.5, 4 |
| PKO | 92 (12/13)∗ | 13.0, 2.0-20, 54∗∗ |
| MAIDS | 88 (7/8)∗ | 13.5, 3.0-21, 56∗∗ |
∗, significantly different from normal group (P ≤ 0.001); ∗∗, significantly different from normal group (P ≤ 0.05).
FIG. 1.
(A) Photomicrograph of a section of retina of a gld mouse 10 days after subretinal inoculation with MCMV. Retinal necrosis is absent, but retinal folding is present as observed in MCMV-inoculated eyes of normal C57BL/6 mice (not shown) (10, 12). (B) Photomicrograph of a section of retina of a PKO mouse 10 days after subretinal inoculation with MCMV. There is full-thickness retinal necrosis that contains cytomegalic cells (arrow) as observed in MCMV-inoculated eyes of mice with MAIDS (not shown) (10, 12). A hematoxylin and eosin stain was used; original magnification, ×200.
Recovery of infectious virus from MCMV-infected eyes of PKO and gld mice.
The presence of MCMV-induced intranuclear inclusions within the cells of both the neurosensory retina and retinal pigment epithelium supports the existence of productive virus infection during the pathogenesis of experimental MCMV retinitis. It has been shown previously that development of retinitis in MCMV-infected eyes of mice with MAIDS correlates with the recovery of large amounts of infectious virus from whole eyes (10, 11). To determine if this correlation extended to MCMV-infected eyes of PKO mice that are susceptible to retinitis, groups of PKO mice, gld mice, MAIDS mice, and normal mice were inoculated subretinally with MCMV. Eight days later, whole eyes were collected, homogenized, and subjected to a quantitative plaque assay for detection of infectious virus. Results are shown in Table 2. In agreement with susceptibility results, MCMV-infected eyes of normal or gld mice that were resistant to retinitis displayed measurable but relatively low levels of infectious MCMV. This was not the case for MCMV-infected eyes of PKO or MAIDS mice that exhibited high susceptibility to retinitis. These eyes harbored high titers of infectious virus consistent with the presence of productive infection during retinitis. These findings support an earlier observation that development of retinitis correlates with high titers of intraocular virus (10, 11) and suggest that PKO mice but not gld mice are unable to clear infectious MCMV from ocular tissues when compared at day 8 after subretinal MCMV inoculation.
TABLE 2.
MCMV titers of whole eyes and whole spleensc
| Mouse type | Titer in:
|
|
|---|---|---|
| Eyesa (PFU/ml/eye) | Liversb (PFU/ml/0.1 g) | |
| Normal | 2.2 log10 | 2.1 log10 |
| gld | 2.3 log10 | 3.6 log10 |
| PKO | 4.5 log10∗ | 6.1 log10∗∗ |
| MAIDS | 4.3 log10∗ | 5.1 log10∗∗ |
Mean titers of three to five whole eyes collected at 6 days after subretinal MCMV inoculation.
Mean titers of five whole livers collected at 5 days after intraperitoneal MCMV inoculation.
∗, statistically different from eyes of normal and gld mice (P < 0.05); ∗∗, statistically different from livers of normal and gld mice (P < 0.05).
Susceptibility of PKO and gld mice to MCMV hepatitis.
One might argue that results obtained following subretinal inoculation of PKO and gld mice are unique to the ocular compartment due to its immune-privileged status (49) and therefore do not reflect a generalized systemic immune response that leads to virus clearance. To explore this possibility, an experiment was performed to determine the ability of PKO and gld mice to clear virus from the liver during MCMV hepatitis following intraperitoneal inoculation of the virus. This route of virus inoculation produces systemic MCMV infection and dissemination of virus to the liver as well as to the spleen, pancreas, lungs, and salivary glands of immunocompetent mice (46). Mice with MAIDS also develop MCMV hepatitis following intraperitoneal virus inoculation and exhibit impaired clearance of infectious virus from the liver (41). Groups of PKO mice, gld mice, MAIDS mice, and normal mice were therefore inoculated intraperitoneally with a sublethal dose (∼103 PFU) of MCMV. Five days later, whole livers were collected from all mice, individually weighed, individually homogenized, and subjected to quantitative plaque assay. Results demonstrated that PKO mice and MAIDS mice were similar in their inability to clear virus from liver tissue (Table 2). In comparison, better virus clearance was observed in the livers of gld and normal mice as evidenced by a significant reduction of virus titers at nearly equivalent levels. Results obtained from a study of MCMV hepatitis involving systemic virus infection therefore mirrored those obtained using the same animal groups to study MCMV retinitis following subretinal virus infection.
Diminished perforin expression in splenic T cells of mice with MAIDS.
Our finding that mice deficient in perforin and the perforin cytotoxic pathway (PKO mice) are susceptible to experimental MCMV retinitis at a level that is equivalent to mice immunosuppressed by retrovirus infection (MAIDS mice) prompted us to hypothesize that MAIDS is associated with a downregulation of the perforin cytotoxic pathway. To test this hypothesis, we developed a competitive RT-PCR assay for perforin mRNA and used this assay to quantify perforin mRNA levels within mouse tissues as a measure of perforin-mediated cytotoxicity. Initial experiments were directed at the measurement and comparison of perforin mRNA levels within the spleens of immunocompetent or MAIDS mice who were infected with MCMV by subretinal inoculation, a route of virus inoculation that induces the production of MCMV-specific splenic T cells (13). Three days following uniocular subretinal MCMV inoculation, lymphocytes were collected from the pooled spleens of both animal groups and enriched for T lymphocytes by passage over nylon wool columns. The enrichment procedure was especially important for the spleens of MAIDS animals, since these spleens exhibit a profound splenomegaly due to B-cell lymphoproliferation and therefore contain a significant number of B lymphocytes (24). Following the enrichment procedure, approximately 75% of the recovered cells from both normal and MAIDS spleens were T lymphocytes as determined by flow cytometry analysis (data not shown). Total RNA was recovered from an equivalent number of splenic T lymphocytes of both groups, and quantitative RT-PCR was performed (5, 7). Results showed that splenic T lymphocytes from MCMV-infected normal mice contained ∼5,000 fg of perforin mRNA per 500 ng of total RNA, whereas an equal number of splenic T lymphocytes from MCMV-infected MAIDS mice contained less than 50 fg of perforin mRNA per 500 ng of total RNA. These data demonstrate that perforin mRNA levels in splenic T lymphocytes are significantly decreased during MAIDS, by ∼100-fold, from perforin mRNA levels in immunocompetent animals.
Diminished perforin expression in MCMV-inoculated eyes of mice with MAIDS.
We next performed experiments to investigate directly the expression of perforin mRNA in MCMV-inoculated eyes early in the course of ocular MCMV infection. Whole MCMV-infected eyes from groups of normal mice or MAIDS mice were collected at 3 days following subretinal MCMV inoculation and were individually subjected to quantitative RT-PCR assay for perforin mRNA expression. Unmanipulated normal eyes from separate uninfected healthy mice served as controls to determine possible background expression of perforin mRNA by the resident cell populations. In the eyes of uninfected normal mice, MCMV-infected normal mice, and MCMV-infected MAIDS mice, the levels of perforin mRNA were < 1, 168 ± 32 (mean plus or minus standard error of the mean), and 3 ± 1 fg of perforin mRNA per 500 ng of total RNA, respectively. The levels of perforin mRNA in the eyes of MCMV-infected normal mice and MCMV-infected MAIDS mice were statistically significantly different. As expected, control unmanipulated normal eyes expressed no detectable perforin mRNA, indicating an extremely low background expression of perforin in the absence of MCMV infection. In comparison, MCMV-inoculated eyes from normal mice that were destined to be resistant to MCMV retinitis demonstrated high levels of detectable perforin mRNA. In sharp contrast, however, MCMV-inoculated eyes from mice with MAIDS that were destined to become susceptible to MCMV retinitis demonstrated significantly lower levels of detectable perforin mRNA (P < 0.05) (Fig. 2). Thus, susceptibility to MCMV retinitis during MAIDS correlates with a significant decrease in perforin mRNA levels within the ocular compartment following subretinal MCMV infection.
FIG. 2.
Ethidium bromide-stained agarose gel of competitive RT-PCR assay for mouse perforin mRNA in individual eyes collected at 3 days after subretinal MCMV inoculation. Top, MCMV-inoculated eye from a healthy C57BL/6 mouse showing equivalence at ∼100 fg of perforin mRNA per 500 ng of total RNA. Bottom, MCMV-inoculated eye from a mouse with MAIDS showing lack of detection of authentic perforin mRNA. (Authentic perforin mRNA = 442 bp; competitive template = 380 bp).
DISCUSSION
Two important findings have emerged from this study. Firstly, we provide the first evidence that the perforin cytotoxic pathway is more important than the Fas/FasL cytotoxic pathway in providing resistance to HCMV retinitis. Approximately 80% of adults worldwide are infected with HCMV, and the virus establishes a lifelong latent infection (20). While it has been generally accepted that healthy persons are resistant to HCMV retinitis due to a strong cellular immune response provided by NK cells and cytotoxic CD8+ T cells (4), the precise immunologic pathway(s) that actually provides this protection has remained unexplored until this study. Our finding that mice deficient in the perforin cytotoxic pathway (PKO mice) are highly susceptible to experimental MCMV retinitis but that mice deficient in the Fas/FasL cytotoxic pathway (gld mice) exhibit absolute resistance to retinitis as occurs in normal, healthy, mice underscores the importance of the perforin pathway in providing protection against the disease. This conclusion is strengthened by the observation that intraocular titers of infectious MCMV are significantly higher in MCMV-infected eyes of PKO mice with histopathologic features of MCMV retinitis but that MCMV-infected eyes of gld and normal mice harbor equivalently low intraocular titers of infectious virus in the absence of retinitis. Whether the perforin cytotoxic pathway predominates in protection against diseases other than retinitis remains to be determined. It is noteworthy, however, that in the present study the livers of PKO mice inoculated intraperitoneally with MCMV exhibited high titers of infectious virus, whereas the livers of gld and normal mice exhibited lower virus titers. Thus, the perforin cytotoxic pathway might also provide protection against virus-induced hepatitis and other systemic diseases in addition to protection against ocular disease.
Secondly, we provide the first evidence that mice with MAIDS are highly susceptible to experimental MCMV retinitis due to downregulation of the perforin cytotoxic pathway as measured by a significant decrease in perforin mRNA levels in MCMV-infected eyes of mice with MAIDS from those in MCMV-infected eyes or unmanipulated eyes of normal mice. Confirmation of this conclusion must await direct testing of the perforin pathway in an in vitro cell killing assay with lymphocytes from mice with MAIDS. It is important, however, that splenic T lymphocytes from mice with MAIDS also exhibited a profound decrease in perforin mRNA levels from those found in splenic T lymphocytes recovered from normal mice.
MAIDS shares many immunopathologic features with AIDS in humans (24). Both immunodeficiencies are caused by pathogenic, species-specific retroviruses. Moreover, both MAIDS and AIDS are characterized by progressive development of chronic generalized lymphadenopathy, polyclonal B-cell activation, hypergammaglobulinemia, a shift in cytokine production from a Th1 profile to a Th2 profile, diminished NK cell and CD8+-T-cell functions, and increased susceptibility to opportunistic infections (24). It is therefore intriguing to speculate that HIV-1-induced AIDS in humans is also associated with a retrovirus-induced downregulation of the perforin cytotoxic pathway, thereby rendering the patient susceptible to HCMV retinitis. Previous studies attempting to correlate immune status and onset and severity of retinitis during AIDS have focused exclusively on the quantification of individual immune cell populations, such as CD4+- and CD8+-T-cell counts (30, 32), but not on functional aspects of the immune response. Findings with an established MAIDS model of experimental MCMV retinitis (10) predict that loss of effector cell function (i.e., the perforin cytotoxic pathway) as opposed to loss of individual effector cell populations might be more important in predisposing to onset of HCMV retinitis in the HIV-1-immunosuppressed patient.
Our results also predict that cytokine immunotherapy that targets the perforin cytotoxic pathway but not the Fas/FasL pathway will restore resistance to retinitis. Previous in vitro studies have shown that exogenous interleukin-2 (IL-2), a Th1 regulatory cytokine of T-cell origin with potent effects on T cells, B cells, and NK cells (47), enhances the depressed NK cell activity and HCMV-specific cytotoxic T-cell activity of peripheral blood mononuclear cells recovered from patients with AIDS (43). Moreover, it has been shown previously that systemic cytokine immunotherapy with IL-2 during MAIDS will restore resistance to MCMV retinitis at a level seen in normal, healthy mice following subretinal MCMV inoculation (12). Experiments are presently in progress to determine if protection provided by IL-2 immunotherapy during MAIDS correlates with an upregulation of perforin mRNA levels. Such a finding would support our working hypothesis that downregulation of the perforin cytotoxic pathway during MAIDS is due indirectly to a retrovirus-induced shift in cytokine production by CD4+ helper T cells that favors a Th2 profile over a Th1 profile.
Why the Fas/FasL cytotoxic pathway does not appear to play a role in protection against experimental MCMV retinitis during MAIDS is unclear. The Fas/FasL system has been recognized as a mechanism of clonal deletion (36) as well as a means for controlling T-cell expansion during immune responses (25). FasL is expressed constitutively in the eye of mice, rats, and humans, where it is displayed abundantly on cells of the cornea, iris, ciliary body, and retina (14). Moreover, it is distributed at or near areas of the eye that comprise the blood-ocular barrier, where there is an opportunity for interaction with inflammatory cells. Thus, FasL expression in the eye is thought to be important in the maintenance of immune privilege as well as immune tolerance in the eye by apoptotic killing of inflammatory cells that invade the eye. In addition to serving as a major factor in promoting corneal allograft survival (50) and regulating subretinal angiogenesis (28), FasL expression in the eye induces apoptotic death in Fas+ lymphoid cells that enter the organ in response to experimental anterior chamber infection of normal C57BL/6 mice with herpes simplex virus type 1 (17). Consequently, inflammatory responses are limited in their spread throughout the eye, generally confined to the site of virus injection. In sharp contrast, herpes simplex virus type 1 anterior chamber inoculation of gld mice results in a rapidly proliferating inflammation that spreads beyond the site of initial virus infection and ultimately consumes the retina. Our finding that gld mice are resistant to experimental MCMV retinitis following subretinal MCMV inoculation suggests that pathways of cytotoxicity may be virus specific in terms of providing protection.
Immunosuppressed patient populations other than those who suffer from HIV-1 infection are also susceptible to a wide spectrum of HCMV diseases. Solid-organ and bone marrow transplant recipients not treated with antiviral chemotherapy are susceptible to HCMV pneumonitis, which accounts for significant mortality ranging from 40 to 70% and 70 to 85% in these patient populations, respectively (19). Surprisingly, less than 5% of transplant patients suffer from HCMV retinitis (19). The converse is true during AIDS, where HCMV pneumonitis is rarely life threatening but where up to 40% of patients are susceptible to HCMV retinitis (45). Thus, different conditions of immunosuppression result in distinct patterns of HCMV disease, an observation that suggests that the pathophysiology of a particular HCMV disease need not be uniform in different immunosuppressed patient populations. One might argue, therefore, that loss of the perforin cytotoxic pathway accounts for susceptibility to HCMV retinitis in AIDS patients only but does not play a role in solid-organ or bone marrow transplant rejection. Recent work by Li and coworkers (31), however, that appeared in the literature as our study was ongoing would speak in favor of a role for the perforin cytotoxic pathway in acute rejection of renal allografts. These investigators developed a quantitative RT-PCR assay similar to ours to measure mRNA for perforin and granzyme B in urinary cells of renal transplant patients and used this assay as a noninvasive means for diagnosing acute rejection of renal allografts. Whether the perforin cytotoxic pathway plays a role in susceptibility to the rare cases of HCMV retinitis in immunosuppressed transplant recipients remains to be determined. Nevertheless, our results, coupled with those of Li and coworkers (31), raise the intriguing possibility that measurement of perforin mRNA is a far better quantification of cellular immunity (and susceptibility to HCMV disease) than measurement of absolute CD4+- and/or CD8+-T-cell numbers and might be used in the clinical setting as a predictor for those immunosuppressed patients who are destined to develop HCMV retinitis.
Acknowledgments
We thank Xiao-Kun Liu, Jason Stroh, and Eleut Hernandez for excellent technical assistance.
This work was supported by NIH grant EY10568 and in part by unrestricted grants from Research to Prevent Blindness and the Pat & Willard Walker Eye Research Center.
REFERENCES
- 1.Atherton, S. S., C. K. Newell, M. Y. Kanter, and S. W. Cousins. 1991. Retinitis in euthymic mice following inoculation of murine cytomegalovirus (MCMV) via the supraciliary route. Curr. Eye Res. 10:667-677. [DOI] [PubMed] [Google Scholar]
- 2.Atherton, S. S., C. K. Newell, M. Y. Kanter, and S. W. Cousins. 1992. T cell depletion increases susceptibility to murine cytomegalovirus retinitis. Investig. Ophthalmol. Vis. Sci. 33:3353-3360. [PubMed] [Google Scholar]
- 3.Bigger, J. E., C. A. Thomas III, and S. S. Atherton. 1998. NK cell modulation of murine cytomegalovirus retinitis. J. Immunol. 160:5826-5831. [PubMed] [Google Scholar]
- 4.Britt, W. J., and C. A. Alford. 1996. Cytomegalovirus, p. 2493-2523. In B. N. Fields, D. M. Knipe, and P. M. Howley (ed.), Fields virology, 3rd ed. Lippincott-Raven Publishers, Philadelphia, Pa.
- 5.Celi, F. S., M. E. Zenilman, and A. R. Shuldiner. 1993. A rapid and versatile method to synthesize internal standards for competitive PCR. Nucleic Acids Res. 21:1047-1056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chang, H. Y., H. Nishotoh, X. Yang, H. Ichijo, and D. Baltimore. 1998. Activation of apoptosis signal-regulating kinase 1 (ASK1) by the adapter protein Daxx. Science 281:1860-1863. [DOI] [PubMed] [Google Scholar]
- 7.Chang-Fang, J., and M. Marina. 1994. Rapid construction of deleted DNA fragments for use as internal standards in competitive PCR. PCR Methods Appl. 3:252-255. [DOI] [PubMed] [Google Scholar]
- 8.Cunningham, E. T., and T. P. Margolis. 1998. Ocular manifestations of HIV infection. N. Engl. J. Med. 339:236-244. [DOI] [PubMed] [Google Scholar]
- 9.Dix, R. D. 1998. Systemic murine cytomegalovirus infection of mice with retrovirus-induced immunodeficiency results in ocular infection but not retinitis. Ophthalm. Res. 30:295-301. [DOI] [PubMed] [Google Scholar]
- 10.Dix, R. D., C. C. Cray, and S. W. Cousins. 1994. Mice immunosuppressed by murine retrovirus infection (MAIDS) are susceptible to cytomegalovirus retinitis. Curr. Eye Res. 13:587-595. [DOI] [PubMed] [Google Scholar]
- 11.Dix, R. D., C. Cray, and S. W. Cousins. 1997. Antibody alone does not prevent experimental cytomegalovirus retinitis in mice with retrovirus-induced immunodeficiency (MAIDS). Ophthalm. Res. 29:381-392. [DOI] [PubMed] [Google Scholar]
- 12.Dix, R. D., M. Giedlin, and S. W. Cousins. 1997. Systemic cytokine immunotherapy for experimental cytomegalovirus retinitis in mice with retrovirus-induced immunodeficiency. Investig. Ophthalmol. Vis. Sci. 38:1411-1417. [PubMed] [Google Scholar]
- 13.Duan, Y., Z. Yi, and S. S. Atherton. 1994. Dissemination and replication of murine cytomegalovirus after supraciliary invoulation in immunosuppressed mice. Investig. Ophthalmol. Vis. Sci. 35:1124-1131. [PubMed] [Google Scholar]
- 14.Ferguson, T. A., and T. S. Griffith. 1997. A vision of cell death: insights into immune privilege. Immunol. Rev. 156:167-184. [DOI] [PubMed] [Google Scholar]
- 15.Gao, E.-R., X.-H. Yu, P.-C. Lin, H. Zhang, and H. J. Kaplan. 1995. Intraocular viral replication after systemic murine cytomegalovirus infection requires immunosuppression. Investig. Ophthalmol. Vis. Sci. 36:2322-2327. [PubMed] [Google Scholar]
- 16.Goldstein, P. 1995. Fas-based T cell-mediated cytotoxicity. Curr. Top. Microbiol. Immunol. 198:25-37. [DOI] [PubMed] [Google Scholar]
- 17.Griffin, T. S., T. Brunner, S. M. Fletcher, D. R. Green, and T. A. Ferguson. 1995. Fas ligand-induced apoptosis as a mechanism of immune privilege. Science 270:1189-1192. [DOI] [PubMed] [Google Scholar]
- 18.Hartley, J. W., T. N. Fredrickson, R. A. Yetter, M. Makino, and H. C. Morse III. 1989. Retrovirus-induced murine acquired immunodeficiency syndrome: natural history of infection and differing susceptibility of inbred mouse strains. J. Virol. 63:1223-1231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ho, M. 1994. Advances in understanding cytomegalovirus infection after transplantation. Transplant. Proc. 26:7-11. [PubMed] [Google Scholar]
- 20.Holland, G. N., A. Tufail, and M. C. Jordan. 1996. Cytomegalovirus disease, p. 1088-1130. In J. S. Pepose, G. N. Holland, and K. R. Wilhelmus (ed.), Ocular infections and immunity. C.V. Mosby Co., Inc., St. Louis, Mo.
- 21.Inoue, Y., P. Minasi, and J. O. Oh. 1993. The role of natural killer cells in murine cytomegalovirus eye infections. Investig. Ophthalmol. Vis. Sci. 34:3353-3360. [PubMed] [Google Scholar]
- 22.Jabs, D. A., and J. G. Bartlett. 1997. AIDS and ophthalmology: a period of transition. Am. J. Ophthalmol. 124:227-233. [DOI] [PubMed] [Google Scholar]
- 23.Jenne, D. E., and J. Tschopp. 1988. Granyzmes, a family of serine proteases released from granules of cytolytic T-lymphocytes upon T-cell receptor stimulation. Immunol. Rev. 103:53-71. [DOI] [PubMed] [Google Scholar]
- 24.Jolicoeur, P. 1991. Murine acquired immunodeficiency syndrome (MAIDS): an animal model to study AIDS pathogenesis. FASEB J. 5:2398-2405. [DOI] [PubMed] [Google Scholar]
- 25.Ju, S.-T., D. J. Panka, H. Cui, R. Ettinger, J. El-Khatib, D. H. Sherr, B. Z. Stanger, and A. Marshak-Rothstein. 1995. Fas(CD95)/FasL interactions required for programmed cell death after T-cell activation. Nature (London) 373:444-448. [DOI] [PubMed] [Google Scholar]
- 26.Kagi, D., B. Ledermann, K. Burki, P. Seiler, B. Odermatt, K. J. Olsen, E. R. Podack, R. M. Zindernagel, and H. Hengartner. 1994. Cytotoxicity mediated by T cells and natural killer cells is greatly impaired in perforin-deficient mice. Nature 369:313-337. [DOI] [PubMed] [Google Scholar]
- 27.Kagi, D., F. Vignaux, B. Ledermann, K. Burki, V. Depraitere, S. Nagata, H. Hengartner, and P. Golstein. 1994. Fas and perforin pathways as major mechanisms of T cell-mediated cytotoxicity. Science 265:528-530. [DOI] [PubMed] [Google Scholar]
- 28.Kaplan, H. J., M. A. Leibole, T. Tezel, and T. A. Ferguson. 1999. Fas ligand (CD95 ligand) controls angiogenesis beneath the retina. Nat. Med. 5:292-297. [DOI] [PubMed] [Google Scholar]
- 29.Kercher, L., and B. M. Mitchell. 2000. Immune transfer protects severely immunosuppressed mice from murine cytomegalovirus retinitis and reduces the viral load in ocular tissue. J. Infect. Dis. 182:652-661. [DOI] [PubMed] [Google Scholar]
- 30.Kupperman, B. D., J. G. Petty, D. D. Richman, W. C. Mathews, S. C. Fullerton, L. S. Rickman, and W. R. Freeman. 1993. Correlation between CD4+ counts and prevalence of cytomegalovirus retinitis and human immunodeficiency virus-related noninfectious retinal vasculopathy in patients with acquired immunodeficiency syndrome. Am. J. Ophthalmol. 115:575-582. [DOI] [PubMed] [Google Scholar]
- 31.Li, B., C. Hartono, R. Ding, V. K. Sharma, R. Ramaswamy, B. Quin, D. Serur, J. Mouradian, J. E. Schwartz, and M. Suthanthiran. 2001. Noninvasive diagnosis of renal-allograft rejection by measurement of messenger RNA for perforin and granzyme B in urine. N. Engl. J. Med. 344:947-954. [DOI] [PubMed] [Google Scholar]
- 32.Lowder, C. Y., C. P. Butler, E. M. Dodds, M. Secic, and C. Recillas-Gispert. 1995. CD8+ T lymphocytes and cytomegalovirus retinitis in patients with the acquired immunodeficiency syndrome. Am. J. Ophthalmol. 120:283-290. [DOI] [PubMed] [Google Scholar]
- 33.Lowin, B., F. Beermann, A. Schmidt, and J. Tschopp. 1994. A null mutation in the perforin gene impairs cytolytic T lymphocyte- and natural killer cell-mediated cytotoxicity. Proc. Natl. Acad. Sci. USA 91:11571-11575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lowin, B., M. Hahne, C. Mattmann, and J. Tschopp. 1994. Cytolytic T-cell cytotoxicity is mediated through perforin and Fas lytic pathways. Nature 370:650-652. [DOI] [PubMed] [Google Scholar]
- 35.Lowin, B., M. C. Peitsch, and J. Tschopp. 1995. Perforin and granzymes: crucial effector molecules in cytolytic T lymphocyte and natural killer cell-mediated cytotoxicity. Curr. Top. Microbiol. Immunol. 198:1-24. [DOI] [PubMed] [Google Scholar]
- 36.Mogil, R. J., L. Fadvanyi, R. Gonzalez-Quintial, R. Miller, G. Mills, A. N. Theofilopoulos, and D. R. Green. 1995. Fas (CD95) participates in peripheral T cell depletion and associated apoptosis in vivo. Int. Immunol. 7:1451-1458. [DOI] [PubMed] [Google Scholar]
- 37.Muzio, M., A. M. Chinnaiyan, F. C. Kischkel, K. O'Rourke, A. Shevchenko, J. Ni, C. Schaffidi, J. D. Bretz, M. Zhang, R. Gentz, M. Mann, P. H. Krammes, M. E. Peter, and V. M. Dixit. 1996. FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death-inducing signaling complex. Cell 85:817-827. [DOI] [PubMed] [Google Scholar]
- 38.Nagata, S., and P. Goldstein. 1995. The Fas death factor. Science 267:1449-1461. [DOI] [PubMed] [Google Scholar]
- 39.Nagata, S., and T. Suda. 1995. Fas and Fas ligand: lpr and gld mutations. Immunol. Today 16:39-43. [DOI] [PubMed] [Google Scholar]
- 40.Palella, F. J., Jr., K. M. Delaney, A. C. Moorman, J. O. Loveless, J. Fuhrer, G. A. Satten, D. J. Aschman, S. D. Holmberg, and the HIV Outpatient Study Investigators. 1998. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. N. Engl. J. Med. 338:853-860. [DOI] [PubMed] [Google Scholar]
- 41.Peacock, C. D., S. D. Olver, and P. Price. 1997. Effect of a retroviral immunodeficiency syndrome on murine cytomegalovirus-induced hepatitis. Am. J. Pathol. 150:1089-1100. [PMC free article] [PubMed] [Google Scholar]
- 42.Podack, E. R. 1995. Perforin, killer cells and gene transfer immunotherapy for cancer. Curr. Top. Microbiol. Immunol. 198:121-130. [DOI] [PubMed] [Google Scholar]
- 43.Rook, A. H., H. Masur, H. C. Lane, W. Frederick, T. Kasahara, A. M. Macher, J. Y. Djeu, J. F. Manischewitz, L. Jackson, A. S. Fauci, and G. V. Quinnan, Jr. 1983. Interleukin-2 enhances the depressed natural killer and cytomegalovirus-specific cytotoxic activities of lymphocytes of patients with the acquired immune deficiency syndrome. J. Clin. Investig. 72:398-403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Russell, J. H., and T. J. Ley. 2002. Lymphocyte-mediated cytotoxicity. Annu. Rev. Immunol. 20:323-370. [DOI] [PubMed] [Google Scholar]
- 45.Schacker, T., and L. Corey. 1997. Herpesvirus infections in human immunodeficiency virus-infected persons, p. 267-280. In V. T. DeVita, Jr., S. Hellman, and S. A. Rosenberg (ed.), AIDS: biology, diagnosis, treatment, and prevention, 4th ed. Lippincott-Raven Publishers, Philadelphia, Pa.
- 46.Shanley, J. D., L. Biczak, and S. J. Forman. 1993. Acute murine cytomegalovirus infection induces lethal hepatitis. J. Infect. Dis. 167:264-269. [DOI] [PubMed] [Google Scholar]
- 47.Smith, K. A. 1988. Interleukin-2: inception, impact, and implications. Science 240:1169-1176. [DOI] [PubMed] [Google Scholar]
- 48.Smyth, M. J., and J. A. Trapani. 1998. The relative role of lymphocyte granule exocytosis versus death receptor-mediated cytotoxicity in viral pathophysiology. J. Virol. 72:1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Streilein, J. W. 1999. Regional immunity and ocular immune privilege, p. 11-38. In J. W. Streilein (ed.), Immune response and the eye, vol. 73. Karger, Basel, Switzerland. [DOI] [PubMed] [Google Scholar]
- 50.Stuart, P. M., T. S. Griffith, N. Usui, J. Pepose, X. Yu, and T. A. Ferguson. 1997. CD95 ligand (FasL)-induced apoptosis is necessary for corneal allograft survival. J. Clin. Investig. 99:396-402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Walsh, C. M., M. Matloubian, C.-C. Liu, R. Ueda, C. G. Kurahara, J. L. Christensen, M. T. F. Huang, J. D.-E. Young, R. Ahmed, and W. R. Clark. 1994. Immune function in mice lacking the perforin gene. Proc. Natl. Acad. Sci. USA 91:10854-10858. [DOI] [PMC free article] [PubMed] [Google Scholar]