Abstract
With patients demanding a greater role in the clinical decision-making process, many researchers are developing and disseminating decision aids for various medical conditions. In this article, we outline the essential elements in the development and evaluation of a decision aid to help patients with atrial fibrillation choose, in consultation with their physicians, appropriate antithrombotic therapy (warfarin, aspirin, or no therapy) to prevent stroke. We also outline possible future directions regarding the implementation and evaluation of this decision aid. This information should enable clinicians to better understand the role that decision aids may have in their interactions with patients.
Keywords: decision aid, atrial fibrillation, warfarin, aspirin
Decision aids are tools designed to help individual patients make clinical decisions that are optimal for them. They are adjuncts to physician counseling and are not replacements for patient-physician encounters. Decision aids are different from general educational materials (such as informational pamphlets) in that they provide more detailed information about the benefits and risks of different therapeutic options, use explicit probabilities to explain the chance of pertinent outcomes (usually tailored to the patients' clinical situations), and ask patients to indicate which therapy they favor.1 With patients demanding greater roles in the clinical decision-making process, many researchers are developing and disseminating decision aids for various medical conditions using a number of different formats, including interactive videodiscs,2 audiobooklets,3 and decision boards.4
We have recently developed a decision aid to help patients with atrial fibrillation decide the appropriate antithrombotic therapy for them. Patients with atrial fibrillation are five times more likely to suffer a stroke compared with those without atrial fibrillation.5 To prevent stroke from atrial fibrillation, both warfarin and aspirin have been extensively studied in randomized trials6–9 and are commonly used in clinical practice. Warfarin is more efficacious but has a greater number of adverse effects and is more inconvenient to use. Numerous studies have shown that many patients who are candidates for warfarin therapy are not receiving it.10–14 This may be because some patients and physicians believe that the benefits of warfarin do not outweigh its disadvantages. The decision regarding whether to take warfarin, aspirin, or neither depends upon the patients' understanding of the risks and benefits of the two medications, and how they value the trade-off between the reduction to the likelihood of stroke and the increase in major hemorrhage.
Previous papers from our Ottawa group have described the general decision support framework used to develop and evaluate decision aids.15,16 However, these papers did not provide detailed information regarding the specific process used in the development of an individual decision aid. Therefore, in this paper, we describe in detail the process used to develop a decision aid for patients with atrial fibrillation who are considering antithrombotic therapy. Although this article deals with antithrombotic therapy for atrial fibrillation, many of the issues are pertinent to the development of decision aids for other therapies.
METHODS
The issues considered when developing our decision aid are outlined in Table 1.
Table 1.
Issues to Consider When Developing aDecision Aid
| 1. Clinical considerations |
| ⋄Choice of therapies |
| ⋄Determining the efficacy and side effects of the therapies |
| ⋄Choice of outcomes |
| ⋄Description of the consequences of the outcomes |
| ⋄Tailoring the probabilities of outcomes to patients' individual circumstances |
| 2. Format of the decision aid |
| ⋄Type of decision aid |
| ⋄Presenting the probabilities |
| ⋄Personal worksheet and values clarification |
| ⋄Physician's manual |
| 3. Pilot testing and approval of the decision aid by patients and physicians |
| 4. Validation of the decision aid |
Clinical Considerations
A number of clinical issues were considered when developing this decision aid.
Choice of Therapies
Warfarin and aspirin clearly needed to be included in the decision aid. The dose of aspirin we decided to use was 325 mg daily as this is the dose commonly used in clinical practice. Indobufen, another antithrombotic agent, was found efficacious in one randomized trial,17 but was not included because it is not widely available.
Efficacy and Side Effects of Warfarin and Aspirin
Individual patient meta-analyses of randomized trials have found that warfarin decreases the relative risk of stroke by 68% (95% confidence limits [CL], 50 to 79)6 and aspirin decreases the risk of stroke by 21% (95% CL, 0 to 38).7 The most serious side effect of these medications is major hemorrhage. Data on the frequency of major hemorrhage can be obtained from the randomized trials in patients with atrial fibrillation,18 cohort studies of patients treated with warfarin in anticoagulation clinics (most of whom did not have atrial fibrillation),19–24 and case control and cohort studies of patients treated with aspirin.25–27 The frequency of major hemorrhage attributable to warfarin was lowest in the randomized trials (an increase of between 0% and 2% per year) and highest in the cohort studies (0.6% to 5% per year). The increase in major bleeding associated with aspirin therapy appeared to be about 0.12% to 0.25% per year.28
Choice of Outcomes
The most common cardioembolic event for patients with atrial fibrillation is a stroke, accounting for about 90% of all clinically detected events.18 Therefore, patients should be informed about the consequences of a stroke. Noncerebral emboli constitute the other 10%, with their presentation being extremely diverse (e.g., mesenteric, retinal, etc.). Therefore, we omitted them, reasoning that they are comparatively rare and that their consequences are similar in severity to a stroke.
For major hemorrhages that occur when taking warfarin and aspirin, intracranial hemorrhages (either intracerebral or subdural) are the most serious but are rare, even in patients taking warfarin if the international normalized ratio (INR) is kept between 2.0 and 3.0; gastrointestinal hemorrhages are the most common. We decided not to describe intracranial hemorrhages, but instead incorporated them as “strokes” when presenting the probabilities of a stroke. We felt that since intracranial hemorrhages present with the symptoms of a stroke (although they are usually more severe than the average nonhemorrhagic stroke), it would be less confusing for patients if they were presented as strokes. Thus, symptomatic gastrointestinal hemorrhages and minor hemorrhages were the side effects of therapy described.
Description of the Consequences
Having determined the therapies and outcomes that would be presented, we then needed to describe their consequences.
Warfarin therapy
The consequences of taking warfarin therapy described were the need for regular blood testing (usually every 3 to 4 weeks), the cost of the drug if it is not covered by insurance, avoiding activities that predispose to head injury, curtailing alcohol intake, and the need to check with a physician before starting new medications.
Aspirin therapy
Relative to warfarin therapy, aspirin does not require regular blood testing and dose adjustments and has fewer drug interactions. Therefore, the consequences of taking aspirin included only the need to take it on a regular basis.
Stroke
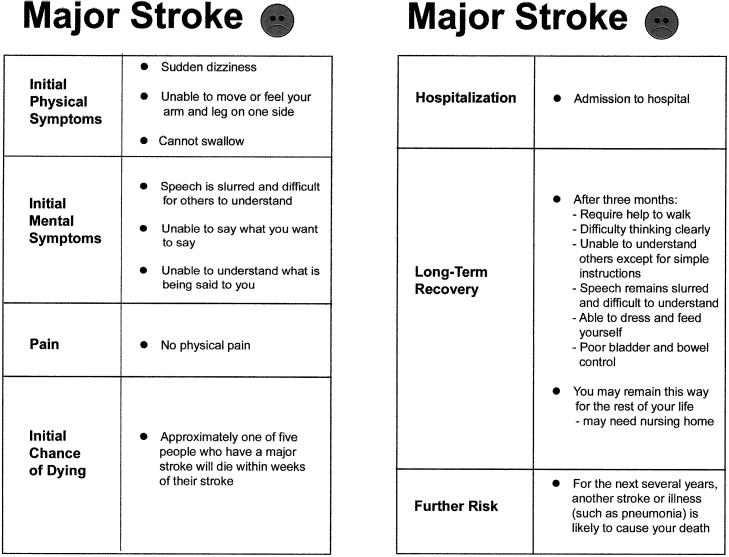
The severity of strokes can range from leaving no residual functional deficits to causing severe disability or death. Thus, describing the consequences of a stroke was one of the most challenging aspects of developing the decision aid. The main concepts we wished to convey were that strokes result in permanent brain damage, patients' recovery can range from full functional recovery to severe dependency or death, the chances of being left functionally dependent or independent are about equal,6 and there is no way of determining which patient will suffer a stroke or how disabling a stroke will be should it occur.
We presented detailed descriptions of the consequences of minor and major strokes, and emphasized that these examples were to illustrate the range of strokes that may occur. Figure 1 illustrates how we summarized the consequences of a major stroke. We hoped that the seven domains described and the descriptions of minor and major strokes would provide the patients with a balanced, reasonably comprehensive, and comprehensible description of the consequences of a stroke.
FIGURE 1.
Description of a major stroke used in the audiobooklet.
Symptomatic gastrointestinal hemorrhage
We described the consequences of a typical upper gastrointestinal hemorrhage (vomiting blood or passing melena, admission to hospital for up to a week, endoscopy, transfusion of 2 units of blood, need to take antiulcer medications for 6 months [the decision aid was developed prior to the widespread use of antibiotic therapy for Helicobacter pylori ], and no permanent residua). We mentioned that some patients died from a gastrointestinal hemorrhage but that this was “rare.” We implied that the consequences of a hemorrhage were similar whether taking warfarin, aspirin, or neither.
Other side effects of warfarin
We indicated that patients taking warfarin have an increase in the risk of minor bleeding episodes, and mentioned easy bruising and nosebleeds as examples.
Other side effects of aspirin
We indicated that patients taking aspirin have a slight increase in nausea and abdominal pain.
Tailoring the Probabilities of the Outcomes to Patients' Individual Circumstances
An advantage of decision aids over general educational materials is that they are tailored to the clinical circumstances of individual patients. The risk of stroke in patients with atrial fibrillation who do not take antithrombotic agents varies considerably, from 1% per year or less in young patients with no other clinical risk factors to at least 12% per year in elderly patients with a recent stroke.18 Therefore, we felt that it was important to develop different versions of the decision aid for various groups of patients. After considering the number of different decision aids that could practically be developed as well as the degree of precision provided by the risk stratification schemes available, we decided to develop four versions. Table 2 shows the risks of stroke over 2 years when taking no antithrombotic therapy, warfarin, or aspirin as presented for the four risk levels.
Table 2.
Risk Stratification Scheme for Audiobooklet Decision Aid
| Risk of Stroke Over 2 Years | |||
|---|---|---|---|
| Risk Level | No Therapy | Aspirin | Warfarin |
| Low | 3% | 2% | 1% |
| <70 years old, no hypertension,no diabetes, no congestive heartfailure, no previous TIA or stroke | |||
| Medium | 8% | 6% | 3% |
| >70 years old, no hypertension,no diabetes, no congestive heartfailure, no previous TIA or stroke, | |||
| or | |||
| <70 years old, and 1 or more ofthe following: hypertension,diabetes, congestive heart failure | |||
| High | 14% | 11% | 5% |
| >70 years old, and one or moreof the following: hypertension,diabetes, congestive heart failure | |||
| Very high | 20% | 16% | 7% |
| Previous TIA or stroke | |||
TIA indicates transient ischemic attack.
One of the most difficult tasks was deciding what to present to patients regarding their likelihood of stroke and major hemorrhage. We recognized that the results of randomized trials are not always directly generalizable to wider populations.29 However, we felt that the reduction in the risk of stroke associated with warfarin therapy was very consistent among studies and that none of these studies used strategies that differed from good clinical practice. Therefore, we used a relative risk reduction of 68%6 for warfarin therapy when deriving the probabilities used in the audiobooklet.
Deciding which value to use for the efficacy of aspirin was more difficult because the relative risk reduction associated with aspirin varied among the published trials. We thought that the most reliable and accurate estimate of the efficacy of aspirin was provided by a pooled analysis of individual patients (relative risk reduction of 21%).7 The decision aid was developed before the publication of two recent meta-analyses8,9 that have revised the estimates of the efficacy of aspirin and warfarin to prevent stroke in patients with atrial fibrillation.
Determining the risk of major hemorrhage in warfarin and aspirin was the most difficult and contentious issue encountered. For warfarin, the reasons for this included that for major hemorrhage, there were differences in the estimates among the randomized trials, differences in the estimates among cohort studies of patients receiving anticoagulation, differences in the estimates of major hemorrhage between the randomized trials and cohort studies, disagreements about whether the randomized trial estimates are generalizable to usual clinical practice, uncertainties about the effect of age upon the risk, and difficulties in determining how much the risk was affected by the method of monitoring the anticoagulation (INR vs prothrombin time [PT] ratio), the target INR (generally lower in the most recently published studies), and the frequency of routine measurement of the INR or PT ratio (varying from every 2 weeks to every 4 to 5 weeks). We eventually decided that an annual risk of 2% (4% over 2 years) was a reasonable estimate of the incremental risk of major hemorrhage attributable to warfarin, judging that this estimate was at the upper limit of the estimates from randomized trials and was still compatible with the cohort data.
For aspirin, the estimates of the risk of major hemorrhage from the randomized trials for patients with atrial fibrillation suffered from the same methodological limitations just described for warfarin. We used the estimates from randomized trials in atrial fibrillation, cohort studies of patients on aspirin, and a meta-analysis of randomized trials of aspirin therapy to derive an estimate of an incremental risk of major nonintracranial hemorrhage of 0.12% per year (or 0.25% over 2 years).
We were convinced that the risk of major hemorrhage increases with age, especially for patients older than 75 years of age.30 Therefore, we indicated that patients younger than 65 years probably have a risk of major hemorrhage that is slightly lower than 4% over 2 years, and patients older than 75 probably have a slightly higher risk. Finally, we decided against making separate decision aids for patients younger than 65 years of age, between 65 and 75, and older than 75, because the evidence for the effect of age upon the risk of major hemorrhage is methodologically weak, and this would have required the development of 12 different versions (four strata for the risk of stroke and three strata for the risk of major hemorrhage), which was clearly impractical.
Format of the Decision Aid
A number of issues were considered regarding the format of the decision aid.
Type of Decision Aid
We developed our decision aid as an audiobooklet, a format that some of us had previously used for a decision aid for women contemplating long-term hormone replacement therapy.3,31 The booklet highlighted key points (similar to a slide presentation) while the audiotape connected the points in a narrative, providing much more detail than the booklet.
Presentation of the Probabilities
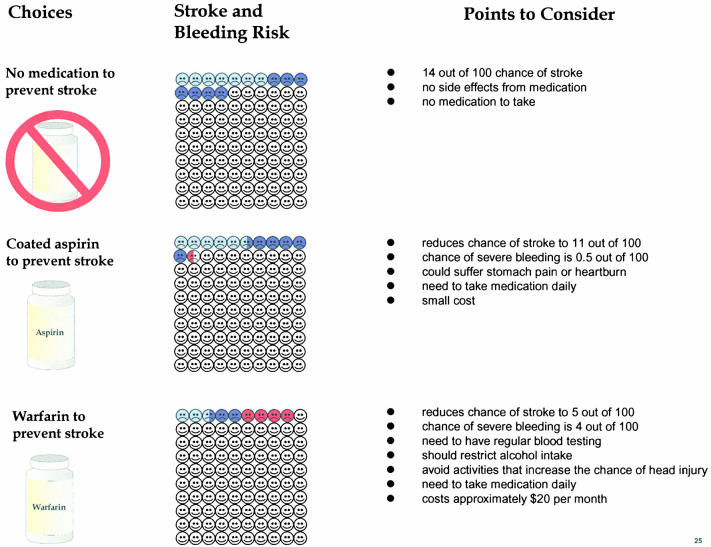
We presented the probabilities of stroke and major hemorrhage using 100 facial icons. Patients who had not suffered either a stroke or hemorrhage were represented with uncolored “happy faces.” Patients who had suffered a stroke were presented as blue “sad faces” (dark blue for major strokes and light blue for minor strokes), and patients who had had a hemorrhage were presented as red “sad faces” Figure 2. Beside each group of 100 icons, we indicated the probabilities of a good and bad outcome, and this was explained in detail in the audiotape (e.g., “This means that you would have a 8 in 100 chance of having a stroke, or a 92 in 100 chance of not having a stroke.”). We felt that presenting the probabilities in this way had a number of advantages. These included providing a clear visual message, presenting the patients with information about the absolute risk of the events, and providing the probabilities of having and not having an event.
FIGURE 2.
Summary pages from the audiobooklet. Light blue = stroke; dark blue = major stroke; red = major hemorrhage.
We provided patients with information about the likelihood of a stroke or major hemorrhage during the next 2 years. We chose 2 years because it was a reasonable compromise between the average length of follow-up in the randomized trials and the fact that both warfarin and aspirin are chronic therapies that most patients take indefinitely.
Personal Worksheet
After the factual information was provided, patients were guided to consider personal issues by completing a worksheet Figure 3. They described their own medical condition, clarified their values (explicitly considered the potential benefits and risks of warfarin and aspirin), recorded any questions or comments, indicated their preferences in the decision-making process, and indicated the therapy they were leaning towards taking. The worksheet was used to convey “at a glance” the pertinent information to their physicians.
FIGURE 3.
Personal worksheet from the audiobooklet. The worksheet was developed before a recent study38 suggested that a predisposition to falling is not a contraindication to anticoagulation in elderly patients with atrial fibrillation.
Physician's Manual
We provided physicians with scientific information regarding the content of the decision aid. This was for their own education, to allow them to determine if they agreed with the presented material, and to ensure that they were aware of the content of the audiobooklet when meeting with their patients. Thus, we developed a “Physician's Manual,” an 8-page summary of the content of the audiobooklet complete with 22 references.
Pilot Testing and Approval by Patients and Physicians
Early in the process of development, pertinent materials were given to 6 family physicians and 2 specialists. They were generally complimentary but had some useful suggestions. Our initial draft suggested that patients taking warfarin should abstain from alcohol, but almost all of the physicians remarked that they allowed their patients to take 1 or 2 alcoholic drinks per day. We changed this section to reflect their suggestions. Some physicians felt that the risk and severity of major hemorrhage was overestimated when taking aspirin and were uncomfortable with the indication that some patients die from a gastrointestinal hemorrhage, while others felt the risk was underestimated. We repeated our review of the literature and subsequently decided to continue mentioning the possibility of death (the risk of death from a major non–central nervous system hemorrhage is up to 10% in some studies).32 We also decided to revise our estimate of an incremental risk of major hemorrhage associated with aspirin from 0.25% to 0.5% over 2 years.
We also pretested the material with 18 patients with atrial fibrillation. In general, the patients liked the audiobooklet, and felt that its length and the amount of detail were appropriate. They had similar comments about alcohol use as the physicians. They also wanted some background information about atrial fibrillation. This led us to develop a brief introductory page providing this information. On the basis of their comments, we also modified the personal worksheet to provide better clarity.
We had initial concerns that the probability of stroke for patients at highest risk and the description of a major stroke (Fig. 2) might distress patients. However, no patient had this difficulty. Most knew someone who had had a major stroke, and thus were not surprised that the consequences could be grave.
Finally, we gave a draft to representatives of DuPont Pharma, one of the project sponsors. They felt that the audiobooklet overemphasized the risks of warfarin and underemphasized the risks of aspirin. After considering their comments, we decided not to make any changes to the risk of major hemorrhage on warfarin, but did change the risk of major hemorrhage when taking aspirin.
Current Format of the Audiobooklet
The entire “audiobooklet package” contains the physician's manual, the booklet, an audiotape, and the personal worksheet. The booklet is 32 pages in length, and it takes about 20 minutes to listen to the tape. Patients listen to the audiotape while reading the booklet, and then complete the personal worksheet. They are free to share the material with anyone they wish. At the clinic visit, the patient and physician review the worksheet, the physician answers any questions, and a decision about the type of antithrombotic therapy is made. Examples of the audiobooklet and text of the audiotape are available at: www.lri.ca.
EVALUATION
Validation of the Decision Aid
In collaboration with the Stroke Prevention in Atrial Fibrillation (SPAF) III investigators, we have recently completed an evaluation of a modified version of our decision aid.33 At the end of the SPAF III aspirin cohort study,34 to inform participating patients of study results and help them decide whether they wished to remain on aspirin or switch to warfarin, we randomized 287 participants to either an audiobooklet explaining the final results of the study plus usual care versus usual care alone. Outcome measures included in this trial were the ability of patients to make a choice regarding the use of antithrombotic therapy, knowledge of different treatments, decisional conflict (patient's perceptions about whether they have the knowledge and support to make an informed and effective decision), satisfaction with the decision-making process, and 6-month adherence with the therapy patients chose. As we previously reported, patients using the audiobooklet were more likely than control patients to make a definitive choice regarding antithrombotic therapy, were more knowledgeable, and were slightly less likely to choose to take warfarin. Decisional conflict, satisfaction, and 6-month adherence were not significantly improved.
DISCUSSION
Development of a decision aid can be a difficult and time-consuming task. This audiobooklet took about a year to develop and involved a multidisciplinary group, including physicians, nurses, and experts in decision making and determining patient preferences.
The results of the SPAF III decision aid trial suggest that the explicit description of the pertinent information tailored to the individual can improve patients' knowledge, allowing them (in conjunction with their physician) to make more informed decisions about the use of antithrombotic therapy. However, this added knowledge may not alleviate patient uncertainty in the decision-making process or allow them to take the chosen therapy with increased confidence. These limitations highlight the importance of physician guidance in clinical decision making and support the use of decision aids as adjuncts to rather than substitutes for the patient-physician interaction. Furthermore, the persons in the SPAF III decision aid trial were also participants in a major clinical trial and thus were likely different from average patients with atrial fibrillation. They likely received more personalized care and were possibly better informed about their condition. Therefore, we believe that the effectiveness of our decision aid should be assessed in a full-scale randomized trial of patients with atrial fibrillation within the setting of usual clinical care.
A recent article35 has summarized the evidence from randomized, controlled trials regarding the efficacy of decision aids, including clinical situations involving patients faced with treatment decisions regarding hormone replacement therapy,3 benign prostatic hypertrophy,36 and coronary artery disease.37 These studies suggest that decision aids, affect the clinical decision-making process. In these trials, compared with patients receiving usual care, patients using decision aids were consistently more knowledgeable and more informed about the pertinent clinical issues. Depending upon the clinical situation, persons using decision aids may or may not report less uncertainty about the choices that they made, and thus had variable decisional conflict levels. Also, satisfaction with the decision-making process has not been consistently improved by use of the decision aids. Some of the other issues regarding decision aid technology await further research including determination of the characteristics of patients who most benefit from using them (including those that may lead to increased anxiety in some patients using decision aids), their cost-effectiveness, and identification of the clinical situations that warrant their use. Another issue is determination of the most effective and efficient method of incorporating new information into them. For example, newer versions of the atrial fibrillation decision aid will need to take into account the information from two recent studies.8,9
Acknowledgments
This study was supported by grants from the Medical Research Council of Canada, the National Institute of Neurological Disorders and Stroke (RO1 NS 24224), Bethesda, Md, and DuPont Pharma.
REFERENCES
- 1.O'connor AM, Fiset V, Rostom A, et al. Systematic review of approaches to support people's decision making about health treatments or screening. 1999. Cochrane Collaboration Review.
- 2.Spunt BS, Deyo RA, Taylor VM, Leek KM, Goldberg HI, Mulley AG. An interactive videodisc program for low back pain patients. Health Educ Res. 1996;11:535–41. doi: 10.1093/her/11.4.535. [DOI] [PubMed] [Google Scholar]
- 3.O'connor AM, Tugwell P, Wells G, et al. Randomized trial of a portable, self-administered decision aid for post-menopausal women considering long-term preventive hormone therapy. Med Decis Making. 1998;18:295–303. doi: 10.1177/0272989X9801800307. [DOI] [PubMed] [Google Scholar]
- 4.Levine MN, Gafni A, Markham B, MacFarlane D. A bedside decision instrument to elicit a patient's preference concerning adjuvant chemotherapy for breast cancer. Ann Intern Med. 1992;117:53–8. doi: 10.7326/0003-4819-117-1-53. [DOI] [PubMed] [Google Scholar]
- 5.Wolf PA, Abbott RD, Kannel WB. Atrial fibrillation as an independent risk factor for stroke: the Framingham study. Stroke. 1991;22:983–8. doi: 10.1161/01.str.22.8.983. [DOI] [PubMed] [Google Scholar]
- 6.Risk factors for stroke and efficacy of antithrombotic therapy in atrial fibrillation: analysis of pooled data from five randomized controlled trials. Arch Intern Med. 1994;154:1449–1457. [PubMed] [Google Scholar]
- 7.The efficacy of aspirin in patients with atrial fibrillation: analysis of pooled data from three randomized trials Atrial Fibrillation Investigators. Arch Intern Med. 1997;157:1237–40. [PubMed] [Google Scholar]
- 8.Hart RG, Benavente O, McBride R, Pearce LA. Antithrombotic therapy to prevent stroke in patients with atrial fibrillation: a meta-analysis. Ann Intern Med. 1999;131:492–501. doi: 10.7326/0003-4819-131-7-199910050-00003. [DOI] [PubMed] [Google Scholar]
- 9.Segal JB, McNamara RL, Miller MR, et al. Prevention of thromboembolism in atrial fibrillation. J Gen Intern Med. 2000;15:56–67. doi: 10.1046/j.1525-1497.2000.04329.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chang HJ, Bell JR, Deroo DB, Kirk JW, Wasson JH Dartmouth Primary Care COOP Project. Physician variation in anticoagulating patients with atrial fibrillation. Arch Intern Med. 1990;150:83–6. [PubMed] [Google Scholar]
- 11.Dow L, Bertagne B. Anticoagulation in patients with atrial fibrillation. Underuse of warfarin is multifactorial. BMJ. 1993;307:1492–3. doi: 10.1136/bmj.307.6917.1492-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brass LM, Krumholz HM, Scinto JD, Mathur D, Radford M. Warfarin use following ischemic stroke among Medicare patients with atrial fibrillation. Arch Intern Med. 1998;158:2093–100. doi: 10.1001/archinte.158.19.2093. [DOI] [PubMed] [Google Scholar]
- 13.Kutner M, Nixon G, Silverstone F. Physicians' attitudes toward oral anticoagulants and antiplatelet agents for stroke prevention in elderly patients with atrial fibrillation. Arch Intern Med. 1991;51:1950–3. [PubMed] [Google Scholar]
- 14.McCrory DC, Matchar DB, Samsa G, Sanders LL, Pritchett ELC. Physician attitudes about anticoagulation for nonvalvular atrial fibrillation in the elderly. Arch Intern Med. 1995;155:277–81. [PubMed] [Google Scholar]
- 15.O'connor AM, Tugwell P, Wells GA, et al. A decision aid for women considering hormone replacement therapy after menopause: decision support framework and evaluation. Patient Educ Couns. 1998;33:267–79. doi: 10.1016/s0738-3991(98)00026-3. [DOI] [PubMed] [Google Scholar]
- 16.O'connor AM, Drake ER, Fiset V, Graham ID, Laupacis A, Tugwell P. The Ottawa patient decision aids. Eff Clin Prac. 1999;2:163–70. [PubMed] [Google Scholar]
- 17.Morocoutti C, Amabile G, Fattapposta F, et al. Indobufen versus warfarin in the secondary prevention of major vascular events in nonrheumatic atrial fibrillation. Stroke. 1997;28:1015–21. doi: 10.1161/01.str.28.5.1015. [DOI] [PubMed] [Google Scholar]
- 18.Laupacis A, Albers G, Dalen J, Dunn MI, Jacobson AK, Singer DE. Antithrombotic therapy in atrial fibrillation. Chest. 1998;114:579S–89S. doi: 10.1378/chest.114.5_supplement.579s. [DOI] [PubMed] [Google Scholar]
- 19.Fihn SD, Callahan CM, Martin DC, et al. The risk for and severity of bleeding complications in elderly patients treated with warfarin. Ann Intern Med. 1996;24:970–9. doi: 10.7326/0003-4819-124-11-199606010-00004. [DOI] [PubMed] [Google Scholar]
- 20.Beyth RJ, Landefeld CS. Outcomes of warfarin therapy: lessons from the real world. Mayo Clin Proc. 1995;70:806–8. doi: 10.4065/70.8.806. [DOI] [PubMed] [Google Scholar]
- 21.Landefeld CS, Beyth RJ. Anticoagulant-related bleeding: clinical epidemiology, prediction, and prevention. Amer J Med. 1994;95:315–28. doi: 10.1016/0002-9343(93)90285-w. [DOI] [PubMed] [Google Scholar]
- 22.van der Meer FJM, Rosendaal FR, Vandenbroucke JP, Briet E. Bleeding complications in oral anticoagulant therapy. An analysis of risk factors. Arch Intern Med. 1993;153:1557–62. doi: 10.1001/archinte.153.13.1557. [DOI] [PubMed] [Google Scholar]
- 23.Gitter MJ, Jaeger TM, Petterson TM, Gersh BJ, Silverstein MD. Bleeding and thromboembolism during anticoagulation therapy: a population-based study in Rochester, Minnesota. Mayo Clin Proc. 1995;70:725–33. doi: 10.4065/70.8.725. [DOI] [PubMed] [Google Scholar]
- 24.Palareti G, Leali N, Coccheri S, et al. Bleeding complications of oral anticoagulant treatment: an inception-cohort, prospective collaborative study (ISCOAT). Lancet. 1996;348:423–42. doi: 10.1016/s0140-6736(96)01109-9. [DOI] [PubMed] [Google Scholar]
- 25.Weil J, Colin-Jones D, Langman M, et al. Prophylactic aspirin and risk of peptic ulcer bleeding. BMJ. 1995;310:827–30. doi: 10.1136/bmj.310.6983.827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roderick PJ, Wilkes HC, Meade TW. The gastrointestinal toxicity of aspirin: an overview of randomized controlled trials. Br J Clin Pharmac. 1993;35:219–26. doi: 10.1111/j.1365-2125.1993.tb05689.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hart RG, Harrison MJG. Aspirin wars. The optimal dose of aspirin to prevent stroke. Stroke. 1996;27:585–7. doi: 10.1161/01.str.27.4.585. [DOI] [PubMed] [Google Scholar]
- 28.Collaborative overview of randomised trials of antiplatelet therapy–I: Prevention of death, myocardial infarction, and stroke by prolonged antiplatelet therapy in various categories of patients Antiplatelet Trialist's Collaboration. BMJ. 1994;308:81–106. [PMC free article] [PubMed] [Google Scholar]
- 29.Blackshear JL, Kopecky SL, Litin SC, Safford RE, Hammill SC. Management of atrial fibrillation in adults: prevention of thromboembolism and symptomatic treatment. Mayo Clin Proc. 1996;71:150–60. doi: 10.4065/71.2.150. [DOI] [PubMed] [Google Scholar]
- 30.Bleeding during antithrombotic therapy in patients with atrial fibrillation The Stroke Prevention in Atrial Fibrillation Investigators. Arch Intern Med. 1996;156:409–16. [PubMed] [Google Scholar]
- 31.O'connor A, Tugwell P, Wells G. Testing a portable, self-administered decision aid for postmenopausal women considering long-term hormone replacement therapy to prevent osteoporosis and heart disease. Med Decis Making. 1994;14:438. doi: 10.1177/0272989X9801800307. Abstract. [DOI] [PubMed] [Google Scholar]
- 32.Van der Meer FJ, Rosendaal FR, Vandenbroucke JP, Briet E. Bleeding complications in oral anticoagulant therapy. Arch Intern Med. 1993;153:1557–62. doi: 10.1001/archinte.153.13.1557. [DOI] [PubMed] [Google Scholar]
- 33.Man-Son-Hing M, Laupacis A, O'connor AM, et al. A patient decision aid regarding antithrombotic therapy for stroke prevention in atrial fibrillation. A randomized controlled trial. JAMA. 1999;282:737–43. doi: 10.1001/jama.282.8.737. [DOI] [PubMed] [Google Scholar]
- 34.Patients with nonvalvular atrial fibrillation at low risk of stroke during treatment with aspirin: Stroke Prevention in Atrial Fibrillation III Study The SPAF II Writing Committee for the Stroke Prevention in Atrial Fibrillation Investigators. JAMA. 1998;279:1273–7. [PubMed] [Google Scholar]
- 35.O'connor AM, Rostom A, Fiset V, et al. Decision aids for patients facing health treatment of screening decisions: systematic review. BMJ. 1999;319:731–4. doi: 10.1136/bmj.319.7212.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Barry MJ, Cherkin DC, Chang Y, Fowler FJ, Skates S. A randomized trial of a multimedia shared decision-making program for men facing a treatment decision for benign prostatic hypertrophy. Dis Manag Clin Outcomes. 1997;1:5–14. [Google Scholar]
- 37.Morgan MW. A Randomized Trial of the Ischemic Heart Disease Shared Decision Making Program: An Evaluation of a Decision Aid. Master's thesis. Toronto, Ontario, Canada: University of Toronto; 1997. [Google Scholar]
- 38.Man-Son-Hing M, Nichol G, Lau A, Laupacis A. Choosing antithrombotic therapy for elderly patients with atrial fibrillation who are at risk for falls. Arch Intern Med. 1999;159:677–85. doi: 10.1001/archinte.159.7.677. [DOI] [PubMed] [Google Scholar]